Pre-Pregnancy Obesity vs. Other Risk Factors in Probability Models of Preeclampsia and Gestational Hypertension
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Methods
2.2. Definitions
2.3. Potential Predictors, Independent Variables
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jaacks, L.M.; Vandevijvere, S.; Pan, A.; McGowan, C.J.; Wallace, C.; Imamura, F.; Mozaffarian, D.; Swinburn, B.; Ezzati, M. The obesity transition: Stages of the global epidemic. Lancet Diabetes Endocrinol. 2019, 7, 231–240. [Google Scholar] [CrossRef]
- Hauspurg, A.; Countouris, M.E.; Jeyabalan, A.; Hubel, C.A.; Roberts, J.M.; Schwarz, E.B.; Catov, J.M. Risk of hypertension and abnormal biomarkers in the first year postpartum associated with hypertensive disorders of pregnancy among overweight and obese women. Pregnancy Hypertens. 2019, 15, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, A.; Patro-Małysza, J.; Kimber-Trojnar, Ż.; Marciniak, B.; Oleszczuk, J.; Leszczyńska-Gorzelak, B. Fetal programming of the metabolic syndrome. Taiwan J. Obstet. Gynecol. 2017, 56, 133–138. [Google Scholar] [CrossRef]
- Staud, F.; Karahoda, R. Trophoblast: The central unit of fetal growth, protection and programming. Int. J. Biochem. Cell Biol. 2018, 105, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Fajersztajn, L.; Veras, M.M. Hypoxia: From Placental Development to Fetal Programming. Birth Defects Res. 2017, 109, 1377–1385. [Google Scholar] [CrossRef]
- Hanafi, M.Y.; Saleh, M.M.; Saad, M.I.; Abdelkhalek, T.M.; Kamel, M.A. Transgenerational effects of obesity and malnourishment on diabetes risk in F2 generation. Mol. Cell. Biochem. 2016, 412, 269–280. [Google Scholar] [CrossRef]
- Lewandowska, M.; Więckowska, B.; Sajdak, S. Pre-Pregnancy Obesity, Excessive Gestational Weight Gain, and the Risk of Pregnancy-Induced Hypertension and Gestational Diabetes Mellitus. J. Clin. Med. 2020, 9, 1980. [Google Scholar] [CrossRef]
- Bartsch, E.; Medcalf, K.E.; Park, A.L.; Ray, J.G. High Risk of Pre-eclampsia Identification Group Clinical risk factors for pre-eclampsia determined in early pregnancy: Systematic review and meta-analysis of large cohort studies. BMJ 2016, 353, i1753. [Google Scholar] [CrossRef]
- Phipps, E.A.; Thadhani, R.; Benzing, T.; Karumanchi, S.A. Author Correction: Pre-eclampsia: Pathogenesis, novel diagnostics and therapies. Nat. Rev. Nephrol. 2019, 15, 386. [Google Scholar] [CrossRef]
- Olson, K.N.; Redman, L.M.; Sones, J.L. Obesity “complements” preeclampsia. Physiol. Genom. 2019, 51, 73–76. [Google Scholar] [CrossRef]
