The Effect of High Polyphenol Extra Virgin Olive Oil on Blood Pressure and Arterial Stiffness in Healthy Australian Adults: A Randomized, Controlled, Cross-Over Study
Abstract
1. Introduction
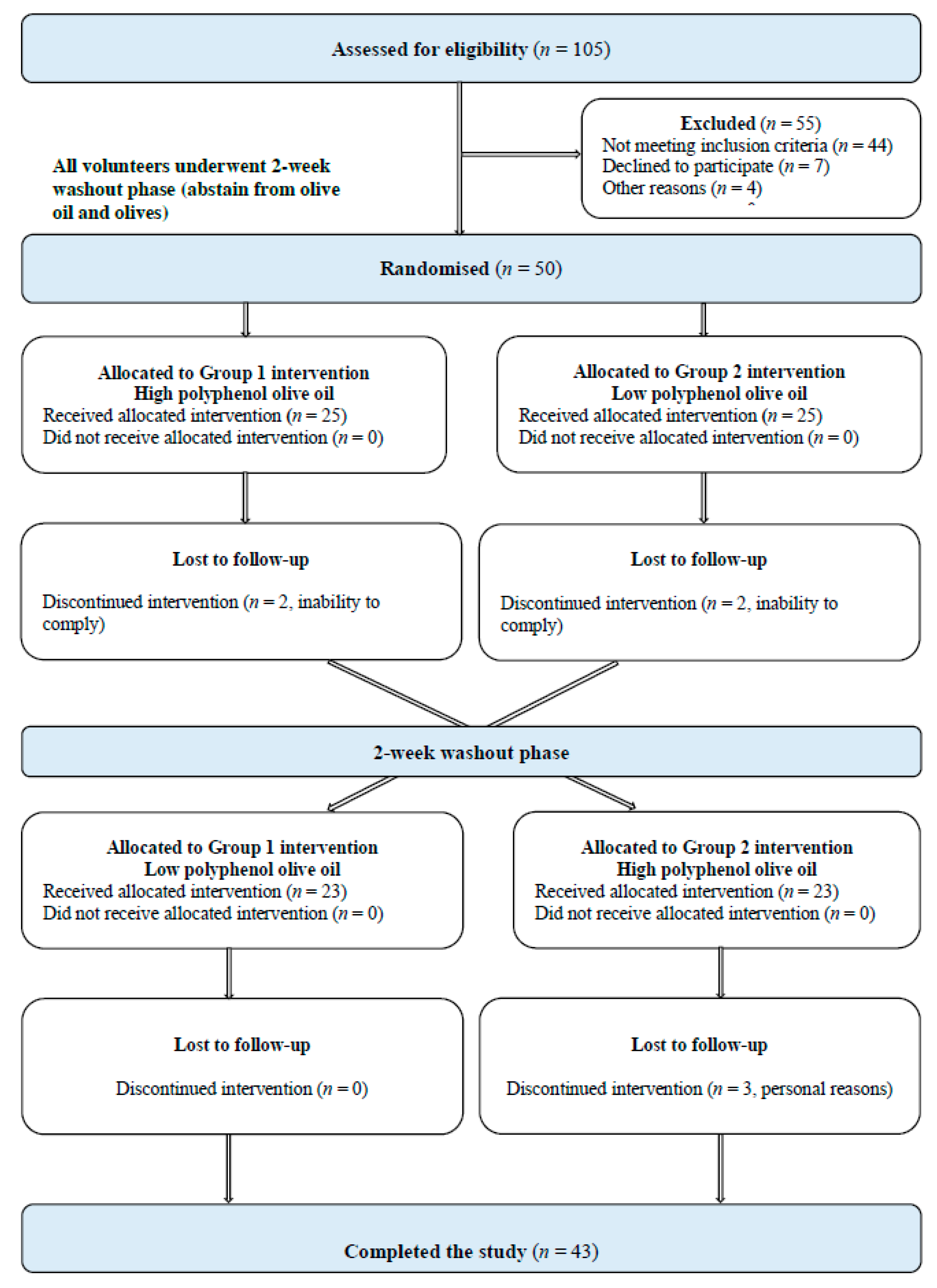
2. Materials and Methods
2.1. Study Population
2.2. Study Design and Procedure
2.3. Measurements
2.3.1. Socio-Demographics, Use of Medication and Dietary Supplements
2.3.2. Dietary Intake
2.3.3. Physical Activity
2.3.4. Anthropometric Measurements
2.3.5. Hemodynamic Indices
Blood Pressure
Arterial Stiffness
2.4. Sample Size Calculation
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics of Study Participants
3.2. Effect of LPOO and HPOO on Dietary Intake and Physical Activity
3.3. Effect of LPOO and HPOO on Anthropometrics
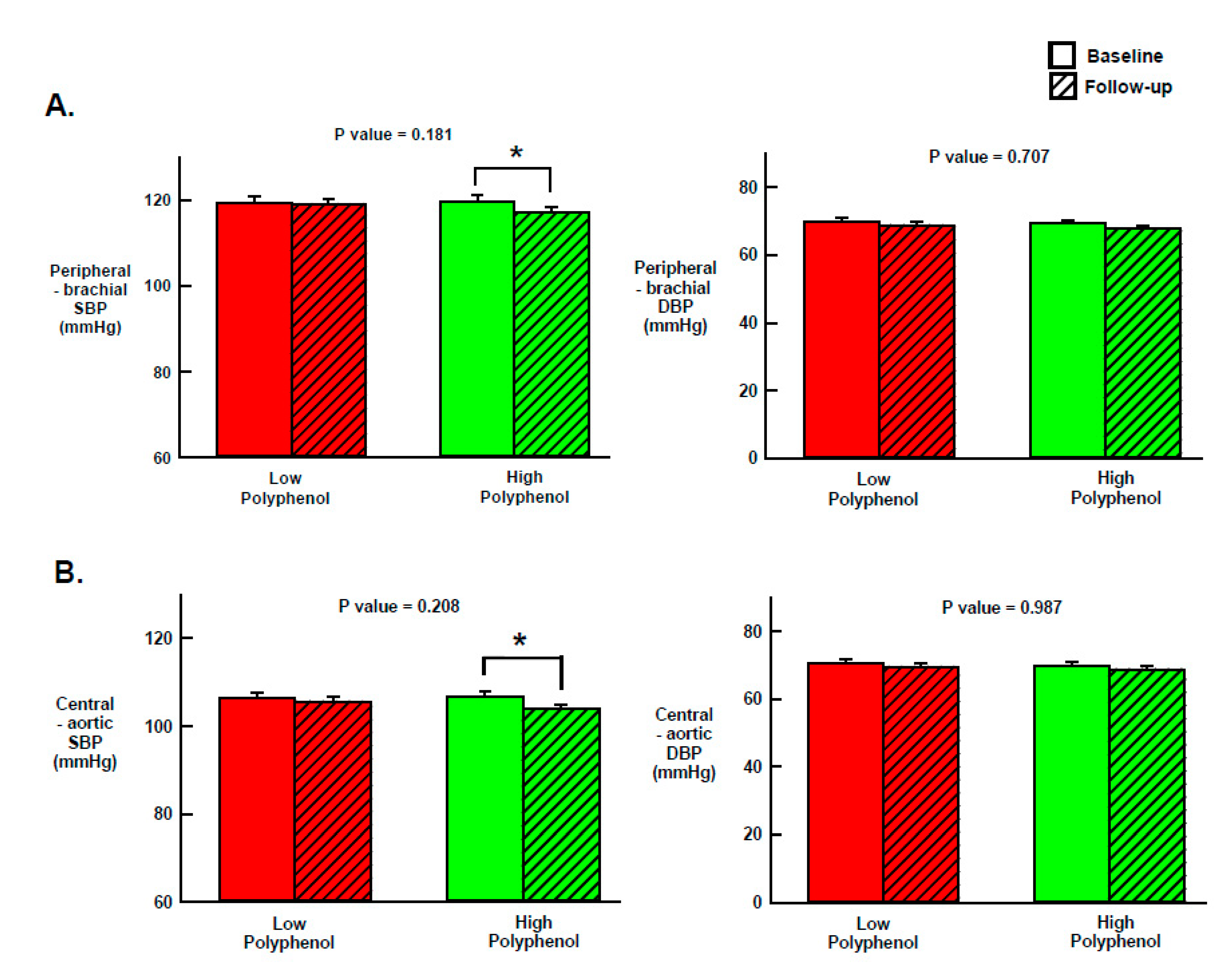
3.4. Effect of LPOO and HPOO on Peripheral BP, Central BP and Arterial Stiffness
3.5. Compliance to Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tzoulaki, I.; Elliott, P.; Kontis, V.; Ezzati, M. Worldwide Exposures to Cardiovascular Risk Factors and Associated Health Effects: Current Knowledge and Data Gaps. Circulation 2016, 133, 2314–2333. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. National Health Survey: First Results 2017–2018. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/4364.0.55.001~2017-18~Main%20Features~Heart,%20stroke%20and%20vascular%20disease~55 (accessed on 16 December 2019).
- Australian Institute of Health and Welfare. The Active Australia Survey: A Guide and Manual for Implementation, Analysis and Reporting. Available online: https://www.aihw.gov.au/reports/physical-activity/active-australia-survey/contents/table-of-contents (accessed on 16 December 2019).
