Hydroxytyrosol as a Promising Ally in the Treatment of Fibromyalgia
Abstract
1. Introduction
2. Materials and Methods
2.1. Fibroblasts Cultures
2.2. Hydroxytyrosol Treatment
2.3. Proteomic Study
2.3.1. Preparation of Protein Samples and nanoLC-MS/MS Analysis
2.3.2. Differential Expression
2.4. Bioinformatic Analysis
2.5. Validation of Relevant Proteins
2.6. Statistical Analysis
3. Results
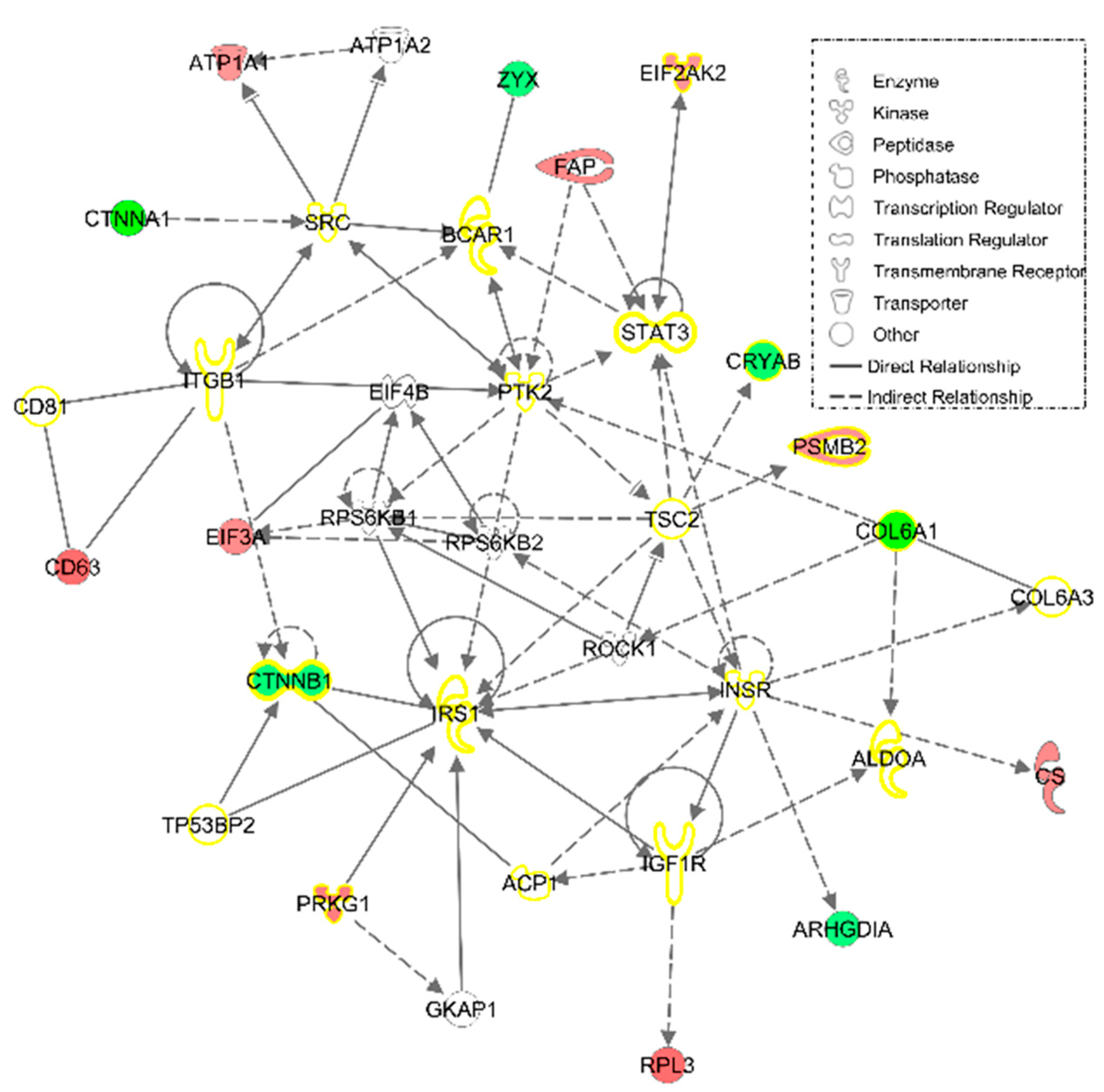
3.1. Proteomic Signature of FM
3.1.1. GO Analysis
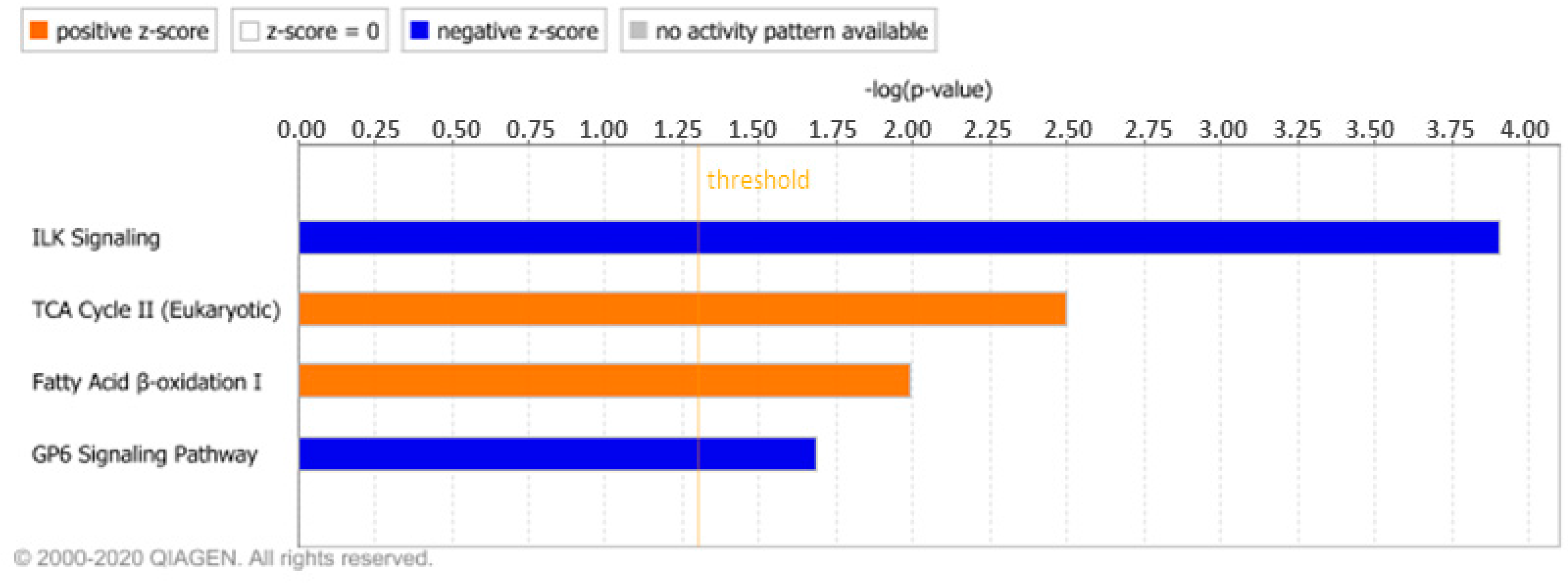
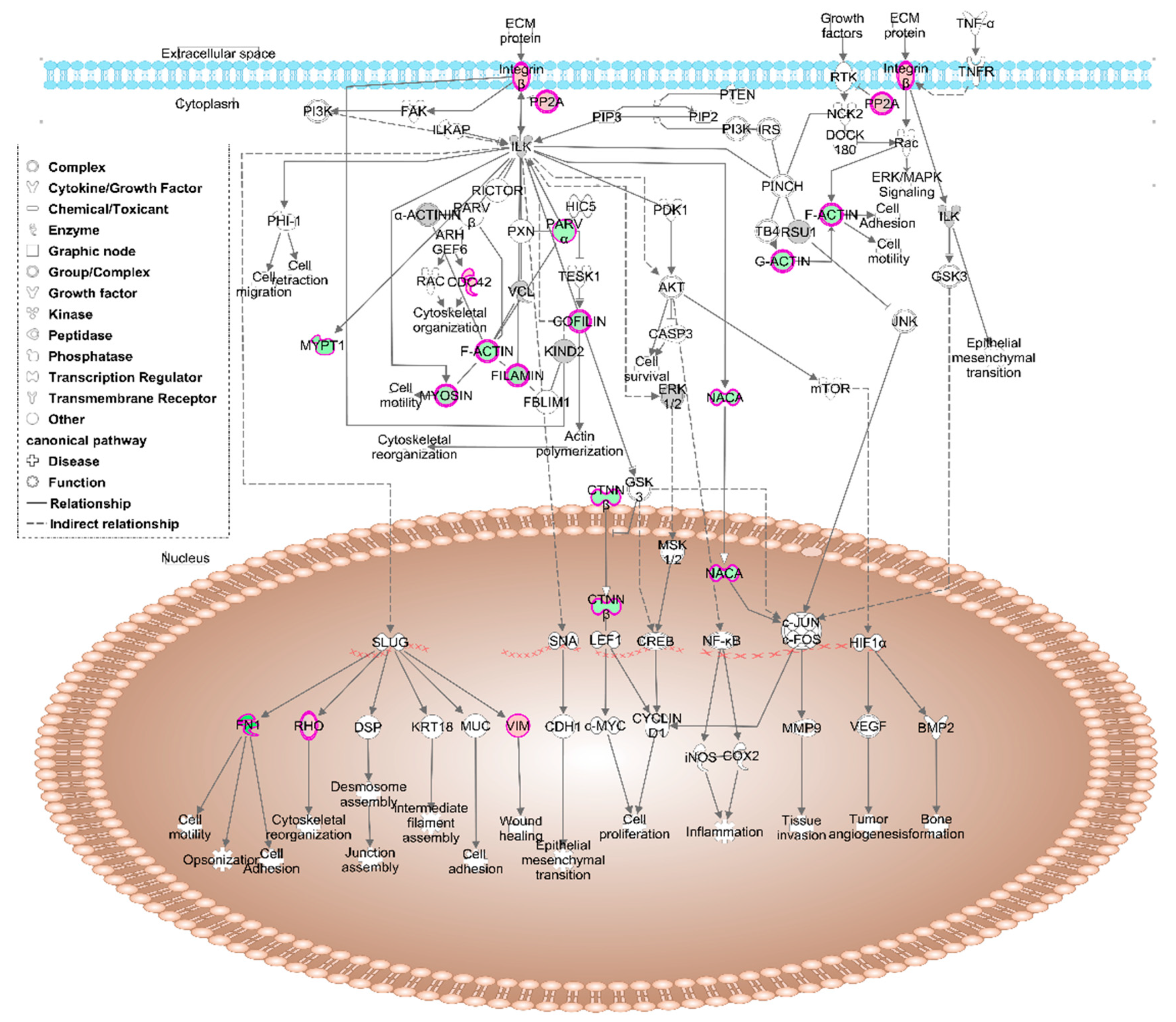
3.1.2. IPA Analysis
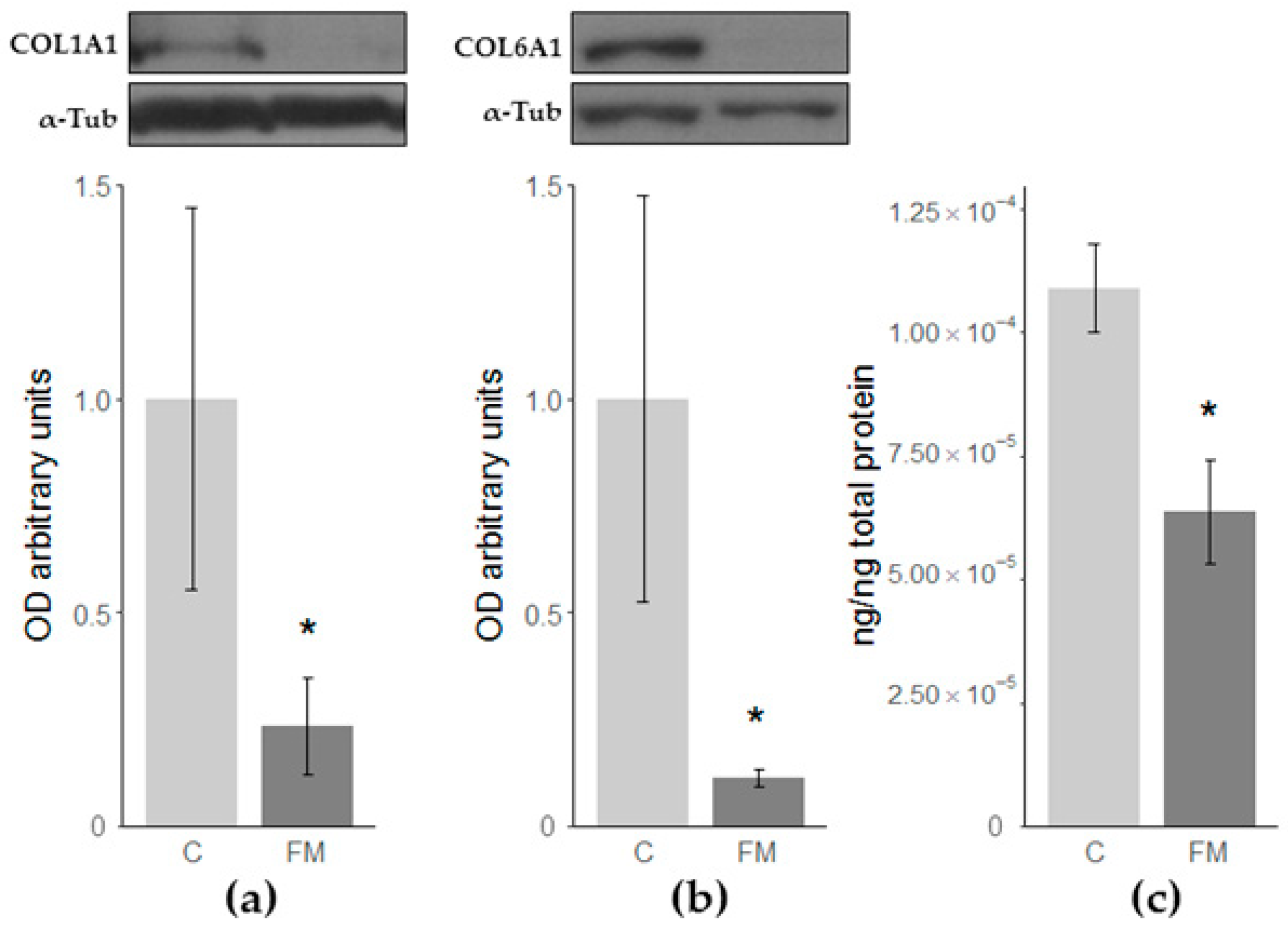
3.1.3. Validations of Collagen Type 1 and 6 alpha 1 Chains and Fibronectin 1
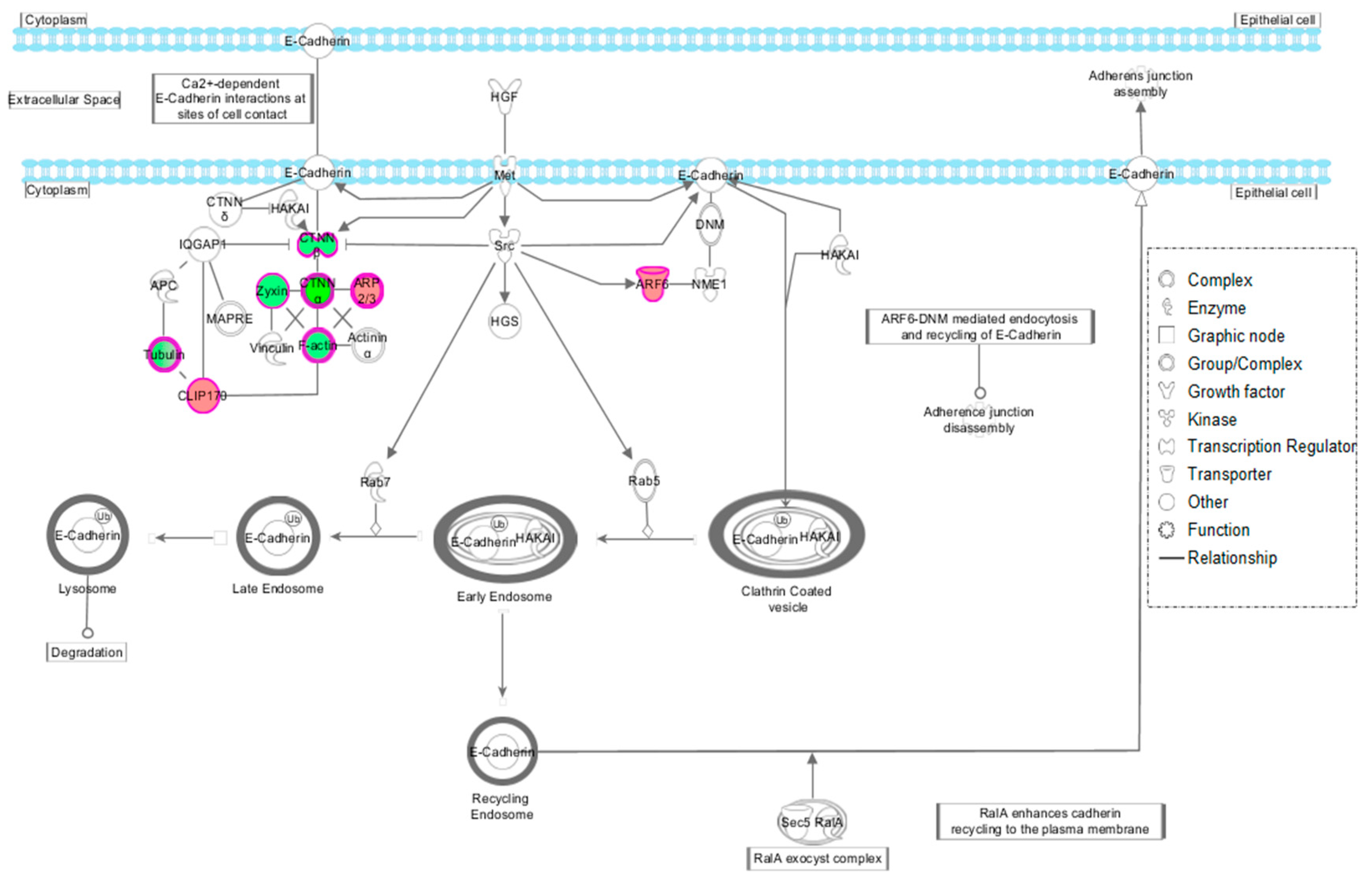
3.2. Effect of HT on Cell Proteome
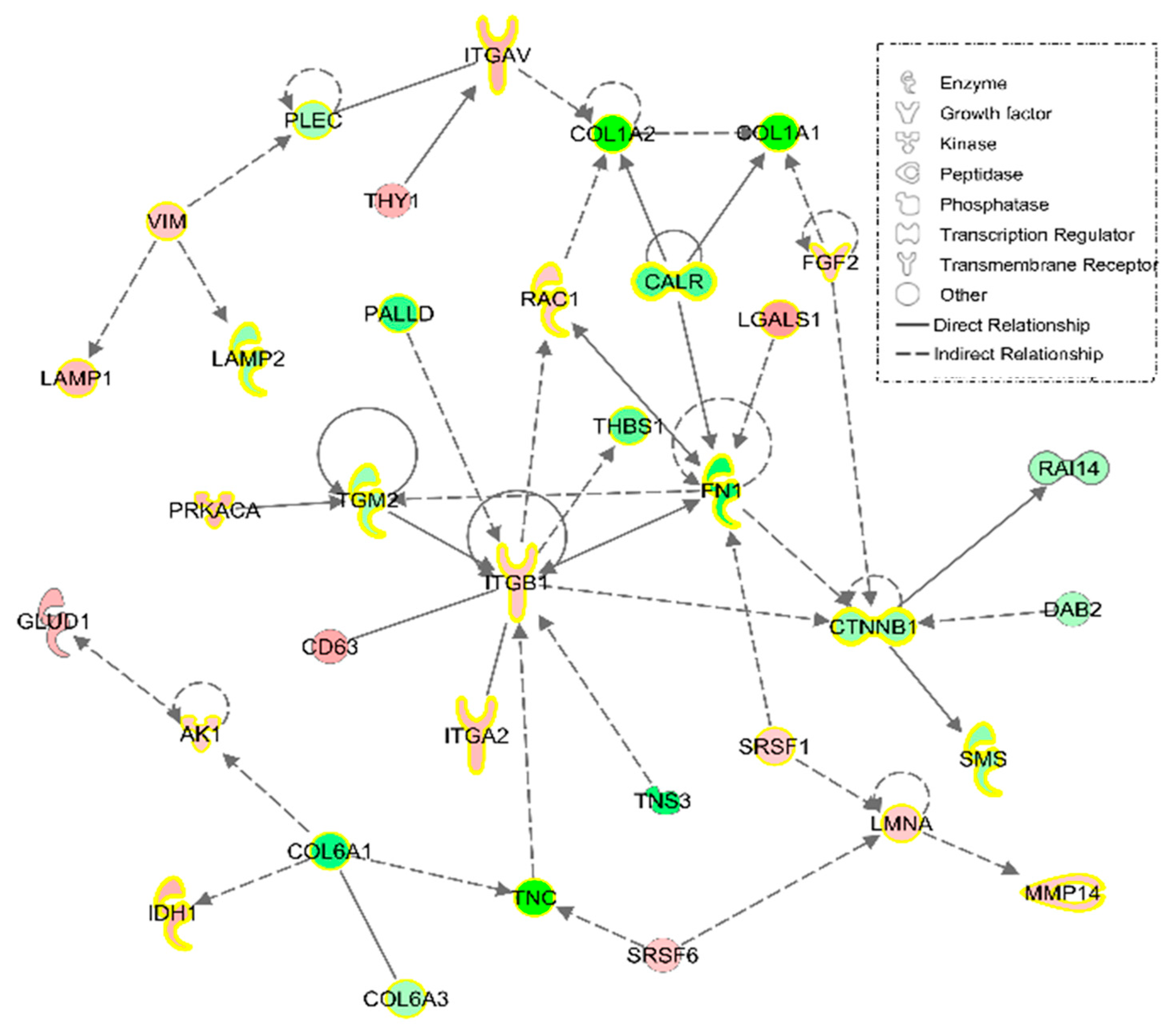
3.2.1. IPA Analysis
3.2.2. Validations of the Reversion of Cofilin 1 and Catenin Beta 1 by HT
4. Discussions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chinn, S.; Caldwell, W.; Gritsenko, K. Fibromyalgia Pathogenesis and Treatment Options Update. Curr. Pain Headache Rep. 2016, 20, 25. [Google Scholar] [CrossRef] [PubMed]
- Häuser, W.; Fitzcharles, M.A. Facts and myths pertaining to fibromyalgia. Dialogues Clin. Neurosci. 2018, 20, 53–62. [Google Scholar]
- Marques, A.P.; Santo, A.; Berssaneti, A.A.; Matsutani, L.A.; Yuan, S.L.K. Prevalence of fibromyalgia: Literature review update. Rev. Bras. Reumatol. Engl. Ed. 2017, 57, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Sicras-Mainar, A.; Rejas, J.; Navarro, R.; Blanca, M.; Morcillo, A.; Larios, R.; Velasco, S.; Villarroya, C. Treating patients with fibromyalgia in primary care settings under routine medical practice: A claim database cost and burden of illness study. Arthritis Res Ther. 2009, 11, R54. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, J.; Grasedyck, K.; Bromm, B. Middle and long latency somatosensory evoked potentials after painful laser stimulation in patients with fibromyalgia syndrome. Electroencephalogr. Clin. Neurophysiol. 1996, 100, 165–168. [Google Scholar] [CrossRef]
- Nijs, J.; Malfliet, A.; Ickmans, K.; Baert, I.; Meeus, M. Treatment of central sensitization in patients with ‘unexplained’ chronic pain: An update. Expert Opin. Pharmacother. 2014, 15, 1671–1683. [Google Scholar] [CrossRef]
- Serra, J.; Collado, A.; Sola, R.; Antonelli, F.; Torres, X.; Salgueiro, M.; Quiles, C.; Bostock, H. Hyperexcitable C nociceptors in fibromyalgia. Ann. Neurol. 2014, 75, 196–208. [Google Scholar] [CrossRef]
- Bozkurt, M.; Caglayan, M.; Oktayoglu, P.; Em, S.; Batmaz, I.; Sariyildiz, M.A.; Nas, K.; Ucar, D.; Yuksel, H.; Sarac, A.J. Serum prolidase enzyme activity and oxidative status in patients with fibromyalgia. Redox Rep. 2014, 19, 148–153. [Google Scholar] [CrossRef]
- La Rubia, M.; Rus, A.; Molina, F.; Del Moral, M.L. Is fibromyalgia-related oxidative stress implicated in the decline of physical and mental health status? Clin. Exp. Rheumatol. 2013, 31, S121–S127. [Google Scholar]
- Maes, M.; Mihaylova, I.; Kubera, M.; Uytterhoeven, M.; Vrydags, N.; Bosmans, E. Lower plasma Coenzyme Q10 in depression: A marker for treatment resistance and chronic fatigue in depression and a risk factor to cardiovascular disorder in that illness. Neuroendocrinol. Lett. 2009, 30, 462–469. [Google Scholar]
- Meeus, M.; Nijs, J.; Hermans, L.; Goubert, D.; Calders, P. The role of mitochondrial dysfunctions due to oxidative and nitrosative stress in the chronic pain or chronic fatigue syndromes and fibromyalgia patients: Peripheral and central mechanisms as therapeutic targets? Expert Opin. Ther. Targets 2013, 17, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Alcocer-Gómez, E.; Garrido-Maraver, J.; Bullón, P.; Marín-Aguilar, F.; Cotán, D.; Carrión, A.M.; Alvarez-Suárez, J.M.; Giampieri, F.; Sánchez-Alcázar, J.A.; Battino, M.; et al. Metformin and caloric restriction induce an AMPK-dependent restoration of mitochondrial dysfunction in fibroblasts from Fibromyalgia patients. Biochim. Biophys. Acta 2015, 1852, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Littlejohn, G.; Guymer, E. Neurogenic inflammation in fibromyalgia. Semin. Immunopathol. 2018, 40, 291–300. [Google Scholar] [CrossRef]
