Effects of Probiotic Strains on Disease Activity and Enteric Permeability in Psoriatic Arthritis–A Pilot Open-Label Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Procedure
2.3. ELISA
2.4. Flow cytometry
2.5. Statistics
3. Results
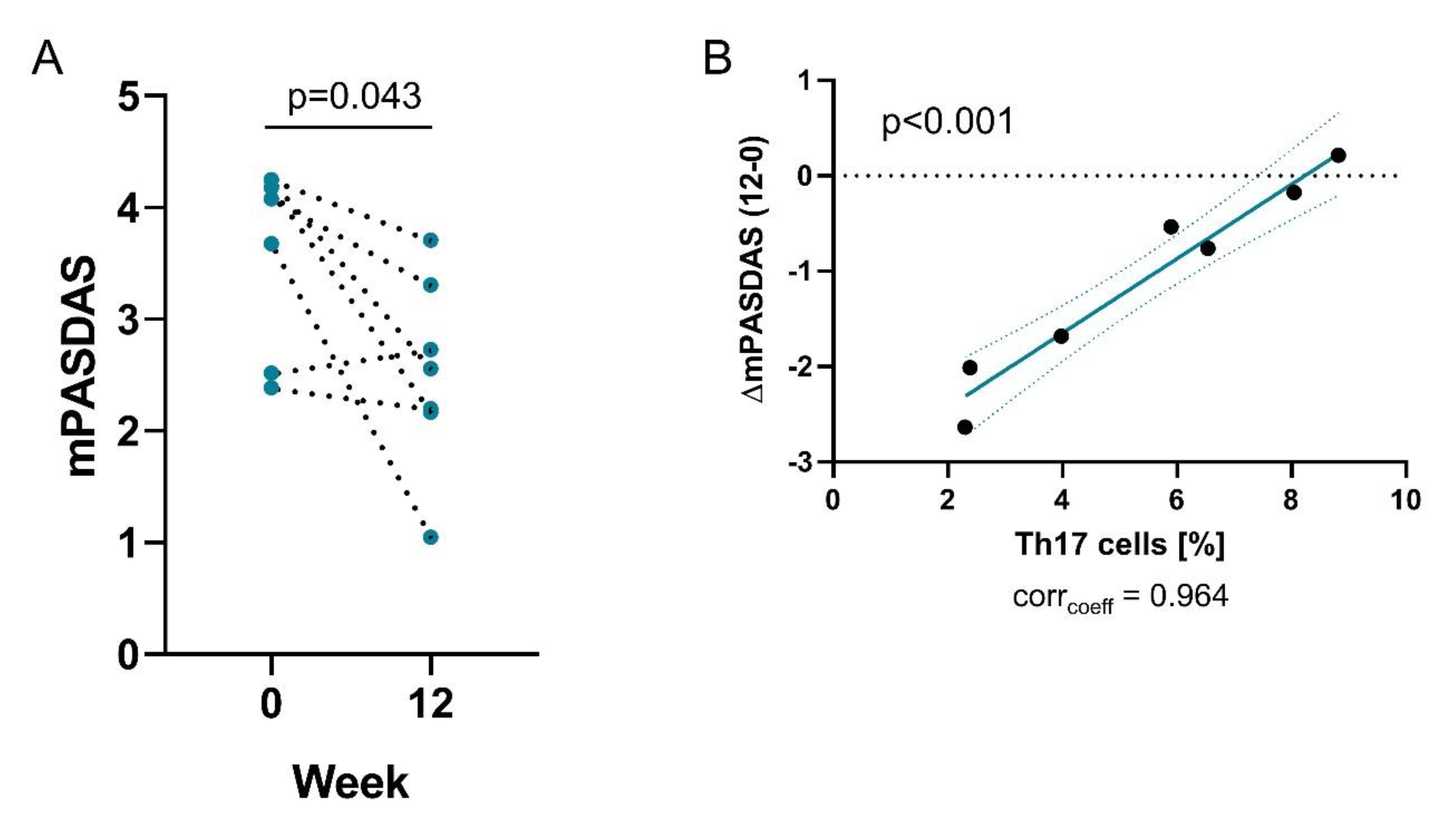
3.1. Probiotics Intake Decreases Disease Activity in PsA Patients
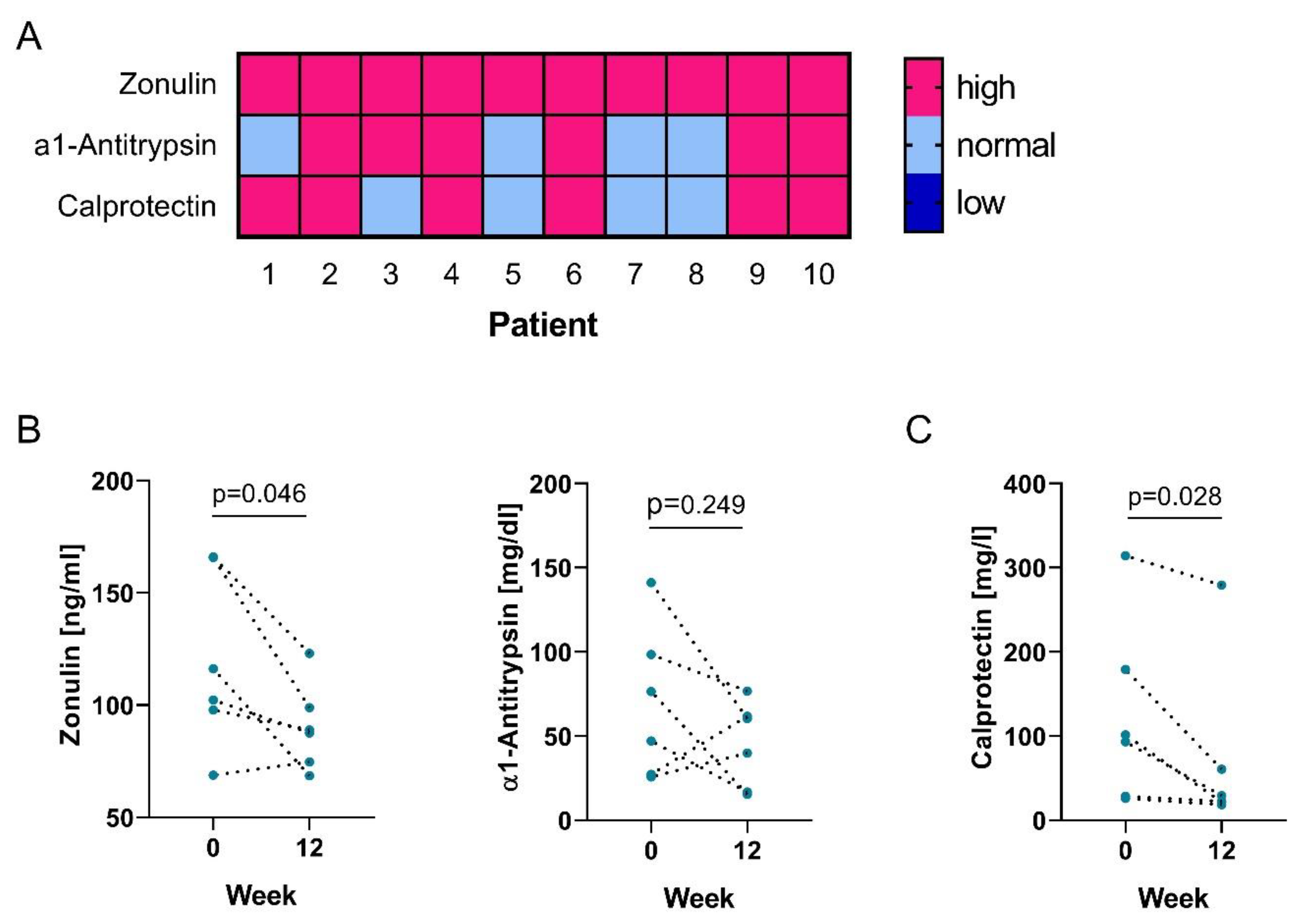
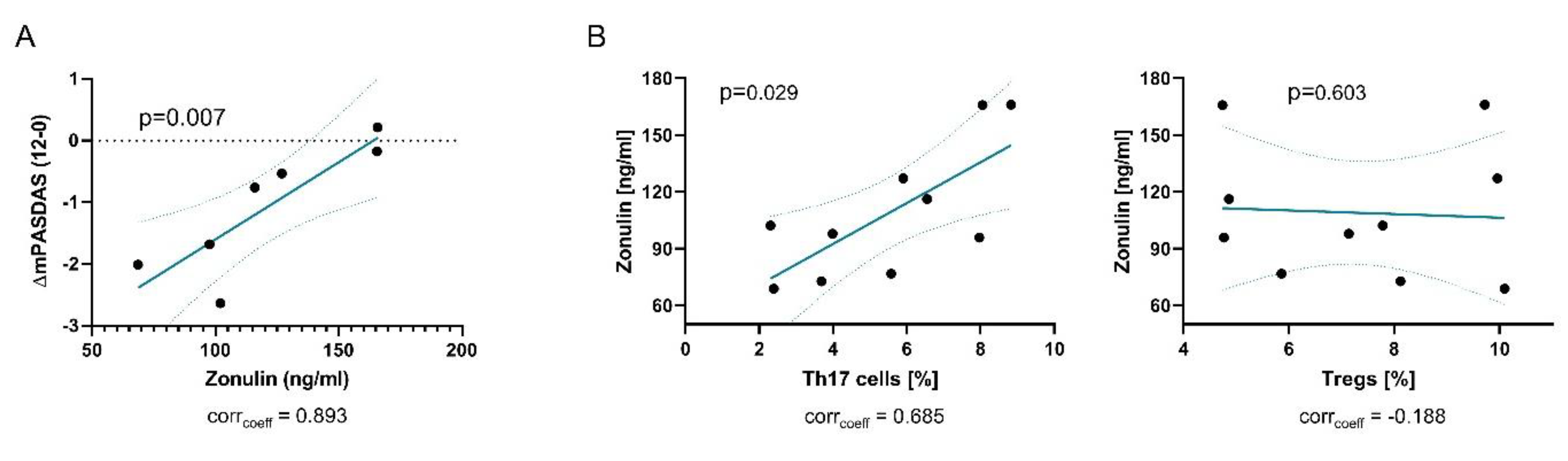
3.2. Intestinal Permeability is Enhanced in PsA Patients
3.3. Intestinal Permeability is Linked to a Th17-Related Immune Response
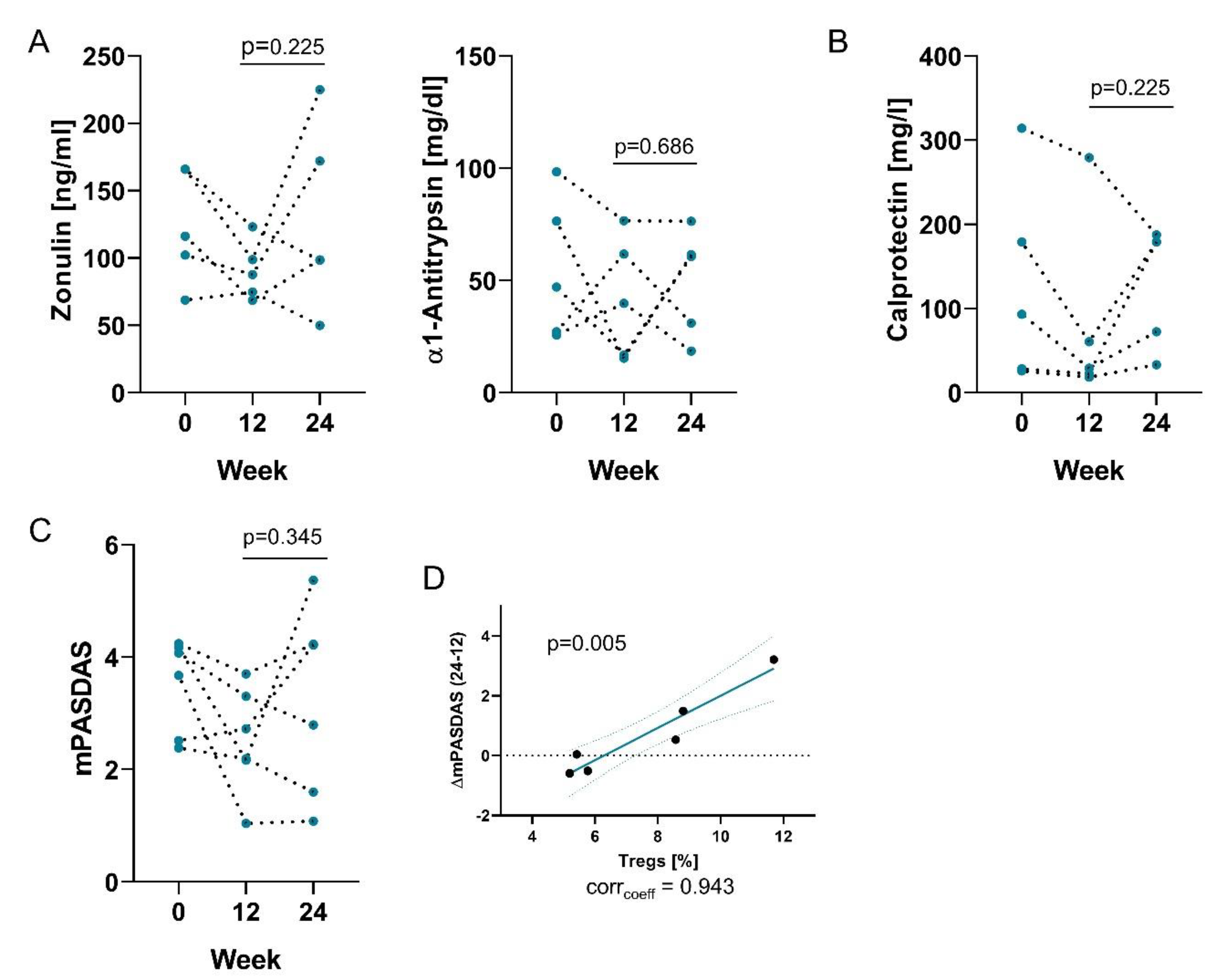
3.4. Effects of Probiotics are not Long-Lasting
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ritchlin, C.T.; Colbert, R.A.; Gladman, D.D. Psoriatic arthritis. N. Engl. J. Med. 2017, 376, 957–970. [Google Scholar] [CrossRef]
- Rath, H.C.; Herfarth, H.H.; Ikeda, J.S.; Grenther, W.B.; Hamm, T.E.; Balish, E.; Taurog, J.D.; Hammer, R.E.; Wilson, K.H.; Sartor, R.B. Normal luminal bacteria, especially bacteroides species, mediate chronic colitis, gastritis, and arthritis in HLA-B27/human β2 microglobulin transgenic rats. J. Clin. Investig. 1996, 98, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M.; et al. Dysbiosis Contributes to Arthritis Development via Activation of Autoreactive T Cells in the Intestine. Arthritis Rheumatol. 2016, 68, 2646–2661. [Google Scholar] [CrossRef] [PubMed]
- Rehaume, L.M.; Mondot, S.; Aguirre De Cárcer, D.; Velasco, J.; Benham, H.; Hasnain, S.Z.; Bowman, J.; Ruutu, M.; Hansbro, P.M.; McGuckin, M.A.; et al. ZAP-70 genotype disrupts the relationship between microbiota and host, leading to spondyloarthritis and ileitis in SKG mice. Arthritis Rheumatol. 2014, 66, 2780–2792. [Google Scholar] [CrossRef] [PubMed]
