Gestational Weight Gain—Re-Examining the Current Paradigm
Abstract
1. Introduction
- Is GWG modifiable through diet and physical activity?
- Does optimal GWG and risk of excess GWG, vary by pre-pregnancy BMI category?
- Is the association between GWG and adverse pregnancy outcomes causal?
2. Materials and Methods
2.1. Is Gestational Weight Gain Modifiable through Antenatal Diet and Physical Activity?
2.2. Does Optimal GWG and Risk of Excess GWG, Vary by Pre-Pregnancy BMI Category?
2.3. Is the Association between GWG and Adverse Pregnancy Outcomes Causal?
3. Results
3.1. Is Gestational Weight Gain Modifiable through Antenatal Diet and Physical Activity?
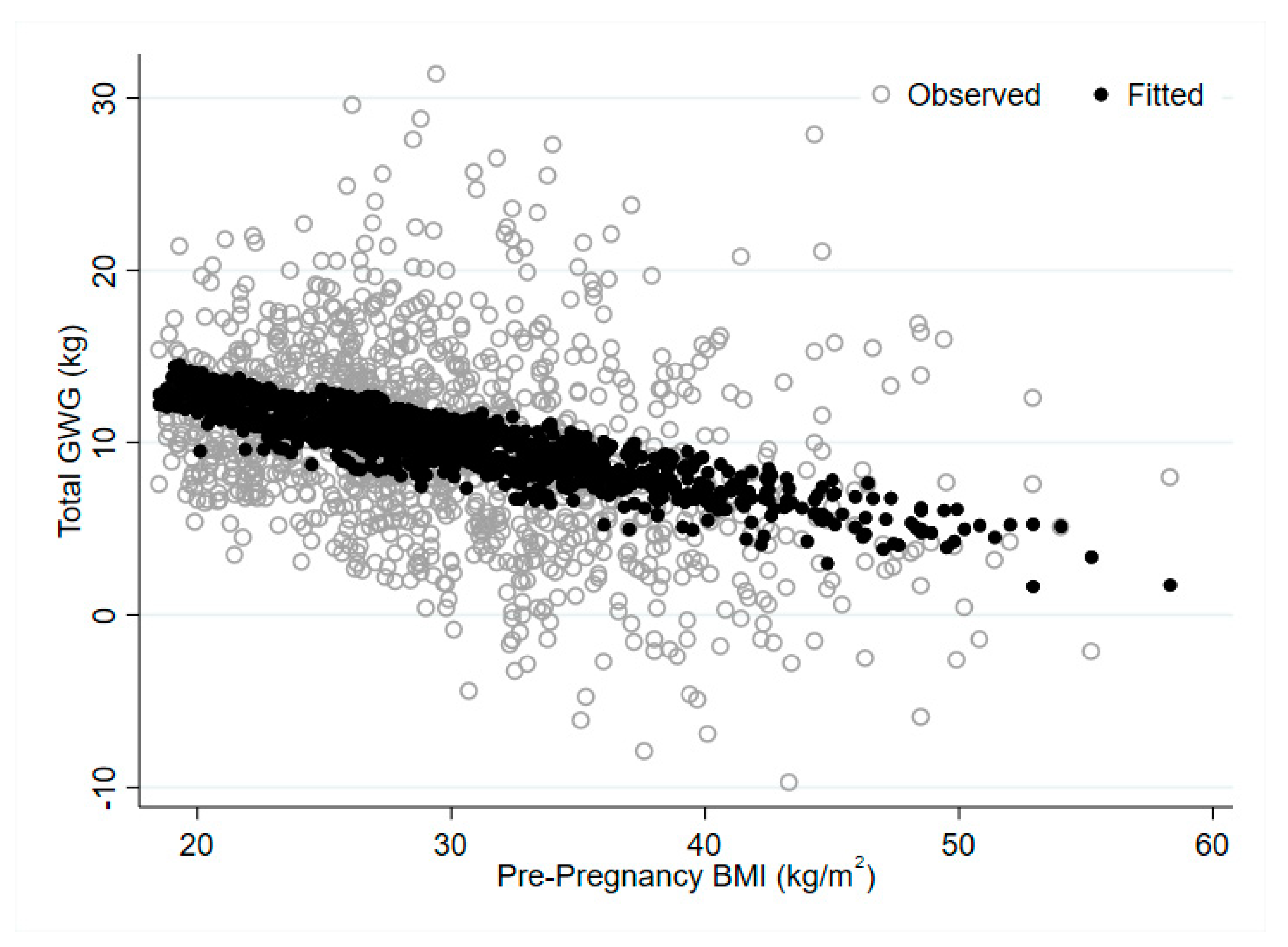
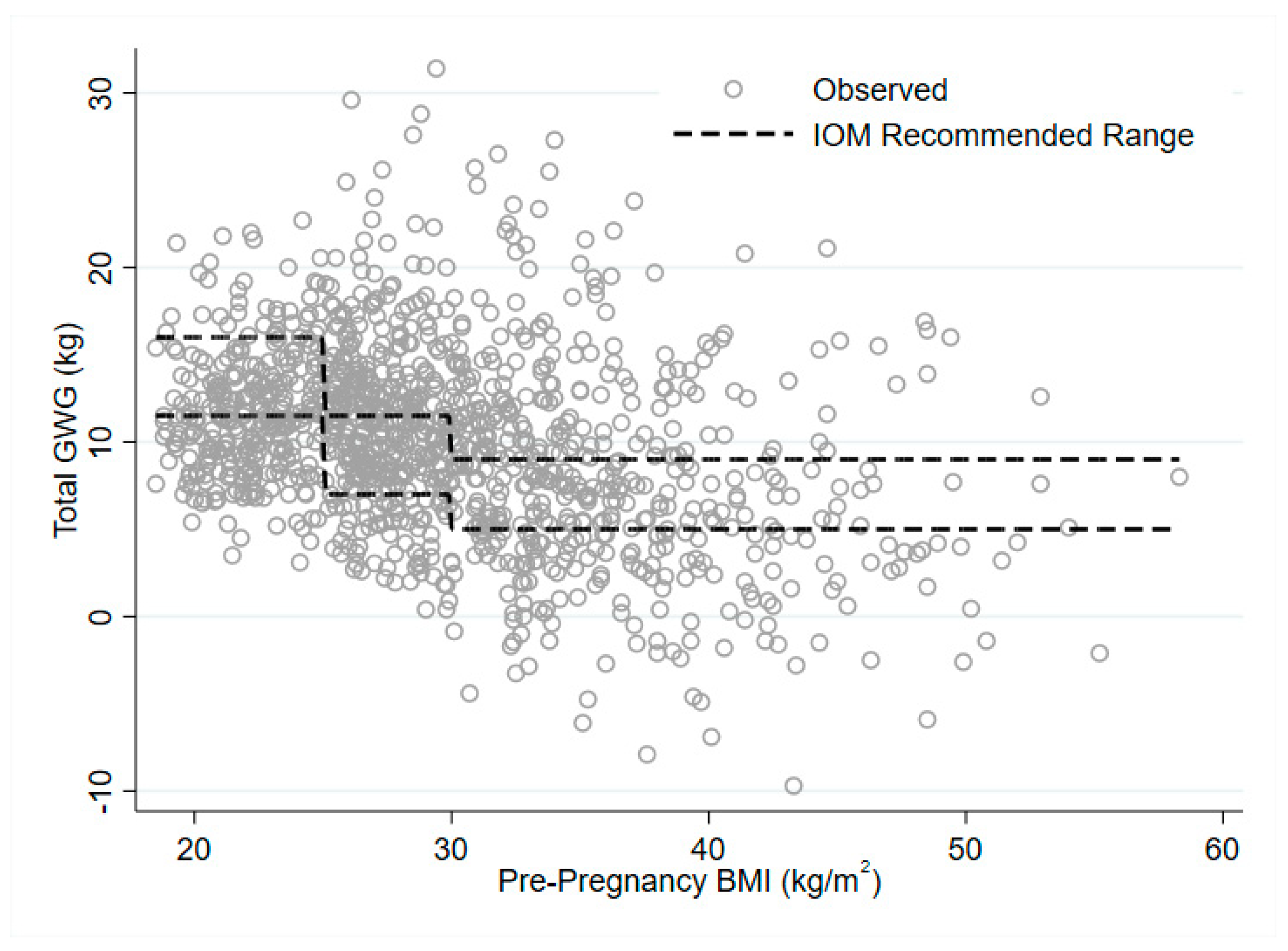
3.2. Does Optimal GWG and Risk of Excess GWG, Vary by Pre-Pregnancy BMI Category?
3.3. Is the Association between GWG and Adverse Pregnancy Outcomes Causal?
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dodd, J.M.; Grivell, R.M.; Nguyen, A.-M.; Chan, A.; Robinson, J.S. Maternal and perinatal health outcomes by body mass index category. Aust. N. Z. J. Obstet. Gynaecol. 2011, 51, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Callaway, L.K.; Prins, J.B.; Chang, A.M.; McIntyre, H.D. The prevalence and impact of overweight and obesity in an australian obstetric population. Med. J. Aust. 2006, 184, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Cedergren, M.I. Effects of gestational weight gain and body mass index on obstetric outcomes in sweden. Int. J. Gynecol. Obstet. 