Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin
Abstract
1. Introduction
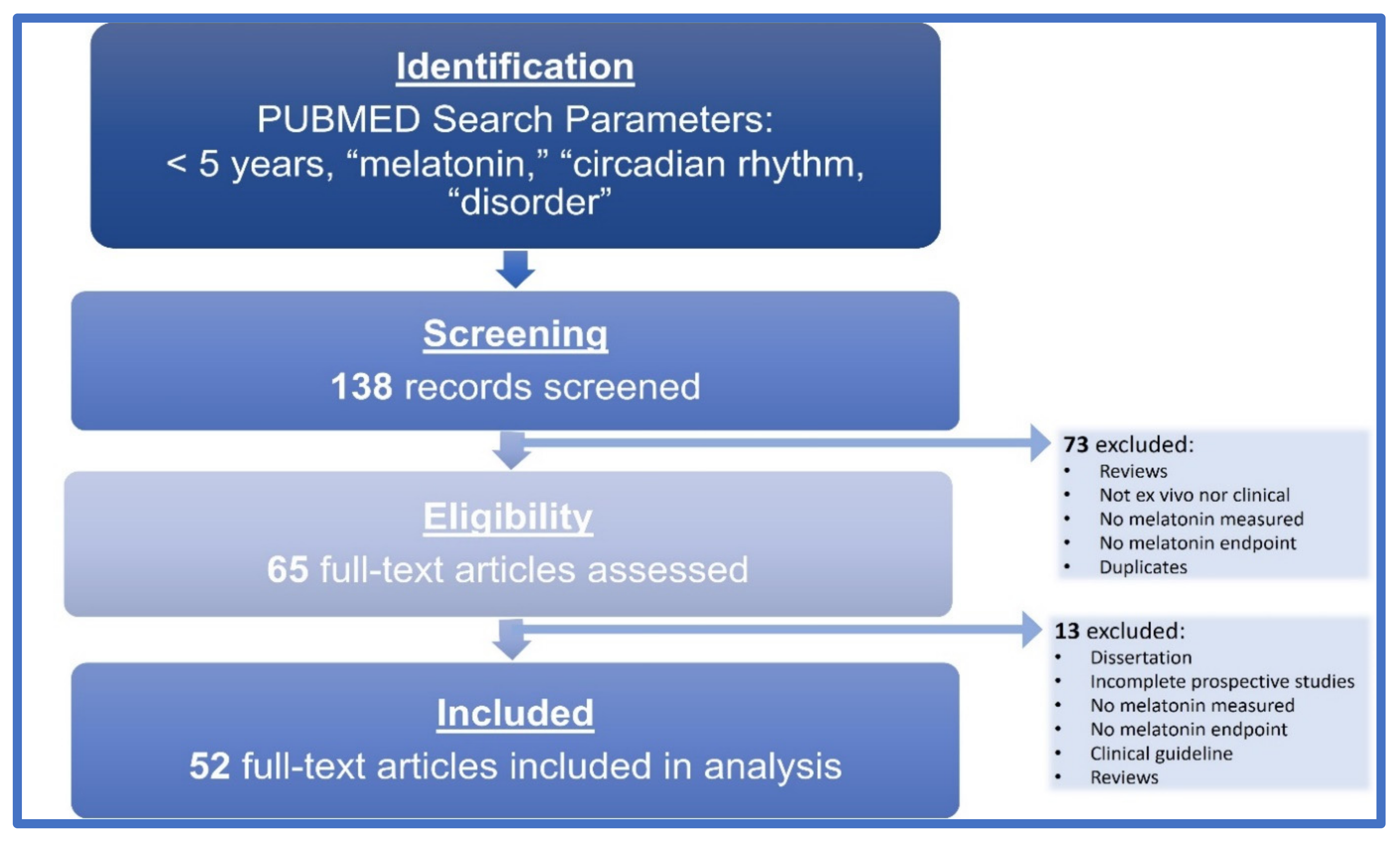
2. Materials and Methods
Concepts and Inclusion Criteria
3. Background
3.1. Daylight Hours
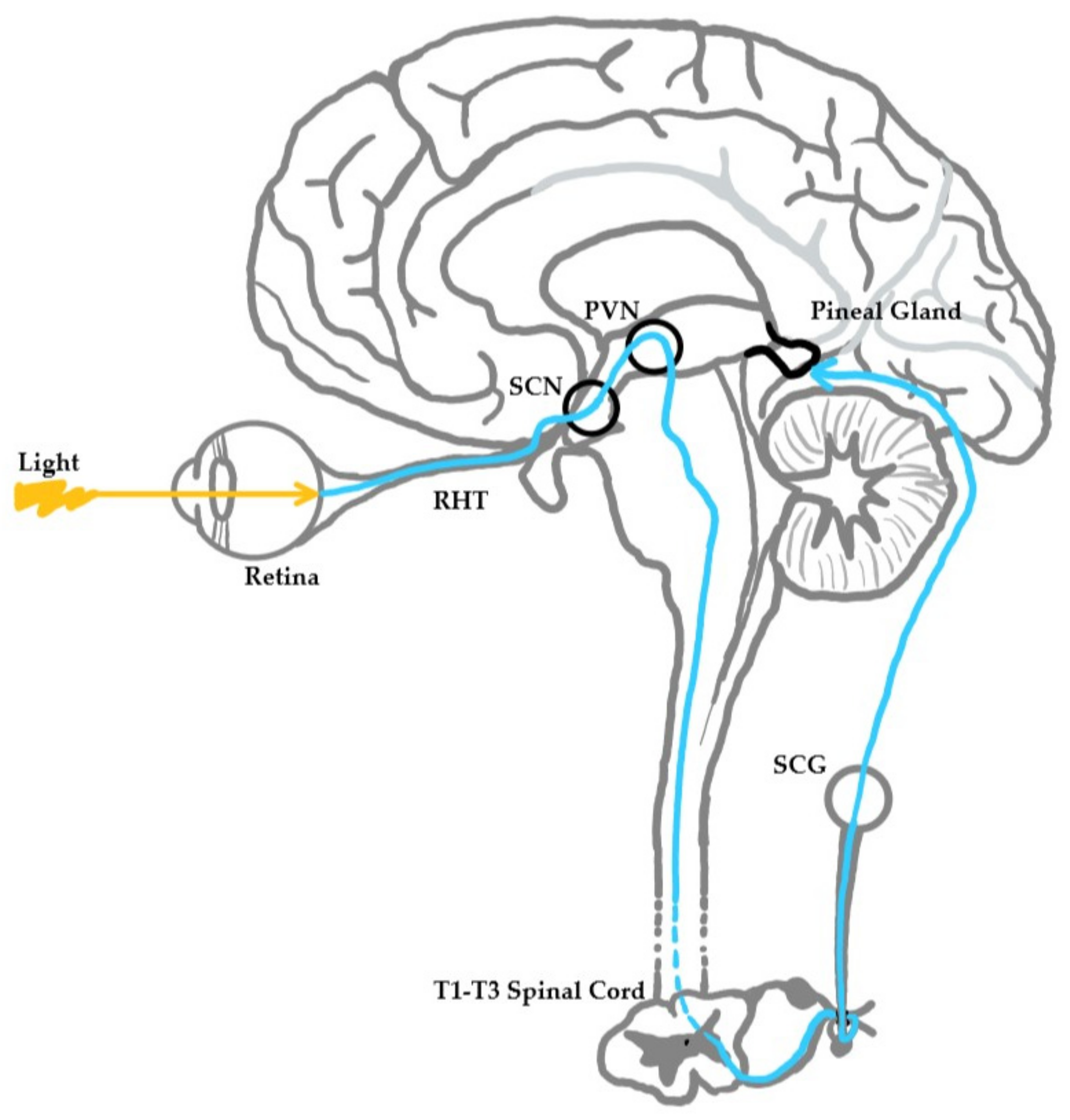
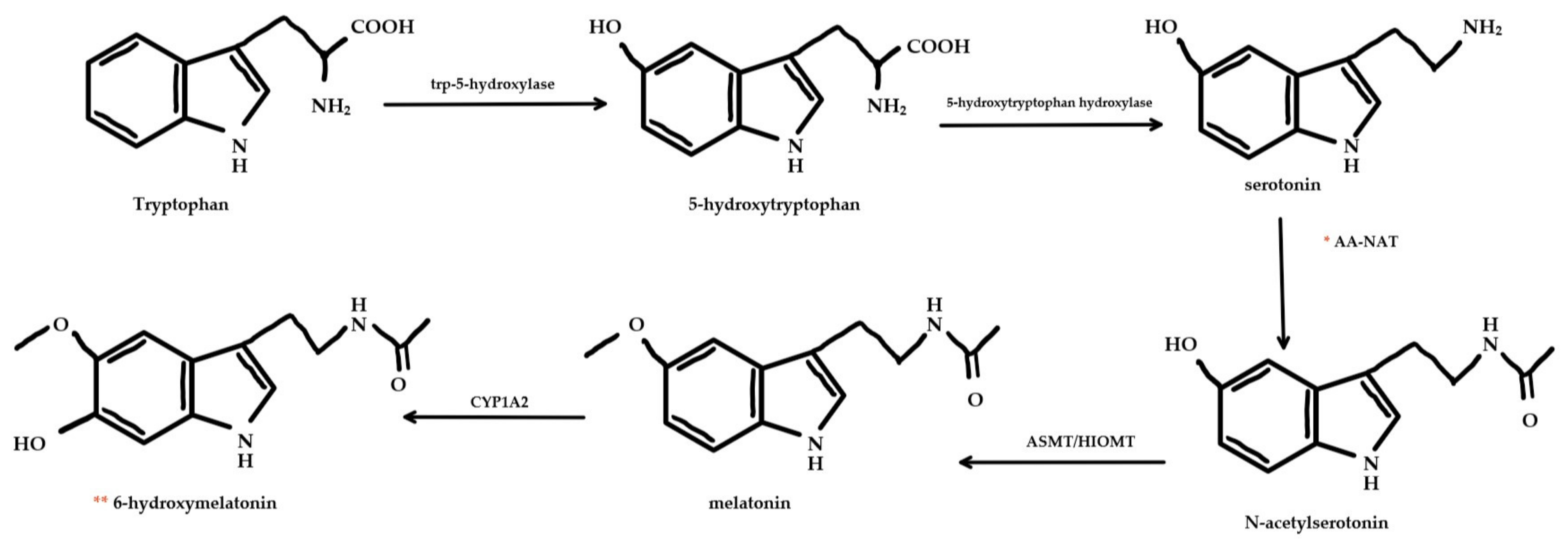
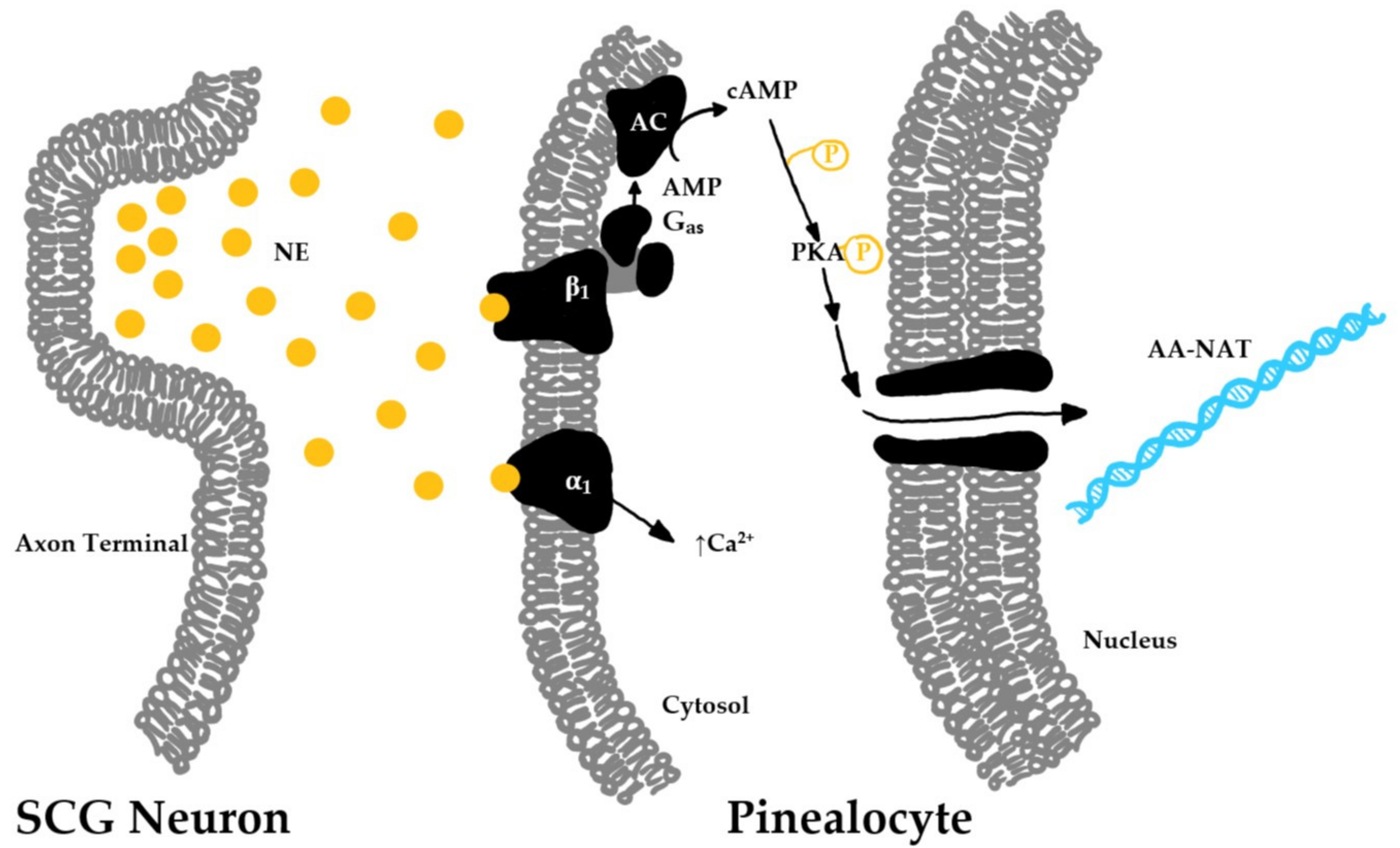
3.2. Endogenous Melatonin
3.3. Exogenous Melatonin
3.4. Dysfunction and Disorders
4. Results
5. Discussion
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Sleep Association. Reviewers and Writers What Is Sleep and Why Is It Important? American Sleep Association: Boston, MA, USA, 2016. [Google Scholar]
- Stevenson, A. How Important Is Sleep? American Sleep Association: Boston, MA, USA, 2020. Available online: https://www.sleepassociation.org/about-sleep/how-important-is-sleep/ (accessed on 18 August 2020).
