Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
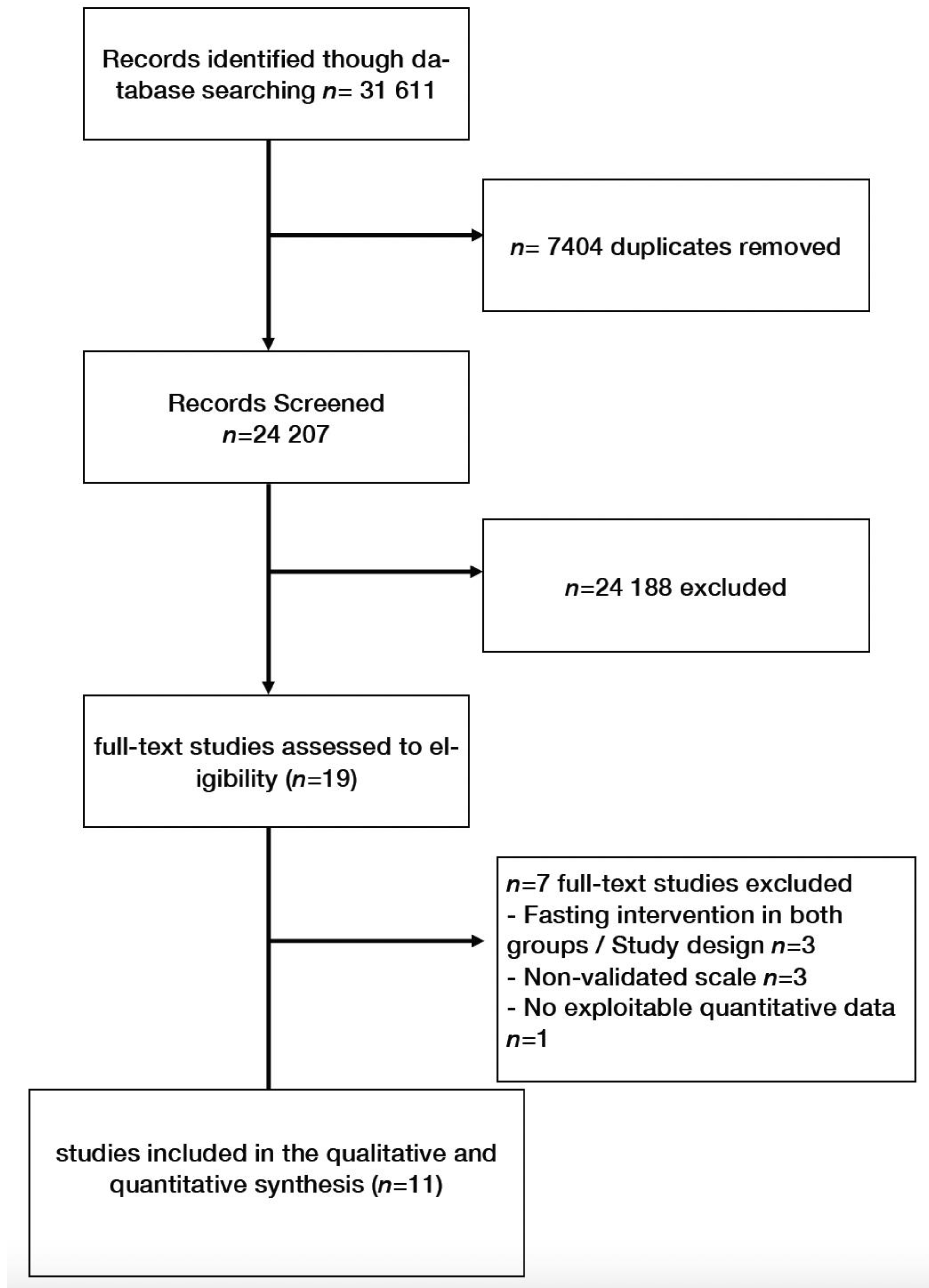
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Eligibility
2.3. Data Extraction
2.4. Study Quality
2.5. Statistical Analyses
2.6. Role of the Funding Source
3. Results
3.1. Characteristics of Studies
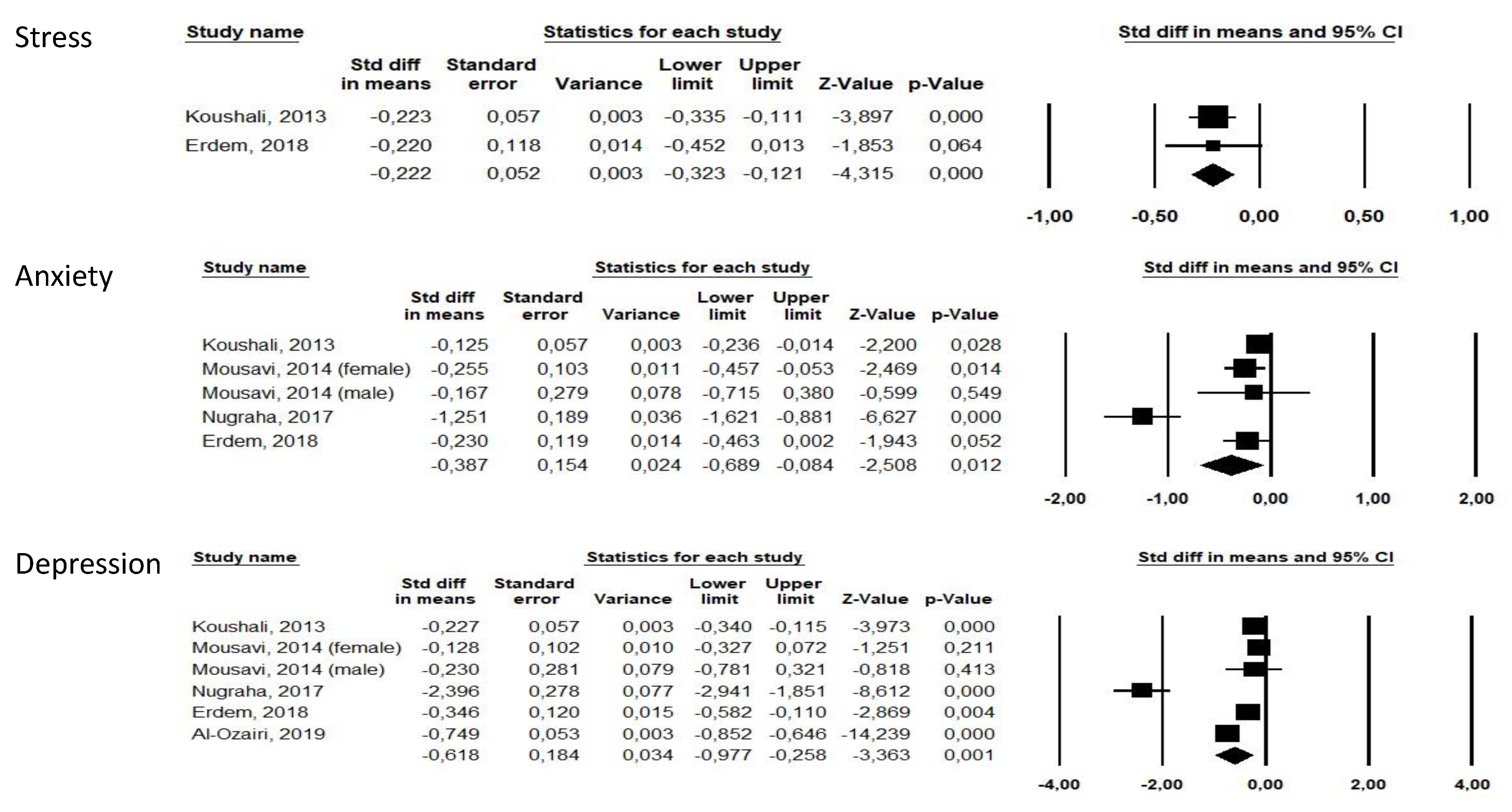
3.2. Ramadan Studies
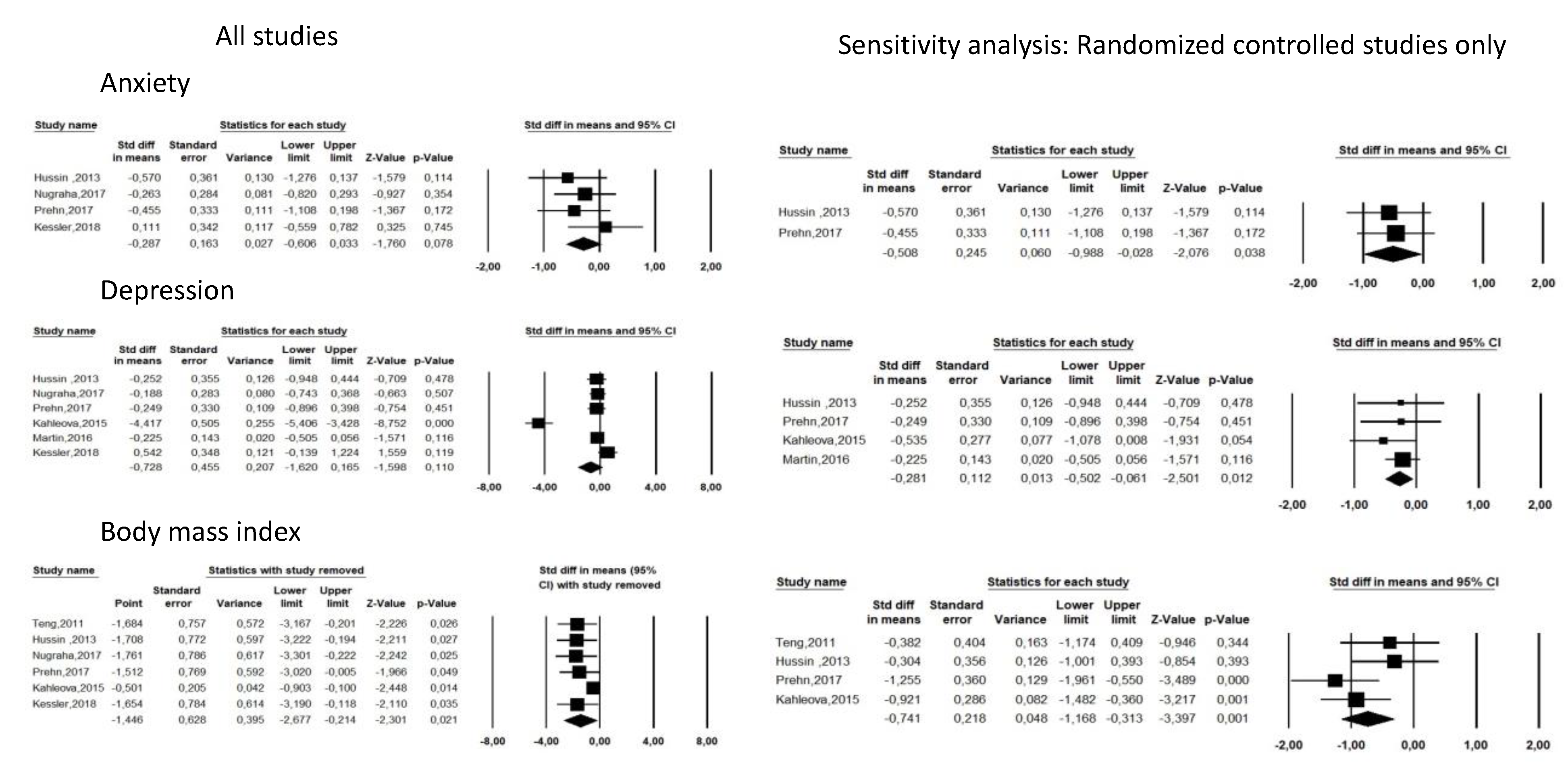
3.3. Fasting Controlled Trials
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A.; et al. Global Burden of 369 Diseases and Injuries in 204 Countries and Territories, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Chevalier, G.; Siopi, E.; Guenin-Macé, L.; Pascal, M.; Laval, T.; Rifflet, A.; Boneca, I.G.; Demangel, C.; Colsch, B.; Pruvost, A.; et al. Effect of gut microbiota on depressive-like behaviors in mice is mediated by the endocannabinoid system. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fond, G.B.; Lagier, J.-C.; Honore, S.; Lancon, C.; Korchia, T.; De Verville, P.-L.S.; Llorca, P.-M.; Auquier, P.; Guedj, E.; Boyer, L. Microbiota-Orientated Treatments for Major Depression and Schizophrenia. Nutrients 2020, 12, 1024. [Google Scholar] [CrossRef] [Green Version]
