Protective Effects of Fermented Paprika (Capsicum annuum L.) on Sodium Iodate-Induced Retinal Damage
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Antibodies
2.2. Fermentation and Preparation of Paprika Samples
2.3. Analysis Physicochemical and Antioxidant Effect
2.4. Cell Culture
2.5. Cell Viability Assay
2.6. Measurement of Intracellular Reactive Oxygen Species (ROS) Increase
2.7. Measurement of NO Production in RAW 264.7 Cells
2.8. Animals and Treatment
2.9. Determination of Antioxidant Activity
2.10. Histology of Ocular Tissues
2.11. Immunoblotting
2.12. Statistical Analysis
3. Results
3.1. Physicochemical Content and Radical Sacavenging Ability
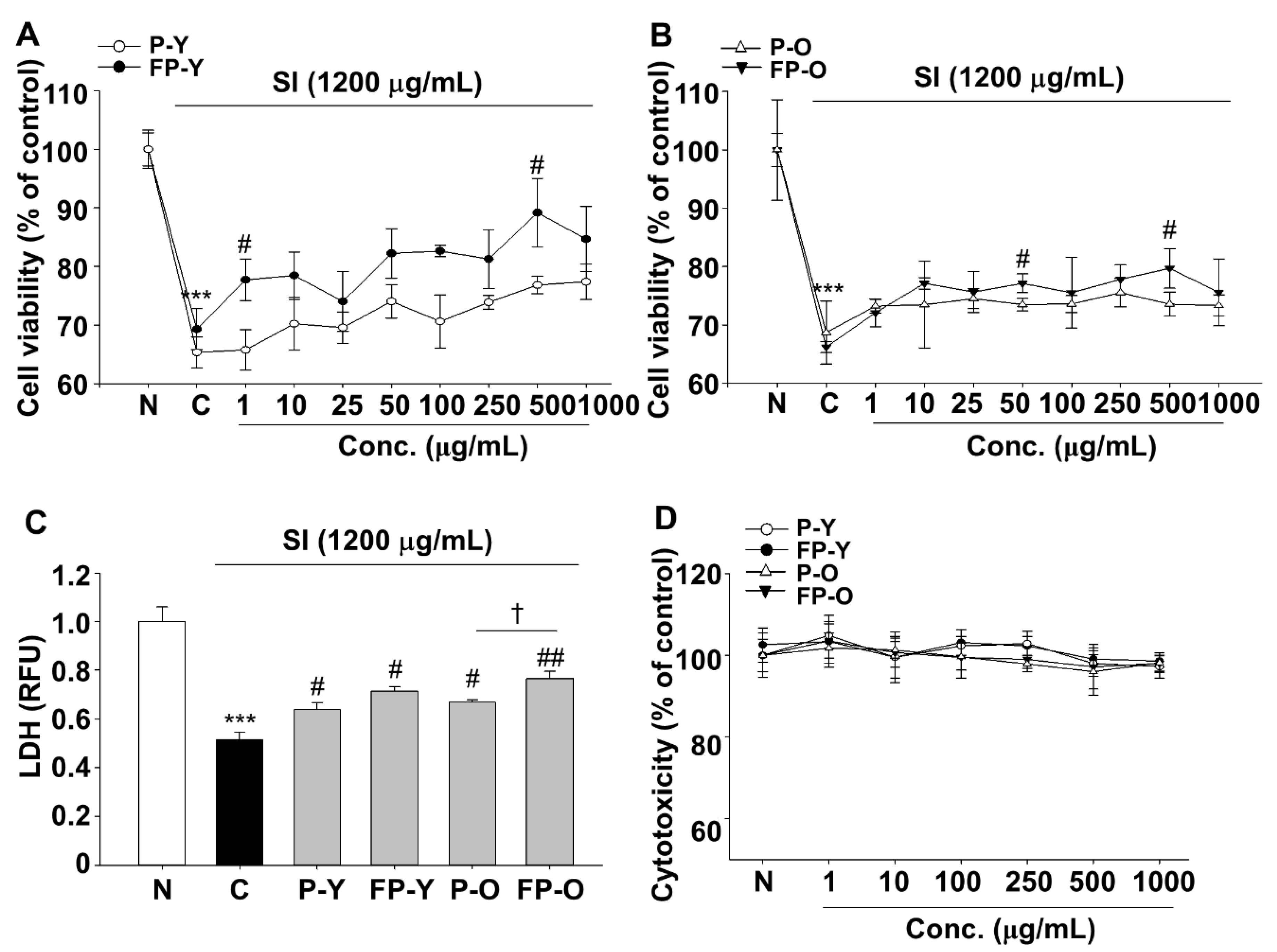
3.2. Effects of Fermented Paprika (FP) on Sodium Iodate (SI)-Induced Damage in Human Retinal Pigment Epithelial Cell line (ARPE-19)
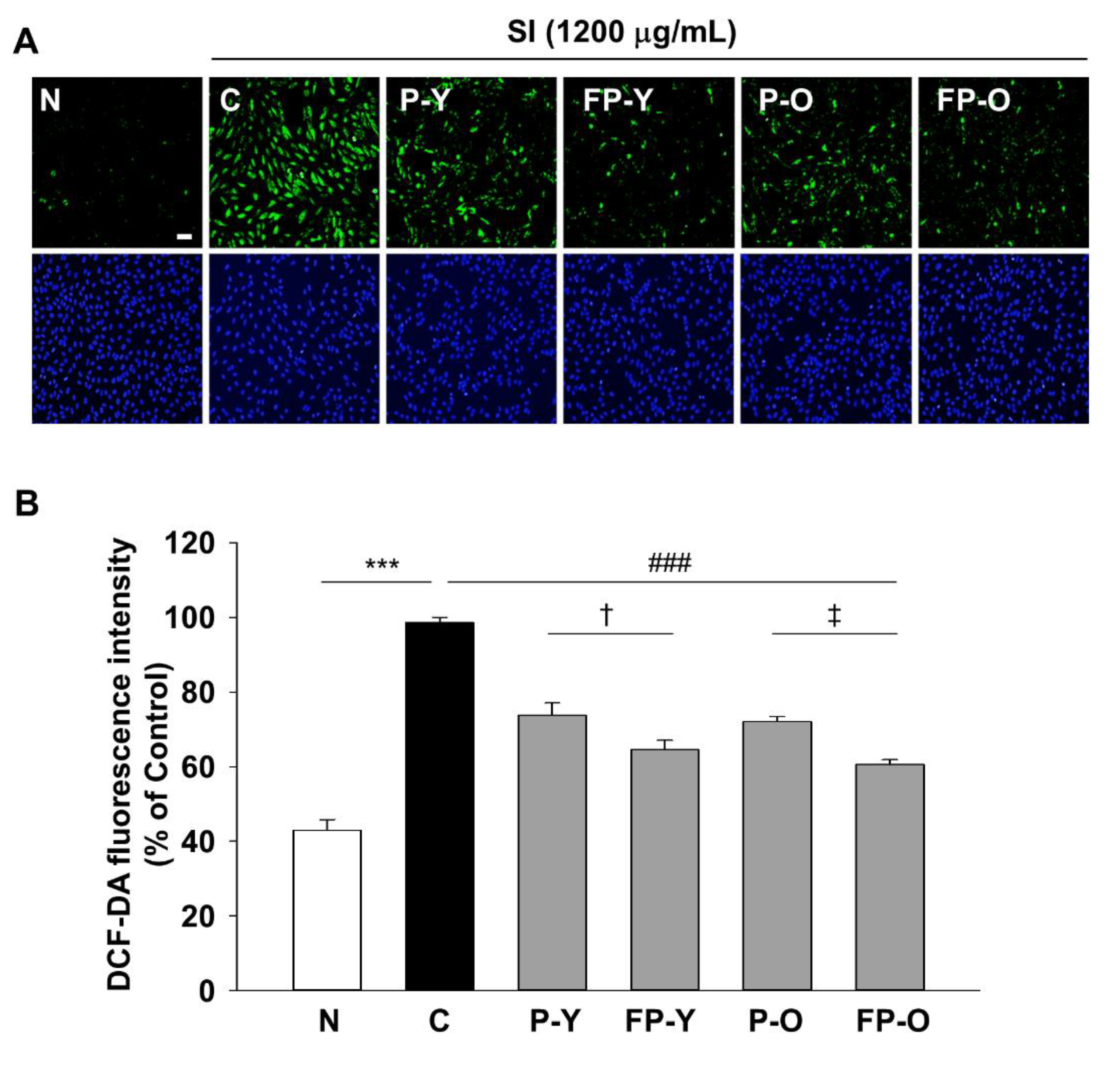
3.3. Effects of FP on SI-Induced Intracellular ROS Generation in ARPE-19 Cells
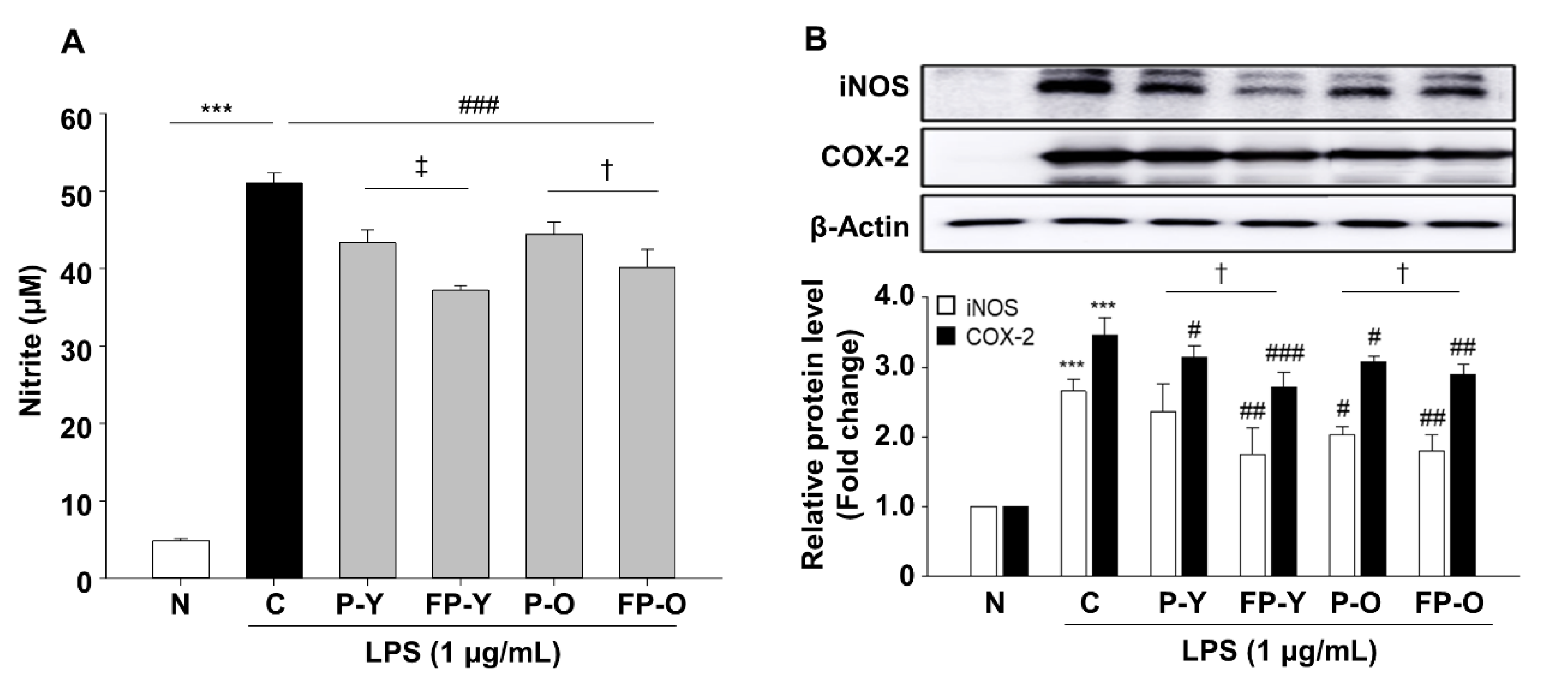
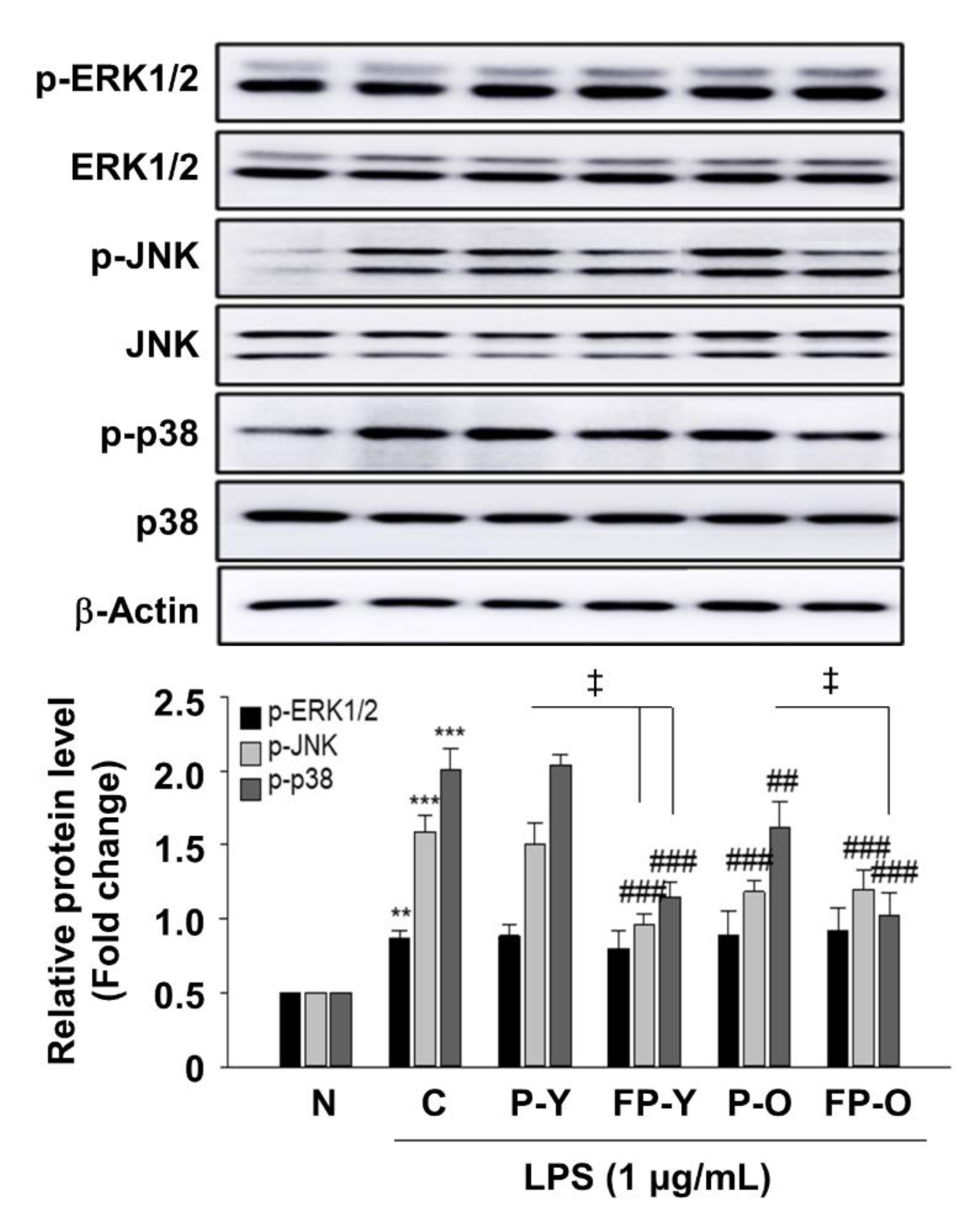
3.4. Effect of FP on Lipopolysaccharide (LPS)-Induced Nitric Oxide (NO) Production in RAW 264.7 Macrophages
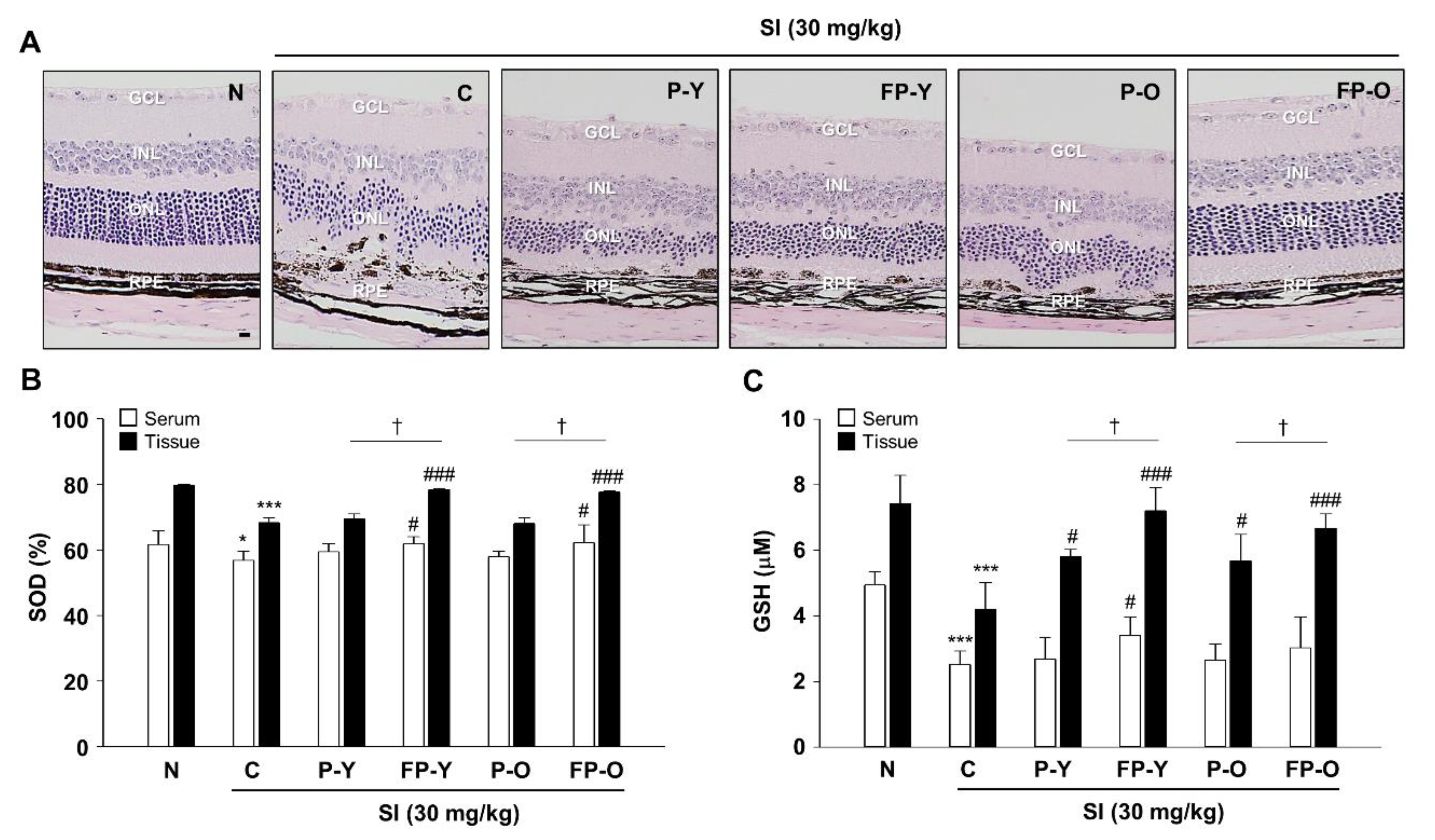
3.5. Preservation Efect of Fermented Paprika Extract (FPE) on Retinal Structure and Oxidative Stress in SI-Induced Retinal Degeneration Mose Model
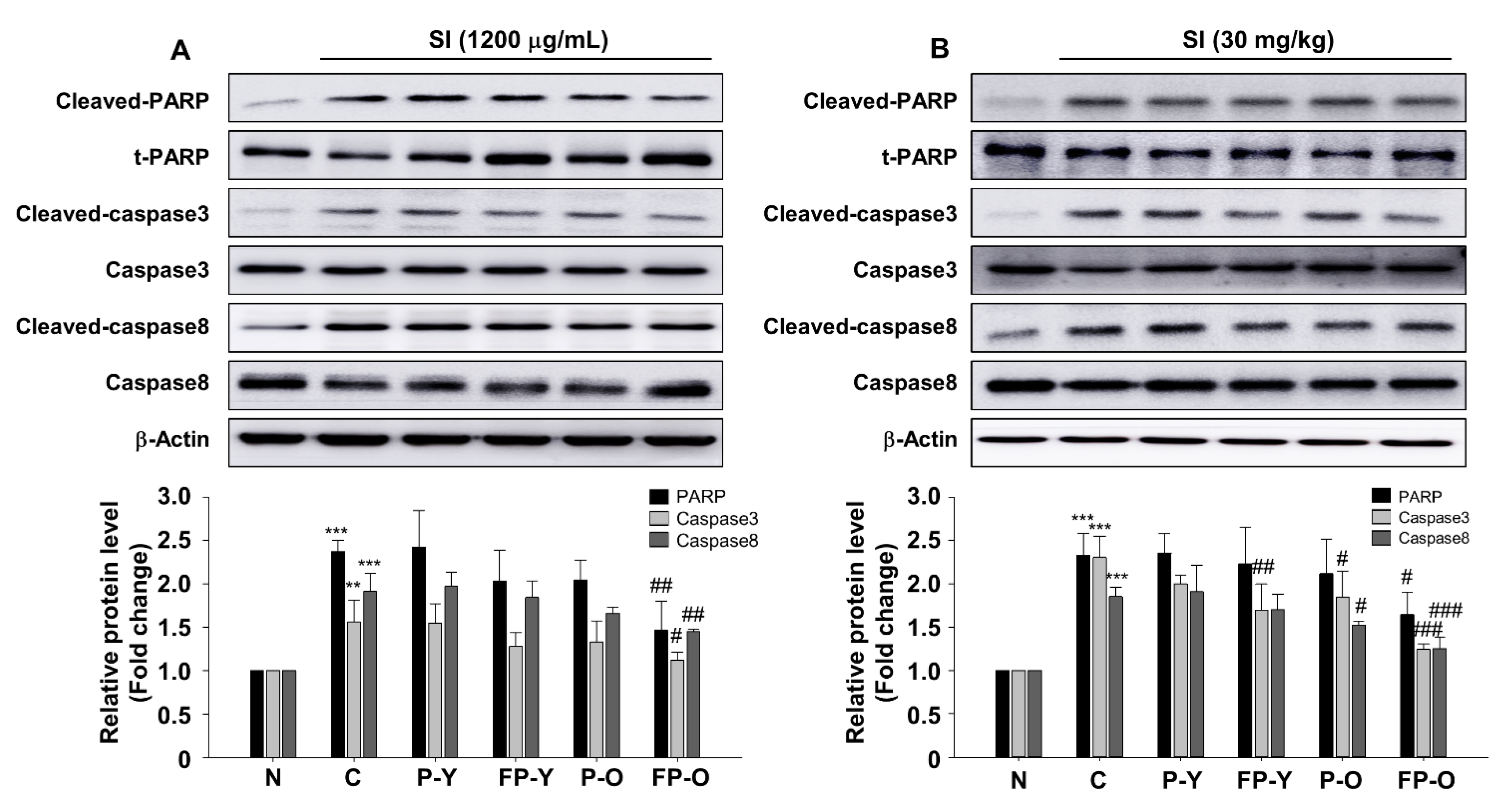
