An Infant Formula with Partially Hydrolyzed Whey Protein Supports Adequate Growth and Is Safe and Well-Tolerated in Healthy, Term Infants: A Randomized, Double-Blind, Equivalence Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participating Centers
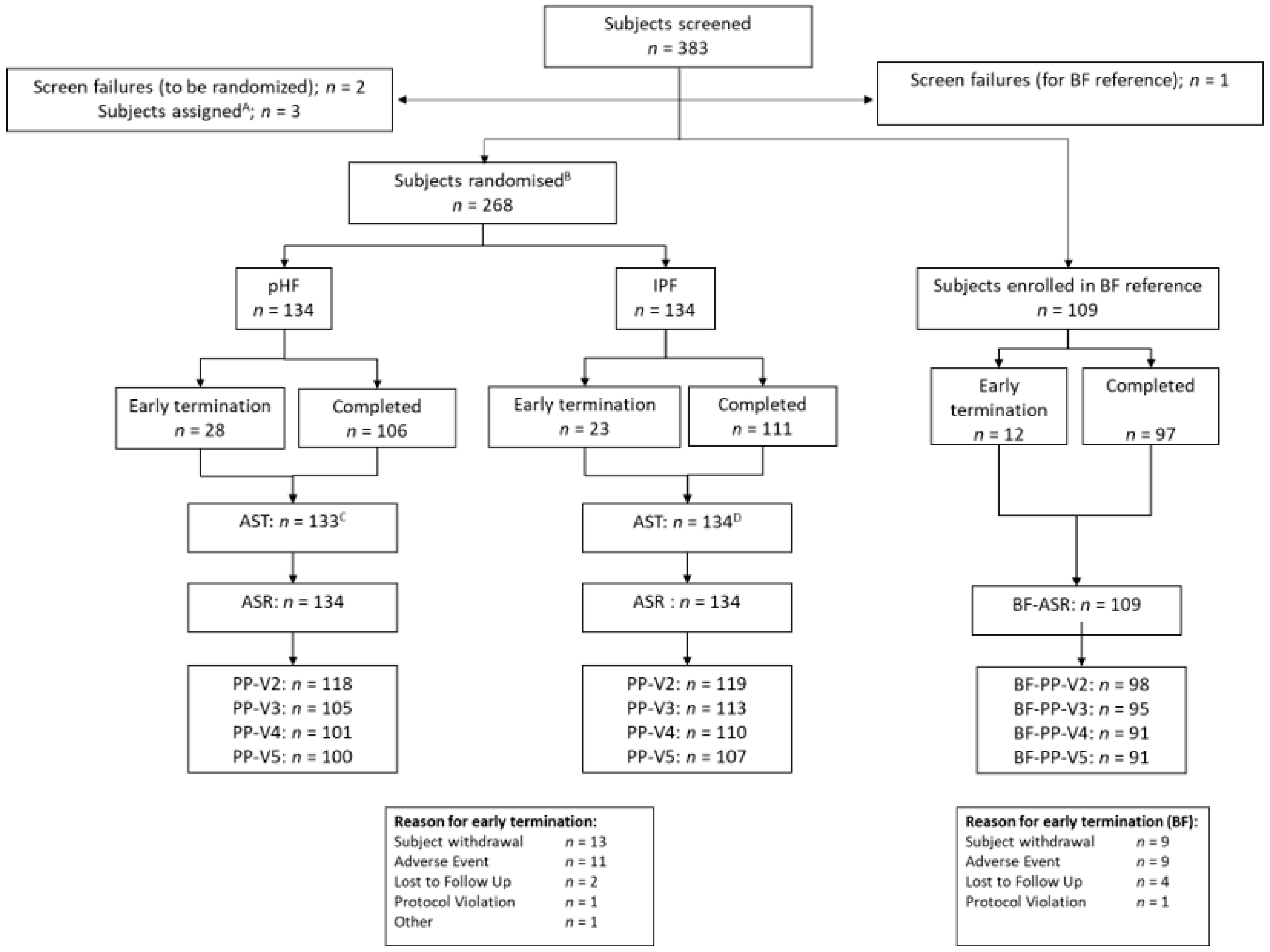
2.2. Subjects and Study Design
2.3. Study Products
2.4. Measurements
2.5. Statistics
3. Results
3.1. Subject Characteristics
3.2. Study Product Intake
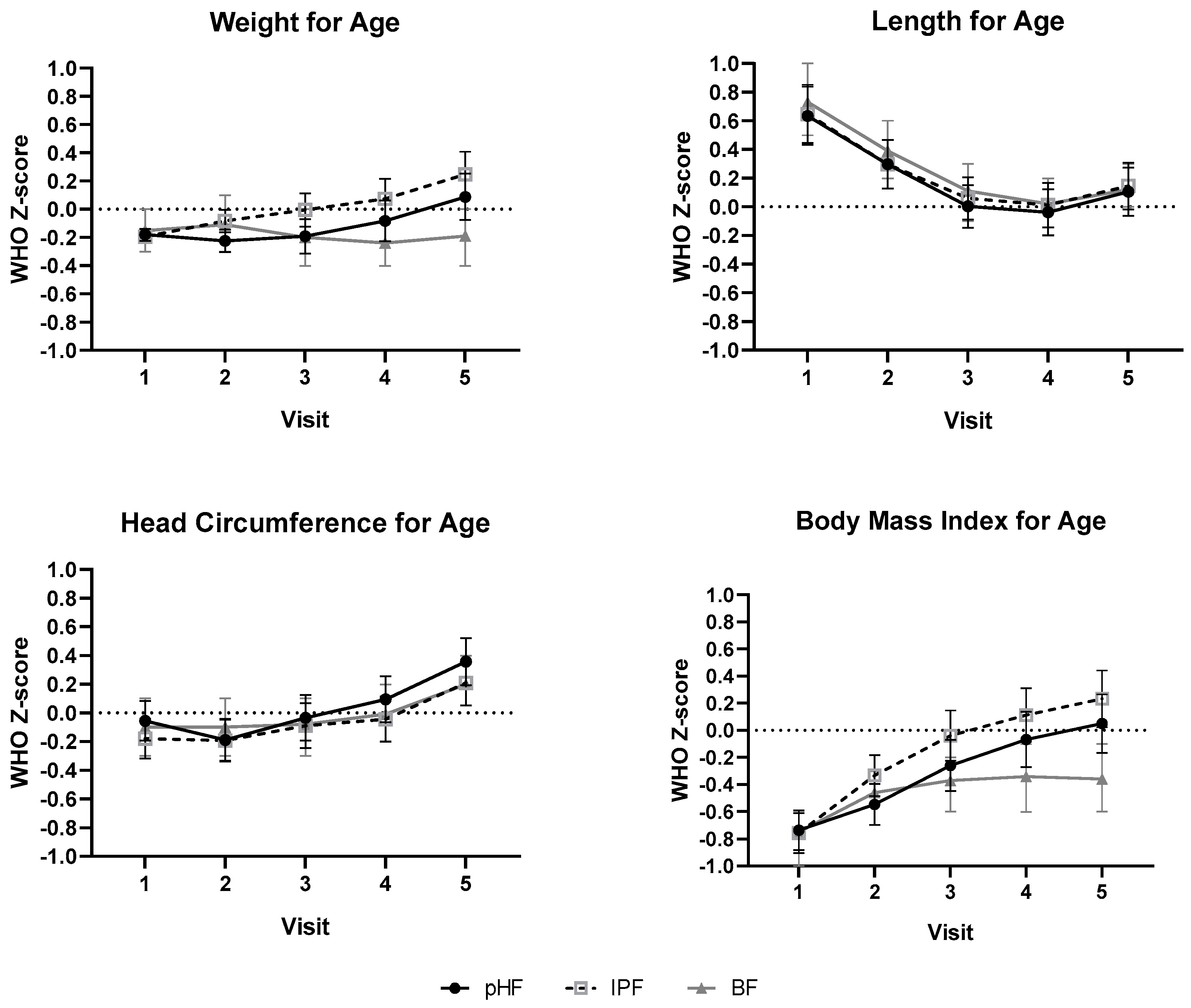
3.3. Growth Outcomes
3.4. Serum Parameters
3.5. Gastrointestinal Tolerance
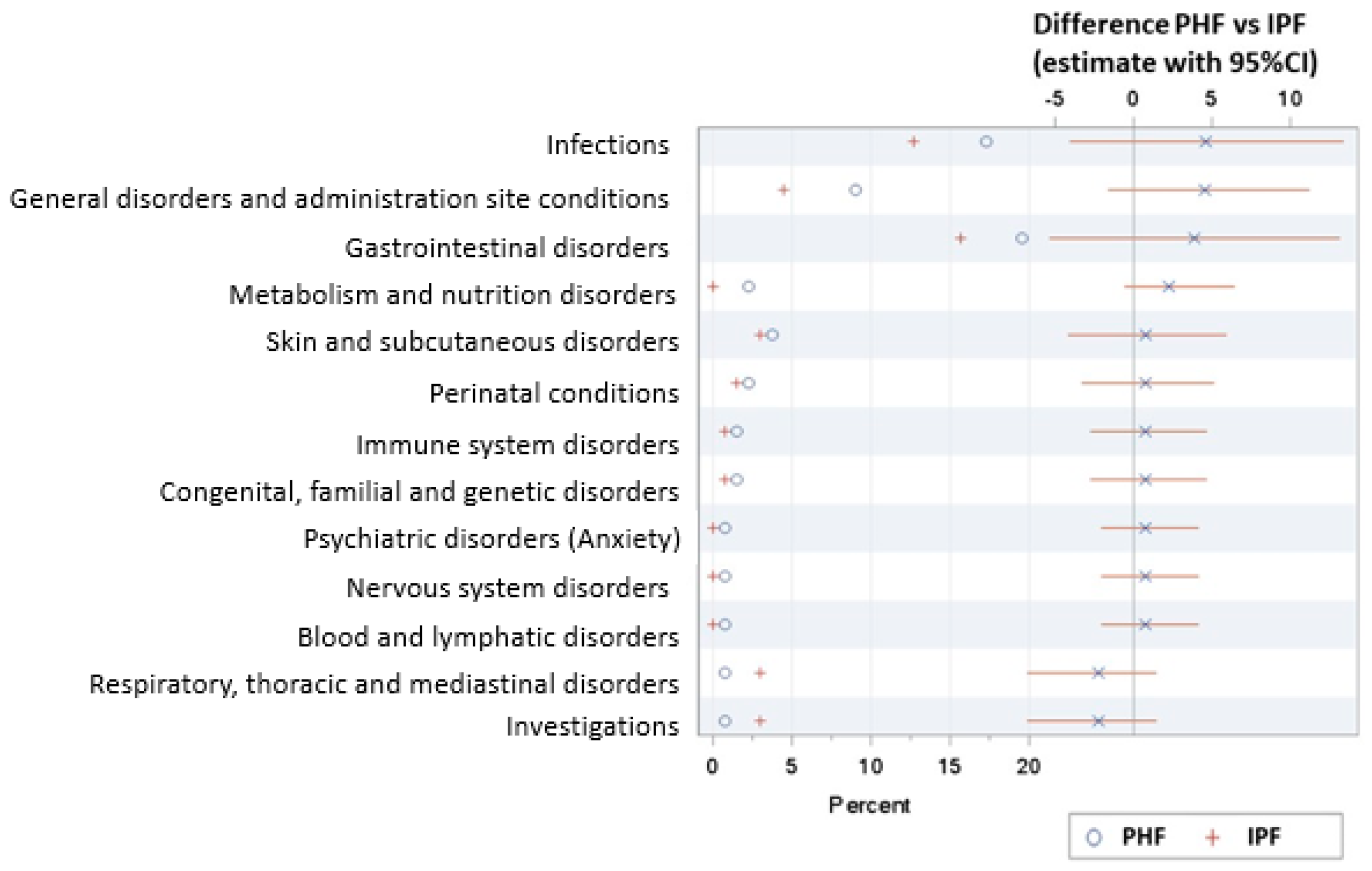
3.6. Adverse Events
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EFSA; Panel on Dietetic Products; Nutrition and Allergies (NDA). Scientific opinion on the essential composition of infant and follow-on formulae. EFSA J. 2014, 12, 3760. [Google Scholar] [CrossRef]
- Oddy, W.H. Breastfeeding, childhood asthma, and allergic disease. Ann. Nutr. Metab. 2017, 70, 26–36. [Google Scholar] [CrossRef]
- Kramer, M.S. Breastfeeding and allergy: The evidence. Ann. Nutr. Metab. 2011, 59, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Muraro, A.; Halken, S.; Arshad, S.H.; Beyer, K.; Dubois, A.E.; Du Toit, G.; Eigenmann, P.A.; Grimshaw, K.E.; Hoest, A.; Lack, G.; et al. EAACI food allergy and anaphylaxis guidelines. Primary prevention of food allergy. Allergy 2014, 69, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, D.M.; Spergel, J.M.; Assa‘ad, A.H.; Pongracic, J.A. Primary prevention of allergic disease through nutritional interventions. J. Allergy Clin. Immunol. Pract. 