Chrononutrition during Pregnancy: A Review on Maternal Night-Time Eating
Abstract
1. Introduction
2. Assessment and Definition of Night Eating
3. Prevalence of Maternal Night Eating
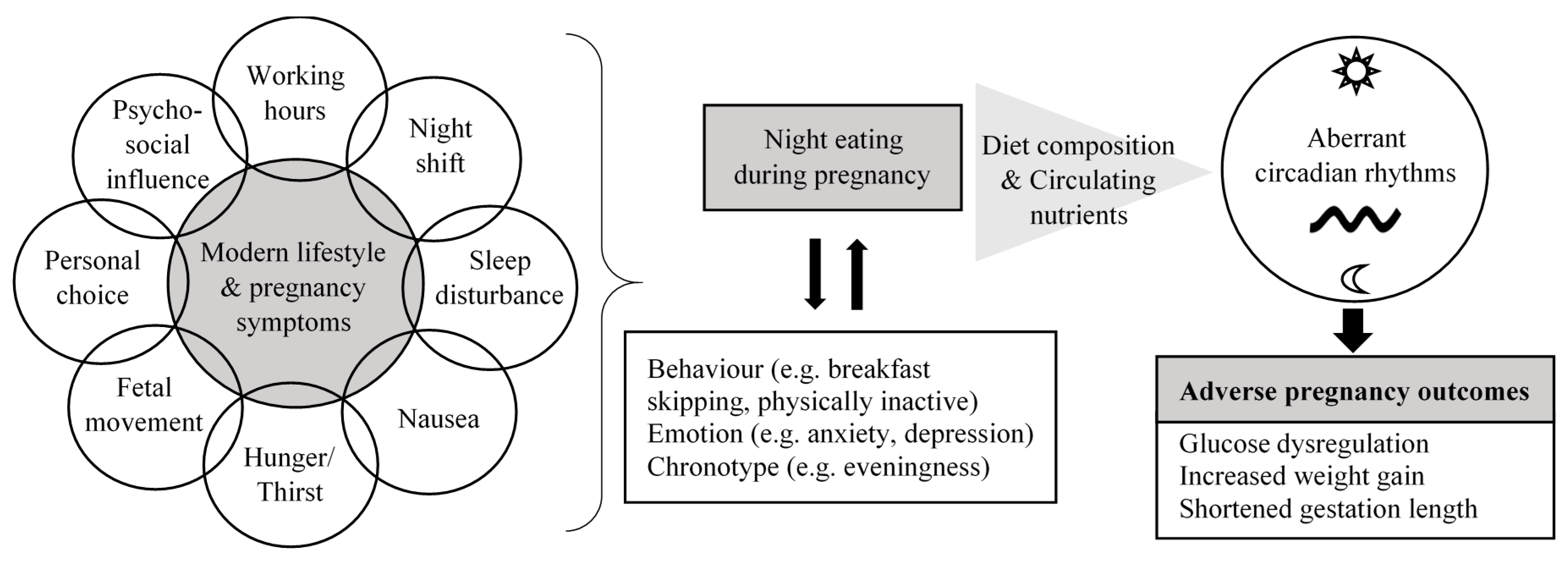
4. Potential Reasons for Maternal Night Eating
5. Characteristics Associated with Maternal Night Eating
6. Nutritional Aspects of Night Eating
7. Night Eating and Maternal Health Outcomes
8. Concluding Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, P.; Oster, H.; Korf, H.W.; Foster, R.G.; Erren, T.C. Food as a circadian time cue—Evidence from human studies. Nat. Rev. Endocrinol. 2020, 16, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D.; Ordovás, J.M.; Scheer, F.A.J.L.; Turek, F.W. Circadian Rhythms, Metabolism, and Chrononutrition in Rodents and Humans123. Adv. Nutr. 2016, 7, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Vargas, N.N.; Espitia-Bautista, E.; Buijs, R.M.; Escobar, C. Shift-work: Is time of eating determining metabolic health? Evidence from animal models. Proc. Nutr. Soc. 2018, 77, 199–215. [Google Scholar] [CrossRef]
- Moran-Ramos, S.; Baez-Ruiz, A.; Buijs, R.M.; Escobar, C. When to eat? The influence of circadian rhythms on metabolic health: Are animal studies providing the evidence? Nutr. Res. Rev. 2016, 29, 180–193. [Google Scholar] [CrossRef] [PubMed]
- McHill, A.W.; Phillips, A.J.; Czeisler, C.A.; Keating, L.; Yee, K.; Barger, L.K.; Garaulet, M.; Scheer, F.A.J.L.; Klerman, E.B. Later circadian timing of food intake is associated with increased body fat. Am. J. Clin. Nutr. 2017, 106, 1213–1219. [Google Scholar] [CrossRef]
- Srour, B.; Plancoulaine, S.; Andreeva, V.A.; Fassier, P.; Julia, C.; Galan, P.; Hercberg, S.; Deschasaux, M.; Latino-Martel, P.; Touvier, M. Circadian nutritional behaviours and cancer risk: New insights from the NutriNet-santé prospective cohort study: Disclaimers. Int. J. Cancer 2018, 143, 2369–2379. [Google Scholar] [CrossRef]
- Ribas-Latre, A.; Eckel-Mahan, K. Interdependence of nutrient metabolism and the circadian clock system: Importance for metabolic health. Mol. Metab. 2016, 5, 133–152. [Google Scholar] [CrossRef]
- Pot, G.K.; Almoosawi, S.; Stephen, A.M. Meal irregularity and cardiometabolic consequences: Results from observational and intervention studies. Proc. Nutr. Soc. 2016, 75, 475–486. [Google Scholar] [CrossRef]
- Asher, G.; Sassone-Corsi, P. Time for Food: The Intimate Interplay between Nutrition, Metabolism, and the Circadian Clock. Cell 2015, 161, 84–92. [Google Scholar] [CrossRef]
- Martin-Fairey, C.A.; Zhao, P.; Wan, L.; Roenneberg, T.; Fay, J.; Ma, X.; McCarthy, R.; Jungheim, E.S.; England, S.K.; Herzog, E.D. Pregnancy Induces an Earlier Chronotype in Both Mice and Women. J. Biol. Rhythm. 2019, 34, 323–331. [Google Scholar] [CrossRef]
