Dietary Oat Bran Reduces Systemic Inflammation in Mice Subjected to Pelvic Irradiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
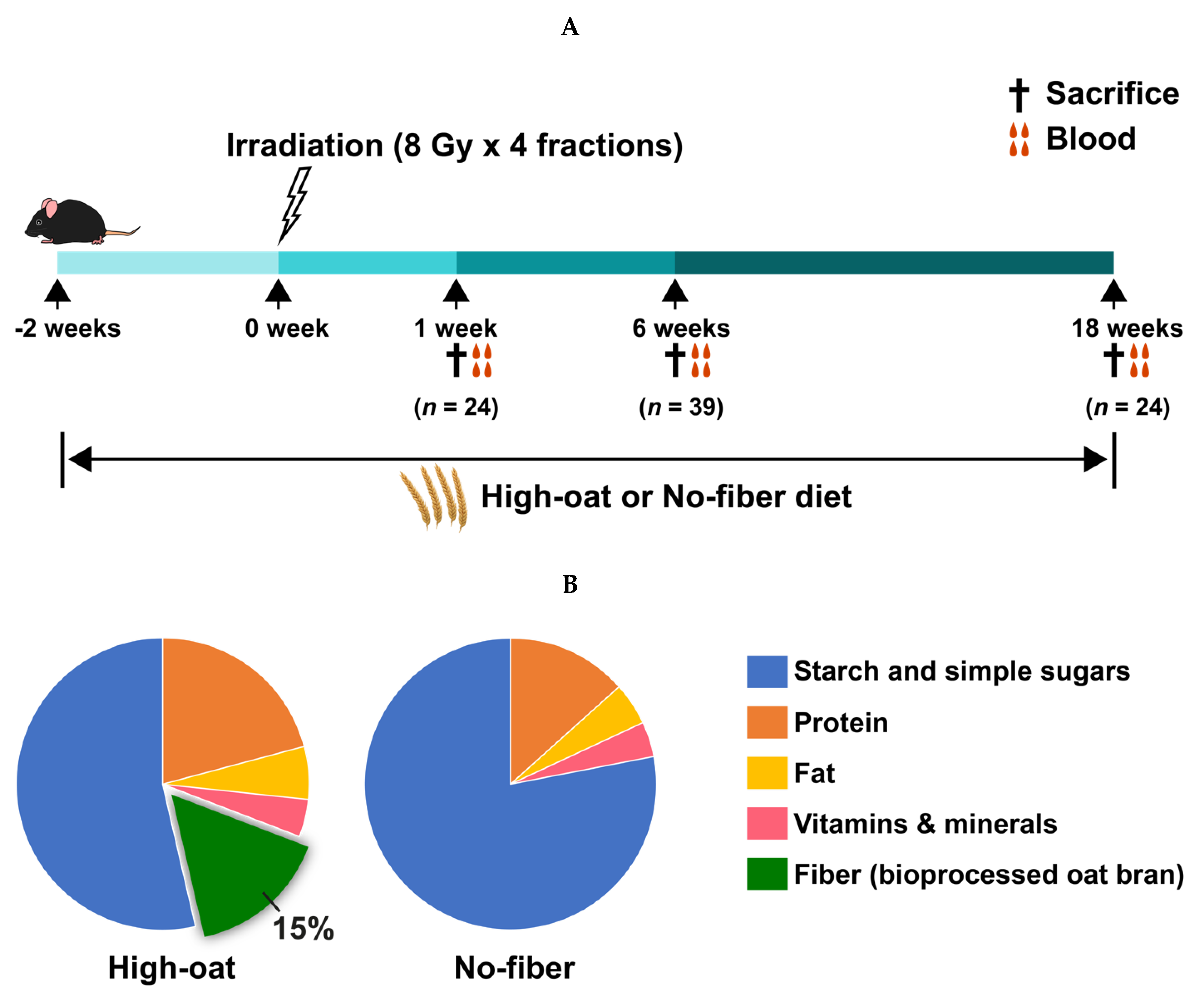
2.2. Experimental Design
2.3. Irradiation Procedure
2.4. Serum Preparation
2.5. Analyses of Serum Cytokines and Chemokines
2.6. Statistical Analysis
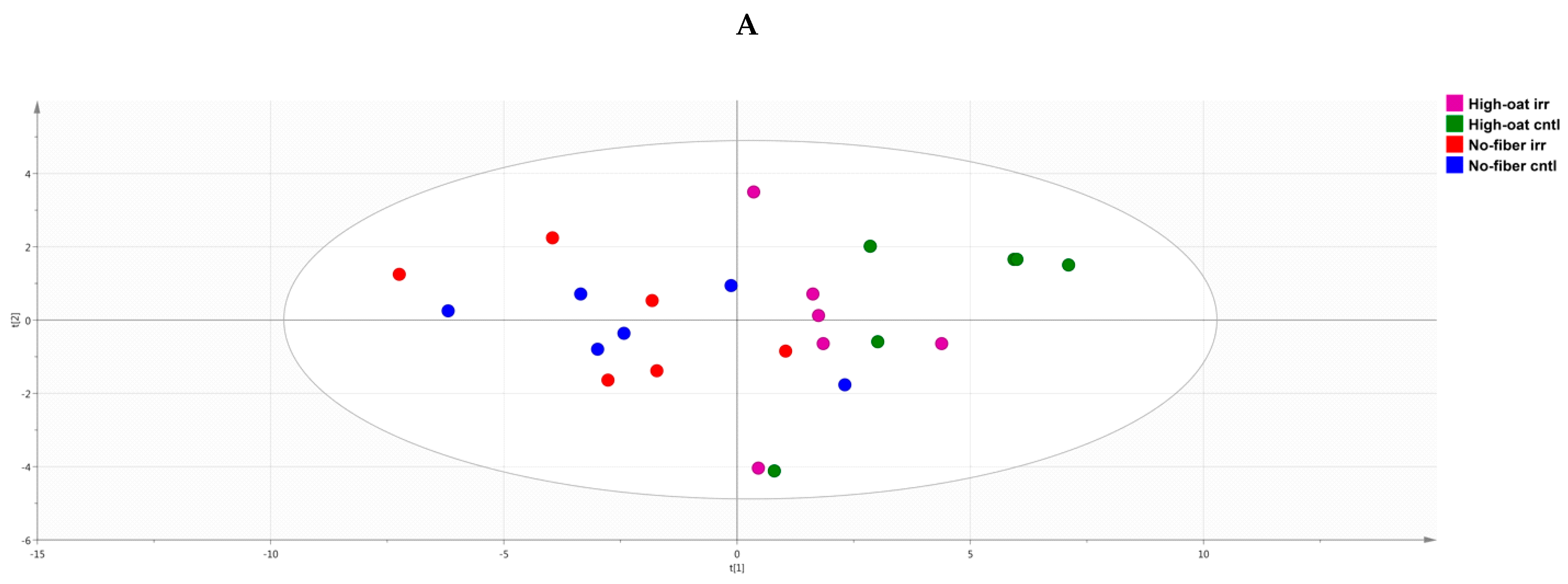
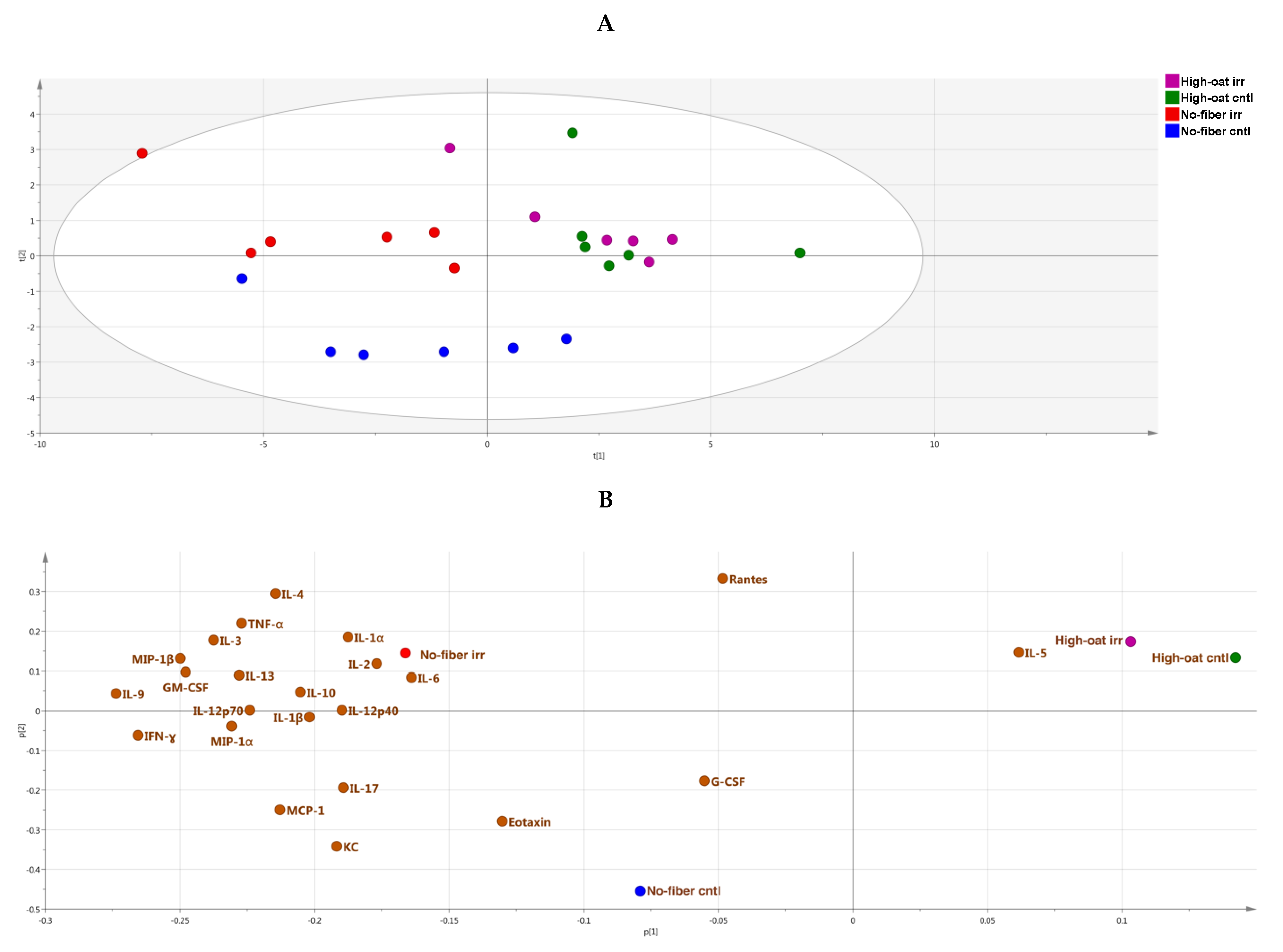
2.7. Principal Component Analysis
2.8. Ingenuity Pathway Analysis
3. Results
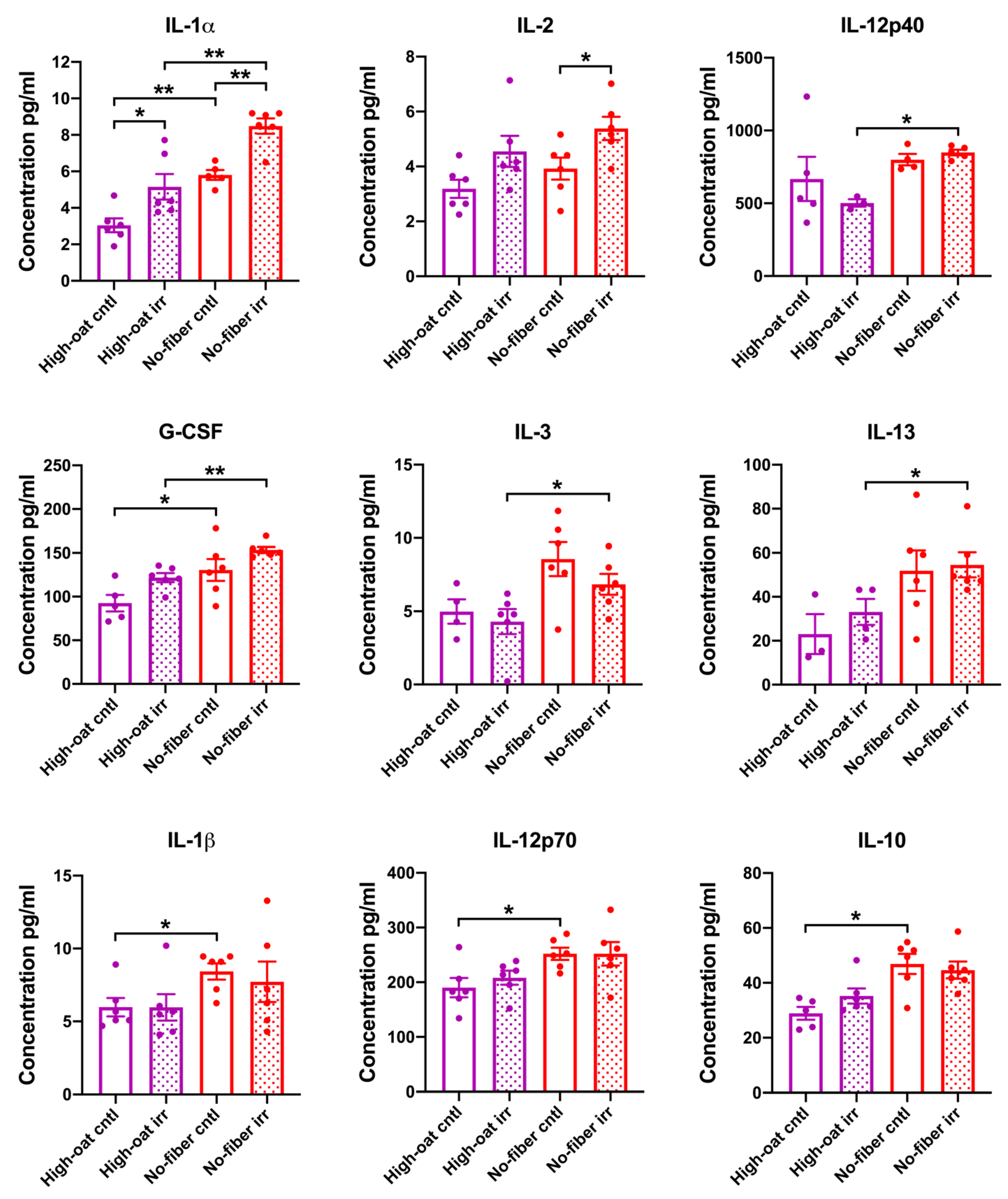
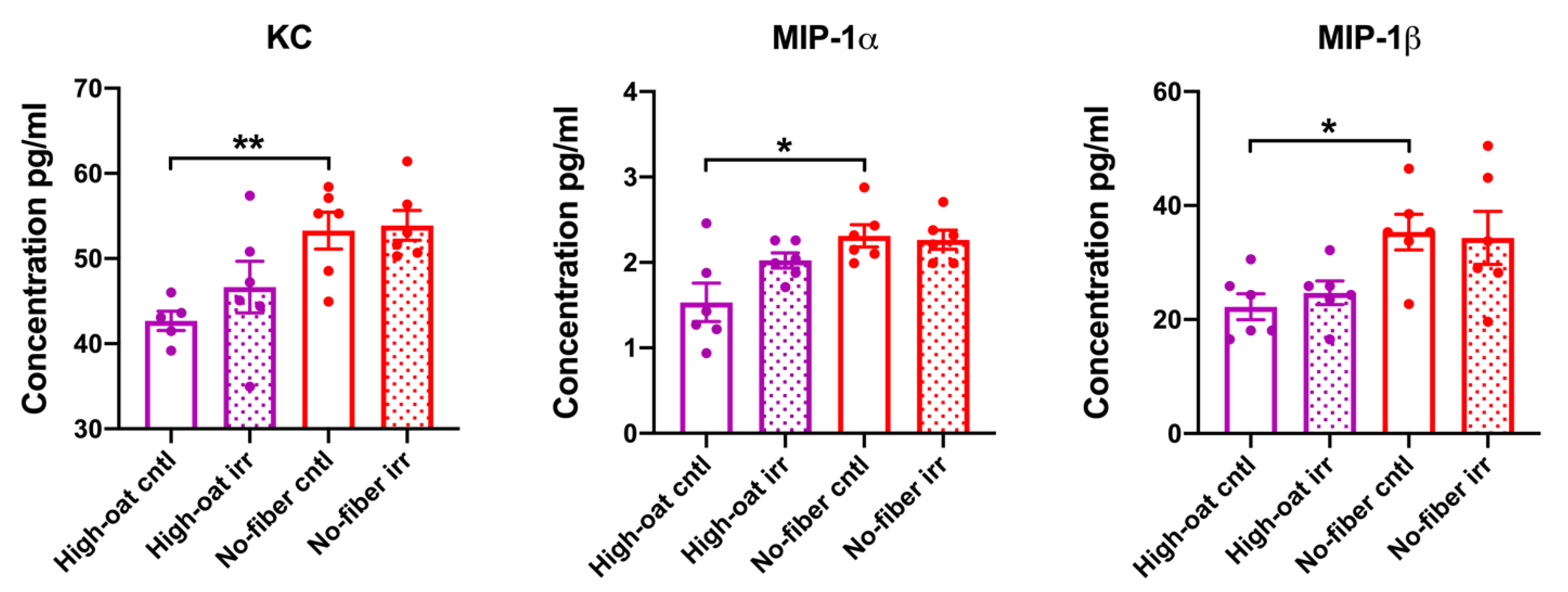
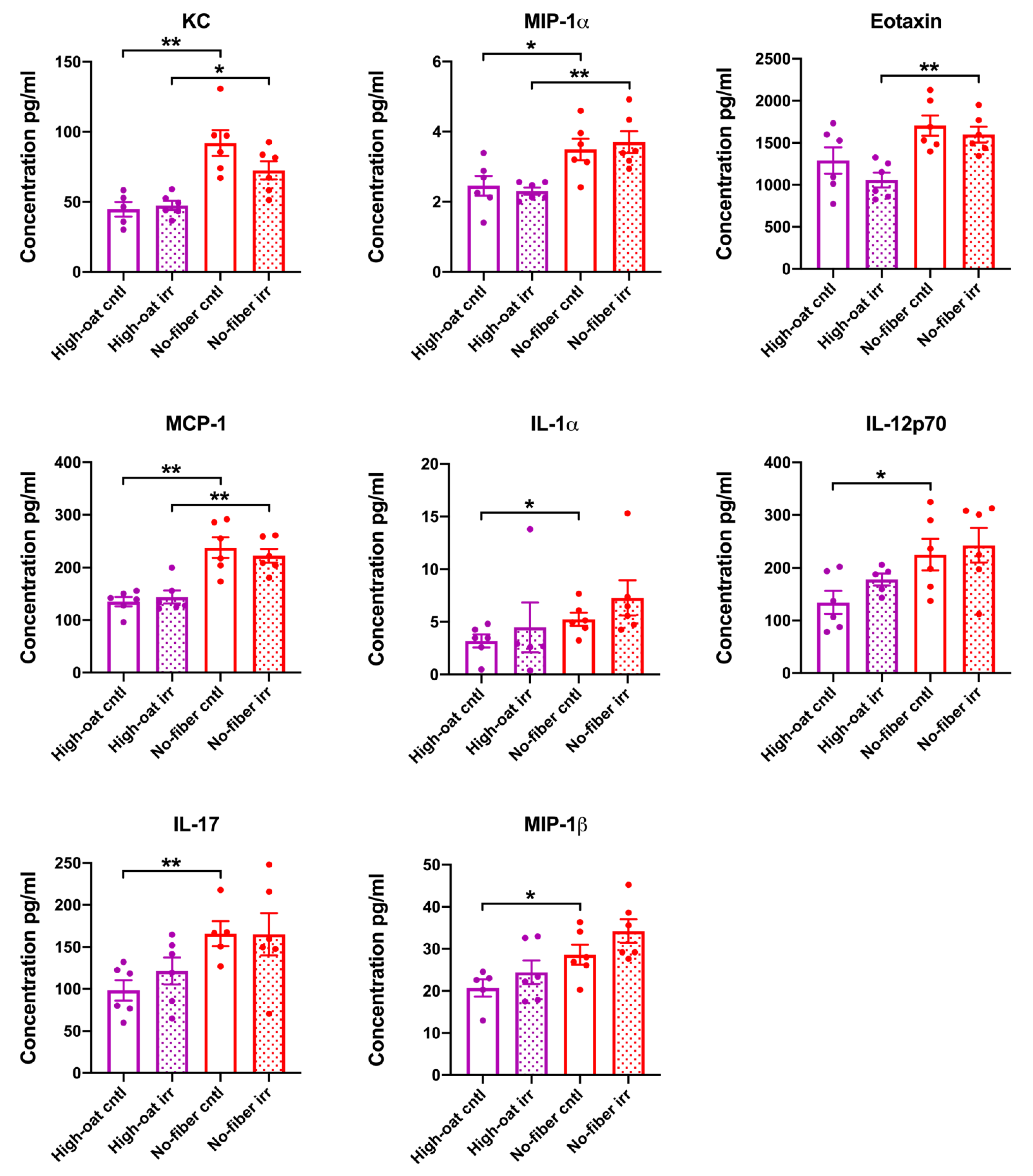
3.1. Serum Cytokine Levels at 1 Week Post-Irradiation
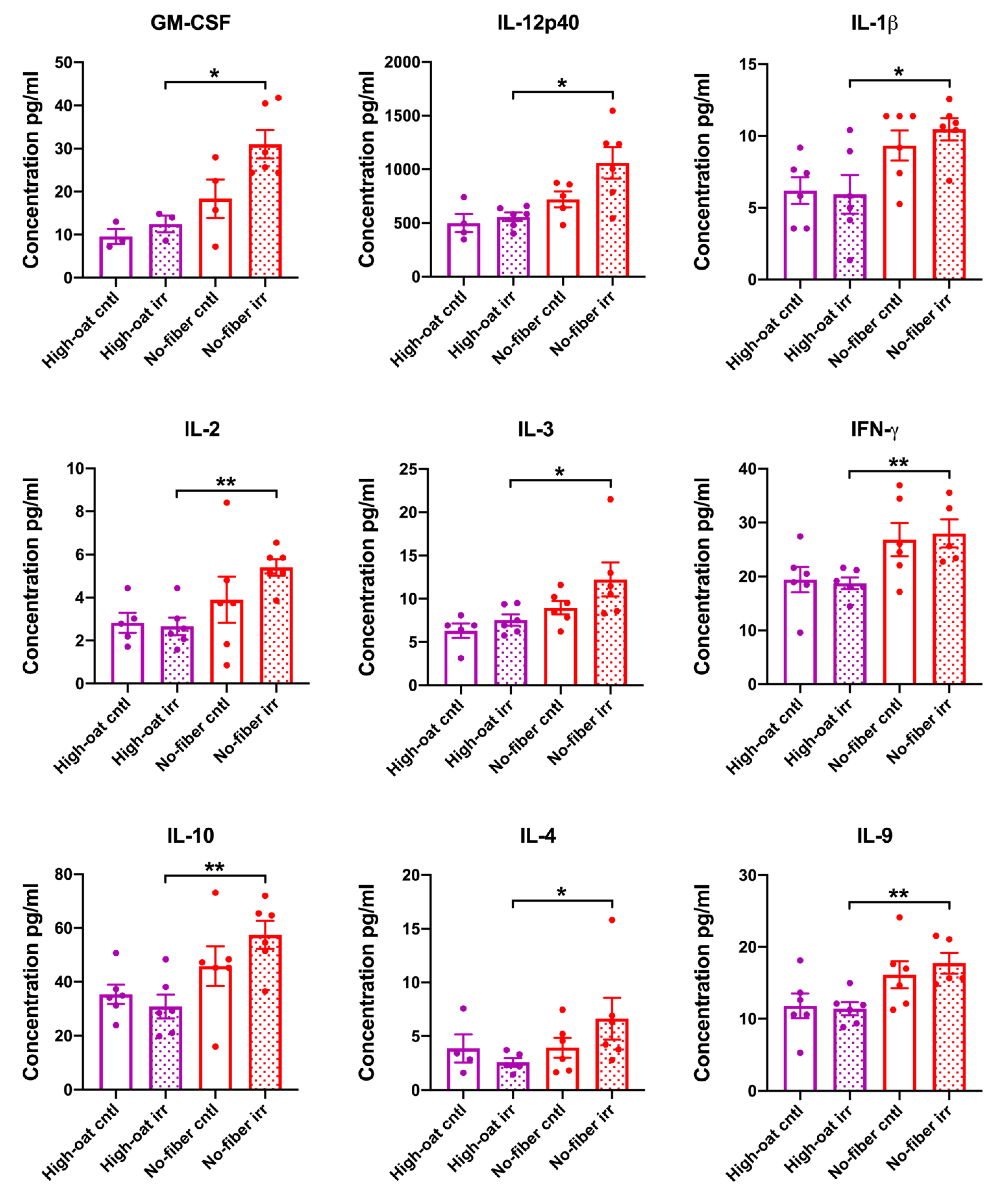
3.2. Serum Cytokine Levels at 6 Weeks Post-Irradiation
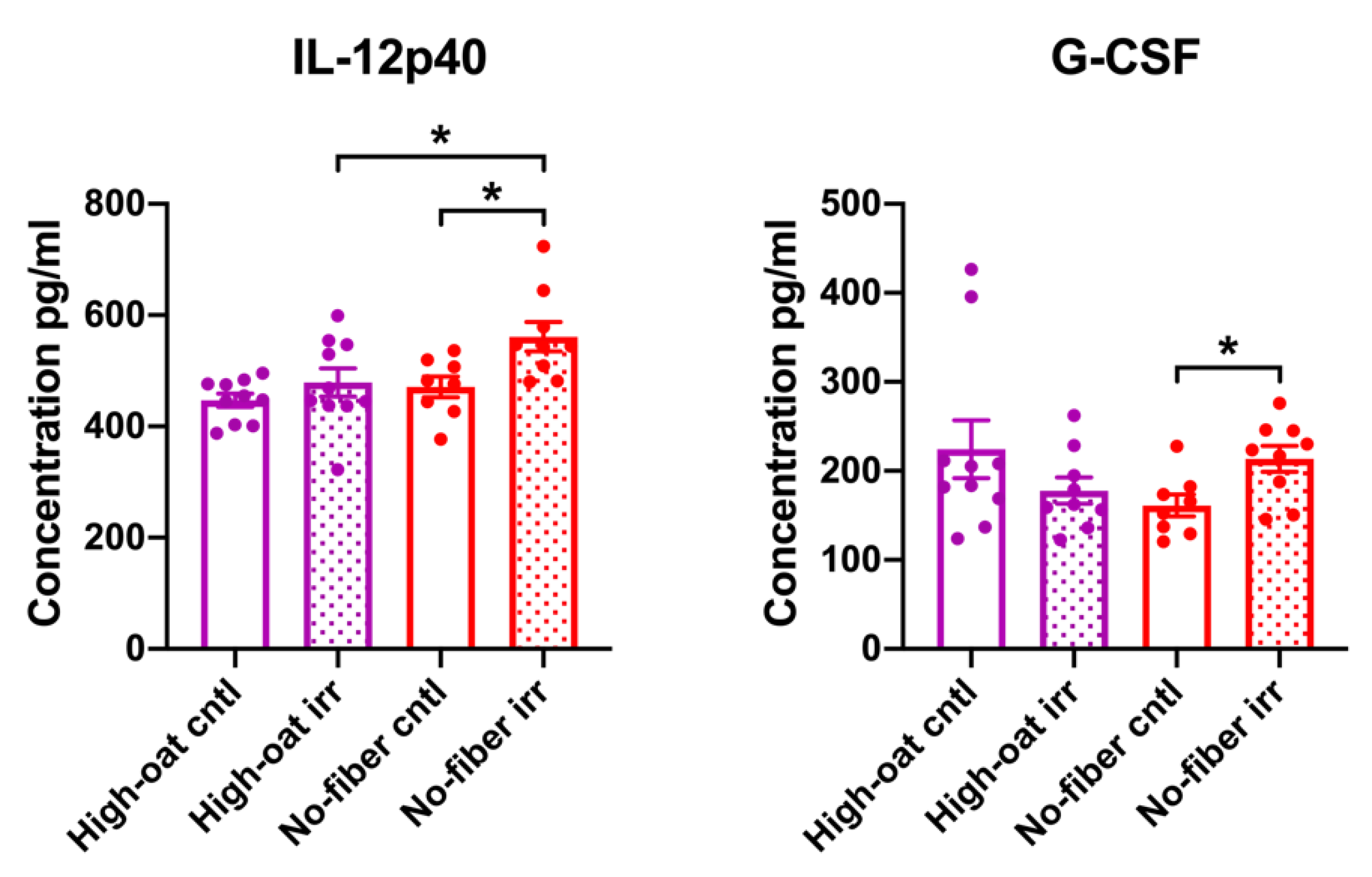
3.3. Serum Cytokine Levels at 18 Weeks Post-Irradiation
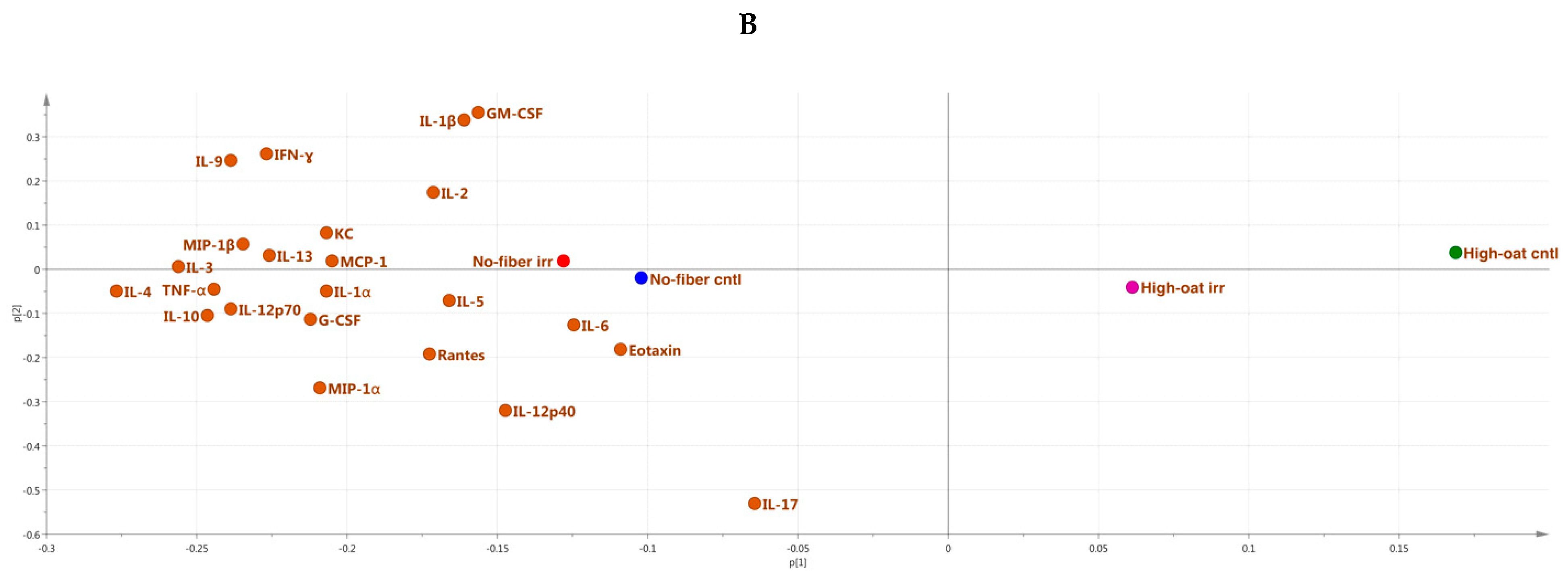
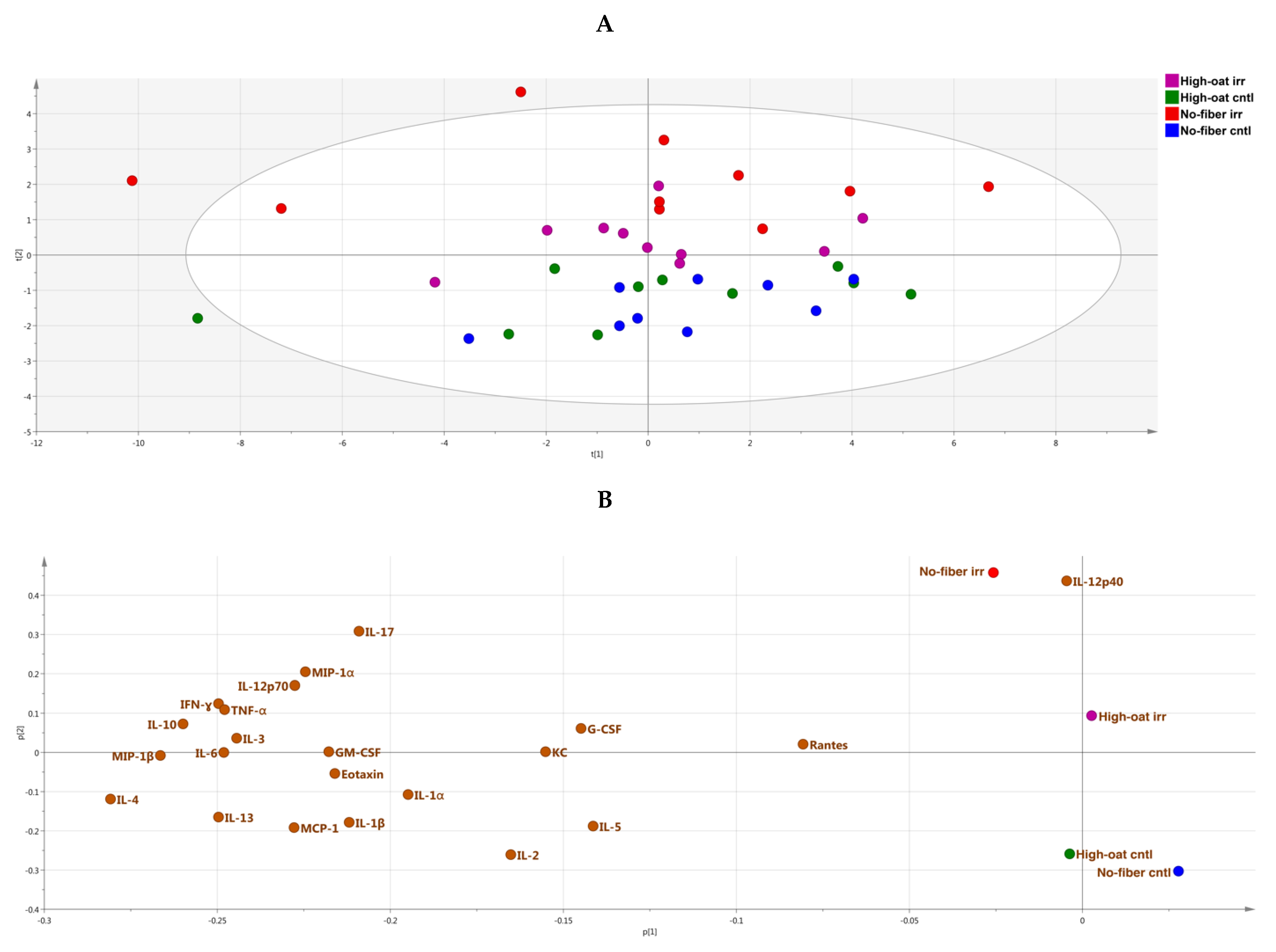
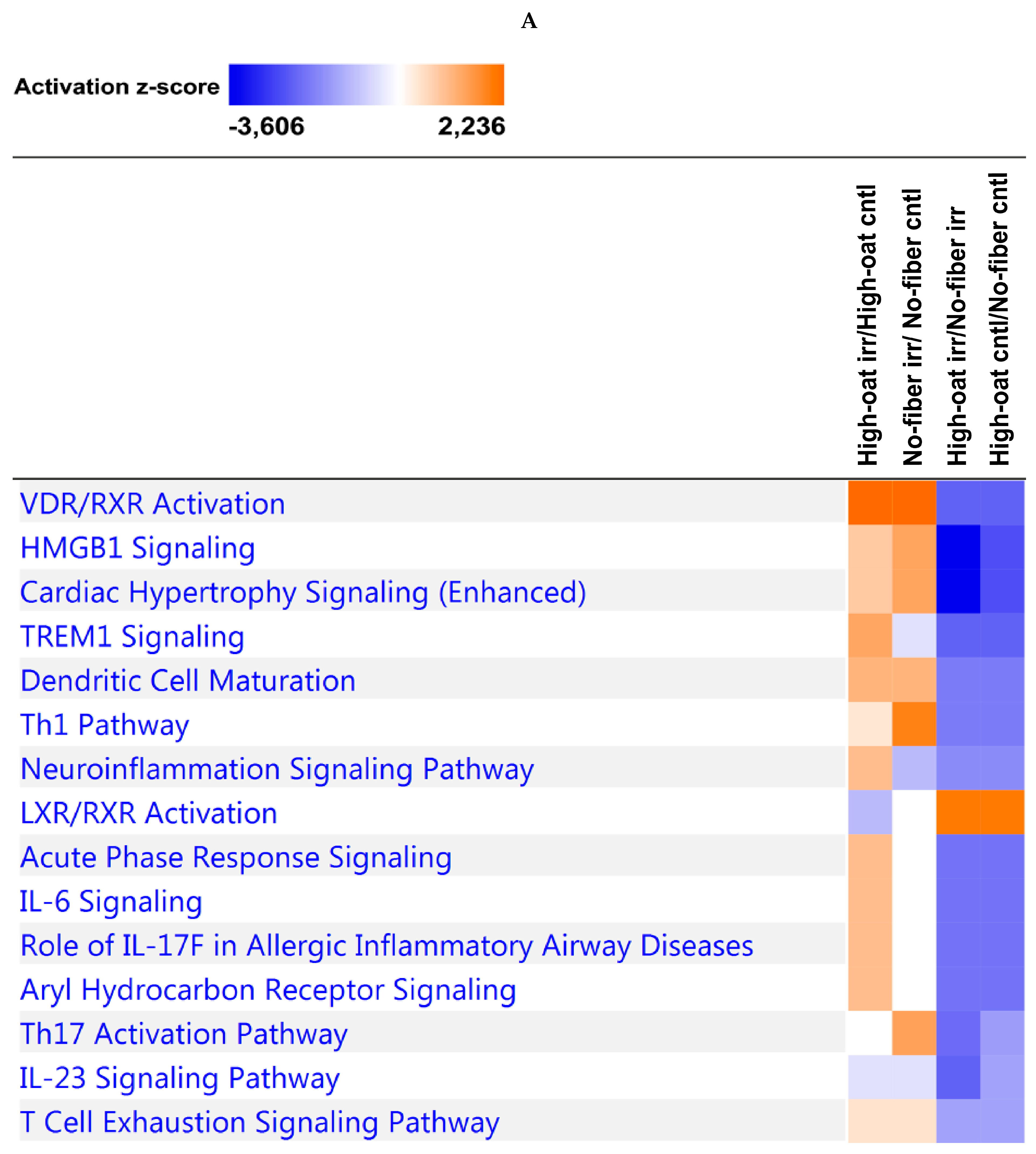
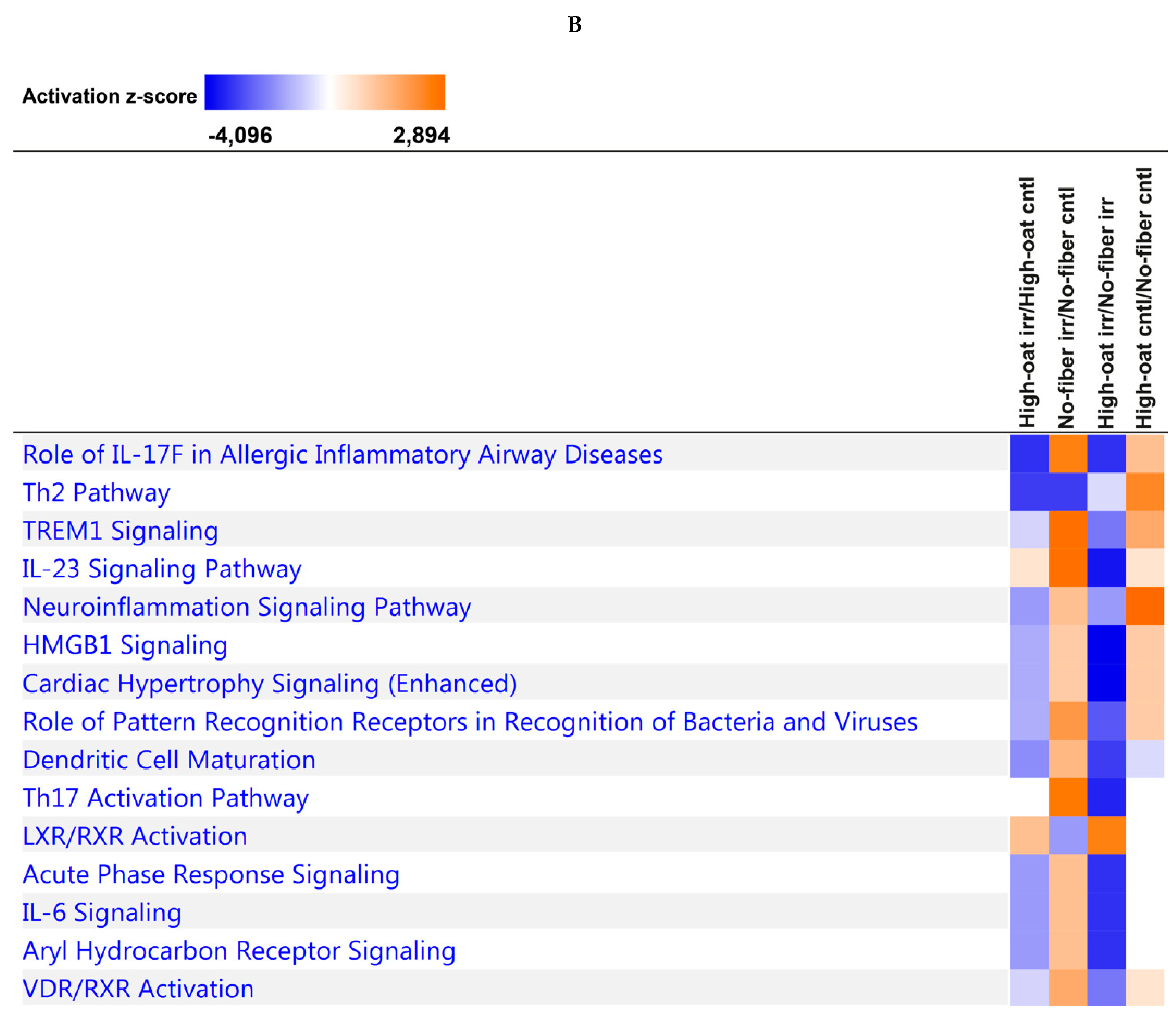
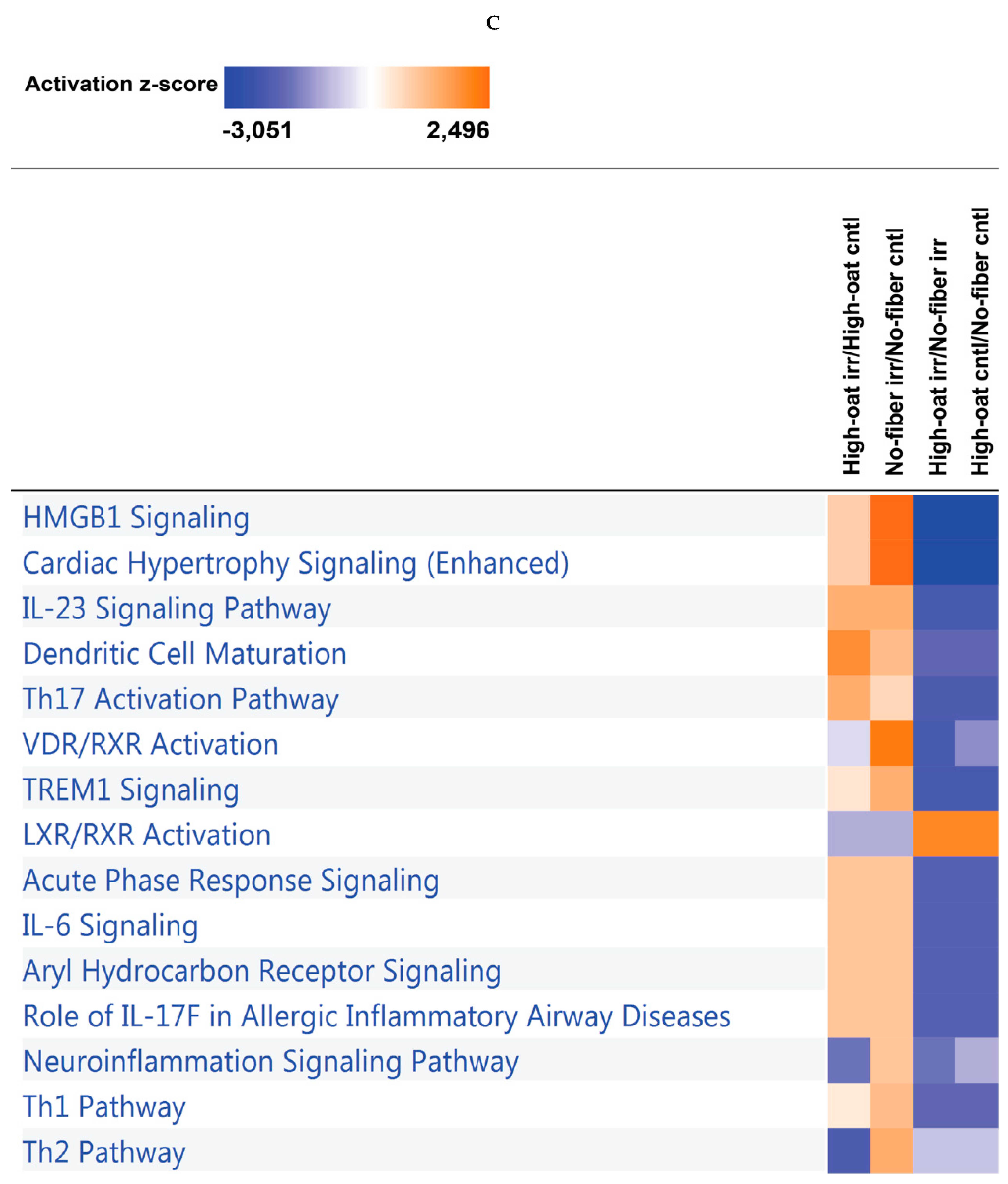
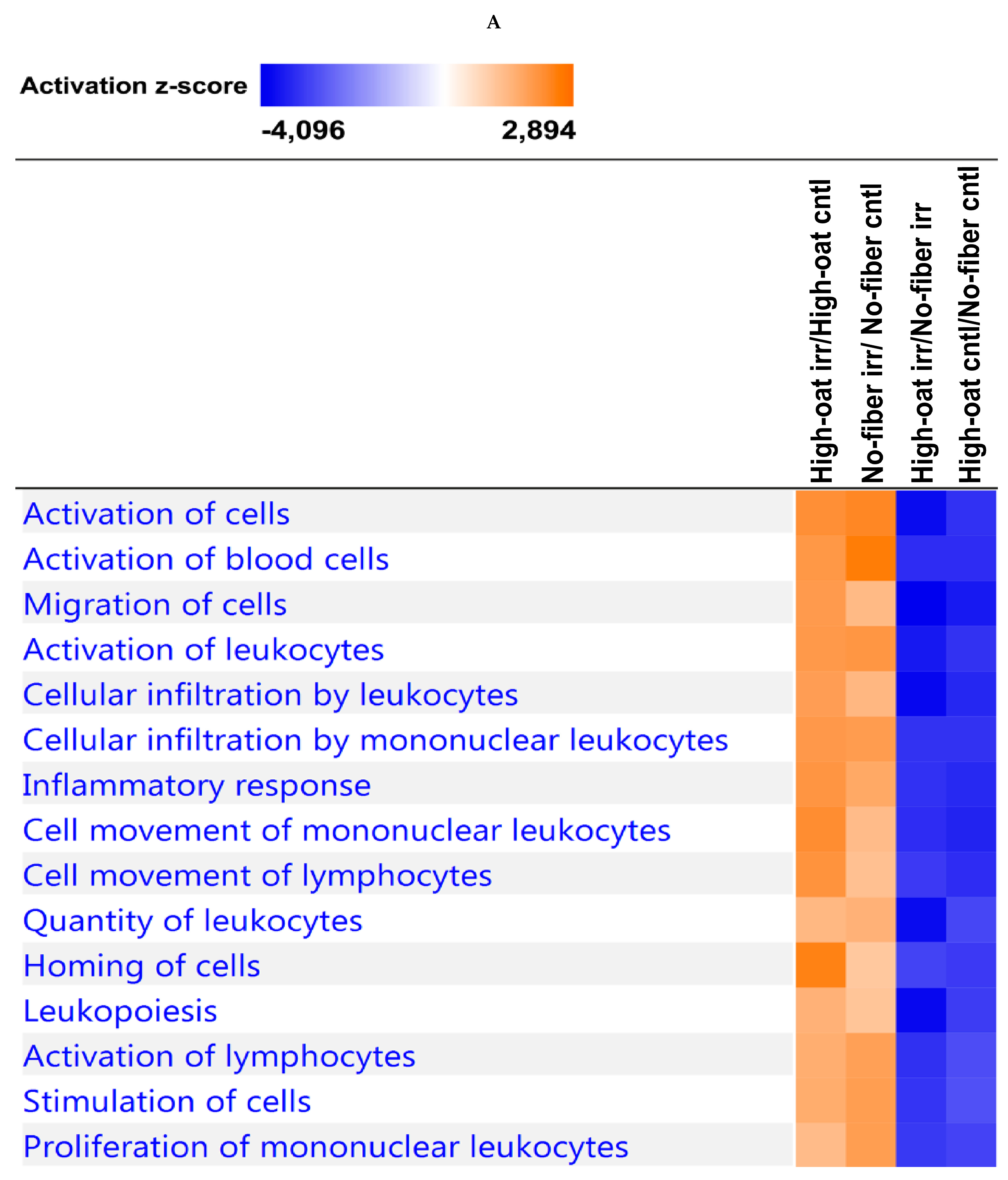
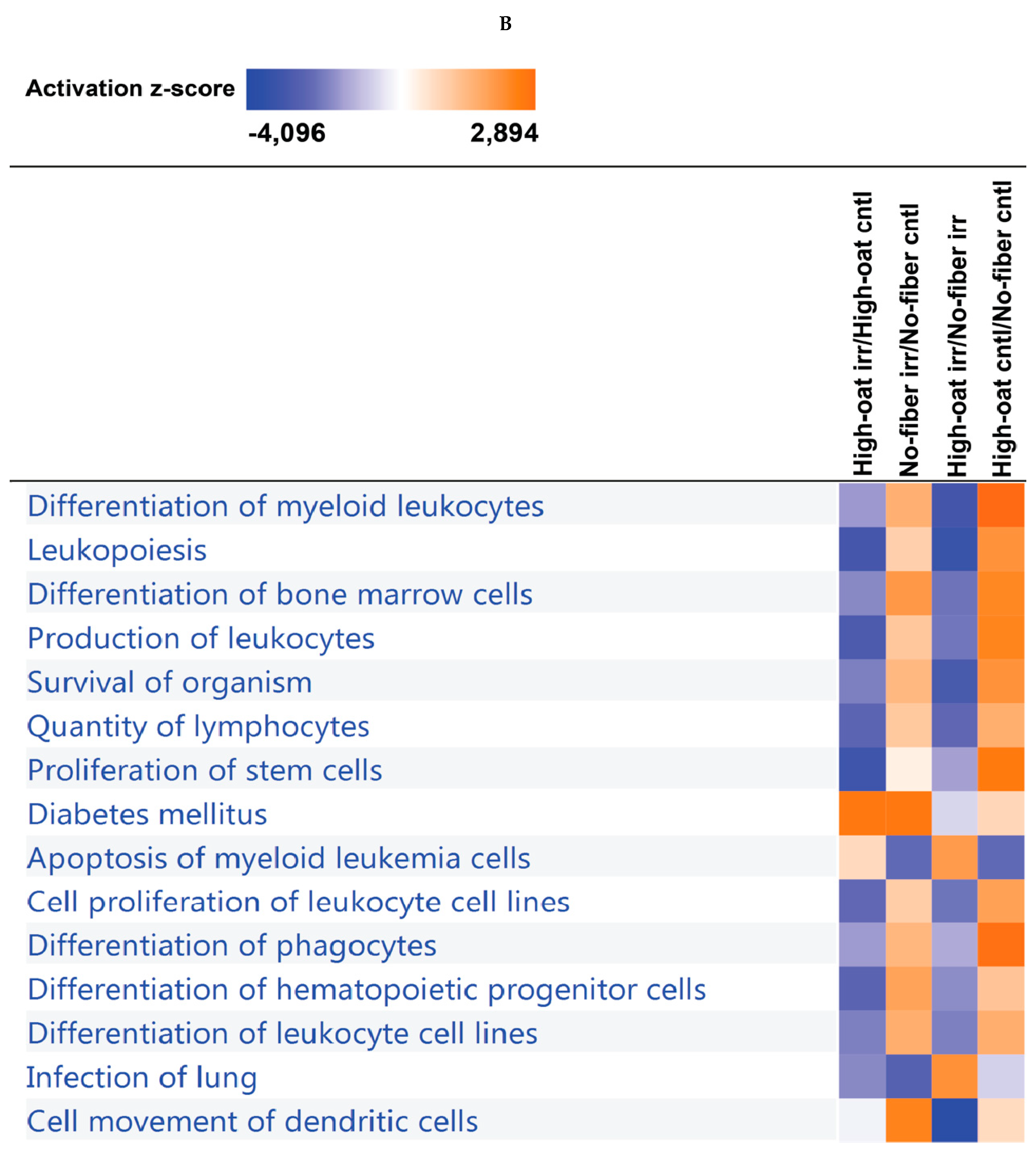
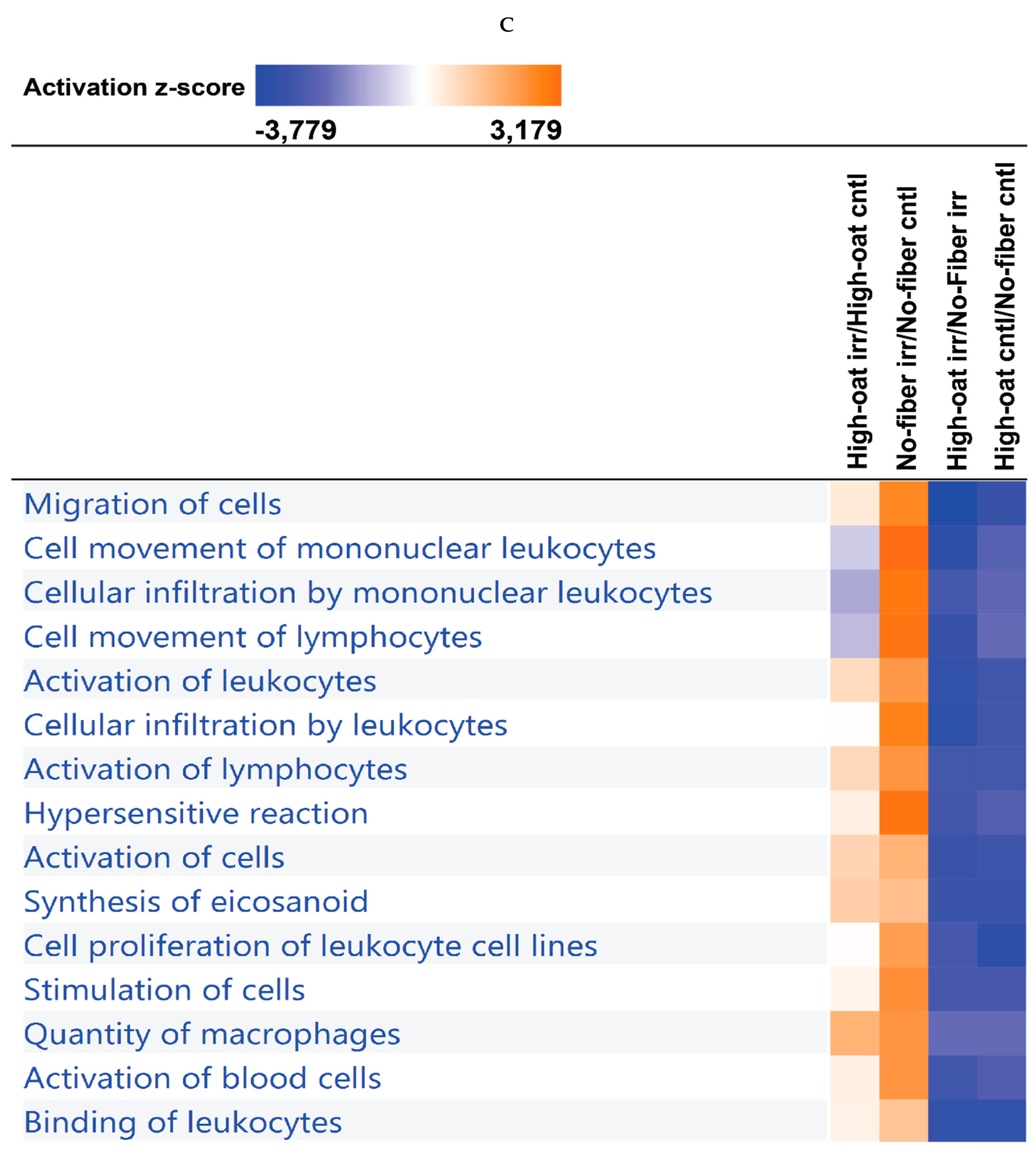
3.4. Ingenuity Pathway Analysis of the Cytokine Profiles
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ringborg, U.; Bergqvist, D.; Brorsson, B.; Cavallin-Stahl, E.; Ceberg, J.; Einhorn, N.; Frodin, J.E.; Jarhult, J.; Lamnevik, G.; Lindholm, C.; et al. The Swedish Council on Technology Assessment in Health Care (SBU) systematic overview of radiotherapy for cancer including a prospective survey of radiotherapy practice in Sweden 2001--summary and conclusions. Acta Oncol. 2003, 42, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Hauer-Jensen, M.; Denham, J.W.; Andreyev, H.J. Radiation enteropathy--pathogenesis, treatment and prevention. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Wedlake, L.J.; Thomas, K.; Lalji, A.; Blake, P.; Khoo, V.S.; Tait, D.; Andreyev, H.J. Predicting late effects of pelvic radiotherapy: Is there a better approach? Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1163–1170. [Google Scholar] [CrossRef]
- Bismar, M.M.; Sinicrope, F.A. Radiation enteritis. Curr. Gastroenterol. Rep. 2002, 4, 361–365. [Google Scholar] [CrossRef]
- Miller, R.C.; Martenson, J.A.; Sargent, D.J.; Kahn, M.J.; Krook, J.E. Acute treatment-related diarrhea during postoperative adjuvant therapy for high-risk rectal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 1998, 41, 593–598. [Google Scholar] [CrossRef]
- Visich, K.L.; Yeo, T.P. The prophylactic use of probiotics in the prevention of radiation therapy-induced diarrhea. Clin. J. Oncol. Nurs. 2010, 14, 467–473. [Google Scholar] [CrossRef]
