Vitamin D Synthesis Following a Single Bout of Sun Exposure in Older and Younger Men and Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Baseline Measurements
2.3. Sunlight Exposure
2.4. Blood Draws and Analyses
2.5. Estimates of UV-Irradiance
2.6. Statistical Procedures
3. Results
3.1. Subject Characteristics
3.2. Sun Exposure and Estimates of UVB Irradiance
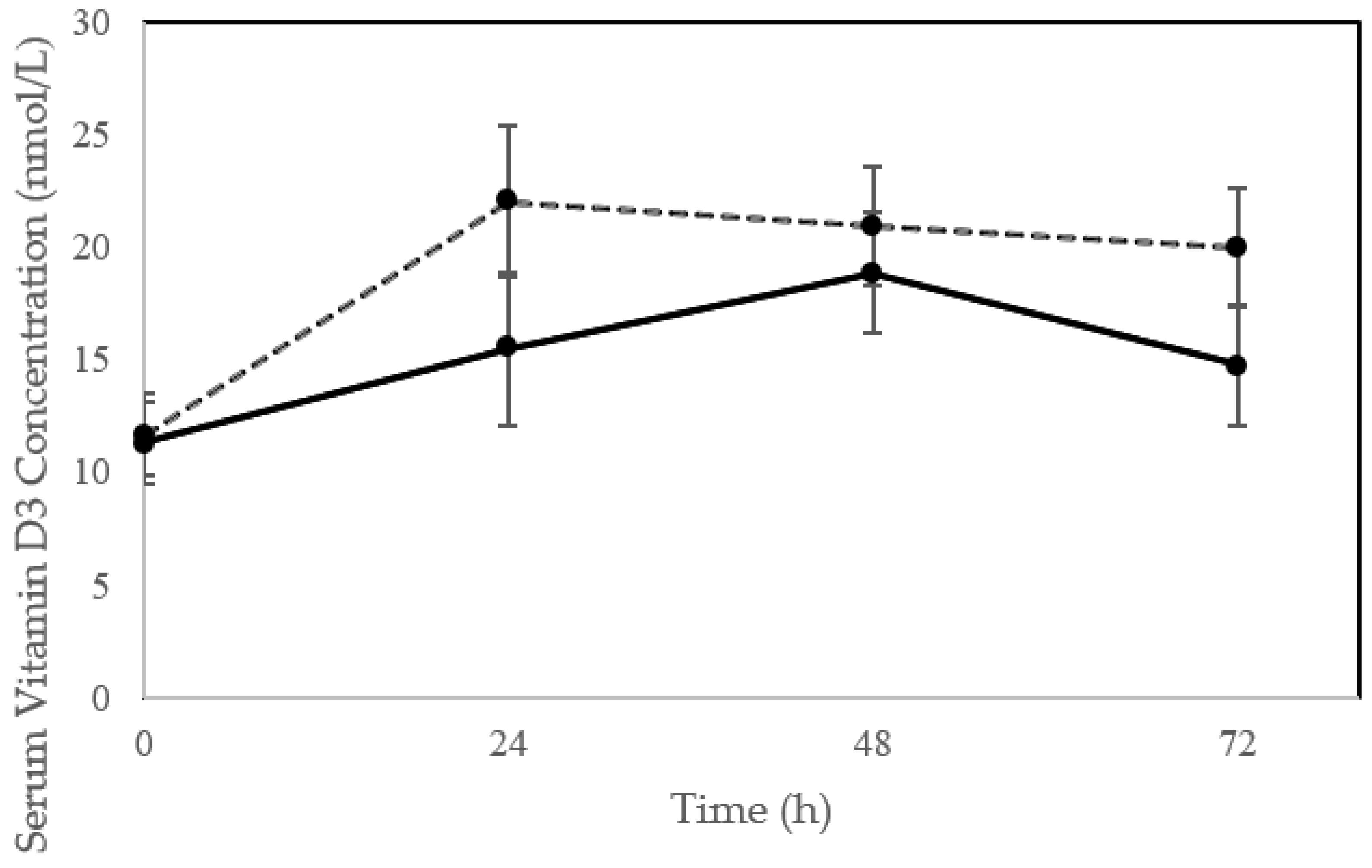
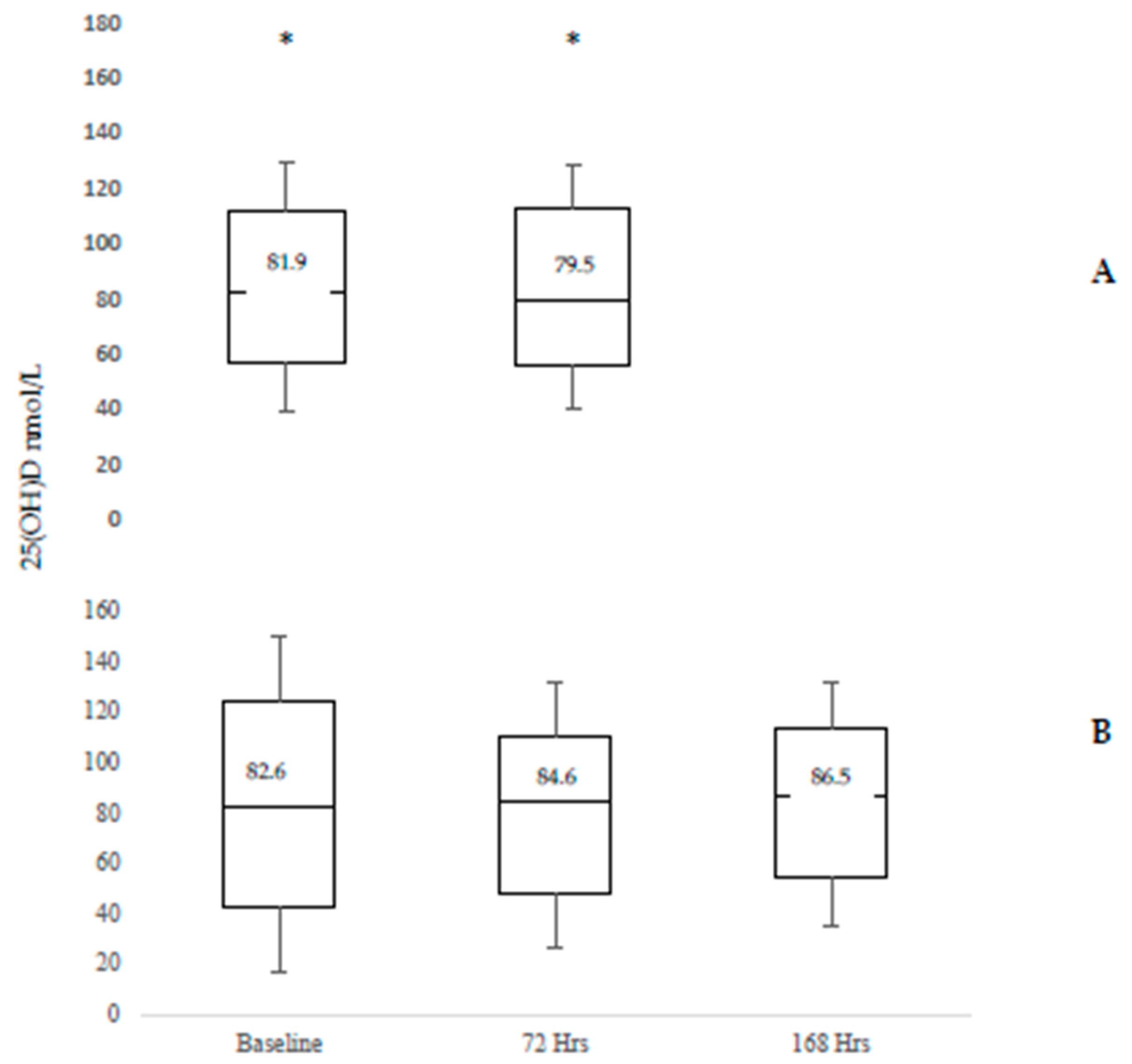
3.3. Impact of Sun Exposure on Serum D3 and 25(OH)D
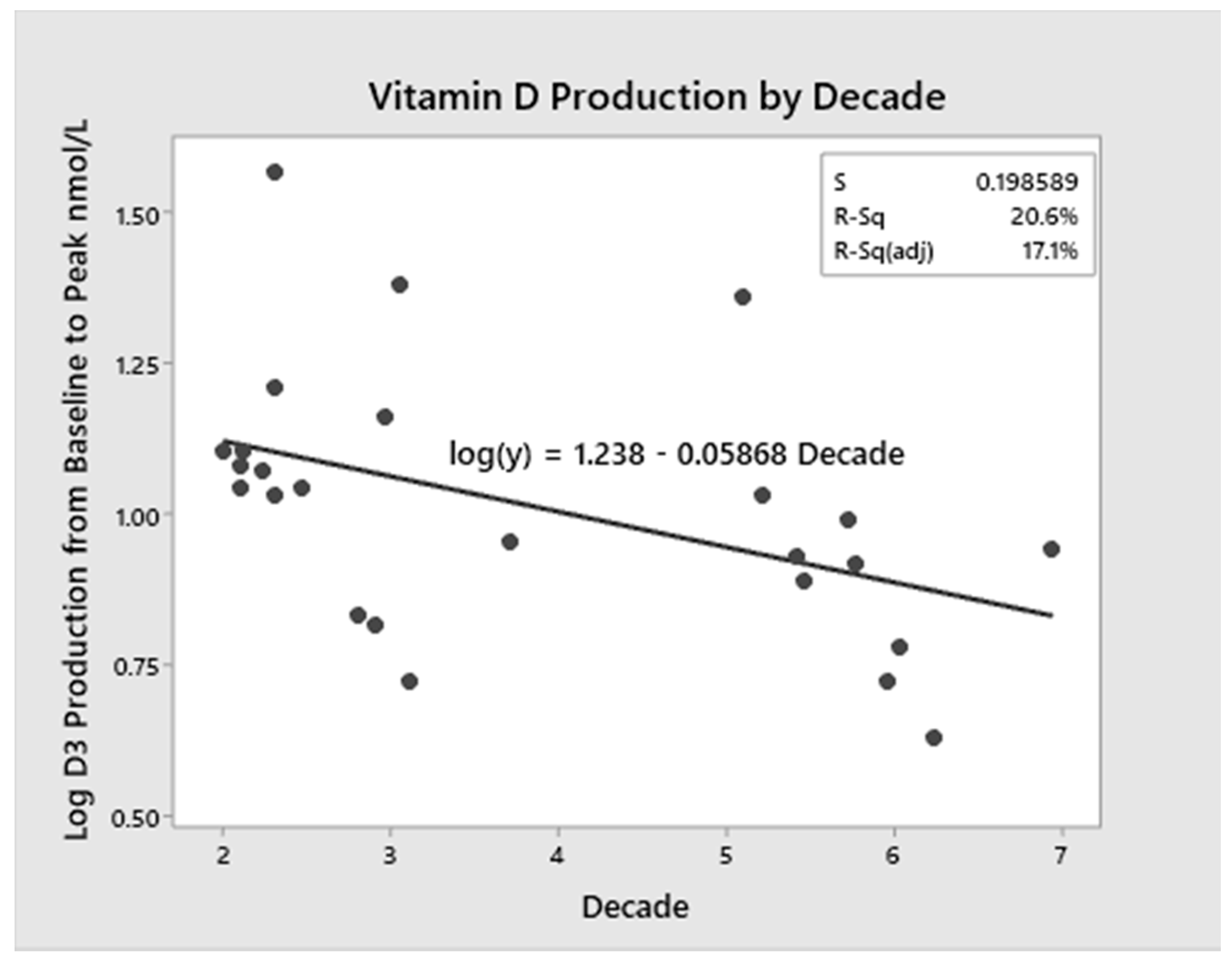
3.4. Predictors of D3 Production
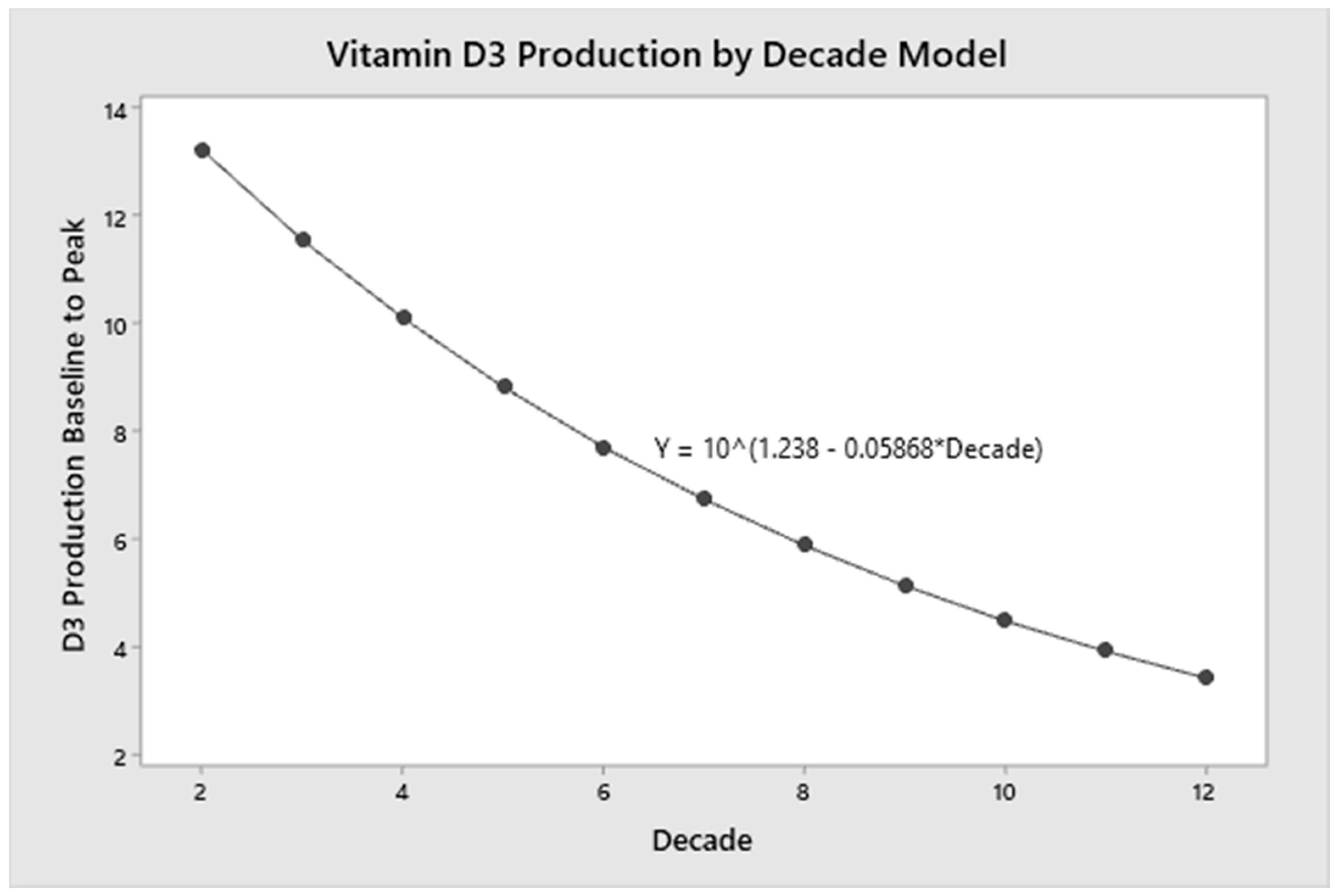
3.5. Modeling of D3 Production with Ageing
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Data Availability
Conflicts of Interest
References
- Wang, J.; Ma, J.; Cheng, G. An accident of acute hydrogen sulfide poisoning when cleaning up a salted vegetables pool. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi 2015, 33, 918–919. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Orces, C.H. Association between leisure-time aerobic physical activity and vitamin D concentrations among US older adults: The NHANES 2007–2012. Aging Clin. Exp. Res. 2019, 31, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Sherman, F.T. Vitamin D deficiency is rampant in older adults. Geriatrics 2008, 63, 9–11. [Google Scholar] [PubMed]
- Ginter, J.K.; Krithika, S.; Gozdzik, A.; Hanwell, H.; Whiting, S.; Parra, E.J. Vitamin D status of older adults of diverse ancestry living in the Greater Toronto Area. BMC Geriatr. 2013, 13, 66. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carrillo-Vega, M.F.; Garcia-Pena, C.; Gutierrez-Robledo, L.M.; Perez-Zepeda, M.U. Vitamin D deficiency in older adults and its associated factors: A cross-sectional analysis of the Mexican Health and Aging Study. Arch. Osteoporos. 