- Young, O.M.; Twedt, R.; Catov, J.M. Pre-pregnancy maternal obesity and the risk of preterm preeclampsia in the American primigravida. Obesity 2016, 24, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Patro Golab, B.; Santos, S.; Voerman, E.; Lawlor, D.A.; Jaddoe, V.W.V.; Gaillard, R. MOCO Study Group Authors Influence of maternal obesity on the association between common pregnancy complications and risk of childhood obesity: An individual participant data meta-analysis. Lancet Child Adolesc. Health 2018, 2, 812–821. [Google Scholar] [CrossRef]
- Poon, L.C.; Shennan, A.; Hyett, J.A.; Kapur, A.; Hadar, E.; Divakar, H.; McAuliffe, F.; da Silva Costa, F.; von Dadelszen, P.; McIntyre, H.D.; et al. The International Federation of Gynecology and Obstetrics (FIGO) initiative on pre-eclampsia: A pragmatic guide for first-trimester screening and prevention. Int. J. Gynaecol. Obstet. 2019, 145, 1–33. [Google Scholar] [CrossRef]
- Shen, M.; Smith, G.N.; Rodger, M.; White, R.R.; Walker, M.C.; Wen, S.W. Comparison of risk factors and outcomes of gestational hypertension and pre-eclampsia. PLoS ONE 2017, 12, e0175914. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics Gestational Hypertension and Preeclampsia: ACOG Practice Bulletin, Number 222. Obstet. Gynecol. 2020, 135, e237–e260. [CrossRef]
- Sufriyana, H.; Wu, Y.-W.; Su, E.C.-Y. Prediction of Preeclampsia and Intrauterine Growth Restriction: Development of Machine Learning Models on a Prospective Cohort. JMIR Med. Inf. 2020, 8, e15411. [Google Scholar] [CrossRef]
- Hromadnikova, I.; Kotlabova, K.; Hympanova, L.; Krofta, L. Gestational hypertension, preeclampsia and intrauterine growth restriction induce dysregulation of cardiovascular and cerebrovascular disease associated microRNAs in maternal whole peripheral blood. Thromb. Res. 2016, 137, 126–140. [Google Scholar] [CrossRef]
- Aryan, L.; Medzikovic, L.; Umar, S.; Eghbali, M. Pregnancy-associated cardiac dysfunction and the regulatory role of microRNAs. Biol. Sex Differ. 2020, 11, 14. [Google Scholar] [CrossRef]
- Elharram, M.; Dayan, N.; Kaur, A.; Landry, T.; Pilote, L. Long-Term Cognitive Impairment After Preeclampsia: A Systematic Review and Meta-analysis. Obstet. Gynecol. 2018, 132, 355–364. [Google Scholar] [CrossRef]
- Sandström, A.; Snowden, J.M.; Höijer, J.; Bottai, M.; Wikström, A.-K. Clinical risk assessment in early pregnancy for preeclampsia in nulliparous women: A population based cohort study. PLoS ONE 2019, 14, e0225716. [Google Scholar] [CrossRef]
- Robillard, P.-Y.; Dekker, G.; Scioscia, M.; Bonsante, F.; Iacobelli, S.; Boukerrou, M.; Hulsey, T.C. Increased BMI has a linear association with late-onset preeclampsia: A population-based study. PLoS ONE 2019, 14, e0223888. [Google Scholar] [CrossRef] [PubMed]
