The Environmental Impact of the Athlete’s Plate Nutrition Education Tool
Abstract
1. Introduction
2. Materials and Methods
2.1. Life Cycle Assessment of the Athlete’s Plate
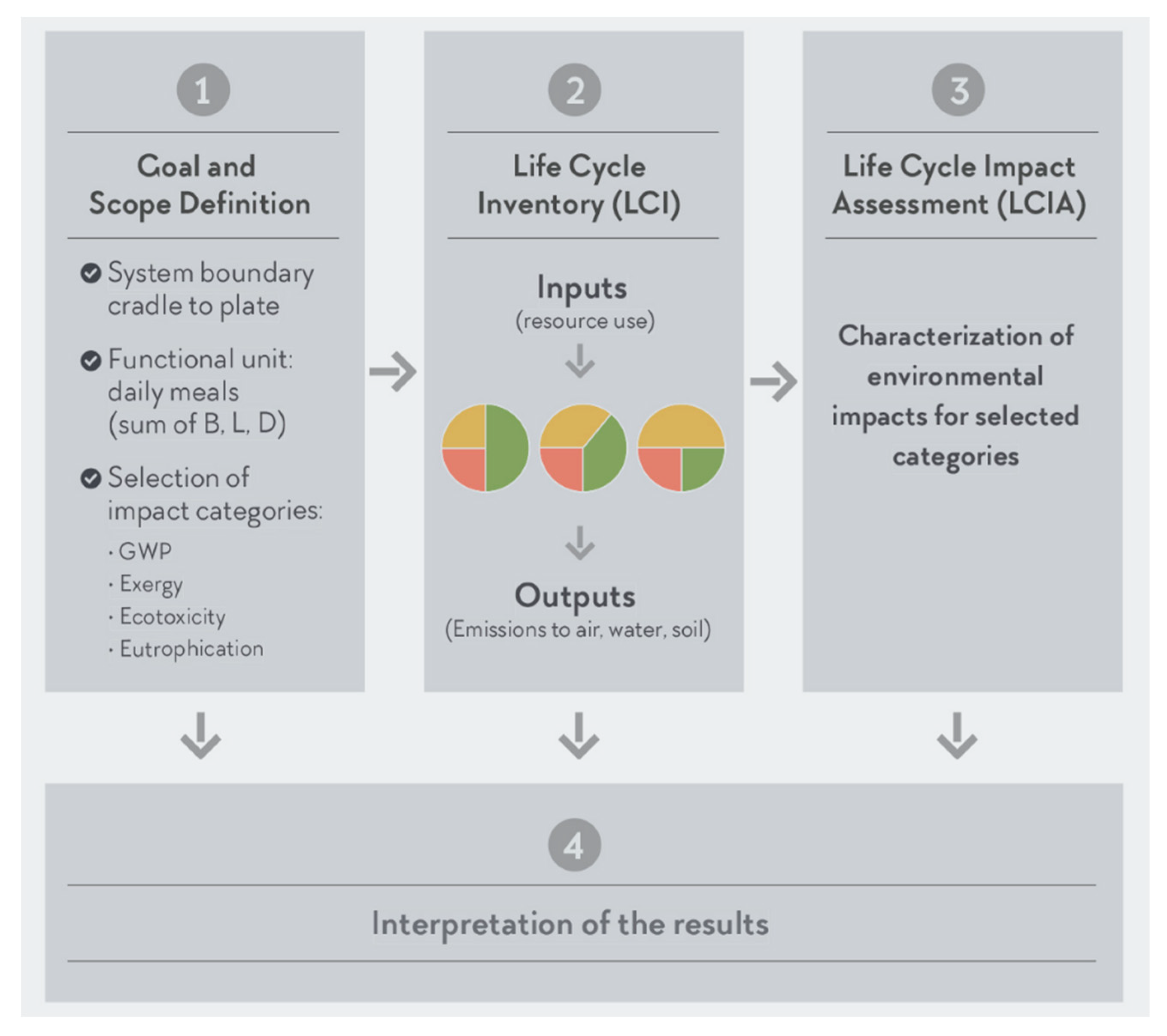
2.1.1. Goal and Scope Definition
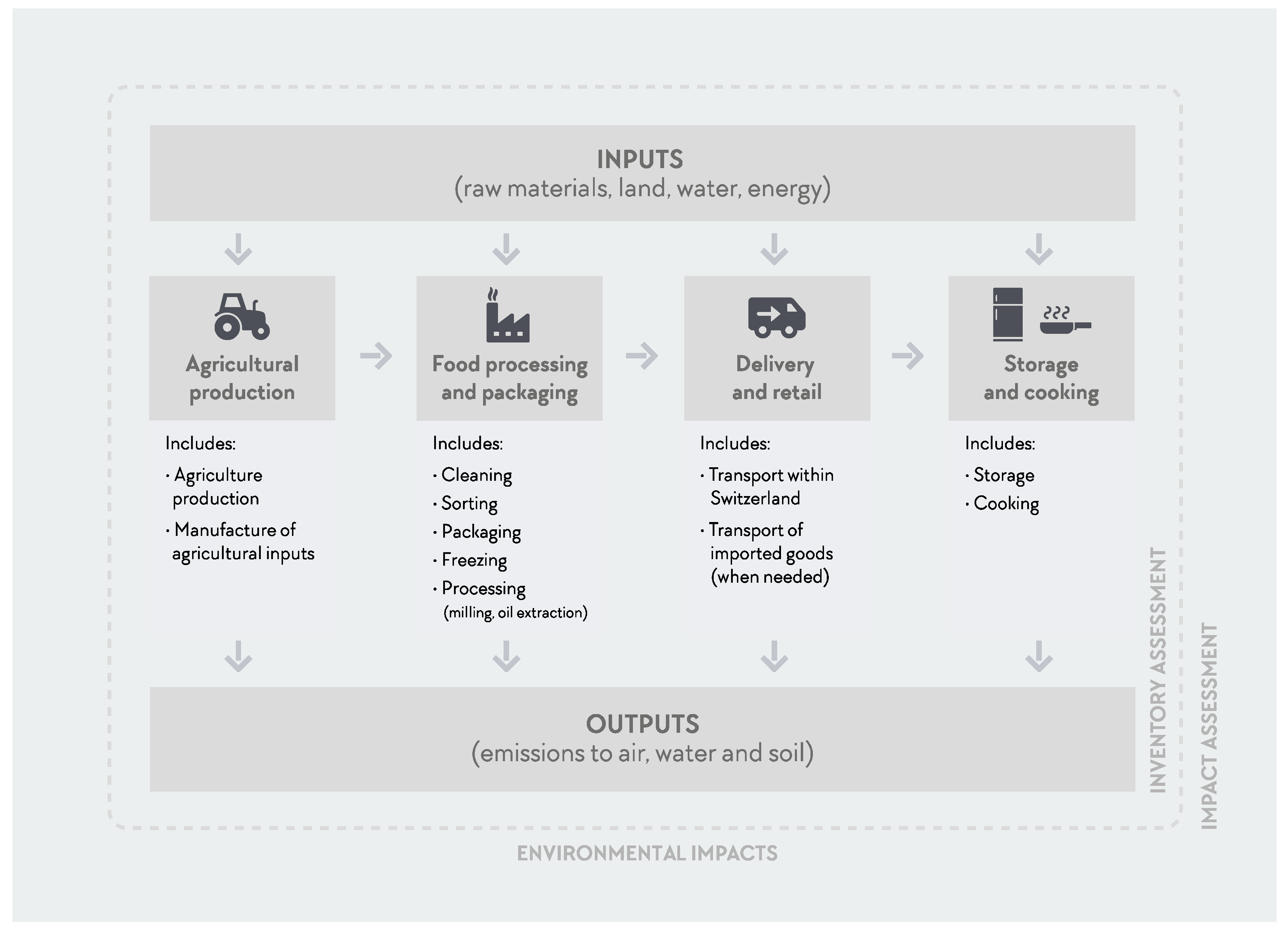
2.1.2. Life Cycle Inventory (LCI)
2.1.3. Life Cycle Impact Assessment (LCIA)
2.1.4. Statistical Analysis
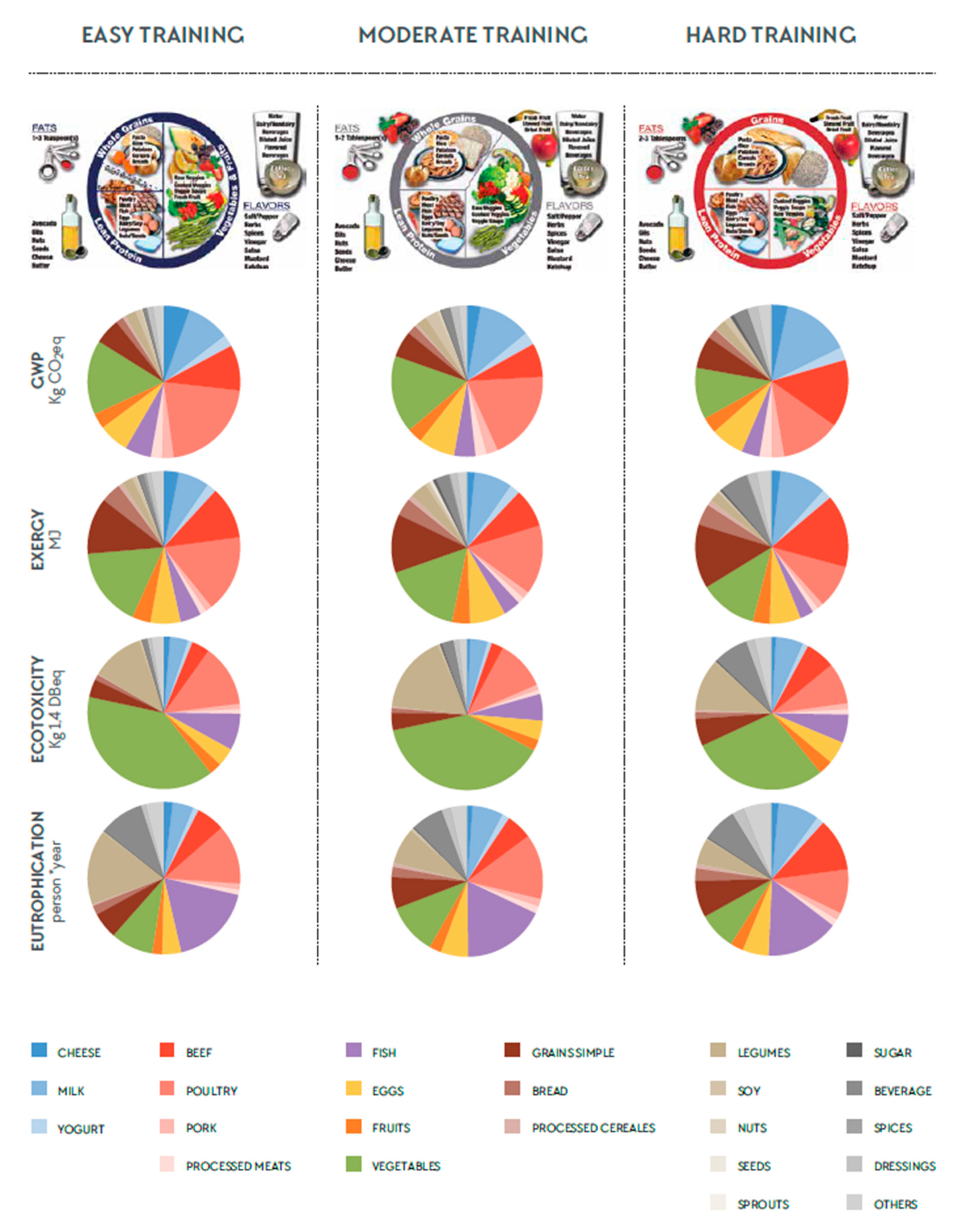
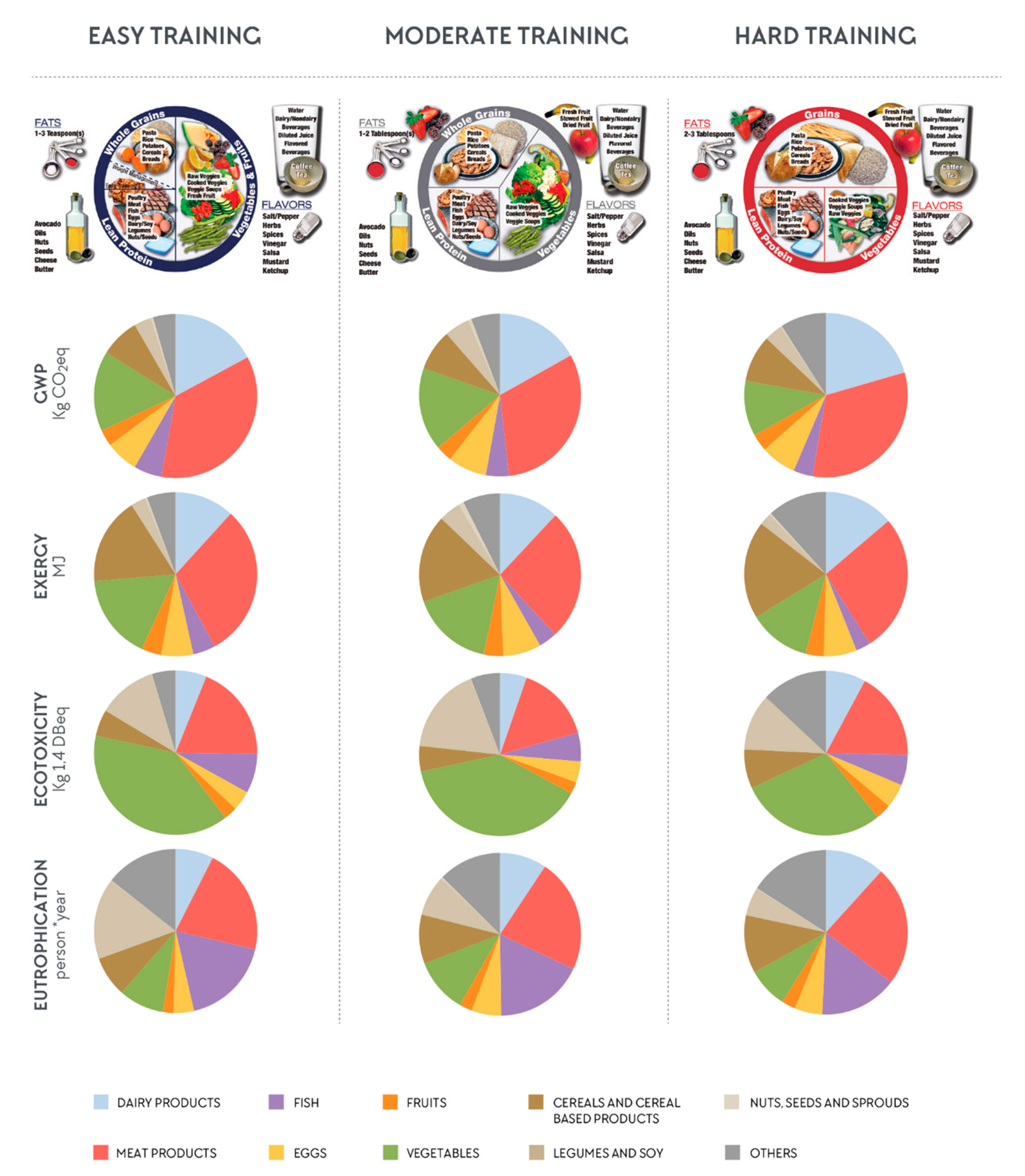
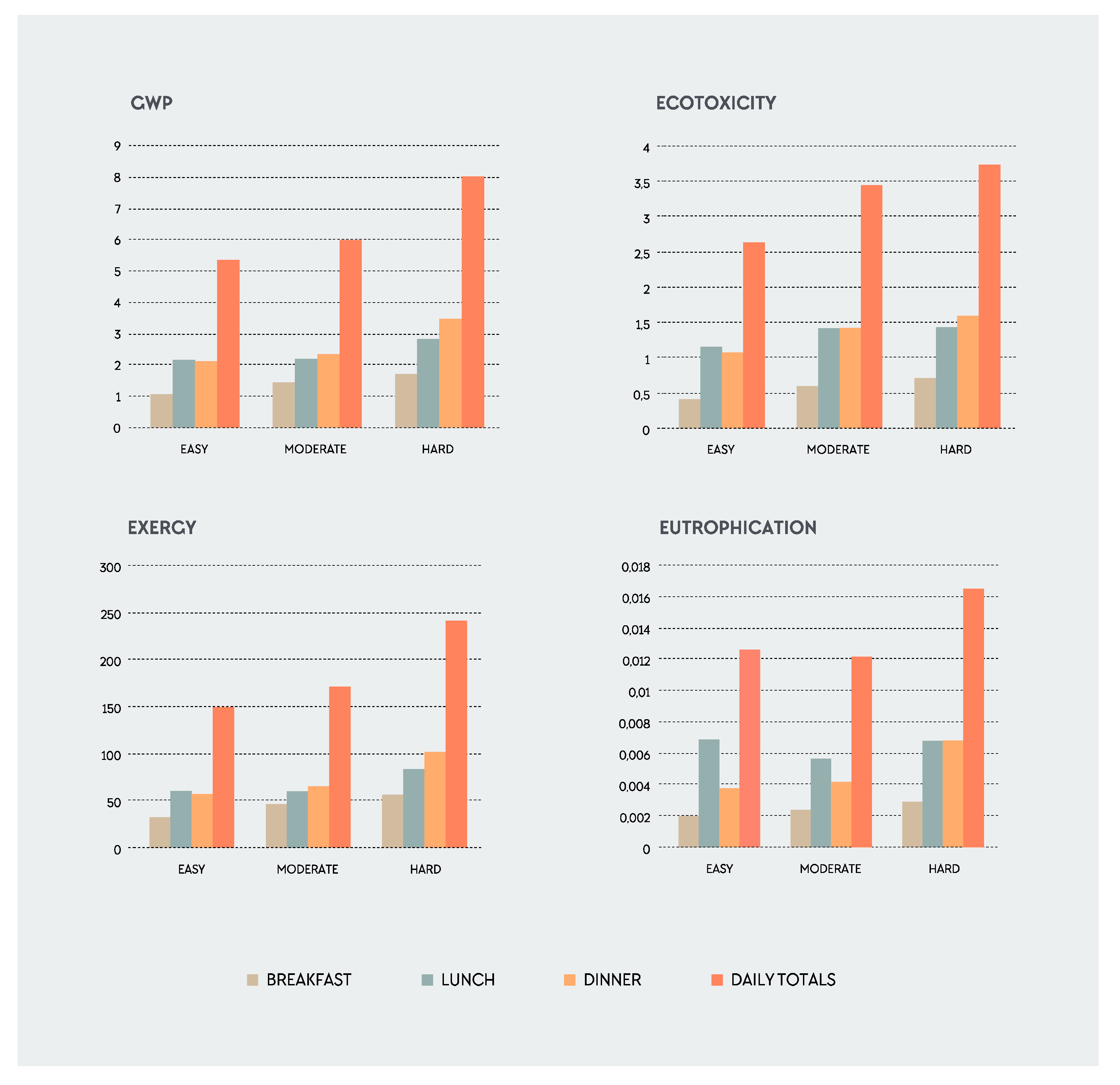
3. Results
3.1. Descriptive Data
3.2. The Influence of Different Foods on the Total Environmental Impact
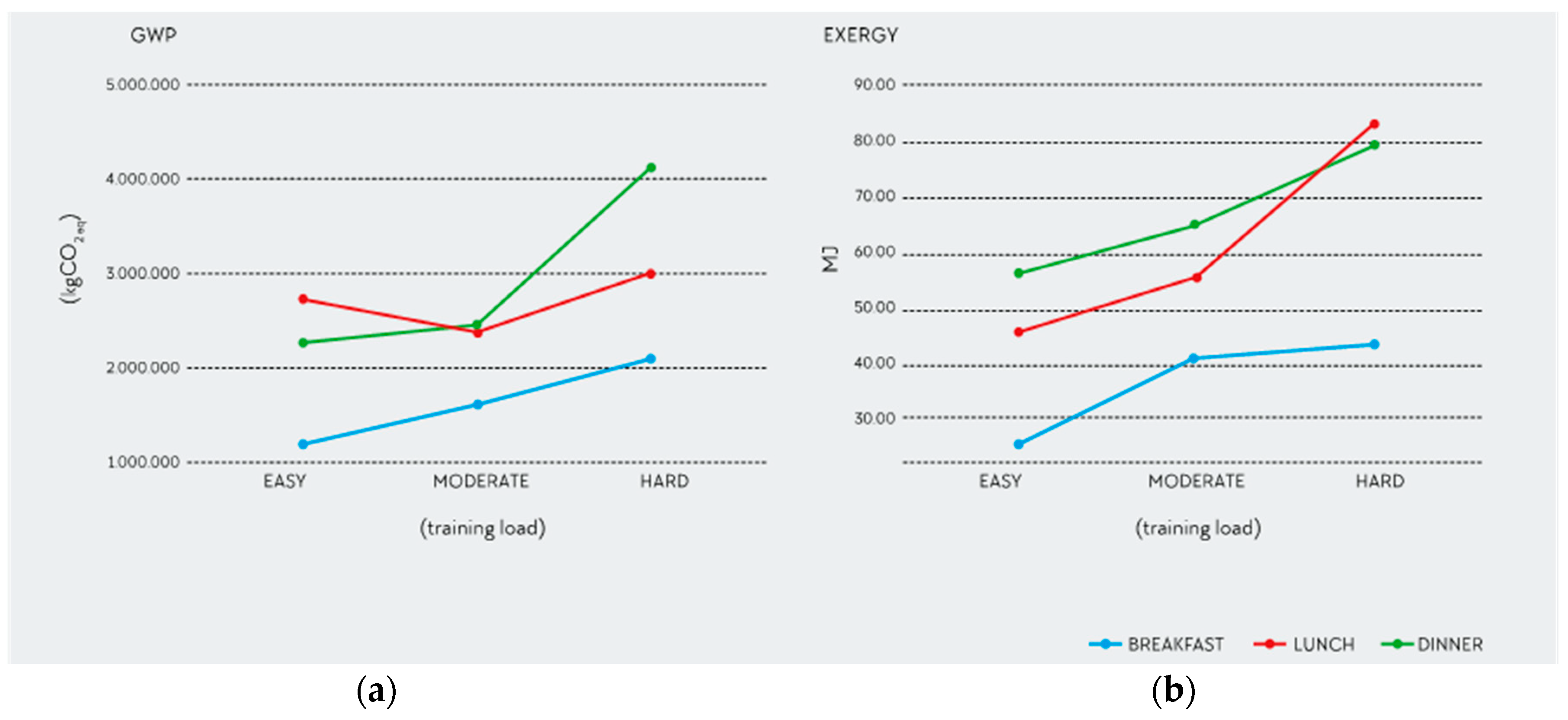
3.3. Sources of Variation among RDs
3.4. Outliers and Consistency of the Data
4. Discussion
4.1. Descriptive Data
4.2. The Influence of Different Foods on the Total Environmental Impact
4.3. Sources of Variation among RDs
4.4. Outliers and Consistency of the Data
4.5. Limitations
4.6. Recommendations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| AGGREGATION OF FOODS | GROUPS INCLUDED | SUBGROUPS |
|---|---|---|
| DAIRY | Milk products | - |
| Yogurt | - | |
| Cheese products | Soft cheese and hard cheese | |
| MEAT | Beef | - |
| Poultry | - | |
| Pork | - | |
| Processed meats | - | |
| FISH | Fish | - |
| EGGS | Egg | - |
| VEGETABLES | Fresh vegetables | Specific inventories for vegetables where created when possible |
| Canned vegetables | Specific inventories for vegetables where created when possible | |
| Frozen vegetables | - | |
| FRUITS | Fresh fruits | Specific inventories for vegetables where created when possible |
| Frozen fruits | Specific inventories for vegetables where created when possible | |
| GRAINS | Grain simple | Includes pasta, rice, bulgur, oats, etc. |
| Bread | - | |
| Processed cereals | Includes breakfast cereals with sugar and processing | |
| LEGUMES | Legumes without soy canned | - |
| Soy beans | - | |
| Tofu | - | |
| NUTS | Nuts in general | - |
| Peanuts | - | |
| Peanut butter | - | |
| SEEDS | Seeds | - |
| SPROUDS | Sprouts | - |
| SUGAR | Honey | - |
| Sugar | - | |
| BEVERAGES | Bottled fruit juice | - |
| Wine | - | |
| Sports drinks | - | |
| SPICES | Spices | - |
| DRESSINGS | Olive oil vinaigrette dressing | - |
| Mayonnaise based dressing | - | |
| OTHERS | Others | - |
| STAGE | ASSUMPTIONS |
|---|---|
| Agriculture production |
|