2017, 12, 8. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Vaes, A.M.M.; van der Zwaluw, N.L.; van Wijngaarden, J.P.; Swart, K.M.A.; Ham, A.C.; van Dijk, S.C.; Enneman, A.W.; Sohl, E.; van Schoor, N.M.; et al. Relative importance of summer sun exposure, vitamin D intake, and genes to vitamin D status in Dutch older adults: The B-PROOF study. J. Steroid Biochem. Mol. Biol. 2016, 164, 168–176. [Google Scholar] [CrossRef]
- Herrick, K.A.; Storandt, R.J.; Afful, J.; Pfeiffer, C.M.; Schleicher, R.L.; Gahche, J.J.; Potischman, N. Vitamin D status in the United States, 2011-2014. Am. J. Clin. Nutr. 2019, 110, 150–157. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and Health: Evolution, Biologic Functions, and Recommended Dietary Intakes for Vitamin D. Vitamin D: Physiology, Molecular Biology, and Clinical Applications, 2nd ed.; Springer: Totowa, NJ, USA, 2010. [Google Scholar]
- Czekalla, C.; Schonborn, K.H.; Lademann, J.; Meinke, M.C. Noninvasive Determination of Epidermal and Stratum Corneum Thickness in vivo Using Two-Photon Microscopy and Optical Coherence Tomography: Impact of Body Area, Age, and Gender. Skin Pharmacol. Physiol. 2019, 32, 142–150. [Google Scholar] [CrossRef]
- Sauermann, K.; Clemann, S.; Jaspers, S.; Gambichler, T.; Altmeyer, P.; Hoffmann, K.; Ennen, J. Age related changes of human skin investigated with histometric measurements by confocal laser scanning microscopy in vivo. Skin Res. Technol. 2002, 8, 52–56. [Google Scholar] [CrossRef]
- MacLaughlin, J.; Holick, M.F. Aging decreases the capacity of human skin to produce vitamin D3. J. Clin. Investig. 1985, 76, 1536–1538. [Google Scholar] [CrossRef] [PubMed]