- Lassale, C.; Batty, G.; Baghdadli, A.; Jacka, F.; Villegas, A.S.; Kivimäki, M.; Akbaraly, T. Healthy dietary indices and risk of depressive outcomes: A systematic review and meta-analysis of observational studies. Mol. Psychiatry 2019, 24, 965–986. [Google Scholar] [CrossRef] [Green Version]
- Matison, A.P.; Mather, K.A.; Flood, V.M.; Reppermund, S. Associations between nutrition and the incidence of depression in middle-aged and older adults: A systematic review and meta-analysis of prospective observational population-based studies. Ageing Res. Rev. 2021, 70, 101403. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Xie, B.; Zhang, H.; He, Q.; Guo, L.; Subramanieapillai, M.; Fan, B.; Lu, C.; McIntyre, R.S. Efficacy of omega-3 PUFAs in depression: A meta-analysis. Transl. Psychiatry 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wadden, T.A.; Mason, G.; Foster, G.D.; Stunkard, A.J.; Prange, A.J. Effects of a very low calorie diet on weight, thyroid hormones and mood. Int. J. Obes. 1990, 14, 249–258. [Google Scholar]
- Wing, R.R.; Marcus, M.D.; Blair, E.H.; Burton, L.R. Psychological Responses of Obese Type II Diabetic Subjects to Very-Low-Calorie Diet. Diabetes Care 1991, 14, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Wadden, T.A.; Stunkard, A.J.; Brownell, K.D.; Day, S.C. A comparison of two very-low-calorie diets: Protein-sparing-modified fast versus protein-formula-liquid diet. Am. J. Clin. Nutr. 1985, 41, 533–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabricatore, A.N.; Wadden, T.A.; Higginbotham, A.J.; Faulconbridge, L.; Nguyen, A.M.; Heymsfield, S.B.; Faith, M.S. Intentional weight loss and changes in symptoms of depression: A systematic review and meta-analysis. Int. J. Obes. 2011, 35, 1363–1376. [Google Scholar] [CrossRef] [Green Version]
- Sadeghirad, B.; Motaghipisheh, S.; Kolahdooz, F.; Zahedi, M.J.; Haghdoost, A. Islamic fasting and weight loss: A systematic review and meta-analysis. Public Health Nutr. 2014, 17, 396–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, L.; Hamilton, S.; Azevedo, L.B.; Olajide, J.; De Brún, C.; Waller, G.; Whittaker, V.; Sharp, T.; Lean, M.; Hankey, C.; et al. Intermittent fasting interventions for treatment of overweight and obesity in adults. JBI Database Syst. Rev. Implement. Rep. 2018, 16, 507–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerrero, A.E.; Martín, I.S.M.; Vilar, E.G.; Martín, M.A.C. Effectiveness