Effects of Dietary Glucose and Fructose on Copper, Iron, and Zinc Metabolism Parameters in Humans
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Subjects and Study Design
2.2. Diet
2.3. Serum Metals and Ceruloplasmin Ferroxidase Activity
2.4. Serum Protein Levels
2.5. Plasma Lipid and Glucose Parameters
2.6. Statistical Analyses
3. Results
3.1. Baseline Measures
3.2. Effects of Glucose, Fructose, HFCS, and Aspartame on Metals and Metal Metabolism Markers
3.3. Correlations of Baseline Serum Metal Concentrations with Baseline Metal Metabolic Markers
3.4. Correlations of Changes in Serum Metal Concentrations with Changes in Metal Metabolism Markers
3.5. Correlations of Changes in Serum Metal Concentrations with Changes in Metabolism Markers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hasan, N.M. Regulation of Copper Transporters in Human Cells. Curr. Top. Membr. 2012, 69, 137–161. [Google Scholar] [CrossRef] [PubMed]
- Morrell, A.; Tallino, S.; Yu, L.; Burkhead, J.L. The role of insufficient copper in lipid synthesis and fatty-liver disease. IUBMB Life 2017, 69, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Lin, Z.; Zhang, Y. The Alteration of Copper Homeostasis in Inflammation Induced by Lipopolysaccharides. Biol. Trace Elem. Res. 2013, 154, 268–274. [Google Scholar] [CrossRef]
- Kim, H.; Son, H.-Y.; Bailey, S.M.; Lee, J. Deletion of hepatic Ctr1 reveals its function in copper acquisition and compensatory mechanisms for copper homeostasis. AJP Gastrointest. Liver Physiol. 2008, 296, G356–G364. [Google Scholar] [CrossRef] [PubMed]
- Lutsenko, S. Copper trafficking to the secretory pathway. Metallomics 2016, 8, 840–852. [Google Scholar] [CrossRef]
- Linder, M.C. Ceruloplasmin and other copper binding components of blood plasma and their functions: An update. Metallomics 2016, 8, 887–905. [Google Scholar] [CrossRef]
- Lutsenko, S.; Barnes, N.L.; Bartee, M.Y.; Dmitriev, O.Y. Function and Regulation of Human Copper-Transporting ATPases. Physiol. Rev. 2007, 87, 1011–1046. [Google Scholar] [CrossRef]
- Członkowska, A.; Litwin, T.; Dusek, P.; Ferenci, P.; Lutsenko, S.; Medici, V.; Rybakowski, J.K.; Weiss, K.H.; Schilsky, M.L. Wilson disease. Nat. Rev. Dis. Prim. 2018, 4, 1–20. [Google Scholar] [CrossRef]
- Roberts, E.A. Update on the Diagnosis and Management of Wilson Disease. Curr. Gastroenterol. Rep. 2018, 20, 1–12. [Google Scholar] [CrossRef]
- Heffern, M.C.; Park, H.M.; Au-Yeung, H.Y.; van de Bittner, G.C.; Ackerman, C.M.; Stahl, A.; Chang, C.J. In vivo bioluminescence imaging reveals copper deficiency in a murine model of nonalcoholic fatty liver disease. Proc. Natl. Acad. Sci. USA 2016, 113, 14219–14224. [Google Scholar] [CrossRef]