- Snijder, M.B.; van Dam, R.M.; Visser, M.; Deeg, D.J.; Dekker, J.M.; Bouter, L.M.; Seidell, J.C.; Lips, P. Adiposity in relation to vitamin D status and parathyroid hormone levels: A population-based study in older men and women. J. Clin. Endocrinol. Metab. 2005, 90, 4119–4123. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.R.; Granic, A.; Aspray, T.J. Vitamin D and Ageing. Subcell Biochem. 2018, 90, 191–220. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.S.; Heath, H., 3rd; Kumar, R.; Riggs, B.L. Impaired vitamin D metabolism with aging in women. Possible role in pathogenesis of senile osteoporosis. J. Clin. Investig. 1984, 73, 1668–1672. [Google Scholar] [CrossRef]
- Veldurthy, V.; Wei, R.; Oz, L.; Dhawan, P.; Jeon, Y.H.; Christakos, S. Vitamin D, calcium homeostasis and aging. Bone Res. 2016, 4, 16041. [Google Scholar] [CrossRef]
- Holick, M.F.; Matsuoka, L.Y.; Wortsman, J. Age, vitamin D, and solar ultraviolet. Lancet 1989, 2, 1104–1105. [Google Scholar] [CrossRef]
- Meehan, M.; Penckofer, S. The Role of Vitamin D in the Aging Adult. J. Aging Gerontol. 2014, 2, 60–71. [Google Scholar] [CrossRef]
- Orces, C.; Lorenzo, C.; Guarneros, J.E. The Prevalence and Determinants of Vitamin D Inadequacy among U.S. Older Adults: National Health and Nutrition Examination Survey 2007–2014. Cureus 2019, 11, e5300. [Google Scholar] [CrossRef]
- Boucher, B.J. The problems of vitamin d insufficiency in older people. Aging Dis. 2012, 3, 313–329. [Google Scholar]
- Chel, V.G.; Ooms, M.E.; Popp-Snijders, C.; Pavel, S.; Schothorst, A.A.; Meulemans, C.C.; Lips, P. Ultraviolet irradiation corrects vitamin D deficiency and suppresses secondary hyperparathyroidism in the elderly. J. Bone Miner. Res. 1998, 13, 1238–1242. [Google Scholar] [CrossRef]