3.6. FP Inhibits SI-Induced Apoptosis in ARPE-19 Cells and Mouse Retina
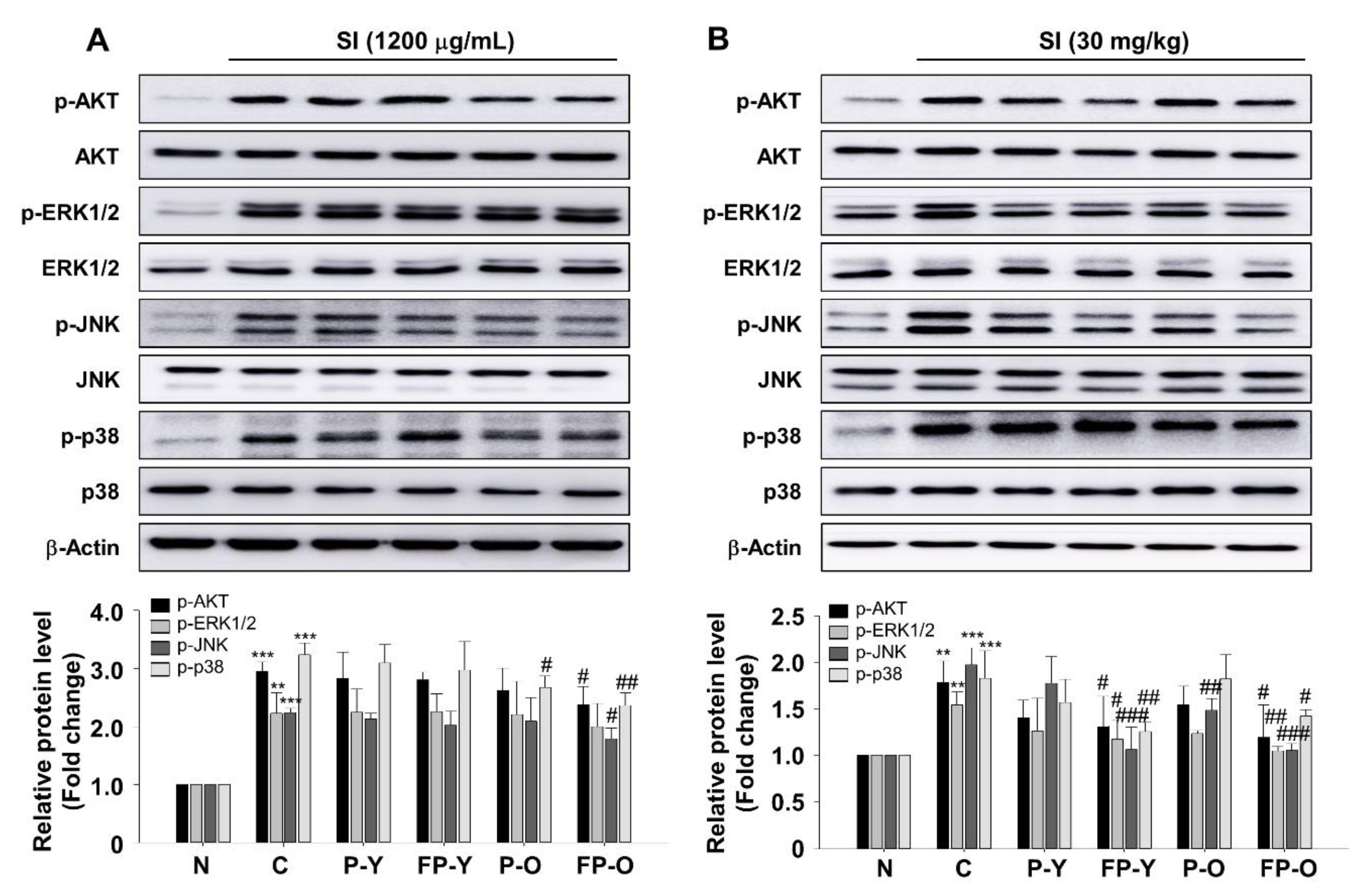
3.7. Effect of FP on SI-Mediated AKT and Mitogen-Activated Protein Kinases (MAPK) Signaling In Vitro and In Vivo
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- van Lookeren Campagne, M.; LeCouter, J.; Yaspan, B.L.; Ye, W. Mechanisms of age-related macular degeneration and therapeutic opportunities. J. Pathol. 2014, 232, 151–164. [Google Scholar] [CrossRef]
- Anderson, R.E.; Rapp, L.M.; Wiegand, R.D. Lipid peroxidation and retinal degeneration. Curr. Eye Res. 1984, 3, 223–227. [Google Scholar] [CrossRef]
- Pujol-Lereis, L.M.; Schäfer, N.; Kuhn, L.B.; Rohrer, B.; Pauly, D. Interrelation between oxidative stress and complement activation in models of age-related macular degeneration. Adv. Exp. Med. Biol. 2016, 854, 87–93. [Google Scholar] [CrossRef]
- Beatty, S.; Koh, H.; Phil, M.; Henson, D.; Boulton, M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv. Ophthalmol. 2000, 45, 115–134. [Google Scholar] [CrossRef]
- Jabbehdari, S.; Handa, J.T. Oxidative stress as a therapeutic target for the prevention and treatment of early age-related macular degeneration. Surv. Ophthalmol. 2020, in press. [Google Scholar] [CrossRef]