2013, 1, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Osborn, D.A.; Sinn, J. Formulas containing hydrolysed protein for prevention of allergy and food intolerance in infants. Cochrane Database Syst. Rev. 2006. [Google Scholar] [CrossRef]
- Boyle, R.J.; Tang, M.L.; Chiang, W.C.; Chua, M.C.; Ismail, I.; Nauta, A.; Hourihane, J.O.B.; Smith, P.; Gold, M.; Ziegler, J.; et al. Prebiotic-supplemented partially hydrolysed cow’s milk formula for the prevention of eczema in high-risk infants: A randomized controlled trial. Allergy 2016, 71, 701–710. [Google Scholar] [CrossRef] [PubMed]
- van’t Land, B.; Schijf, M.; van Esch, B.C.; van Bergenhenegouwen, J.; Bastiaans, J.; Schouten, B.; Boon, L.; Garssen, J. Regulatory T-cells have a prominent role in the immune modulated vaccine response by specific oligosaccharides. Vaccine 2010, 28, 5711–5717. [Google Scholar] [CrossRef]
- Schouten, B.; van Esch, B.C.; Hofman, G.A.; Boon, L.; Knippels, L.M.; Willemsen, L.E.; Garssen, J. Oligosaccharide-induced whey-specific CD25(+) regulatory T-cells are involved in the suppression of cow milk allergy in mice. J. Nutr. 2010, 140, 835–841. [Google Scholar] [CrossRef]
- Van Esch, B.C.; Schouten, B.; de Kivit, S.; Hofman, G.A.; Knippels, L.M.; Willemsen, L.E.; Garssen, J. Oral tolerance induction by partially hydrolyzed whey protein in mice is associated with enhanced numbers of Foxp3+ regulatory T-cells in the mesenteric lymph nodes. Pediatr. Allergy Immunol. 2011, 22, 820–826. [Google Scholar] [CrossRef]