- Zisapel, N. New Perspectives on the Role of Melatonin in Human Sleep, Circadian Rhythms and Their Regulation. Br. J. Pharamcol. 2018, 175, 3190–3199. [Google Scholar] [CrossRef]
- Bjorness, T.E.; Greene, R.W. Adenosine and Sleep. Curr. Neuropharmacol. 2009, 7, 238–245. [Google Scholar] [CrossRef]
- Mohammad-Zadeh, L.F.; Moses, L.; Gwaltney-Brant, S.M. Serotonin: A Review. J. Vet. Pharmacol. Ther. 2008, 31, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, S.; Sawhney, G.; Pandhi, P. The Therapeutic Potential of Melatonin: A Review of the Science. Medscape Gen. Med. 2004, 6, 46. [Google Scholar]
- Mayo Clinic Staff. Melatonin; Mayo Clinic: Rochester, MN, USA, 2021. [Google Scholar]
- Decker, K. 2019’s Biggest Ingredient Sales Surprises. Nutr. Outlook 2020, 23, 62–63. [Google Scholar]
- Vom Brocke, J.; Simons, A.; Niehaves, B.; Niehaves, B.; Reimer, K. Reconstructing the Giant: On the Importance of Rigour in Documenting the Literature Search Process. In Proceedings of the 17th European Conference on Information Systems (ECIS), Verona, Italy, 8–10 June 2009. [Google Scholar]
- Circadian Rhythms. 2020. Available online: https://www.nigms.nih.gov (accessed on 18 August 2020).
- Redlin, U. Neural Basis and Biological Function of Masking by Light in Mammals: Suppression of Melatonin and Locomotor Activity. Chronobiol. Int. 2001, 18, 737–758. [Google Scholar] [CrossRef]
- Carlson, E. Resetting Our Clocks: New Details about How the Body Tells Time. Available online: https://www.nigms.nih.gov/education/Inside-Life-Science/Pages/Resetting-Our-Clocks-New-Details-About-How-the-Body-Tells-Time.aspx (accessed on 18 August 2020).
- Reiter, R.J.; Meltz, M.L.; Herman, T.S. Melatonin: Possible Mechanisms Involved in Its “radioprotective” Effect. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 1998, 404, 187–189. [Google Scholar]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an Antioxidant: Under Promises but over Delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef]
- Hardeland, R. Melatonin in Aging and Disease-Multiple Consequences of Reduced Secretion, Options and Limits of Treatment. Aging Dis. 2012, 3, 194–225. [Google Scholar]
- Paul, M.A.; Love, R.J.; Hawton, A.; Brett, K.; McCreary, D.R.; Arendt, J. Sleep Deficits in the High Arctic Summer in Relation to Light Exposure and Behaviour: Use of Melatonin as a Countermeasure. Sleep Med. 2015, 16, 406–413. [Google Scholar] [CrossRef]
- Arendt, J.; Middleton, B. Human Seasonal and Circadian Studies in Antarctica (Halley, 75° S). Gen. Comp. Endocrinol. 2018, 258, 250–258. [Google Scholar] [CrossRef]
- Pattyn, N.; Mairesse, O.; Cortoos, A.; Marcoen, N.; Neyt, X.; Meeusen, R. Sleep during an Antarctic Summer Expedition: New Light on “Polar Insomnia”. J. Appl. Physiol. 2017, 122, 788–794. [Google Scholar] [CrossRef][Green Version]
- Resuehr, D.; Wu, G.; Johnson, R.L.; Young, M.E.; Hogenesch, J.B.; Gamble, K.L. Shift Work Disrupts Circadian Regulation of the Transcriptome in Hospital Nurses. J. Biol. Rhythm. 2019, 34, 167–177. [Google Scholar] [CrossRef]
- Razavi, P.; Devore, E.E.; Bajaj, A.; Lockley, S.W.; Figueiro, M.G.; Ricchiuti, V.; James Gauderman, W.; Hankinson, S.E.; Willett, W.C.; Schernhammer, E.S. Shift Work, Chronotype, and Melatonin Rhythm in Nurses. Cancer Epidemiol. Biomark. Prev. 2019, 28, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Papantoniou, K. Night Shift Work and Its Carcinogenicity. Lancet Oncol. 2019, 20, e550. [Google Scholar] [CrossRef]
- Haim, A.; Portnov, B.A. Light Pollution as a New Risk Factor for Human Breast and Prostate Cancers; Springer: Berlin/Heidelberg, Germany, 2013; ISBN 9789400762206. [Google Scholar]
- Tähkämö, L.; Partonen, T.; Pesonen, A.K. Systematic Review of Light Exposure Impact on Human Circadian Rhythm. Chronobiol. Int. 2019, 36, 151–170. [Google Scholar] [CrossRef]
- Chang, A.M.; Aeschbach, D.; Duffy, J.F.; Czeisler, C.A. Evening Use of Light-Emitting EReaders Negatively Affects Sleep, Circadian Timing, and next-Morning Alertness. Proc. Natl. Acad. Sci. USA 2015, 112, 1232–1237. [Google Scholar] [CrossRef]
- Lockley, S.W.; Brainard, G.C.; Czeisler, C.A. High Sensitivity of the Human Circadian Melatonin Rhythm to Resetting by Short Wavelength Light. J. Clin. Endocrinol. Metab. 2003, 88, 4502–4505. [Google Scholar] [CrossRef]
- Green, A.; Cohen-Zion, M.; Haim, A.; Dagan, Y. Evening Light Exposure to Computer Screens Disrupts Human Sleep, Biological Rhythms, and Attention Abilities. Chronobiol. Int. 2017, 34, 855–865. [Google Scholar] [CrossRef]
- Knufinke, M.; Fittkau-Koch, L.; Møst, E.I.S.; Kompier, M.A.J.; Nieuwenhuys, A. Restricting Short-Wavelength Light in the Evening to Improve Sleep in Recreational Athletes–A Pilot Study. Eur. J. Sport Sci. 2019, 19, 728–735. [Google Scholar] [CrossRef]
- Lewis, S.R.; Pritchard, M.W.; Schofield-Robinson, O.J.; Alderson, P.; Smith, A.F. Melatonin for the Promotion of Sleep in Adults in the Intensive Care Unit. Cochrane Database Syst. Rev. 2018, 2018, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin: Pharmacology, Functions and Therapeutic Benefits. Curr. Neuropharmacol. 2017, 15, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; López, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal Melatonin: Sources, Regulation, and Potential Functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef] [PubMed]