of an intermittent fasting diet versus continuous energy restriction on anthropometric measurements, body composition and lipid profile in overweight and obese adults: A meta-analysis. Eur. J. Clin. Nutr. 2021, 75, 1024–1039. [Google Scholar] [CrossRef] [PubMed]
- De Cabo, R.; Mattson, M.P. Effects of intermittent fasting on health, aging, and disease. N. Engl. J. Med. 2019, 381, 2541–2551. [Google Scholar] [CrossRef] [PubMed]
- Fond, G.; Macgregor, A.; Leboyer, M.; Michalsen, A. Fasting in mood disorders: Neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013, 209, 253–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gergelyfi, M.; Sanz-Arigita, E.J.; Solopchuk, O.; Dricot, L.; Jacob, B.; Zénon, A. Mental fatigue correlates with depression of task-related network and augmented DMN activity but spares the reward circuit. NeuroImage 2021, 243, 118532. [Google Scholar] [CrossRef]
- Kahleova, H.; Belinova, L.; Hill, M.; Pelikanova, T. Do patients with type 2 diabetes still need to eat snacks? Eur. J. Clin. Nutr. 2015, 69, 755–756. [Google Scholar] [CrossRef]
- NIH Study Quality Assessment Tools|NHLBI, NIH 2021. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 1 September 2021).