- Boyle, R.J.; Ierodiakonou, D.; Khan, T.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Jarrold, K.; Afxentiou, T.; Reeves, T.; Cunha, S.; et al. Hydrolysed formula and risk of allergic or autoimmune disease: Systematic review and meta-analysis. BMJ 2016, 352, i974. [Google Scholar] [CrossRef]
- Osborn, D.A.; Sinn, J.K.; Jones, L.J. Infant formulas containing hydrolysed protein for prevention of allergic disease. Cochrane Database Syst. Rev. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Picaud, J.C.; Rigo, J.; Normand, S.; Lapillonne, A.; Reygrobellet, B.; Claris, O.; Salle, B.L. Nutritional efficacy of preterm formula with a partially hydrolyzed protein source: A randomized pilot study. J. Pediatr. Gastroenterol. Nutr. 2001, 32, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Hernell, O.; Lonnerdal, B. Nutritional evaluation of protein hydrolysate formulas in healthy term infants: Plasma amino acids, hematology, and trace elements. Am. J. Clin. Nutr. 2003, 78, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Rigo, J.; Senterre, J. Metabolic balance studies and plasma amino acid concentrations in preterm infants fed experimental protein hydrolysate preterm formulas. Acta Paediatr. 1994, 405, 98–104. [Google Scholar] [CrossRef]
- Mennella, J.A.; Ventura, A.K.; Beauchamp, G.K. Differential growth patterns among healthy infants fed protein hydrolysate or cow-milk formulas. Pediatrics 2011, 127, 110–118. [Google Scholar] [CrossRef]