- Klevay, L.M. Is the Western diet adequate in copper? J. Trace Elem. Med. Biol. 2011, 25, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.H.C.; Riddell, L.J.; Nowson, C.A.; Booth, A.O.; Szymlek-Gay, E.A. Iron and zinc nutrition in the economically-developed world: A review. Nutrients 2013, 5, 3184–3211. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhang, C.; Bu, J. Relationship between selected serum metallic elements and obesity in children and adolescent in the U.S. Nutrients 2017, 9, 104. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Liu, C.N.; Wolf, R.M.; Ralle, M.; Dev, S.; Pierson, H.; Askin, F.; Steele, K.E.; Magnuson, T.H.; Schweitzer, M.A.; et al. Obesity is associated with copper elevation in serum and tissues. Metallomics 2019, 11, 1363–1371. [Google Scholar] [CrossRef]
- Aigner, E.; Strasser, M.; Haufe, H.; Sonnweber, T.; Hohla, F.; Stadlmayr, A.; Solioz, M.; Tilg, H.; Patsch, W.; Weiss, G.; et al. A role for low hepatic copper concentrations in nonalcoholic fatty liver disease. Am. J. Gastroenterol. 2010, 105, 1978–1985. [Google Scholar] [CrossRef]
- Song, M.; Schuschke, D.A.; Zhou, Z.; Chen, T.; Pierce, W.M.; Wang, R.; Johnson, W.T.; McClain, C.J. High fructose feeding induces copper deficiency in Sprague-Dawley rats: A novel mechanism for obesity related fatty liver. J. Hepatol. 2012, 56, 433–440. [Google Scholar] [CrossRef]
- Lane, D.J.R.; Ayton, S.; Bush, A.I. Iron and Alzheimer’s Disease: An Update on Emerging Mechanisms. J. Alzheimers Dis. 2018, 64, S379–S395. [Google Scholar] [CrossRef]
- Datz, C.; Müller, E.; Aigner, E. Iron overload and non-alcoholic fatty liver disease. Miner. Endocrinol. 2017, 42, 173–183. [Google Scholar]
- Brabin, B.J.; Hakimi, M.; Pelletier, D. An Analysis of Anemia and Pregnancy-Related Maternal Mortality. J. Nutr. 2001, 131, 604S–615S. [Google Scholar] [CrossRef]
- Burggraf, L.W.; Hansen, S.D.; Bleckmann, C.A. Metabolic inhibition by transition metal ions in a slow-growing, toluene-enriched microbial population. Environ. Toxicol. Water Qual. 1998, 13, 249–261. [Google Scholar] [CrossRef]
- Salaye, L.; Bychkova, I.; Sink, S.; Kovalic, A.J.; Bharadwaj, M.S.; Lorenzo, F.; Jain, S.; Harrison, A.V.; Davis, A.T.; Turnbull, K.; et al. A Low Iron Diet Protects from Steatohepatitis in a Mouse Model. Nutrients 2019, 11, 2172. [Google Scholar] [CrossRef] [PubMed]