| Processing |
Due to the difficulty of accounting of all the individualities of processing steps due to the wide range of ingredients considered for this study, some assumption were considered on this life-cycle stage:
|
| Packaging |
|
| Transport |
|
| Storage |
|
| Cooking |
|
| GWP (kg CO2 eq) | Exergy (MJ) | Ecotoxicity (kg 1.4DB eq) | Eutrophication (person × year) | |
|---|---|---|---|---|
| GWP | 1 | 0.86 | 0.69 | 0.26 |
| Exergy | 0.86 | 1 | 0.45 | 0.28 |
| Ecotoxicity | 0.69 | 0.45 | 1 | 0.44 |
| Eutrophication | 0.26 | 0.28 | 0.44 | 1 |
References
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Notarnicola, B.; Sala, S.; Anton, A.O.; Mclaren, S.J.; Saouter, E.; Sonesson, U. The role of life cycle assessment in supporting sustainable agri-food systems: A review of the challenges. J. Clean. Prod. 2016. [Google Scholar] [CrossRef]
- Ridoutt, B.G.; Hendrie, G.A.; Noakes, M. Dietary strategies to reduce environmental impact: A critical review of the evidence. Adv. Nutr. 2017, 8, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, S.J.; Campbell, B.M.; Ingram, J.S.I. Climate Change and Food Systems. Annu. Rev. Environ. Resour. 2012, 37, 195–222. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013. [Google Scholar]
- Sabaté, J.; Sranacharoenpong, K.; Harwatt, H.; Wien, M.; Soret, S. The environmental cost of protein food choices. Public Health Nutr. 2014, 18, 1–7. [Google Scholar] [CrossRef]
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT–Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Soret, S.; Sabate, J. Sustainability of plant-based diets: Back to the future. Am. J. Clin. Nutr. 2014, 100, 476–482. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Hall, K.D.; McPherson, K.; Finegood, D.T.; Moodie, M.L.; Gortmaker, S.L. The global obesity pandemic: Shaped by global drivers and local environments. Lancet 2011, 378, 804–814. [Google Scholar] [CrossRef]
- Tilman, D.; Clark, M. Global diets link environmental sustainability and human health. Nature 2014, 515, 518–522. [Google Scholar] [CrossRef]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K.; Alaejos, M.; et al. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Kraak, V.I.; Allender, S.; Atkins, V.J.; Baker, P.I.; Bogard, J.R.; Brinsden, H.; Calvillo, A.; De Schutter, O.; Devarajan, R.; et al. The Global Syndemic of Obesity, Undernutrition, and Climate Change: The Lancet Commission report. Lancet 2019, 393, 791–846. [Google Scholar] [CrossRef]
