Therapeutic Potential of Natural Products in Treatment of Cervical Cancer: A Review
Abstract
1. Introduction
2. Methods
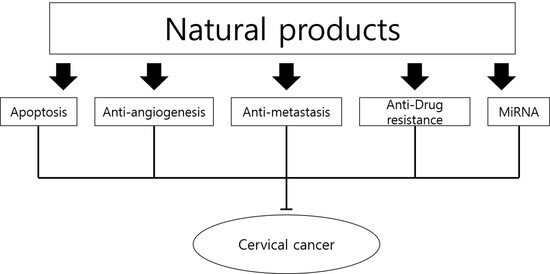
3. Results
3.1. Apoptosis
3.1.1. Compounds
3.1.2. Extracts
3.2. Anti-angiogenesis
3.3. Anti-Metastasis
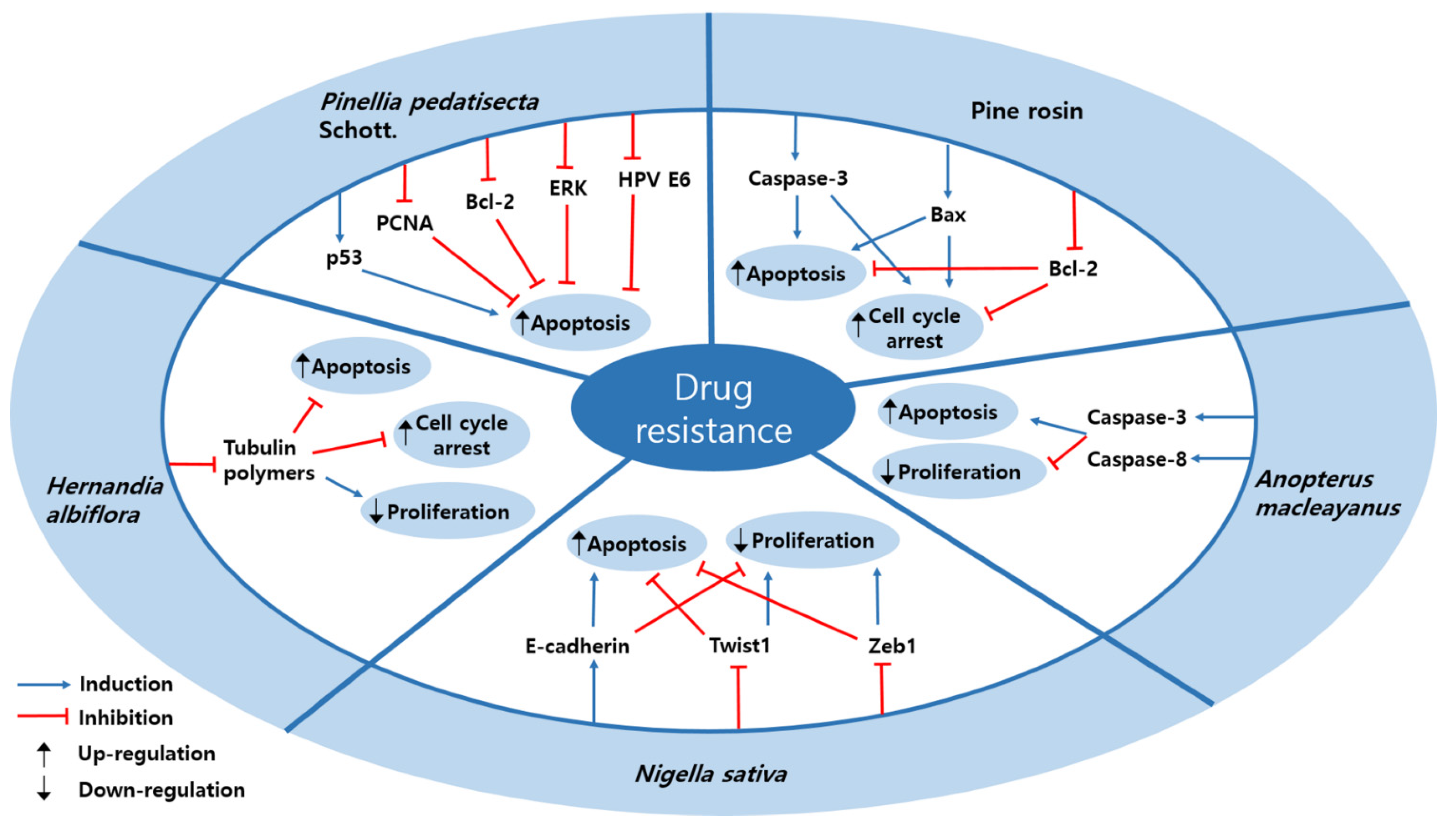
3.4. Drug Resistance
3.5. MicroRNA Regulation
4. Previous Studies
5. Limitation and Strength
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Urasa, M.; Darj, E. Knowledge of cervical cancer and screening practices of nurses at a regional hospital in Tanzania. Afr. Health Sci. 2011, 11, 48–57. [Google Scholar] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Byun, S.J.; Kim, Y.S.; Nam, J.-H. Disease courses in patients with residual tumor following concurrent chemoradiotherapy for locally advanced cervical cancer. Gynecol. Oncol. 2017, 144, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Hertlein, M.L.; Lenhard, M.; Kirschenhofer, A.; Kahlert, S.; Mayr, D.; Burges, A.; Friese, K. Cetuximab monotherapy in advanced cervical cancer: A retrospective study with five patients. Arch. Gynecol. Obstet. 2011, 283, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, J.; Hardy-Bessard, A.-C.; Deslandres, M.; Lavau-Denes, S.; Largillier, R.; Roemer-Becuwe, C.; Weber, B.; Guillemet, C.; Paraiso, D.; Pujade-Lauraine, E. Cetuximab, topotecan and cisplatin for the treatment of advanced cervical cancer: A phase II GINECO trial. Gynecol. Oncol. 2009, 113, 16–20. [Google Scholar] [CrossRef]
- Gaffney, D.K.; Winter, K.; Dicker, A.P.; Miller, B.; Eifel, P.J.; Ryu, J.; Avizonis, V.; Fromm, M.; Greven, K. A Phase II study of acute toxicity for Celebrex™ (celecoxib) and chemoradiation in patients with locally advanced cervical cancer: Primary endpoint analysis of RTOG. Int. J. Radiat. Oncol. 2007, 67, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Herrera, F.G.; Chan, P.; Doll, C.; Milosevic, M.; Oza, A.; Syed, A.; Pintilie, M.; Levin, W.; Manchul, L.; Fyles, A. A prospective phase I–II trial of the cyclooxygenase-2 inhibitor celecoxib in patients with carcinoma of the cervix with biomarker assessment of the tumor microenvironment. Int. J. Radiat. Oncol. 2007, 67, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Broutet, N.; Eckert, L.; Ullrich, A.; Bloem, P. Comprehensive Cervical Cancer Control: A Guide to Essential Practice; World Health Organization: Geneva, Switzerland, 2014; pp. 1–378. [Google Scholar]
- Lee, J.; Jeong, M.I.; Kim, H.-R.; Park, H.; Moon, W.-K.; Kim, B. Plant Extracts as Possible Agents for Sequela of Cancer Therapies and Cachexia. Antioxidants 2020, 9, 836. [Google Scholar] [CrossRef]
- Federico, C.; Sun, J.; Muz, B.; Alhallak, K.; Cosper, P.F.; Muhammad, N.; Jeske, A.; Hinger, A.; Markovina, S.; Grigsby, P.; et al. Localized Delivery of Cisplatin to Cervical Cancer Improves Its Therapeutic Efficacy and Minimizes Its Side-Effect Profile. Int. J. Radiat. Oncol. 2020. [Google Scholar] [CrossRef]
- Shen, D.-W.; Pouliot, L.M.; Hall, M.D.; Gottesman, M.M. Cisplatin Resistance: A Cellular Self-Defense Mechanism Resulting from Multiple Epigenetic and Genetic Changes. Pharmacol. Rev. 2012, 64, 706–721. [Google Scholar] [CrossRef]
- Sun, C.; Brown, A.J.; Jhingran, A.; Frumovitz, M.; Ramondetta, L.; Bodurka, D.C. Patient Preferences for Side Effects Associated With Cervical Cancer Treatment. Int. J. Gynecol. Cancer 2014, 24, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Momtazi-Borojeni, A.A.; Ghasemi, F.; Hesari, A.; Majeed, M.; Caraglia, M.; Sahebkar, A. Anti-Cancer and Radio-Sensitizing Effects of Curcumin in Nasopharyngeal Carcinoma. Curr. Pharm. Des. 2018, 24, 2121–2128. [Google Scholar] [CrossRef] [PubMed]
- Nasreen, S.; Safeer, S.; Dar, K.K.; Andleeb, S.; Ejaz, M.; Khan, M.A.; Ali, S. Etiology of hepatocellular carcinoma and treatment through medicinal plants: A comprehensive review. Orient. Pharm. Exp. Med. 2018, 18, 187–197. [Google Scholar] [CrossRef]
- Ezzat, S.M.; Shouman, S.A.; ElKhoely, A.; Attia, Y.M.; Elsesy, M.E.; El Senousy, A.S.; Choucry, M.A.; El Gayed, S.H.; El Sayed, A.A.; Sattar, E.A.; et al. Anticancer potentiality of lignan rich fraction of six Flaxseed cultivars. Sci. Rep. 2018, 8, 544. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.; Li, J.; Huang, C.; Huang, T.; Zhang, M.; Huang, L. miR-300/FA2H affects gastric cancer cell proliferation and apoptosis. Open Med. 2020, 15, 882–889. [Google Scholar] [CrossRef]
- Escuin, D.; López-Vilaró, L.; Bell, O.; Mora, J.; Moral, A.; Pérez, J.I.; Arqueros, C.; Cajal, T.R.Y.; Lerma, E.; Barnadas, A. MicroRNA-1291 Is Associated With Locoregional Metastases in Patients With Early-Stage Breast Cancer. Front. Genet. 2020, 11, 562114. [Google Scholar] [CrossRef]
- Lim, H.J.; Park, M.N.; Kim, C.; Kang, B.; Song, H.-S.; Lee, H.; Kim, S.-H.; Shim, B.S.; Kim, B. MiR-657/ATF2 Signaling Pathway Has a Critical Role in Spatholobus suberectus Dunn Extract-Induced Apoptosis in U266 and U937 Cells. Cancers 2019, 11, 150. [Google Scholar] [CrossRef]