- Chel, V.G.; Ooms, M.E.; Pavel, S.; de Gruijl, F.; Brand, A.; Lips, P. Prevention and treatment of vitamin D deficiency in Dutch psychogeriatric nursing home residents by weekly half-body UVB exposure after showering: A pilot study. Age Ageing 2011, 40, 211–214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Corless, D.; Gupta, S.P.; Switala, S.; Barragry, J.M.; Boucher, B.J.; Cohen, R.D.; Diffey, B.L. Response of plasma-25-hydroxyvitamin D to ultraviolet irradiation in long-stay geriatric patients. Lancet 1978, 2, 649–651. [Google Scholar] [CrossRef]
- Reid, I.R.; Gallagher, D.J.; Bosworth, J. Prophylaxis against vitamin D deficiency in the elderly by regular sunlight exposure. Age Ageing 1986, 15, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Baggerly, C.A.; Cuomo, R.E.; French, C.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Heaney, R.P.; Holick, M.F.; Hollis, B.W.; McDonnell, S.L.; et al. Sunlight and Vitamin D: Necessary for Public Health. J. Am. Coll. Nutr. 2015, 34, 359–365. [Google Scholar] [CrossRef]
- Vainio, H.; Miller, A.B.; Bianchini, F. An international evaluation of the cancer-preventive potential of sunscreens—Meeting held at Lyon, France, 10–18, 2000. Int. J. Cancer 2000, 88, 838–842. [Google Scholar] [CrossRef]
- American Cancer Society Skin Cancer Facts. Available online: http://www.cancer.org/cancer/cancercauses/sunanduvexposure/skin-cancer-facts (accessed on 5 February 2017).
- (US Department of Health and Human Services). The Surgeon General’s Call to Action to Prevent Skin Cancer. Available online: https://www.hhs.gov/sites/default/files/call-to-action-prevent-skin-cancer.pdf (accessed on 23 July 2020).
- Seleh, N. MDLinx A Daily Dose of This Can Help Fight Chronic Conditions. Available online: https://www.mdlinx.com/article/a-daily-dose-of-this-can-help-fight-chronic-conditions/P5i9BWT7H9W1VJGPntebl (accessed on 29 May 2020).
- Hossein-Nezhad, A.; Holick, M.F. Vitamin D for health: A global perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef]