- Kim, H.-R.; Jeong, D.-H.; Kim, S.; Lee, S.-W.; Sin, H.-S.; Yu, K.-Y.; Jeong, S.-I.; Kim, S.-Y. Fermentation of blackberry with L. plantarum JBMI F5 enhance the protection effect on UVB-mediated photoaging in human foreskin fibroblast and hairless mice through regulation of MAPK/NF-κB signaling. Nutrients 2019, 11, 2429. [Google Scholar] [CrossRef] [PubMed]
- Chinskey, N.D.; Besirli, C.G.; Zacks, D.N. Retinal cell death and current strategies in retinal neuroprotection. Curr. Opin. Ophthalmol. 2014, 25, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Ao, J.; Wood, J.P.; Chidlow, G.; Gillies, M.C.; Casson, R.J. Retinal pigment epithelium in the pathogenesis of age-related macular degeneration and photobiomodulation as a potential therapy? Clin. Exp. Ophthalmol. 2018, 46, 670–686. [Google Scholar] [CrossRef] [PubMed]
- Faghiri, Z.; Bazan, N.G. PI3K/Akt and mTOR/p70S6K pathways mediate neuroprotectin D1-induced retinal pigment epithelial cell survival during oxidative stress-induced apoptosis. Exp. Eye Res. 2010, 90, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Liu, J.; Wang, Z.; Yu, L.L.; Wang, J. Piceatannol protects human retinal pigment epithelial cells against hydrogen peroxide induced oxidative stress and apoptosis through modulating PI3K/Akt signaling pathway. Nutrients 2019, 11, 1515. [Google Scholar] [CrossRef]