- Guarnieri, M.; Balmes, J.R. Outdoor Air Pollution and Asthma. Lancet 2014, 383. [Google Scholar] [CrossRef]
- Manisalidis, I.; Stavropoulou, E.; Stavropoulos, A.; Bezirtzoglou, E. Environmental and Health Impacts of Air Pollution: A Review. Front. Public Health 2020, 8, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Nemecek, T.; Jungbluth, N.; Canals, L.M.; Schenck, R. Environmental impacts of food consumption and nutrition: Where are we and what is next? Int. J. Life Cycle Assess. 2016, 607–620. [Google Scholar] [CrossRef]
- Batlle-Bayer, L.; Bala, A.; García-Herrero, I.; Lemaire, E.; Song, G.; Aldaco, R.; Fullana-i-Palmer, P. The Spanish Dietary Guidelines: A potential tool to reduce greenhouse gas emissions of current dietary patterns. J. Clean. Prod. 2019, 213, 588–598. [Google Scholar] [CrossRef]
- Castañé, S.; Antón, A. Assessment of the nutritional quality and environmental impact of two food diets: A Mediterranean and a vegan diet. J. Clean. Prod. 2018, 167, 929–937. [Google Scholar] [CrossRef]
- van Dooren, C.; Aiking, H. Defining a nutritionally healthy, environmentally friendly, and culturally acceptable Low Lands Diet. In Proceedings of the 9th International Conference on Life Cycle Assessment in the Agri-Food Sector (LCA Food 2014), San Francisco, CA, USA, 8–10 October 2014. [Google Scholar]
- Scarborough, P.; Appleby, P.N.; Mizdrak, A.; Briggs, A.D.M.; Travis, R.C.; Bradbury, K.E.; Key, T.J. Dietary greenhouse gas emissions of meat-eaters, fish-eaters, vegetarians and vegans in the UK. Clim. Chang. 2014, 125, 179–192. [Google Scholar] [CrossRef]
- Masset, G.; Soler, L.G.; Vieux, F.; Darmon, N. Identifying sustainable foods: The relationship between environmental impact, nutritional quality, and prices of foods representative of the french diet. J. Acad. Nutr. Diet. 2014, 114, 862–869. [Google Scholar] [CrossRef]
- Ripple, W.J.; Smith, P.; Haberl, H.; Montzka, S.A.; McAlpine, C.; Boucher, D.H. Ruminants, climate change and climate policy. Nat. Clim. Chang. 2014, 4, 2–5. [Google Scholar] [CrossRef]
- Soret, S.; Mejia, A.; Batech, M.; Jaceldo-Siegl, K.; Harwatt, H.; Sabaté, J. Climate change mitigation and health effects of varied dietary patterns in real-life settings throughout North America. Am. J. Clin. Nutr. 2014, 100, 490–495. [Google Scholar] [CrossRef]