- Baron, E.D.; Suggs, A.K. Introduction to photobiology. Dermatol. Clin. 2014, 32, 255–266. [Google Scholar] [CrossRef]
- Jager, N.; Schope, J.; Wagenpfeil, S.; Bocionek, P.; Saternus, R.; Vogt, T.; Reichrath, J. The Impact of UV-dose, Body Surface Area Exposed and Other Factors on Cutaneous Vitamin D Synthesis Measured as Serum 25(OH)D Concentration: Systematic Review and Meta-analysis. Anticancer Res. 2018, 38, 1165–1171. [Google Scholar] [CrossRef]
- Holick, M.F. Sunlight and Vitamin D for bone health and prevention of autoimmune diseases, cancers and cardioavascular disease. Am. J. Clin. Nutr. 2004, 80, 1678S–1688S. [Google Scholar] [CrossRef]
- Lagunova, Z.; Porojnicu, A.C.; Aksnes, L.; Holick, M.F.; Iani, V.; Bruland, O.S.; Moan, J. Effect of vitamin D supplementation and ultraviolet B exposure on serum 25-hydroxyvitamin D concentrations in healthy volunteers: A randomized, crossover clinical trial. Br. J. Dermatol. 2013, 169, 434–440. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Mosteller, R.D. Simplified calculation of body-surface area. N. Engl. J. Med. 1987, 317, 1098. [Google Scholar] [CrossRef] [PubMed]
- Larson-Meyer, D.E.; Douglas, C.S.; Thomas, J.J.; Johnson, E.C.; Barcal, J.N.; Heller, J.E.; Hollis, B.W.; Halliday, T.M. Validation of a Vitamin D Specific Questionnaire to Determine Vitamin D Status in Athletes. Nutrients 2019, 11, 2732. [Google Scholar] [CrossRef] [PubMed]
- Moan, J.; Dahlback, A.; Porojnicu, A.C. At what time should one go out in the sun? Adv. Exp. Med. Biol. 2008, 624, 86–88. [Google Scholar] [CrossRef]
- Grigalavicius, M.; Moan, J.; Dahlback, A.; Juzeniene, A. Daily, seasonal, and latitudinal variations in solar ultraviolet A and B radiation in relation to vitamin D production and risk for skin cancer. Int. J. Dermatol. 2016, 55, e23–e28. [Google Scholar] [CrossRef]