- Rzehak, P.; Sausenthaler, S.; Koletzko, S.; Reinhardt, D.; von Berg, A.; Kramer, U.; Berdel, D.; Bollrath, C.; Grubl, A.; Bauer, C.P.; et al. Short- and long-term effects of feeding hydrolyzed protein infant formulas on growth at < or = 6 y of age: Results from the German Infant Nutritional Intervention Study. Am. J. Clin. Nutr. 2009, 89, 1846–1856. [Google Scholar] [CrossRef]
- American Academy of Pediatrics. Clinical Testing of Infant Formulas with Respect to Nutritional Suitability for Term Infants; American Academy: Elk Grove Village, IL, USA, 1988. [Google Scholar]
- Rigo, J.; Schoen, S.; Verghote, M.; van Overmeire, B.; Marion, W.; Abrahamse-Berkeveld, M.; Alliet, P. Partially hydrolysed whey-based formulae with reduced protein content support adequate infant growth and are well tolerated: Results of a randomised controlled trial in healthy term infants. Nutrients 2019, 11, 1654. [Google Scholar] [CrossRef]
- EFSA; European Food Safety Authority. Commission Delegated Regulation (EU) 2016/127 of 25 September 2015 Supplementing Regulation (EU) NO 609/2013 of the European Parliament and of the Council as Regards the Specific Compositional and Information Requirements for Infant Formula and Follow-on Formula and as Regards Requirements on Information Relating to Infant and Young Child Feeding. Available online: https://www.fsai.ie/uploadedFiles/Reg2016_127.pdf (accessed on 22 November 2019).