- Nugraha, B.; Ghashang, S.K.; Hamdan, I.; Gutenbrunner, C. Effect of Ramadan fasting on fatigue, mood, sleepiness, and health-related quality of life of healthy young men in summer time in Germany: A prospective controlled study. Appetite 2017, 111, 38–45. [Google Scholar] [CrossRef]
- Teng, N.I.M.F.; Shahar, S.; Manaf, Z.A.; Das, S.K.; Taha, C.S.C.; Ngah, W.Z.W. Efficacy of fasting calorie restriction on quality of life among aging men. Physiol. Behav. 2011, 104, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Hussin, N.M.; Shahar, S.; Teng, N.I.M.F.; Ngah, W.Z.W.; Das, S.K. Efficacy of Fasting and Calorie Restriction (FCR) on mood and depression among ageing men. J. Nutr. Health Aging 2013, 17, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Koushali, A.N.; Hajiamini, Z.; Ebadi, A.; Bayat, N.; Khamseh, F. Effect of Ramadan fasting on emotional reactions in nurses. Iran. J. Nurs. Midwifery Res. 2013, 18, 232–236. [Google Scholar]
- Mousavi, S.A.; Rezaei, M.; Baghni, S.A.; Seifi, M. Effect of fasting on mental health in the general population of Kermanshah, Iran. J. Nutr. Health 2014, 2, 65–70. [Google Scholar]
- Martin, C.K.; Bhapkar, M.; Pittas, A.G.; Pieper, C.F.; Das, S.K.; Williamson, D.A.; Scott, T.; Redman, L.M.; Stein, R.; Gilhooly, C.H.; et al. Effect of Calorie Restriction on Mood, Quality of Life, Sleep, and Sexual Function in Healthy Nonobese Adults. JAMA Intern. Med. 2016, 176, 743–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prehn, K.; Von Schwartzenberg, R.J.; Mai, K.; Zeitz, U.; Witte, A.V.; Hampel, D.; Szela, A.-M.; Fabian, S.; Grittner, U.; Spranger, J.; et al. Caloric Restriction in Older Adults—Differential Effects of Weight Loss and Reduced Weight on Brain Structure and Function. Cereb. Cortex 2016, 27, 1765–1778. [Google Scholar] [CrossRef] [PubMed]
- Erdem, O. The investigation of the effects of Ramadan fasting on the mood state of healthy volunteer persons. Fam. Pract. Palliat. CARE 2018, 3, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Kessler, C.S.; Stange, R.; Schlenkermann, M.; Jeitler, M.; Michalsen, A.; Selle, A.; Raucci, F.; Steckhan, N. A nonrandomized controlled clinical pilot trial on 8 wk of intermittent fasting (24 h/wk). Nutrition 2018, 46, 143–152.e2. [Google Scholar] [CrossRef] [PubMed]
- Al-Ozairi, E.; Alawadhi, M.M.; Al-Ozairi, A.; Taghadom, E.; Ismail, K.; Isamil, K. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabetes Res. Clin. pract. 2019, 153, 145–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Secades-Villa, R.; González-Roz, A.; García-Pérez, Á.; Becoña, E. Psychological, pharmacological, and combined smoking cessation interventions for smokers with current depression: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0188849. [Google Scholar] [CrossRef]
- Ioannou, M.; Wartenberg, C.; Greenbrook, J.T.V.; Larson, T.; Magnusson, K.; Schmitz, L.; Sjögren, P.; Stadig, I.; Szabó, Z.; Steingrimsson, S. Sleep deprivation as treatment for depression: Systematic review and meta-analysis. Acta Psychiatr. Scand. 2021, 143, 22–35. [Google Scholar] [CrossRef]
- Silva, D.A.; Coutinho, E.D.S.F.; Ferriani, L.O.; Viana, M.C. Depression subtypes and obesity in adults: A systematic review and meta-analysis. Obes. Rev. 2019, 21, e12966. [Google Scholar] [CrossRef] [PubMed]