- Tamaki, M.; Fujitani, Y.; Hara, A.; Uchida, T.; Tamura, Y.; Takeno, K.; Kawaguchi, M.; Watanabe, T.; Ogihara, T.; Fukunaka, A.; et al. The diabetes-susceptible gene SLC30A8/ZnT8 regulates hepatic insulin clearance. J. Clin. Investig. 2013, 123, 4513–4524. [Google Scholar] [CrossRef] [PubMed]
- Daru, J.; Colman, K.; Stanworth, S.J.; de La Salle, B.; Wood, E.M.; Pasricha, S.R. Serum ferritin as an indicator of iron status: What do we need to know? Am. J. Clin. Nutr. 2017, 106, 1634S–1639S. [Google Scholar] [CrossRef] [PubMed]
- Viveiros, A.; Finkenstedt, A.; Schaefer, B.; Mandorfer, M.; Scheiner, B.; Lehner, K.; Tobiasch, M.; Reiberger, T.; Tilg, H.; Edlinger, M.; et al. Transferrin as a predictor of survival in cirrhosis. Liver Transpl. 2018, 24, 343–351. [Google Scholar] [CrossRef]
- Coleman, J.E. Zinc Proteins: Enzymes, Storage Proteins, Transcription Factors, and Replication Proteins. Annu. Rev. Biochem. 1992, 61, 897–946. [Google Scholar] [CrossRef]
- Ahmed, U.; Latham, P.S.; Oates, P.S. Interactions between hepatic iron and lipid metabolism with possible relevance to steatohepatitis. World J. Gastroenterol. 2012, 18, 4651–4658. [Google Scholar] [CrossRef]
- Meerarani, P.; Reiterer, G.; Toborek, M.; Hennig, B. Zinc modulates PPARgamma signaling and activation of porcine endothelial cells. J. Nutr. 2003, 133, 3058–3064. [Google Scholar] [CrossRef]
- Temple, K.A.; Cohen, R.N.; Wondisford, S.R.; Yu, C.; Deplewski, D.; Wondisford, F.E. An intact DNA-binding domain is not required for peroxisome proliferator-activated receptor γ (PPARγ) binding and activation on some PPAR response elements. J. Biol. Chem. 2005, 280, 3529–3540. [Google Scholar] [CrossRef]
- Kelly, D.; Crotty, G.; O’Mullane, J.; Stapleton, M.; Sweeney, B.; O’Sullivan, S.S. The Clinical Utility of a Low Serum Ceruloplasmin Measurement in the Diagnosis of Wilson Disease. Ir. Med. J. 2016, 109, 341–343. [Google Scholar]
- Twomey, P.J.; Viljoen, A.; Reynolds, T.M.; Wierzbicki, A.S. Non-ceruloplasmin-bound copper in routine clinical practice in different laboratories. J. Trace Elem. Med. Biol. Organ Soc. Min. Trace Elem. 2008, 22, 50–53. [Google Scholar] [CrossRef]
- Hellman, N.E.; Gitlin, J.D. Ceruloplasmin metabolism and function. Annu. Rev. Nutr. 2002, 22, 439–458. [Google Scholar] [CrossRef] [PubMed]
- Członkowska, A.; Rodo, M.; Wierzchowska-Ciok, A.; Smolinski, L.; Litwin, T. Accuracy of the radioactive copper incorporation test in the diagnosis of Wilson disease. Liver Int. 2018, 38, 1860–1866. [Google Scholar] [CrossRef] [PubMed]