- Turck, D.; Bresson, J.-L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Scientific and technical guidance for the preparation and presentation of an application for authorisation of an infant and/or follow-on formula manufactured from protein hydrolysates. EFSA J. 2017, 15, 4779. [Google Scholar]
- Villar, J.; Cheikh Ismail, L.; Victora, C.G.; Ohuma, E.O.; Bertino, E.; Altman, D.G.; Lambert, A.; Papageorghiou, A.T.; Carvalho, M.; Jaffer, Y.A.; et al. International standards for newborn weight, length, and head circumference by gestational age and sex: The newborn cross-sectional study of the intergrowth-21st Project. Lancet 2014, 384, 857–868. [Google Scholar] [CrossRef]
- Group, W.M.G.R.S. WHO Child growth standards based on length/height, weight and age. Acta Paediatr. 2006, 450, 76–85. [Google Scholar]
- WHO. The Optimal Duration of Exclusive Breastfeeding: Report of an Expert Consultation. Available online: http://www.who.int/nutrition/publications/infantfeeding/Who_NHD_01.09/en/ (accessed on 10 July 2020).
- Bekkali, N.; Hamers, S.L.; Reitsma, J.B.; Van Toledo, L.; Benninga, M.A. Infant stool form scale: Development and results. J. Pediatr. 2009, 154, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, B.; Hellmuth, C.; Haiden, N.; Olbertz, D.; Hamelmann, E.; Vusurovic, M.; Fleddermann, M.; Roehle, R.; Knoll, A.; Koletzko, B.; et al. Hydrolyzed Formula With Reduced Protein Content Supports Adequate Growth: A Randomized Controlled Noninferiority Trial. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, E.E.; Jeter, J.M.; Drulis, J.M.; Nelson, S.E.; Haschke, F.; Steenhout, P.; Brown, C.; Maire, J.-C.; Hager, C. Formula with reduced content of improved, partially hydrolyzed protein and probiotics: Infant growth and health. Mon. Kinderheilkd. 2003, 151, S65–S71. [Google Scholar] [CrossRef]
- Scholtens, P.A.; Goossens, D.A.; Staiano, A. Stool characteristics of infants receiving short-chain galacto-oligosaccharides and long-chain fructo-oligosaccharides: A review. World J. Gastroenterol. 2014, 20, 13446–13452. [Google Scholar] [CrossRef]
- Knol, J.; Scholtens, P.; Kafka, C.; Steenbakkers, J.; Gro, S.; Helm, K.; Klarczyk, M.; Schopfer, H.; Bockler, H.M.; Wells, J. Colon microflora in infants fed formula with galacto- and fructo-oligosaccharides: More like breast-fed infants. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 36–42. [Google Scholar] [CrossRef]
- Arslanoglu, S.; Moro, G.E.; Schmitt, J.; Tandoi, L.; Rizzardi, S.; Boehm, G. Early dietary intervention with a mixture of prebiotic oligosaccharides reduces the incidence of allergic manifestations and infections during the first two years of life. J. Nutr. 2008, 138, 1091–1095. [Google Scholar] [CrossRef]
- Moro, G.; Arslanoglu, S.; Stahl, B.; Jelinek, J.; Wahn, U.; Boehm, G. A mixture of prebiotic oligosaccharides reduces the incidence of atopic dermatitis during the first six months of age. Arch. Dis. Child 2006, 91, 814–819. [Google Scholar] [CrossRef]