- Kim, C.; Song, H.-S.; Park, H.; Kim, B. Activation of ER Stress-Dependent miR-216b Has a Critical Role in Salvia miltiorrhiza Ethanol-Extract-Induced Apoptosis in U266 and U937 Cells. Int. J. Mol. Sci. 2018, 19, 1240. [Google Scholar] [CrossRef]
- Phuah, N.H.; Azmi, M.N.; Awang, K.; Nagoor, N.H. Down-Regulation of MicroRNA-210 Confers Sensitivity towards 1’S-1’-Acetoxychavicol Acetate (ACA) in Cervical Cancer Cells by Targeting SMAD. Mol. Cells 2017, 40, 291–298. [Google Scholar] [CrossRef]
- Noh, S.; Choi, E.; Hwang, C.-H.; Jung, J.H.; Kim, S.-H.; Kim, B. Dietary Compounds for Targeting Prostate Cancer. Nutritients 2019, 11, 2401. [Google Scholar] [CrossRef]
- Lowe, S.W.; Lin, A.W. Apoptosis in cancer. Carcinog. 2000, 21, 485–495. [Google Scholar] [CrossRef]
- Li, F.-Y.; Wang, X.; Duan, W.-G.; Lin, G.-S. Synthesis and In Vitro Anticancer Activity of Novel Dehydroabietic Acid-Based Acylhydrazones. Molecules 2017, 22, 1087. [Google Scholar] [CrossRef] [PubMed]
- Vishnu, V.R.; Renjith, R.S.; Mukherjee, A.; Anil, S.R.; Sreekumar, J.; Jyothi, A.N.; Alummoottil, J.N. Comparative Study on the Chemical Structure and In Vitro Antiproliferative Activity of Anthocyanins in Purple Root Tubers and Leaves of Sweet Potato (Ipomoea batatas). J. Agric. Food Chem. 2019, 67, 2467–2475. [Google Scholar] [CrossRef] [PubMed]
- Piboonprai, K.; Khumkhrong, P.; Khongkow, M.; Yata, T.; Ruangrungsi, N.; Chansriniyom, C.; Iempridee, T. Anticancer activity of arborinine from Glycosmis parva leaf extract in human cervical cancer cells. Biochem. Biophys. Res. Commun. 2018, 500, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, Y.; Wu, Q.; Guan, Y.; Wu, X. Therapeutic effects of β-elemene via attenuation of the Wnt/β-catenin signaling pathway in cervical cancer cells. Mol. Med. Rep. 2018, 17, 4299–4306. [Google Scholar] [CrossRef] [PubMed]
- Rehana, D.; Mahendiran, D.; Kumar, R.S.; Rahiman, A.K. Evaluation of antioxidant and anticancer activity of copper oxide nanoparticles synthesized using medicinally important plant extracts. Biomed. Pharmacother. 2017, 89, 1067–1077. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Q.; Yang, L.; Xia, X.; Zhu, R.; Chen, S.; Wang, M.; Cheng, L.; Wu, X.; Wang, S. Curcumin-Loaded TPGS/F127/P123 Mixed Polymeric Micelles for Cervical Cancer Therapy: Formulation, Characterization, and In Vitro and In Vivo Evaluation. J. Biomed. Nanotechnol. 2017, 13, 1631–1646. [Google Scholar] [CrossRef]
- Moreira, T.F.; Sorbo, J.M.; Souza, F.D.O.; Fernandes, B.C.; Ocampos, F.M.M.; De Oliveira, D.M.S.; Arcaro, C.A.; Assis, R.P.; Barison, A.; Miguel, O.G.; et al. Emodin, Physcion, and Crude Extract of Rhamnus sphaerosperma var. pubescens Induce Mixed Cell Death, Increase in Oxidative Stress, DNA Damage, and Inhibition of AKT in Cervical and Oral Squamous Carcinoma Cell Lines. Oxidative Med. Cell. Longev. 2018, 2018, 1–18. [Google Scholar] [CrossRef]
- Yang, J.; Fa, J.; Li, B. Apoptosis induction of epifriedelinol on human cervical cancer cell line. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 80–86. [Google Scholar] [CrossRef][Green Version]
- Das, A.; Harshadha, K.; Dhinesh Kannan, S.; Hari Raj, K.; Jayaprakash, B. Evaluation of therapeutic potential of eugenol-a natural derivative of Syzygium aromaticum on cervical cancer. APJCP 2018, 19, 1977. [Google Scholar]
- Chen, X.; Song, L.; Hou, Y.; Li, F. Reactive oxygen species induced by icaritin promote DNA strand breaks and apoptosis in human cervical cancer cells. Oncol. Rep. 2018, 41, 765–778. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-Y.; Schelz, Z.; Tóth, B.; Vasas, A.; Ocsovszki, I.; Chang, F.-R.; Hohmann, J.; Zupkó, I.; Wang, H.-C. Investigation of natural phenanthrenes and the antiproliferative potential of juncusol in cervical cancer cell lines. Phytomedicine 2018, 58, 152770. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-L.; Zhang, Y.-S.; Zhang, F.; Zhang, Y.-Y.; Thakur, K.; Zhang, J.-G.; Wei, Z.-J. Methyl protodioscin from Polygonatum sibiricum inhibits cervical cancer through cell cycle arrest and apoptosis induction. Food Chem. Toxicol. 2019, 132, 110655. [Google Scholar] [CrossRef]
- Al-Otaibi, W.A.; Alkhatib, M.H.; Wali, A.N. Cytotoxicity and apoptosis enhancement in breast and cervical cancer cells upon coadministration of mitomycin C and essential oils in nanoemulsion formulations. Biomed. Pharmacother. 2018, 106, 946–955. [Google Scholar] [CrossRef]
- Latif, A.D.; Gonda, T.; Vágvölgyi, M.; Kúsz, N.; Kulmány, Á.; Ocsovszki, I.; Zomborszki, Z.P.; Zupkó, I.; Hunyadi, A. Synthesis and In Vitro Antitumor Activity of Naringenin Oxime and Oxime Ether Derivatives. Int. J. Mol. Sci. 2019, 20, 2184. [Google Scholar] [CrossRef]