- Westhoek, H.; Lesschen, J.P.; Rood, T.; Wagner, S.; De Marco, A.; Murphy-Bokern, D.; Leip, A.; van Grinsven, H.; Sutton, M.A.; Oenema, O. Food choices, health and environment: Effects of cutting Europe’s meat and dairy intake. Glob. Environ. Chang. 2014, 26, 196–205. [Google Scholar] [CrossRef]
- Castellani, V.; Sala, S.; Benini, L. Hotspots analysis and critical interpretation of food life cycle assessment studies for selecting eco-innovation options and for policy support. J. Clean. Prod. 2017, 140, 556–568. [Google Scholar] [CrossRef]
- FAO. Sustainable Diets and Biodiversity; Food and Agriculture Organisation of the United Nations (FAO): Rome, Italy, 2010. [Google Scholar] [CrossRef]
- Steffen, W.; Richardson, K.; Rockström, J.; Cornell, S.E.; Fetzer, I.; Bennett, E.M.; Biggs, R.; Carpenter, S.R.; De Vries, W.; De Wit, C.A.; et al. Planetary boundaries: Guiding human development on a changing planet. Science 2015, 347, 1259855. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. American College of Sports Medicine Joint Position Statement. Nutrition and Athletic Performance. Med. Sci. Sports Exerc. 2016, 48, 543–568. [Google Scholar] [CrossRef] [PubMed]
- Daly, R.M.; Connell, S.L.O.; Mundell, N.L.; Grimes, C.A.; Dunstan, D.W.; Nowson, C.A. Protein-enriched diet, with the use of lean red meat, combined with progressive resistance training enhances lean tissue mass and muscle strength and reduces circulating IL-6 concentrations in elderly women: A cluster randomized controlled trial 1. Am. J. Clin. Nutr. 2014, 99, 899–910. [Google Scholar] [CrossRef]
- Nowson, C.; O’Connell, S. Protein Requirements and Recommendations for Older People: A Review. Nutrients 2015, 7, 6874–6899. [Google Scholar] [CrossRef]
- Te Morenga, L.; Mann, J. The role of high-protein diets in body weight management and health. Br. J. Nutr. 2012, 108, S130–S138. [Google Scholar] [CrossRef]
- Garnett, T. Plating up solutions. Science 2016, 353, 1202–1204. [Google Scholar] [CrossRef]
- Lynch, H.; Johnston, C.; Wharton, C. Plant-based diets: Considerations for environmental impact, protein quality, and exercise performance. Nutrients 2018, 10, 1841. [Google Scholar] [CrossRef]
- Meyer, N.; Reguant-closa, A. “Eat as If You Could Save the Planet and Win !” Sustainability Integration into Nutrition for Exercise. Nutrients 2017, 9, 412. [Google Scholar] [CrossRef]