- Wharfe, M.D.; Mark, P.J.; Wyrwoll, C.S.; Smith, J.T.; Yap, C.; Clarke, M.W.; Waddell, B.J. Pregnancy-induced adaptations of the central circadian clock and maternal glucocorticoids. J. Endocrinol. 2015, 228, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Wharfe, M.D.; Wyrwoll, C.S.; Waddell, B.J.; Mark, P.J. Pregnancy-induced changes in the circadian expression of hepatic clock genes: Implications for maternal glucose homeostasis. Am. J. Physiol. Metab. 2016, 311, E575–E586. [Google Scholar] [CrossRef] [PubMed]
- Gamble, K.L.; Resuehr, D.; Johnson, C.H. Shift Work and Circadian Dysregulation of Reproduction. Front. Endocrinol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Hammer, P.; Flachs, E.; Specht, I.O.; Pinborg, A.; Petersen, S.; Larsen, A.; Hougaard, K.; Hansen, J.; Hansen, Å.; Kolstad, H.; et al. Night work and hypertensive disorders of pregnancy: A national register-based cohort study. Scand. J. Work. Environ. Health 2018, 44, 403–413. [Google Scholar] [CrossRef]
- Fong, M.; Caterson, I.D.; Madigan, C.D. Are large dinners associated with excess weight, and does eating a smaller dinner achieve greater weight loss? A systematic review and meta-analysis. Br. J. Nutr. 2017, 118, 616–628. [Google Scholar] [CrossRef]
- Chandler-Laney, P.; Schneider, C.; Gower, B.A.; Granger, W.M.; Mancuso, M.S.; Biggio, J. Association of late-night carbohydrate intake with glucose tolerance among pregnant African American women. Matern. Child Nutr. 2015, 12, 688–698. [Google Scholar] [CrossRef]
- Loy, S.L.; Cheng, T.S.; Colega, M.T.; Cheung, Y.B.; Godfrey, K.M.; Gluckman, P.D.; Kwek, K.; Saw, S.M.; Chong, Y.-S.; Padmapriya, N.; et al. Predominantly night-time feeding and maternal glycaemic levels during pregnancy. Br. J. Nutr. 2016, 115, 1563–1570. [Google Scholar] [CrossRef]
- Gontijo, C.; Balieiro, L.C.T.; Teixeira, G.P.; Fahmy, W.M.; Crispim, C.A.; Maia, Y.; Araújo, G.C.; Tibiletti, B.L.C.; Pereira, T.G.; Makin, F.W.; et al. Higher energy intake at night effects daily energy distribution and contributes to excessive weight gain during pregnancy. Nutrition 2020, 74, 110756. [Google Scholar] [CrossRef]
- Englund-Ögge, L.; Birgisdóttir, B.E.; Sengpiel, V.; Brantsæter, A.L.; Haugen, M.; Myhre, R.; Meltzer, H.M.; Jacobsson, B. Meal frequency patterns and glycemic properties of maternal diet in relation to preterm delivery: Results from a large prospective cohort study. PLoS ONE 2017, 12, e0172896. [Google Scholar] [CrossRef]
- Allison, K.C.; Wrotniak, B.H.; Pare, E.; Sarwer, D.B. Psychosocial Characteristics and Gestational Weight Change among Overweight, African American Pregnant Women. Obstet. Gynecol. Int. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Damla Deniz, Ç.; Özler, S.; Sayın, F.K.; Eryılmaz, M.A. Associations between night eating syndrome and metabolic parameters in pregnant women. J. Turk. Soc. Obstet. Gynecol. 2019, 16, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Wołyńczyk-Gmaj, D.; Różańska-Walędziak, A.; Ziemka, S.; Ufnal, M.; Brzezicka, A.; Gmaj, B.; Januszko, P.; Fudalej, S.; Czajkowski, K.; Wojnar, M. Insomnia in Pregnancy is Associated with Depressive Symptoms and Eating at Night. J. Clin. Sleep Med. 2017, 13, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Loy, S.L.; Chan, J.K.Y.; Wee, P.H.; Colega, M.T.; Cheung, Y.B.; Godfrey, K.M.; Kwek, K.; Saw, S.M.; Chong, Y.