- Mease, P.; Van Der Heijde, D.; Landewé, R.; Mpofu, S.; Rahman, P.; Tahir, H.; Singhal, A.; Boettcher, E.; Navarra, S.; Meiser, K.; et al. Secukinumab improves active psoriatic arthritis symptoms and inhibits radiographic progression: Primary results from the randomised, double-blind, phase III FUTURE 5 study. Ann. Rheum. Dis. 2018, 77, 890–897. [Google Scholar] [CrossRef]
- Zhang, M.; Zhou, Q.; Dorfman, R.G.; Huang, X.; Fan, T.; Zhang, H.; Zhang, J.; Yu, C. Butyrate inhibits interleukin-17 and generates Tregs to ameliorate colorectal colitis in rats. BMC Gastroenterol. 2016, 16, 1–9. [Google Scholar] [CrossRef]
- Scher, J.U.; Ubeda, C.; Artacho, A.; Attur, M.; Isaac, S.; Reddy, S.M.; Marmon, S.; Neimann, A.; Brusca, S.; Patel, T.; et al. Decreased bacterial diversity characterizes the altered gut microbiota in patients with psoriatic arthritis, resembling dysbiosis in inflammatory bowel disease. Arthritis Rheumatol. 2015, 67, 128–139. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, Y.; Xu, Y.; Li, P.; Ma, H.; Li, X.; Li, M. The correlation between intestinal dysbiosis and the development of ankylosing spondylitis. Microb. Pathog. 2019, 132, 188–192. [Google Scholar] [CrossRef]
- Ciccia, F.; Guggino, G.; Rizzo, A.; Alessandro, R.; Luchetti, M.M.; Milling, S.; Saieva, L.; Cypers, H.; Stampone, T.; Di Benedetto, P.; et al. Dysbiosis and zonulin upregulation alter gut epithelial and vascular barriers in patients with ankylosing spondylitis. Ann. Rheum. Dis. 2017, 76, 1123–1132. [Google Scholar] [CrossRef]