- Kyosseva, S.V. Targeting MAPK signaling in age-related macular degeneration. Ophthalmol. Eye Dis. 2016, 8, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.R.; Azevedo, J.; Pereira, M.J.; Valentão, P.; Andrade, P.B. Chemical assessment and antioxidant capacity of pepper (Capsicum annuum L.) seeds. Food Chem. Toxicol. 2013, 53, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Xu, Z.; Wu, C.T.; Janes, M.; Prinyawiwatkul, W.; No, H.K. Antioxidant activities of different colored sweet bell peppers (Capsicum annuum L.). J. Food. Sci. 2007, 72, S98–S102. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.A.; Mahmood, T.; Ali, M.; Saeed, A.; Maalik, A. Pharmacological importance of an ethnobotanical plant: Capsicum annuum L. Nat. Prod. Res. 2014, 28, 1267–1274. [Google Scholar] [CrossRef]
- Hur, S.J.; Lee, S.Y.; Kim, Y.-C.; Choi, I.; Kim, G.-B. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chem. 2014, 160, 346–356. [Google Scholar] [CrossRef]
- Torino, M.I.; Limón, R.I.; Martínez-Villaluenga, C.; Mäkinen, S.; Pihlanto, A.; Vidal-Valverde, C.; Frias, J. Antioxidant and antihypertensive properties of liquid and solid state fermented lentils. Food Chem. 2013, 136, 1030–1037. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Chun, O.K. Comparison of ABTS/DPPH assays for the detection of antioxidant capacity in foods. FASEB J. 2010, 24, 535–539. [Google Scholar] [CrossRef]
- Eigler, A.; Moeller, J.; Endres, S. Exogenous and endogenous nitric oxide attenuates tumor necrosis factor synthesis in the murine macrophage cell line RAW 264.7. J. Immunol. 1995, 154, 4048–4054. [Google Scholar]