-S.; Natarajan, P.; et al. Maternal Circadian Eating Time and Frequency Are Associated with Blood Glucose Concentrations during Pregnancy. J. Nutr. 2016, 147, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Wal, J.S.V. Night eating syndrome: A critical review of the literature. Clin. Psychol. Rev. 2012, 32, 49–59. [Google Scholar] [CrossRef]
- Allison, K.C.; Lundgren, J.D.; O’Reardon, J.P.; Martino, N.S.; Sarwer, D.B.; Wadden, T.A.; Crosby, R.D.; Engel, S.G.; Stunkard, A.J. The Night Eating Questionnaire (NEQ): Psychometric properties of a measure of severity of the Night Eating Syndrome. Eat. Behav. 2008, 9, 62–72. [Google Scholar] [CrossRef]
- Almoosawi, S.; Vingeliene, S.; Karagounis, L.G.; Pot, G.K. Chrono-nutrition: A review of current evidence from observational studies on global trends in time-of-day of energy intake and its association with obesity. Proc. Nutr. Soc. 2016, 75, 487–500. [Google Scholar] [CrossRef]
- Forbes, L.; Graham, J.; Berglund, C.; Bell, R. Dietary Change during Pregnancy and Women’s Reasons for Change. Nutrients 2018, 10, 1032. [Google Scholar] [CrossRef]
- Mc Morrow, L.; Ludbrook, A.; MacDiarmid, J.; Olajide, D. Perceived barriers towards healthy eating and their association with fruit and vegetable consumption. J. Public Heal. 2016, 39, 330–338. [Google Scholar] [CrossRef]
- Escoto, K.H.; Laska, M.N.; Larson, N.; Neumark-Sztainer, D.; Hannan, P.J. Work hours and perceived time barriers to healthful eating among young adults. Am. J. Heal. Behav. 2012, 36, 786–796. [Google Scholar] [CrossRef]
- Han, K.; Choi-Kwon, S.; Kim, K.S. Poor dietary behaviors among hospital nurses in Seoul, South Korea. Appl. Nurs. Res. 2016, 30, 38–44. [Google Scholar] [CrossRef]
- Sayle, A.E.; Wilcox, A.J.; Weinberg, C.R.; Baird, D.D. A prospective study of the onset of symptoms of pregnancy. J. Clin. Epidemiol. 2002, 55, 676–680. [Google Scholar] [CrossRef]
- Kroeger, E.; Carson, T.L.; Baskin, M.L.; Langaigne, A.; Schneider, C.; Bertrand, B.; Herbey, I.I.; Harper, L.M.; Biggio, J.; Chandler-Laney, P.C. Reasons for Late-Night Eating and Willingness to Change: A Qualitative Study in Pregnant Black Women. J. Nutr. Educ. Behav. 2019, 51, 598–607. [Google Scholar] [CrossRef]
- Kızılırmak, A.; Taşhan, S.T.; Kartal, B. Insomnia in Pregnancy and Factors Related to Insomnia. Sci. World J. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Alonso, A.M.; Trabalón-Pastor, M.; Chedraui, P.; Pérez-López, F.R. Factors related to insomnia and sleepiness in the late third trimester of pregnancy. Arch. Gynecol. Obstet. 2012, 286, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Galgani, J.E.; Moro, C.; Ravussin, E. Metabolic flexibility and insulin resistance. Am. J. Physiol. Metab. 2008, 295, E1009–E1017. [Google Scholar] [CrossRef] [PubMed]