2006, 93, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Rong, K.; Yu, K.; Han, X.; Szeto, I.M.; Qin, X.; Wang, J.; Ning, Y.; Wang, P.; Ma, D. Pre-pregnancy bmi, gestational weight gain and postpartum weight retention: A meta-analysis of observational studies. Public Health Nutr. 2014, 20, 2172–2182. [Google Scholar] [CrossRef]
- Rooney, B.L.; Mathiason, M.A.; Schauberger, C.W. Predictors of obesity in childhood, adolescence, and adulthood in a birth cohort. Matern. Child Health J. 2010. [Google Scholar] [CrossRef]
- Wrotniak, B.H.; Shults, J.; Butts, S.; Stettler, N. Gestational weight gain and risk of overweight in the offspring at age 7 y in a multicenter, multiethnic cohort study. Am. J. Clin. Nutr. 2008, 87, 1818–1824. [Google Scholar] [CrossRef] [PubMed]
- Cnattingius, S.; Villamor, E.; Lagerros, Y.T.; Wikström, A.K.; Granath, F. High birth weight and obesity-a vicious circle across generations. Int. J. Obes. 2012, 36, 1320–1324. [Google Scholar] [CrossRef]
- Institute of Medicine Subcommittee on Nutritional Status and Weight Gain in Pregnancy. In Nutrition during Pregnancy; National Academy Press: Washington, DC, USA, 1990.
- Institute of Medicine. Weight Gain during Pregnancy: Reexamining the Guidelines; Institute of Medicine: Washington, DC, USA, 2009. [Google Scholar]