- Li, W.F.; Huang, Y.Q.; Feng, Y.Q. Association between central haemodynamics and risk of all-cause mortality and cardiovascular disease: A systematic review and meta-analysis. J. Hum. Hypertens 2019, 33, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Zieman, S.J.; Melenovsky, V.; Kass, D.A. Mechanisms, pathophysiology, and therapy of arterial stiffness. Arter. Thromb. Vasc. Biol. 2005, 25, 932–943. [Google Scholar] [CrossRef]
- Shirwany, N.A.; Zou, M.H. Arterial stiffness: A brief review. Acta Pharm. Sin. 2010, 31, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Van Bortel, L.M.; Laurent, S.; Boutouyrie, P.; Chowienczyk, P.; Cruickshank, J.K.; De Backer, T.; Filipovsky, J.; Huybrechts, S.; Mattace-Raso, F.U.; Protogerou, A.D.; et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J. Hypertens 2012, 30, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, G.; Meyer, D.; Hardman, R.J.; Macpherson, H.; Scholey, A.B.; Pipingas, A. Physical Fitness and Aortic Stiffness Explain the Reduced Cognitive Performance Associated with Increasing Age in Older People. J. Alzheimers Dis. 2018, 63, 1307–1316. [Google Scholar] [CrossRef]
- Correction to: 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2018, 72, e33. [CrossRef]
- Collins, C.; Burrows, T.; Rollo, M. Dietary Patterns and Cardiovascular Disease Outcomes: An Evidence Check rapid review brokered by the Sax Institute for the National Heart Foundation of Australia. Available online: http://www.saxinstitute.org.au (accessed on 2 June 2020).
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing evidence on benefits of adherence to the Mediterranean diet on health: An updated systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1189–1196. [Google Scholar] [CrossRef]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Covas, M.I.; Fiol, M.; Gomez-Gracia, E.; Lopez-Sabater, M.C.; Vinyoles, E.; et al. Effects of a Mediterranean-style diet on cardiovascular risk factors: A randomized trial. Ann. Intern. Med. 2006, 145, 1–11. [Google Scholar] [CrossRef]
- Davis, C.R.; Hodgson, J.M.; Woodman, R.; Bryan, J.; Wilson, C.; Murphy, K.J. A Mediterranean diet lowers blood pressure and improves endothelial function: Results from the MedLey randomized intervention trial. Am. J. Clin. Nutr. 2017, 105, 1305–1313. [Google Scholar] [CrossRef]
- Mahmud, A.; Feely, J. Divergent effect of acute and chronic alcohol on arterial stiffness. Am. J. Hypertens 2002, 15, 240–243. [Google Scholar] [CrossRef]
- Karatzi, K.N.; Papamichael, C.M.; Karatzis, E.N.; Papaioannou, T.G.; Aznaouridis, K.A.; Katsichti, P.P.; Stamatelopoulos, K.S.; Zampelas, A.; Lekakis, J.P.; Mavrikakis, M.E. Red wine acutely induces favorable effects on wave reflections and central pressures in coronary artery disease patients. Am. J. Hypertens 2005, 18, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, C.M.; Karatzi, K.N.; Papaioannou, T.G.; Karatzis, E.N.; Katsichti, P.; Sideris, V.; Zakopoulos, N.; Zampelas, A.; Lekakis, J.P. Acute combined effects of olive oil and wine on pressure wave reflections: Another beneficial influence of the Mediterranean diet antioxidants? J. Hypertens 2008, 26, 223–229. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Naska, A.; Orfanos, P.; Trichopoulos, D.; Mountokalakis, T.; Trichopoulou, A. Olive oil, the Mediterranean diet, and arterial blood pressure: The Greek European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am. J. Clin. Nutr. 2004, 80, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Fito, M.; Cladellas, M.; de la Torre, R.; Marti, J.; Alcantara, M.; Pujadas-Bastardes, M.; Marrugat, J.; Bruguera, J.; Lopez-Sabater, M.C.; Vila, J.; et al. Antioxidant effect of virgin olive oil in patients with stable coronary heart disease: A randomized, crossover, controlled, clinical trial. Atherosclerosis 2005, 181, 149–158. [Google Scholar] [CrossRef] [PubMed]