- Woimant, F.; Djebrani-Oussedik, N.; Poujois, A. New tools for Wilson’s disease diagnosis: Exchangeable copper fraction. Ann. Transl. Med. 2019, 7, S70. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Medici, V.; Bremer, A.A.; Lee, V.; Lam, H.D.; Nunez, M.V.; Chen, G.X.; Keim, N.L.; Havel, P.J. A dose-response study of consuming high-fructose corn syrup–sweetened beverages on lipid/lipoprotein risk factors for cardiovascular disease in young adults. Am. J. Clin. Nutr. 2015, 101, 1144–1154. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Bremer, A.A.; Medici, V.; Nakajima, K.; Ito, Y.; Nakano, T.; Chen, G.; Fong, T.H.; Lee, V.; Menorca, R.I.; et al. Consumption of fructose and high fructose corn syrup increase postprandial triglycerides, LDL-cholesterol, and apolipoprotein-B in young men and women. J. Clin. Endocrinol. Metab. 2011, 96, E1596. [Google Scholar] [CrossRef]
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [CrossRef]
- Ramanujam, V.M.S. Riboflavin as an Oral Tracer for Monitoring Compliance in Clinical Research. Open Biomark. J. 2011, 4, 1–7. [Google Scholar] [CrossRef][Green Version]
- Olusi, S.; Al-Awadhi, A.; Abiaka, C.; Abraham, M.; George, S. Serum copper levels and not zinc are positively associated with serum leptin concentrations in the healthy adult population. Biol. Trace Elem. Res. 2003, 91, 137–144. [Google Scholar] [CrossRef]
- Tallino, S.; Duffy, M.; Ralle, M.; Cortés, M.P.; Latorre, M.; Burkhead, J.L. Nutrigenomics analysis reveals that copper deficiency and dietary sucrose up-regulate inflammation, fibrosis and lipogenic pathways in a mature rat model of nonalcoholic fatty liver disease. J. Nutr. Biochem. 2015, 26, 996–1006. [Google Scholar] [CrossRef]
- Porcu, C.; Antonucci, L.; Barbaro, B.; Illi, B.; Nasi, S.; Martini, M.; Licata, A.; Miele, L.; Grieco, A.; Balsano, C. Copper/MYC/CTR1 interplay: A dangerous relationship in hepatocellular carcinoma. Oncotarget 2018, 9, 9325–9343. [Google Scholar] [CrossRef]
- Aigner, E.; Theurl, I.; Haufe, H.; Seifert, M.; Hohla, F.; Scharinger, L.; Stickel, F.; Mourlane, F.; Weiss, G.; Datz, C. Copper Availability Contributes to Iron Perturbations in Human Nonalcoholic Fatty Liver Disease. Gastroenterology 2008, 135, 680–688. [Google Scholar] [CrossRef]
- Song, M.; Li, X.; Zhang, X.; Shi, H.; Vos, M.B.; Wei, X.; Wang, Y.; Gao, H.; Rouchka, E.C.; Yin, X.; et al. Dietary copper-fructose interactions alter gut microbial activity in male rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 314, G119–G130. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, L.; Cotruvo, J.A.; Chan, J.; Kaluarachchi, H.; Muchenditsi, A.; Pendyala, V.S.; Jia, S.; Aron, A.T.; Ackerman, C.M.; Vander Wal, M.N.; et al. Copper regulates cyclic-AMP-dependent lipolysis. Nat. Chem. Biol. 2016, 12, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Prohaska, J.R. Changes in Cu,Zn-Superoxide Dismutase, Cytochrome c Oxidase, Glutathione Peroxidase and Glutathione Transferase Activities in Copper-Deficient Mice and Rats. J. Nutr. 1991, 121, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, C.H.; Reeve, V.E.; Wright, R. Copper deficiency in the rat. Effect on the ultrastructure of hepatocytes. Aust. J. Exp. Biol. Med. Sci. 1973, 51, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Vos, M.; McClain, C. Copper-Fructose Interactions: A Novel Mechanism in the Pathogenesis of NAFLD. Nutrients 2018, 10, 1815. [Google Scholar] [CrossRef]
- Fields, M.; Lewis, C.G. Dietary Fructose but Not Starch is Responsible for Hyperlipidemia Associated with Copper Deficiency in Rats: Effect of High-Fat Diet. J. Am. Coll. Nutr. 1999, 18, 83–87. [Google Scholar] [CrossRef]
- Morrell, A.; Tripet, B.P.; Eilers, B.J.; Tegman, M.; Thompson, D.; Copié, V.; Burkhead, J.L. Copper modulates sex-specific fructose hepatoxicity in nonalcoholic fatty liver disease (NALFD) Wistar rat models. J. Nutr. Biochem. 2020, 78, 108316. [Google Scholar] [CrossRef]
- Hieronimus, B.; Griffen, S.C.; Keim, N.L.; Bremer, A.A.; Berglund, L.; Nakajima, K.; Havel, P.J.; Stanhope, K.L. Effects of Fructose or Glucose on Circulating ApoCIII and Triglyceride and Cholesterol Content of Lipoprotein Subfractions in Humans. J. Clin. Med. 2019, 8, 913. [Google Scholar] [CrossRef]
- Jiang, B.; Liu, G.; Zheng, J.; Chen, M.; Maimaitiming, Z.; Chen, M.; Liu, S.; Jiang, R.; Fuqua, B.K.; Dunaief, J.L.; et al. Hephaestin and ceruloplasmin facilitate iron metabolism in the mouse kidney. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Navarro, J.A.; Schneuwly, S. Copper and zinc homeostasis: Lessons from Drosophila melanogaster. Front. Genet. 2017, 8, 223. [Google Scholar]
- Latorre, M.; Low, M.; Gárate, E.; Reyes-Jara, A.; Murray, B.E.; Cambiazo, V.; González, M. Interplay between copper and zinc homeostasis through the transcriptional regulator Zur in Enterococcus faecalis. Metallomics 2015, 7, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Panemangalore, M.; Bebe, F.N. Effect of high dietary zinc on plasma ceruloplasmin and erythrocyte superoxide dismutase activities in copper-depleted and repleted rats. Biol. Trace Elem. Res. 1996, 55, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Ausk, K.J.; Ioannou, G.N. Is obesity associated with anemia of chronic disease? A population-based study. Obesity 2008, 16, 2356–2361. [Google Scholar] [CrossRef]