- Wischmeyer, P.E.; McDonald, D.; Knight, R. Role of the microbiome, probiotics, and “dysbiosis therapy” in critical illness. Curr. Opin. Crit. Care 2016, 22, 347–353. [Google Scholar] [CrossRef]
- Tsai, Y.L.; Lin, T.L.; Chang, C.J.; Wu, T.R.; Lai, W.F.; Lu, C.C.; Lai, H.C. Probiotics, prebiotics and amelioration of diseases. J. Biomed. Sci. 2019, 26, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Navarro-López, V.; Martínez-Andrés, A.; Ramírez-Boscà, A.; Ruzafa-Costas, B.; Núñez-Delegido, E.; Carrión-Gutiérrez, M.A.; Prieto-Merino, D.; Codoñer-Cortés, F.; Ramón-Vidal, D.; Genovés-Martínez, S.; et al. Efficacy and safety of oral administration of a mixture of probiotic strains in patients with psoriasis: A randomized controlled clinical trial. Acta Derm. Venereol. 2019, 99, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Perruccio, A.V.; Got, M.; Li, S.; Ye, Y.; Gladman, D.D.; Chandran, V. Development of a Modified Psoriatic Arthritis Disease Activity Score Using the Medical Outcomes Study Short Form 12 as a Measure of Quality of Life. Arthritis Care Res. 2020, 72, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Bosch, P.; Husic, R.; Ficjan, A.; Gretler, J.; Lackner, A.; Graninger, W.B.; Duftner, C.; Hermann, J.; Dejaco, C. Evaluating current definitions of low disease activity in psoriatic arthritis using ultrasound. Rheumatol. (UK) 2019, 58, 2212–2220. [Google Scholar] [CrossRef]
- Fessler, J.; Raicht, A.; Husic, R.; Ficjan, A.; Duftner, C.; Schwinger, W.; Dejaco, C.; Schirmer, M. Premature senescence of T-cell subsets in axial spondyloarthritis. Ann. Rheum. Dis. 2015. [Google Scholar] [CrossRef]