- Schomerus, C.; Korf, H.W. Mechanisms Regulating Melatonin Synthesis in the Mammalian Pineal Organ. Ann. N. Y. Acad. Sci. 2005, 1057, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.B.P.; Spessert, R.; Vollrath, L. Molecular Components and Mechanism of Adrenergic Signal Transduction in Mammalian Pineal Gland: Regulation of Melatonin Synthesis. Indian J. Exp. Biol. 2005, 43, 115–149. [Google Scholar]
- Duffy, J.F.; Cain, S.W.; Chang, A.M.; Phillips, A.J.K.; Münch, M.Y.; Gronfier, C.; Wyatt, J.K.; Dijk, D.J.; Wright, K.P.; Czeisler, C.A. Sex Difference in the Near-24-Hour Intrinsic Period of the Human Circadian Timing System. Proc. Natl. Acad. Sci. USA 2011, 108, 15602–15608. [Google Scholar] [CrossRef] [PubMed]
- Gunn, P.J.; Middleton, B.; Davies, S.K.; Revell, V.L.; Skene, D.J. Sex Differences in the Circadian Profiles of Melatonin and Cortisol in Plasma and Urine Matrices under Constant Routine Conditions. Chronobiol. Int. 2016, 33, 39–50. [Google Scholar] [CrossRef]
- Wever, R.A. Sex Differences in Human Circadian Rhythms: Intrinsic Periods and Sleep Fractions. Experientia 1984, 40, 1226–1234. [Google Scholar] [CrossRef]
- Czeisler, C.A.; Duffy, J.F.; Shanahan, T.L.; Brown, E.N.; Mitchell, J.F.; Rimmer, D.W.; Ronda, J.M.; Silva, E.J.; Allan, J.S.; Emens, J.S.; et al. Stability, Precision, and near-24-Hour Period of the Human Circadian Pacemaker. Science 1999, 284, 2177–2181. [Google Scholar] [CrossRef]
- Miyamoto, M. Pharmacology of Ramelteon, a Selective MT1/MT2 Receptor Agonist: A Novel Therapeutic Drug for Sleep Disorders. CNS Neurosci. Ther. 2009, 15, 32–51. [Google Scholar] [CrossRef]
- Andersen, L.P.H.; Werner, M.U.; Rosenkilde, M.M.; Harpsøe, N.G.; Fuglsang, H.; Rosenberg, J.; Gögenur, I. Pharmacokinetics of Oral and Intravenous Melatonin in Healthy Volunteers. BMC Pharmacol. Toxicol. 2016, 17, 8. [Google Scholar] [CrossRef]
- Zhdanova, I.V.; Geiger, D.A.; Schwagerl, A.L.; Leclair, O.U.; Killiany, R.; Taylor, J.A.; Rosene, D.L.; Moss, M.B.; Madras, B.K. Melatonin Promotes Sleep in Three Species of Diurnal Nonhuman Primates. Physiol. Behav. 2002, 75, 523–529. [Google Scholar] [CrossRef]
- Ferracioli-Oda, E.; Qawasmi, A.; Bloch, M.H. Meta-Analysis: Melatonin for the Treatment of Primary Sleep Disorders. PLoS ONE 2013, 8, e63773. [Google Scholar] [CrossRef] [PubMed]
- Barchas, J.; DaCosta, F.; Spector, S. Acute Pharmacology of Melatonin. Nature 1967, 214, 919–920. [Google Scholar] [CrossRef] [PubMed]
- Melatonin: What You Need To Know. NCCIH. Available online: https://www.nccih.nih.gov/health/melatonin-what-you-need-to-know (accessed on 7 December 2020).
- Sleep Disorders: Types, Causes, Symptoms & Diagnosis. Available online: https://my.clevelandclinic.org/health/articles/11429-common-sleep-disorders (accessed on 25 October 2020).
- Circadian Rhythm Sleep Disorders: Types, Symptoms and Management. Available online: https://my.clevelandclinic.org/health/diseases/12115-circadian-rhythm-disorders (accessed on 25 October 2020).
- Allega, O.R.; Leng, X.; Vaccarino, A.; Skelly, M.; Lanzini, M.; Paz Hidalgo, M.; Soares, C.N.; Kennedy, S.H.; Frey, B.N. Performance of the Biological Rhythms Interview for Assessment in Neuropsychiatry: An Item Response Theory and Actigraphy Analysis. J. Affect. Disord. 2018, 225, 54–63. [Google Scholar] [CrossRef]
- Baandrup, L.; Fasmer, O.B.; Glenthøj, B.Y.; Jennum, P.J. Circadian Rest-Activity Rhythms during Benzodiazepine Tapering Covered by Melatonin versus Placebo Add-on: Data Derived from a Randomized Clinical Trial. BMC Psychiatry 2016, 16, 348. [Google Scholar] [CrossRef]
- Baker, E.K.; Richdale, A.L.; Hazi, A.; Prendergast, L.A. Assessing the Dim Light Melatonin Onset in Adults with Autism Spectrum Disorder and No Comorbid Intellectual Disability. J. Autism Dev. Disord. 2017, 47, 2120–2137. [Google Scholar] [CrossRef]
- Bock, D.E.; Roach-Fox, E.; Seabrook, J.A.; Rieder, M.J.; Matsui, D. Sleep-Promoting Medications in Children: Physician Prescribing Habits in Southwestern Ontario, Canada. Sleep Med. 2016, 17, 52–56. [Google Scholar] [CrossRef]
- Bradley, A.J.; Webb-Mitchell, R.; Hazu, A.; Slater, N.; Middleton, B.; Gallagher, P.; McAllister-Williams, H.; Anderson, K.N. Sleep and Circadian Rhythm Disturbance in Bipolar Disorder. Psychol. Med. 2017, 47, 1678–1689. [Google Scholar] [CrossRef]
- Büber, A.; Çakaloz, B.; Işildar, Y.; Ünlü, G.; Bostanci, H.E.; Aybek, H.; Herken, H. Increased Urinary 6-Hydroxymelatoninsulfate Levels in Attention Deficit Hyperactivity Disorder Diagnosed Children and Adolescent. Neurosci. Lett. 2016, 617, 195–200. [Google Scholar] [CrossRef]
- Burgess, H.J.; Park, M.; Wyatt, J.K.; Fogg, L.F. Home Dim Light Melatonin Onsets with Measures of Compliance in Delayed Sleep Phase Disorder. J. Sleep Res. 2016, 25, 314–317. [Google Scholar] [CrossRef]
- Burgess, H.J.; Park, M.; Wyatt, J.K.; Rizvydeen, M.; Fogg, L.F. Sleep and Circadian Variability in People with Delayed Sleep-Wake Phase Disorder versus Healthy Controls. Sleep Med. 2017, 25, 33–39. [Google Scholar] [CrossRef]
- Burgess, H.J.; Rizvydeen, M.; Kimura, M.; Pollack, M.H.; Hobfoll, S.E.; Rajan, K.B.; Burns, J.W. An Open Trial of Morning Bright Light Treatment among US Military Veterans with Chronic Low Back Pain: A Pilot Study. Pain Med. 2019, 20, 770–778. [Google Scholar] [CrossRef]
- Carpenter, J.S.; Abelmann, A.C.; Hatton, S.N.; Robillard, R.; Hermens, D.F.; Bennett, M.R.; Lagopoulos, J.; Hickie, I.B. Pineal Volume and Evening Melatonin in Young People with Affective Disorders. Brain Imaging Behav. 2017, 11, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Carriere, C.; Coste, O.; Meiffred-Drouet, M.C.; Barat, P.; Thibault, H. Sleep Disorders in Obese Children Are Not Limited to Obstructive Sleep Apnoea Syndrome. Acta Paediatr. Int. J. Paediatr. 2018, 107, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.Y.; McGlashan, E.M.; Vidafar, P.; Phillips, A.J.K.; Cain, S.W. Advanced Melatonin Onset Relative to Sleep in Women with Unmedicated Major Depressive Disorder. Chronobiol. Int. 2019, 36, 1373–1383. [Google Scholar] [CrossRef] [PubMed]