- Gooley, J.J. Circadian regulation of lipid metabolism. Proc. Nutr. Soc. 2016, 75, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Gluck, M.E.; Venti, C.A.; Salbe, A.D.; Votruba, S.B.; Krakoff, J. Higher 24-h respiratory quotient and higher spontaneous physical activity in nighttime eaters. Obesity 2010, 19, 319–323. [Google Scholar] [CrossRef]
- Hibi, M.; Masumoto, A.; Naito, Y.; Kiuchi, K.; Yoshimoto, Y.; Matsumoto, M.; Katashima, M.; Oka, J.; Ikemoto, S. Nighttime snacking reduces whole body fat oxidation and increases LDL cholesterol in healthy young women. Am. J. Physiol. Integr. Comp. Physiol. 2013, 304, R94–R101. [Google Scholar] [CrossRef]
- Kelly, K.P.; McGuinness, O.P.; Buchowski, M.; Hughey, J.J.; Chen, H.; Powers, J.; Page, T.; Johnson, C.H. Eating breakfast and avoiding late-evening snacking sustains lipid oxidation. PLoS Biol. 2020, 18, e3000622. [Google Scholar] [CrossRef]
- Rynders, C.A.; Bergouignan, A.; Kealey, E.; Bessesen, D.H. Ability to adjust nocturnal fat oxidation in response to overfeeding predicts 5-year weight gain in adults. Obesity 2017, 25, 873–880. [Google Scholar] [CrossRef]
- Schmidt, S.L.; Kealey, E.H.; Horton, T.J.; VonKaenel, S.; Bessesen, D.H. The effects of short-term overfeeding on energy expenditure and nutrient oxidation in obesity-prone and obesity-resistant individuals. Int. J. Obes. 2012, 37, 1192–1197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scheer, F.A.J.L.; Morris, C.J.; Shea, S. The internal circadian clock increases hunger and appetite in the evening independent of food intake and other behaviors. Obesity 2013, 21, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Patrick, J.; Campbell, M.K.; Carmichael, L.; Natale, R.; Richardson, B. Patterns of gross fetal body movements over 24-hour observation intervals during the last 10weeks of pregnancy. Am. J. Obstet. Gynecol. 1982, 142, 363–371. [Google Scholar] [CrossRef]
- Stone, P.; Burgess, W.; McIntyre, J.P.R.; Gunn, A.J.; Lear, C.A.; Bennet, L.; Mitchell, E.A.; Thompson, J.M.D. An investigation of fetal behavioural states during maternal sleep in healthy late gestation pregnancy: An observational study. J. Physiol. 2017, 595, 7441–7450. [Google Scholar] [CrossRef]
- Bradford, B.; Maude, R. Fetal response to maternal hunger and satiation—Novel finding from a qualitative descriptive study of maternal perception of fetal movements. BMC Pregnancy Childbirth 2014, 14, 1–9. [Google Scholar] [CrossRef][Green Version]
- McDermott, M.S.; Oliver, M.; Svenson, A.; Simnadis, T.; Beck, E.J.; Coltman, T.; Iverson, D.; Caputi, P.; Sharma, R. The theory of planned behaviour and discrete food choices: A systematic review and meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Orloff, N.C.; Flammer, A.; Hartnett, J.; Liquorman, S.; Samelson, R.; Hormes, J.M. Food cravings in pregnancy: Preliminary evidence for a role in excess gestational weight gain. Appetite 2016, 105, 259–265. [Google Scholar] [CrossRef]