- George, E.S.; Marshall, S.; Mayr, H.L.; Trakman, G.L.; Tatucu-Babet, O.A.; Lassemillante, A.M.; Bramley, A.; Reddy, A.J.; Forsyth, A.; Tierney, A.C.; et al. The effect of high-polyphenol extra virgin olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2019, 59, 2772–2795. [Google Scholar] [CrossRef] [PubMed]
- Marx, W.; George, E.S.; Mayr, H.L.; Thomas, C.J.; Sarapis, K.; Moschonis, G.; Kennedy, G.; Pipingas, A.; Willcox, J.C.; Prendergast, L.A.; et al. Effect of high polyphenol extra virgin olive oil on markers of cardiovascular disease risk in healthy Australian adults (OLIVAUS): A protocol for a double-blind randomised, controlled, cross-over study. Nutr. Diet. 2019. [Google Scholar] [CrossRef]
- Ghanbari, R.; Anwar, F.; Alkharfy, K.M.; Gilani, A.H.; Saari, N. Valuable nutrients and functional bioactives in different parts of olive (Olea europaea L.)—A review. Int. J. Mol. Sci. 2012, 13, 3291–3340. [Google Scholar] [CrossRef]
- Souza, P.A.L.; Marcadenti, A.; Portal, V.L. Effects of Olive Oil Phenolic Compounds on Inflammation in the Prevention and Treatment of Coronary Artery Disease. Nutrients 2017, 9. [Google Scholar] [CrossRef]
- Godos, J.; Sinatra, D.; Blanco, I.; Mulè, S.; La Verde, M.; Marranzano, M. Association between Dietary Phenolic Acids and Hypertension in a Mediterranean Cohort. Nutrients 2017, 9, 1069. [Google Scholar] [CrossRef]
- Moreno-Luna, R.; Muñoz-Hernandez, R.; Miranda, M.L.; Costa, A.F.; Jimenez-Jimenez, L.; Vallejo-Vaz, A.J.; Muriana, F.J.G.; Villar, J.; Stiefel, P. Olive Oil Polyphenols Decrease Blood Pressure and Improve Endothelial Function in Young Women with Mild Hypertension. Am. J. Hypertens. 2012, 25, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Krause, M.; Schmucker, C.; Hoffmann, G.; Rucker, G.; Meerpohl, J.J. Impact of different types of olive oil on cardiovascular risk factors: A systematic review and network meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1030–1039. [Google Scholar] [CrossRef]
- Hohmann, C.D.; Cramer, H.; Michalsen, A.; Kessler, C.; Steckhan, N.; Choi, K.; Dobos, G. Effects of high phenolic olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Phytomedicine 2015, 22, 631–640. [Google Scholar] [CrossRef]
- Sarapis, K.; Thomas, C.J.; Shivantha, S.; George, E.S.; Marx, W.; Mayr, H.L.; Kennedy, G.; Pipingas, A.; Willcox, J.C.; Prendergast, L.A.; et al. The Effect of High Polyphenol Extra Virgin Olive Oil on Markers of Cardiovascular Disease Risk in Healthy Australian Adults (OLIVAUS): Results from a double blind, randomised, controlled cross-over feasibility pilot study. In Proceedings of the Dietitians Association of Australia 36th National Conference, Gold Coast, Australia, 12–14 August 2019. [Google Scholar]
- WHO. Physical status: The use and interpretation of anthropometry. In Report of a WHO Expert Consultation; World Health Organization: Geneva, The Swiss, 1995. [Google Scholar]
- WHO. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation; World Health Organization: Geneva, The Swiss, 8–11 December 2008. [Google Scholar]
- Hernaez, A.; Fernandez-Castillejo, S.; Farras, M.; Catalan, U.; Subirana, I.; Montes, R.; Sola, R.; Munoz-Aguayo, D.; Gelabert-Gorgues, A.; Diaz-Gil, O.; et al. Olive oil polyphenols enhance high-density lipoprotein function in humans: A randomized controlled trial. Arter. Thromb Vasc. Biol. 2014, 34, 2115–2119. [Google Scholar] [CrossRef] [PubMed]
