The Effects of Legume Consumption on Markers of Glycaemic Control in Individuals with and without Diabetes Mellitus: A Systematic Literature Review of Randomised Controlled Trials
Abstract
1. Introduction
2. Methods
2.1. Eligibility and Exclusion Criteria
2.2. Search Strategy
2.3. Study Selection, Data Extraction, and Quality Assessment
2.4. Data Analysis
3. Results
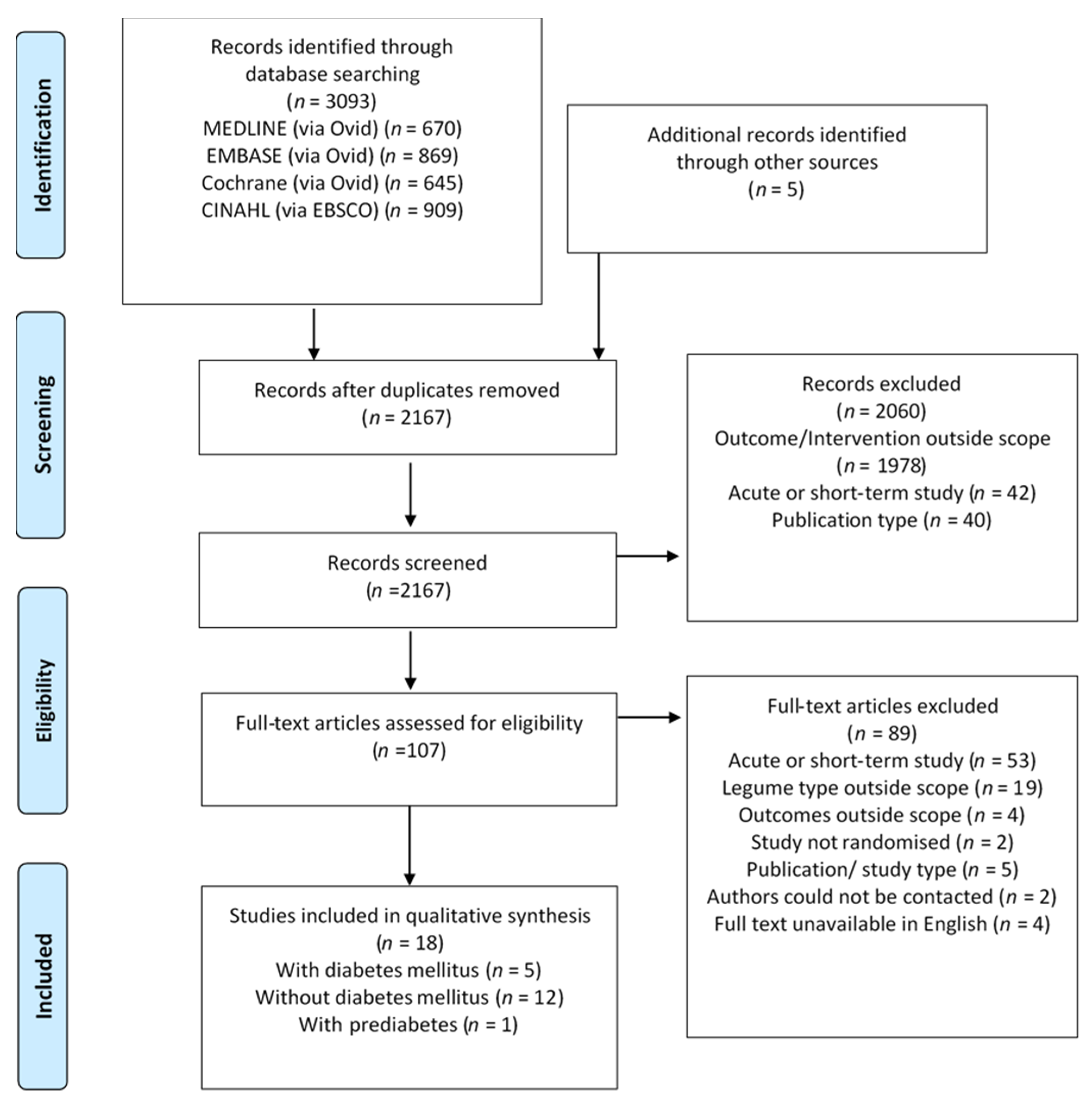
3.1. Search Results and Study Selection
3.2. Study Characteristics
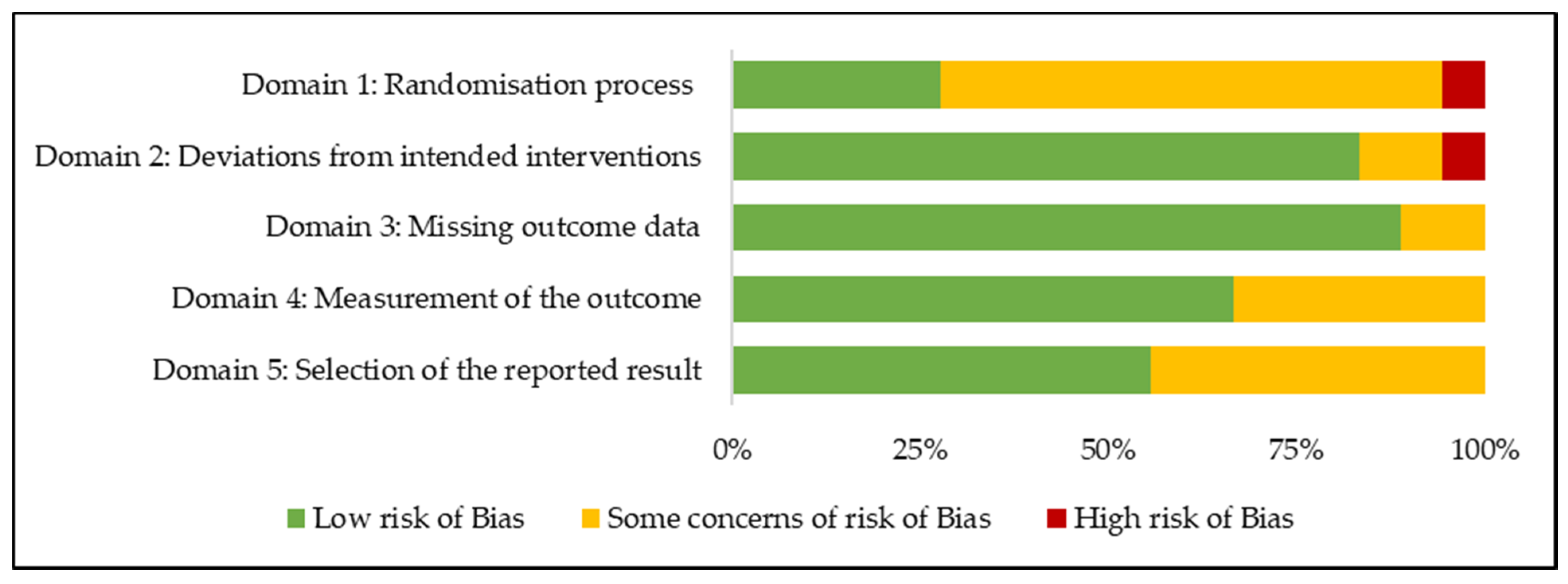
3.3. Risk of Bias
3.4. GRADE Assessment
3.5. Effect of Intervention on the Outcome
3.5.1. Individuals with Diabetes Mellitus
3.5.2. Individuals without Diabetes Mellitus
3.5.3. Individuals with Prediabetes
4. Discussion
4.1. Strengths and Limitations
4.2. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization. Definition and Classification of Commodities: Pulses and Derived Products. Available online: http://www.fao.org/es/faodef/fdef04e.htm (accessed on 2 March 2020).
- Marinangeli, C.P.F.; Curran, J.; Barr, S.I.; Slavin, J.; Puri, S.; Swaminathan, S.; Tapsell, L.; Patterson, C.A. Enhancing nutrition with pulses: Defining a recommended serving size for adults. Nutr. Rev. 2017, 75, 990–1006. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Carbohydrates in Human Nutrition. Report of a Joint FAO/WHO Expert Consultation; Food and Agriculture Organization: Rome, Italy, 1998. [Google Scholar]
- Schwingshackl, L.; Hoffmann, G.; Iqbal, K.; Schwedhelm, C.; Boeing, H. Food groups and intermediate disease markers: A systematic review and network meta-analysis of randomized trials. Am. J. Clin. Nutr. 2018, 108, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Bechthold, A.; Boeing, H.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; De Henauw, S.; Michels, N.; Devleesschauwer, B.; Schlesinger, S.; et al. Food groups and risk of coronary heart disease, stroke and heart failure: A systematic review and dose-response meta-analysis of prospective studies. Crit. Rev. Food Sci. Nutr. 2017, 59, 1071–1090. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Lampousi, A.-M.; Knüppel, S.; Iqbal, K.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of all-cause mortality: A systematic review and meta-analysis of prospective studies. Am. J. Clin. Nutr. 2017, 105, 1462–1473. [Google Scholar] [CrossRef]
- National Health and Medical Research Council. Australian Dietary Guidelines. Available online: https://www.nhmrc.gov.au/_files_nhmrc/file/publications/n55_australian_dietary_guidelines1.pdf (accessed on 17 June 2020).
- Health Canada. Canada’s Dietary Guidelines for Health Professionals and Policy Makers. Available online: https://food-guide.canada.ca/en/guidelines/ (accessed on 17 June 2020).
- US Department of Health and Human Services and US Department of Agriculture. 2015–2020 Dietary Guidelines for Americans. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 17 June 2020).
- National Health and Medical Research Council. A Modelling System to Inform the Revision of the Australian Guide to Healthy Eating; National Health and Medical Research Council: Canberra, Australia, 2011.
- Guariguata, L.; Whiting, D.; Hambleton, I.R.; Beagley, J.; Linnenkamp, U.; Shaw, J. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pr. 2014, 103, 137–149. [Google Scholar] [CrossRef]
- Tanamas, S.; Magliano, D.; Lynch, B.; Sethi, P.; Willenberg, L.; Polkinghorne, K.; Chadban, S.; Dunstan, D.; Shaw, J. AusDiab 2012 The Australian Diabetes, Obesity and Lifestyle Study; Baker Heart and Diabetes Institute: Melbourne, Australia, 2013. [Google Scholar]
- Australian Bureau of Statistics. 4364.0.55.001—National Health Survey First Results, 2017–2018. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/4364.0.55.0012017-18?OpenDocument#Publications (accessed on 9 June 2020).