- Mastrangelo, F.; Frydas, I.; Ronconi, G.; Kritas, S.K.; Tettamanti, L.; Caraffa, A.; D’Ovidio, C.; Younes, A.; Gallenga, C.E.; Conti, P. Low-grade chronic inflammation mediated by mast cells in fibromyalgia: Role of IL-37. J. Biol. Regul. Homeost. Agents 2018, 32, 195–198. [Google Scholar]
- Backryd, E.; Tanum, L.; Lind, A.L.; Larsson, A.; Gordh, T. Evidence of both systemic inflammation and neuroinflammation in fibromyalgia patients, as assessed by a multiplex protein panel applied to the cerebrospinal fluid and to plasma. J. Pain Res. 2017, 10, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Stournaras, D.; Petrovic, K. Fibromyalgia treatment and management considerations for nurse practitioners. J. Am. Assoc. Nurse Pract. 2019, 31, 371–377. [Google Scholar] [CrossRef]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Hauser, W.; Fluss, E.; Choy, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef]
- Atzeni, F.; Talotta, R.; Masala, I.F.; Giacomelli, C.; Conversano, C.; Nucera, V.; Lucchino, B.; Iannuccelli, C.; Di Franco, M.; Bazzichi, L. One year in review 2019: Fibromyalgia. Clin. Exp. Rheumatol. 2019, 37, 3–10. [Google Scholar]
- Bjorklund, G.; Dadar, M.; Chirumbolo, S.; Aaseth, J. Fibromyalgia and nutrition: Therapeutic possibilities? Biomed. Pharmacother. 2018, 103, 531–538. [Google Scholar] [CrossRef]
- Gaforio, J.J.; Visioli, F.; Alarcón-de-la-Lastra, C.; Castañer, O.; Delgado-Rodríguez, M.; Fitó, M.; Hernández, A.F.; Huertas, J.R.; Martínez-González, M.A.; Menéndez, J.A.; et al. Virgin Olive Oil and Health: Summary of the III International Conference on Virgin Olive Oil and Health Consensus Report, JAEN (Spain) 2018. Nutrients 2019, 11, 2039. [Google Scholar] [CrossRef]
- Rus, A.; Molina, F.; Ramos, M.M.; Martinez-Ramirez, M.J.; Del Moral, M.L. Extra Virgin Olive Oil Improves Oxidative Stress, Functional Capacity, and Health-Related Psychological Status in Patients With Fibromyalgia: A Preliminary Study. Biol. Res. Nurs. 2017, 19, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Robles-Almazán, M.; Pulido-Morán, M.; Moreno-Fernández, J.; Ramírez-Tortosa, C.; Rodríguez-García, C.; Quiles, J.L.; Ramírez-Tortosa, M. Hydroxytyrosol: Bioavailability, toxicity, and clinical applications. Food Res. Int. 2018, 105, 654–667. [Google Scholar] [CrossRef] [PubMed]
- Charoenprasert, S.; Mitchell, A. Factors influencing phenolic compounds in table olives (Olea europaea). J. Agric. Food Chem. 2012, 60, 7081–7095. [Google Scholar] [CrossRef]
- Rigacci, S.; Stefani, M. Nutraceutical Properties of Olive Oil Polyphenols. An Itinerary from Cultured Cells through Animal Models to Humans. Int. J. Mol. Sci. 2016, 17, 843. [Google Scholar]
- Cicerale, S.; Conlan, X.A.; Sinclair, A.J.; Keast, R.S. Chemistry and health of olive oil phenolics. Crit. Rev. Food Sci. Nutr. 2009, 49, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Granados-Principal, S.; El-Azem, N.; Pamplona, R.; Ramirez-Tortosa, C.; Pulido-Moran, M.; Vera-Ramirez, L.; Quiles, J.L.; Sanchez-Rovira, P.; Naudi, A.; Portero-Otin, M.; et al. Hydroxytyrosol ameliorates oxidative stress and mitochondrial dysfunction in doxorubicin-induced cardiotoxicity in rats with breast cancer. BioChem. Pharmacol. 2014, 90, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Fuccelli, R.; Fabiani, R.; Rosignoli, P. Hydroxytyrosol Exerts Anti-Inflammatory and Anti-Oxidant Activities in a Mouse Model of Systemic Inflammation. Molecules 2018, 23, 3212. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, Z.; Wu, X.; Mao, A.; Chang, F.; Deng, X.; Gao, H.; Ouyang, C.; Dery, K.J.; Le, K.; et al. Discovery of potential new gene variants and inflammatory cytokine associations with fibromyalgia syndrome by whole exome sequencing. PLoS ONE 2013, 8, e65033. [Google Scholar] [CrossRef]