- Bondia-Pons, I.; Schroder, H.; Covas, M.I.; Castellote, A.I.; Kaikkonen, J.; Poulsen, H.E.; Gaddi, A.V.; Machowetz, A.; Kiesewetter, H.; Lopez-Sabater, M.C. Moderate consumption of olive oil by healthy European men reduces systolic blood pressure in non-Mediterranean participants. J. Nutr. 2007, 137, 84–87. [Google Scholar] [CrossRef]
- Martin-Pelaez, S.; Castaner, O.; Konstantinidou, V.; Subirana, I.; Munoz-Aguayo, D.; Blanchart, G.; Gaixas, S.; de la Torre, R.; Farre, M.; Saez, G.T.; et al. Effect of olive oil phenolic compounds on the expression of blood pressure-related genes in healthy individuals. Eur. J. Nutr. 2017, 56, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Rodriguez, E.; Lima-Cabello, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Roca, M.; Espejo-Calvo, J.A.; Gil-Extremera, B.; Soria-Florido, M.; de la Torre, R.; et al. Effects of Virgin Olive Oils Differing in Their Bioactive Compound Contents on Metabolic Syndrome and Endothelial Functional Risk Biomarkers in Healthy Adults: A Randomized Double-Blind Controlled Trial. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Castaner, O.; Fito, M.; Lopez-Sabater, M.C.; Poulsen, H.E.; Nyyssonen, K.; Schroder, H.; Salonen, J.T.; De la Torre-Carbot, K.; Zunft, H.F.; De la Torre, R.; et al. The effect of olive oil polyphenols on antibodies against oxidized LDL. A randomized clinical trial. Clin. Nutr. 2011, 30, 490–493. [Google Scholar] [CrossRef]
- Protogerou, A.D.; Papaioannou, T.G.; Blacher, J.; Papamichael, C.M.; Lekakis, J.P.; Safar, M.E. Central blood pressures: Do we need them in the management of cardiovascular disease? Is it a feasible therapeutic target? J. Hypertens 2007, 25, 265–272. [Google Scholar] [CrossRef]
- Tanaka, H.; Safar, M.E. Influence of lifestyle modification on arterial stiffness and wave reflections. Am. J. Hypertens 2005, 18, 137–144. [Google Scholar] [CrossRef]
- McCarron, D.A.; Reusser, M.E. Body weight and blood pressure regulation. Am. J. Clin. Nutr. 1996, 63, 423s–425s. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.J.; Ho, S.C.; Su, X.; Liu, Z.M. Changes in Body Weight From Young Adulthood to Middle Age and Its Association With Blood Pressure and Hypertension: A Cross-Sectional Study in Hong Kong Chinese Women. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- D’Elia, L.; La Fata, E.; Giaquinto, A.; Strazzullo, P.; Galletti, F. Effect of dietary salt restriction on central blood pressure: A systematic review and meta-analysis of the intervention studies. J. Clin. Hypertens (Greenwich) 2020. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yin, L.; Peng, Y.; Liu, X.; Cao, X.; Wang, Y.; Yang, P.; Li, X.; Chen, Z. The association of blood pressure with estimated urinary sodium, potassium excretion and their ratio in hypertensive, normotensive, and hypotensive Chinese adults. Asia Pac. J. Clin. Nutr. 2020, 29, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Schutten, J.C.; Joosten, M.M.; de Borst, M.H.; Bakker, S.J.L. Magnesium and Blood Pressure: A Physiology-Based Approach. Adv. Chronic Kidney Dis. 2018, 25, 244–250. [Google Scholar] [CrossRef]
- Pamuk, N.; Akkan, T.; Dagdeviren, M.; Koca, A.O.; Beyan, E.; Ertugrul, D.T.; Altay, M. Central and peripheral blood pressures and arterial stiffness increase in hypoparathyroidism. Arch. Endocrinol Metab. 2020. [Google Scholar] [CrossRef]
- Tripoli, E.; Giammanco, M.; Tabacchi, G.; Di Majo, D.; Giammanco, S.; La Guardia, M. The phenolic compounds of olive oil: Structure, biological activity and beneficial effects on human health. Nutr. Res. Rev. 2005, 18, 98–112. [Google Scholar] [CrossRef]