- The Royal Australian College of General Practitioners. General Practice Management of Type 2 Diabetes: 2016–18; RACGP: Melbourne, Australia, 2016. [Google Scholar]
- Sievenpiper, J.L.; Kendall, C.W.; Esfahani, A.; Wong, J.M.; Carleton, A.J.; Jiang, H.Y.; Bazinet, R.P.; Vidgen, E.; Jenkins, D.J. Effect of non-oil-seed pulses on glycaemic control: A systematic review and meta-analysis of randomised controlled experimental trials in people with and without diabetes. Diabetologia 2009, 52, 1479–1495. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; The PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 349, g7647. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.N.; Akerman, A.P.; Mann, J. Dietary fibre and whole grains in diabetes management: Systematic review and meta-analyses. PLoS Med. 2020, 17, e1003053. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; Vist, G.E.; Glasziou, P.; Akl, E.A.; Skoetz, N.; Guyatt, G.H. Chapter 14: Completing ‘Summary of Findings’ Tables and Grading the Certainty of the Evidence. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (Updated July 2019). Available online: www.training.cochrane.org/handbook (accessed on 17 June 2020).
- GRADE Working Group. Grading quality of evidence and strength of recommendations. BMJ 2004, 328, 1490. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (Updated July 2019). Available online: www.training.cochrane.org/handbook (accessed on 1 April 2020).
- Knopp, J.L.; Holder-Pearson, L.; Chase, J.G. Insulin Units and Conversion Factors: A Story of Truth, Boots, and Faster Half-Truths. J. Diabetes Sci. Technol. 2018, 13, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh-Rostami, Z.; Hemmatdar, Z.; Pishdad, G.R.; Faghih, S. Moderate Consumption of Red Meat, Compared to Soy or Non-Soy Legume, Has No Adverse Effect on Cardio-Metabolic Factors in Patients with Type 2 Diabetes. Exp. Clin. Endocrinol. Diabetes 2019, 17. [Google Scholar] [CrossRef]
- Hosseinpour-Niazi, S.; Mirmiran, P.; Hedayati, M.; Azizi, F. Substitution of red meat with legumes in the therapeutic lifestyle change diet based on dietary advice improves cardiometabolic risk factors in overweight type 2 diabetes patients: A cross-over randomized clinical trial. Eur. J. Clin. Nutr. 2014, 69, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Augustin, L.S.A.; Mitchell, S.; Sahye-Pudaruth, S.; Mejia, S.B.; Chiavaroli, L.; Mirrahimi, A.; Ireland, C.; Bashyam, B.; et al. Effect of Legumes as Part of a Low Glycemic Index Diet on Glycemic Control and Cardiovascular Risk Factors in Type 2 Diabetes Mellitus. Arch. Intern. Med. 2012, 172, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Shams, H.; Tahbaz, F.; Entezari, M.h.; Abadi, A. Effects of cooked lentils on glycemic control and blood lipids of patients with type 2 diabetes. ARYA Atheroscler. J. 2008, 4, 1–5. [Google Scholar]
- Simpson, H.C.; Simpson, R.W.; Lousley, S.; Carter, R.D.; Geekie, M.; Hockaday, T.D.; Mann, J.I. A high carbohydrate leguminous fibre diet improves all aspects of diabetic control. Lancet 1981, 317, 1–5. [Google Scholar] [CrossRef]
- Kim, S.J.; De Souza, R.J.; Choo, V.L.; Ha, V.; I Cozma, A.; Chiavaroli, L.; Mirrahimi, A.; Mejia, S.B.; Di Buono, M.; Bernstein, A.M.; et al. Effects of dietary pulse consumption on body weight: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2016, 103, 1213–1223. [Google Scholar] [CrossRef]
- Abete, I.; Parra, D.; Martínez, J.A. Legume-, Fish-, or High-Protein-Based Hypocaloric Diets: Effects on Weight Loss and Mitochondrial Oxidation in Obese Men. J. Med. Food 2009, 12, 100–108. [Google Scholar] [CrossRef]