- Omani-Samani, R.; Ranjbaran, M.; Amini, P.; Esmailzadeh, A.; Sepidarkish, M.; Almasi-Hashiani, A. Adverse maternal and neonatal outcomes in women with preeclampsia in Iran. J. Matern. Fetal Neonatal Med. 2019, 32, 212–216. [Google Scholar] [CrossRef] [PubMed]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Pencina, M.J.; D’Agostino, R.B.; D’Agostino, R.B.; Vasan, R.S. Evaluating the added predictive ability of a new marker: From area under the ROC curve to reclassification and beyond. Stat. Med. 2008, 27, 157–172; discussion 207–212. [Google Scholar] [CrossRef]
- Cook, N.R. Quantifying the added value of new biomarkers: How and how not. Diagn. Progn. Res. 2018, 2, 14. [Google Scholar] [CrossRef]
- Lewandowska, M.; Sajdak, S.; Lubiński, J. Serum Selenium Level in Early Healthy Pregnancy as a Risk Marker of Pregnancy Induced Hypertension. Nutrients 2019, 11, 1028. [Google Scholar] [CrossRef]
- Tarca, A.L.; Romero, R.; Benshalom-Tirosh, N.; Than, N.G.; Gudicha, D.W.; Done, B.; Pacora, P.; Chaiworapongsa, T.; Panaitescu, B.; Tirosh, D.; et al. The prediction of early preeclampsia: Results from a longitudinal proteomics study. PLoS ONE 2019, 14, e0217273. [Google Scholar] [CrossRef]
- Van Esch, J.J.A.; van Heijst, A.F.; de Haan, A.F.J.; van der Heijden, O.W.H. Early-onset preeclampsia is associated with perinatal mortality and severe neonatal morbidity. J. Matern. Fetal Neonatal Med. 2017, 30, 2789–2794. [Google Scholar] [CrossRef]
- Erez, O.; Romero, R.; Maymon, E.; Chaemsaithong, P.; Done, B.; Pacora, P.; Panaitescu, B.; Chaiworapongsa, T.; Hassan, S.S.; Tarca, A.L. The prediction of late-onset preeclampsia: Results from a longitudinal proteomics study. PLoS ONE 2017, 12, e0181468. [Google Scholar] [CrossRef]
- Ługowska, K.; Kolanowski, W. The Nutritional Behaviour of Pregnant Women in Poland. Int. J. Environ. Res. Public Health 2019, 16, 4357. [Google Scholar] [CrossRef]
- LifeCycle Project-Maternal Obesity and Childhood Outcomes Study Group; Voerman, E.; Santos, S.; Inskip, H.; Amiano, P.; Barros, H.; Charles, M.-A.; Chatzi, L.; Chrousos, G.P.; Corpeleijn, E.; et al. Association of Gestational Weight Gain With Adverse Maternal and Infant Outcomes. JAMA 2019, 321, 1702–1715. [Google Scholar] [CrossRef] [PubMed]
- Hillesund, E.R.; Seland, S.; Bere, E.; Sagedal, L.R.; Torstveit, M.K.; Lohne-Seiler, H.; Vistad, I.; Øverby, N.C. Preeclampsia and gestational weight gain in the Norwegian Fit for Delivery trial. BMC Res. Notes 2018, 11, 282. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.Y.; Roberts, V.H.J.; Frias, A.E.; Brace, R.A. Effects of maternal western-style diet on amniotic fluid volume and amnion VEGF profiles in a nonhuman primate model. Physiol. Rep. 2018, 6, e13894. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska, M.; Więckowska, B.; Sajdak, S.; Lubiński, J. First Trimester Microelements and their Relationships with Pregnancy Outcomes and Complications. Nutrients 2020, 12, 1108. [Google Scholar] [CrossRef] [PubMed]