- Rogozińska, E.; Zamora, J.; Marlin, N.; Betrán, A.P.; Astrup, A.; Bogaerts, A.; Cecatti, J.G.; Dodd, J.M.; Facchinetti, F.; Geiker, N.R.; et al. Gestational weight gain outside the institute of medicine recommendations and adverse pregnancy outcomes: Analysis using individual participant data from randomised trials. BMC Pregnancy Childbirth 2019, 19, 322. [Google Scholar] [CrossRef]
- Goldstein, R.F.; Abell, S.K.; Ranasinha, S.; Misso, M.; Boyle, J.A.; Black, M.H.; Li, N.; Hu, G.; Corrado, F.; Rode, L.; et al. Association of gestational weight gain with maternal and infant outcomes: A systematic review and meta-analysis. JAMA 2017, 317, 2207–2225. [Google Scholar] [CrossRef]
- US Centre for Disease Control. Weight Gain during Pregnancy. Available online: https://www.cdc.gov/reproductivehealth/maternalinfanthealth/pregnancy-weight-gain.htm (accessed on 3 June 2019).
- Australian Government Department of Health. Pregnancy Care Guidelines. Available online: https://www.health.gov.au/resources/publications/pregnancy-care-guidelines (accessed on 3 June 2019).
- World Health Organisation. Who Recommendations on Antenatal Care for a Positive Pregnancy Experience. Available online: http://www.who.int/reproductivehealth/publications/maternal_perinatal_health/anc-positive-pregnancy-experience/en/ (accessed on 3 June 2019).