- Abeysekara, S.; Chilibeck, P.D.; Vatanparast, H.; Zello, G.A. A pulse-based diet is effective for reducing total and LDL-cholesterol in older adults. Br. J. Nutr. 2012, 108, S103–S110. [Google Scholar] [CrossRef]
- Alizadeh, M.; Gharaaghaji, R.; Gargari, B.P. The Effects of Legumes on Metabolic Features, Insulin Resistance and Hepatic Function Tests in Women with Central Obesity: A Randomized Controlled Trial. Int. J. Prev. Med. 2014, 5, 710–720. [Google Scholar]
- Crujeiras, A.B.; Parra, D.; Abete, I.; Martínez, J.A. A hypocaloric diet enriched in legumes specifically mitigates lipid peroxidation in obese subjects. Free Radic. Res. 2007, 41, 498–506. [Google Scholar] [CrossRef]
- Gravel, K.; Lemieux, S.; Asselin, G.; Dufresne, A.; Lemay, A.; Forest, J.-C.; Dodin, S. Effects of pulse consumption in women presenting components of the metabolic syndrome: A randomized controlled trial. Mediterr. J. Nutr. Metab. 2010, 3, 143–151. [Google Scholar] [CrossRef]
- Hermsdorff, H.H.M.; Zulet, M.A.; Abete, I.; Martínez, J.A. A legume-based hypocaloric diet reduces proinflammatory status and improves metabolic features in overweight/obese subjects. Eur. J. Nutr. 2010, 50, 61–69. [Google Scholar] [CrossRef]
- Kazemi, M.; McBreairty, L.E.; Chizen, D.R.; Pierson, R.A.; Chilibeck, P.D.; Zello, G.A. A Comparison of a Pulse-Based Diet and the Therapeutic Lifestyle Changes Diet in Combination with Exercise and Health Counselling on the Cardio-Metabolic Risk Profile in Women with Polycystic Ovary Syndrome: A Randomized Controlled Trial. Nutrients 2018, 10, 1387. [Google Scholar] [CrossRef]
- Mollard, R.; Luhovyy, B.L.; Panahi, S.; Núñez, M.; Hanley, A.; Anderson, G.H. Regular consumption of pulses for 8 weeks reduces metabolic syndrome risk factors in overweight and obese adults. Br. J. Nutr. 2012, 108, S111–S122. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.; Cehun, M.; Chronopoulos, A. Effects of long-term consumption and single meals of chickpeas on plasma glucose, insulin, and triacylglycerol concentrations. Am. J. Clin. Nutr. 2004, 79, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Saraf-Bank, S.; Esmaillzadeh, A.; Faghihimani, E.; Azadbakht, L. Effects of Legume-Enriched Diet on Cardiometabolic Risk Factors among Individuals at Risk for Diabetes: A Crossover Study. J. Am. Coll. Nutr. 2015, 35, 1–10. [Google Scholar] [CrossRef]
- Tonstad, S.; Malik, N.; Haddad, E. A high-fibre bean-rich diet versus a low-carbohydrate diet for obesity. J. Hum. Nutr. Diet. 2013, 27, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Winham, D.M.; Hutchins, A. Baked bean consumption reduces serum cholesterol in hypercholesterolemic adults. Nutr. Res. 2007, 27, 380–386. [Google Scholar] [CrossRef]
- Winham, D.M.; Hutchins, A.M.; Johnston, C.S. Pinto bean consumption reduces biomarkers for heart disease risk. J. Am. Coll. Nutr. 2007, 26, 243–249. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Rind, D.; Devereaux, P.J.; Montori, V.M.; Freyschuss, B.; Vist, G.; et al. GRADE guidelines 6. Rating the quality of evidence—Imprecision. J. Clin. Epidemiol. 2011, 64, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Rosner, B. Fundamentals of Biostatistics, 8th ed.; Cengage Learning: Boston, MA, USA, 2015; pp. 403–405. [Google Scholar]
- National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand. Available online: https://www.nrv.gov.au/nutrients/dietary-fibre (accessed on 7 March 2020).
- Röder, P.V.; Wu, B.; Liu, Y.; Han, W. Pancreatic regulation of glucose homeostasis. Exp. Mol. Med. 2016, 48, e219. [Google Scholar] [CrossRef]
- Ferreira, H.; Vasconcelos, M.; Gil, A.M.; Pinto, E. Benefits of pulse consumption on metabolism and health: A systematic review of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 1–12. [Google Scholar] [CrossRef]