- Borchers, A.T.; Selmi, C.; Meyers, F.J.; Keen, C.L.; Gershwin, M.E. Probiotics and immunity. J. Gastroenterol. 2009, 44, 26–46. [Google Scholar] [CrossRef]
- Esmaeili, S.A.; Mahmoudi, M.; Momtazi, A.A.; Sahebkar, A.; Doulabi, H.; Rastin, M. Tolerogenic probiotics: Potential immunoregulators in Systemic Lupus Erythematosus. J. Cell. Physiol. 2017, 232, 1994–2007. [Google Scholar] [CrossRef]
- Hajavi, J.; Esmaeili, S.A.; Varasteh, A.R.; Vazini, H.; Atabati, H.; Mardani, F.; Momtazi-Borojeni, A.A.; Hashemi, M.; Sankian, M.; Sahebkar, A. The immunomodulatory role of probiotics in allergy therapy. J. Cell. Physiol. 2019, 234, 2386–2398. [Google Scholar] [CrossRef]
- Malíčková, K.; Francová, I.; Lukáš, M.; Kolář, M.; Králíková, E.; Bortlík, M.; Ďuricová, D.; Štěpánková, L.; Zvolská, K.; Pánková, A.; et al. Fecal zonulin is elevated in Crohn’s disease and in cigarette smokers. Pract. Lab. Med. 2017, 9, 39–44. [Google Scholar] [CrossRef]
- Asmar, R.E.l.; Panigrahi, P.; Bamford, P.; Berti, I.; Not, T.; Coppa, G.V.; Catassi, C.; Fasano, A. Host-dependent zonulin secretion causes the impairment of the small intestine barrier function after bacterial exposure. Gastroenterology 2002, 123, 1607–1615. [Google Scholar] [CrossRef]
- Rajilić-Stojanović, M.; Biagi, E.; Heilig, H.G.H.J.; Kajander, K.; Kekkonen, R.A.; Tims, S.; De Vos, W.M. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1792–1801. [Google Scholar] [CrossRef] [PubMed]