- Steineck, G.; Skokic, V.; Sjoberg, F.; Bull, C.; Alevronta, E.; Dunberger, G.; Bergmark, K.; Wilderang, U.; Oh, J.H.; Deasy, J.O.; et al. Identifying radiation-induced survivorship syndromes affecting bowel health in a cohort of gynecological cancer survivors. PLoS ONE 2017, 12, e0171461. [Google Scholar] [CrossRef]
- Andreyev, H.J. Gastrointestinal problems after pelvic radiotherapy: The past, the present and the future. Clin. Oncol. 2007, 19, 790–799. [Google Scholar] [CrossRef]
- Richter, K.K.; Fagerhol, M.K.; Carr, J.C.; Winkler, J.M.; Sung, C.C.; Hauer-Jensen, M. Association of granulocyte transmigration with structural and cellular parameters of injury in experimental radiation enteropathy. Radiat. Oncol. Investig. 1997, 5, 275–282. [Google Scholar] [CrossRef]
- Wang, J.; Zheng, H.; Sung, C.C.; Hauer-Jensen, M. The synthetic somatostatin analogue, octreotide, ameliorates acute and delayed intestinal radiation injury. Int. J. Radiat. Oncol. Biol. Phys. 1999, 45, 1289–1296. [Google Scholar] [CrossRef]
- Linard, C.; Marquette, C.; Mathieu, J.; Pennequin, A.; Clarencon, D.; Mathe, D. Acute induction of inflammatory cytokine expression after gamma-irradiation in the rat: Effect of an NF-kappaB inhibitor. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, D.E.; Haimovitz-Friedman, A.; Kufe, D.W.; Fuks, Z.; Weichselbaum, R.R. The role of cytokines in radiation oncology. Important Adv. Oncol. 1993, 71–80. [Google Scholar]
- Beetz, A.; Messer, G.; Oppel, T.; van Beuningen, D.; Peter, R.U.; Kind, P. Induction of interleukin 6 by ionizing radiation in a human epithelial cell line: Control by corticosteroids. Int. J. Radiat. Biol. 1997, 72, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Woloschak, G.E.; Chang-Liu, C.M.; Jones, P.S.; Jones, C.A. Modulation of gene expression in Syrian hamster embryo cells following ionizing radiation. Cancer Res. 1990, 50, 339–344. [Google Scholar] [PubMed]
- Wang, J.; Robbins, M.E. Radiation-induced alteration of rat mesangial cell transforming growth factor-beta and expression of the genes associated with the extracellular matrix. Radiat. Res. 1996, 146, 561–568. [Google Scholar] [CrossRef]
- Rubin, P.; Johnston, C.J.; Williams, J.P.; McDonald, S.; Finkelstein, J.N. A perpetual cascade of cytokines postirradiation leads to pulmonary fibrosis. Int. J. Radiat. Oncol. Biol. Phys. 1995, 33, 99–109. [Google Scholar] [CrossRef]
- Sprung, C.N.; Forrester, H.B.; Siva, S.; Martin, O.A. Immunological markers that predict radiation toxicity. Cancer Lett. 2015, 368, 191–197. [Google Scholar] [CrossRef]
- Schaue, D.; Kachikwu, E.L.; McBride, W.H. Cytokines in radiobiological responses: A review. Radiat. Res. 2012, 178, 505–523. [Google Scholar] [CrossRef]