- Goff, H.D.; Repin, N.; Fabek, H.; El Khoury, D.; Gidley, M.J. Dietary fibre for glycaemia control: Towards a mechanistic understanding. Bioact. Carbohydr. Diet. Fibre 2018, 14, 39–53. [Google Scholar] [CrossRef]
- Figueira, N.; Curtain, F.; Beck, E.; Grafenauer, S. Consumer Understanding and Culinary Use of Legumes in Australia. Nutrients 2019, 11, 1575. [Google Scholar] [CrossRef] [PubMed]
| Study | Design a and Duration | n (I/C) b | Characteristics c | (M/F) d | Age (Years) | Anti-Diabetic Medication e | Legume Type f Dose (g/day) g Control | %E (CHO: Fat: Pro) h Dietary Fibre i (g/day) | Energy Balance |
|---|---|---|---|---|---|---|---|---|---|
| Hassanzadeh-Rostami et al. 2019 [23] | P 8 wks | (20/23) | T2DM, BMI I: 27.3 ± 3.4 C: 26.5 ± 3.2 | (13/32) # | I: 59.6 ± 6.0 C: 56.1 ± 7.2 | Yes | Legume: NR 77 Control: Legume free | I: 52:32:17 C: 56:29:16 | Isocaloric |
| I: 17.3 ± 4.7 C: 17.2 ± 4.7 | |||||||||
| Hosseinpour-Niazi et al. 2015 [24] | C 8 wks | 31 | T2DM, BMI: I: 27.7 ± 3.3 C: 27.8 ± 3.3 | (7/24) | 58.1 ± 6.0 | Yes, ≥3 months | Legume: Mixed (L, CP, B, P) 83 Control: Legume free | I: 54:32:14 C: 52:34:15 | Isocaloric |
| I: 31.4 ± 8.4 C: 26.9 ± 7.2 | |||||||||
| Jenkins et al. 2012 [25] | P 13 wks | (60/61) | T2DM, BMI: I: 31.4 ± 7 C:29.9 ± 5.5 | (61/60) | I: 58.0 ± 10.1 C: 61.0 ± 7.8 | Yes, ≥2 months | Legume: Mixed (L, CP, B) 190 Control: Wheat-based diet | I: 45:31:23 C: 48:29:21 | Isocaloric |
| I: 39.4 ± 13.1 C: 26.9 ± 5.2 | |||||||||
| Shams et al. 2010 [26] | C 6 wks | 30 | T2DM, BMI: 28.9 ± 4.1 | NR | 50.2 ± 3.8 | NR | Legumes: Lentils 50 Control: Legume free | I: 48:31:18 C: 53:28:20 | Isocaloric |
| I: 28.6 ± 3.4 C: 23.3 ± 6.4 | |||||||||
| Simpson et al. 1981 [27] | C 6 wks | 18 | T2DM BMI: NR | (10/8) | 52.5 ± 12.3 | Yes, (n = 15) | Legumes: Beans (Mixed) NR Control: Low CHO | I: 61:18:21 C: 40:39:21 | Isocaloric |
| I: 96.6 C: 17.6 | |||||||||
| Simpson et al. 1981 [27] | C 6 wks | 9 | T1DM BMI: NR | (4/5) | 41.2 ± 14.8 | Insulin (n = 9) | Legumes: Beans (Mixed) NR Control: Low CHO | I: 61:18:21 C: 40:18:21 | Isocaloric |
| I: 96.6 C: 17.6 |
| Study | Design a and Duration | n (I/C) b | Characteristics c | (M/F) d | Age (Years) | Anti-Diabetic Medication e | Legume Type f Dose (g/day) g Control | %E (CHO: Fat: Pro) h Dietary Fibre i (g/day) | Energy Balance |
|---|---|---|---|---|---|---|---|---|---|
| Abete et al. 2009 [29] | P 8 wks | (8/10) | Obese, BMI: 31.8 ± 4.1 | (18/0) | 38.0 ± 7.0 | NR | Legumes: NR NR Control: No legumes | I: 52:30:18 C: 51:33:19 | Hypocaloric |
| I: 26.5 ± 15.3 C: 20.3 ± 17.1 | |||||||||
| Abeysekara et al. 2012 [30] | C 8 wks | 87 | BMI: 27.5 ± 4.5 | (30/57) | 59.7 ± 6.3 | (n = 3) | Legumes: Mixed (L, CP, B, P) 250 Control: Usual diet | I: 48:37:15 C: 47:38:16 | Isocaloric |
| I: 30.0 ± 15.0 C: 22.0 ± 10.0 | |||||||||
| Alizadeh et al. 2014 [31] | P 6 wks | (17/17) | WC > 88 cm BMI: NR | (0/34) | 36.1 ± 8.2 | Nil | Legumes: Mixed (L, CP, P, B) 190 Control: No legumes | I: 55:30:15 C: 55:30:15 | Hypocaloric |
| NR | |||||||||
| Crujeiras et al. 2007 [32] | P 8 wks | (15/15) | Obese, BMI: 32.0 ± 5.3 | (17/13) | 36.0 ± 8.0 | Nil | Legumes: Mixed (L, CP, B, P) NR Control: No legumes | I: 50:33:19 C: 51:31:19 | Hypocaloric |
| I: 25.0 ± 6.0 C: 18.0 ± 5.0 | |||||||||
| Gravel et al. 2010 [33] | P 16 wks | (60/54) | 2 risk factors for MetSyn, BMI: I: 29.6 ± 4.5 C: 30.1 ± 5.7 | (0/114) | I: 52.5 ± 7.5 C: 50.0 ± 9.6 | Nil | Legume: Mixed (L, CP, B, P) 81 Control: No legumes | I: 49:33:17 C: 49:32:18 | Isocaloric |
| I: 22.9 ± 10.4 C: 18.2 ± 9.2 | |||||||||