- Silva, S.; Bronze, M.R.; Figueira, M.E.; Siwy, J.; Mischak, H.; Combet, E.; Mullen, W. Impact of a 6-wk olive oil supplementation in healthy adults on urinary proteomic biomarkers of coronary artery disease, chronic kidney disease, and diabetes (types 1 and 2): A randomized, parallel, controlled, double-blind study. Am. J. Clin. Nutr. 2015, 101, 44–54. [Google Scholar] [CrossRef]
- Sonmez, A.; Celebi, G.; Erdem, G.; Tapan, S.; Genc, H.; Tasci, I.; Ercin, C.N.; Dogru, T.; Kilic, S.; Uckaya, G.; et al. Plasma apelin and ADMA Levels in patients with essential hypertension. Clin. Exp. Hypertens 2010, 32, 179–183. [Google Scholar] [CrossRef]
| Total Sample (n = 50) | Males (n = 17) | Females (n = 33) | p-Value * | |
|---|---|---|---|---|
| Socio-Demographics | Mean (SD) | Mean (SD) | Mean (SD) | |
| Age (years) | 38.5 (13.9) | 33.4 (11.6) | 41.2 (14.4) | 0.058 |
| Education (years) | 17.3 (3.5) | 17.4 (3.7) | 17.2 (3.5) | 0.895 |
| Highest Level of Education | n (%) | n (%) | n (%) | 0.241 |
| Secondary School | 2 (4.0) | 0 (0.0) | 2 (6.1) | |
| Tertiary | 43 (86.0) | 17 (100.0) | 26 (78.8) | |
| Trade | 2 (4.0) | 0 (0.0) | 2 (6.1) | |
| Other | 3 (6.0) | 0 (0.0) | 3 (9.1) | |
| Country of Birth | 0.798 | |||
| Australia, NZ, Pacific Islanders | 35 (70.0) | 11 (64.7) | 24 (72.7) | |
| Europe | 5 (10.0) | 2 (11.8) | 3 (9.1) | |
| South America | 4 (8.0) | 1 (5.9) | 3 (9.1) | |
| Middle East and Asia | 6 (12.0) | 3 (17.6) | 3 (9.1) | |
| Anthropometrics | Mean (SD) | Mean (SD) | Mean (SD) | |
| Height (cm) | 168.9 (9.6) | 179.3 (6.8) | 163.6 (5.8) | <0.001 |
| Weight (Kg) | 70.7 (12.8) | 79.6 (9.6) | 66.1 (11.9) | <0.001 |
| BMI (kg/m2) | 24.7 (3.5) | 24.7 (2.4) | 24.6 (3.9) | 0.915 |
| Waist Circumference (cm) | 86.9 (11.2) | 88.9 (8.7) | 85.9 (12.3) | 0.364 |
| Weight Status Categories † | n (%) | n (%) | n (%) | |
| Underweight | 1 (2.0) | 1 (3.0) | 0 (0) | 0.649 |
| Normal Weight | 25 (50.0) | 16 (48.5) | 9 (52.9) | |
| Overweight | 22 (44.0) | 14 (42.4) | 8 (47.1) | |
| Obese | 2 (4.0) | 2 (6.1) | 0 (0) | |
| Waist Circumference Categories ‡ | ||||
| Normal | 25 (50.0) | 13 (39.4) | 12 (70.6) | 0.105 |
| High Risk | 8 (16.0) | 6 (18.2) | 2 (11.8) | |
| Very High Risk | 17 (34.0) | 14 (42.4) | 3 (17.6) | |
| Hemodynamic Indices | ||||
| Peripheral Blood Pressure | Mean (SD) | Mean (SD) | Mean (SD) | |
| Peripheral SBP (mmHg) | 120.0 (13.4) | 121.7 (9.1) | 119.1 (15.2) | 0.454 |
| Peripheral DBP (mmHg) | 69.9 (8.4) | 69.7 (8.9) | 70.0 (8.3) | 0.904 |
| Peripheral Blood Pressure Categories § | n (%) | n (%) | n (%) | |
| Normal Blood Pressure | 27 (54.0) | 20 (60.6) | 7 (41.2) | 0.399 |
| Elevated Blood Pressure | 9 (18.0) | 4 (12.1) | 5 (29.4) | |
| Hypertension Stage 1 | 10 (20.0) | 6 (18.2) | 4 (23.5) | |
| Hypertension Stage 2 | 4 (8.0) | 3 (9.1) | 1 (5.9) | |
| Central Blood Pressure | Mean (SD) | Mean (SD) | Mean (SD) | |
| Central Aortic SBP (mmHg) | 106.8 (13.3) | 106.9 (8.5) | 106.8 (15.3) | 0.971 |
| Central Aortic DBP (mmHg) | 70.6 (8.7) | 70.3 (9.1) | 70.8 (9.7) | 0.843 |
| Pulse Pressure (mmHg) | 36.0 (8.9) | 36.2 (7.9) | 35.9 (9.5) | 0.930 |
| Heart Rate (bpm) | 61.5 (10.2) | 58.1 (8.9) | 63.2 (10.4) | 0.092 |
| Systemic Arterial Stiffness | ||||
| Augmented Pressure (mmHg) | 6.8 (6.8) | 4.8 (4.2) | 7.8 (7.6) | 0.077 |
| Augmented Index (%) | 16.6 (14.9) | 12.2 (9.4) | 18.9 (16.8) | 0.077 |
| Pulse Wave Velocity (m/s) | 9.5 (1.4) | 9.5 (1.3) | 9.5 (1.5) | 0.933 |
| Baseline Mean (SEM) | Follow Up Mean (SEM) | Mean Change (95% CI) (Time Effect) | p-Value (Treatment * Time Effect) | |
|---|---|---|---|---|
| Energy intake (KJ/day) | ||||
| Low Polyphenol OO (n = 50) | 8712.8 (328.3) | 10518.9 (344.4) | 1806.1 (1075.4 to 2536.8) | 0.940 |