- Grigolon, R.B.; Trevizol, A.P.; Gerchman, F.; Bambokian, A.D.; Magee, T.; McIntyre, R.S.; Gomes, F.A.; Brietzke, E.; Mansur, R.B. Is Obesity A Determinant Of Success With Pharmacological Treatment For Depression? A Systematic Review, Meta-Analysis And Meta-Regression. J. Affect. Disord. 2021, 287, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Garcia, G.D.; Pompeo, D.A.; Eid, L.P.; Cesarino, C.B.; Pinto, M.H.; Gonçalves, L.W.P. Relationship between anxiety, depressive symptoms and compulsive overeating disorder in patients with cardiovascular diseases. Rev. Lat.-Am. Enferm. 2018, 26, 3040. [Google Scholar] [CrossRef] [PubMed]
- Ensminger, D.C.; Salvador-Pascual, A.; Arango, B.G.; Allen, K.N.; Vázquez-Medina, J.P. Fasting ameliorates oxidative stress: A review of physiological strategies across life history events in wild vertebrates. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2021, 256, 110929. [Google Scholar] [CrossRef] [PubMed]
| Study | Country | N | N F | N C | N(%) Men | Design | Population | Fasting Intervention | Controls | Endpoint * | Scales ** | Authors’ Conclusion | Adverse Events | N Dropout Fasting | N Dropout Controls |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Koushali (2013) [22] | Iran | 313 | 313 | NA | 177(56.5%) | OBS | Hospital nurses | Ramadan | NA | 1 to 2 | Anxiety: DASS21 Depression: DASS21 | Depression and stress were significantly reduced (p < 0.05) but not anxiety. | md | NA | NA |
| Mousavi (2014) [23] | Iran | 110 | 110 | NA | 13(11.8%) | OBS | Residents of Kermanshah city | Ramadan | NA | MD | Anxiety: GHQ subscore Depression: GHQ subscore Stress: GHQ | Significant reduction in anxiety (p = 0.011) but no significant reduction in depression (p > 0.05) after Ramadan. | md | NA | NA |
| Erdem (2018) [26] | Turkey | 73 | 73 | NA | 63(86.3%) | OBS | Muslim healthy volunteers | Ramadan | NA | 0 | Stress: DASS-42 Anxiety: DASS anxiety Depression: DASS | Significant reduction in depression (p = 0.001), anxiety (p = 0.01) and stress (p = 0.002) scores after Ramadan. | md | NA | NA |
| Al-Ozairi (2019) [28] | Kuwait | 463 | 463 | NA | 251(54.2%) | OBS | Type 2 diabetes Muslim patients ≥21 years | Ramadan | NA | 4–6 | Depression: PHQ-9 | Significant reduction in depressive symptoms after Ramadan (p < 0.05). | md | NA | NA |
| Nugraha (2017) [19] | Germany | 50 | 25 | 25 | 50(100%) | CT | Healthy male volunteers ≥ 18 years (mostly students) | Ramadan | No fasting and no other intervention | 1 | Anxiety: HADS Depression: BDI-II | Significant reduction in depressive symptoms after Ramadan (p < 0.05). | Increased fatigue during first week of Ramadan, then decreased fatigue during week 2 to 4 but decreased sleepiness during whole Ramadan. | 3/28(10.7%) (2 time schedule, 1 other reason) | 2/28(7.6%) (other reason) |
| Teng (2011) [20] | Malaysia | 25 | 12 | 13 | 25(100%) | CT | Healthy men aged 50 to 70 years, BMI 23.0 to 29.9 kg/m2 | Reduction in 300 to 500 kcal/day from thei habitual energy intake + two days of Muslim sunnah * fasting per week 12 weeks | No fasting and no other intervention | 0 | Depression: BDI-II | Non-significant reduction in depressive symptoms after fasting intervention (p > 0.05). | Adverse events were not reported but 2 participants were unable to follow the fasting intervention | 2/14(14.2%) (unable to follow the fasting intervention) | 1/14(7.1%) (personal reasons) |
| Hussin (2013) [21] | Malaysia | 32 | 16 | 16 | 32(100%) | RCT | Healthy men aged 50 to 70 years, BMI 23.0 to 29.9 kg/m2 | Reduction of 300 to 500 kcal/day from thei habitual energy intake + two days of Muslim sunnah * fasting per week 12 weeks | No fasting and no other intervention | 0 | Depression: BDI-II. Fatigue: POMS | Non-significant reduction in depressive symptoms after fasting intervention (p > 0.05). | No reported adverse events. | 0(0%) | 1/16(6.2%) |