- Crowley, S.J.; Suh, C.; Molina, T.A.; Fogg, L.F.; Sharkey, K.M.; Carskadon, M.A. Estimating the Dim Light Melatonin Onset of Adolescents within a 6-h Sampling Window: The Impact of Sampling Rate and Threshold Method. Sleep Med. 2016, 20, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, K.; Jansson-Fröjmark, M.; Broman, J.E.; Markström, A. Light Therapy With Scheduled Rise Times in Young Adults With Delayed Sleep Phase Disorder: Therapeutic Outcomes and Possible Predictors. Behav. Sleep Med. 2018, 16, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Vallim, J.R.D.S.; Amaral, F.G.D.; Cipolla-Neto, J.; D’Almeida, V. Rhythmic changes in Fabry disease: Inversion and non-oscillatory pattern in 6-sulfatoxymelatonin daily profile. Chronobiol Int. 2019, 36, 470–480. [Google Scholar] [CrossRef]
- Bumb, J.M.; Mier, D.; Noelte, I.; Schredl, M.; Kirsch, P.; Hennig, O.; Liebrich, L.; Fenske, S.; Alm, B.; Sauer, C.; et al. Associations of Pineal Volume, Chronotype and Symptom Severity in Adults with Attention Deficit Hyperactivity Disorder and Healthy Controls. Eur. Neuropsychopharmacol. 2016, 26, 1119–1126. [Google Scholar] [CrossRef][Green Version]
- Esaki, Y.; Kitajima, T.; Ito, Y.; Koike, S.; Nakao, Y.; Tsuchiya, A.; Hirose, M.; Iwata, N. Wearing Blue Light-Blocking Glasses in the Evening Advances Circadian Rhythms in the Patients with Delayed Sleep Phase Disorder: An Open-Label Trial. Chronobiol. Int. 2016, 33, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Fargason, R.E.; Fobian, A.D.; Hablitz, L.M.; Paul, J.R.; White, B.A.; Cropsey, K.L.; Gamble, K.L. Correcting Delayed Circadian Phase with Bright Light Therapy Predicts Improvement in ADHD Symptoms: A Pilot Study. J. Psychiatr. Res. 2017, 91, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Ferri, L.; Filardi, M.; Moresco, M.; Pizza, F.; Vandi, S.; Antelmi, E.; Toni, F.; Zucchelli, M.; Pierangeli, G.; Plazzi, G. Non-24-Hour Sleep-Wake Rhythm Disorder and Melatonin Secretion Impairment in a Patient with Pineal Cyst. J. Clin. Sleep Med. 2017, 13, 1355–1357. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Flynn-Evans, E.E.; Lockley, S.W. A Pre-Screening Questionnaire to Predict Non-24-Hour Sleep-Wake Rhythm Disorder (N24HSWD) among the Blind. J. Clin. Sleep Med. 2016, 12, 703–710. [Google Scholar] [CrossRef]
- Fukuda, T.; Haraguchi, A.; Takahashi, M.; Nakaoka, T.; Fukazawa, M.; Okubo, J.; Ozaki, M.; Kanatome, A.; Ohya, R.; Miura, Y.; et al. A Randomized, Double-Blind and Placebo-Controlled Crossover Trial on the Effect of l-Ornithine Ingestion on the Human Circadian Clock. Chronobiol. Int. 2018, 35, 1445–1455. [Google Scholar] [CrossRef]
- Ghaziuddin, N.; Shamseddeen, W.; Bertram, H.; McInnis, M.; Wilcox, H.C.; Mitchell, P.B.; Fullerton, J.M.; Roberts, G.M.P.; Glowinski, A.L.; Kamali, M.; et al. Salivary Melatonin Onset in Youth at Familial Risk for Bipolar Disorder. Psychiatry Res. 2019, 274, 49–57. [Google Scholar] [CrossRef]
- Gobert, F.; Luauté, J.; Raverot, V.; Cotton, F.; Dailler, F.; Claustrat, B.; Perrin, F.; Gronfier, C. Is Circadian Rhythmicity a Prerequisite to Coma Recovery? Circadian Recovery Concomitant to Cognitive Improvement in Two Comatose Patients. J. Pineal Res. 2019, 66, e12555. [Google Scholar] [CrossRef]
- Hamers, P.C.M.; Evenhuis, H.M.; Hermans, H. A Multicenter Randomized Controlled Trial for Bright Light Therapy in Adults with Intellectual Disabilities and Depression: Study Protocol and Obstacle Management. Res. Dev. Disabil. 2017, 60, 96–106. [Google Scholar] [CrossRef]
- Lovato, N.; Micic, G.; Gradisar, M.; Ferguson, S.A.; Burgess, H.J.; Kennaway, D.J.; Lack, L. Can the Circadian Phase Be Estimated from Self-Reported Sleep Timing in Patients with Delayed Sleep Wake Phase Disorder to Guide Timing of Chronobiologic Treatment? Chronobiol. Int. 2016, 33, 1376–1390. [Google Scholar] [CrossRef]
- McGlashan, E.M.; Nandam, L.S.; Vidafar, P.; Mansfield, D.R.; Rajaratnam, S.M.W.; Cain, S.W. The SSRI Citalopram Increases the Sensitivity of the Human Circadian System to Light in an Acute Dose. Psychopharmacology 2018, 235, 3201–3209. [Google Scholar] [CrossRef]
- McGlashan, E.M.; Coleman, M.Y.; Vidafar, P.; Phillips, A.J.K.; Cain, S.W. Decreased Sensitivity of the Circadian System to Light in Current, but Not Remitted Depression. J. Affect. Disord. 2019, 256, 386–392. [Google Scholar] [CrossRef]
- Micic, G.; Lovato, N.; Gradisar, M.; Burgess, H.J.; Ferguson, S.A.; Lack, L. Circadian Melatonin and Temperature Taus in Delayed Sleep-wake Phase Disorder and Non-24-hour Sleep-wake Rhythm Disorder Patients: An Ultradian Constant Routine Study. J. Biol. Rhythms. 2016, 31, 387–405. [Google Scholar] [CrossRef]
- Micic, G.; Lovato, N.; Gradisar, M.; Lack, L.C. Personality Differences in Patients with Delayed Sleep–Wake Phase Disorder and Non-24-h Sleep–Wake Rhythm Disorder Relative to Healthy Sleepers. Sleep Med. 2017, 30, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Miyata, R.; Tanuma, N.; Sakuma, H.; Hayashi, M. Circadian Rhythms of Oxidative Stress Markers and Melatonin Metabolite in Patients with Xeroderma Pigmentosum Group A. Oxidative Med. Cell. Longev. 2016, 2016, 5741517. [Google Scholar] [CrossRef] [PubMed]