- Ding, N.H.; Li, J.J.; Sun, L.Q. Molecular mechanisms and treatment of radiation-induced lung fibrosis. Curr. Drug Targets 2013, 14, 1347–1356. [Google Scholar] [CrossRef]
- Provatopoulou, X.; Athanasiou, E.; Gounaris, A. Predictive markers of radiation pneumonitis. Anticancer Res. 2008, 28, 2421–2432. [Google Scholar]
- Kovacs, C.J.; Daly, B.M.; Evans, M.J.; Johnke, R.M.; Lee, T.K.; Karlsson, U.L.; Allison, R.; Eaves, G.S.; Biggs, L.M. Cytokine profiles in patients receiving wide-field + prostate boost radiotherapy (xRT) for adenocarcinoma of the prostate. Cytokine 2003, 23, 151–163. [Google Scholar] [CrossRef]
- McBride, W.H.; Chiang, C.S.; Olson, J.L.; Wang, C.C.; Hong, J.H.; Pajonk, F.; Dougherty, G.J.; Iwamoto, K.S.; Pervan, M.; Liao, Y.P. A sense of danger from radiation. Radiat. Res. 2004, 162, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Burkitt, D.P.; Walker, A.R.; Painter, N.S. Effect of dietary fibre on stools and the transit-times, and its role in the causation of disease. Lancet 1972, 2, 1408–1412. [Google Scholar] [CrossRef]
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- McCullogh, J.S.; Ratcliffe, B.; Mandir, N.; Carr, K.E.; Goodlad, R.A. Dietary fibre and intestinal microflora: Effects on intestinal morphometry and crypt branching. Gut 1998, 42, 799–806. [Google Scholar] [CrossRef]
- Sanchez, J.I.; Marzorati, M.; Grootaert, C.; Baran, M.; Van Craeyveld, V.; Courtin, C.M.; Broekaert, W.F.; Delcour, J.A.; Verstraete, W.; Van de Wiele, T. Arabinoxylan-oligosaccharides (AXOS) affect the protein/carbohydrate fermentation balance and microbial population dynamics of the Simulator of Human Intestinal Microbial Ecosystem. Microb. Biotechnol. 2009, 2, 101–113. [Google Scholar] [CrossRef]
- Mackie, A.R.; Macierzanka, A.; Aarak, K.; Rigby, N.M.; Parker, R.; Channell, G.A.; Harding, S.E.; Bajka, B.H. Sodium alginate decreases the permeability of intestinal mucus. Food Hydrocoll. 2016, 52, 749–755. [Google Scholar] [CrossRef]
- Pavadhgul, P.; Bumrungpert, A.; Harjani, Y.; Kurilich, A. Oat porridge consumption alleviates markers of inflammation and oxidative stress in hypercholesterolemic adults. Asia Pac. J. Clin. Nutr. 2019, 28, 260–265. [Google Scholar]
- McGough, C.; Baldwin, C.; Frost, G.; Andreyev, H.J. Role of nutritional intervention in patients treated with radiotherapy for pelvic malignancy. Br. J. Cancer 2004, 90, 2278–2287. [Google Scholar] [CrossRef]
- Hamad, A.; Fragkos, K.C.; Forbes, A. A systematic review and meta-analysis of probiotics for the management of radiation induced bowel disease. Clin. Nutr. 2013, 32, 353–360. [Google Scholar] [CrossRef]