- Breij, L.M.; Abrahamse-Berkeveld, M.; Vandenplas, Y.; Jespers, S.N.J.; de Mol, A.C.; Khoo, P.C.; Kalenga, M.; Peeters, S.; van Beek, R.H.T.; Norbruis, O.F.; et al. An infant formula with large, milk phospholipid-coated lipid droplets containing a mixture of dairy and vegetable lipids supports adequate growth and is well tolerated in healthy, term infants. Am. J. Clin. Nutr. 2019, 109, 586–596. [Google Scholar] [CrossRef]
- Huet, F.; Abrahamse-Berkeveld, M.; Tims, S.; Simeoni, U.; Beley, G.; Savagner, C.; Vandenplas, Y.; Hourihane, J.O. Partly fermented infant formulae with specific oligosaccharides support adequate infant growth and are well-tolerated. J. Pediatr. Gastroenterol. Nutr. 2016, 63, e43. [Google Scholar] [CrossRef]
- Heinig, M.J.; Nommsen, L.A.; Peerson, J.M.; Lonnerdal, B.; Dewey, K.G. Energy and protein intakes of breast-fed and formula-fed infants during the first year of life and their association with growth velocity: The darling study. Am. J. Clin. Nutr. 1993, 58, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Kouwenhoven, S.M.P.; Antl, N.; Finken, M.J.J.; Twisk, J.W.R.; van der Beek, E.M.; Abrahamse-Berkeveld, M.; van de Heijning, B.J.M.; Schierbeek, H.; Holdt, L.M.; van Goudoever, J.B.; et al. A modified low-protein infant formula supports adequate growth in healthy, term infants: A randomized, double-blind, equivalence trial. Am. J. Clin. Nutr. 2019, 111, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Lonnerdal, B. Infant formula and infant nutrition: Bioactive proteins of human milk and implications for composition of infant formulas. Am. J. Clin. Nutr. 2014, 99, 712S–717S. [Google Scholar] [CrossRef] [PubMed]
| Per 100 mL | PHF | IPF |
|---|---|---|
| Fat (g) | 3.4 | 3.4 |
| Saturates | 1.6 | 1.5 |
| Monounsaturates | 1.2 | 1.3 |
| Polyunsaturates | 0.5 | 0.5 |
| Linoleic acid (mg) | 419 | 445 |
| Alpha linolenic acid (mg) | 77 | 83 |
| Arachidonic acid (mg) | 11 | 11 |
| Docosahexaenoic acid (mg) | 6.4 | 10.6 |
| Protein (g) | 1.5 | 1.3 |
| Whey protein (g) | 1.5 | 0.8 |
| Casein (g) | 0.5 | |
| Carbohydrates (g) | 7.2 | 7.5 |
| scGOS/lcFOS (9:1) (g) | 0.8 | 0.8 |
| Unit | PHF (N = 118) | IPF (N = 119) | Breastfed (N = 98) | |
|---|---|---|---|---|
| Sex | ||||
| Male | n (%) | 53 (45%) | 52 (44%) | 52 (53%) |
| Female | n (%) | 65 (55%) | 67 (56%) | 46 (47%) |
| Country | ||||
| Germany | n (%) | 7 (6%) | 5 (4%) | 4 (4%) |
| Spain | n (%) | 25 (21%) | 25 (21%) | 22 (22%) |
| Finland | n (%) | 0 (0%) | 0 (0%) | 8 (8%) |
| France | n (%) | 5 (4%) | 5 (4%) | 4 (4%) |
| Poland | n (%) | 81 (69%) | 83 (70%) | 60 (61%) |
| Netherlands | n (%) | 0 (0%) | 1 (1%) | 0 (0%) |
| Age at baseline (d) | ||||
| Mean age (d) | Mean (SD) | 8 (4) | 8 (5) | 9 (4) |
| Birth characteristics | ||||
| Weight (g) | Mean (SD) | 3360 (369) | 3367 (370) | 3376 (334) |
| Length(cm) | Mean (SD) | 53 (3) | 53 (3) | 53 (3) |
| Head circumference (cm) | Mean (SD) | 34 (1) | 35 (1) | 34 (1) |