- Foster, C.; Florhaug, J.A.; Franklin, J.; Gottschall, L.; Hrovatin, L.A.; Parker, S.; Doleshal, P.; Dodge, C. A New Approach to Monitoring Exercise Training. J. Strength Cond. Res. 2001, 15, 109–115. [Google Scholar] [PubMed]
- Reguant-Closa, A.; Harris, M.M.; Lohman, T.G.; Meyer, N.L. Validation of the Athlete’s Plate Nutrition Educational Tool: Phase I. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Churchward-Venne, T.A.; Witard, O.; Breen, L.; Burd, N.A.; Tipton, K.D.; Phillips, S.M. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; van Loon, L.J.C. Dietary protein for athletes: From requirements to optimum adaptation. J. Sports Sci. 2011, 29, S29–S38. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. A Brief Review of Higher Dietary Protein Diets in Weight Loss: A Focus on Athletes. Sport Med. 2014, 44, 149–153. [Google Scholar] [CrossRef]
- Spendlove, J.; Mitchell, L.; Gifford, J.; Hackett, D.; Slater, G.; Cobley, S.; O’Connor, H. Dietary Intake of Competitive Bodybuilders. Sports Med. 2015, 45, 1041–1063. [Google Scholar] [CrossRef]
- Burd, N.A.; Gorissen, S.H.; van Vliet, S.; Snijders, T.; van Loon, L.J. Differences in postprandial protein handling after beef compared with milk ingestion during postexercise recovery: A randomized controlled trial. Am. J. Clin. Nutr. 2015, 102, 828–836. [Google Scholar] [CrossRef]
- Babault, N.; Païzis, C.; Deley, G.; Guérin-Deremaux, L.; Saniez, M.-H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, Placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12, 3. [Google Scholar] [CrossRef]
- van Vliet, S.; Burd, N.A.; van Loon, L.J. The Skeletal Muscle Anabolic Response to Plant-versus Animal-Based Protein Consumption. J. Nutr. 2015. [Google Scholar] [CrossRef]
- Berryman, C.E.; Lieberman, H.R.; Fulgoni, V.L.; Pasiakos, S.M. Protein Intake Trends and Conformity With the Dietary Reference Intakes in the United States: Analysis of the National Health and Nutrition Examination Survey, 2001–2014. Am. J. Clin. Nutr. 2018, 108. [Google Scholar] [CrossRef]
- Meyer, N.L.; Reguant-Closa, A.; Nemecek, T. Sustainable Diets for Athletes. Curr. Nutr. Rep. 2020, 1–16. [Google Scholar] [CrossRef]
- ISO 14040:Environmental Management-Life Cycle Assessment-Principles and Framework; ISO (the International Organization for Standardization): Geneva, Switzerland, 2006.