- Jones, K.D.; Gelbart, T.; Whisenant, T.C.; Waalen, J.; Mondala, T.S.; Iklé, D.N.; Salomon, D.R.; Bennett, R.M.; Kurian, S.M. Genome-wide expression profiling in the peripheral blood of patients with fibromyalgia. Clin. Exp. Rheumatol. 2016, 34, S89–S98. [Google Scholar]
- Ramírez-Tejero, J.A.; Martínez-Lara, E.; Rus, A.; Camacho, M.V.; Del Moral, M.L.; Siles, E. Insight into the biological pathways underlying fibromyalgia by a proteomic approach. J. Proteom. 2018, 186, 47–55. [Google Scholar] [CrossRef]
- Ciregia, F.; Giacomelli, C.; Giusti, L.; Boldrini, C.; Piga, I.; Pepe, P.; Consensi, A.; Gori, S.; Lucacchini, A.; Mazzoni, M.R.; et al. Putative salivary biomarkers useful to differentiate patients with fibromyalgia. J. Proteom. 2019, 190, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Khoonsari, P.E.; Musunri, S.; Herman, S.; Svensson, C.I.; Tanum, L.; Gordh, T.; Kultima, K. Systematic analysis of the cerebrospinal fluid proteome of fibromyalgia patients. J. Proteom. 2019, 190, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Alcocer-Gómez, E.; Marín-Aguilar, F.; Rybkina, T.; Cotán, D.; Pérez-Pulido, A.; Alvarez-Suarez, J.M.; Battino, M.; Sánchez-Alcazar, J.A.; Carrión, A.M.; et al. Mutation in cytochrome b gene of mitochondrial DNA in a family with fibromyalgia is associated with NLRP3-inflammasome activation. J. Med. Genet. 2016, 53, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Officioso, A.; Alzoubi, K.; Lang, F.; Manna, C. Hydroxytyrosol inhibits phosphatidylserine exposure and suicidal death induced by mercury in human erythrocytes: Possible involvement of the glutathione pathway. Food Chem. Toxicol. 2016, 89, 47–53. [Google Scholar] [CrossRef]
- Abate, M.; Pisanti, S.; Caputo, M.; Citro, M.; Vecchione, C.; Martinelli, R. 3-Hydroxytyrosol Promotes Angiogenesis In Vitro by Stimulating Endothelial Cell Migration. Int. J. Mol. Sci. 2020, 21, 3657. [Google Scholar] [CrossRef]
- Zorić, N.; Kopjar, N.; Rodriguez, J.V.; Tomić, S.; Kosalec, I. Protective effects of olive oil phenolics oleuropein and hydroxytyrosol against hydrogen peroxide-induced DNA damage in human peripheral lymphocytes. Acta Pharm. 2021, 71, 131–141. [Google Scholar]
- Wiśniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef]
- Thomas, P.D.; Campbell, M.J.; Kejariwal, A.; Mi, H.; Karlak, B.; Daverman, R.; Diemer, K.; Muruganujan, A.; Narechania, A. PANTHER: A library of protein families and subfamilies indexed by function. Genome Res. 2003, 13, 2129–2141. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis, 1st ed.; Springer: New York, NY, USA, 2016. [Google Scholar]
- Kuhlman, P.A.; Hughes, C.A.; Bennett, V.; Fowler, V.M. A new function for adducin. Calcium/calmodulin-regulated capping of the barbed ends of actin filaments. J. Biol. Chem. 1996, 271, 7986–7991. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; McCully, M.E.; Bhattacharya, N.; Butler, B.; Sept, D.; Cooper, J.A. Structure/function analysis of the interaction of phosphatidylinositol 4,5-bisphosphate with actin-capping protein: Implications for how capping protein binds the actin filament. J. Biol. Chem. 2007, 282, 5871–5879. [Google Scholar] [CrossRef] [PubMed]
- Bereiter-Hahn, J.; Voth, M.; Mai, S.; Jendrach, M. Structural implications of mitochondrial dynamics. Biotechnol. J. 2008, 3, 765–780. [Google Scholar] [CrossRef] [PubMed]
- Kuzmicic, J.; Del Campo, A.; López-Crisosto, C.; Morales, P.E.; Pennanen, C.; Bravo-Sagua, R.; Hechenleitner, J.; Zepeda, R.; Castro, P.F.; Verdejo, H.E.; et al. Mitochondrial dynamics: A potential new therapeutic target for heart failure. Revista Española de Cardiología 2011, 64, 916–923. [Google Scholar] [CrossRef]