| Hermsdorff et al. 2011 [34] | P 8 wks | (15/15) | Obese, BMI: 32.5 ± 4.5 | (17/13) | 36.0 ± 8.0 | Nil | Legumes: Mixed (L, CP, B, P) 113 Control: No legumes | I: 50:33:19 C: 51:31:19 | Hypocaloric |
| I: 26.0 ± 6.0 C: 18.0 ± 5.0 | |||||||||
| Kazemi et al. 2018 [35] | P 16 wks | (30/31) | PCOS, BMI: I: 33.3 ± 9.0 C: 34.0 ± 9.8 | (0/61) | I: 27.0 ± 4.6 C: 26.9 ± 4.4 | Metformin (I/C, n = 18/20) | Legumes: Mixed (L, CP, B, P) ~244 Control: Legume free TLC | I: 57:30:16 C: 54:29:18 | Isocaloric |
| I: 33.3 ± 8.2 C: 24.5 ± 9.5 | |||||||||
| Mollard et al. 2012 [36] | P 8 wks | (19/21) | Overweight/Obese, BMI: 32.8 ± 4.4 | (11/29) | 45.5 ± 6.3 | Nil | Legumes: Mixed (L, CP, B, P) 128 Control: No legumes | I: 55:29:16 C: 51:32:17 | Isocaloric |
| I: 28.9 ± 9.1 C: 21.4 ± 6.4 | |||||||||
| Nestel et al. 2004 [37] | C 6 wks | 19 | Healthy subjects, BMI 25.6 ± 3.2 | (9/10) | 56.6 ± 7.6 | NR | Legumes: Chickpeas 140 Control: Wheat-based foods | I: 47:30:19 C: 44:31:19 | Isocaloric |
| I: 33.0 ± 8.0 C: 26.0 ± 13.0 | |||||||||
| Saraf-Bank et al. 2016 [38] | C 6 wks | 26 | 1° relatives w/T2DM, BMI: I: 28.7 ± 4.1 C: 29.0 ± 4.5 | (12/14) | 50.0 ± 6.6 | Nil | Legumes: Mixed (L, B) 111 Control: No legumes | I: 66:20:16 C: 67:19:17 | Isocaloric |
| I: 38.4 ± 14.4 C: 32.3 ± 15.0 | |||||||||
| Tonstad et al. 2014 [39] ** | P 16 wks | (64/59) | Obese, T2DM (n = 35), BMI: I: 36.6 ± 3.8 C: 36.3 ± 4.1 | (45/128) # | I: 47.7 ± 10.2 C: 49.1 ± 11.2 | Nil | Legumes: Beans (Mixed) 285 Control: Low CHO | I: 52:28:19 C: 32:42:27 | Isocaloric |
| I: 37.1 ± 21.9 C: 17.3 ± 10.2 | |||||||||
| Winham et al. 2007 [40] (BB) | C 8 wks | 23 | Hyperlipidaemia, BMI: 27.4 ± 4.3 | (10/13) | 45.9 ± 10.5 | NR | Legumes: Navy beans 95 Control: Carrots | I: 51:31:17 C: 51:33:17 | Isocaloric |
| I: 25.5 ± 17.1 C: 20.7 ± 16.2 |
| Study | Design a and Duration | n (I/C) b | Characteristics c | (M/F) d | Age (Years) | Anti-Diabetic Medication e | Legume Type f Dose g (g/day) Control | %E (CHO: Fat: Pro) h Dietary Fibre i (g/day) | Energy Balance |
|---|---|---|---|---|---|---|---|---|---|
| Winhman et al. 2007 [41] | C 8 wks | 16 | Mild-mod IR, BMI: 27.8 ± 0.9 | (7/9) | 43.0 ± 12.0 | NR | Legume: Pinto Beans 95 Control: Carrots | I: 51:32:15 C: 50:32:17 | Isocaloric |
| I: 23.0 ± 15.6 C: 21.0 ± 15.6 | |||||||||
| Winham et al. 2007 [41] | C 8 wks | 16 | Mild-mod IR, BMI: 27.8 ± 0.9 | (7/9) | 43.0 ± 12.0 | NR | Legume: Black-eyed peas 95 Control: Carrots | I: 53:31:16 C: 50:32:17 | Isocaloric |
| I: 19.0 ± 15.7 C: 21.0 ± 15.6 |
| Outcome, n Studies, (I/C) | Criteria for Downgrading Quality | Assessment and Justification | Quality of Evidence a |
|---|---|---|---|
| FBG, 4 studies, (141/145) | Initial Quality | High; Randomised Controlled Trials only | Very Low |
| Risk of bias | Evidence not downgraded; Two studies were rated as ‘low RoB’, and two were rated as ‘some concerns’. Limitations were not serious | ||
| Inconsistency | Evidence not downgraded; Visual inspection identified consistency within size of effect | ||
| Indirectness | Downgrade by one level; Population (T2DM; direct), intervention (Legume dose (g/day) varied between studies), comparisons (control interventions varied between studies), outcomes (FBG; direct) | ||
| Imprecision | Downgrade by one level; Insufficient sample size according to OIS | ||
| Publication bias | Downgrade by one level; Grey literature sources were not included in defined search strategy | ||
| HbA1c, 3 studies, (98/102) | Initial Quality | High; Randomised Controlled Trials only | Very Low |
| Risk of bias | Evidence not downgraded; One study rated as ‘low RoB’, one ‘some concerns’ and one ‘high’ due to absence of wash-out period, contribution of study was small. Limitations were not serious | ||