- Ganz, T. Hepcidin—A Peptide Hormone at the Interface of Innate Immunity and Iron Metabolism. In Antimicrobial Peptides and Human Disease; Springer: Berlin, Germany, 2006; Volume 306, pp. 183–198. [Google Scholar]
- Zhou, B.; Ren, H.; Zhou, X.; Yuan, G. Associations of iron status with apolipoproteins and lipid ratios: A cross-sectional study from the China Health and Nutrition Survey. Lipids Health Dis. 2020, 19, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mak, C.M.; Lam, C.-W. Diagnosis of Wilson’s disease: A comprehensive review. Crit. Rev. Clin. Lab. Sci. 2008, 45, 263–290. [Google Scholar] [CrossRef]
- Pollack, S.; Kaufman, R.M.; Crosby, W.H. Iron Absoption: Effects of Sugars and Reducing Agents. Blood 1964, 24, 577–581. [Google Scholar] [CrossRef]
- Christides, T.; Sharp, P. Sugars increase non-heme iron bioavailability in human epithelial intestinal and liver cells. PLoS ONE 2013, 8, e83031. [Google Scholar] [CrossRef]
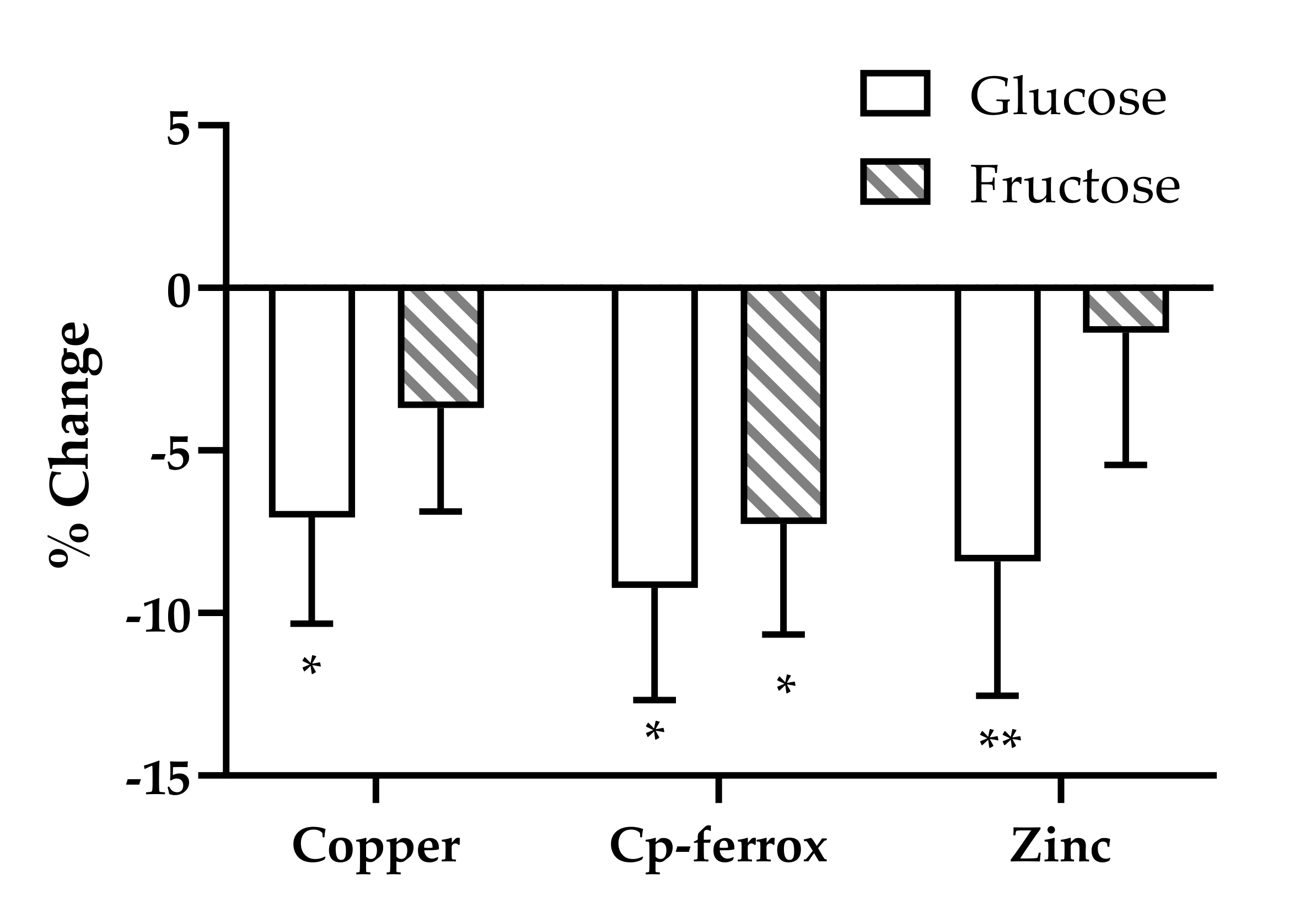
| Glucose n = 28 | Fructose n = 28 | HFCS n = 28 | Aspartame n = 23 | Total n = 107 | |
|---|---|---|---|---|---|
| BMI | 25.4 ± 0.7 | 25.8 ± 0.7 | 24.9 ± 0.8 | 24.8 ± 0.7 | 25.3 ± 0.4 |
| Age | 26.8 ± 1.2 | 26.0 ± 1.1 | 26.8 ± 1.2 | 25.4 ± 1.3 | 26.3 ± 1.6 |
| Sex | 15 M/13 F | 15 M/13 F | 15 M/13 F | 11 M/12 F | 56 M/51 F |
| % M/F | 53.6/46.4 | 53.6/46.4 | 53.6/46.4 | 47.9/53.1 | 52.3/47.7 |
| Aspartame | Glucose | Fructose | HFCS | |||||
|---|---|---|---|---|---|---|---|---|
| Week 0 | Week 2 | Week 0 | Week 2 | Week 0 | Week 2 | Week 0 | Week 2 | |
| Cp (µg/mL) | 773 ± 125 | 1095 ± 287 | 920 ± 157 | 815 ± 125 | 1098 ± 258 | 805 ± 198 | 885 ± 146 | 984 ± 156 |
| Cp-ferrox (U/mL) | 26.8 ± 2.5 | 26.8 ±2.3 | 28.2 ± 2.2 | 26.1 ± 1.8 | 28.8 ± 1.5 | 27.7 ± 1.5 | 29.5 ± 2.3 | 27.5 ± 2.1 |
| Transferrin (mg/dL) | 93.6 ± 6.6 | 84.87 ± 6.5 | 104.2 ± 4.4 | 104.4 ± 6.2 | 101.2 ± 5.1 | 94.0 ± 5.8 | 109.2 ± 5.7 | 94.0 ± 5.4 |
| Ferritin (ng/mL) | 37.7 ± 10.2 | 28.9 ± 7.54 | 48.7 ± 7.9 | 36.8 ± 7.1 | 45.6 ± 8.4 | 28.7 ± 5.7 | 41.9 ± 6.0 | 31.1 ± 5.3 |
| Copper (ppb) | 912 ± 49 | 881 ± 41 | 976 ± 55 | 869 ± 43 | 953 ± 34 | 898 ± 32 | 1019 ± 55 | 935 ± 49 |
| Iron (ppb) | 1029 ± 77 | 726 ± 68 | 920 ± 82 | 825 ± 78 | 1037 ± 87 | 796 ± 53 | 1007 ± 74 | 750 ± 59 |
| Zinc (ppb) | 975 ± 37 | 957 ± 30 | 1012 ± 24 | 932 ± 33 | 983 ± 24 | 969 ± 24 | 991 ± 33 | 897 ± 22 |
| Beverage | Sex | BMI | Time | Beverage × time | |
|---|---|---|---|---|---|