- Cordero, M.D.; de Miguel, M.; Carmona-Lopez, I.; Bonal, P.; Campa, F.; Moreno-Fernandez, A.M. Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuroendocrinol. Lett. 2010, 31, 169–173. [Google Scholar]
- Cordero, M.D.; De Miguel, M.; Moreno Fernandez, A.M.; Carmona Lopez, I.M.; Garrido Maraver, J.; Cotan, D.; Gomez Izquierdo, L.; Bonal, P.; Campa, F.; Bullon, P.; et al. Mitochondrial dysfunction and mitophagy activation in blood mononuclear cells of fibromyalgia patients: Implications in the pathogenesis of the disease. Arthritis Res. Ther. 2010, 12, R17. [Google Scholar] [CrossRef]
- Favero, G.; Bonomini, F.; Franco, C.; Rezzani, R. Mitochondrial Dysfunction in Skeletal Muscle of a Fibromyalgia Model: The Potential Benefits of Melatonin. Int. J. Mol. Sci. 2019, 20, 765. [Google Scholar] [CrossRef]
- Izai, K.; Uchida, Y.; Orii, T.; Yamamoto, S.; Hashimoto, T. Novel fatty acid beta-oxidation enzymes in rat liver mitochondria. I. Purification and properties of very-long-chain acyl-coenzyme A dehydrogenase. J. Biol. Chem. 1992, 267, 1027–1033. [Google Scholar]
- Kamijo, T.; Aoyama, T.; Miyazaki, J.; Hashimoto, T. Molecular cloning of the cDNAs for the subunits of rat mitochondrial fatty acid beta-oxidation multienzyme complex. Structural and functional relationships to other mitochondrial and peroxisomal beta-oxidation enzymes. J. Biol. Chem. 1993, 268, 26452–26460. [Google Scholar]
- Nelson, D.L.; Cox, M.M. Lehninger: Principios de Bioquímica, 4th ed.; Ediciones Omega S.A.: Barcelona, Spain, 2015. [Google Scholar]
- Gelse, K.; Poschl, E.; Aigner, T. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef]
- Gajko-Galicka, A. Mutations in type I collagen genes resulting in osteogenesis imperfecta in humans. Acta Biochim. Pol. 2002, 49, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Zigrino, P.; Brinckmann, J.; Niehoff, A.; Lu, Y.; Giebeler, N.; Eckes, B.; Kadler, K.E.; Mauch, C. Fibroblast-Derived MMP-14 Regulates Collagen Homeostasis in Adult Skin. J. Investig. Dermatol. 2016, 136, 1575–1583. [Google Scholar] [CrossRef] [PubMed]
- Korpos, E.; Wu, C.; Sorokin, L. Multiple roles of the extracellular matrix in inflammation. Curr. Pharm. Des. 2009, 15, 1349–1357. [Google Scholar] [CrossRef] [PubMed]
- Araki, Y.; Mimura, T. Matrix Metalloproteinase Gene Activation Resulting from Disordred Epigenetic Mechanisms in Rheumatoid Arthritis. Int. J. Mol. Sci. 2017, 18, 905. [Google Scholar] [CrossRef] [PubMed]
- Coskun Benlidayi, I. Role of inflammation in the pathogenesis and treatment of fibromyalgia. Rheumatol. Int. 2019, 39, 781–791. [Google Scholar] [CrossRef]
- Weathington, N.M.; van Houwelingen, A.H.; Noerager, B.D.; Jackson, P.L.; Kraneveld, A.D.; Galin, F.S.; Folkerts, G.; Nijkamp, F.P.; Blalock, J.E. A novel peptide CXCR ligand derived from extracellular matrix degradation during airway inflammation. Nat. Med. 2006, 12, 317–323. [Google Scholar] [CrossRef]
- Almine, J.F.; Wise, S.G.; Hiob, M.; Singh, N.K.; Tiwari, K.K.; Vali, S.; Abbasi, T.; Weiss, A.S. Elastin sequences trigger transient proinflammatory responses by human dermal fibroblasts. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2013, 27, 3455–3465. [Google Scholar] [CrossRef]
- Pasparakis, M.; Vandenabeele, P. Necroptosis and its role in inflammation. Nature 2015, 517, 311–320. [Google Scholar] [CrossRef]
- Vanegas, O.C.; Bertini, E.; Zhang, R.Z.; Petrini, S.; Minosse, C.; Sabatelli, P.; Giusti, B.; Chu, M.L.; Pepe, G. Ullrich scleroatonic muscular dystrophy is caused by recessive mutations in collagen type VI. Proc. Natl. Acad. Sci. USA 2001, 98, 7516–7521. [Google Scholar] [CrossRef]
- Nugent, A.E.; Speicher, D.M.; Gradisar, I.; McBurney, D.L.; Baraga, A.; Doane, K.J.; Horton, W.E., Jr. Advanced osteoarthritis in humans is associated with altered collagen VI expression and upregulation of ER-stress markers Grp78 and bag-1. J. Histochem. Cytochem. 2009, 57, 923–931. [Google Scholar] [CrossRef]