- Souza, F.D.O.; Sorbo, J.M.; Regasini, L.O.; Rosa, J.C.; Czernys, É.D.S.; Valente, V.; Moreira, T.F.; Navegante, G.; Fernandes, B.C.; Soares, C.P. Nitensidine B affects proteins of the glycolytic pathway and induces apoptosis in cervical carcinoma cells immortalized by HPV. Phytomedicine 2018, 48, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, J.-X.; Wang, X.-Q.; Liu, X.-K.; Chen, X.-X.; Zhang, B.; He, Z.-D.; Liu, D.-Z.; Chen, L.-X.; Wang, L.-W.; et al. Notoginsenoside R7 suppresses cervical cancer via PI3K/PTEN/Akt/mTOR signaling. Oncotarget 2017, 8, 109487–109496. [Google Scholar] [CrossRef]
- Che, Y.; Li, J.; Li, Z.; Li, J.; Wang, S.; Yan, Y.; Zou, K.; Zou, L. Osthole enhances antitumor activity and irradiation sensitivity of cervical cancer cells by suppressing ATM/NF-κB signaling. Oncol. Rep. 2018, 40, 737–747. [Google Scholar] [CrossRef]
- Selvan, D.A.; Mahendiran, D.; Kumar, R.S.; Rahiman, A.K. Garlic, green tea and turmeric extracts-mediated green synthesis of silver nanoparticles: Phytochemical, antioxidant and in vitro cytotoxicity studies. J. Photochem. Photobiol. B Biol. 2018, 180, 243–252. [Google Scholar] [CrossRef]
- Xie, Z.; Wei, Y.; Xu, J.; Lei, J.; Yu, J. Alkaloids from Piper nigrum Synergistically Enhanced the Effect of Paclitaxel against Paclitaxel-Resistant Cervical Cancer Cells through the Downregulation of Mcl-1. J. Agric. Food Chem. 2019, 67, 5159–5168. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, D.; Liu, W.; Li, C.; Hao, L.; Zhang, G.; Deng, S.; Yang, R.; Qin, J.K.; Li, J.; et al. Cytotoxic Activity and Related Mechanisms of Prenylflavonoids Isolated from Mallotus conspurcatus Croizat. Chem. Biodivers. 2019, 16, e1800465. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-L.; Lee, C.-H.; Chen, C.-M.; Cheng, C.-W.; Chen, P.-N.; Ying, T.-H.; Hsieh, Y.-H. Protodioscin induces apoptosis through ROS-mediated endoplasmic reticulum stress via the JNK/p38 activation pathways in human cervical cancer cells. Cell. Physiol. Biochem. 2018, 46, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Mukherjee, S.; Vanmanen, J.; Banerjee, P.; Fata, J.E. Dietary Polyphenols, Resveratrol and Pterostilbene Exhibit Antitumor Activity on an HPV E6-Positive Cervical Cancer Model: An in vitro and in vivo Analysis. Front. Oncol. 2019, 9, 352. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Qu, D.; Fu, R.; Guo, M.; Qin, Y.; Guo, J.; Chen, Y. A Tf-modified tripterine-loaded coix seed oil microemulsion enhances anti-cervical cancer treatment. Int. J. Nanomed. 2018, 13, 7275–7287. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Khan, A.; Wei, C.; Cheng, J.; Chen, H.; Yang, L.; Ijaz, I.; Fu, J. Thymoquinone Inhibits the Migration and Invasive Characteristics of Cervical Cancer Cells SiHa and CaSki In Vitro by Targeting Epithelial to Mesenchymal Transition Associated Transcription Factors Twist1 and Zeb1. Molecules 2017, 22, 2105. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, M.; Tsering, J.; Li, H.; Li, S.; Li, Y.; Liu, Y.; Hu, X. An Integrated Study on the Antitumor Effect and Mechanism of Triphala Against Gynecological Cancers Based on Network Pharmacological Prediction and In Vitro Experimental Validation. Integr. Cancer Ther. 2018, 17, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Phuah, N.H.; Azmi, M.N.; Awang, K.; Nagoor, N.H. Suppression of microRNA-629 enhances sensitivity of cervical cancer cells to 1′S-1′-acetoxychavicol acetate via regulating RSU1. OncoTargets Ther. 2017, 10, 1695–1705. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Yang, W.; Fan, Y.; Dehaen, W.; Li, Y.; Li, H.-J.; Wang, W.; Zheng, Q.; Huai, Q.-Y. Design and synthesis of the novel oleanolic acid-cinnamic acid ester derivatives and glycyrrhetinic acid-cinnamic acid ester derivatives with cytotoxic properties. Bioorganic Chem. 2019, 88, 102951. [Google Scholar] [CrossRef]
- Hassan, A.H.; Choi, E.; Yoon, Y.M.; Lee, K.W.; Yoo, S.Y.; Cho, M.C.; Yang, J.S.; Kim, H.I.; Hong, J.Y.; Shin, J.-S.; et al. Natural products hybrids: 3,5,4’-Trimethoxystilbene-5,6,7-trimethoxyflavone chimeric analogs as potential cytotoxic agents against diverse human cancer cells. Eur. J. Med. Chem. 2018, 161, 559–580. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, Y.; Liu, Y.; Xu, W.; Yang, Y.; Hao, Y.; Tao, L. A natural product enhances apoptosis via mitochondria/caspase-mediated pathway in HeLa cells. J. Cell. Biochem. 2019, 120, 16811–16823. [Google Scholar] [CrossRef]
- Fiandalo, M.; Kyprianou, N. Caspase control: Protagonists of cancer cell apoptosis. Exp. Oncol. 2012, 34, 165–175. [Google Scholar] [PubMed]
- Kang, M.H.; Reynolds, C.P. Bcl-2 Inhibitors: Targeting Mitochondrial Apoptotic Pathways in Cancer Therapy. Clin. Cancer Res. 2009, 15, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- Huh, W.K.; Gomez-Navarro, J.; Arafat, W.O.; Xiang, J.; Mahasreshti, P.J.; Alvarez, R.D.; Barnes, M.N.; Curiel, D.T. Bax-Induced Apoptosis as a Novel Gene Therapy Approach for Carcinoma of the Cervix. Gynecol. Oncol. 2001, 83, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Swanepoel, B.; Venables, L.; Octavian-Tudorel, O.; Nitulescu, G.M.; Van De Venter, M. In Vitro Anti-proliferative Activity and Mechanism of Action of Anemone nemorosa. Int. J. Mol. Sci. 2019, 20, 1217. [Google Scholar] [CrossRef]