- Gaillard, G.; Nemecek, T. Swiss agricultural life cycle assessment (SALCA): An integrated environmental assessment concept for agriclture. In Proceedings of the Int. Conf. “Integrated Environmental Assessment of Agriculture and Sustainable Development, Setting the Agenda for Science and Policy”, Egmon aan Zee, The Netherlands, 10–12 March 2009; pp. 134–135. [Google Scholar]
- Saarinen, M.; Fogelholm, M.; Tahvonen, R.; Kurppa, S. Taking nutrition into account within the life cycle assessment of food products. J. Clean. Prod. 2017, 149, 828–844. [Google Scholar] [CrossRef]
- Sonesson, U.; Davis, J.; Hallström, E.; Woodhouse, A. Dietary-dependent nutrient quality indexes as a complementary functional unit in LCA: A feasible option? J. Clean. Prod. 2019, 211, 620–627. [Google Scholar] [CrossRef]
- Van Kernebeek, H.R.J.; Oosting, S.J.; Feskens, E.J.M.; Gerber, P.J.; De Boer, I.J.M. The effect of nutritional quality on comparing environmental impacts of human diets. J. Clean. Prod. 2014, 73, 88–99. [Google Scholar] [CrossRef]
- Fulgoni, V.L.; Keast, D.R.; Drewnowski, A. Development and validation of the nutrient-rich foods index: A tool to measure nutritional quality of foods. J. Nutr. 2009, 139, 1549–1554. [Google Scholar] [CrossRef]
- Berardy, A.; Johnston, C.S.; Plukis, A.; Vizcaino, M.; Wharton, C. Integrating protein quality and quantity with environmental impacts in life cycle assessment. Sustainability 2019, 11, 2747. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Bösch, M.E.; Hellweg, S.; Huijbregts, M.A.J.; Frischknecht, R. Applying cumulative exergy demand (CExD) indicators to the ecoinvent database. Int. J. Life Cycle Assess. 2007, 12, 181–190. [Google Scholar] [CrossRef]
- Alvarenga, R.A.F.; Dewulf, J.; Van Langenhove, H.; Huijbregts, M.A.J. Exergy-based accounting for land as a natural resource in life cycle assessment. Int. J. Life Cycle Assess. 2013, 18, 939–947. [Google Scholar] [CrossRef]
- Guinée, J.; Gorrée, M.; Heijungs, R.; Huppes, G.; Klejin, R.; de Koning, A.; van Oers, L.; Wegener Sleeswijk, A.; Suh, S.; Udo de Haes, H.; et al. Life Cycle Assessment-An Operational Guide to the Iso Standards; Den Haag and Leiden: Leiden, The Netherlands, 2001. [Google Scholar] [CrossRef]
- Fantke, P.; Jolliet, O. Life cycle human health impacts of 875 pesticides. Int. J. Life Cycle Assess. 2016, 21, 722–733. [Google Scholar] [CrossRef]
- Hayer, F.; Bockstaller, C.; Gaillard, G.; Mamy, L.; Nemecek, T.; Strassemeyer, J. Multi-criteria comparison of eco-toxicity models focused on pesticides. In Proceedings of the 7th International Conference on Life Cycle Assessment in the Agri-Food Sector, Bari, Italy, 22–24 September 2010. [Google Scholar]
- Hauschild, M.; Pooting, J. Spatial Differentiation in Life Cycle Impact Assessment-the EDIP 2003 Methodology; Miljøstyrelsen: Copenhagen, Denmark, 2005. [Google Scholar]
- Batlle-Bayer, L.; Bala, A.; Lemaire, E.; Albertí, J.; García-Herrero, I.; Aldaco, R.; Fullana-i-Palmer, P. An energy- and nutrient-corrected functional unit to compare LCAs of diets. Sci. Total Environ. 2019, 671, 175–179. [Google Scholar] [CrossRef]
- Masset, G.; Vieux, F.; Darmon, N. Which functional unit to identify sustainable foods? Public Health Nutr. 2015, 18, 2488–2497. [Google Scholar] [CrossRef] [PubMed]
- Sonesson, U.; Davis, J.; Flysjö, A.; Gustavsson, J.; Witthöft, C. Protein quality as functional unit–A methodological framework for inclusion in life cycle assessment of food. J. Clean. Prod. 2017, 140, 470–478. [Google Scholar] [CrossRef]
- Burke, L.M.; Hawley, J.A.; Wong, S.H.S.; Jeukendrup, A.E. Carbohydrates for training and competition. J. Sports Sci. 2011, 29 (Suppl. 1), S17–S27. [Google Scholar] [CrossRef] [PubMed]
- Barnard, N.; Goldman, D.; Loomis, J.; Kahleova, H.; Levin, S.; Neabore, S.; Batts, T. Plant-Based Diets for Cardiovascular Safety and Performance in Endurance Sports. Nutrients 2019, 11, 130. [Google Scholar] [CrossRef]
- Burd, N.A.; Beals, J.W.; Martinez, I.G.; Salvador, A.F.; Skinner, S.K. Food-First Approach to Enhance the Regulation of Post-exercise Skeletal Muscle Protein Synthesis and Remodeling. Sports Med. 2019, 49, 59–68. [Google Scholar] [CrossRef]
- Viner, R.T.; Harris, M.; Berning, J.R.; Meyer, N.L. Energy Availability and Dietary Patterns of Adult Male and Female Competitive Cyclists with Lower than Expected Bone Mineral Density. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 594–602. [Google Scholar] [CrossRef]
- Manore, M.M.; Patton-Lopez, M.M.; Meng, Y.; Sung Wong, S. Sport Nutrition Knowledge, Behaviors and Beliefs of High School Soccer Players. Nutrients 2017, 9, 350. [Google Scholar] [CrossRef]
- Derbyshire, E.J. Flexitarian Diets and Health: A Review of the Evidence-Based Literature. Front. Nutr. 2017, 3, 1–8. [Google Scholar] [CrossRef]
- American Dietetic Association. Position of the American Dietetic Association: Vegetarian diets. J. Am. Diet. Assoc. 2009, 109, 1266. [Google Scholar] [CrossRef]
- Jeukendrup, A.E. Training the Gut for Athletes. Sports Med. 2017, 47, 101–110. [Google Scholar] [CrossRef]
- de Oliveira, E.P.; Buruni, R.C.; Jeukendrup, A.E. Gastrointestinal Complaints During Exercise: Prevalence, Etiology, and Nutritional Recommendations. Sports Med. 2014, 44, S79–S85. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Livingstone, M.B.E. Greenhouse gas emissions of self-selected diets in the UK and their association with diet quality: Is energy under-reporting a problem? Nutr. J. 2018, 17, 27. [Google Scholar] [CrossRef]
- Vieux, F.; Soler, L.-G.; Touazi, D.; Darmon, N. High nutritional quality is not associated with low greenhouse gas emissions in self-selected diets of French adults. Am. J. Clin. Nutr. 2013. [Google Scholar] [CrossRef] [PubMed]
- Hyland, J.J.; Henchion, M.; McCarthy, M.; McCarthy, S.N. The climatic impact of food consumption in a representative sample of Irish adults and implications for food and nutrition policy. Public Health Nutr. 2017, 20, 726–738. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.M.; Slater, G.; Broad, E.M.; Haukka, J.; Modulon, S.; Hopkins, W.G. Eating patterns and meal frequency of elite Australian athletes. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 521. [Google Scholar] [CrossRef] [PubMed]