- Chiquet, M.; Birk, D.E.; Bonnemann, C.G.; Koch, M. Collagen XII: Protecting bone and muscle integrity by organizing collagen fibrils. Int. J. Biochem. Cell Biol. 2014, 53, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Zamurs, L.K.; Idoate, M.A.; Hanssen, E.; Gomez-Ibanez, A.; Pastor, P.; Lamande, S.R. Aberrant mitochondria in a Bethlem myopathy patient with a homozygous amino acid substitution that destabilizes the collagen VI alpha2(VI) chain. J. Biol. Chem. 2015, 290, 4272–4281. [Google Scholar] [CrossRef] [PubMed]
- Parapia, L.A.; Jackson, C. Ehlers-Danlos syndrome—A historical review. Br. J. Haematol. 2008, 141, 32–35. [Google Scholar] [CrossRef]
- Hermanns-Lê, T.; Piérard, G.E.; Angenot, P. Fibromyalgia: An unrecognized Ehlers-Danlos syndrome hypermobile type? Revue Medicale de Liege 2013, 68, 22–24. [Google Scholar] [PubMed]
- Hicks, D.; Farsani, G.T.; Laval, S.; Collins, J.; Sarkozy, A.; Martoni, E.; Shah, A.; Zou, Y.; Koch, M.; Bonnemann, C.G.; et al. Mutations in the collagen XII gene define a new form of extracellular matrix-related myopathy. Hum. Mol. Genet. 2014, 23, 2353–2363. [Google Scholar] [CrossRef]
- Zou, Y.; Zwolanek, D.; Izu, Y.; Gandhy, S.; Schreiber, G.; Brockmann, K.; Devoto, M.; Tian, Z.; Hu, Y.; Veit, G.; et al. Recessive and dominant mutations in COL12A1 cause a novel EDS/myopathy overlap syndrome in humans and mice. Hum. Mol. Genet. 2014, 23, 2339–2352. [Google Scholar] [CrossRef]
- Lee, J.O.; Bankston, L.A.; Arnaout, M.A.; Liddington, R.C. Two conformations of the integrin A-domain (I-domain): A pathway for activation? Structure 1995, 3, 1333–1340. [Google Scholar] [CrossRef]
- Brakebusch, C.; Fassler, R. The integrin-actin connection, an eternal love affair. EMBO J. 2003, 22, 2324–2333. [Google Scholar] [CrossRef]
- Juliano, R.L.; Reddig, P.; Alahari, S.; Edin, M.; Howe, A.; Aplin, A. Integrin regulation of cell signalling and motility. Biochem. Soc. Trans. 2004, 32, 443–446. [Google Scholar] [CrossRef]
- Yen, C.-F.; Wang, H.-S.; Lee, C.-L.; Liao, S.-K. Roles of integrin-linked kinase in cell signaling and its perspectives as a therapeutic target. Gynecol. Minim. Invasive Ther. 2014, 3, 67–72. [Google Scholar] [CrossRef]
- Nieswandt, B.; Watson, S.P. Platelet-collagen interaction: Is GPVI the central receptor? Blood 2003, 102, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Furie, B.; Furie, B.C. Mechanisms of thrombus formation. N. Engl. J. Med. 2008, 359, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Miró-Casas, E.; Covas, M.I.; Farré, M.; Fitó, M.; Ortuño, J.; Weinbrenner, T.; Roset, P.; de la Torre, R. Hydroxytyrosol disposition in humans. Clin. Chem. 2003, 49, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Auñón-Calles, D.; Canut, L.; Visioli, F. Toxicological evaluation of pure hydroxytyrosol. Food Chem. Toxicol. 2013, 55, 498–504. [Google Scholar] [CrossRef]
- Auñón-Calles, D.; Giordano, E.; Bohnenberger, S.; Visioli, F. Hydroxytyrosol is not genotoxic in vitro. Pharmacol. Res. 2013, 74, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Colica, C.; Di Renzo, L.; Trombetta, D.; Smeriglio, A.; Bernardini, S.; Cioccoloni, G.; de Miranda, R.C.; Gualtieri, P.; Sinibaldi Salimei, P.; De Lorenzo, A. Antioxidant Effects of a Hydroxytyrosol-Based Pharmaceutical Formulation on Body Composition, Metabolic State, and Gene Expression: A Randomized Double-Blinded, Placebo-Controlled Crossover Trial. Oxidative Med. Cell. Longev. 2017, 2017, 2473495. [Google Scholar] [CrossRef] [PubMed]
- El Sayegh, T.Y.; Kapus, A.; McCulloch, C.A. Beyond the epithelium: Cadherin function in fibrous connective tissues. FEBS Lett. 2007, 581, 167–174. [Google Scholar] [CrossRef]