- Dwarka, D.; Thaver, V.; Naidu, M.; Koorbanally, N.A.; Baijnath, A.H. In vitro chemo-preventative activity of strelitzia nicolai aril extract containing bilirubin. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 147–156. [Google Scholar] [CrossRef][Green Version]
- Lee, K.M.; Lee, K.; Choi, Y.K.; Choi, Y.J.; Seo, H.S.; Ko, S.G. SH003-induced G1 phase cell cycle arrest induces apoptosis in HeLa cervical cancer cells. Mol. Med. Rep. 2017, 16, 8237–8244. [Google Scholar] [CrossRef]
- Dos Santos, K.M.; Gomes, I.N.F.; Silva-Oliveira, R.J.; Pinto, F.E.; Oliveira, B.G.; Chagas, R.C.R.; Romão, W.; Reis, R.M.; Ribeiro, R.I.M.D.A. Bauhinia variegata candida Fraction Induces Tumor Cell Death by Activation of Caspase-3, RIP, and TNF-R1 and Inhibits Cell Migration and Invasion In Vitro. BioMed Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Suh, S.-S.; Kim, S.-M.; Kim, J.E.; Hong, J.-M.; Lee, S.G.; Youn, U.J.; Han, S.J.; Kim, I.C.; Kim, S. Anticancer activities of ethanol extract from the Antarctic freshwater microalga, Botryidiopsidaceae sp. BMC Complement. Altern. Med. 2017, 17, 509. [Google Scholar] [CrossRef]
- Suh, S.-S.; Yang, E.J.; Lee, S.G.; Youn, U.J.; Han, S.J.; Kim, I.-C.; Kim, S. Bioactivities of ethanol extract from the Antarctic freshwater microalga, Chloromonas sp. Int. J. Med Sci. 2017, 14, 560–569. [Google Scholar] [CrossRef]
- Prasad, R.; Rana, N.K.; Koch, B. Dendrobium chrysanthum ethanolic extract induces apoptosis via p53 up-regulation in HeLa cells and inhibits tumor progression in mice. J. Complement. Integr. Med. 2017, 14, 14. [Google Scholar] [CrossRef]
- Ma, J.F.; Wei, P.F.; Guo, C.; Shi, Y.P.; Lv, Y.; Qiu, L.X.; Wen, L.P. The Ethyl Acetate Extract of Gynura formosana Kitam. Leaves Inhibited Cervical Cancer Cell Proliferation via Induction of Autophagy. BioMed Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Kuriakose, G.C.; M, D.L.; Bp, A.; Rs, H.K.; Th, A.K.; Ananthaswamy, K.; Chelliah, J. Extract of Penicillium sclerotiorum an endophytic fungus isolated from Cassia fistula L. induces cell cycle arrest leading to apoptosis through mitochondrial membrane depolarization in human cervical cancer cells. Biomed. Pharmacother. 2018, 105, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Dan, V.M.; Muralikrishnan, B.; Sanawar, R.; S, V.J.; Burkul, B.B.; Srinivas, K.P.; Lekshmi, A.; Pradeep, N.S.; Dastager, S.G.; Santhakumari, B.; et al. Streptomyces sp. metabolite(s) promotes Bax mediated intrinsic apoptosis and autophagy involving inhibition of mTOR pathway in cervical cancer cell lines. Sci. Rep. 2018, 8, 2810. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.T.; Zhu, Z.; Bai, Q.; Xiao, H.; Wakefield, M.R.; Fang, Y. Blueberry as a Potential Radiosensitizer for Treating Cervical Cancer. Pathol. Oncol. Res. 2019, 25, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Zhang, M.; Yao, S.; Zhang, M.; Peng, J.; Guiling with the Pinellia Pedatisecta (PE) Advisory Group; Xu, C.-J.; Ye, Y.; Gui, S. Immune modulation of a lipid-soluble extract of Pinellia pedatisecta Schott in the tumor microenvironment of an HPV + tumor-burdened mouse model. J. Ethnopharmacol. 2018, 225, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Khazaei, S.; Ramachandran, V.; Hamid, R.A.; Esa, N.M.; Etemad, A.; Moradipoor, S.; Patimah, I. Flower extract of Allium atroviolaceum triggered apoptosis, activated caspase-3 and down-regulated antiapoptotic Bcl-2 gene in HeLa cancer cell line. Biomed. Pharmacother. 2017, 89, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Esposito, T.; Sansone, F.; Franceschelli, S.; Del Gaudio, P.; Picerno, P.; Aquino, R.P.; Mencherini, T. Hazelnut (Corylus avellana L.) Shells Extract: Phenolic Composition, Antioxidant Effect and Cytotoxic Activity on Human Cancer Cell Lines. Int. J. Mol. Sci. 2017, 18, 392. [Google Scholar] [CrossRef]
- Mannarreddy, P.; Denis, M.; Munireddy, D.; Pandurangan, R.; Thangavelu, K.P.; Venkatesan, K. Cytotoxic effect of Cyperus rotundus rhizome extract on human cancer cell lines. Biomed. Pharmacother. 2017, 95, 1375–1387. [Google Scholar] [CrossRef]
- Vijayarathna, S.; Chen, Y.; Kanwar, J.R.; Sasidharan, S. Standardized Polyalthia longifolia leaf extract (PLME) inhibits cell proliferation and promotes apoptosis: The anti-cancer study with various microscopy methods. Biomed. Pharmacother. 2017, 91, 366–377. [Google Scholar] [CrossRef]
- Sul‘ain, M.D.; Fashihah Zakaria, M.F.J. Anti-Proliferative Effects of Methanol and Water Extracts of Pyrrosia piloselloides on the Hela Human Cervical Carcinoma Cell Line. APJCP 2019, 20, 185. [Google Scholar]