| Kahleova (2015) [17] | Czech Rebublic | 54 | 27 | 27 | 29(54 %) | RCT | Patient with type 2 diabetes, mean age 59.4 years, mean BMI 32.6 kg/m2 | Time Restricted feeding (14 h fasting/day) + caloric restriction 12 weeks | 6 meals/day (3 meals + 3 snacks) | 0 | Depression: BDI-II | Significant reduction in depression score was decreased in the fasting group (p < 0.05), and feelings of hunger were greater than in the control group. Quality of life increased (p < 0.01) comparably under both regimens. | No reported adverse events. | 3/27(11.1%) (1 personal reasons, 2 lack of motivation) | 4/27(14.8%) (2 personal reasons, 2 lack of motivation) |
| Martin(2016) [24] | USA | 218 | 143 | 75 | 66(30%) | RCT | Healthy men aged 20 to 50 years and women aged 20 to 47 years, with a BMI between 22.0 and 28.0 | 25% Caloric Restriction 104 weeks | No fasting and no other intervention | 0 | Depression: BDI-II Fatigue: POMS | Significant improvement in the depression score (p < 0.05), tension (p < 0.01), and General health (p < 0.001). | No reported adverse events but 3/117(2.6%) participants of the fasting group were removed for safety reasons (not detailed). | 26/143(18.2%) (8 withdrew consent, 6 moved away from study site, 6 for personal and other reasons, 3 women became pregnant, 3 withdrawn for safety) | 5/75(6.7%) (3 women became pregnant, 1 withdrew consent) |
| Prehn (2017) [25] | Germany | 37 | 19 | 18 | 0(0%) | RCT | Older obese women, mean age 61 years, mean BMI 35 | Low calorie diet (800 kcal/J) 12 weeks | No fasting and no other intervention | 0 | Anxiety: STAI Depression: BDI-II | Reduction in Beck’s depression score (p < 0.001) and anxiety score (p < 0.004) in the fasting group. | No reported adverse events but 6 subjects were excluded for instruction failure without details. | 5/23(21.7%) (personal reasons) | 5/24(20.8%) |
| Kessler (2018) [27] | Germany | 36 | 22 | 14 | 14(39)% | CT | Healthy volunteers | Fixed fasting day per week for 8 weeks, a fixed week day 8 weeks | 2 groups counseling sessions for healthy diet + waiting list for fasting intervention | 0 | Anxiety: HADS-A Depression: HADS-D Fatigue: POMS | Significant within- group differences in the fasting group were observed after 6 months for the HADS total score, and the HADS depression and anxiety subscales, the POMS total score (including subscales for positive mood and vigor). | Adverse events: headache, migraine, nausea, ravenousness, circulatory disturbance, hunger, general feeling of weakness, tiredness, stomach ache, meteorism, heartburn, and cold sensations in the body. | N = 4/22 (9.1%) (2 declined to further participate, 2 lost of follow-up) | N = 2/14 (14.2%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berthelot, E.; Etchecopar-Etchart, D.; Thellier, D.; Lancon, C.; Boyer, L.; Fond, G. Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3947. https://doi.org/10.3390/nu13113947
Berthelot E, Etchecopar-Etchart D, Thellier D, Lancon C, Boyer L, Fond G. Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis. Nutrients. 2021; 13(11):3947. https://doi.org/10.3390/nu13113947
Chicago/Turabian StyleBerthelot, Elisa, Damien Etchecopar-Etchart, Dimitri Thellier, Christophe Lancon, Laurent Boyer, and Guillaume Fond. 2021. "Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis" Nutrients 13, no. 11: 3947. https://doi.org/10.3390/nu13113947
APA StyleBerthelot, E., Etchecopar-Etchart, D., Thellier, D., Lancon, C., Boyer, L., & Fond, G. (2021). Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis. Nutrients, 13(11), 3947. https://doi.org/10.3390/nu13113947