- Striegel-Moore, R.H.; Franko, D.L.; Thompson, D.; Affenito, S.; Kraemer, H.C.; Weissman, R. Night Eating: Prevalence and Demographic Correlates. Obesity 2006, 14, 139–147. [Google Scholar] [CrossRef]
- Potter, G.D.M.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Nutrition and the circadian system. Br. J. Nutr. 2016, 116, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Gómez-Abellán, P. Timing of food intake and obesity: A novel association. Physiol. Behav. 2014, 134, 44–50. [Google Scholar] [CrossRef]
- Loy, S.L.; Cheung, Y.B.; Cai, S.; Colega, M.T.; Godfrey, K.M.; Chong, Y.-S.; Shek, L.P.-C.; Tan, K.H.; Chong, M.F.-F.; Yap, F.; et al. Maternal night-time eating and sleep duration in relation to length of gestation and preterm birth. Clin. Nutr. 2020, 39, 1935–1942. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-W.; Shin, D. Association of Night Eating with Depression and Depressive Symptoms in Korean Women. Int. J. Environ. Res. Public Health 2019, 16, 4831. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.S.G.; Cañavate, R.; Hernández, C.M.; Cara-Salmerón, V.; Morante, J.J.H. The association among chronotype, timing of food intake and food preferences depends on body mass status. Eur. J. Clin. Nutr. 2016, 71, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, G.P.; Mota, M.C.; Crispim, C.A. Eveningness is associated with skipping breakfast and poor nutritional intake in Brazilian undergraduate students. Chronobiol. Int. 2017, 35, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Kronholm, E.; Tapanainen, H.; Kontto, J.; Männistö, S. Chronotype differences in timing of energy and macronutrient intakes: A population-based study in adults. Obesity 2017, 25, 608–615. [Google Scholar] [CrossRef]
- Kutsuma, A.; Nakajima, K.; Suwa, K. Potential Association between Breakfast Skipping and Concomitant Late-Night-Dinner Eating with Metabolic Syndrome and Proteinuria in the Japanese Population. Scientifica 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- De Castro, J.M. Circadian rhythms of the spontaneous meal pattern, macronutrient intake, and mood of humans. Physiol. Behav. 1987, 40, 437–446. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Ijedema, M.J.; Wijckmans-Duijsens, N.E. The role of macronutrient selection in determining patterns of food intake in obese and non-obese women. Eur. J. Clin. Nutr. 1996, 50, 580–591. [Google Scholar]
- Gallant, A.; Lundgren, J.D.; Drapeau, V. Nutritional Aspects of Late Eating and Night Eating. Curr. Obes. Rep. 2013, 3, 101–107. [Google Scholar] [CrossRef]
- Mazri, F.H.; Manaf, Z.A.; Shahar, S.; Ludin, A.F.M. The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. Int. J. Environ. Res. Public Heal. 2019, 17, 68. [Google Scholar] [CrossRef]