- Panicker, N.G.; Balhamar, S.O.M.S.; Akhlaq, S.; Qureshi, M.M.; Rizvi, T.S.; Al-Harrasi, A.; Hussain, J.; Mustafa, F. Identification and Characterization of the Caspase-Mediated Apoptotic Activity of Teucrium mascatense and an Isolated Compound in Human Cancer Cells. Molecules 2019, 24, 977. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.J.; Ashworth, A. Targeted therapy for cancer using PARP inhibitors. Curr. Opin. Pharmacol. 2008, 8, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Waxman, D.J. Combination of antiangiogenesis with chemotherapy for more effective cancer treatment. Mol. Cancer Ther. 2008, 7, 3670–3684. [Google Scholar] [CrossRef]
- Rajabi, M.; Mousa, S.A. The Role of Angiogenesis in Cancer Treatment. Biomediences 2017, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Tomao, S.; Tomao, F.; Rossi, L.; Zaccarelli, E.; Caruso, D.; Zoratto, F.; Panici, P.B.; Papa, A. Angiogenesis and antiangiogenic agents in cervical cancer. OncoTargets Ther. 2014, 7, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Shivamadhu, M.C.; Srinivas, B.K.; Jayarama, S.; Chandrashekaraiah, S.A. Anti-cancer and anti-angiogenic effects of partially purified lectin from Praecitrullus fistulosus fruit on in vitro and in vivo model. Biomed. Pharmacother. 2017, 96, 1299–1309. [Google Scholar] [CrossRef]
- Seifaddinipour, M.; Farghadani, R.; Namvar, F.; Bin Mohamad, J.; Kadir, H.A. Cytotoxic Effects and Anti-Angiogenesis Potential of Pistachio (Pistacia vera L.) Hulls against MCF-7 Human Breast Cancer Cells. Molecules 2018, 23, 110. [Google Scholar] [CrossRef]
- Foda, H.D.; Zucker, S. Matrix metalloproteinases in cancer invasion, metastasis and angiogenesis. Drug Discov. Today 2001, 6, 478–482. [Google Scholar] [CrossRef]
- Weber, G.F. Why does cancer therapy lack effective anti-metastasis drugs? Cancer Lett. 2013, 328, 207–211. [Google Scholar] [CrossRef]
- Chanvorachote, P.; Chamni, S.; Ninsontia, C.; Phiboonchaiyanan, P.P. Potential Anti-metastasis Natural Compounds for Lung Cancer. Anticancer Res. 2016, 36, 5707–5718. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, J.; Qin, X.; Huang, H.; Nie, C. Astragaloside IV inhibits the invasion and metastasis of SiHa cervical cancer cells via the TGF-β1-mediated PI3K and MAPK pathways. Oncol. Rep. 2019, 41, 2975–2986. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Q.; Lu, J.-L.; Liang, Y.-R.; Li, Q.-S. Suppressive Effects of EGCG on Cervical Cancer. Molecules 2018, 23, 2334. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-Y.; Lee, C.-H.; Chiou, H.-L.; Lin, C.-L.; Chen, P.-N.; Lin, M.-T.; Hsieh, Y.-H.; Chou, M.-C. Praeruptorin-b inhibits 12-o-tetradecanoylphorbol-13-acetate-induced cell invasion by targeting akt/nf-kappab via matrix metalloproteinase-2/-9 expression in human cervical cancer cells. Cell Physiol. Biochem. 2019, 52, 1255–1266. [Google Scholar] [PubMed]
- Lee, C.-Y.; Yang, S.-F.; Wang, P.-H.; Su, C.-W.; Hsu, H.-F.; Tsai, H.-T.; Hsiao, Y.-H. Antimetastatic effects of Terminalia catappa leaf extracts on cervical cancer through the inhibition of matrix metalloprotein-9 and MAPK pathway. Environ. Toxicol. 2019, 34, 60–66. [Google Scholar] [CrossRef]
- Onder, T.T.; Gupta, P.B.; Mani, S.A.; Yang, J.; Lander, E.S.; Weinberg, R.A. Loss of E-Cadherin Promotes Metastasis via Multiple Downstream Transcriptional Pathways. Cancer Res. 2008, 68, 3645–3654. [Google Scholar] [CrossRef]
- Vasan, N.; Baselga, J.; Hyman, D.M. A view on drug resistance in cancer. Nat. Cell Biol. 2019, 575, 299–309. [Google Scholar] [CrossRef]
- Gillet, J.-P.; Gottesman, M.M. Mechanisms of Multidrug Resistance in Cancer. In cryoEM; Springer Science and Business Media LLC: Berlin, Germany, 2010; Volume 596, pp. 47–76. [Google Scholar]
- Faustino, C.; Neto, Í.; Fonte, P.; Macedo, A. Cytotoxicity and Chemotherapeutic Potential of Natural Rosin Abietane Diterpenoids and their Synthetic Derivatives. Curr. Pharm. Des. 2018, 24, 4362–4375. [Google Scholar] [CrossRef]
- Levrier, C.; Rockstroh, A.; Gabrielli, B.; Kavallaris, M.; Lehman, M.; Davis, R.A.; Sadowski, M.C.; Nelson, C. Discovery of thalicthuberine as a novel antimitotic agent from nature that disrupts microtubule dynamics and induces apoptosis in prostate cancer cells. Cell Cycle 2018, 17, 652–668. [Google Scholar] [CrossRef]
- Levrier, C.; Sadowski, M.C.; Rockstroh, A.; Gabrielli, B.; Kavallaris, M.; Lehman, M.; Davis, R.A.; Nelson, C. 6α-Acetoxyanopterine: A Novel Structure Class of Mitotic Inhibitor Disrupting Microtubule Dynamics in Prostate Cancer Cells. Mol. Cancer Ther. 2016, 16, 3–15. [Google Scholar] [CrossRef]
- Ho, C.S.; Yap, S.H.; Phuah, N.H.; In, L.L.; Nagoor, N.H. MicroRNAs associated with tumour migration, invasion and angiogenic properties in A549 and SK-Lu1 human lung adenocarcinoma cells. Lung Cancer 2014, 83, 154–162. [Google Scholar] [CrossRef]
- Di Leva, G.; Croce, C.M. miRNA profiling of cancer. Curr. Opin. Genet. Dev. 2013, 23, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Huang, S.; Ding, J.; Zhao, Y.; Liang, L.; Liu, T.; Zhan, R.; He, X. Multiple microRNAs modulate p21Cip1/Waf1 expression by directly targeting its 3′ untranslated region. Oncogene 2010, 29, 2302–2308. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.B. MicroRNA (miRNA) in cancer. Cancer Cell Int. 2015, 15, 1–6. [Google Scholar] [CrossRef] [PubMed]