- International Weight Management in Pregnancy (i-WIP) Collaborative Group. Effect of diet and physical activity based interventions in pregnancy on gestational weight gain and pregnancy outcomes: Meta-analysis of individual participant data from randomised trials. BMJ 2017, 358, j3119. [Google Scholar] [CrossRef]
- Peaceman, A.M.; Clifton, R.G.; Phelan, S.; Gallagher, D.; Evans, M.; Redman, L.M.; Knowler, W.C.; Joshipura, K.; Haire-Joshu, D.; Yanovski, S.Z.; et al. Lifestyle interventions limit gestational weight gain in women with overweight or obesity: Life-moms prospective meta-analysis. Obesity 2018, 26, 1396–1404. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.M.; Turnbull, D.A.; McPhee, A.J.; Deussen, A.R.; Grivell, R.M.; Yelland, L.N.; Crowther, C.A.; Wittert, G.; Owens, J.A.; Robinson, J.S. Antenatal lifestyle advice for women who are overweight or obese: The limit randomised trial. BMJ 2014, 348, g1285. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.M.; Louise, J.; Deussen, A.R.; Grivell, R.M.; Dekker, G.; McPhee, A.J.; Hague, W. Effect of metformin in addition to dietary and lifestyle advice for pregnant women who are overweight or obese: The grow randomised, double-blind placebo-controlled trial. Lancet Diabetes Endocrinol. 2019, 7, 15–24. [Google Scholar] [CrossRef]
- Dodd, J.M.; Deussen, A.R.; Louise, J. A randomised trial to optimise gestational weight gain and improve maternal and infant health outcomes through antenatal dietary, lifestyle and exercise advice: The optimise randomised trial. Nutrients 2019, 11, 2911. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.M.; Cramp, C.S.; Sui, Z.; Yelland, L.N.; Deussen, A.R.; Grivell, R.M.; Moran, L.J.; Crowther, C.A.; Turnbull, D.A.; McPhee, A.J.; et al. Effects of antenatal lifestyle advice for women who are overweight or obese on maternal diet and physical activity: The limit randomised trial. BMC Med. 2014, 12, 161. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.M.; Deussen, A.R.; Mohamad, I.; Rifas-Shiman, S.L.; Yelland, L.N.; Louise, J.; McPhee, A.J.; Turnbull, D.A.; Grivell, R.M.; Owens, J.A.; et al. The effect of antenatal lifestyle advice for women who are overweight or obese on neonatal health: The limit randomised trial. BMC Med. 2014, 12, 163. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. Socio-Economic Indexes for Areas (SEIFA) 2006. Available online: https://www.ausstats.abs.gov.au/Ausstats/subscriber.nsf/0/72283F45CB86E5FECA2574170011B271/$File/2039055001_socio-economic%20indexes%20for%20areas%20(seifa)%20-%20technical%20paper_2006.pdf (accessed on 1 February 2019).
- Royston, P.; Sauerbrei, W. A new approach to modelling interactions between treatment and continuous covariates in clinical trials by using fractional polynomials. Stat. Med. 2004, 23, 2509–2525. [Google Scholar] [CrossRef]
- Royston, P.; Sauerbrei, W. Multivariable Model Building: A Pragmatic Approach to Regression Analysis Based on Fractional Polynomials for Continuous Variables; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- VanderWeele, T.J.; Vansteelandt, S. Mediation analysis with multiple mediators. Epidemiol. Method 2014, 2, 95–115. [Google Scholar] [CrossRef]