- Wei, X.; Luo, M.; Xu, L.; Zhang, Y.; Lin, X.; Kong, P.; Liu, H. Production of fibrinolytic enzyme from Bacillus amyloliquefaciens by fermentation of chickpeas, with the evaluation of the anticoagulant and antioxidant properties of chickpeas. J. Agric. Food Chem. 2011, 59, 3957–3963. [Google Scholar] [CrossRef]
- Yan, Y.; Zhang, F.; Chai, Z.; Liu, M.; Battino, M.; Meng, X. Mixed fermentation of blueberry pomace with L. rhamnosus GG and L. plantarum-1: Enhance the active ingredient, antioxidant activity and health-promoting benefits. Food Chem. Toxicol. 2019, 131, 110541. [Google Scholar] [CrossRef]
- Stanisavljević, N.; Samardžić, J.; Janković, T.; Šavikin, K.; Mojsin, M.; Topalović, V.; Stevanović, M. Antioxidant and antiproliferative activity of chokeberry juice phenolics during in vitro simulated digestion in the presence of food matrix. Food Chem. 2015, 175, 516–522. [Google Scholar] [CrossRef] [PubMed]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell. Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef] [PubMed]
- Enzmann, V.; Row, B.W.; Yamauchi, Y.; Kheirandish, L.; Gozal, D.; Kaplan, H.J.; McCall, M.A. Behavioral and anatomical abnormalities in a sodium iodate-induced model of retinal pigment epithelium degeneration. Exp. Eye Res. 2006, 82, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; An, Y.; He, X.; Zhang, D.; He, W. Protection of Kaempferol on oxidative stress-induced retinal pigment epithelial cell damage. Oxidative Med. Cell. Longev. 2018, 2018. [Google Scholar] [CrossRef]