- Heeney, D.D.; Gareau, M.G.; Marco, M.L. Intestinal Lactobacillus in health and disease, a driver or just along for the ride? Curr. Opin. Biotechnol. 2018, 49, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lv, J.; Pan, L.; Zhang, Y. Roles and applications of probiotic Lactobacillus strains. Appl. Microbiol. Biotechnol. 2018, 102, 8135–8143. [Google Scholar] [CrossRef] [PubMed]
- Cua, D.J.; Sherlock, J.P. Gut microbiota strikes “back”. Nat. Med. 2011, 17, 1055–1056. [Google Scholar] [CrossRef]
- Schaubeck, M.; Clavel, T.; Calasan, J.; Lagkouvardos, I.; Haange, S.B.; Jehmlich, N.; Basic, M.; Dupont, A.; Hornef, M.; Von Bergen, M.; et al. Dysbiotic gut microbiota causes transmissible Crohn’s disease-like ileitis independent of failure in antimicrobial defence. Gut 2016, 65, 225–237. [Google Scholar] [CrossRef]
- Breban, M.; Tap, J.; Leboime, A.; Said-Nahal, R.; Langella, P.; Chiocchia, G.; Furet, J.P.; Sokol, H. Faecal microbiota study reveals specific dysbiosis in spondyloarthritis. Ann. Rheum. Dis. 2017, 76, 1614–1622. [Google Scholar] [CrossRef]
- Manichanh, C.; Rigottier-Gois, L.; Bonnaud, E.; Gloux, K.; Pelletier, E.; Frangeul, L.; Nalin, R.; Jarrin, C.; Chardon, P.; Marteau, P.; et al. Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut 2006, 55, 205–211. [Google Scholar] [CrossRef]
- Luu, M.; Pautz, S.; Kohl, V.; Singh, R.; Romero, R.; Lucas, S.; Hofmann, J.; Raifer, H.; Vachharajani, N.; Carrascosa, L.C.; et al. The short-chain fatty acid pentanoate suppresses autoimmunity by modulating the metabolic-epigenetic crosstalk in lymphocytes. Nat. Commun. 2019, 10, 760. [Google Scholar] [CrossRef]
- Leblhuber, F.; Steiner, K.; Schuetz, B.; Fuchs, D.; Gostner, J.M. Probiotic Supplementation in Patients with Alzheimer’s Dementia—An Explorative Intervention Study. Curr. Alzheimer Res. 2018, 15, 1106–1113. [Google Scholar] [CrossRef]