- Helmer, A.C.; Jansen, C.H. Vitamin D precursors removed from human skin by washing. Stud. Inst. Divi Thomae 1937, 1, 207–216. [Google Scholar]
- Kollias, N.; Ruvolo, E., Jr.; Sayre, R.M. The value of the ratio of UVA to UVB in sunlight. Photochem. Photobiol. 2011, 87, 1474–1475. [Google Scholar] [CrossRef]
- Adams, J.S.; Clemens, T.L.; Parrish, J.A.; Holick, M.F. Vitamin-D synthesis and metabolism after ultraviolet irradiation of normal and vitamin-D-deficient subjects. N. Engl J. Med. 1982, 306, 722–725. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and Health: Evolution, Biologic Functions, and Recommended Dietary Intakes for Vitamin D. Clin. Rev. Bone Miner. Metab. 2009, 7, 2–19. [Google Scholar] [CrossRef]
- Datta, P.; Bogh, M.K.; Olsen, P.; Eriksen, P.; Schmedes, A.V.; Grage, M.M.; Philipsen, P.A.; Wulf, H.C. Increase in serum 25-hydroxyvitamin-D3 in humans after solar exposure under natural conditions compared to artificial UVB exposure of hands and face. Photochem. Photobiol. Sci. 2012, 11, 1817–1824. [Google Scholar] [CrossRef]
- Terushkin, V.; Bender, A.; Psaty, E.L.; Engelsen, O.; Wang, S.Q.; Halpern, A.C. Estimated equivalency of vitamin D production from natural sun exposure versus oral vitamin D supplementation across seasons at two US latitudes. J. Am. Acad. Dermatol. 2010, 62, 929. e1–929. e9. [Google Scholar] [CrossRef] [PubMed]
- Holick, M. Sunlight “D”ilemma: Risk of skin cancer or bone disease and muscle weakness. Lancet 2001, 357, 3. [Google Scholar] [CrossRef]
- Makrantonaki, E.; Zouboulis, C.C. William J. Cunliffe Scientific Awards. Characteristics and pathomechanisms of endogenously aged skin. Dermatology 2007, 214, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Moragas, A.; Castells, C.; Sans, M. Mathematical morphologic analysis of aging-related epidermal changes. Anal. Quant. Cytol. Histol. 