- Jebeile, H.; Mijatovic, J.; Louie, J.C.Y.; Prvan, T.; Brand-Miller, J.C. A systematic review and metaanalysis of energy intake and weight gain in pregnancy. Am. J. Obstet. Gynecol. 2016, 214, 465–483. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Siega-Riz, A.M.; Simhan, H.N.; Himes, K.P.; Abrams, B. Severe obesity, gestational weight gain, and adverse birth outcomes. Am. J. Clin. Nutr. 2010, 91, 1642–1648. [Google Scholar] [CrossRef]
- Hinkle, S.N.; Sharma, A.J.; Dietz, P.M. Gestational weight gain in obese mothers and associations with fetal growth. Am. J. Clin. Nutr. 2010, 92, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Altman, D.G.; Royston, P. The cost of dichotomising continuous variables. BMJ 2006, 332, 1080. [Google Scholar] [CrossRef] [PubMed]
- Bennette, C.; Vickers, A. Against quantiles: Categorization of continuous variables in epidemiologic research, and its discontents. BMC Med. Res. Methodol. 2012, 12, 21. [Google Scholar] [CrossRef] [PubMed]
- Froslie, K.F.; Roislien, J.; Laake, P.; Henriksen, T.; Qvigstad, E.; Veierod, M.B. Categorisation of continuous exposure variables revisited. A response to the hyperglycaemia and adverse pregnancy outcome (HAPO) study. BMC Med. Res. Methodol. 2010, 10, 103. [Google Scholar] [CrossRef]
- Dodd, J.M. Pregnancy: Managing obesity during pregnancy-what are the options? Nat. Rev. Endocrinol. 2015. [Google Scholar] [CrossRef]
- Hanson, M.A.; Barker, M.; Bustreo, F.; Dodd, J.M.; Kumanyika, S.; Norris, S.; Steegers, E.; Stephenson, J.; Thangaratinam, S.; Yang, H. Interventions to prevent preconception and maternal obesity. Lancet Diabetes Endocrinol. 2017, 5, 65–76. [Google Scholar] [CrossRef]
| Outcome | LIMIT | GRoW | OPTIMISE |
|---|---|---|---|
| LGA a | 0.90 (0.77, 1.07) | 0.87 (0.62, 1.23) | 0.88 (0.51, 1.52) |
| Birth weight > 4 kg a | 0.82 (0.68, 0.99) | 0.97 (0.65, 1.47) | 0.91 (0.54, 1.55) |
| Birth weight (g) b | −6.90 (−55.47, 41.67) | −13.01 (−106.45, 80.44) | −78.39 (−164.00, 7.22) |
| Birth weight z-score b | −0.05 (−0.14, 0.03) | −0.06 (−0.24, 0.12) | −0.04 (−0.18, 0.09) |
| Total GWG (kg) b | −0.04 (−0.55, 0.48) | −1.18 (−2.37, 0.01) | −0.37 (−0.97, 0.23) |
| Average Weekly GWG (kg) b | 0.00 (−0.03, 0.03) | −0.08 (−0.14, −0.02) | −0.03 (−0.06, 0.01) |
| GWG below recommendations a | 0.99 (0.84, 1.15) | 1.46 (1.10, 1.94) | 0.85 (0.60, 1.21) |
| GWG above recommendations a | 0.99 (0.89, 1.10) | 0.84 (0.65, 1.09) | 0.58 (0.32, 1.04) |
| Preterm Birth a | 0.74 (0.54, 1.03) | 0.79 (0.40, 1.58) | 1.14 (0.64, 2.03) |
| Caesarean Section a | 0.95 (0.85, 1.06) | 0.80 (0.64, 1.00) | 0.95 (0.72, 1.26) |
| Characteristic | LIMIT Standard Care a | OPTIMISE Standard Care a | LIMIT Combined Groups b | OPTIMISE Combined Groups b | GroW Metformin c | GroW Placebo c |
|---|---|---|---|---|---|---|
| Overall Numbers | n = 870 | n = 294 | n = 1767 | n = 585 | n = 195 | n = 184 |