| n = 10 | |
|---|---|
| Age at inclusion, years, median (range) | 58 (51–72) |
| Disease duration, years, median (range) | 11 (2–33) |
| Female sex, n | 7 |
| Current smoker, n | 3 |
| Anti-TNF, n | 3 |
| Anti-IL-17, n (%) | 2 |
| Methotrexate, n (%) | 2 |
| NSAID, n (%) | 5 |
| PASI, median (range) | 0.3 (0–4.2) |
| mPASDAS, median (range) | 3.9 (2.51–4.53) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haidmayer, A.; Bosch, P.; Lackner, A.; D’Orazio, M.; Fessler, J.; Stradner, M.H. Effects of Probiotic Strains on Disease Activity and Enteric Permeability in Psoriatic Arthritis–A Pilot Open-Label Study. Nutrients 2020, 12, 2337. https://doi.org/10.3390/nu12082337
Haidmayer A, Bosch P, Lackner A, D’Orazio M, Fessler J, Stradner MH. Effects of Probiotic Strains on Disease Activity and Enteric Permeability in Psoriatic Arthritis–A Pilot Open-Label Study. Nutrients. 2020; 12(8):2337. https://doi.org/10.3390/nu12082337
Chicago/Turabian StyleHaidmayer, Andreas, Philipp Bosch, Angelika Lackner, Monica D’Orazio, Johannes Fessler, and Martin H Stradner. 2020. "Effects of Probiotic Strains on Disease Activity and Enteric Permeability in Psoriatic Arthritis–A Pilot Open-Label Study" Nutrients 12, no. 8: 2337. https://doi.org/10.3390/nu12082337
APA StyleHaidmayer, A., Bosch, P., Lackner, A., D’Orazio, M., Fessler, J., & Stradner, M. H. (2020). Effects of Probiotic Strains on Disease Activity and Enteric Permeability in Psoriatic Arthritis–A Pilot Open-Label Study. Nutrients, 12(8), 2337. https://doi.org/10.3390/nu12082337