- Wang, J.; Iacovelli, J.; Spencer, C.; Saint-Geniez, M. Direct effect of sodium iodate on neurosensory retina. Invest. Ophthalmol. Vis. Sci. 2014, 55, 1941–1953. [Google Scholar] [CrossRef]
- Balmer, J.; Zulliger, R.; Roberti, S.; Enzmann, V. Retinal cell death caused by sodium iodate involves multiple caspase-dependent and caspase-independent cell-death pathways. Int. J. Mol. Sci. 2015, 16, 15086–15103. [Google Scholar] [CrossRef]
- Liu, C.; Li, Y.; Peng, M.; Laties, A.M.; Wen, R. Activation of caspase-3 in the retina of transgenic rats with the rhodopsin mutation s334ter during photoreceptor degeneration. J. Neurosci. 1999, 19, 4778–4785. [Google Scholar] [CrossRef]
- Sanvicens, N.; Gómez-Vicente, V.; Masip, I.; Messeguer, A.; Cotter, T.G. Oxidative stress-induced apoptosis in retinal photoreceptor cells is mediated by calpains and caspases and blocked by the oxygen radical scavenger CR-6. J. Biol. Chem. 2004, 279, 39268–39278. [Google Scholar] [CrossRef]
- Gastman, B.R.; Johnson, D.E.; Whiteside, T.L.; Rabinowich, H. Tumor-induced apoptosis of T lymphocytes: Elucidation of intracellular apoptotic events. Blood 2000, 95, 2015–2023. [Google Scholar] [CrossRef]