| Study Centre | ||||||
| WCH | 388 (44.60) | 294 (100.00) | 792 (44.82) | 585 (100.00) | 86 (44.10) | 83 (45.11) |
| LMH | 212 (24.37) | 439 (24.84) | 90 (46.15) | 86 (46.74) | ||
| FMC | 270 (31.03) | 536 (30.33) | 19 (9.74) | 15 (8.15) | ||
| BMI: Median (IQR) | 30.9 (27.6, 35.4) | 22.2 (20.9, 23.5) | 31.0 (27.8, 35.8) | 22.2 (20.9, 23.6) | 32.60 (28.50, 37.70) | 32.27 (29.04, 36.80) |
| BMI Category: N (%) | ||||||
| 18.5–24.9 | 294 (100.00) | 585 (100.00) | 65 (33.33) | 57 (30.98) | ||
| 25.0–29.9 | 386 (44.37) | 752 (42.56) | 55 (28.21) | 61 (33.15) | ||
| 30.0–34.9 | 247 (28.39) | 507 (28.69) | 45 (23.08) | 35 (19.02) | ||
| 35.0–39.9 | 137 (15.75) | 317 (17.94) | 30 (15.38) | 31 (16.85) | ||
| ≥40.0 | 100 (11.49) | 191 (10.81) | ||||
| Parity: N (%) | 68 (34.87) | 65 (35.33) | ||||
| 0 | 351 (40.34) | 171 (58.16) | 717 (40.58) | 343 (58.63) | 127 (65.13) | 119 (64.67) |
| 1+ | 519 (59.66) | 123 (41.84) | 1050 (59.42) | 242 (41.37) | ||
| Smoking N (%) | 156 (84.78) | 181 (92.82) | ||||
| Nonsmoker | 755 (86.78) | 280 (95.24) | 1528 (86.47) | 562 (96.07) | 26 (14.13) | 13 (6.67) |
| Smoker | 98 (11.26) | 12 (4.08) | 210 (11.88) | 21 (3.59) | 2 (1.09) | 1 (0.51) |
| Missing | 17 (1.95) | 2 (0.68) | 29 (1.64) | 2 (0.34) | 30.18 (5.73) | 30.12 (5.47) |
| Age at Trial Entry: Mean (SD) | 29.57 (5.41) | 31.51 (4.91) | 29.50 (5.41) | 31.62 (4.68) | ||
| IRSD Quintile: N (%) | 55 (28.21) | 66 (35.87) | ||||
| Q1 (most disadvantaged) | 250 (28.74) | 57 (19.39) | 529 (29.94) | 99 (16.92) | 58 (29.74) | 51 (27.72) |
| Q2 | 214 (24.60) | 83 (28.23) | 432 (24.45) | 157 (26.84) | 22 (11.28) | 23 (12.50) |
| Q3 | 131 (15.06) | 30 (10.20) | 270 (15.28) | 76 (12.99) | 42 (21.54) | 32 (17.39) |
| Q4 | 141 (16.21) | 75 (25.51) | 264 (14.94) | 147 (25.13) | 18 (9.23) | 12 (6.52) |
| Q5 (least disadvantaged) | 134 (15.40) | 49 (16.67) | 271 (15.34) | 106 (18.12) |
| Characteristic | LIMIT Estimate (95% CI) a | LIMIT p Value | OPTIMISE Estimate (95% CI) b | OPTMISE p Value |
|---|---|---|---|---|
| Energy (kJ): +100 kJ | 0.142 * | 0.403 * | ||
| BL c | 0.01 (−0.02, 0.03) | 0.528 | −0.00 (−0.02, 0.02) | 0.645 |
| 28 w | −0.03 (−0.05, −0.00) | 0.020 | 0.02 (−0.01, 0.05) | 0.121 |
| 36 w | 0.02 (−0.01, 0.04) | 0.170 | −0.02 (−0.05, 0.01) | 0.185 |
| Carbohydrate (g): +10 g | 0.130 * | 0.433 * | ||
| BL | 0.01 (−0.06, 0.08) | 0.755 | −0.05 (−0.13, 0.03) | 0.237 |
| 28 w | −0.08 (−0.14, −0.01) | 0.029 | 0.05 (−0.03, 0.14) | 0.233 |
| 36 w | 0.06 (−0.00, 0.13) | 0.070 | 0.02 (−0.07, 0.10) | 0.696 |
| Fiber (g): +10 g | 0.360 * | 0.506 * | ||
| BL | 0.44 (−0.06, 0.95) | 0.085 | −0.26 (−0.75, 0.23) | 0.303 |
| 28 w | −0.19 (−0.74, 0.36) | 0.504 | 0.36 (−0.20, 0.92) | 0.209 |
| 36 w | −0.04 (−0.59, 0.52) | 0.899 | −0.21 (−0.83, 0.40) | 0.495 |
| Total Fat (g): +10 g | 0.637 * | 0.096 * | ||
| BL | 0.00 (−0.24, 0.25) | 0.982 | 0.07 (−0.14, 0.27) | 0.522 |
| 28 w | −0.17 (−0.44, 0.10) | 0.225 | 0.19 (−0.07, 0.46) | 0.151 |
| 36 w | 0.09 (−0.18, 0.36) | 0.515 | −0.35 (−0.62, −0.08) | 0.012 |
| Protein (g): +10 g | 0.293 * | 0.053 * | ||
