Dietary Fibre Consensus from the International Carbohydrate Quality Consortium (ICQC)
Abstract
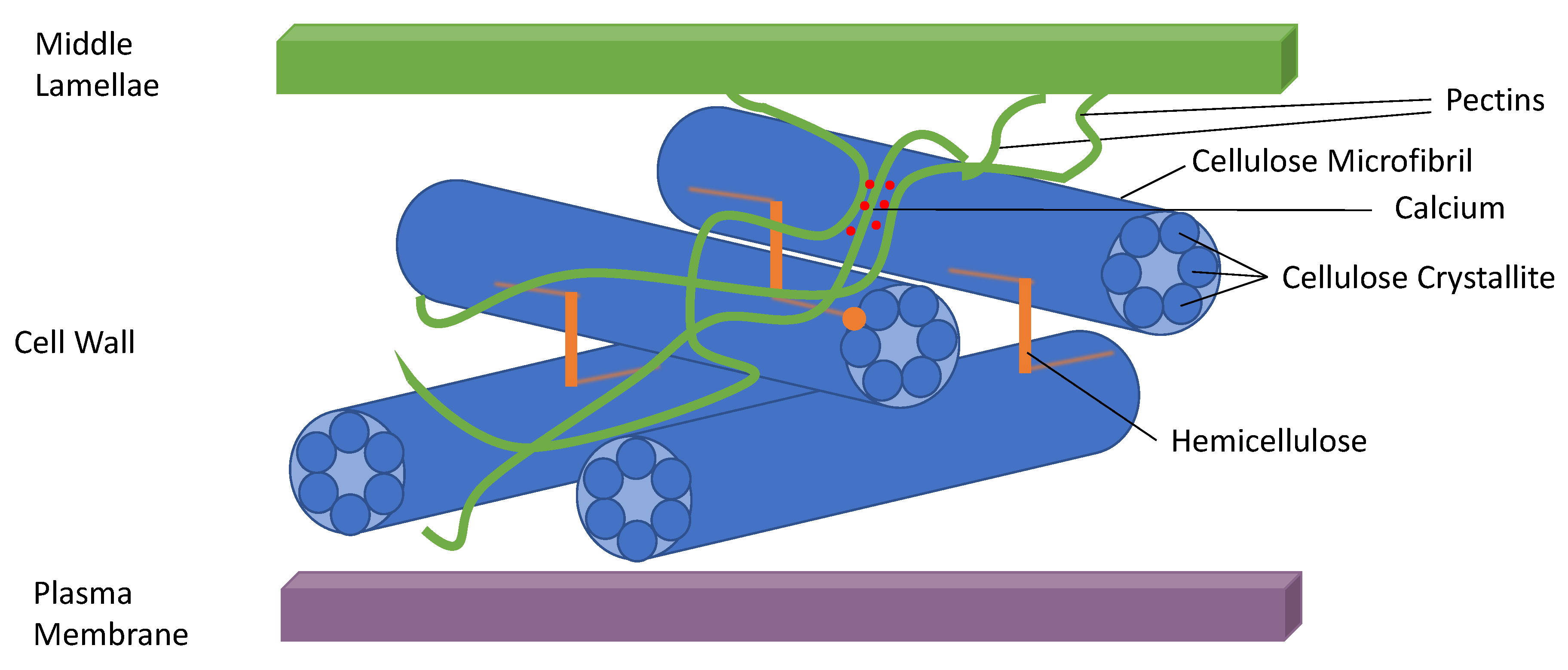
1. Introduction
2. Definitions
3. Health Benefits
4. Recommendations to the Public and to Health Professionals
5. Recommendations to the Food Industry
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burkitt, D.P.; Trowell, H.C. Refined Carbohydrate Food and Disease; Academic Press: London, UK, 1975. [Google Scholar]
- Trowell, H.C.; Burkitt, D.P. The development of the concept of dietary fibre. Mol. Asp. Med. 1987, 9, 7–15. [Google Scholar] [CrossRef]
- Ha, M.A.; Jarvis, M.C.; Mann, J.I. A definition for dietary fibre. Eur. J. Clin. Nutr. 2000, 54, 861–864. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jarvis, M.C. Plant cell walls: Supramolecular assemblies. Food Hydrocoll. 2011, 25, 257–262. [Google Scholar] [CrossRef]
- Grundy, M.M.; Edwards, C.H.; Mackie, A.R.; Gidley, M.J.; Butterworth, P.J.; Ellis, P.R. Re-evaluation of the mechanisms of dietary fibre and implications for macronutrient bioaccessibility, digestion and postprandial metabolism. Br. J. Nutr. 2016, 116, 816–833. [Google Scholar] [CrossRef] [PubMed]
- Rastall, R.F.; Javier Moreno, F.J.; Hernandez-Hernandez, O. Dietary carbohydrate digestibility and metabolic effects in human health. Front. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Food Standards Australia. Available online: http://www.foodstandards.gov.au/code/applications/Documents/A277%20IR(FULL).pdf (accessed on 21 August 2020).