- Volman, J.J.; Mensink, R.P.; Ramakers, J.D.; de Winther, M.P.; Carlsen, H.; Blomhoff, R.; Buurman, W.A.; Plat, J. Dietary (1-->3), (1-->4)-beta-D-glucans from oat activate nuclear factor-kappaB in intestinal leukocytes and enterocytes from mice. Nutr. Res. 2010, 30, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Rascon, A. Method for Preparing a Liquid Oat Base and Products Prepared by the Method. 2017. Available online: https://patentimages.storage.googleapis.com/85/36/3d/df43a7fe5c21aa/EP2996492B1.pdf (accessed on 10 October 2019).
- Motulsky, H.J.; Brown, R.E. Detecting outliers when fitting data with nonlinear regression-a new method based on robust nonlinear regression and the false discovery rate. BMC Bioinform. 2006, 7, 123. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.E. A User’s Guide to Principal Components; John Wiley & Sons: Hoboken, NJ, USA, 1991. [Google Scholar]
- Wold, S.; Esbensen, K.; Geladi, P. Principal component analysis. Chemom. Intell. Lab. Syst. 1987, 2, 37–52. [Google Scholar] [CrossRef]
- Kettaneh, N.; Berglund, A.; Wold, S. PCA and PLS with very large data sets. Comput. Stat. Data Anal. 2005, 48, 69–85. [Google Scholar] [CrossRef]
- Kramer, A.; Green, J.; Pollard, J., Jr.; Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 2014, 30, 523–530. [Google Scholar] [CrossRef]
- Dirksen, S.R.; Kirschner, K.F.; Belyea, M.J. Association of symptoms and cytokines in prostate cancer patients receiving radiation treatment. Biol. Res. Nurs. 2014, 16, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Okunieff, P.; Cornelison, T.; Mester, M.; Liu, W.; Ding, I.; Chen, Y.; Zhang, H.; Williams, J.P.; Finkelstein, J. Mechanism and modification of gastrointestinal soft tissue response to radiation: Role of growth factors. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 273–278. [Google Scholar] [CrossRef]
- Zhang, M.; Yin, L.; Zhang, K.; Sun, W.; Yang, S.; Zhang, B.; Salzman, P.; Wang, W.; Liu, C.; Vidyasagar, S.; et al. Response patterns of cytokines/chemokines in two murine strains after irradiation. Cytokine 2012, 58, 169–177. [Google Scholar] [CrossRef]
- Bull, C.; Malipatlolla, D.; Kalm, M.; Sjoberg, F.; Alevronta, E.; Grander, R.; Sultanian, P.; Persson, L.; Bostrom, M.; Eriksson, Y.; et al. A novel mouse model of radiation-induced cancer survivorship diseases of the gut. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G456–G466. [Google Scholar] [CrossRef]
- Malipatlolla, D.K.; Patel, P.; Sjoberg, F.; Devarakonda, S.; Kalm, M.; Angenete, E.; Lindskog, E.B.; Grander, R.; Persson, L.; Stringer, A.; et al. Long-term mucosal injury and repair in a murine model of pelvic radiotherapy. Sci. Rep. 2019, 9, 13803. [Google Scholar] [CrossRef]
- Andreyev, J. Gastrointestinal symptoms after pelvic radiotherapy: A new understanding to improve management of symptomatic patients. Lancet Oncol. 2007, 8, 1007–1017. [Google Scholar] [CrossRef]