1993, 15, 75–82. [Google Scholar]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinants of vitamin D insufficiency: A genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Bogh, M.K.B.; Schmedes, A.V.; Philipsen, P.A.; Thieden, E.; Wulf, H.C. Vitamin D Production after UVB Exposure Depends on Baseline Vitamin D and Total cholesterol but Not on skin Pigmentation. J. Investig. Dermatol. 2009, 130, 546–553. [Google Scholar] [CrossRef]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J. Bone Miner. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef]
- Savastano, S.; Barrea, L.; Savanelli, M.C.; Nappi, F.; Di Somma, C.; Orio, F.; Colao, A. Low vitamin D status and obesity: Role of nutritionist. Rev. Endocr. Metab. Dis. 2017, 18, 215–225. [Google Scholar] [CrossRef]
- Abboud, M.; Rybchyn, M.S.; Rizk, R.; Fraser, D.R.; Mason, R.S. Sunlight exposure is just one of the factors which influence vitamin D status. Photochem. Photobiol. Sci. 2017, 16, 302–313. [Google Scholar] [CrossRef]
- Girgis, C.M.; Clifton-Bligh, R.J.; Turner, N.; Lau, S.L.; Gunton, J.E. Effects of Vitamin D in Skeletal Muscle: Falls, Strength, Athletic Performance and Insulin Sensitivity. Clin. Endocrinol. (Oxf.) 2013. [Google Scholar] [CrossRef] [PubMed]
| Total Group | Younger Adults | Older Adults | P * | |
|---|---|---|---|---|
| Sex (M/F) | 11/19 | 9/9 | 2/10 | — |
| Age (years) | 38.4 ± 10.4 (20–69) | 25.1 ± 4.8 (20–37) | 58.3 ± 5.1 (50–69) | 0.0001 * |
| Skin Type (II/III) | 3/27 | 3/15 | 0/12 | — |
| Hair (Bl/Br/R) | 7/21/2 | 5/12/1 | 2/9/1 | — |
| Mass (kg) | 71.2 ± 15.2 (50.1–106.1 | 71.8 ± 13.4 (54.2–102.1) | 70.4 ± 18.2 (50.1–106) | 0.816 |
| Height (cm) | 170.4 ± 7.9 (156.5–186.0) | 173.3 ± 7.4 (156.5–186) | 166.2 ± 6.8 (159.0–179) | 0.013 * |
| BMI (kg/m2) | 24.4 ± 4.5 (18.7–39.4) | 23.8 ± 3.2 (20.0–30.0) | 25.4 ± 6.1 (18.7–39.4) | 0.34 |