| Caesarean section | n (%) | 59 (50%) | 59 (50%) | 37 (38%) |
| Gestational age (wk) | Mean (SD) | 39.2 (1.2) | 39.2 (1.3) | 39.4 (1.1) |
| Parental characteristics | ||||
| Maternal age (y) | Mean (SD) | 29.5 (5.4) | 30.7 (5.2) | 31.9 (4.3) |
| Maternal university education (yes) | n (%) | 40 (34%) | 47 (40%) | 71 (72%) |
| Maternal BMI (kg/m2) | Mean (SD) | 24.0 (3.9) | 24.8 (4.8) | 23.5 (3.9) |
| Paternal BMI (kg/m2) | Mean (SD) | 26.4 (3.4) | 26.4 (3.0) | 26.7 (3.6) |
| Formula Intake | Visit | PHF | IPF |
|---|---|---|---|
| Average daily intake, mL/d | 2 | 618 (112) | 658 (127) |
| 3 | 744 (153) | 741 (152) | |
| 4 | 802 (156) | 784 (130) | |
| 5 | 880 (183) | 848 (155) | |
| Average daily intake per kg body weight, mL/kg/d | 2 | 151 (31) | 157 (28) |
| 3 | 148 (30) | 142 (26) | |
| 4 | 133 (23) | 128 (22) | |
| 5 | 131 (25) | 125 (24) |
| Outcome Parameter | Postnatal Age | PHF # (n = 118) | IPF # (n = 119) | Breastfed Reference § (n = 97) |
|---|---|---|---|---|
| Weight (g) | Baseline | 3393 ± 11 | 3396 ± 11 | 3423 ± 15 |
| 4 weeks | 4130 ± 20 | 4197 ± 20 | 4157 ± 24 | |
| 8 weeks | 5069 ± 35 a | 5196 ± 34 b | 5060 ± 40 a | |
| 13 weeks | 6075 ± 48 a | 6229 ± 48 b | 5952 ± 55 a | |
| 17 weeks | 6746 ± 60 b | 6882 ± 59 b | 6475 ± 67 a | |
| Length (cm) | Baseline | 52.2 ± 0.2 | 52.2 ± 0.2 | 52.5 ± 0.2 |
| 4 weeks | 54.4 ± 0.2 | 54.4 ± 0.2 | 54.7 ± 0.2 | |
| 8 weeks | 57.3 ± 0.1 | 57.4 ± 0.1 | 57.5 ± 0.2 | |
| 13 weeks | 60.6 ± 0.2 | 60.7 ± 0.2 | 60.7 ± 0.2 | |
| 17 weeks | 63.0 ± 0.2 | 63.1 ± 0.2 | 63.0 ± 0.2 | |
| Head circumference (cm) | Baseline | 34.9 ± 0.1 | 34.9 ± 0.1 | 34.9 ± 0.1 |
| 4 weeks | 36.5 ± 0.1 | 36.4 ± 0.1 | 36.4 ± 0.1 | |
| 8 weeks | 38.4 ± 0.1 | 38.3 ± 0.1 | 38.2 ± 0.1 | |
| 13 weeks | 40.3 ± 0.1 | 40.1 ± 0.1 | 40.1 ± 0.1 | |
| 17 weeks | 41.4 ± 0.1 a | 41.2 ± 0.1 a,b | 41.1 ± 0.1 b |
| Serum Parameter 2 | PHF Group (N = 27) | IPF Group (N = 38) | Breastfed Reference (N = 38) |
|---|---|---|---|
| Albumin, g/L | 37.73 (2.89) | 38.46 (2.02) | 40.15 (3.00) |
| Calcium, mmol/L | 2.66 (0.11) | 2.69 (0.06) | 2.72 (0.09) |
| Phosphate, mmol/L | 2.13 (2.03; 2.27) | 2.16 (2.04; 2.28) | 2.03 (1.88; 2.11) |
| Ferritin, ug/L 3 | 97.5 (57.9; 145.0) | 78.2 (61.1; 124.0) | 106.0 (80.1; 170.0) |
| Magnesium, mmol/L | 0.94 (0.05) | 0.94 (0.06) | 0.94 (0.05) |
| Urea, mmol/L | 3.0 (2.5; 3.2) | 2.6 (2.4; 3.2) | 1.9 (1.7; 2.3) |
| Parameter | Age | Severity | PHF (N = 133) | IPF (N = 134) | Breastfed (N = 109) |
|---|---|---|---|---|---|
| Stool frequency (n/d) 2 | 4 weeks | 1.9 (1.1;3.1) | 1.9 (1.3;2.6) | 5.2 (3.4;6.3) | |
| 8 weeks | 1.6 (1.0;2.1) | 1.3 (0.9;2.0) | 2.7 (1.6;4.9) | ||
| 13 weeks | 1.4 (0.9;2.0) | 1.2 (0.9;1.9) | 1.9 (1.0;3.6) | ||
| 17 weeks | 1.4 (1.0;2.0) | 1.4 (1.0;1.9) | 1.4 (0.9;2.9) | ||
| Diarrhea occurrence (n, %) 3,4 | 4 weeks | 8 (7%) | 4 (3%) | 37 (36%) | |
| 8 weeks | 5 (5%) | 3 (3%) | 29 (30%) | ||
| 13 weeks | 4 (4%) | 3 (3%) | 23 (24%) | ||