- Morris, K.A.; Haboubi, N.Y. Pelvic radiation therapy: Between delight and disaster. World J. Gastrointest. Surg. 2015, 7, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, E.J. Fibrogenic cytokines: The role of immune mediators in the development of scar tissue. Immunol. Today 1991, 12, 17–23. [Google Scholar] [CrossRef]
- Indaram, A.V.; Visvalingam, V.; Locke, M.; Bank, S. Mucosal cytokine production in radiation-induced proctosigmoiditis compared with inflammatory bowel disease. Am. J. Gastroenterol. 2000, 95, 1221–1225. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Kruger, C.; Steigleder, T.; Weber, D.; Pitzer, C.; Laage, R.; Aronowski, J.; Maurer, M.H.; Gassler, N.; Mier, W.; et al. The hematopoietic factor G-CSF is a neuronal ligand that counteracts programmed cell death and drives neurogenesis. J. Clin. Investig. 2005, 115, 2083–2098. [Google Scholar] [CrossRef] [PubMed]
- Nicola, N.A.; Metcalf, D.; Matsumoto, M.; Johnson, G.R. Purification of a factor inducing differentiation in murine myelomonocytic leukemia cells. Identification as granulocyte colony-stimulating factor. J. Biol. Chem. 1983, 258, 9017–9023. [Google Scholar]
- Carulli, G. Effects of recombinant human granulocyte colony-stimulating factor administration on neutrophil phenotype and functions. Haematologica 1997, 82, 606–616. [Google Scholar]
- Yasuda, H.; Ajiki, Y.; Shimozato, T.; Kasahara, M.; Kawada, H.; Iwata, M.; Shimizu, K. Therapeutic efficacy of granulocyte colony-stimulating factor alone and in combination with antibiotics against Pseudomonas aeruginosa infections in mice. Infect. Immun. 1990, 58, 2502–2509. [Google Scholar] [CrossRef]
- Tanji, N.; Kikugawa, T.; Ochi, T.; Taguchi, S.; Sato, H.; Sato, T.; Sugahara, T.; Hamada, H.; Asai, S.; Matsumoto, A. Circulating Cytokine Levels in Patients with Prostate Cancer: Effects of Neoadjuvant Hormonal Therapy and External-beam Radiotherapy. Anticancer Res. 2015, 35, 3379–3383. [Google Scholar]
- Suchecka, D.; Gromadzka-Ostrowska, J.; Żyła, E.; Harasym, J.P.; Oczkowski, M. Selected physiological activities and health promoting properties of cereal β-glucans. A review. J. Anim. Feed Sci. 2017, 26, 183–191. [Google Scholar] [CrossRef]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta glucan: Health benefits in obesity and metabolic syndrome. J. Nutr. Metab. 2012, 2012, 851362. [Google Scholar] [CrossRef]
- Yun, C.H.; Estrada, A.; Van Kessel, A.; Park, B.C.; Laarveld, B. Beta-glucan, extracted from oat, enhances disease resistance against bacterial and parasitic infections. FEMS Immunol. Med. Microbiol. 2003, 35, 67–75. [Google Scholar] [CrossRef]