- de Moura, M.D.; de Se Silva, J.; de Oliveira, R.A.G.; de Diniz, M.; Barbosa-Filho, J.M. Natural products reported as potential inhibitors of uterine cervical neoplasia. Acta Farm. Bonaer. 2002, 21, 67–74. [Google Scholar]
- AL, A.O. Dietary supplements as a treatment for cervical cancer: A systematic review. Nutr. Hosp. 2013, 28, 1770–1780. [Google Scholar]
- Wang, S.; Zheng, C.; Peng, C.; Zhang, H.; Jiang, Y.-P.; Han, T.; Qin, L.-P. Plants and cervical cancer: An overview. Expert Opin. Investig. Drugs 2013, 22, 1133–1156. [Google Scholar] [CrossRef]
- Roy, M.; Mukherjee, A.; Sarkar, R.; Mukherjee, S.; Biswas, J. In Search of Natural Remediation for Cervical Cancer. Anti-Cancer Agents Med. Chem. 2014, 15, 57–65. [Google Scholar] [CrossRef]
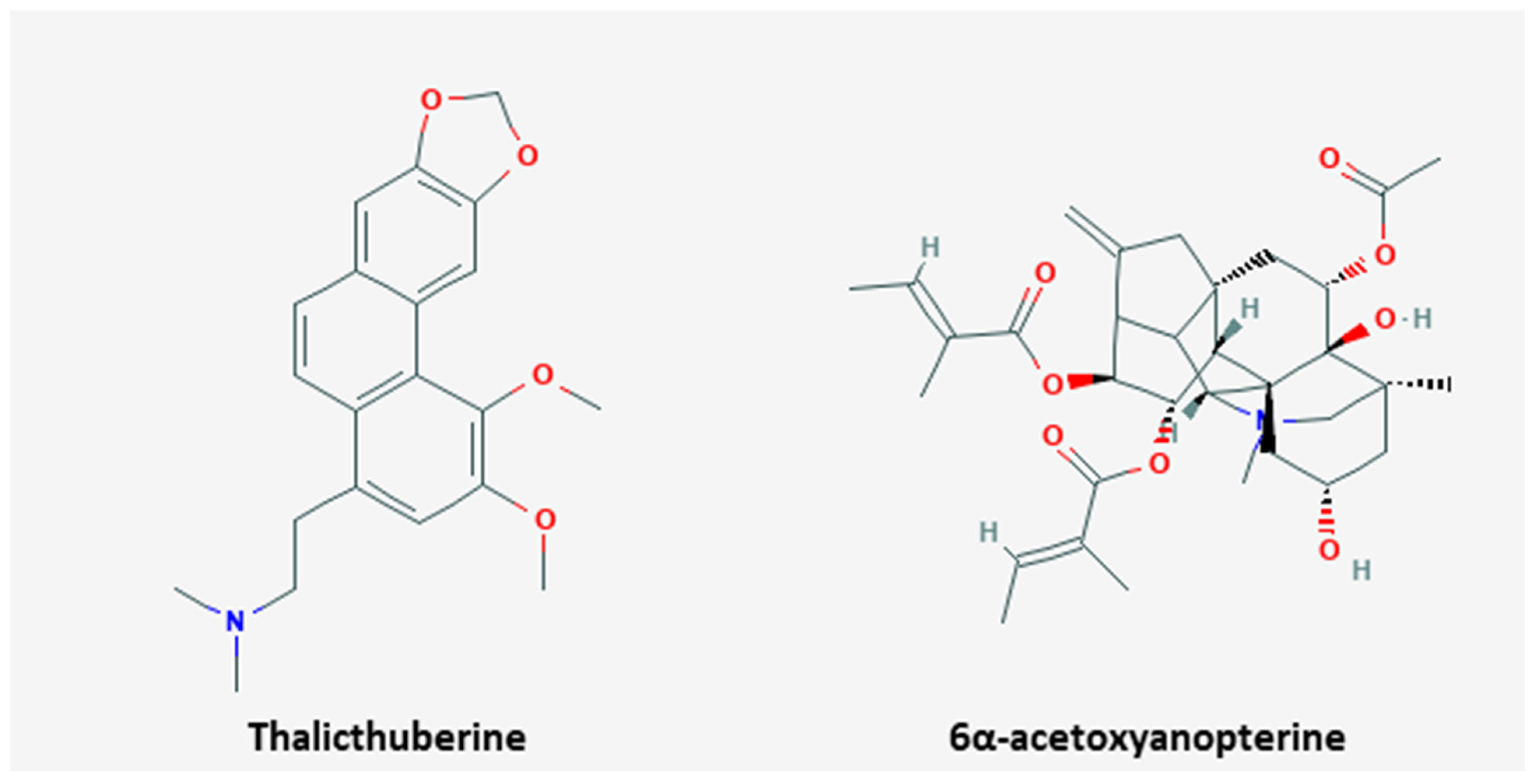
| Classification | Compound | Source | Cell Line/ Animal Model | Dose; Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
| Etc. | Acylhydrazone | HeLa | 2.21 µM; 48 h | Inhibition of cancer activity | [23] | ||
| Plant | Anthocyanins | Root tubers and leaves of Ipomoea batatas | HeLa | 100, 200 µg/mL; 48 h | Induction of apoptosis, cell cycle arrest | ↑CFP/YFP | [24] |
| Plant | Arborinine | Glycosmis parva | HeLa | 110 μg/mL; 24 h | Induction of apoptosis Inhibition of migration | ↑caspase-3, -7 ↓Bcl2-L1 | [25] |
| Plant | β-elemene | Curcuma zedoaria | SiHa | 30, 40, 50 μg/mL; 24, 48, 72 h | Inhibition of proliferation and migration Induction of cell cycle arrest and apoptosis | ↑p15, p53, Bax ↓cyclin D1, Bcl-2, MMP-2, -9, β-catenin, TCF7, c-Myc | [26] |
| Plant | Copper oxide nanoparticles | Azadirachta indica, Hibiscus rosa-sinensis, Murraya koenigii, Moringa oleifera, Tamarindus indica | HeLa | 2, 5, 10, 25, 50, 100 μg/mL; 48 h | Inhibition of oxidative stress Induction of apoptosis | [27] | |
| Plant | Curcumin | Curcuma longa | HeLa, C57BL/6, BALB/c | In vitro: 2 μg/mL; 48 h In vivo: 25 mg/kg | Induction of apoptosis and cell cycle arrest | ↑p53, cytochrome c, PARP, caspase-3, -7, -9 ↓Bcl-2, NF-κB | [28] |
| Plant | Emodin | Rhamnus sphaerosperma var. pubescens | SiHa, C33A | 46.3, 92.8, 185 μg/mL; 6, 12, 24 h | Induction of, apoptosis | ↓NO-, O2-, HOCl/OCl-, p-Akt | [29] |
| Plant | Epifriedelinol | Aster tataricus, Vitex peduncularis Wall. | HeLa | 50, 100, 250, 500, 1000 μg/mL; 72 h | Induction of apoptosis | ↑caspase-3, -8, -9 ↓Bcl-2, -xL, survivin | [30] |
| Plant | Eugenol | Syzygium aromaticum | HeLa, SiHa | 12.5, 25 µM; 24, 48 h | Induction of apoptosis | ↑Bax, PARP, caspase-3, ROS ↓Bcl-2, XIAP | [31] |
| Plant | Icaritin | Epimedium | HeLa, SiHa | HeLa: 12.5, 25 µM; 24, 48, 72 h SiHa: 17, 34 µM; 24, 48, 72 h | Induction of apoptosis Inhibition of proliferation | ↑ROS, Bax, c-caspase-3, -9 ↓Bcl-2, XIAP | [32] |
| Plant | Juncusol | Juncus inflexu | HeLa, SiHa, CaSki | 1, 3, 10, 30 µM; 24, 48, 72 h | Induction of apoptosis Inhibition of proliferation | ↑caspase-3, -8, -9 ↓EGFR, tubulin polymerization | [33] |
| Plant | Methyl protodioscin | Rhizoma of Polygonatum sibiricum | HeLa | 18.31, 40, 49 µM; 24 h | Induction of apoptosis and cell cycle arrest Inhibition of proliferation | ↑ ROS | [34] |
| Plant | Mitomycin C | Ginger, Frankincense | HeLa | 10 µg/mL; 24 h | Induction of apoptosis Inhibition of proliferation | [35] | |
| Etc. | Naringenin oxime | HeLa, SiHa | HeLa: 12, 24 µM; 24 h SiHa: 18, 36 µM; 24 h | Induction of apoptosis Inhibition of proliferation | ↑caspase-3 | [36] | |
| Naringenin oxime ether | |||||||
| Plant | Nitensidine B | Leaves of Pterogyne nitens Tul. | HPV16, SiHa | 30, 60, 120 µM; 6, 12, 24 h | Induction of apoptosis | ↑caspase-3, -7 ↓aldolase A, alpha-enolase, pyruvate kinase, glyceraldehyde 3-p-dehydrogenase | [37] |
| Plant | Notoginsenoside R7 | Panax notoginseng | HeLa, BALB/c | In vitro: 5, 10, 20, 40 μM; 24, 36, 48 h In vivo: 5, 10 mg/kg | Induction of apoptosis Inhibition of proliferation | ↑Bax, p-PTEN, Akt ↓Bcl-2, -xL, caspase-3, -9, raptor | [38] |
| Plant | Osthole | Cnidiummonnieri (L.) Cusson | HeLa, SiHa, C-33A, CaSki | 40, 80, 120, 160, 200, 240 µM; 24, 48 h | Induction of apoptosis Inhibition of proliferation | ↑Bax, c-caspase-3, -9 proteins, E-cadherin, H2AX ↓Bcl-2, MMP-2, -9, β-catenin, vimentin, N-cadherin, IKKα, p-IKKα, p65, p-p65, p50, NF-κB | [39] |
| Plant | Physcion | Rhamnus sphaerosperma var. pubescens | SiHa, C33A | 43.8, 87.5, 175 μg/mL; 6, 12, 24 h | Induction of apoptosis | ↓HOCl/OCl-, p-Akt | [29] |
| Plant | Phyto-synthesis of silver nanoparticles | Garlic, Green tea, Turmeric | HeLa | 2, 5, 10, 25, 50, 100 μg/mL; 48 h | Induction of apoptosis | ↓free radical | [40] |
| Plant | Piperine | Piper nigrum L. | HeLa, PTX | 50 µM; 6, 24, 72 h with paclitaxel | Induction of apoptosis | ↑Bax, Bcl-2, c-PARP, caspase-3 ↓p-Akt, Mcl-1 | [41] |