- Moderie, C.; van der Maren, S.; Dumont, M. Circadian Phase, Dynamics of Subjective Sleepiness and Sensitivity to Blue Light in Young Adults Complaining of a Delayed Sleep Schedule. Sleep Med. 2017, 34, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.M.; Sletten, T.L.; Magee, M.; Gordon, C.; Lovato, N.; Bartlett, D.J.; Kennaway, D.J.; Lack, L.C.; Grunstein, R.R.; Lockley, S.W.; et al. Prevalence of Circadian Misalignment and Its Association with Depressive Symptoms in Delayed Sleep Phase Disorder. Sleep 2017, 40, zsw002. [Google Scholar] [CrossRef]
- Murray, J.M.; Phillips, A.J.K.; Magee, M.; Sletten, T.L.; Gordon, C.; Lovato, N.; Bei, B.; Bartlett, D.J.; Kennaway, D.J.; Lack, L.C.; et al. Sleep Regularity Is Associated with Sleep-Wake and Circadian Timing, and Mediates Daytime Function in Delayed Sleep-Wake Phase Disorder. Sleep Med. 2019, 58, 93–101. [Google Scholar] [CrossRef]
- Naegel, S.; Huhn, J.I.; Gaul, C.; Diener, H.C.; Obermann, M.; Holle, D. No Pattern Alteration in Single Nocturnal Melatonin Secretion in Patients With Hypnic Headache: A Case–Control Study. Headache 2017, 57, 648–653. [Google Scholar] [CrossRef]
- Ogłodek, E.A.; Just, M.J.; Szromek, A.R.; Araszkiewicz, A. Melatonin and Neurotrophins NT-3, BDNF, NGF in Patients with Varying Levels of Depression Severity. Pharmacol. Rep. 2016, 68, 945–951. [Google Scholar] [CrossRef]
- Parry, B.L.; Meliska, C.J.; Lopez, A.M.; Sorenson, D.L.; Martinez, L.F.; Orff, H.J.; Hauger, R.L.; Kripke, D.F. Early versus Late Wake Therapy Improves Mood More in Antepartum versus Postpartum Depression by Differentially Altering Melatonin-Sleep Timing Disturbances. J. Affect. Disord. 2019, 245, 608–616. [Google Scholar] [CrossRef]
- Robillard, R.; Carpenter, J.S.; Rogers, N.L.; Fares, S.; Grierson, A.B.; Hermens, D.F.; Naismith, S.L.; Mullin, S.J.; Feilds, K.L.; Glozier, N.; et al. Circadian Rhythms and Psychiatric Profiles in Young Adults with Unipolar Depressive Disorders. Transl. Psychiatry 2018, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.S.; Giacheti, C.M.; Dornelas, L.S.; Silva, N.C.; Souza, A.L.D.M.; Guissoni Campos, L.M.; Pinato, L. Day/Night Melatonin Content in Cerebral Palsy. Neurosci. Lett. 2018, 686, 23–27. [Google Scholar] [CrossRef]
- Shimada, M.; Seki, H.; Samejima, M.; Hayase, M.; Shirai, F. Salivary Melatonin Levels and Sleep-Wake Rhythms in Pregnant Women with Hypertensive and Glucose Metabolic Disorders: A Prospective Analysis. BioScience Trends 2016, 10, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Slyepchenko, A.; Allega, O.R.; Leng, X.; Minuzzi, L.; Eltayebani, M.M.; Skelly, M.; Sassi, R.B.; Soares, C.N.; Kennedy, S.H.; Frey, B.N. Association of Functioning and Quality of Life with Objective and Subjective Measures of Sleep and Biological Rhythms in Major Depressive and Bipolar Disorder. Aust. N. Z. J. Psychiatry 2019, 53, 683–696. [Google Scholar] [CrossRef] [PubMed]
- Solaiman, S.S.; Agrawal, R. Non-24-Hour Sleep-Wake Circadian Rhythm Disorder in a Sighted Male with Normal Functioning. J. Clin. Sleep Med. 2018, 14, 483–484. [Google Scholar] [CrossRef]
- Solheim, B.; Olsen, A.; Kallestad, H.; Langsrud, K.; Bjorvatn, B.; Gradisar, M.; Sand, T. Cognitive Performance in DSWPD Patients upon Awakening from Habitual Sleep Compared with Forced Conventional Sleep. J. Sleep Res. 2019, 28, e12730. [Google Scholar] [CrossRef]
- Maria, S.; Swanson, M.H.; Enderby, L.T.; D’Amico, F.; Enderby, B.; Samsonraj, R.M.; Dudakovic, A.; van Wijnen, A.J.; Witt-Enderby, P.A. Melatonin-micronutrients Osteopenia Treatment Study (MOTS): A Translational Study Assessing Melatonin, Strontium (Citrate), Vitamin D3 and Vitamin K2 (MK7) on Bone Density, Bone Marker Turnover and Health Related Quality of Life in Postmenopausal Osteopen. Aging 2017, 9, 256–285. [Google Scholar] [CrossRef]
- Swanson, G.R.; Siskin, J.; Gorenz, A.; Shaikh, M.; Raeisi, S.; Fogg, L.; Forsyth, C.; Keshavarzian, A. Disrupted Diurnal Oscillation of Gut-Derived Short Chain Fatty Acids in Shift Workers Drinking Alcohol: Possible Mechanism for Loss of Resiliency of Intestinal Barrier in Disrupted Circadian Host. Transl. Res. 2020, 221, 97–109. [Google Scholar] [CrossRef]
- van der Maren, S.; Moderie, C.; Duclos, C.; Paquet, J.; Daneault, V.; Dumont, M. Daily Profiles of Light Exposure and Evening Use of Light-Emitting Devices in Young Adults Complaining of a Delayed Sleep Schedule. J. Biol. Rhythm. 2018, 33, 192–202. [Google Scholar] [CrossRef]
- Watson, L.A.; Phillips, A.J.K.; Hosken, I.T.; McGlashan, E.M.; Anderson, C.; Lack, L.C.; Lockley, S.W.; Rajaratnam, S.M.W.; Cain, S.W. Increased Sensitivity of the Circadian System to Light in Delayed Sleep–Wake Phase Disorder. J. Physiol. 2018, 596, 6249–6261. [Google Scholar] [CrossRef]
- Weissová, K.; Škrabalová, J.; Skálová, K.; Červená, K.; Bendová, Z.; Miletínová, E.; Kopřivová, J.; Šonka, K.; Dudysová, D.; Bartoš, A.; et al. Circadian Rhythms of Melatonin and Peripheral Clock Gene Expression in Idiopathic REM Sleep Behavior Disorder. Sleep Med. 2018, 52, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.; Reid, K.J.; Braun, R.I.; Abbott, S.M.; Zee, P.C. Habitual Light Exposure Relative to Circadian Timing in Delayed Sleep-Wake Phase Disorder. Sleep 2018, 41, zsy166. [Google Scholar] [CrossRef] [PubMed]
- Zuculo, G.M.; Gonçalves, B.S.B.; Brittes, C.; Menna-Barreto, L.; Pinato, L. Melatonin and Circadian Rhythms in Autism: Case Report. Chronobiol. Int. 2017, 34, 527–530. [Google Scholar] [CrossRef] [PubMed]
| Author | Number of Participants | Treatment | Measurement | Results |