- Erdman, K.A.; Tunnicliffe, J.; Lun, V.M.; Reimer, R.A. Eating patterns and composition of meals and snacks in elite Canadian athletes. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 210. [Google Scholar] [CrossRef] [PubMed]
- Masset, G.; Vieux, F.; Verger, E.O.; Soler, L.G.; Touazi, D.; Darmon, N. Reducing energy intake and energy density for a sustainable diet: A study based on self-selected diets in French adults. Am. J. Clin. Nutr. 2014, 99, 1460–1469. [Google Scholar] [CrossRef] [PubMed]
- Nemecek, T.; Dubois, D.; Huguenin-Elie, O.; Gaillard, G. Life cycle assessment of Swiss farming systems: I. Integrated and organic farming. Agric. Syst. 2011, 104, 217–232. [Google Scholar] [CrossRef]
- Nemecek, T.; Huguenin-Elie, O.; Dubois, D.; Gaillard, G.; Schaller, B.; Chervet, A. Life cycle assessment of Swiss farming systems: II. Extensive and intensive production. Agric. Syst. 2011, 104, 233–245. [Google Scholar] [CrossRef]
- Eshel, G.; Shepon, A.; Makov, T.; Milo, R. Land, irrigation water, greenhouse gas, and reactive nitrogen burdens of meat, eggs, and dairy production in the United States. Proc. Natl. Acad. Sci. USA 2014. [Google Scholar] [CrossRef]
- Lunn, W.R.; Pasikos, S.M.; Colletto, M.R.; Karfonta, K.E.; Carbone, J.W.; Anderson, J.M.; Rodriguez, N.R. Chocolate Milk and Endurance Exercise Recovery. Med. Sci. Sports Exerc. 2012, 44, 682–691. [Google Scholar] [CrossRef]
- Elliot, T.A.; Cree, M.G.; Sanford, A.P.; Wolfe, R.R.; Tipton, K.D. Milk ingestion stimulates net muscle protein synthesis following resistance exercise. Med. Sci. Sports Exerc. 2006, 38, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.; Phillips, S.M. Protein Recommendations for Weight Loss in Elite Athletes: A Focus on Body Composition and Performance. Int. J. Sport Nutr. Exerc. Metab. 2017, 32, 1–26. [Google Scholar] [CrossRef]
- Clifton, P.M.; Condo, D.; Keogh, J.B.; Skov, A.R.; Toubro, S.; Rønn, B.; Holm, L.; Astrup, A.; Weigle, D.S.; Breen, P.A.; et al. Long term weight maintenance after advice to consume low carbohydrate, higher protein diets—a systematic review and meta analysis. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Halton, T.L.; Hu, F.B. The Effects of High Protein Diets on Thermogenesis, Satiety and Weight Loss: A Critical Review. J. Am. Coll. Nutr. 2004, 23, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Doering, T.M.; Reaburn, P.R.; Phillips, S.M.; Jenkins, D.G. Post-Exercise Dietary Protein Strategies to Maximize Skeletal Muscle Repair and Remodeling in Masters Endurance: A Review. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 168–178. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Campbell, W.W.; Jacques, P.F.; Kritchevsky, S.B.; Moore, L.L.; Rodriguez, N.R.; van Loon, L.J. Protein and healthy aging. Am. J. Clin. Nutr. 2015, 101, 1339S–1345S. [Google Scholar] [CrossRef]
- Rowlands, D.S.; Nelson, A.R.; Phillips, S.M.; Faulkner, J.A.; Clarke, J.; Burd, N.A.; Moore, D.; Stellingwerff, T. Protein-Leucine Fed Dose Effects on Muscle Protein Synthesis after Endurance Exercise. Med. Sci. Sports Exerc. 2015, 47. [Google Scholar] [CrossRef]
- Jäger, R.; Zaragoza, J.; Purpura, M.; Iametti, S.; Marengo, M.; Tinsley, G.M.; Anzalone, A.J.; Oliver, J.M.; Fiore, W.; Biffi, A.; et al. Probiotic Administration Increases Amino Acid Absorption from Plant Protein: A Placebo-Controlled, Randomized, Double-Blind, Multicenter, Crossover Study. Probiotics Antimicrob. Proteins 2020. [Google Scholar] [CrossRef]
- Cabrera-Ramírez, A.H.; Luzardo-Ocampo, I.; Ramírez-Jiménez, A.K.; Morales-Sánchez, E.; Campos-Vega, R.; Gaytán-Martínez, M. Effect of the nixtamalization process on the protein bioaccessibility of white and red sorghum flours during in vitro gastrointestinal digestion. Food Res. Int. 2020, 134, 109234. [Google Scholar] [CrossRef]
- Kamau, E.H.; Nkhata, S.G.; Ayua, E.O. Extrusion and nixtamalization conditions influence the magnitude of change in the nutrients and bioactive components of cereals and legumes. Food Sci. Nutr. 2020, 8, 1753–1765. [Google Scholar] [CrossRef] [PubMed]
- Ciuris, C.; Lynch, H.M.; Wharton, C.; Johnston, C.S. A Comparison of Dietary Protein Digestibility, Based on DIAAS Scoring, in Vegetarian and Non-Vegetarian Athletes. Nutrients 2019, 11, 3016. [Google Scholar] [CrossRef] [PubMed]
- Jovanov, P.; Đorđić, V.; Obradović, B.; Barak, O.; Pezo, L.; Marić, A.; Sakač, M. Prevalence, knowledge and attitudes towards using sports supplements among young athletes. J. Int. Soc. Sports Nutr. 2019, 16, 27. [Google Scholar] [CrossRef]
- Madden, R.; Shearer, J.; Parnell, J. Evaluation of Dietary Intakes and Supplement Use in Paralympic Athletes. Nutrients 2017, 9, 1266. [Google Scholar] [CrossRef] [PubMed]
- Abbey, E.L.; Wright, C.J.; Kirkpatrick, C.M. Nutrition practices and knowledge among NCAA Division III football players. J. Int. Soc. Sports Nutr. 2017, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, S.; Beals, J.W.; Martinez, I.G.; Skinner, S.K.; Burd, N.A. Achieving optimal post-exercise muscle protein remodeling in physically active adults through whole food consumption. Nutrients 2018, 10, 224. [Google Scholar] [CrossRef]
- Kårlund, A.; Gómez-Gallego, C.; Turpeinen, A.M.; Palo-Oja, O.M.; El-Nezami, H.; Kolehmainen, M. Protein supplements and their relation with nutrition, microbiota composition and health: Is more protein always better for sportspeople? Nutrients 2019, 11, 829. [Google Scholar] [CrossRef]
- Clark, A.; Mach, N. Exercise-induced stress behavior, gut-microbiota-brain axis and diet: A systematic review for athletes. J. Int. Soc. Sports Nutr. 2016, 13, 1–21. [Google Scholar] [CrossRef]
- Xu, Z.; Knight, R. Dietary effects on human gut microbiome diversity. Br. J. Nutr. 2015, 113, S1–S5. [Google Scholar] [CrossRef]
- Churchward-Venne, T.A.; Pinckaers, P.J.M.; van Loon, J.J.A.; van Loon, L.J.C. Consideration of insects as a source of dietary protein for human consumption. Nutr. Rev. 2017, 75, 1035–1045. [Google Scholar] [CrossRef]
- van Huis, A.; van Itterbeeck, J.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects. Future Prospects for Food and Feed Security; Food and Agriculture Organiation of the United Nations (FAO): Rome, Italy, 2013. [Google Scholar] [CrossRef]
- Canning, P.; Charles, A.; Huang, S.; Polenske, K.R.; Waters, A. Energy Use in the U.S. Food System; The U.S. Department of Agriculture (USDA): Washington, DC, USA, 2010.