- Davis, J.M.; Murphy, E.A.; Brown, A.S.; Carmichael, M.D.; Ghaffar, A.; Mayer, E.P. Effects of oat beta-glucan on innate immunity and infection after exercise stress. Med. Sci. Sports Exerc. 2004, 36, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Inan, M.S.; Rasoulpour, R.J.; Yin, L.; Hubbard, A.K.; Rosenberg, D.W.; Giardina, C. The luminal short-chain fatty acid butyrate modulates NF-kappaB activity in a human colonic epithelial cell line. Gastroenterology 2000, 118, 724–734. [Google Scholar] [CrossRef]
- Liu, B.; Lin, Q.; Yang, T.; Zeng, L.; Shi, L.; Chen, Y.; Luo, F. Oat beta-glucan ameliorates dextran sulfate sodium (DSS)-induced ulcerative colitis in mice. Food Funct. 2015, 6, 3454–3463. [Google Scholar] [CrossRef] [PubMed]
- Trompette, A.; Gollwitzer, E.S.; Pattaroni, C.; Lopez-Mejia, I.C.; Riva, E.; Pernot, J.; Ubags, N.; Fajas, L.; Nicod, L.P.; Marsland, B.J. Dietary Fiber Confers Protection against Flu by Shaping Ly6c(-) Patrolling Monocyte Hematopoiesis and CD8(+) T Cell Metabolism. Immunity 2018, 48, 992–1005. [Google Scholar] [CrossRef]
- Bermudez-Brito, M.; Sahasrabudhe, N.M.; Rosch, C.; Schols, H.A.; Faas, M.M.; de Vos, P. The impact of dietary fibers on dendritic cell responses in vitro is dependent on the differential effects of the fibers on intestinal epithelial cells. Mol. Nutr. Food Res. 2015, 59, 698–710. [Google Scholar] [CrossRef]
- Donatto, F.F.; Prestes, J.; Frollini, A.B.; Palanch, A.C.; Verlengia, R.; Cavaglieri, C.R. Effect of oat bran on time to exhaustion, glycogen content and serum cytokine profile following exhaustive exercise. J. Int. Soc. Sports Nutr. 2010, 7, 32. [Google Scholar] [CrossRef]
- Wilczak, J.; Blaszczyk, K.; Kamola, D.; Gajewska, M.; Harasym, J.P.; Jalosinska, M.; Gudej, S.; Suchecka, D.; Oczkowski, M.; Gromadzka-Ostrowska, J. The effect of low or high molecular weight oat beta-glucans on the inflammatory and oxidative stress status in the colon of rats with LPS-induced enteritis. Food Funct. 2015, 6, 590–603. [Google Scholar] [CrossRef]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef]
- Eastaff-Leung, N.; Mabarrack, N.; Barbour, A.; Cummins, A.; Barry, S. Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. J. Clin. Immunol. 2010, 30, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Hu, E.D.; Chen, D.Z.; Wu, J.L.; Lu, F.B.; Chen, L.; Zheng, M.H.; Li, H.; Huang, Y.; Li, J.; Jin, X.Y.; et al. High fiber dietary and sodium butyrate attenuate experimental autoimmune hepatitis through regulation of immune regulatory cells and intestinal barrier. Cell Immunol. 