- Yang, M.; So, K.F.; Lam, W.C.; Lo, A.C.Y. Novel programmed cell death as therapeutic targets in age-related macular degeneration? Int. J. Mol. Sci. 2020, 21, 7279. [Google Scholar] [CrossRef]
- Singh, P.K.; Kumar, A. Mitochondria mediates caspase-dependent and independent retinal cell death in Staphylococcus aureus endophthalmitis. Cell Death Discov. 2016, 2, 16034. [Google Scholar] [CrossRef]
- Wang, Z.; Bartosh, T.; Ding, M.; Roque, R. MAPKs Modulate RPE response to oxidative stress. J. Med. Bioeng. 2014, 3, 67–73. [Google Scholar]
- Yang, Y.; Qin, Y.J.; Yip, Y.W.Y.; Chan, K.P.; Chu, K.O.; Chu, W.K.; Ng, T.K.; Pang, C.P.; Chan, S.O. Green tea catechins are potent anti-oxidants that ameliorate sodium iodate-induced retinal degeneration in rats. Sci. Rep. 2016, 6, 29546. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Teng, J.; Lyu, Y.; Hu, X.; Zhao, Y.; Wang, M. Enhanced antioxidant activity for apple juice fermented with Lactobacillus plantarum ATCC14917. Molecules 2018, 24, 51. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Lin, M.-W.; Liang, C.-J.; Wang, S.-H. The anti-inflammatory effects and mechanisms of Eupafolin in Lipopolysaccharide-induced inflammatory responses in RAW264.7 macrophages. PLoS ONE 2016, 11, e0158662. [Google Scholar] [CrossRef] [PubMed]
| TPC (GAE, mg/g) | TFC (QE, mg/g) | VC (mg/100 g) | DPPH Radical Scavenging Activity (%) | ABTS Radical Scavenging Activity (%) | |