- FAO. Food and Agriculture Organization of the United Nations, Codex Alimentarius Commission FAO/WHO Distribution of the Report of the 30th Session of the Codex Committee on Nutrition and Foods for Special Dietary Uses; (ALINORM 09/32/26); FAO: Rome, Italy, 2009. [Google Scholar]
- Scientific Advisory Committee on Nutrition (SACN) 2015. Carbohydrates and Health; London TSO, TSO Norwich NR3 1GN, UK. Available online: https://www.gov.uk/government/publications/sacn-carbohydrates-and-health-report (accessed on 21 August 2020).
- Jones, J.M. CODEX-aligned dietary fiber definitions help to bridge the ‘fiber gap’. Nutr. J. 2014, 13, 34. [Google Scholar] [CrossRef]
- FDA. 2018. The Declaration of Certain Isolated or Synthetic Non-Digestible Carbohydrates as Dietary Fiber on Nutrition and Supplement Facts Labels: Guidance for Industry. Available online: https://www.fda.gov/media/113663/download (accessed on 21 August 2020).
- Jenkins, D.J.; Kendall, C.W.; Axelsen, M.; Augustin, L.S.; Vuksan, V. Viscous and nonviscous fibres, nonabsorbable and low glycaemic index carbohydrates, blood lipids and coronary heart disease. Curr. Opin. Lipidol. 2000, 11, 49–56. [Google Scholar] [CrossRef]
- Capuano, E. The behavior of dietary fiber in the gastrointestinal tract determines its physiological effect. Crit. Rev. Food Sci. Nutr. 2017, 57, 3543–3564. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Leeds, A.R.; Gassull, M.A.; Haisman, P.; Dilawari, J.; Goff, D.V.; Metz, G.L.; Alberti, K.G. Dietary fibres, fibre analogues, and glucose tolerance: Importance of viscosity. Br. Med. J. 1978, 1, 1392–1394. [Google Scholar] [CrossRef]
- Liu, F.; Prabhakar, M.; Ju, J.; Long, H.; Zhou, H.W. Effect of inulin-type fructans on blood lipid profile and glucose level: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2017, 71, 9–20. [Google Scholar] [CrossRef]
- Livesey, G.; Tagami, H. Interventions to lower the glycemic response to carbohydrate foods with a low-viscosity fiber (resistant maltodextrin): Meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2009, 89, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Livesey, G.; Taylor, R.; Hulshof, T.; Howlett, J. Glycemic response and health a systematic review and meta-analysis: Relations between dietary glycemic properties and health outcomes. Am. J. Clin. Nutr. 2008, 87, 258S–268S. [Google Scholar] [CrossRef] [PubMed]
- Salmeron, J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Wing, A.L.; Willett, W.C. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. JAMA 1997, 277, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Ding, M.; Sampson, L.; Willett, W.C.; Manson, J.E.; Wang, M.; Rosner, B.; Hu, F.B.; Sun, Q. Intake of whole grain foods and risk of type 2 diabetes: Results from three prospective cohort studies. BMJ 2020, 370, m2206. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Augustin, L.S.; Martini, M.C.; Axelsen, M.; Faulkner, D.; Vidgen, E.; Parker, T.; Lau, H.; Connelly, P.W.; et al. Effect of wheat bran on glycemic control and risk factors for cardiovascular disease in type 2 diabetes. Diabetes Care 2002, 25, 1522–1528. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Kendall, C.W.; McKeown-Eyssen, G.; Josse, R.G.; Silverberg, J.; Booth, G.L.; Vidgen, E.; Josse, A.R.; Nguyen, T.H.; Corrigan, S.; et al. Effect of a low-glycemic index or a high-cereal fiber diet on type 2 diabetes: A randomized trial. JAMA 2008, 300, 2742–2753. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Jones, P.J.; Lamarche, B.; Kendall, C.W.; Faulkner, D.; Cermakova, L.; Gigleux, I.; Ramprasath, V.; de Souza, R.; Ireland, C.; et al. Effect of a dietary portfolio of cholesterol-lowering foods given at 2 levels of intensity of dietary advice on serum lipids in hyperlipidemia: A randomized controlled trial. JAMA 2011, 306, 831–839. [Google Scholar] [CrossRef]