- Goebel, A.; Buhner, S.; Schedel, R.; Lochs, H.; Sprotte, G. Altered intestinal permeability in patients with primary fibromyalgia and in patients with complex regional pain syndrome. Rheumatology 2008, 47, 1223–1227. [Google Scholar] [CrossRef]
- Groschwitz, K.R.; Hogan, S.P. Intestinal barrier function: Molecular regulation and disease pathogenesis. J. Allergy Clin. Immunol. 2009, 124, 3–20; quiz 21–22. [Google Scholar] [CrossRef]
- Trujillo, J.; Molina-Jijon, E.; Medina-Campos, O.N.; Rodriguez-Munoz, R.; Reyes, J.L.; Loredo, M.L.; Barrera-Oviedo, D.; Pinzon, E.; Rodriguez-Rangel, D.S.; Pedraza-Chaverri, J. Curcumin prevents cisplatin-induced decrease in the tight and adherens junctions: Relation to oxidative stress. Food Funct. 2016, 7, 279–293. [Google Scholar] [CrossRef]
- Pirozzi, C.; Lama, A.; Simeoli, R.; Paciello, O.; Pagano, T.B.; Mollica, M.P.; Di Guida, F.; Russo, R.; Magliocca, S.; Canani, R.B.; et al. Hydroxytyrosol prevents metabolic impairment reducing hepatic inflammation and restoring duodenal integrity in a rat model of NAFLD. J. Nutr. BioChem. 2016, 30, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Zheng, A.; Li, H.; Xu, J.; Cao, K.; Li, H.; Pu, W.; Yang, Z.; Peng, Y.; Long, J.; Liu, J.; et al. Hydroxytyrosol improves mitochondrial function and reduces oxidative stress in the brain of db/db mice: Role of AMP-activated protein kinase activation. Br. J. Nutr. 2015, 113, 1667–1676. [Google Scholar] [CrossRef] [PubMed]
- Calabriso, N.; Gnoni, A.; Stanca, E.; Cavallo, A.; Damiano, F.; Siculella, L.; Carluccio, M.A. Hydroxytyrosol Ameliorates Endothelial Function under Inflammatory Conditions by Preventing Mitochondrial Dysfunction. Oxidative Med. Cell. Longev. 2018, 2018, 9086947. [Google Scholar] [CrossRef] [PubMed]
- Anelli, T.; Sitia, R. Protein quality control in the early secretory pathway. EMBO J. 2008, 27, 315–327. [Google Scholar] [CrossRef]
- Rozpȩdek, W.; Pytel, D.; Mucha, B.; Leszczńyska, H.; Diehl, J.A.; Majsterek, I. The Role of the PERK/eIF2alpha/ATF4/CHOP Signaling Pathway in Tumor Progression During Endoplasmic Reticulum Stress. Curr. Mol. Med. 2016, 16, 533–544. [Google Scholar] [CrossRef]
- Giordano, E.; Davalos, A.; Nicod, N.; Visioli, F. Hydroxytyrosol attenuates tunicamycin-induced endoplasmic reticulum stress in human hepatocarcinoma cells. Mol. Nutr. Food Res. 2014, 58, 954–962. [Google Scholar] [CrossRef]
- Wang, N.; Liu, Y.; Ma, Y.; Wen, D. Hydroxytyrosol ameliorates insulin resistance by modulating endoplasmic reticulum stress and prevents hepatic steatosis in diet-induced obesity mice. J. Nutr. BioChem. 2018, 57, 180–188. [Google Scholar] [CrossRef]
- Azios, N.G.; Krishnamoorthy, L.; Harris, M.; Cubano, L.A.; Cammer, M.; Dharmawardhane, S.F. Estrogen and resveratrol regulate Rac and Cdc42 signaling to the actin cytoskeleton of metastatic breast cancer cells. Neoplasia 2007, 9, 147–158. [Google Scholar] [CrossRef]
- García-Vilas, J.A.; Quesada, A.R.; Medina, M.Á. Hydroxytyrosol targets extracellular matrix remodeling by endothelial cells and inhibits both ex vivo and in vivo angiogenesis. Food Chem. 2017, 221, 1741–1746. [Google Scholar] [CrossRef]
- Arikawa-Hirasawa, E.; Le, A.H.; Nishino, I.; Nonaka, I.; Ho, N.C.; Francomano, C.A.; Govindraj, P.; Hassell, J.R.; Devaney, J.M.; Spranger, J.; et al. Structural and functional mutations of the perlecan gene cause Schwartz-Jampel syndrome, with myotonic myopathy and chondrodysplasia. Am. J. Hum. Genet. 2002, 70, 1368–1375. [Google Scholar] [CrossRef]
- Stattin, E.L.; Wiklund, F.; Lindblom, K.; Onnerfjord, P.; Jonsson, B.A.; Tegner, Y.; Sasaki, T.; Struglics, A.; Lohmander, S.; Dahl, N.; et al. A missense mutation in the aggrecan C-type lectin domain disrupts extracellular matrix interactions and causes dominant familial osteochondritis dissecans. Am. J. Hum. Genet. 2010, 86, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Mehraban, F.; Finegan, C.K.; Moskowitz, R.W. Serum keratan sulfate. Quantitative and qualitative comparisons in inflammatory versus noninflammatory arthritides. Arthritis Rheum. 1991, 34, 383–392. [Google Scholar] [PubMed]