- Gontijo, C.; Cabral, B.B.M.; Balieiro, L.C.T.; Teixeira, G.P.; Fahmy, W.M.; Maia, Y.C.D.P.; Crispim, C.A. Time-related eating patterns and chronotype are associated with diet quality in pregnant women. Chronobiol. Int. 2018, 36, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Han, C.Y.; Colega, M.; Quah, E.P.L.; Chan, Y.H.; Godfrey, K.M.; Kwek, K.; Saw, S.-M.; Gluckman, P.D.; Chong, Y.-S.; Chong, M.F.F.; et al. A healthy eating index to measure diet quality in pregnant women in Singapore: A cross-sectional study. BMC Nutr. 2015, 1, 1. [Google Scholar] [CrossRef]
- Shiraishi, M.; Haruna, M.; Matsuzaki, M. Effects of skipping breakfast on dietary intake and circulating and urinary nutrients during pregnancy. Asia Pac. J. Clin. Nutr. 2019, 28, 99–105. [Google Scholar] [PubMed]
- Cheng, T.S.; Loy, S.L.; Cheung, Y.B.; Cai, S.; Colega, M.T.; Godfrey, K.M.; Chong, Y.-S.; Tan, K.H.; Shek, L.P.; Lee, Y.S.; et al. Plasma Vitamin D Deficiency Is Associated with Poor Sleep Quality and Night-Time Eating at Mid-Pregnancy in Singapore. Nutrients 2017, 9, 340. [Google Scholar] [CrossRef]
- Hernández, E.; Kim, M.; Kim, W.G.; Yoon, J. Nutritional aspects of night eating and its association with weight status among Korean adolescents. Nutr. Res. Pract. 2016, 10, 448–455. [Google Scholar] [CrossRef]
- Saad, A.; Man, C.D.; Nandy, D.K.; Levine, J.A.; Bharucha, A.E.; Rizza, R.A.; Basu, R.; Carter, R.E.; Cobelli, C.; Kudva, Y.C.; et al. Diurnal Pattern to Insulin Secretion and Insulin Action in Healthy Individuals. Diabetes 2012, 61, 2691–2700. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-J.; Lee, J.; Kim, J.-M.; Lee, H.A.; Kim, S.-H.; Kim, Y. A Study of Snack Consumption, Night-Eating Habits, and Nutrient Intake in Gestational Diabetes Mellitus. Clin. Nutr. Res. 2013, 2, 42–51. [Google Scholar] [CrossRef][Green Version]
- Loy, S.L.; Cheung, Y.B.; Colega, M.T.; Chia, A.-R.; Han, C.Y.; Godfrey, K.M.; Chong, Y.-S.; Shek, L.P.; Tan, K.H.; Lek, N.; et al. Associations of Circadian Eating Pattern and Diet Quality with Substantial Postpartum Weight Retention. Nutrients 2019, 11, 2686. [Google Scholar] [CrossRef]
- Goo, R.; Moore, J.; Greenberg, E.; Alazraki, N. Circadian variation in gastric emptying of meals in humans. Gastroenterology 1987, 93, 515–518. [Google Scholar] [CrossRef]
- Shaw, E.; Leung, G.K.W.; Jong, J.; Coates, A.M.; Blair, M.; Huggins, C.E.; Huggins, C.E.; Dorrian, J.; Banks, S.; Coates, A.M.; et al. The Impact of Time of Day on Energy Expenditure: Implications for Long-Term Energy Balance. Nutrients 2019, 11, 2383. [Google Scholar] [CrossRef]
- Paoli, A.; Tinsley, G.M.; Bianco, A.; Moro, T. The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting. Nutrients 2019, 11, 719. [Google Scholar] [CrossRef]