| 17 weeks | 7 (7%) | 8 (7%) | 19 (20%) | ||
| Regurgitation occurrence (n, %) 4 | 4 weeks | Occasionally | 101 (82%) | 101 (82%) | 96 (94%) |
| Commonly | 48 (39%) | 41 (33%) | 62 (61%) | ||
| Frequently | 23 (19%) | 21 (17%) | 41 (40%) | ||
| 8 weeks | Occasionally | 84 (76%) | 94 (78%) | 87 (89%) | |
| Commonly | 32 (29%) | 38 (32%) | 50 (51%) | ||
| Frequently | 16 (14%) | 19 (16%) | 37 (38%) | ||
| 13 weeks | Occasionally | 65 (61%) | 79 (68%) | 78 (80%) | |
| Commonly | 30 (28%) | 28 (24%) | 42 (43%) | ||
| Frequently | 14 (13%) | 16 (14%) | 28 (29%) | ||
| 17 weeks | Occasionally | 60 (56%) | 68 (61%) | 75 (77%) | |
| Commonly | 22 (21%) | 29 (26%) | 39 (40%) | ||
| Frequently | 7 (6%) | 18 (16%) | 24 (25%) | ||
| Vomiting occurrence (n, %) 4 | 4 weeks | ≥1 day | 42 (34%) | 31 (25%) | 28 (28%) |
| ≥2–3 days | 10 (8%) | 9 (7%) | 11 (11%) | ||
| 8 weeks | ≥1 day | 23 (21%) | 24 (20%) | 26 (27%) | |
| ≥2–3 days | 7 (6%) | 5 (4%) | 10 (10%) | ||
| 13 weeks | ≥1 day | 14 (13%) | 19 (16%) | 17 (18%) | |
| ≥2–3 days | 2 (2%) | 5 (4%) | 5 (5%) | ||
| 17 weeks | ≥1 day | 10 (9%) | 17 (15%) | 16 (17%) | |
| ≥2–3 days | 3 (3%) | 5 (5%) | 3 (3%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Picaud, J.-C.; Pajek, B.; Arciszewska, M.; Tarczón, I.; Escribano, J.; Porcel, R.; Adelt, T.; Hassink, E.; Rijnierse, A.; Abrahamse-Berkeveld, M.; et al. An Infant Formula with Partially Hydrolyzed Whey Protein Supports Adequate Growth and Is Safe and Well-Tolerated in Healthy, Term Infants: A Randomized, Double-Blind, Equivalence Trial. Nutrients 2020, 12, 2072. https://doi.org/10.3390/nu12072072
Picaud J-C, Pajek B, Arciszewska M, Tarczón I, Escribano J, Porcel R, Adelt T, Hassink E, Rijnierse A, Abrahamse-Berkeveld M, et al. An Infant Formula with Partially Hydrolyzed Whey Protein Supports Adequate Growth and Is Safe and Well-Tolerated in Healthy, Term Infants: A Randomized, Double-Blind, Equivalence Trial. Nutrients. 2020; 12(7):2072. https://doi.org/10.3390/nu12072072
Chicago/Turabian StylePicaud, Jean-Charles, Barbara Pajek, Malgorzata Arciszewska, Izabela Tarczón, Joaquin Escribano, Rocio Porcel, Thomas Adelt, Elly Hassink, Anneke Rijnierse, Marieke Abrahamse-Berkeveld, and et al. 2020. "An Infant Formula with Partially Hydrolyzed Whey Protein Supports Adequate Growth and Is Safe and Well-Tolerated in Healthy, Term Infants: A Randomized, Double-Blind, Equivalence Trial" Nutrients 12, no. 7: 2072. https://doi.org/10.3390/nu12072072
APA StylePicaud, J.-C., Pajek, B., Arciszewska, M., Tarczón, I., Escribano, J., Porcel, R., Adelt, T., Hassink, E., Rijnierse, A., Abrahamse-Berkeveld, M., Korczowski, B., & on behalf of the TENUTO Study Group. (2020). An Infant Formula with Partially Hydrolyzed Whey Protein Supports Adequate Growth and Is Safe and Well-Tolerated in Healthy, Term Infants: A Randomized, Double-Blind, Equivalence Trial. Nutrients, 12(7), 2072. https://doi.org/10.3390/nu12072072