| Body Fat (%) | 30.3 ± 10.3 (11.9–54.0) | 28.8 ± 9.4 11.9–47.7) | 32.5 ± 11.5 (12.7–54.0) | 0.35 |
| Vitamin D Intake (IU) | 187 ± 182 (20–828) | 183.5 ± 167.4 (20–828) | 151.3 ± 96.2 (64–385) | 0.64 |
| Sun Exposure (h/month) | 14.5 ± 15.2 (0–60) | 13.4 ± 17.4 (0–60) | 16.2 ± 16.9 (2–60) | 0.63 |
| Baseline Serum D3 (nmol/L) | 15.0 ± 19.5 (3.7–110.1) | 18.0 ± 24.2 (3.7–110.1) | 10.7 ± 8.2 (3.7–30.0) | 0.33 |
| Baseline Serum D2 (nmol/L) | <3.7 | <3.7 | <3.7 | — |
| Baseline Serum 25(OH)D (nmol/L) | 84.9 ± 26.0 (42.4–166.5) | 86.6 ± 6.3 (56.7–166.5) | 82.6 ± 7.5 (42.4–123.5) | 0.69 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chalcraft, J.R.; Cardinal, L.M.; Wechsler, P.J.; Hollis, B.W.; Gerow, K.G.; Alexander, B.M.; Keith, J.F.; Larson-Meyer, D.E. Vitamin D Synthesis Following a Single Bout of Sun Exposure in Older and Younger Men and Women. Nutrients 2020, 12, 2237. https://doi.org/10.3390/nu12082237
Chalcraft JR, Cardinal LM, Wechsler PJ, Hollis BW, Gerow KG, Alexander BM, Keith JF, Larson-Meyer DE. Vitamin D Synthesis Following a Single Bout of Sun Exposure in Older and Younger Men and Women. Nutrients. 2020; 12(8):2237. https://doi.org/10.3390/nu12082237
Chicago/Turabian StyleChalcraft, Jenna R., Linda M. Cardinal, Perry J. Wechsler, Bruce W. Hollis, Kenneth G. Gerow, Brenda M. Alexander, Jill F. Keith, and D. Enette Larson-Meyer. 2020. "Vitamin D Synthesis Following a Single Bout of Sun Exposure in Older and Younger Men and Women" Nutrients 12, no. 8: 2237. https://doi.org/10.3390/nu12082237
APA StyleChalcraft, J. R., Cardinal, L. M., Wechsler, P. J., Hollis, B. W., Gerow, K. G., Alexander, B. M., Keith, J. F., & Larson-Meyer, D. E. (2020). Vitamin D Synthesis Following a Single Bout of Sun Exposure in Older and Younger Men and Women. Nutrients, 12(8), 2237. https://doi.org/10.3390/nu12082237