- Mertoglu, C.; Gunay, M.; Yerligok, O. Could Endocan, a Marker of Inflammation and Endothelial Dysfunction, be a New Diagnostic Marker for Fibromyalgia? Clinica y Laboratorio 2018, 64, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Larsson, A.; Palstam, A.; Lofgren, M.; Ernberg, M.; Bjersing, J.; Bileviciute-Ljungar, I.; Gerdle, B.; Kosek, E.; Mannerkorpi, K. Resistance exercise improves muscle strength, health status and pain intensity in fibromyalgia—A randomized controlled trial. Arthritis Res. Ther. 2015, 17, 161. [Google Scholar] [CrossRef]
- Jeschonneck, M.; Grohmann, G.; Hein, G.; Sprott, H. Abnormal microcirculation and temperature in skin above tender points in patients with fibromyalgia. Rheumatology 2000, 39, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.A.; Pardo, J.V.; Pasley, J.D. Review of overlap between thermoregulation and pain modulation in fibromyalgia. Clin. J. Pain 2014, 30, 544–555. [Google Scholar] [CrossRef]
- Kuroshima, A.; Habara, Y.; Uehara, A.; Murazumi, K.; Yahata, T.; Ohno, T. Cross adaption between stress and cold in rats. Pflug. Archiv. Eur. J. Physiol. 1984, 402, 402–408. [Google Scholar] [CrossRef]
- Julien, N.; Goffaux, P.; Arsenault, P.; Marchand, S. Widespread pain in fibromyalgia is related to a deficit of endogenous pain inhibition. Pain 2005, 114, 295–302. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Wang, F.; You, L.; Xu, P.; Cao, Y.; Chen, L.; Wen, J.; Guo, X.; Cui, X.; et al. Identification of intracellular peptides associated with thermogenesis in human brown adipocytes. J. Cell. Physiol. 2019, 234, 7104–7114. [Google Scholar] [CrossRef]
| PANTHER GO-Slim Biological Process | GO ID | Fold Enrichment | FDR |
|---|---|---|---|
| actin filament depolymerisation | GO:0030042 | 15.97 | 5.04 × 10−2 |
| COPII-coated vesicle budding | GO:0090114 | 15.97 | 4.95 × 10−2 |
| bone morphogenesis | GO:0060349 | 13.78 | 2.02 × 10−2 |
| chondrocyte differentiation | GO:0002062 | 11.71 | 2.74 × 10−2 |
| connective tissue development | GO:0061448 | 11.16 | 2.86 × 10−2 |
| cartilage development | GO:0051216 | 11.16 | 2.80 × 10−2 |
| bone development | GO:0060348 | 10.65 | 3.10 × 10−2 |
| cellular protein complex disassembly | GO:0043624 | 8.57 | 9.17 × 10−3 |
| ribosomal small subunit biogenesis | GO:0042274 | 7.74 | 5.72 × 10−3 |
| translational initiation | GO:0006413 | 7.51 | 3.03 × 10−2 |
| protein-containing complex disassembly | GO:0032984 | 7.10 | 3.29 × 10−3 |
| cytoplasmic translation | GO:0002181 | 6.76 | 2.15 × 10−2 |
| cellular amino acid metabolic process | GO:0006520 | 6.71 | 4.41 × 10−6 |
| translational elongation | GO:0006414 | 6.25 | 4.29 × 10−9 |
| translation | GO:0006412 | 6.25 | 3.22 × 10−9 |
| peptide biosynthetic process | GO:0043043 | 6.17 | 2.77 × 10−9 |
| amide biosynthetic process | GO:0043604 | 5.88 | 2.93 × 10−9 |
| ribonucleoprotein complex assembly | GO:0022618 | 5.70 | 1.03 × 10−3 |
| carboxylic acid catabolic process | GO:0046395 | 5.54 | 2.14 × 10−2 |
| organic acid catabolic process | GO:0016054 | 5.54 | 2.09 × 10−2 |
| Symbol | Entrez Gene Name | UniProt Accession | q-Value | Fold Change | Pathway |
|---|---|---|---|---|---|
| PTPA | protein phosphatase 2 phosphatase activator | PTPA_HUMAN | 3.82 × 10−5 | 1.44 | ILK |
| VIM | vimentin | VIME_HUMAN | 1.16 × 10−4 | 1.40 | |
| ITGB1 | integrin subunit beta 1 | ITB1_HUMAN | 3.26 × 10−3 | 1.38 | |
| CDC42 | cell division cycle 42 | CDC42_HUMAN | 5.28 × 10−3 | 1.22 | |
| MYH9 | myosin heavy chain 9 | MYH9_HUMAN | 2.27 × 10−3 | −1.31 | |
| MYL6 | myosin light chain 6 | MYL6_HUMAN | 2.05 × 10−2 | −1.31 | |
| NACA | nascent polypeptide associated complex subunit alpha | NACAM_HUMAN | 1.00 × 10−3 | −1.47 | |
| PARVA | parvin alpha | PARVA_HUMAN | 4.29 × 10−4 | −1.47 | |