- Izaola, O.; de Luis, D.; Sajoux, I.; Domingo, J.C.; Vidal, M. Inflammation and obesity (lipoinflammation). Nutr. Hosp. 2015, 31, 2352–2358. [Google Scholar] [CrossRef]
- Saltiel, A.R.; Olefsky, J.M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Investig. 2017, 127, 1–4. [Google Scholar] [CrossRef]
- Staff, A.C. The two-stage placental model of preeclampsia: An update. J. Reprod. Immunol. 2019, 134–135, 1–10. [Google Scholar] [CrossRef]
- Rozas-Villanueva, M.F.; Casanello, P.; Retamal, M.A. Role of ROS/RNS in Preeclampsia: Are Connexins the Missing Piece? Int. J. Mol. Sci. 2020, 21, 4698. [Google Scholar] [CrossRef]
- Ahmad, I.M.; Zimmerman, M.C.; Moore, T.A. Oxidative stress in early pregnancy and the risk of preeclampsia. Pregnancy Hypertens. 2019, 18, 99–102. [Google Scholar] [CrossRef]
- Haram, K.; Mortensen, J.H.; Myking, O.; Magann, E.F.; Morrison, J.C. The Role of Oxidative Stress, Adhesion Molecules and Antioxidants in Preeclampsia. Curr. Hypertens. Rev. 2019, 15, 105–112. [Google Scholar] [CrossRef]
- Wright, D.; Tan, M.Y.; O’Gorman, N.; Poon, L.C.; Syngelaki, A.; Wright, A.; Nicolaides, K.H. Predictive performance of the competing risk model in screening for preeclampsia. Am. J. Obstet. Gynecol. 2019, 220, 199. [Google Scholar] [CrossRef] [PubMed]
- Antwi, E.; Amoakoh-Coleman, M.; Vieira, D.L.; Madhavaram, S.; Koram, K.A.; Grobbee, D.E.; Agyepong, I.A.; Klipstein-Grobusch, K. Systematic review of prediction models for gestational hypertension and preeclampsia. PLoS ONE 2020, 15, e0230955. [Google Scholar] [CrossRef] [PubMed]
- Antwi, E.; Klipstein-Grobusch, K.; Browne, J.L.; Schielen, P.C.; Koram, K.A.; Agyepong, I.A.; Grobbee, D.E. Improved prediction of gestational hypertension by inclusion of placental growth factor and pregnancy associated plasma protein-a in a sample of Ghanaian women. Reprod. Health 2018, 15, 56. [Google Scholar] [CrossRef]
- Founds, S.A.; Tsigas, E.; Ren, D.; Barmada, M.M. Associating Symptom Phenotype and Genotype in Preeclampsia. Biol. Res. Nurs. 2018, 20, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ananth, C.V.; Jablonski, K.; Myatt, L.; Roberts, J.M.; Tita, A.T.N.; Leveno, K.J.; Reddy, U.M.; Varner, M.W.; Thorp, J.M.; Mercer, B.M.; et al. Risk of Ischemic Placental Disease in Relation to Family History of Preeclampsia. Am. J. Perinatol. 2019, 36, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Serrano, N.C.; Quintero-Lesmes, D.C.; Dudbridge, F.; Leon, L.J.; Hingorani, A.D.; Williams, D.J.; Casas, J.P. Family history of pre-eclampsia and cardiovascular disease as risk factors for pre-eclampsia: The GenPE case-control study. Hypertens. Pregnancy 2020, 39, 56–63. [Google Scholar] [CrossRef]
- Umesawa, M.; Kobashi, G. Epidemiology of hypertensive disorders in pregnancy: Prevalence, risk factors, predictors and prognosis. Hypertens. Res. 2017, 40, 213–220. [Google Scholar] [CrossRef]