| Plant | Prenylflavonoids C1 | Mallotus conspurcatus | HeLa | 30 μM; 24 h | Induction of apoptosis | ↑EGFP, ROS, Bcl-2, cytochrome c, Apaf-1, caspase-3, -9 ↓c-Myc, hTERT | [42] |
| Prenylflavonoids C5 | 10 μM; 24 h | ||||||
| Plant | Protodioscin | Dioscoreae rhizome | HeLa, C33A | 4 μM; 24, 48 h | Induction of apoptosis and mitochondrial dysfunction | ↑JNK, p38, PERK, ATF4, Bax, caspase-3, -8, -9, PARP ↓Bcl-2 | [43] |
| Etc. | Pterostilbene | HPV E6, TC1, C57Bl/6 | In vitro: 30 µM; 48 h In vivo: 1 mM; 5 days | Induction of cell cycle arrest | ↑caspase-3 ↓PCNA, VEGF | [44] | |
| Resveratrol | |||||||
| Plant | Tf-CT-ME | Tripterygium wilfordii | HeLa | 0.5, 1, 2 µg/mL; 24 h | Induction of cell cycle arrest and apoptosis Inhibition of proliferation | ↑c-caspase-3 ↓Bcl-2/Bax | [45] |
| Seed | Thymoquinone | Nigella sativa | SiHa, CaSki | 10, 20, 40 μM; 24, 36, 48 h | Inhibition of migration and invasion | ↑Bax, E-cadherin ↓Bcl-2, Twist1, vimentin | [46] |
| Plant | Triphala | Terminalia chebula Retz., Terminalia bellerica (Gaertn) Roxb., Phyllanthus emblica Linn. | HeLa | 25-150 μg/mL; 48 h | Induction of apoptosis | ↑ERK, p53 ↓c-Myc, cyclin D1, p-Akt, p-NF-κB, p56, p-p44/42, MAPK | [47] |
| Plant | 1′S-1′-acetoxychavicol acetate | Alpinia conchigera | CaSki, SiHa | 20, 30 μM; 6, 12, 48 h | Induction of apoptosis | ↑RSU1, GAPDH | [48] |
| Plant | 2D of oleanolic acid and glycyrrhetinic acid | Ligustri Lucidi Fructus, Glycyrrhiza uralensis | HeLa | 2, 4 µM; 24, 48 h | Induction of apoptosis Inhibition of proliferation | ↑ROS | [49] |
| 3O of oleanolic acid and glycyrrhetinic acid | 1, 2 μM; 48 h | ||||||
| Etc. | 3,5,4′-trimethoxystilbene | HeLa | 10 µM; 48 h | Induction of apoptosis | [50] | ||
| 5,6,7-trimethoxyflavone | |||||||
| Plant | 5′-epi-SPA-6952A | Streptomyces diastatochromogenes | HeLa | 2, 4, 8, 16 µg/mL; 24 h | Induction of apoptosis and cell cycle arrest Inhibition of proliferation | ↑Bax/Bcl-2, cytochrome c, caspase-3, -9, c-PARP, p53 ↓MMP | [51] |
| Classification | Extract | Source | Cell Line/ Animal Model | Dose; Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
| Plant | Aqueous extract | Anemone nemorosa | HeLa | 20.33 ± 2.480 μg/mL; 24, 48 h | Induction of apoptosis Inhibition of proliferation | ↑PS translocation, c-caspase-3, -8, ROS (24 h) ↓MMP, ROS (48 h) | [55] |
| Plant | Aril extract | Strelitzia nicolai | HeLa | 250 μg/mL; 24, 48, 72 h | Inhibition of oxidative stress Induction of apoptosis | [56] | |
| Plant | Ethanol extract | Astragalus membranaceus, Angelica gigas, Trichosanthes kirilowii Maximowicz. | HeLa | 100, 200, 400 μg/mL; 24, 48 h | Induction of apoptosis and cell cycle arrest Inhibition of cell viability | ↑c-caspase-3, -8, PARP-1 ↓Bax, cyclin D, CDK2, CDK4, CDK6, p27 | [57] |
| Plant | Ethanol extract | Bauhinia variegate candida | HeLa | 15 μg/mL; 24 h | Inhibition of cell viability and migration Induction of apoptosis | ↑c-caspase-3, -8, RIP, TNF-R1 ↓MMP-2, MMP-9 | [58] |
| Plant | Ethanol extract | Botryidiopsidaceae species | HeLa | 6.25, 12.5, 25, 50 μg/mL; 24 h | Inhibition of oxidative stress and migration Induction of apoptosis | ↑p53, c-caspase-3 ↓Bcl-2 | [59] |
| Plant | Ethanol extract | Chloromonas species | HeLa | 12.5, 25 μg/mL; 24, 72 h | Inhibition of oxidative stress Induction of apoptosis | ↑c-caspase-3, p53 ↓Bcl-2 | [60] |
| Plant | Ethanol extract | Dendrobium chrysanthum | HeLa, Swiss albino mice | In vitro: 450 μg/mL; 24 h In vivo: 50, 100 mg/kg | Induction of apoptosis | ↑Bax, p53 ↓Bcl-2 | [61] |
| Plant | Ethanol extract | Rhamnus sphaerosperma var. pubescens | SiHa, C33A | 25, 50, 100 μg/mL; 6, 12, 24 h | Induction of apoptosis | ↓HOCl/OCl-, p-Akt | [29] |
| Plant | Ethyl acetate extract | Gynura formosana Kitam. | HeLa | 30 μg/mL; 72 h | Inhibition of proliferation | ↑ LC3-II/LC3-I, ↓P62/GAPDH, MCM7/GAPDH | [62] |
| Fungus | Ethyl acetate extract | Penicillium sclerotiorum | HeLa | 5, 25, 50 μg/mL; 24 h | Induction of apoptosis and cell cycle arrest | ↑Bax, p53, Apaf-1 ↓Bcl-2 | [63] |
| Plant | Ethyl acetate extract | Streptomyces species | SiHa | 20, 40, 60 μg/mL; 24 h | Induction of apoptosis and autophagy | ↑caspase-3, -9, Bax, LC3-Ⅱ ↓PARP, LC3-Ⅰ, Beclin1, p62 | [64] |
| Plant | Extract | Blueberry | SiHa | 50 mg/mL; 24 h with 4 Gy radiotherapy | Enhancement of radiotherapy | ↑p53 ↓cyclin D, E, p21, survivin | [65] |
| Plant | Lipid-soluble extract | Pinellia pedatisecta Schott. | HPV+TC-1, C57BL/6 | In vitro: 500 μg/mL; 72, 120 h In vivo: 10, 20 mg/kg | Induction of cell cycle arrest and apoptosis | ↑ β-catenin, c-Myc, cyclin D1, PPAR1 ↓Th2, Th17 | [66] |
| Plant | Methanol extract | Allium atroviolaceum | HeLa | 20, 40, 60, 80, 100 μg/mL; 24, 48, 72 h | Induction of cell cycle arrest | ↑caspase-3, -5, -9 ↓Bcl-2, CDK1, p53 | [67] |
| Plant | Methanol extract | Corylus avellane L. | HeLa | 250, 500 μg/mL; 24 h | Inhibition of oxidative stress Induction of apoptosis | ↑caspase-3 ↓PARP-1 | [68] |