- Drewnowski, A.; Rehm, C.D.; Martin, A.; Verger, E.O.; Voinnesson, M.; Imbert, P. Energy and nutrient density of foods in relation to their carbon footprint. Am. J. Clin. Nutr. 2015, 101, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Gillen, J.B.; Trommelen, J.; Wardenaar, F.C.; Brinkmans, N.Y.; Versteegen, J.J.; Jonvik, K.L.; Kapp, C.; de Vries, J.H.M.; van Den Borne, J.J.G.C.; Gibala, M.J.; et al. Dietary protein intake and distribution patterns of well-trained Dutch athletes. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Basiotis, P.P.; Welsh, S.O.; Cronin, F.J.; Kelsay, J.L.; Mertz, W. Number of days of food intake records required to estimate individual and group nutrient intakes with defined confidence. J. Nutr. 1987, 117, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Tuomisto, H.L.; Hodge, I.D.; Riordan, P.; Macdonald, D.W. Does organic farming reduce environmental impacts?–A meta-analysis of European research. J. Environ. Manag. 2012, 112, 309–320. [Google Scholar] [CrossRef]
- U.S. Energy Information Administration. U.S. Energy Facts Explained-Consumption and Production-U.S. Energy Information Administration (EIA). Available online: https://www.eia.gov/energyexplained/us-energy-facts/ (accessed on 26 October 2019).
- Weber, C.L.; Matthews, S.H. Food-Miles and the Relative Climate Impacts of Food Choices in the United States. Environ. Sci. Technol. 2008, 42, 3508–3513. [Google Scholar] [CrossRef]



| Environmental Impact Categories | Description |
|---|---|
| GWP (kg CO2 eq) |
|
| Exergy (MJ) |
|
| Ecotoxicity (kg 1,4DB eq) |
|
| Eutrophication (aquatic and terrestrial) (person × year) |
|
| Easy | Moderate | Hard | ||
|---|---|---|---|---|
| Mean ± SD (n = 72) | Mean ± SD (n = 72) | Mean ± SD (n = 72) | ||
| Total food weight (kg) | 2.6 ± 0.6 | 2.4 ± 0.7 | 2.7 ± 0.7 | |
| GWP (kg CO2 eq) | Per Plate | 5.3 ± 1.9 | 6.0 ± 1.1 | 8.0 ± 1.9 |
| Per kg | 2.6 ± 0.8 | 2.5 ± 0.4 | 2.5 ± 0.3 | |
| 1000 kcal | 3.1 ± 1.0 | 2.6 ± 0.5 | 2.5 ± 0.5 | |
| Exergy (MJ) | Per Plate | 149.0 ± 64.1 | 171.0 ± 40.1 | 241.1 ± 73.7 |
| Per kg | 72.8 ± 26.2 | 71.9 ± 19.6 | 73.7 ± 13.6 | |
| 1000 kcal | 84.7 ± 25.2 | 75.1 ± 17.4 | 76.1 ± 16.2 | |
| Ecotoxicity (kg 1,4DB eq) | Per Plate | 2.6 ± 1.2 | 3.4 ± 1.6 | 3.7 ± 1.6 |
| Per kg | 1.3 ± 0.5 | 1.4 ± 0.5 | 1.1 ± 0.4 | |
| 1000 kcal | 1.6 ± 0.9 | 1.5 ± 0.7 | 1.2 ± 0.6 | |
| Eutrophication (person × year) | Per Plate | 0.0125 ± 0.0083 | 0.0121 ± 0.0037 | 0.0165 ± 0.0061 |
| Per kg | 0.0079 ± 0.0079 | 0.0056 ± 0.0023 | 0.0058 ± 0.0028 | |
| 1000 kcal | 0.0077 ± 0.0061 | 0.0053 ± 0.0016 | 0.0053 ± 0.0018 | |
| DIETITIAN | GWP (kg CO2 eq) | EXERGY (MJ) | ECOTOXICITY (kg 1.4DB eq) | EUTROPHICATION (person × year) | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| 1 | 6.1 | 2.2 | 180.6 | 64.9 | 2.8 | 1.0 | 0.0155 | 0.0033 |
| 2 | 6.0 | 1.9 | 155.0 | 27.2 | 4.1 | 1.5 | 0.0134 | 0.0059 |
| 3 | 6.3 | 1.4 | 162.3 | 29.3 | 2.8 | 0.7 | 0.0090 | 0.0019 |
| 4 | 6 | 1 | 169.2 | 19.2 | 3.2 | 1.1 | 0.0127 | 0.0023 |
| 5 | 6.3 | 1.7 | 169.3 | 42.2 | 3.4 | 1.7 | 0.0182 | 0.0137 |
| 6 | 4.9 | 0.6 | 140.4 | 11.7 | 1.9 | 0.5 | 0.0086 | 0.0008 |
| 7 | 5.8 | 1.0 | 157.5 | 25.8 | 2.3 | 0.4 | 0.0111 | 0.0044 |
| 8 | 7.3 | 1.1 | 194.1 | 31.5 | 3.4 | 0.6 | 0.0114 | 0.0023 |
| 9 | 6.0 | 1.5 | 183.8 | 70.0 | 3.1 | 1.4 | 0.0165 | 0.0055 |
| 10 | 7.3 | 1.8 | 208.8 | 45.6 | 4.1 | 1.5 | 0.0165 | 0.0057 |
| 11 | 7.2 | 1.4 | 214.3 | 41.6 | 5.4 | 1.7 | 0.0188 | 0.0054 |
| 12 | 8.0 | 3.8 | 309.1 | 114.7 | 2.7 | 1.0 | 0.0130 | 0.0059 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reguant-Closa, A.; Roesch, A.; Lansche, J.; Nemecek, T.; Lohman, T.G.; Meyer, N.L. The Environmental Impact of the Athlete’s Plate Nutrition Education Tool. Nutrients 2020, 12, 2484. https://doi.org/10.3390/nu12082484
Reguant-Closa A, Roesch A, Lansche J, Nemecek T, Lohman TG, Meyer NL. The Environmental Impact of the Athlete’s Plate Nutrition Education Tool. Nutrients. 2020; 12(8):2484. https://doi.org/10.3390/nu12082484
Chicago/Turabian StyleReguant-Closa, Alba, Andreas Roesch, Jens Lansche, Thomas Nemecek, Timothy G Lohman, and Nanna L Meyer. 2020. "The Environmental Impact of the Athlete’s Plate Nutrition Education Tool" Nutrients 12, no. 8: 2484. https://doi.org/10.3390/nu12082484
APA StyleReguant-Closa, A., Roesch, A., Lansche, J., Nemecek, T., Lohman, T. G., & Meyer, N. L. (2020). The Environmental Impact of the Athlete’s Plate Nutrition Education Tool. Nutrients, 12(8), 2484. https://doi.org/10.3390/nu12082484