- Ross, K.M.; Dunkel Schetter, C.; McLemore, M.R.; Chambers, B.D.; Paynter, R.A.; Baer, R.; Feuer, S.K.; Flowers, E.; Karasek, D.; Pantell, M.; et al. Socioeconomic Status, Preeclampsia Risk and Gestational Length in Black and White Women. J. Racial Ethn. Health Dispar. 2019, 6, 1182–1191. [Google Scholar] [CrossRef]
- Howe, L.; Hammer, E.; Badger, G.; Bernstein, I.M. Effect of Pregnancy Interval on Second Pregnancy Blood Pressure Following Prior Preeclampsia. Reprod. Sci. 2018, 25, 727–732. [Google Scholar] [CrossRef]
- Sohlberg, S.; Stephansson, O.; Cnattingius, S.; Wikström, A.-K. Maternal body mass index, height, and risks of preeclampsia. Am. J. Hypertens. 2012, 25, 120–125. [Google Scholar] [CrossRef]
- Phipps, E.; Prasanna, D.; Brima, W.; Jim, B. Preeclampsia: Updates in Pathogenesis, Definitions, and Guidelines. Clin. J. Am. Soc. Nephrol. 2016, 11, 1102–1113. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Syngelaki, A.; Maiz, N.; Zinevich, Y.; Nicolaides, K.H. Maternal age and adverse pregnancy outcome: A cohort study. Ultrasound Obstet. Gynecol. 2013, 42, 634–643. [Google Scholar] [CrossRef] [PubMed]
| Controls (n = 775) | GH (n = 113) | PE (n = 24) | |||
|---|---|---|---|---|---|
| Variables | Mean (SD) or n (%) | Mean (SD) or n (%) | p * | Mean (SD) or n (%) | p * |
| Maternal age (years) | 33.5 (4.8) | 35.0 (4.3) | 0.005 | 34.1 (5.0) | 0.434 |
| Primiparous women | 318 (41.0%) | 53 (46.9%) | 0.237 | 12 (50.0%) | 0.380 |
| Prior GH/PE | 4 (0.5%) | 12 (10.6%) | <0.001 | 3 (12.5%) | <0.001 |
| Aspirin (for prophylaxis) | 7 (0.9%) | 2 (1.8%) | 0.390 | 2 (8.3%) | <0.001 |
| Infertility treatment | 29 (3.7%) | 8 (7.1%) | 0.097 | 3 (12.5%) | 0.031 |
| Interpregnancy interval ** | 4.4 (4.0) | 5.6 (4.3) | 0.031 | 7.4 (5.3) | 0.023 |
| Smoking in I trimester | 37 (4.8%) | 17 (15.0%) | <0.001 | 3 (12.5%) | 0.084 |
| Education <12 years ** | 48 (7%)1 | 16 (16.3%) | 0.002 | 6 (27.3%) | <0.001 |
| Lower financial status ** | 95 (26.1%) | 28 (43.8%) | <0.001 | 8 (57.1%) | 0.007 |
| Family history | |||||
| Hypertension in the mother | 137 (17.8%) | 31 (27.7%) | 0.013 | 11 (45.8%) | <0.001 |
| Pre-pregnancy BMI (kg/m2) | 23.3 (4.1) | 26.7 (5.3) | <0.001 | 26.5 (6.2) | 0.008 |
| GWG (kg) | 13.4 (5.3) | 14.6 (8.0) | 0.115 | 15.1(8.2) | 0.612 |
| Fetal sex, daughter | 370 (47.7%) | 58 (51.3%) | 0.476 | 11 (45.8%) | 0.853 |
| Gestational age at childbirth (week) | 38.9 (1.6) | 38.3 (2.2) | 0.016 | 35.1 (3.7) | <0.001 |
| Preterm birth <37th week | 41 (5.3%) | 11 (9.7%) | 0.060 | 13 (54.2%) | <0.001 |
| Preterm birth <34th week | 10 (1.3%) | 7 (6.2%) | <0.001 | 6 (25.0%) | <0.001 |
| Newborn birthweight (g) | 3416.5 (511.7) | 3174.1 (734.3) | 0.001 | 2294.2 (927.5) | <0.001 |
| IUGR cases | 11 (1.4%) | 6 (5.5%) | 0.004 | 4 (18.2%) | <0.001 |