| p-value | p-value | p-value | p-value | p-value | |
| Cp | 0.972 | 0.615 | 0.289 | 0.945 | 0.087 |
| Cp-ferrox | 0.873 | 0.005 | 0.255 | <0.001 | 0.161 |
| Copper | 0.203 | <0.001 | <0.001 | <0.001 | 0.331 |
| Zinc | 0.737 | <0.001 | 0.558 | <0.001 | 0.084 |
| Iron | 0.916 | 0.001 | 0.039 | <0.001 | 0.442 |
| Ferritin | 0.878 | <0.001 | 0.177 | <0.001 | 0.284 |
| Transferrin | 0.104 | 0.355 | 0.590 | 0.003 | 0.209 |
| BMI < 25 (n = 51) | BMI ≥ 25 (n = 56) | p-Value | ||
| Copper | Baseline | 918 ± 33 | 995 ± 30 | 0.08 |
| Intervention | 847 ± 29 | 936 ± 29 | 0.03 | |
| Iron | Baseline | 1107 ± 64 | 899 ± 47 | 0.008 |
| Intervention | 779 ± 53 | 776 ± 40 | 0.97 | |
| Copper | Iron | Zinc | Cp-ferrox | Cp | Transferrin | Ferritin | ||
|---|---|---|---|---|---|---|---|---|
| Copper | r | −0.131 | −0.082 | 0.520 | 0.125 | 0.014 | −0.237 | |
| p | 0.184 | 0.411 | <0.001 | 0.204 | 0.890 | 0.016 | ||
| Iron | r | −0.131 | 0.048 | −0.098 | 0.016 | 0.350 | 0.241 | |
| p | 0.184 | 0.628 | 0.321 | 0.872 | <0.001 | 0.014 | ||
| Zinc | r | −0.0815 | 0.048 | 0.132 | −0.208 | 0.169 | −0.088 | |
| p | 0.411 | 0.628 | 0.178 | 0.033 | 0.087 | 0.376 | ||
| Cp-ferrox | r | 0.520 | −0.098 | 0.132 | −0.452 | 0.028 | −0.153 | |
| p | <0.001 | 0.321 | 0.178 | <0.001 | 0.778 | 0.119 | ||
| Cp | r | 0.125 | 0.016 | −0.208 | −0.452 | −0.032 | −0.014 | |
| p | 0.204 | 0.872 | 0.033 | <0.001 | 0.746 | 0.889 | ||
| Transferrin | r | 0.014 | 0.350 | 0.169 | 0.028 | −0.032 | 0.103 | |
| p | 0.890 | <0.001 | 0.087 | 0.778 | 0.746 | 0.295 | ||
| Ferritin | r | −0.237 | 0.241 | −0.088 | −0.153 | −0.014 | 0.103 | |
| p | 0.016 | 0.014 | 0.376 | 0.119 | 0.889 | 0.295 |
| Copper | Iron | Zinc | Cp-ferrox | Cp | Transferrin | Ferritin | ||
|---|---|---|---|---|---|---|---|---|
| Copper | r | 0.114 | 0.358 | 0.572 | 0.235 | −0.046 | 0.069 | |
| p | 0.251 | <0.001 | <0.001 | 0.016 | 0.642 | 0.492 | ||
| Iron | r | 0.114 | 0.204 | 0.040 | −0.032 | 0.445 | 0.049 | |
| p | 0.251 | 0.036 | 0.687 | 0.745 | <0.001 | 0.624 | ||
| Zinc | r | 0.358 | 0.204 | 0.309 | 0.173 | 0.191 | −0.091 | |
| p | <0.001 | 0.036 | 0.687 | 0.078 | 0.052 | 0.360 | ||
| Cp-ferrox | r | 0.572 | 0.040 | 0.309 | 0.187 | 0.093 | 0.041 | |
| p | <0.001 | 0.687 | 0.687 | 0.056 | 0.346 | 0.676 | ||
| Cp | r | 0.235 | −0.032 | 0.173 | 0.187 | 0.017 | 0.143 | |
| p | 0.016 | 0.745 | 0.078 | 0.056 | 0.864 | 0.147 | ||
| Transferrin | r | −0.046 | 0.445 | 0.191 | 0.093 | 0.017 | 0.045 | |
| p | 0.642 | <0.001 | 0.052 | 0.346 | 0.864 | 0.648 | ||
| Ferritin | r | 0.069 | 0.049 | −0.091 | 0.041 | 0.143 | 0.045 | |