| High Polyphenol OO (n = 50) | 8892.6 (328.3) | 10659.2 (344.4) | 1766.6 (1035.9 to 2497.3) | |
| p-value (Treatment effect) | 0.700 | 0.774 | ||
| Protein intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 102.0 (5.5) | 100.7 (5.2) | −1.3 (−14.3 to 11.8) | 0.924 |
| High Polyphenol OO (n = 50) | 97.4 (5.5) | 97.0 (5.3) | −0.4 (−13.4 to 12.7) | |
| p-value (Treatment effect) | 0.558 | 0.619 | ||
| Carbohydrates (g/day) | ||||
| Low Polyphenol OO (n = 50) | 214.8 (10.1) | 213.4 (11.1) | −1.5 (−23.5 to 20.6) | 0.972 |
| High Polyphenol OO (n = 50) | 219.9 (10.1) | 217.8 (11.1) | −2.0 (−24.0 to 20.0) | |
| p-value (Treatment effect) | 0.726 | 0.776 | ||
| Total fat intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 79.9 (4.0) | 129.2 (4.5) | 49.3 (41.1 to 57.4) | 0.571 |
| High Polyphenol OO (n = 50) | 84.3 (4.0) | 130.3 (4.5) | 46.0 (37.8 to 54.1) | |
| p-value (Treatment effect) | 0.441 | 0.870 | ||
| SFA intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 27.7 (1.5) | 35.1 (1.9) | 7.4 (4.0 to 10.8) | 0.707 |
| High Polyphenol OO (n = 50) | 28.8 (1.5) | 35.3 (1.9) | 6.5 (3.1 to 9.9) | |
| p-value (Treatment effect) | 0.620 | 0.953 | ||
| MUFA intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 30.6 (1.7) | 67.3 (1.9) | 36.8 (33.2 to 40.3) | 0.514 |
| High Polyphenol OO (n = 50) | 31.8 (1.7) | 67.0 (1.9) | 35.1 (31.6 to 38.6) | |
| p-value (Treatment effect) | 0.605 | 0.877 | ||
| PUFA intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 14.6 (1.0) | 17.7 (1.0) | 3.1 (1.0 to 5.1) | 0.971 |
| High Polyphenol OO (n = 50) | 15.7 (1.0) | 18.7 (1.0) | 3.0 (1.0 to 5.1) | |
| p-value (Treatment effect) | 0.483 | 0.469 | ||
| Fibre intake (g/day) | ||||
| Low Polyphenol OO (n = 50) | 29.7 (1.7) | 30.7 (1.8) | 0.9 (−3.1 to 4.9) | 0.314 |
| High Polyphenol OO (n = 50) | 29.6 (1.7) | 33.5 (1.8) | 3.8 (−0.2 to 7.8) | |
| p-value (Treatment effect) | 0.963 | 0.268 | ||
| Sodium intake (mg/day) | ||||
| Low Polyphenol OO (n = 50) | 2611.1 (269.5) | 2287.1 (168.9) | −324.0 (−878.2 to 230.3) | 0.994 |
| High Polyphenol OO (n = 50) | 3096.7 (269.5) | 2775.5 (168.9) | −321.2 (−875.5 to 233.1) | |
| p-value (Treatment effect) | 0.206 | 0.044 | ||
| Potassium intake (mg/day) | ||||
| Low Polyphenol OO (n = 50) | 3486,7 (227.0) | 3389.3 (170.5) | −97.3 (−631.1 to 436.4) | 0.488 |
| High Polyphenol OO (n = 50) | 3334.4 (227.0) | 3501.9 (170.5) | 167.5 (−366.2 to 701.3) | |
| p-value (Treatment effect) | 0.636 | 0.642 | ||
| Magnesium intake (mg/day) | ||||
| Low Polyphenol OO (n = 50) | 574.2 (92.9) | 446.0 (18.9) | −128.2 (−308.4 to 52.0) | 0.271 |
| High Polyphenol OO (n = 50) | 433.6 (92.9) | 447.6 (18.9) | 14.0 (−166.2 to 194.2) | |
| p-value (Treatment effect) | 0.287 | 0.953 | ||
| Calcium intake (mg/day) | ||||
| Low Polyphenol OO (n = 50) | 1005.0 (92.3) | 1056.2 (95.6) | 51.1 (−205.1 to 307.3) | 0.916 |
| High Polyphenol OO (n = 50) | 977.3 (92.3) | 1009.2 (95.6) | 31.9 (−224.3 to 288.1) | |
| p-value (Treatment effect) | 0.832 | 0.729 | ||
| Caffeine intake (mg/day) | ||||
| Low Polyphenol OO (n = 46) | 199.4 (54.1) | 182.0 (35.6) | −17.4 (−122.3 to 87.6) | 0.612 |
| High Polyphenol OO (n = 43) | 242.8 (56.0) | 186.7 (36.8) | −56.1 (−164.6 to 52.5) | |
| p-value (Treatment effect) | 0.578 | 0.926 |
| Baseline Mean (SEM) | Follow Up Mean (SEM) | Mean Change (95% CI) (Time Effect) | p-Value (Treatment * Time Effect) | |
|---|---|---|---|---|
| Weight (kg) | ||||
| Low Polyphenol OO (n = 50) | 70.8 (1.5) | 71.2 (1.5) | 0.4 (0.2 to 0.7) | 0.163 |
| High Polyphenol OO (n = 50) | 70.7 (1.5) | 70.9 (1.5) | 0.2 (−0.1 to 0.4) | |
| p-value (Treatment * effect) | 0.993 | 0.902 | ||
| Height (cm) | ||||