|---|---|---|---|---|
| Allega et al., 2018 [45] | Total N = 103 MDD = 32 BD = 21 HC = 40 | No Treatment | Activity, 6-sulphatoxymelatonin (urine) | Total BRIAN score correlated with wake after sleep onset, total activity count during sleep, and urinary 6-sulphatoxymelatonin. BRIAN |
| Baandrup et al., 2016 [46] | Total N = 48 PRM = 20 Placebo = 28 | 2 mg PRM or Placebo | Active-rest cycle | Melatonin can aid disrupted circadian cycle caused by benzodiazepine withdraw |
| Baker et al., 2017 [47] | ASD Only = 16 ASD with comorbidities = 12 Controls = 32 | No treatment | Activity, salivary melatonin | Lower mean melatonin lower in the ASD with comorbidities |
| Bock et al., 2016 [48] | N = 67 | No treatment | Pharmacotherapy in pediatric insomnia | Use of over-the-counter medicine and prescription have been recommended, however only 20% have formal training in pediatric sleep disorders |
| Bradley et al., 2017 [49] | BD = 46 Control = 42 | No treatment | Sleep, melatonin, mood | 50% of BD patients had abnormal sleep and lower melatonin secretion levels |
| Buber et al., 2016 [50] | ADHD = 27 Control = 28 | No treatment | 6-sulphatoxymelatonin | Melatonin production increased in ADHD patients |
| Burgess et al., 2016 [51] | DSWPD = 32 | No treatment | Salivary melatonin | Demonstrated feasibility of at home salivary melatonin collection |
| Burgess et al., 2017 [52] | DSWPD = 22 Control = 18 | No treatment | Salivary melatonin | Individuals with DSWPD had more variable sleep times |
| Burgess et al., 2019 [53] | N = 37 | Bright light treatment | Pain, mood, sleep, circadian timing | Morning bright light treatment reduced pain intensity, post-traumatic stress disorder symptoms and increased sleep-in veterans |
| Carpenter et al., 2017 [54] | N = 50 | No treatment | Salivary melatonin | Melatonin levels are related to social and occupational functioning, and timing and length of sleep |
| Carriere et al., 2018 [55] | N = 126 | Various for sleep apnea | Polysomnography | Less than 18% of subjects received melatonin for sleep disorders |
| Coleman et al., 2019 [56] | Major depressive N = 8 Previous episode N = 9 Controls = 31 | No treatment | Salivary melatonin | Actively depressed individuals had earlier melatonin onset |
| Crowley et al., 2016 [57] | N = 66 adolescents School enrollment = 34 Summer enrollment = 32 | No treatment | Salivary melatonin | Development of a method to determine circadian phase in older adolescents |
| Danielsson et al., 2018 [58] | DSWPD = 57 | Light therapy | Salivary melatonin | Daily use of the lamp assisted in prediction of sleep onset and offset |
| Vallim JRDS et al., 2018 [59] | N = 17 | No treatment | Urine 6-sulfatoxymelatonin | Alterations in the circadian rhythm of patients with Fabry diseases |
| Bumb et al., 2016 [60] | ADHD = 74 Controls = 86 | No treatment | Pineal gland volume | Pineal gland volume smaller in unmedicated ADHD patients |
| Esaki et al., 2016 [61] | DSWPD = 9 | Amber glasses | Activity, salivary melatonin | Use of amber glasses advanced melatonin and sleep onset |
| Fargason et al., 2017 [62] | ADHD = 16 | Bright light therapy | Salivary melatonin | Bright light therapy advanced melatonin onset, and decreased ADHD symptoms |
| Ferri et al., 2017 [63] | N = 1 | Melatonin | Activity, serum melatonin | Treatment with melatonin realigned sleep-wake rhythm |
| Flynn-Evans et al., 2016 [64] | N = 127 blind women | No treatment | Urine 6-sulfatoxymelatonin | Developed a potential treatment model |
| Fukuda et al., 2018 [65] | N = 28 (male = 22, female = 6) | 500 mg L-ornithine or placebo | Salivary melatonin, sleep quality | Melatonin onset delayed after ingestion of L-ornithine |
| Ghaziuddin et al., 2019 [66] | High Risk BD = 6 Low Risk BP = 6 | No treatment | Salivary melatonin | High risk BD had earlier melatonin onset, and Low Risk BD spent more time in deep sleep |
| Gobert et al., 2019 [67] | Persistent Disorder of Consciousness = 2 | No treatment | Urine 6- sulfatoxymelatonin | Circadian timing system was functional, and in sync with the environmental light-dark cycle |
| Hamers et al., 2017 [68] | 10,000 lux = 57 <499 lux = 57 Standard care = 57 | Bright light treatment | Salivary melatonin, cortisol in scalp | Inconclusive |
| Lovato et al., 2016 [69] | DSWPD = 24 | No treatment | Body temperature, salivary melatonin | Self-reported sleep timing may predict therapeutically relevant circadian phase |
| McGlashan et al., 2018 [70] | N = 12 | Citalopram or placebo | Salivary melatonin | Citalopram had an effect on melatonin suppression response to light, including a 47% increase in suppression observed after an acute dose of citalopram |
| McGlashan et al., 2019 [71] | Depression = 16 Controls = 31 | No treatment | Salivary melatonin | Patients with current depression had lower levels of melatonin suppression to light |
| Micic et al., 2016 [72] | DSWPD = 36 (17 male, 9 female) | No treatment | Core temperature, salivary melatonin | DSWPD patients have significantly longer melatonin rhythm and temperature taus |
| Micic et al., 2017 [73] | DSWPD = 16 N24SWD = 3 Control = 14 | No treatment | Personality factors, Sleep/wake cycle, salivary melatonin | Compared to controls, DSWPD patients had nigher neuroticism, lower extraversion, conscientiousness and agreeableness. |
| Miyata et al., 2016 [74] | XPA = 8 Controls = 8 | No treatment | Urine 6- sulfatoxymelatonin, DNA damage markers | XPA patients had a lower peak melatonin, increase in DNA damage markers |