|---|---|---|---|---|---|
| P-Y | 81.59 ± 0.01 | 8.78 ± 0.04 | 1.98 ± 0.02 | 24.29 ± 0.56 | 48.25 ± 1.21 |
| P-O | 70.52 ± 0.11 | 8.00 ± 0.09 | 1.88 ± 0.02 | 25.09 ± 0.86 | 50.70 ± 0.39 |
| FP-Y | 84.17 ± 0.54 | 9.59 ± 0.85 | 2.05 ± 0.07 | 34.51 ** ± 0.54 | 55.14 ** ± 1.28 |
| FP-O | 81.53 ± 0.40 | 8.69 ± 0.29 | 2.03 # ± 0.03 | 28.26 ## ± 1.22 | 53.45 # ± 0.24 |
| AA | - | - | 95.52 ± 0.12 | - | |
| Trolox | - | - | - | 94.66 ± 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.-R.; Kim, S.; Lee, S.-W.; Sin, H.-S.; Kim, S.-Y. Protective Effects of Fermented Paprika (Capsicum annuum L.) on Sodium Iodate-Induced Retinal Damage. Nutrients 2021, 13, 25. https://doi.org/10.3390/nu13010025
Kim H-R, Kim S, Lee S-W, Sin H-S, Kim S-Y. Protective Effects of Fermented Paprika (Capsicum annuum L.) on Sodium Iodate-Induced Retinal Damage. Nutrients. 2021; 13(1):25. https://doi.org/10.3390/nu13010025
Chicago/Turabian StyleKim, Ha-Rim, Sol Kim, Sang-Wang Lee, Hong-Sig Sin, and Seon-Young Kim. 2021. "Protective Effects of Fermented Paprika (Capsicum annuum L.) on Sodium Iodate-Induced Retinal Damage" Nutrients 13, no. 1: 25. https://doi.org/10.3390/nu13010025
APA StyleKim, H.-R., Kim, S., Lee, S.-W., Sin, H.-S., & Kim, S.-Y. (2021). Protective Effects of Fermented Paprika (Capsicum annuum L.) on Sodium Iodate-Induced Retinal Damage. Nutrients, 13(1), 25. https://doi.org/10.3390/nu13010025