| CFL1 | cofilin 1 | COF1_HUMAN | 3.53 × 10−3 | −1.50 | |
| ACTB | actin beta | ACTB_HUMAN | 3.82 × 10−5 | −1.52 | |
| CTNNB1 | catenin beta 1 | CTNB1_HUMAN | 5.75 × 10−4 | −1.64 | |
| MYL9 | myosin light chain 9 | MYL9_HUMAN | 3.16 × 10−4 | −1.73 | |
| CFL2 | cofilin 2 | COF2_HUMAN | 1.32 × 10−3 | −1.73 | |
| PPP1R12A | protein phosphatase 1 regulatory subunit 12A | MYPT1_HUMAN | 1.04 × 10−3 | −1.86 | |
| FLNC | filamin C | FLNC_HUMAN | 4.22 × 10−4 | −1.95 | |
| FN1 | fibronectin 1 | FINC_HUMAN | 1.62 × 10−5 | −3.31 | |
| ACAA2 | acetyl-CoA acyltransferase 2 | THIM_HUMAN | 3.67 × 10−4 | 1.82 | βOX |
| HADH | hydroxyacyl-CoA dehydrogenase | HCDH_HUMAN | 1.80 × 10−2 | 1.82 | |
| HADHB | hydroxyacyl-CoA dehydrogenase trifunctional multienzyme complex subunit beta | ECHB_HUMAN | 2.57 × 10−4 | 1.49 | |
| HADHA | hydroxyacyl-CoA dehydrogenase trifunctional multienzyme complex subunit alpha | ECHA_HUMAN | 2.46 × 10−2 | 1.48 | |
| CS | citrate synthase | CISY_HUMAN | 7.93 × 10−3 | 1.72 | TCA |
| MDH2 | malate dehydrogenase 2 | MDHM_HUMAN | 4.67 × 10−3 | 1.57 | |
| OGDH | oxoglutarate dehydrogenase | ODO1_HUMAN | 6.99 × 10−3 | 1.53 | |
| DLST | dihydrolipoamide S-succinyltransferase | ODO2_HUMAN | 1.08 × 10−3 | 1.21 | |
| RAC1 | Rac family small GTPase 1 | RAC1_HUMAN | 1.06 × 10−2 | 1.35 | GP6 |
| COL6A2 | collagen type VI alpha 2 chain | CO6A2_HUMAN | 1.07 × 10−3 | −1.22 | |
| COL6A3 | collagen type VI alpha 3 chain | CO6A3_HUMAN | 2.43 × 10−2 | −1.40 | |
| COL6A1 | collagen type VI alpha 1 chain | CO6A1_HUMAN | 1.60 × 10−4 | −2.76 | |
| TLN1 | talin 1 | TLN1_HUMAN | 3.00 × 10−4 | −3.70 | |
| COL12A1 | collagen type XII alpha 1 chain | COCA1_HUMAN | 1.04 × 10−4 | −4.90 | |
| COL1A2 | collagen type I alpha 2 chain | CO1A2_HUMAN | 1.34 × 10−5 | −7.38 | |
| COL1A1 | collagen type I alpha 1 chain | CO1A1_HUMAN | 1.49 × 10−3 | −11.06 |
| KEGG ID | Name | N° of Proteins |
|---|---|---|
| hsa01100 | Metabolic pathways | 115 |
| hsa05200 | Pathways in cancer | 44 |
| hsa04217 | Necroptosis | 39 |
| hsa05165 | Human papillomavirus infection | 34 |
| hsa04510 | Focal adhesion | 33 |
| KEGG ID | Name | N° of Proteins | Proteins Abbreviation |
|---|---|---|---|
| hsa01100 | Metabolic pathways | 12 | ATPK, AT5F1, BIEA, CISY, C1TC, DHB12, GFPT1, GLGB, G6PI, PURA2, STT3B, TALDO |
| hsa04141 | Protein processing in endoplasmic reticulum | 8 | CRYAB, DNAJ2, SC23A, SC31A, STT3B, ERO1A, E2AK2, SAR1A |
| hsa04810 | Regulation of actin cytoskeleton | 5 | ACTB, ARC1B, COF1, COF2, MYPT1 |
| hsa04714 | Thermogenesis | 5 | ACTB, ATPK, AT5F1, KAPCA, KGP1 |
| hsa05205 | Proteoglycans in cancer | 5 | ACTB, CD63, CTNB1, MYPT1, KAPCA |
| Symbol | CONTROL | FM | FM+HT |
|---|---|---|---|
| COF1 | 1,41 × 10−7 ± 6,69 × 10−9 | 2,24 × 10−7 ± 1,73 × 10−9 *** | 2,10 × 10−7 ± 8,46 × 10−10 *** |
| CTNB1 | 7,15 × 10−2 ± 2,45 × 10−3 | 5,11 × 10−2 ± 1,21 × 10−3 ** | 6,64 × 10−2 ± 1,205 × 10−3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Tejero, J.A.; Martínez-Lara, E.; Peinado, M.Á.; del Moral, M.L.; Siles, E. Hydroxytyrosol as a Promising Ally in the Treatment of Fibromyalgia. Nutrients 2020, 12, 2386. https://doi.org/10.3390/nu12082386
Ramírez-Tejero JA, Martínez-Lara E, Peinado MÁ, del Moral ML, Siles E. Hydroxytyrosol as a Promising Ally in the Treatment of Fibromyalgia. Nutrients. 2020; 12(8):2386. https://doi.org/10.3390/nu12082386
Chicago/Turabian StyleRamírez-Tejero, Jorge A., Esther Martínez-Lara, M Ángeles Peinado, María Luisa del Moral, and Eva Siles. 2020. "Hydroxytyrosol as a Promising Ally in the Treatment of Fibromyalgia" Nutrients 12, no. 8: 2386. https://doi.org/10.3390/nu12082386
APA StyleRamírez-Tejero, J. A., Martínez-Lara, E., Peinado, M. Á., del Moral, M. L., & Siles, E. (2020). Hydroxytyrosol as a Promising Ally in the Treatment of Fibromyalgia. Nutrients, 12(8), 2386. https://doi.org/10.3390/nu12082386