2018, 328, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.R.; Wolff, B.S.; Lukkahatai, N.; Espina, A.; Saligan, L.N. Exploratory Investigation of Early Biomarkers for Chronic Fatigue in Prostate Cancer Patients Following Radiation Therapy. Cancer Nurs. 2017, 40, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, R.; Kawamura, Y.I.; Kato, R.; Mizutani, N.; Toyama-Sorimachi, N.; Dohi, T. IL-13 receptor alpha2 promotes epithelial cell regeneration from radiation-induced small intestinal injury in mice. Gastroenterology 2006, 131, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.I.; Horton, J.A.; Ramalingam, T.R.; White, A.O.; Chung, E.J.; Hudak, K.E.; Scroggins, B.T.; Arron, J.R.; Wynn, T.A.; Citrin, D.E. IL-13 is a therapeutic target in radiation lung injury. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Gerassy-Vainberg, S.; Blatt, A.; Danin-Poleg, Y.; Gershovich, K.; Sabo, E.; Nevelsky, A.; Daniel, S.; Dahan, A.; Ziv, O.; Dheer, R.; et al. Radiation induces proinflammatory dysbiosis: Transmission of inflammatory susceptibility by host cytokine induction. Gut 2018, 67, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Symon, Z.; Goldshmidt, Y.; Picard, O.; Yavzori, M.; Ben-Horin, S.; Alezra, D.; Barshack, I.; Chowers, Y. A murine model for the study of molecular pathogenesis of radiation proctitis. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 242–250. [Google Scholar] [CrossRef]
- Kanarek, N.; Grivennikov, S.I.; Leshets, M.; Lasry, A.; Alkalay, I.; Horwitz, E.; Shaul, Y.D.; Stachler, M.; Voronov, E.; Apte, R.N.; et al. Critical role for IL-1beta in DNA damage-induced mucositis. Proc. Natl. Acad. Sci. USA 2014, 111, E702–E711. [Google Scholar] [CrossRef]
- Li, W.; Wang, G.; Cui, J.; Xue, L.; Cai, L. Low-dose radiation (LDR) induces hematopoietic hormesis: LDR-induced mobilization of hematopoietic progenitor cells into peripheral blood circulation. Exp. Hematol. 2004, 32, 1088–1096. [Google Scholar] [CrossRef]
- Christensen, E.; Pintilie, M.; Evans, K.R.; Lenarduzzi, M.; Menard, C.; Catton, C.N.; Diamandis, E.P.; Bristow, R.G. Longitudinal cytokine expression during IMRT for prostate cancer and acute treatment toxicity. Clin. Cancer Res. 2009, 15, 5576–5583. [Google Scholar] [CrossRef]
- Chuang, S.C.; Vermeulen, R.; Sharabiani, M.T.; Sacerdote, C.; Fatemeh, S.H.; Berrino, F.; Krogh, V.; Palli, D.; Panico, S.; Tumino, R.; et al. The intake of grain fibers modulates cytokine levels in blood. Biomarkers 2011, 16, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Takemura, N.; Kurashima, Y.; Mori, Y.; Okada, K.; Ogino, T.; Osawa, H.; Matsuno, H.; Aayam, L.; Kaneto, S.; Park, E.J.; et al. Eosinophil depletion suppresses radiation-induced small intestinal fibrosis. Sci. Transl. Med. 2018, 10, eaan0333. [Google Scholar] [CrossRef]