| p | 0.492 | 0.624 | 0.360 | 0.676 | 0.147 | 0.648 |
| PostprandialTG | TG AUC | Fasting apoCIII | Postprandial apoCIII | Glucose Amplitude | Insulin Amplitude | Lactate Amplitude | ||
|---|---|---|---|---|---|---|---|---|
| Copper | r | −0.001 | −0.069 | −0.250 | −0.053 | 0.028 | −0.116 | −0.026 |
| p | 0.994 | 0.504 | 0.011 | 0.606 | 0.788 | 0.261 | 0.800 | |
| Iron | r | 0.029 | −0.059 | −0.165 | −0.142 | 0.066 | 0.069 | 0.074 |
| p | 0.777 | 0.565 | 0.109 | 0.169 | 0.525 | 0.504 | 0.473 | |
| Zinc | r | 0.010 | −0.061 | 0.023 | −0.036 | 0.059 | 0.080 | 0.150 |
| p | 0.922 | 0.555 | 0.823 | 0.728 | 0.566 | 0.436 | 0.144 | |
| Cp-ferrox | r | 0.010 | 0.095 | −0.089 | 0.0172 | −0.260 | −0.270 | −0.300 |
| p | 0.334 | 0.359 | 0.389 | 0.868 | 0.007 | 0.007 | 0.003 | |
| Cp | r | −0.056 | −0.060 | 0.050 | 0.005 | 0.085 | −0.077 | −0.121 |
| p | 0.586 | 0.559 | 0.630 | 0.959 | 0.411 | 0.457 | 0.240 | |
| Transferrin | r | −0.047 | −0.069 | −0.078 | −0.200 | −0.104 | −0.059 | 0.007 |
| p | 0.651 | 0.507 | 0.451 | 0.046 | 0.312 | 0.570 | 0.947 | |
| Ferritin | r | −0.220 | −0.230 | −0.290 | −0.290 | −0.210 | 0.096 | −0.21 |
| p | 0.025 | 0.021 | 0.003 | 0.003 | 0.934 | 0.351 | 0.039 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harder, N.H.O.; Hieronimus, B.; Stanhope, K.L.; Shibata, N.M.; Lee, V.; Nunez, M.V.; Keim, N.L.; Bremer, A.; Havel, P.J.; Heffern, M.C.; et al. Effects of Dietary Glucose and Fructose on Copper, Iron, and Zinc Metabolism Parameters in Humans. Nutrients 2020, 12, 2581. https://doi.org/10.3390/nu12092581
Harder NHO, Hieronimus B, Stanhope KL, Shibata NM, Lee V, Nunez MV, Keim NL, Bremer A, Havel PJ, Heffern MC, et al. Effects of Dietary Glucose and Fructose on Copper, Iron, and Zinc Metabolism Parameters in Humans. Nutrients. 2020; 12(9):2581. https://doi.org/10.3390/nu12092581
Chicago/Turabian StyleHarder, Nathaniel H. O., Bettina Hieronimus, Kimber L. Stanhope, Noreene M. Shibata, Vivien Lee, Marinelle V. Nunez, Nancy L. Keim, Andrew Bremer, Peter J. Havel, Marie C. Heffern, and et al. 2020. "Effects of Dietary Glucose and Fructose on Copper, Iron, and Zinc Metabolism Parameters in Humans" Nutrients 12, no. 9: 2581. https://doi.org/10.3390/nu12092581
APA StyleHarder, N. H. O., Hieronimus, B., Stanhope, K. L., Shibata, N. M., Lee, V., Nunez, M. V., Keim, N. L., Bremer, A., Havel, P. J., Heffern, M. C., & Medici, V. (2020). Effects of Dietary Glucose and Fructose on Copper, Iron, and Zinc Metabolism Parameters in Humans. Nutrients, 12(9), 2581. https://doi.org/10.3390/nu12092581