| Inconsistency | Evidence not downgraded; Visual inspection identified consistency within size of effect | ||
| Indirectness | Downgrade by one level; Population (T2DM; consistent), intervention (Legume dose (g/day) varied between studies), comparisons (control interventions varied between studies), outcomes (HbA1c; direct) | ||
| Imprecision | Downgrade by one level; Insufficient sample size according to OIS | ||
| Publication bias | Downgrade by one level; Grey literature sources were not included in defined search strategy |
| Study | n (I/C) | FBG Baseline (mmol/L) | FBG Endpoint (mmol/L) | Statistical Significance | |
| Within-Group | Between-Group | ||||
| Hassanzadeh-Rostami et al. 2019 [23] | I (n = 20) C (n = 23) | 7.99 (6.37, 8.82) * 9.60 (6.38, 13.1) | 7.38 (6.22, 8.44) 8.21 (6.60, 9.64) | NS NS | NS |
| Hosseinpour-Niazi et al. 2015 [24] | I (n = 31) C (n = 31) | 7.94 ± 3.09 8.19 ± 2.97 | 6.35 ± 2.26 7.11 ± 2.75 | p < 0.05 p < 0.05 | p < 0.001 |
| Jenkins et al. 2012 [25] | I (n = 60) C (n = 61) | 7.83 ± 1.30 7.44 ± 1.52 | 7.33 ± 1.30 7.05 ± 1.29 | p < 0.05 NS | p = 0.01 |
| Shams et al. 2010 [26] | I (n = 30) C (n = 30) | 8.56 ± 0.82 8.58 ± 0.69 | 8.43 ± 0.70 8.50 ± 0.57 | p < 0.05 NS | p < 0.05 |
| Study | n (I/C) | FBI Baseline (pmol/L) | FBI Endpoint (pmol/L) | Statistical Significance | |
| Within-Group | Between-Group | ||||
| Hassanzadeh-Rostami et al. 2019 [23] | I (n = 20) C (n = 23) | 15.7 (9.10, 35.4) * 8.20 (5.60, 11.3) | 13.9 (8.20, 23.8) 7.50 (4.90, 10.6) | NS NS | p = 0.02 |
| Hosseinpour-Niazi et al. 2015 [24] | I (n = 31) C (n = 31) | 48.6 ± 20.0 45.0 ± 23.4 | 27.0 ± 10.0 36.0 ± 16.7 | p < 0.05 p < 0.05 | p = 0.006 |
| Study | n (I/C) | HbA1c Baseline (%) | HbA1c Endpoint (%) | Statistical Significance | |
| Within-Group | Between-Group | ||||
| Hassanzadeh-Rostami et al. 2019 [23] | I (n = 20) C (n = 23) | 7.70 (7.00, 9.10) * 9.50 (8.10, 11.3) | 7.60 (7.00, 9.30) 9.00 (7.70, 11.5) | NS NS | p = 0.04 |
| Jenkins et al. 2012 [25] | I (n = 60) C (n = 61) | 7.40 ± 0.58 7.20 ± 0.59 | 6.90 ± 0.58 6.90 ± 0.39 | p < 0.05 NS | p < 0.01 |
| Simpson et al. 1981 [27] (T2DM) | I (n = 18) C (n = 18) | NR NR | 8.60 ± 1.60 9.60 ± 2.30 | NR NR | p < 0.02 |
| Simpson et al. 1981 [27] (T1DM) | I (n = 9) C (n = 9) | NR NR | 9.80 ± 1.80 10.0 ± 2.30 | NR NR | NS |
| Study | n (I/C) | 2-h PPG Baseline (mmol/L) | 2-h PPG Endpoint (mmol/L) | Statistical Significance | |
| Within-Group | Between-Group | ||||
| Simpson et al. 1981 [27] (T2DM) | I (n = 18) C (n = 18) | NR NR | 8.10 ± 1.60 9.10 ± 2.30 | NR NR | p < 0.05 |
| Simpson et al. 1981 [27] (T1DM) | I (n = 9) C (n = 9) | NR NR | 9.10 ± 3.30 12.2 ± 3.40 | NR NR | p < 0.02 |
| Study | n (I/C) | FBG Baseline (mmol/L) | FBG Endpoint (mmol/L) | Statistical Significance | |
|---|---|---|---|---|---|
| Within-Group | Between-Group | ||||
| Abete et al. 2009 [29] | I (n = 8) C (n = 10) | NR NR | NR NR | p < 0.05 NS | NS |
| Abeysekara et al. 2012 [30] | I (n = 87) C (n = 87) | 4.37 ± 1.40 4.47 ± 1.92 | 4.39 ± 1.36 4.17 ± 1.51 | NS NS | NS |
| Alizadeh et al. 2014 [31] | I (n = 17) C (n = 17) | 5.09 ± 1.33 5.12 ± 1.46 | 5.12 ± 1.43 5.21 ± 1.43 | NS NS | NS |
| Crujeiras et al. 2007 [32] | I (n = 15) C (n = 15) | NR NR | NR NR | NS NS | NS |
| Gravel et al. 2010 [33] | I (n = 60) C (n = 54) | 5.30 ± 0.64 5.20 ± 0.54 | 5.28 ± 0.69 5.34 ± 0.61 | NS NS | NS |
| Hermsdorff et al. 2011 [34] | I (n = 15) C (n = 15) | 5.17 ± 0.32 5.13 ± 0.53 | 5.13 ± 0.29 4.98 ± 0.37 | NS NS | NS |
| Kazemi et al. 2018 [35] | I (n = 30) C (n = 31) | 5.00 ± 1.50 5.60 ± 1.40 | 4.60 ± 1.30 4.80 ± 1.60 | p < 0.01 p < 0.01 | NS |