| Cesarean section | 306 (39.5%) | 54 (47.8%) | 0.093 | 22 (91.7%) | <0.001 |
| GDM | 121 (15.6%) | 22 (19.5%) | 0.298 | 3 (12.5%) | 0.678 |
| Gestational diabetes mellitus | 121 (15.6%) | 22 (19.5%) | 0.298 | 3 (12.5%) | 0.678 |
| PE beginning <32th week | 7 (29.2%) | ||||
| PE beginning ≥34th week | - | - | - | 13 (54.2%) | - |
| Blood pressure before pregnancy | |||||
| Systolic (mmHg) | 104.9 (10.4) | 123.5 (8.5) | <0.001 | 123.3 (8.7) | <0.001 |
| Diastolic (mmHg) | 64.5 (8.7) | 76.8 (7.7) | <0.001 | 78.1 (8.6) | <0.001 |
| Blood pressure after delivery | |||||
| Systolic (mmHg) | 107.9 (10.8) | 157.2 (17.6) | <0.001 | 170.3 (17.5) | <0.001 |
| Diastolic (mmHg) | 66.8 (8.8) | 99.6 (9.3) | <0.001 | 106.8 (15.8) | <0.001 |
| Variables | GH OR (95% CI) * | p ** | PE OR (95% CI) * | p ** |
|---|---|---|---|---|
| Continuous variables: | ||||
| Pre-pregnancy BMI (kg/m2) | 1.16 (1.11–1.21) | <0.001 | 1.15 (1.06–1.24) | <0.001 |
| Maternal age (years) | 1.07 (1.03–1.12) | 0.003 | 1.03 (0.94–1.12) | 0.539 |
| GWG (kg) | 1.04 (1.00–1.07) | 0.040 | 1.06 (0.98–1.14) | 0.145 |
| Other variables: | ||||
| Prior GH/PE (vs. others) | 22.90 (7.3–72.4) | <0.001 | 27.54 (5.8–130.8) | <0.001 |
| BMI ≥ 30 kg/m2 (vs. normal BMI) | 5.60 (3.32–9.43) | <0.001 | 9.21 (3.52–24.11) | <0.001 |
| Smoking in I trimester (vs. others) | 3.53 (1.92–6.52) | <0.001 | 2.85 (0.81–9.99) | 0.102 |
| Age ≥40 years (vs. 25–29 years) | 3.23 (1.41–7.38) | 0.005 | 1.11 (0.1–12.49) | 0.933 |
| Education <12 years vs. others ** | 2.50 (1.37–4.57) | 0.003 | 5.05 (1.92–13.31) | 0.001 |
| GWG above the range (vs. normal) | 2.45 (1.53–3.92) | <0.001 | 1.57 (0.62–3.97) | 0.337 |
| Financial status (1-2-3) vs. others ** | 2.36 (1.46–3.80) | <0.001 | 3.58 (1.49–8.59) | 0.004 |
| Urogenital infection (vs. others) | 2.12 (1.31–3.43) | 0.002 | 0.61 (0.14–2.65) | 0.513 |
| No multivitamins (vs. others) # | 2.11 (1.41–3.15) | <0.001 | 0.90 (0.39–2.08) | 0.806 |
| Family history of Hypertension (H) | ||||
| H in the father (vs. controls) *** | 2.06 (1.29–3.28) | 0.003 | 1.84 (0.62–5.47) | 0.274 |
| H in the mother (vs. controls) *** | 1.90 (1.18–3.06) | 0.008 | 3.98 (1.66–9.57) | 0.002 |
| Interpregnancy interval (years) | ||||
| ≥11 years (vs. 1 year) | 2.02 (0.90–4.58) | 0.091 | 6.63 (1.18–37.29) | 0.032 |
| Hypothyroidism (vs. others) | 2.01 (1.23–3.29) | 0.006 | 2.36 (0.91–6.09) | 0.076 |
| Infertility treatment (vs. others) | 1.96 (0.87–4.4) | 0.103 | 3.68 (1.04–13.03) | 0.044 |
| In vitro fertilization (vs. others) | 1.92 (0.76–4.84) | 0.167 | 3.11 (0.69–14.06) | 0.140 |
| Primiparity (vs. multiparity) | 1.27 (0.85–1.89) | 0.238 | 1.22 (0.54–2.75) | 0.638 |
| Variables | GH AUC | p * | PE AUC | p * |
|---|---|---|---|---|
| Pre-pregnancy BMI (kg/m2) | 0.698 | <0.001 | 0.660 | 0.008 |