| BL | 0.08 (−0.12, 0.28) | 0.440 | −0.03 (−0.23, 0.16) | 0.752 |
| 28 w | −0.22 (−0.44, 0.00) | 0.054 | 0.25 (0.01, 0.48) | 0.038 |
| 36 w | 0.09 (−0.12, 0.30) | 0.389 | −0.31 (−0.55, −0.07) | 0.011 |
| Sugars (g): +10 g | 0.165 * | 0.370 * | ||
| BL | 0.08 (−0.03, 0.20) | 0.166 | −0.02 (−0.14, 0.11) | 0.802 |
| 28 w | −0.10 (−0.21, 0.01) | 0.088 | 0.13 (−0.02, 0.29) | 0.091 |
| 36 w | 0.08 (−0.03, 0.19) | 0.167 | −0.06 (−0.19, 0.08) | 0.400 |
| HEI: +10 units | 0.862 * | 0.350 * | ||
| BL | 0.36 (−0.49, 1.22) | 0.403 | −0.31 (−1.11, 0.49) | 0.451 |
| 28 w | −0.04 (−0.83, 0.76) | 0.926 | −0.29 (−1.03, 0.45) | 0.447 |
| 36 w | −0.14 (−0.93, 0.65) | 0.728 | 0.57 (−0.10, 1.23) | 0.094 |
| METs: +1000 | 0.944 * | 0.721 * | ||
| BL | −0.02 (−0.17, 0.12) | 0.760 | 0.00 (−0.14, 0.14) | 0.986 |
| 28 w | 0.00 (−0.16, 0.16) | 0.996 | −0.04 (−0.20, 0.13) | 0.669 |
| 36 w | −0.03 (−0.18, 0.12) | 0.704 | −0.04 (−0.18, 0.09) | 0.549 |
| Excess GWG | Proportion a | RR (95% CI) b | p Value |
|---|---|---|---|
| BMI Category: | 0.032 * | ||
| BMI 18.5–24.9 | 0.18 (0.00, 0.37) | 1.17 (0.96, 1.42) | 0.117 |
| BMI 25.0–29.9 | 0.38 (0.32, 0.43) | 0.88 (0.81, 0.96) | 0.004 |
| BMI ≥ 30.0 | 0.46 (0.42, 0.51) | 0.94 (0.92, 0.97) | <0.001 |
| Outcome | Estimate (95% CI) at GWG = 10 kg a | Estimate (95% CI): Effect of 1 kg Increase in GWG b | p Value |
|---|---|---|---|
| Birth weight z-score | 0.170 * | ||
| BMI 20 kg/m2 | −0.08 (−0.15, −0.01) | 0.06 (0.05, 0.08) | <0.001 |
| BMI 25 kg/m2 | 0.11 (0.06, 0.16) | 0.06 (0.05, 0.07) | <0.001 |
| BMI 30 kg/m2 | 0.31 (0.27, 0.35) | 0.05 (0.05, 0.06) | <0.001 |
| BMI 35 kg/m2 | 0.50 (0.45, 0.56) | 0.05 (0.04, 0.06) | <0.001 |
| Outcome | Type of Effect a | Estimate (95% CI) | p Value |
|---|---|---|---|
| Infant large for gestational age (LGA) b | Total Effect | 0.97 (0.55, 1.71) | 0.907 |
| Direct Effect | 1.11 (0.62, 1.99) | 0.717 | |
| Indirect Effect | 0.87 (0.73, 1.03) | 0.103 | |
| Caesarean Section b | Total Effect | 0.56 (0.36, 0.86) | 0.008 |
| Direct Effect | 0.57 (0.37, 0.88) | 0.011 | |
| Indirect Effect | 0.99 (0.94, 1.04) | 0.613 | |
| Birth weight z-score c | Total Effect | −0.01 (−0.21, 0.19) | 0.928 |
| Direct Effect | 0.05 (−0.14, 0.25) | 0.587 | |
| Indirect Effect | −0.06 (−0.14, 0.01) | 0.090 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Louise, J.; Deussen, A.R.; Dodd, J.M. Gestational Weight Gain—Re-Examining the Current Paradigm. Nutrients 2020, 12, 2314. https://doi.org/10.3390/nu12082314
Louise J, Deussen AR, Dodd JM. Gestational Weight Gain—Re-Examining the Current Paradigm. Nutrients. 2020; 12(8):2314. https://doi.org/10.3390/nu12082314
Chicago/Turabian StyleLouise, Jennie, Andrea R. Deussen, and Jodie M. Dodd. 2020. "Gestational Weight Gain—Re-Examining the Current Paradigm" Nutrients 12, no. 8: 2314. https://doi.org/10.3390/nu12082314
APA StyleLouise, J., Deussen, A. R., & Dodd, J. M. (2020). Gestational Weight Gain—Re-Examining the Current Paradigm. Nutrients, 12(8), 2314. https://doi.org/10.3390/nu12082314