- Edwards, C.H.; Grundy, M.M.L.; Grassby, T.; Vasilopoulou, D.; Frost, G.S.; Butterworth, P.J.; Berry, S.E.E.; Sanderson, J.; Ellis, P.R. Manipulation of starch bioaccessibility in wheat endosperm to regulate starch digestion, postprandial glycemia, insulinemia, and gut hormone responses: A randomized controlled trial in healthy ileostomy participants. Am. J. Clin. Nutr. 2015, 102, 791–800. [Google Scholar] [CrossRef]
- Livesey, G.; Wilkinson, J.A.; Roe, M.; Faulks, R.; Clark, S.; Brown, J.C.; Kennedy, H.; Elia, M. Influence of the physical form of barley grain on the digestion of its starch in the human small intestine and implications for health. Am. J. Clin. Nutr. 1995, 61, 75–81. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wesson, V.; Wolever, T.M.; Jenkins, A.L.; Kalmusky, J.; Guidici, S.; Csima, A.; Josse, R.G.; Wong, G.S. Wholemeal versus wholegrain breads: Proportion of whole or cracked grain and the glycaemic response. BMJ 1988, 297, 958–960. [Google Scholar] [CrossRef]
- Wolever, T.M.; Schrade, K.B.; Vogt, J.A.; Tsihlias, E.B.; McBurney, M.I. Do colonic short-chain fatty acids contribute to the long-term adaptation of blood lipids in subjects with type 2 diabetes consuming a high-fiber diet? Am. J. Clin. Nutr. 2002, 75, 1023–1030. [Google Scholar] [CrossRef]
- Augustin, L.S.; Kendall, C.W.; Jenkins, D.J.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef]
- Aslam, M.F.; Ellis, P.; Berry, S.E.; Latunde-Dada, G.O.; Sharp, P.A. Enhancing mineral bioavailability from cereals: Current strategies and future perspectives. Nutr. Bull. 2018, 43, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Delcour, J.A.; Aman, P.; Courtin, C.M.; Hamaker, B.R.; Verbeke, K. Prebiotics, Fermentable Dietary Fiber, and Health Claims. Adv. Nutr. 2016, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Stanton, C.; Lane, J.A.; Smith, G.J.; Ross, R.P. Precision Nutrition and the Microbiome, Part I: Current State of the Science. Nutrients 2019, 11, 923. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Lane, J.A.; Smith, G.J.; Grimaldi, K.A.; Ross, R.P.; Stanton, C. Precision Nutrition and the Microbiome Part II: Potential Opportunities and Pathways to Commercialisation. Nutrients 2019, 11, 1468. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Willett, W.C.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Augustin, L.S.A.; Aas, A.-M.; Astrup, A.; Atkinson, F.S.; Baer-Sinnott, S.; Barclay, A.W.; Brand-Miller, J.C.; Brighenti, F.; Bullo, M.; Buyken, A.E.; et al. Dietary Fibre Consensus from the International Carbohydrate Quality Consortium (ICQC). Nutrients 2020, 12, 2553. https://doi.org/10.3390/nu12092553
Augustin LSA, Aas A-M, Astrup A, Atkinson FS, Baer-Sinnott S, Barclay AW, Brand-Miller JC, Brighenti F, Bullo M, Buyken AE, et al. Dietary Fibre Consensus from the International Carbohydrate Quality Consortium (ICQC). Nutrients. 2020; 12(9):2553. https://doi.org/10.3390/nu12092553
Chicago/Turabian StyleAugustin, Livia S. A., Anne-Marie Aas, Arnie Astrup, Fiona S. Atkinson, Sara Baer-Sinnott, Alan W. Barclay, Jennie C. Brand-Miller, Furio Brighenti, Monica Bullo, Anette E. Buyken, and et al. 2020. "Dietary Fibre Consensus from the International Carbohydrate Quality Consortium (ICQC)" Nutrients 12, no. 9: 2553. https://doi.org/10.3390/nu12092553
APA StyleAugustin, L. S. A., Aas, A.-M., Astrup, A., Atkinson, F. S., Baer-Sinnott, S., Barclay, A. W., Brand-Miller, J. C., Brighenti, F., Bullo, M., Buyken, A. E., Ceriello, A., Ellis, P. R., Ha, M.-A., Henry, J. C., Kendall, C. W. C., La Vecchia, C., Liu, S., Livesey, G., Poli, A., ... Jenkins, D. J. A. (2020). Dietary Fibre Consensus from the International Carbohydrate Quality Consortium (ICQC). Nutrients, 12(9), 2553. https://doi.org/10.3390/nu12092553