| Plant | Methanol extract | Cyperus rotundus | HeLa | 25, 50, 100 μg/mL; 24, 48 h | Induction of apoptosis | [69] | |
| Plant | Methanol extract | Polyalthia longifolia | HeLa | 22 μg/mL; 6, 12, 24, 36 h | Induction of apoptosis | ↑Bax, BAD, caspase-3, p21, p53 ↓Bcl-2 | [70] |
| Plant | Methanol extract | Pyrrosia piloselloides | HeLa, | 16.25 μg/mL; 24, 48, 72 h | Inhibition of proliferation | [71] | |
| Plant | Methanol extract | Teucrium mascatense | HeLa | 25, 50, 125, 250 μg/mL; 72 h | Induction of apoptosis Inhibition of proliferation | ↑c-caspase-7, -8, -9, PARP | [72] |
| Classification | Compound/ Extract | Source | Cell Line/ Animal Model | Dose; Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
| Fruit | PfLP | Praecitrullus fistulosus | HeLa Swiss Albino mice | In vitro: 50 µg/mL; 24 h In vivo: 10 mg/kg | Induction of apoptosis Inhibition of angiogenesis | ↓ MMP-2, -9 | [77] |
| Plant | Purified flaxseed hydrolysate | Lignan | HeLa | 17.4 µg/mL; 48 h | Induction of apoptosis Inhibition of angiogenesis and metastasis | ↑ caspase-3 ↓ MMP-2, VEGF | [15] |
| Fungus | Ethyl acetate extract | Penicillium sclerotiorum | HeLa | 7.75 µg/mL; 24 h | Induction of cell cycle arrest and apoptosis Inhibition of angiogenesis | ↑ Bax, p53, Apaf-1 ↓ Bcl-2 | [63] |
| Plant | Ethyl acetate extract | Pistacia vera L. | CaSki | 81.17 ± 2.87 µg/mL; 72 h | Induction of apoptosis Inhibition of angiogenesis | ↓ TNF, Bcl-2, IAP, TRAF | [78] |
| Classification | Compound/ Extract | Source | Cell Line/ Animal Model | Dose; Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
| Plant | Astragaloside IV | Radix Astragali | SiHa | 200 µg/mL; 24 h | Inhibition of cell metastasis | ↑ E-cadherin ↓ p38, PI3K | [82] |
| Plant | Epigallocatechingallate | Green tea | HeLa | 50 µg/mL; 48 h | Inhibition of cell metastasis and proliferation Induction of apoptosis | ↓ MMP-2, -9, VEGF | [83] |
| Plant | Praeruptorin B | Peucedanum praeruptorum Dunn. | HeLa, SiHa | 40, 60 µM; 24 h | Inhibition of cell metastasis | ↓ NF-κB, MMP-2, -9 | [84] |
| Seed | Thymoquinone | Nigella sativa | CaSki, HeLa | 5 µM; 24 h | Induction of apoptosis, migration and invasion | ↑ E-cadherin ↓ Twist1, Zeb1 | [46] |
| Plant | Ethanol extract | Bauhinia variegata candida | HeLa | 25 µg/mL; 24 h | Inhibition of cell viability, migration and invasion | ↓ MMP-2, -9 | [58] |
| Plant | Ethanol extract | Terminalia catappa | HeLa, SiHa | 25, 50, 75 µg/mL; 24 h | Inhibition of cell metastasis | ↓ MMP-9, ERK1/2 | [85] |
| Classification | Compound/Extract | Source | Cell Line/ Animal Model | Dose; Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
| Plant | Rosin abietane diterpenoid | Pine rosin | HeLa | 1.08 ± 0.12 μM | Induction of apoptosis and cell cycle arrest | ↑ caspase-3, Bax ↓ Bcl-2 | [89] |
| Plant | Thalicthuberine | Hernandia albiflora | HeLa | 2.5 µM; 72 h | Induction of apoptosis and cell cycle arrest Inhibition of proliferation | ↓ tubulin polymers | [90] |
| Seed | Thymoquinone | Nigella sativa | SiHa, CaSki | 5, 10 µM; 24, 36 h | Induction of apoptosis Inhibition of proliferation | ↑ E-cadherin ↓ Twist1, Zeb1 | [46] |
| Plant | 6α-acetoxyanopterine | Anopterus macleayanus | HeLa | 3.2, 11.6 nM; 48 h | Induction of apoptosis Inhibition of proliferation | ↑ caspase-3, -8 | [91] |
| Plant | Lipid-soluble extract | Pinellia pedatisecta Schott. | CaSki, HeLa | 5000 µg/mL; 3 days | Induction of apoptosis | ↑ P53 ↓ ERK, Bcl-2, PCNA, HPV E6 | [66] |
| Classification | Compound/Extract | Source | Cell Line/ Animal Model | Dose/Duration | Efficacy | Mechanism | Reference |
|---|---|---|---|---|---|---|---|
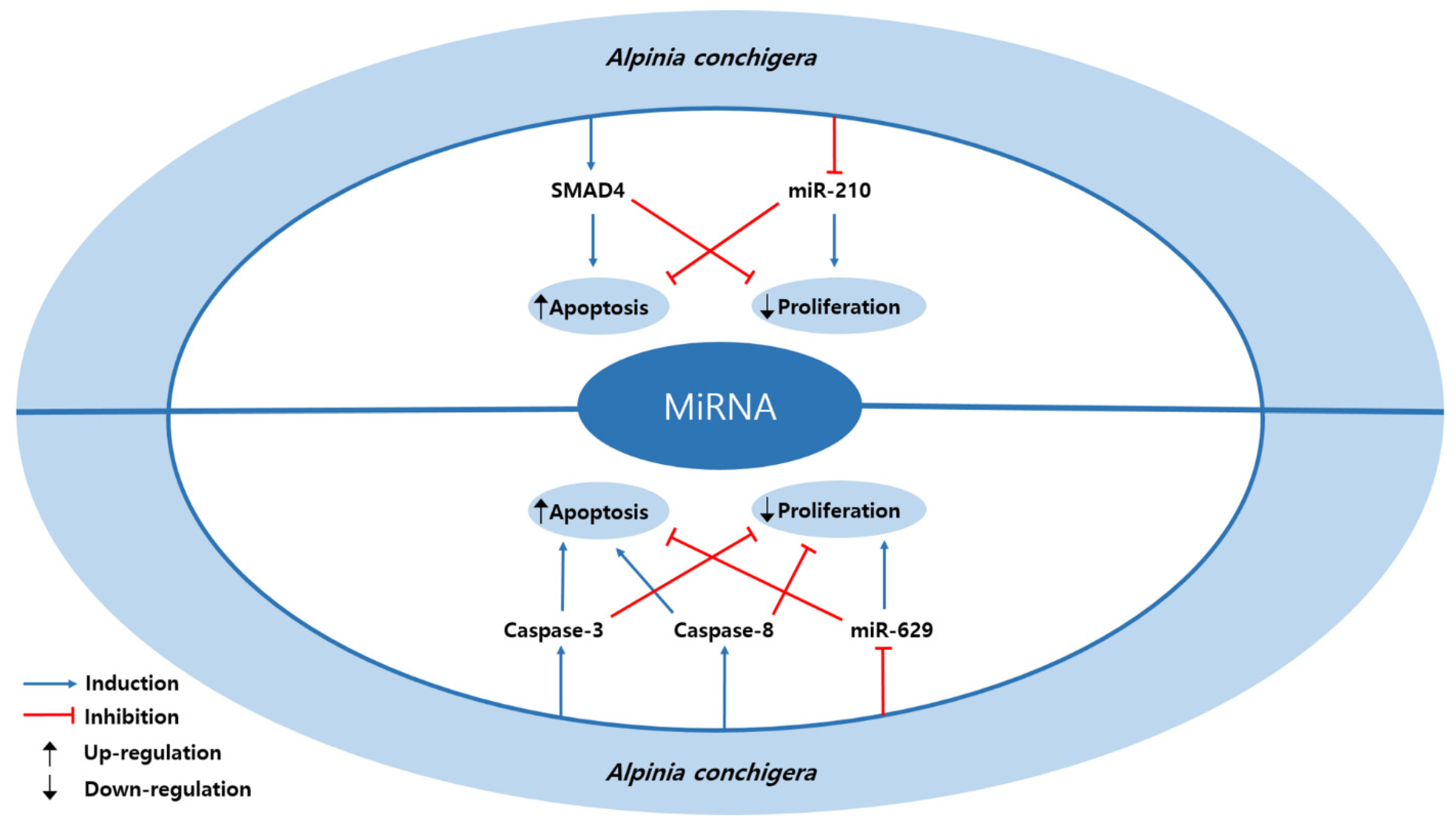
| Plant | 1′S-1′-acetoxychavicol acetate | Alpinia conchigera | SiHa, CaSki | 20, 30 µM; 6, 12 h | Induction of apoptosis Inhibition of proliferation | ↑ SMAD4 ↓ miR-210 | [20] |
| Plant | 1′S-1′-acetoxychavicol acetate | Alpinia conchigera | SiHa, CaSki | 20, 30 µM; 6, 12, 48 h | Induction of apoptosis Inhibition of proliferation | ↑ caspase-3, -8 ↓ miR-629 | [48] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.-H.; Kim, M.; Lee, S.; Jung, W.; Kim, B. Therapeutic Potential of Natural Products in Treatment of Cervical Cancer: A Review. Nutrients 2021, 13, 154. https://doi.org/10.3390/nu13010154
Park S-H, Kim M, Lee S, Jung W, Kim B. Therapeutic Potential of Natural Products in Treatment of Cervical Cancer: A Review. Nutrients. 2021; 13(1):154. https://doi.org/10.3390/nu13010154
Chicago/Turabian StylePark, Seung-Hyeok, Minsun Kim, Somi Lee, Woojin Jung, and Bonglee Kim. 2021. "Therapeutic Potential of Natural Products in Treatment of Cervical Cancer: A Review" Nutrients 13, no. 1: 154. https://doi.org/10.3390/nu13010154
APA StylePark, S.-H., Kim, M., Lee, S., Jung, W., & Kim, B. (2021). Therapeutic Potential of Natural Products in Treatment of Cervical Cancer: A Review. Nutrients, 13(1), 154. https://doi.org/10.3390/nu13010154