| Low Polyphenol OO (n = 50) | 168.9 (0.9) | 169.0 (0.9) | 0.1 (−0.2 to 0.4) | 0.890 |
| High Polyphenol OO (n = 50) | 168.9 (0.9) | 169.0 (0.9) | 0.1 (−0.1 to 0.4) | |
| p-value (Treatment effect) | 0.974 | 0.992 | ||
| BMI (kg/m2) | ||||
| Low Polyphenol OO (n = 50) | 24.7 (0.4) | 24.8 (0.4) | 0.1 (−0.01 to 0.2) | 0.305 |
| High Polyphenol OO (n = 50) | 24.7 (0.4) | 24.7 (0.4) | 0.02 (−0.1 to 0.1) | |
| p-value (Treatment effect) | 0.993 | 0.897 | ||
| Waist circumference (cm) | ||||
| Low Polyphenol OO (n = 50) | 87.1 (1.3) | 87.4 (1.2) | 0.3 (−0.1 to 0.7) | 0.501 |
| High Polyphenol OO (n = 50) | 87.1 (1.3) | 87.3 (1.2) | 0.1 (−0.2 to 0.5) | |
| p-value (Treatment effect) | 1.000 | 0.919 |
| Baseline Mean (SEM) | Follow Up Mean (SEM) | Mean Change (95% CI) (Time Effect) | p-Value (Treatment * Time Effect) | |
|---|---|---|---|---|
| Pulse pressure (mmHg) | ||||
| Low Polyphenol OO (n = 50) | 35.7 (1.0) | 36.4 (1.0) | 0.7 (−0.8 to 2.1) | 0.296 |
| High Polyphenol OO (n = 50) | 36.3 (1.0) | 35.9 (1.0) | −0.4 (−1.9 to 1.1) | |
| p-value (Treatment effect) | 0.653 | 0.723 | ||
| Pulse rate (bpm) | ||||
| Low Polyphenol OO (n = 50) | 61.1 (1.3) | 59.4 (1.4) | −1.7 (−3.9 to 0.4) | 0.403 |
| High Polyphenol OO (n = 50) | 61.0 (1.3) | 60.6 (1.4) | −0.4 (−2.6 to 1.7) | |
| p-value (Treatment effect) | 0.954 | 0.553 | ||
| Augmented pressure (mmHg) | ||||
| Low Polyphenol OO (n = 50) | 6.5 (0.7) | 6.0 (0.7) | −0.5 (−1.6 to 0.6) | 0.987 |
| High Polyphenol OO (n = 50) | 6.9 (0.7) | 6.3 (0.7) | −0.5 (−1.6 to 0.5) | |
| p-value (Treatment effect) | 0.692 | 0.714 | ||
| Augmented index (%) | ||||
| Low Polyphenol OO (n = 50) | 16.2 (1.7) | 14.6 (1.8) | −1.7 (−4.2 to 0.8) | 0.807 |
| High Polyphenol OO (n = 50) | 16.6 (1.7) | 15.4 (1.8) | −1.2 (−3.7 to 1.3) | |
| p-value (Treatment effect) | ||||
| Pulse wave velocity (m/s) | ||||
| Low Polyphenol OO (n = 50) | 9.6 (0.1) | 9.5 (0.1) | −0.03 (−0.3 to 0.2) | 0.926 |
| High Polyphenol OO (n = 50) | 9.5 (0.1) | 9.4 (0.1) | −0.05 (−0.3 to 0.2) | |
| p-value (Treatment effect) | 0.679 | 0.608 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarapis, K.; Thomas, C.J.; Hoskin, J.; George, E.S.; Marx, W.; Mayr, H.L.; Kennedy, G.; Pipingas, A.; Willcox, J.C.; Prendergast, L.A.; et al. The Effect of High Polyphenol Extra Virgin Olive Oil on Blood Pressure and Arterial Stiffness in Healthy Australian Adults: A Randomized, Controlled, Cross-Over Study. Nutrients 2020, 12, 2272. https://doi.org/10.3390/nu12082272
Sarapis K, Thomas CJ, Hoskin J, George ES, Marx W, Mayr HL, Kennedy G, Pipingas A, Willcox JC, Prendergast LA, et al. The Effect of High Polyphenol Extra Virgin Olive Oil on Blood Pressure and Arterial Stiffness in Healthy Australian Adults: A Randomized, Controlled, Cross-Over Study. Nutrients. 2020; 12(8):2272. https://doi.org/10.3390/nu12082272
Chicago/Turabian StyleSarapis, Katerina, Colleen J. Thomas, Johanna Hoskin, Elena S. George, Wolfgang Marx, Hannah L. Mayr, Greg Kennedy, Andrew Pipingas, Jane C. Willcox, Luke A. Prendergast, and et al. 2020. "The Effect of High Polyphenol Extra Virgin Olive Oil on Blood Pressure and Arterial Stiffness in Healthy Australian Adults: A Randomized, Controlled, Cross-Over Study" Nutrients 12, no. 8: 2272. https://doi.org/10.3390/nu12082272
APA StyleSarapis, K., Thomas, C. J., Hoskin, J., George, E. S., Marx, W., Mayr, H. L., Kennedy, G., Pipingas, A., Willcox, J. C., Prendergast, L. A., Itsiopoulos, C., & Moschonis, G. (2020). The Effect of High Polyphenol Extra Virgin Olive Oil on Blood Pressure and Arterial Stiffness in Healthy Australian Adults: A Randomized, Controlled, Cross-Over Study. Nutrients, 12(8), 2272. https://doi.org/10.3390/nu12082272