- Abdel-Messeih, P.L.; Nosseir, N.M.; Bakhe, O.H. Evaluation of inflammatory cytokines and oxidative stress markers in prostate cancer patients undergoing curative radiotherapy. Cent. Eur. J. Immunol. 2017, 42, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Buttner, C.; Skupin, A.; Reimann, T.; Rieber, E.P.; Unteregger, G.; Geyer, P.; Frank, K.H. Local production of interleukin-4 during radiation-induced pneumonitis and pulmonary fibrosis in rats: Macrophages as a prominent source of interleukin-4. Am. J. Respir. Cell Mol. Biol. 1997, 17, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, Y.; Hirashima, N.; Nakanishi, M.; Furuno, T. Inhibition of degranulation and cytokine production in bone marrow-derived mast cells by hydrolyzed rice bran. Inflamm. Res. 2010, 59, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Gerlach, K.; Hwang, Y.; Nikolaev, A.; Atreya, R.; Dornhoff, H.; Steiner, S.; Lehr, H.A.; Wirtz, S.; Vieth, M.; Waisman, A.; et al. TH9 cells that express the transcription factor PU.1 drive T cell-mediated colitis via IL-9 receptor signaling in intestinal epithelial cells. Nat. Immunol. 2014, 15, 676–686. [Google Scholar] [CrossRef]
- Willett, C.G.; Ooi, C.J.; Zietman, A.L.; Menon, V.; Goldberg, S.; Sands, B.E.; Podolsky, D.K. Acute and late toxicity of patients with inflammatory bowel disease undergoing irradiation for abdominal and pelvic neoplasms. Int. J. Radiat. Oncol. Biol. Phys. 2000, 46, 995–998. [Google Scholar] [CrossRef]
- Lotze, M.T.; Tracey, K.J. High-mobility group box 1 protein (HMGB1): Nuclear weapon in the immune arsenal. Nat. Rev. Immunol. 2005, 5, 331–342. [Google Scholar] [CrossRef]
- Zhou, J.; Chai, F.; Lu, G.; Hang, G.; Chen, C.; Chen, X.; Shi, J. TREM-1 inhibition attenuates inflammation and tumor within the colon. Int. Immunopharmacol. 2013, 17, 155–161. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, P.; Malipatlolla, D.K.; Devarakonda, S.; Bull, C.; Rascón, A.; Nyman, M.; Stringer, A.; Tremaroli, V.; Steineck, G.; Sjöberg, F. Dietary Oat Bran Reduces Systemic Inflammation in Mice Subjected to Pelvic Irradiation. Nutrients 2020, 12, 2172. https://doi.org/10.3390/nu12082172
Patel P, Malipatlolla DK, Devarakonda S, Bull C, Rascón A, Nyman M, Stringer A, Tremaroli V, Steineck G, Sjöberg F. Dietary Oat Bran Reduces Systemic Inflammation in Mice Subjected to Pelvic Irradiation. Nutrients. 2020; 12(8):2172. https://doi.org/10.3390/nu12082172
Chicago/Turabian StylePatel, Piyush, Dilip Kumar Malipatlolla, Sravani Devarakonda, Cecilia Bull, Ana Rascón, Margareta Nyman, Andrea Stringer, Valentina Tremaroli, Gunnar Steineck, and Fei Sjöberg. 2020. "Dietary Oat Bran Reduces Systemic Inflammation in Mice Subjected to Pelvic Irradiation" Nutrients 12, no. 8: 2172. https://doi.org/10.3390/nu12082172
APA StylePatel, P., Malipatlolla, D. K., Devarakonda, S., Bull, C., Rascón, A., Nyman, M., Stringer, A., Tremaroli, V., Steineck, G., & Sjöberg, F. (2020). Dietary Oat Bran Reduces Systemic Inflammation in Mice Subjected to Pelvic Irradiation. Nutrients, 12(8), 2172. https://doi.org/10.3390/nu12082172