- Lawson, C.C.; Whelan, E.A.; Hibert, E.N.; Grajewski, B.; Spiegelman, D.; Rich-Edwards, J.W. Occupational factors and risk of preterm birth in nurses. Am. J. Obstet. Gynecol. 2008, 200, 51.e1–51.e8. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, F.; Vera, J.; Venegas, C.; Pino, F.; Lagunas, C. Circadian System and Melatonin Hormone: Risk Factors for Complications during Pregnancy. Obstet. Gynecol. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Reschke, L.; McCarthy, R.; Herzog, E.D.; Fay, J.C.; Jungheim, E.S.; England, S.K. Chronodisruption: An untimely cause of preterm birth? Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 52, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F. Meals and snacking, diet quality and energy balance. Physiol. Behav. 2014, 134, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Kinsey, A.W.; Ormsbee, M.J. The Health Impact of Nighttime Eating: Old and New Perspectives. Nutrients 2015, 7, 2648–2662. [Google Scholar] [CrossRef]
- Brand-Miller, J.C.; Buyken, A. Mapping postprandial responses sets the scene for targeted dietary advice. Nat. Med. 2020, 26, 828–830. [Google Scholar] [CrossRef]
- Berry, S.E.; Valdes, A.M.; Drew, D.A.; Asnicar, F.; Mazidi, M.; Wolf, J.; Capdevila, J.; Hadjigeorgiou, G.; Davies, R.; Al Khatib, H.; et al. Human postprandial responses to food and potential for precision nutrition. Nat. Med. 2020, 26, 964–973. [Google Scholar] [CrossRef]
- Siega-Riz, A.M.; Herrmann, T.S.; Savitz, D.A.; Thorp, J.M. Frequency of Eating During Pregnancy and Its Effect on Preterm Delivery. Am. J. Epidemiol. 2001, 153, 647–652. [Google Scholar] [CrossRef]
- Hennessy, M.D.; Volpe, S.L.; Sammel, M.D.; Gennaro, S. Skipping Meals and Less Walking among African Americans Diagnosed with Preterm Labor. J. Nurs. Sch. 2010, 42, 147–155. [Google Scholar] [CrossRef]
- Hernández-Díaz, S.; Boeke, C.E.; Romans, A.T.; Young, B.; Margulis, A.V.; McElrath, T.F.; Ecker, J.L.; Bateman, B.T. Triggers of spontaneous preterm delivery—Why today? Paediatr. Périnat. Epidemiol. 2014, 28, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, T.; Suzuki, K.; Kondo, N.; Yamagata, Z. Association of Maternal Lifestyles Including Smoking During Pregnancy with Childhood Obesity. Obesity 2007, 15, 3133–3139. [Google Scholar] [CrossRef]
- Haga, C.; Kondo, N.; Suzuki, K.; Sato, M.; Ando, D.; Yokomichi, H.; Tanaka, T.; Yamagata, Z. Developmental Trajectories of Body Mass Index Among Japanese Children and Impact of Maternal Factors during Pregnancy. PLoS ONE 2012, 7, e51896. [Google Scholar] [CrossRef] [PubMed]
- Varcoe, T.J.; Gatford, K.L.; Kennaway, D.J. Maternal circadian rhythms and the programming of adult health and disease. Am. J. Physiol. Integr. Comp. Physiol. 2018, 314, R231–R241. [Google Scholar] [CrossRef] [PubMed]
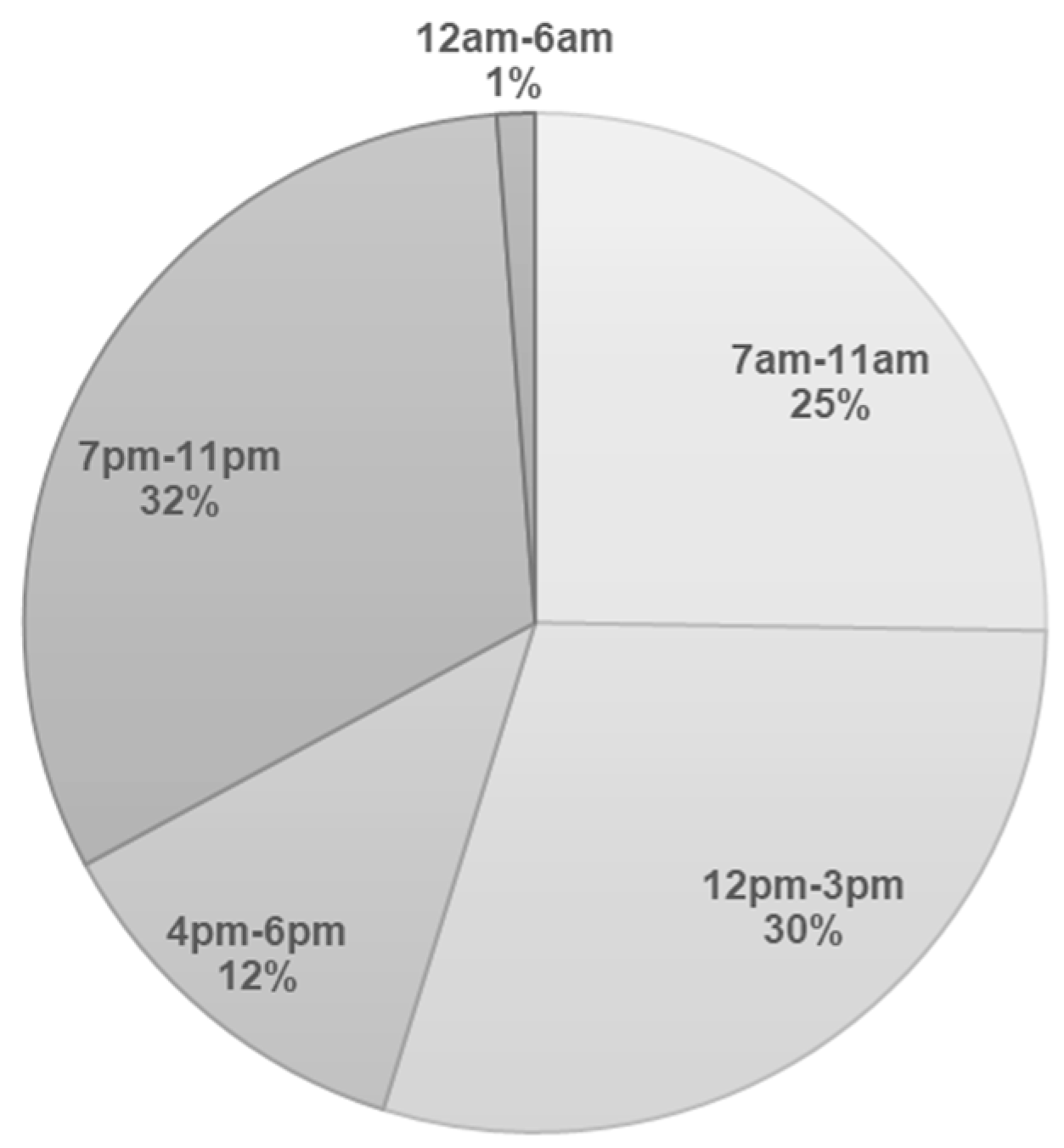
| Reference | Year of Publication | Country | Diet Assessment Time-Point | Sample Size | Gestation Weeks | Method of Dietary Assessment | Definition of Night-Time Eating |
|---|---|---|---|---|---|---|---|
| Chandler-Laney et al. [16] | 2015 | United States (African American) | Single | 40 | 32–34 | 2-day food diary | Higher TDEI during 8:00 p.m.–5:59 a.m. |
| Loy et al. [17] | 2016 | Singapore | Single | 985 | 26–28 | Single 24-h recall | More than 50% of TDEI during 7:00 p.m.–6:59 a.m. |