| Moderie et al., 2017 [75] | N = 14 | No treatment | Salivary melatonin | Melatonin onset was delayed and increase in sleepiness in those with a delayed bedtime |
| Murray et al., 2017 [76] | N = 182 | No treatment | Sleep diary, activity, salivary melatonin | Melatonin onset occurred later in individuals with circadian DSWPD, along with increased odds of mild depressive symptoms |
| Murray et al., 2019 [77] | DSWPD = 20 Controls = 16 | No treatment | Salivary melatonin | DSWPD patients had reduced reaction times, greater response speed variability in the morning |
| Naegel et al., 2017 [78] | Hypnic headache = 9 Control = 9 | No treatment | Serum melatonin at specific times, and headache attacks | No difference in melatonin secretions between headache and control patients |
| Oglodek et al., 2016 [79] | Severely depressed = 40 Moderate depression = 40 Mild depression = 40 Control = 40 | No treatment | Salivary melatonin | Highest melatonin at 3:00AM in severely depressed females, but lower in patients with mild and moderate depression |
| Parry et al., 2019 [80] | Antepartum = 26 Postpartum = 24 | Early night wake therapy vs. late-night wake therapy | Plasma melatonin, mood | Early wake time improved mood in antepartum, late wake time improved mood more in postpartum individuals |
| Robillard et al., 2018 [81] | Unipolar depressive = 35 Healthy controls = 15 | No treatment | Salivary melatonin, body temperature | Delayed circadian rhythm found in 40% of individuals with depressive disorder |
| Santos et al., 2018 [82] | Cerebral Palsy = 33 | No treatment | Salivary melatonin | Cerebral Palsy patients had higher diurnal and lower nocturnal melatonin |
| Shimada et al., 2016 [83] | Pregnant women with pregnancy-related complications = 58 | No treatment | Salivary melatonin | Pregnant women with hypertensive or glucose disorder complications had lower melatonin secretion through the day |
| Slyepchenko et al., 2019 [84] | Total N = 111 MDD = 38 BD = 33 Controls = 40 | No Treatment | 6-sulfatoxymelatonin | Levels of 6-sulfatoxymelatonin were lower in BD patients |
| Solaiman and Agrawal 2018 [85] | Total N = 111 MDD = 38 BD = 33 Controls = 40 | No Treatment | 6-sulfatoxymelatonin | Individuals with DSWPD have more irregular sleep, caused by timing of sleep relative to circadian phase |
| Solheim et al., 2019 [86] | N24SWD = 1 | 3 mg melatonin | Sleep diary | Sleep disorder under adequate control |
| Maria et al., 2017 [87] | MDD = 30 | 20–40 mg of fluoxetine | Salivary melatonin | Advancement of melatonin onset relative to sleep is associated with more severe depression symptoms in men, where for women a shorter window is associated with more severe depression symptoms after 2 weeks of fluoxetine |
| Swanson et al., 2020 [88] | Alcohol use disorder, no liver disease = 20 Control day workers = 11 Control night workers = 11 | 0.5 g of alcohol/kg body weight for 7 days after work, prior to bedtime | Plasma melatonin, rest-activity rhythm | Chronic and moderate consumption of alcohol for 1 week disrupted circadian rhythm |
| Van der Maren et al., 2018 [89] | N = 28 Delayed group = 14 Non-delayed group = 14 | No treatment | Salivary melatonin | In delayed group, there was a higher exposure to white and blue light after melatonin onset |
| Watson et al., 2018 [90] | DSWPD = 12 Controls = 12 | No treatment | Salivary melatonin, Phase shifting | Greater phase delay shift and increased light sensitivity in DSWPD patients. |
| Weissova et al., 2018 [91] | RBD patients = 10 Controls = 10 | No treatment | EEG, EOG, EMG, ECG, circadian rhythm analysis, Serum melatonin | Melatonin profile in RBD patients was delayed 2 h compared to controls, and dispersed melatonin range |
| Wilson et al., 2018 [92] | DSWPD = 12 Control = 12 | No treatment | Plasma melatonin | DSWPD individuals had a later light exposure pattern |
| Zuculo et al., 2017 [93] | N = 1 | 3 mg melatonin | Activity, behavior | Melatonin aids in synchronizing endogenous rhythms |
| Sleep or Circadian Disorder | Time Criteria |
|---|---|
| Insomnia | Episodic: >1 month and <3 months; Persistent: >3 months; Recurrent: >2 episodes within the space of 1 year |
| Hypersomnolence Disorder | Acute: <1 month; Subacute: 1–3 months; Persistent: >3 months |
| Narcolepsy | 3 episodes/week over 3 months |
| Obstructive Sleep Apnea Hypopnea | 5 obstructive apneas/hour of sleep |
| Central Sleep Apnea | 5 central apneas/hour of sleep |
| Sleep-related Hypoventilation | No time criteria |
| Circadian Rhythm Sleep-Wake Disorders (6 subtypes–Delayed Sleep Phase type, Advanced Sleep Phase type, Irregular Sleep-Wake type, Non-24-h Sleep-Wake type, Shift Work type, and Unspecified type) | Episodic: >1 month and <3 months; Persistent: >3 months; Recurrent: >2 episodes within the space of 1 year |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasey, C.; McBride, J.; Penta, K. Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. Nutrients 2021, 13, 3480. https://doi.org/10.3390/nu13103480
Vasey C, McBride J, Penta K. Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. Nutrients. 2021; 13(10):3480. https://doi.org/10.3390/nu13103480
Chicago/Turabian StyleVasey, Clayton, Jennifer McBride, and Kayla Penta. 2021. "Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin" Nutrients 13, no. 10: 3480. https://doi.org/10.3390/nu13103480
APA StyleVasey, C., McBride, J., & Penta, K. (2021). Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. Nutrients, 13(10), 3480. https://doi.org/10.3390/nu13103480