| Maternal age (years) | 0.581 | 0.005 | 0.547 | 0.435 |
| GWG (kg) | 0.546 | 0.115 | 0.530 | 0.613 |
| Maternal age + primiparity | 0.600 | 0.001 | 0.539 | 0.520 |
| GH | ||||||
|---|---|---|---|---|---|---|
| Base Model (Maternal Age + Primiparity) | AUC Base 0.600 | |||||
| Extended Models (Base Model + Listed Variables) | AUC Extended | p-Value * (Extended vs. Base) | IDI | p-Value * (Extended vs. Base) | NRI | p-Value * (Extended vs. Base) |
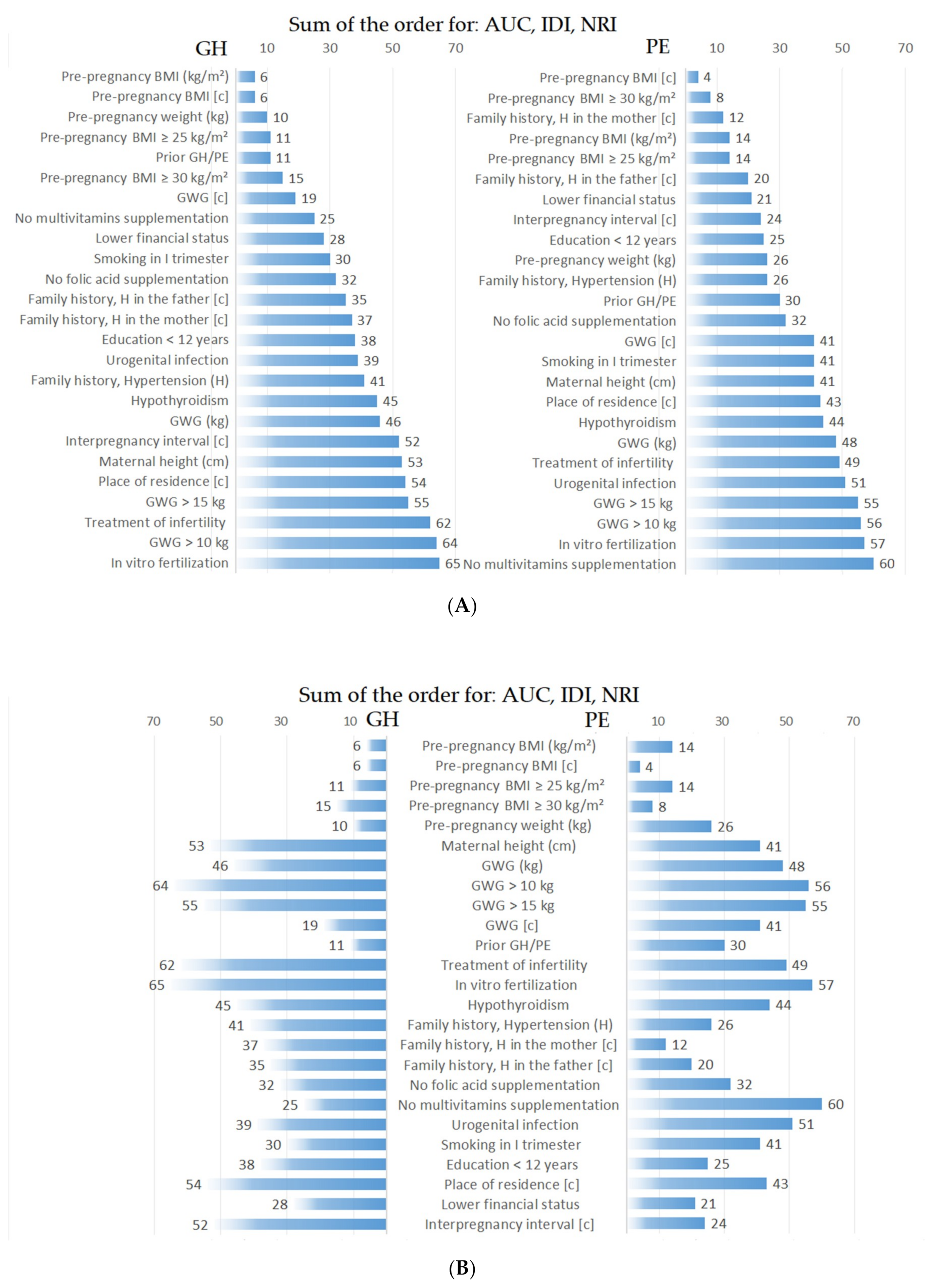
| Pre-pregnancy BMI (kg/m2) | 0.716 | <0.001 | 0.064 | <0.001 | 0.542 | <0.001 |
| Pre-pregnancy BMI (c) | 0.704 | <0.001 | 0.058 | <0.001 | 0.571 | <0.001 |
| Pre-pregnancy weight (kg) | 0.697 | <0.001 | 0.044 | <0.001 | 0.544 | <0.001 |
| Pre-pregnancy BMI ≥ 25 kg/m2 | 0.685 | 0.001 | 0.041 | <0.001 | 0.571 | <0.001 |
| Pre-pregnancy BMI ≥ 30 kg/m2 | 0.663 | 0.002 | 0.045 | <0.001 | 0.399 | <0.001 |
| Prior GH/PE | 0.656 | 0.002 | 0.068 | <0.001 | 0.433 | <0.001 |
| GWG (c) | 0.653 | 0.024 | 0.027 | <0.001 | 0.419 | <0.001 |
| Financial status | 0.648 | 0.022 | 0.015 | 0.007 | 0.250 | 0.003 |
| No multivitamins | 0.646 | 0.039 | 0.016 | 0.001 | 0.368 | <0.001 |
| Smoking in I trimester | 0.646 | 0.025 | 0.024 | 0.003 | 0.205 | 0.003 |
| Education <12 years | 0.643 | 0.018 | 0.01 | 0.043 | 0.159 | 0.019 |
| Family history; H in the mother (c) | 0.639 | 0.034 | 0.004 | 0.19 | 0.236 | 0.019 |
| Family history; H in the father (c) | 0.638 | 0.052 | 0.008 | 0.046 | 0.279 | 0.005 |
| No folic acid supplementation | 0.634 | 0.101 | 0.013 | <0.001 | 0.312 | <0.001 |
| Urogenital infection | 0.632 | 0.075 | 0.01 | 0.022 | 0.220 | 0.009 |
| Family history of Hypertension | 0.631 | 0.064 | 0.004 | 0.194 | 0.236 | 0.019 |
| Interpregnancy interval (c) | 0.619 | 0.166 | 0.004 | 0.086 | −0.014 | 0.891 |
| Hypothyroidism | 0.618 | 0.253 | 0.008 | 0.032 | 0.195 | 0.017 |
| GWG (kg) | 0.616 | 0.402 | 0.011 | 0.009 | 0.149 | 0.139 |
| Maternal height (cm) | 0.613 | 0.22 | 0.003 | 0.137 | 0.125 | 0.213 |
| Place of residence (c) | 0.611 | 0.318 | 0.002 | 0.238 | 0.144 | 0.094 |
| GWG > 15 kg | 0.607 | 0.551 | 0.004 | 0.077 | 0.113 | 0.248 |
| Treatment of infertility | 0.601 | 0.621 | 0.0007 | 0.562 | −0.074 | 0.386 |
| In vitro fertilization | 0.6 | 0.925 | 0.0005 | 0.6 | −0.12 | 0.151 |
| GWG > 10 kg | 0.598 | 0.599 | 0.0003 | 0.454 | −0.011 | 0.903 |
| PE | ||||||
|---|---|---|---|---|---|---|
| Base Model (Maternal Age + Primiparity) | AUC Base 0.539 | |||||
| Extended Models (Base Model + Listed Variables) | AUC Extended | p-Value *(Extended vs. Base) | IDI | p-Value * (Extended vs. Base) | NRI | p-Value * (Extended vs. Base) |
| Pre-pregnancy BMI (c) | 0.726 | 0.017 | 0.034 | 0.002 | 0.688 | 0.001 |
| Family history of H in the mother (c) | 0.717 | 0.009 | 0.013 | 0.020 | 0.563 | 0.006 |