| Gontijio et al. [18] | 2020 | Brazil | Multiple | 100 | 4–12; 20–26; 30–37 | Three 24-h recalls per trimester | Above the median of TDEI during 7:00 p.m.–5:59 a.m. |
| Englund-Ögge et al. [19] | 2017 | Norway | Single | 65,487 | 17–22 | Questions asking frequency of eating events | Evening meal pattern as characterized by intake of supper and night meals |
| Allison et al. [20] | 2012 | United States (African American) | Single | 120 | 14–24 | Night Eating Questionnaire | Night Eating Syndrome |
| Damla Deniz et al. [21] | 2019 | Turkey | Single | 148 | 28–38 | Night Eating Questionnaire | Night Eating Syndrome |
| Wołyńczyk-Gmaj et al. [22] | 2017 | Poland | Single | 266 | 28–41 | Single question | Awake at night and eat |
| Component | Day Eaters (n = 834) | Night Eaters (n = 140) | |
|---|---|---|---|
| Mean ± SE | Mean ± SE | p-Value | |
| Total fruit | 2.4 ± 0.1 | 1.7 ± 0.3 | 0.045 |
| Whole fruit | 2.2 ± 0.1 | 1.6 ± 0.3 | 0.091 |
| Total vegetables | 2.4 ± 0.1 | 2.2 ± 0.2 | 0.535 |
| Dark green leafy and orange vegetables | 2.0 ± 0.1 | 1.9 ± 0.3 | 0.898 |
| Total rice and alternatives | 8.8 ± 0.1 | 8.5 ± 0.3 | 0.312 |
| Whole grains | 1.2 ± 0.1 | 0.5 ± 0.4 | 0.047 |
| Total protein foods | 7.7 ± 0.1 | 8.1 ± 0.4 | 0.325 |
| Dairy | 5.6 ± 0.2 | 5.1 ± 0.6 | 0.448 |
| Total fat | 6.4 ± 0.2 | 5.8 ± 0.5 | 0.306 |
| Saturated fat | 6.4 ± 0.1 | 5.7 ± 0.4 | 0.100 |
| Use of antenatal supplements | 1.7 ± 0.2 | 1.0 ± 0.5 | 0.160 |
| HEI-SG | 52.0 ± 0.6 | 46.9 ± 1.8 | 0.008 |
| Day Eaters (n = 730) | Night Eaters (n = 122) | ||
|---|---|---|---|
| Plasma Nutrients | Mean ± SE | Mean ± SE | p-Value |
| Vitamin B6, nmol/L | 78.0 ± 2.7 | 64.2 ± 8.0 | 0.107 |
| Vitamin B12, pmol/L | 299.7 ± 5.1 | 272.9 ± 15.2 | 0.126 |
| Folate, nmol/L | 17.3 ± 0.9 | 13.4 ± 2.7 | 0.010 |
| Vitamin D (25OHD), nmol/L | 78.4 ± 1.2 | 71.3 ± 3.7 | 0.025 |
| Ferritin, ng/mL | 26.1 ± 0.6 | 28.7 ± 1.7 | 0.079 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loy, S.L.; Loo, R.S.X.; Godfrey, K.M.; Chong, Y.-S.; Shek, L.P.-C.; Tan, K.H.; Chong, M.F.-F.; Chan, J.K.Y.; Yap, F. Chrononutrition during Pregnancy: A Review on Maternal Night-Time Eating. Nutrients 2020, 12, 2783. https://doi.org/10.3390/nu12092783
Loy SL, Loo RSX, Godfrey KM, Chong Y-S, Shek LP-C, Tan KH, Chong MF-F, Chan JKY, Yap F. Chrononutrition during Pregnancy: A Review on Maternal Night-Time Eating. Nutrients. 2020; 12(9):2783. https://doi.org/10.3390/nu12092783
Chicago/Turabian StyleLoy, See Ling, Rachael Si Xuan Loo, Keith M. Godfrey, Yap-Seng Chong, Lynette Pei-Chi Shek, Kok Hian Tan, Mary Foong-Fong Chong, Jerry Kok Yen Chan, and Fabian Yap. 2020. "Chrononutrition during Pregnancy: A Review on Maternal Night-Time Eating" Nutrients 12, no. 9: 2783. https://doi.org/10.3390/nu12092783
APA StyleLoy, S. L., Loo, R. S. X., Godfrey, K. M., Chong, Y.-S., Shek, L. P.-C., Tan, K. H., Chong, M. F.-F., Chan, J. K. Y., & Yap, F. (2020). Chrononutrition during Pregnancy: A Review on Maternal Night-Time Eating. Nutrients, 12(9), 2783. https://doi.org/10.3390/nu12092783