| 6-month follow-up | I (n = 16) C (n = 16) | 5.30 ± 1.70 5.50 ± 1.50 | 4.90 ± 0.20 5.30 ± 0.90 | NS NS | NS |
| 12-month follow-up | I (n = 12) C (n = 13) | 5.20 ± 1.10 5.50 ± 1.40 | 4.90 ± 0.60 5.30 ± 0.50 | NS NS | NS |
| Mollard et al. 2012 [36] | I (n = 19) C (n = 21) | NR NR | NR NR | NS NS | NS |
| Nestel et al. 2004 [37] | I (n = 19) C (n = 19) | 5.20 ± 0.40 5.20 ± 0.40 | 4.90 ± 0.40 5.10 ± 0.50 | NS NS | NS |
| Saraf-Bank et al. 2016 [38] | I (n = 26) C (n = 26) | 5.35 ± 2.08 5.28 ± 2.34 | 5.38 ± 2.28 5.42 ± 2.28 | NS NS | NS |
| Tonstad et al. 2014 [39] | I (n = 64) C (n = 59) | 5.60 ± 1.90 5.90 ± 1.90 | 5.30 ± 1.60 5.60 ± 1.50 | NS NS | NS |
| 12-month follow-up | I (n = 30) C (n = 24) | 5.30 ± 1.10 5.30 ± 0.80 | 5.30 ± 0.90 5.30 ± 0.80 | NS NS | NS |
| Winham et al. 2007 [40] (BB) | I (n = 23) C (n = 23) | 5.61 ± 1.81 5.77 ± 2.21 | 5.49 ± 1.81 5.77 ± 1.81 | NS NS | NS |
| Study | n (I/C) | FBI Baseline (pmol/L) | FBI Endpoint (pmol/L) | Statistical Significance | |
|---|---|---|---|---|---|
| Within-Group | Between-Group | ||||
| Abete et al. 2009 [29] | I (n = 8) C (n = 10) | NR NR | NR NR | NS NS | NS |
| Abeysekara et al. 2012 [30] | I (n = 87) C (n = 87) | 75.7 ± 74.0 82.6 ± 72.2 | 74.8 ± 71.0 71.3 ± 107 | NS NS | NS |
| Alizadeh et al. 2014 [31] | I (n = 17) C (n = 17) | 113 ± 27.2 109 ± 69.3 | 114 ± 37.1 107 ± 47.0 | NS NS | NS |
| Crujeiras et al. 2007 [32] | I (n = 15) C (n = 15) | NR NR | NR NR | NS NS | NS |
| Gravel et al. 2010 [33] | I (n = 60) C (n = 54) | 89.3 ± 44.8 81.2 ± 37.3 | 88.7 ± 43.4 88.3 ± 51.4 | NS NS | NS |
| Hermsdorff et al. 2011 [34] | I (n = 15) C (n = 15) | 45.0 ± 22.8 63.0 ± 60.0 | 35.4 ± 24.0 49.2 ± 25.8 | NS NS | NS |
| Kazemi et al. 2018 [35] | I (n = 30) C (n = 31) | 84.0 ± 68.4 94.2 ± 74.4 | 60.0 ± 46.2 76.2 ± 61.8 | p < 0.01 p < 0.01 | NS |
| 6-month follow up | I (n = 16) C (n = 16) | 81.0 ± 76.8 87.6 ± 21.6 | 79.8 ± 67.2 100 ± 54.6 | NS NS | NS |
| 12-month follow-up | I (n = 12) C (n = 13) | 97.8 ± 91.8 108 ± 104 | 84.6 ± 57.6 99.0 ± 60.0 | p < 0.02 p < 0.02 | NS |
| Mollard et al. 2012 [36] | I (n = 19) C (n = 21) | NR NR | NR NR | NS NS | NS |
| Nestel et al. 2004 [37] | I (n = 19) C (n = 19) | 39.6 ± 21.6 39.6 ± 21.6 | 47.4 ± 27.0 49.2 ± 28.2 | NS NS | NS |
| Winham et al. 2007 [40] (BB) | I (n = 23) C (n = 23) | 126 ± 57.6 156 ± 143.9 | 120 ± 57.6 126 ± 57.6 | NS NS | NS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bielefeld, D.; Grafenauer, S.; Rangan, A. The Effects of Legume Consumption on Markers of Glycaemic Control in Individuals with and without Diabetes Mellitus: A Systematic Literature Review of Randomised Controlled Trials. Nutrients 2020, 12, 2123. https://doi.org/10.3390/nu12072123
Bielefeld D, Grafenauer S, Rangan A. The Effects of Legume Consumption on Markers of Glycaemic Control in Individuals with and without Diabetes Mellitus: A Systematic Literature Review of Randomised Controlled Trials. Nutrients. 2020; 12(7):2123. https://doi.org/10.3390/nu12072123
Chicago/Turabian StyleBielefeld, Dale, Sara Grafenauer, and Anna Rangan. 2020. "The Effects of Legume Consumption on Markers of Glycaemic Control in Individuals with and without Diabetes Mellitus: A Systematic Literature Review of Randomised Controlled Trials" Nutrients 12, no. 7: 2123. https://doi.org/10.3390/nu12072123
APA StyleBielefeld, D., Grafenauer, S., & Rangan, A. (2020). The Effects of Legume Consumption on Markers of Glycaemic Control in Individuals with and without Diabetes Mellitus: A Systematic Literature Review of Randomised Controlled Trials. Nutrients, 12(7), 2123. https://doi.org/10.3390/nu12072123