| Pre-pregnancy BMI ≥ 30 kg/m2 | 0.703 | 0.009 | 0.032 | 0.004 | 0.600 | 0.002 |
| Pre-pregnancy BMI ≥ 25 kg/m2 | 0.679 | 0.042 | 0.012 | 0.011 | 0.575 | 0.005 |
| Pre-pregnancy BMI (kg/m2) | 0.678 | 0.107 | 0.02 | 0.019 | 0.510 | 0.013 |
| Family history of H in the father (c) | 0.658 | 0.094 | 0.007 | 0.059 | 0.447 | 0.029 |
| Financial status | 0.648 | 0.068 | 0.012 | 0.039 | 0.422 | 0.030 |
| Interpregnancy interval (c) | 0.646 | 0.138 | 0.007 | 0.072 | 0.405 | 0.032 |
| Pre-pregnancy weight (kg) | 0.642 | 0.233 | 0.012 | 0.041 | 0.373 | 0.071 |
| Family history of hypertension (H) | 0.638 | 0.119 | 0.005 | 0.069 | 0.447 | 0.029 |
| Education <12 years | 0.629 | 0.05 | 0.018 | 0.034 | 0.376 | 0.034 |
| No folic acid supplementation | 0.625 | 0.032 | 0.005 | 0.010 | 0.373 | 0.028 |
| Prior GH/PE | 0.622 | 0.097 | 0.050 | 0.080 | 0.098 | 0.602 |
| Maternal height (cm) | 0.589 | 0.289 | 0.002 | 0.253 | 0.157 | 0.448 |
| Smoking in I trimester | 0.588 | 0.319 | 0.004 | 0.266 | 0.155 | 0.255 |
| GWG (c) | 0.586 | 0.407 | 0.002 | 0.205 | 0.238 | 0.249 |
| Place of residence (c) | 0.586 | 0.187 | 0.001 | 0.161 | 0.174 | 0.302 |
| Treatment of infertility | 0.585 | 0.163 | 0.005 | 0.251 | −0.121 | 0.436 |
| Urogenital infection | 0.568 | 0.188 | 0.0008 | 0.258 | 0.091 | 0.428 |
| GWG (kg) | 0.565 | 0.72 | 0.006 | 0.165 | 0.055 | 0.791 |
| GWG > 15 kg | 0.563 | 0.507 | 0.0004 | 0.577 | 0.084 | 0.675 |
| No multivitamins supplementation | 0.557 | 0.319 | −0.0001 | 0.870 | 0.05 | 0.803 |
| In vitro fertilization | 0.555 | 0.174 | 0.003 | 0.372 | −0.183 | 0.239 |
| GWG > 10 kg | 0.555 | 0.49 | 0.0001 | 0.792 | 0.094 | 0.631 |
| Hypothyroidism | 0.553 | 0.655 | 0.005 | 0.124 | 0.252 | 0.157 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewandowska, M.; Więckowska, B.; Sajdak, S.; Lubiński, J. Pre-Pregnancy Obesity vs. Other Risk Factors in Probability Models of Preeclampsia and Gestational Hypertension. Nutrients 2020, 12, 2681. https://doi.org/10.3390/nu12092681
Lewandowska M, Więckowska B, Sajdak S, Lubiński J. Pre-Pregnancy Obesity vs. Other Risk Factors in Probability Models of Preeclampsia and Gestational Hypertension. Nutrients. 2020; 12(9):2681. https://doi.org/10.3390/nu12092681
Chicago/Turabian StyleLewandowska, Małgorzata, Barbara Więckowska, Stefan Sajdak, and Jan Lubiński. 2020. "Pre-Pregnancy Obesity vs. Other Risk Factors in Probability Models of Preeclampsia and Gestational Hypertension" Nutrients 12, no. 9: 2681. https://doi.org/10.3390/nu12092681
APA StyleLewandowska, M., Więckowska, B., Sajdak, S., & Lubiński, J. (2020). Pre-Pregnancy Obesity vs. Other Risk Factors in Probability Models of Preeclampsia and Gestational Hypertension. Nutrients, 12(9), 2681. https://doi.org/10.3390/nu12092681





