Comparison of Two Dietary Supplements for Treatment of Uric Acid Renal Lithiasis: Citrate vs. Citrate + Theobromine
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
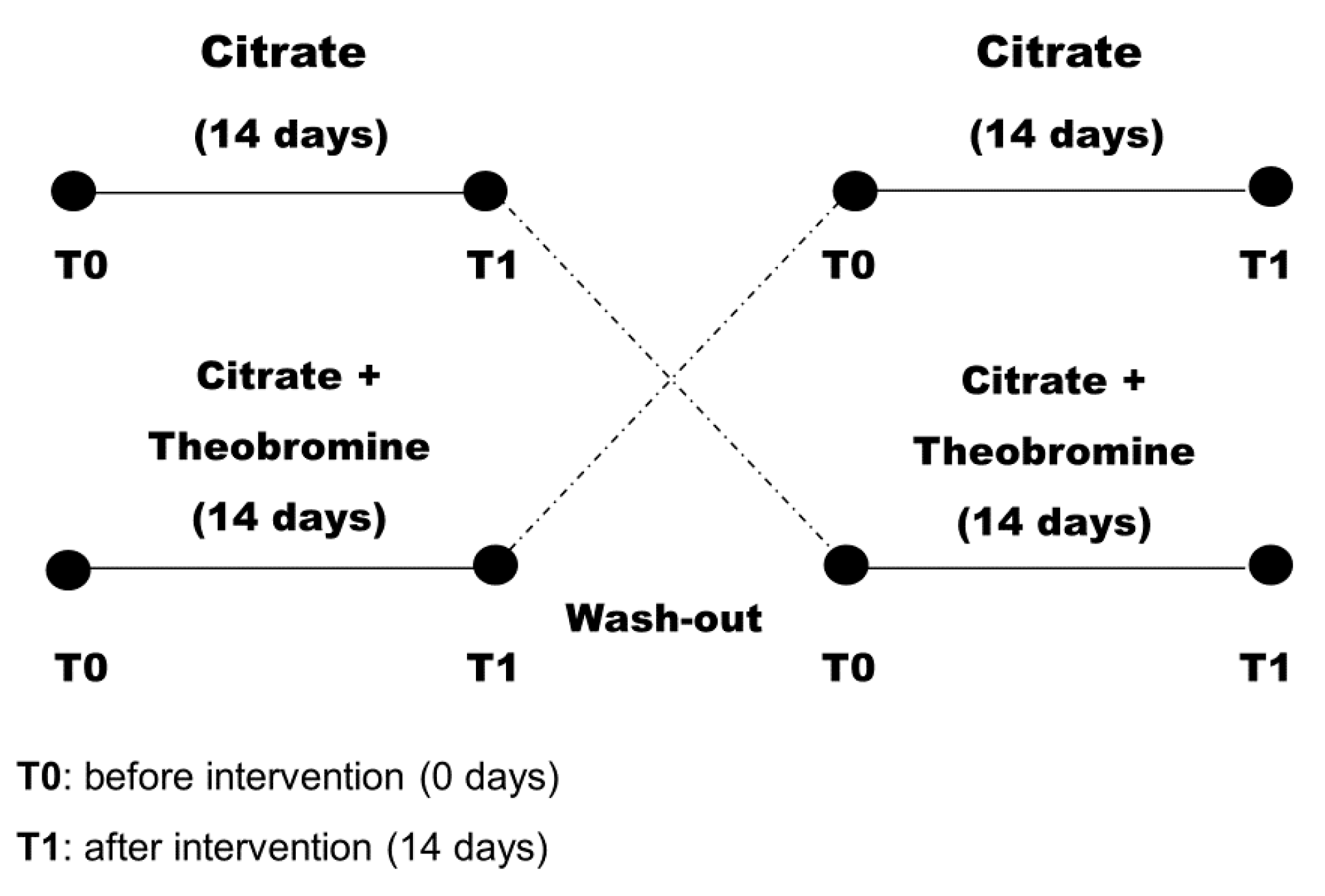
2.2. Treatments
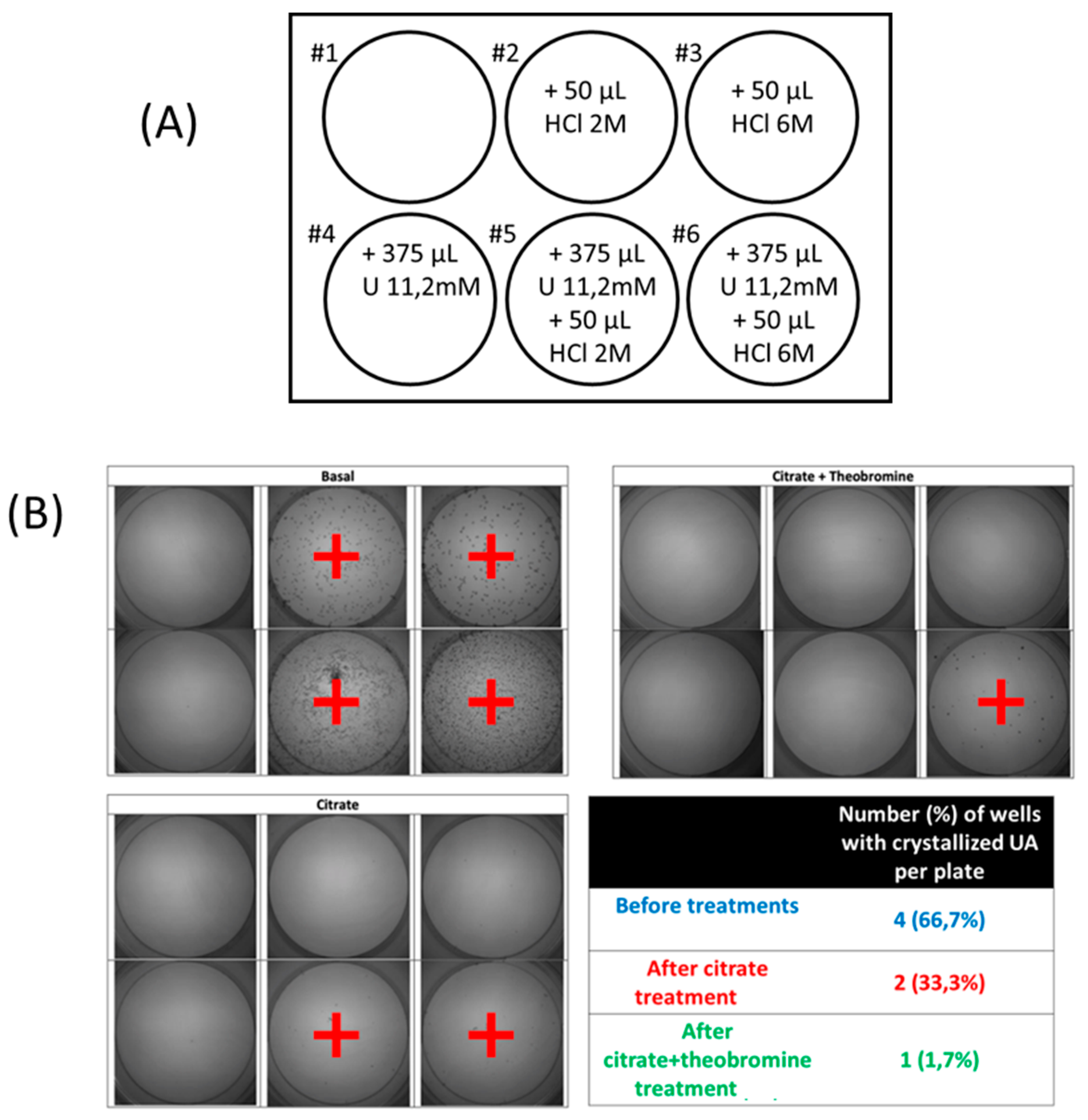
2.3. Outcome Parameters
2.4. Statistical Analysis
2.5. Ethical Considerations
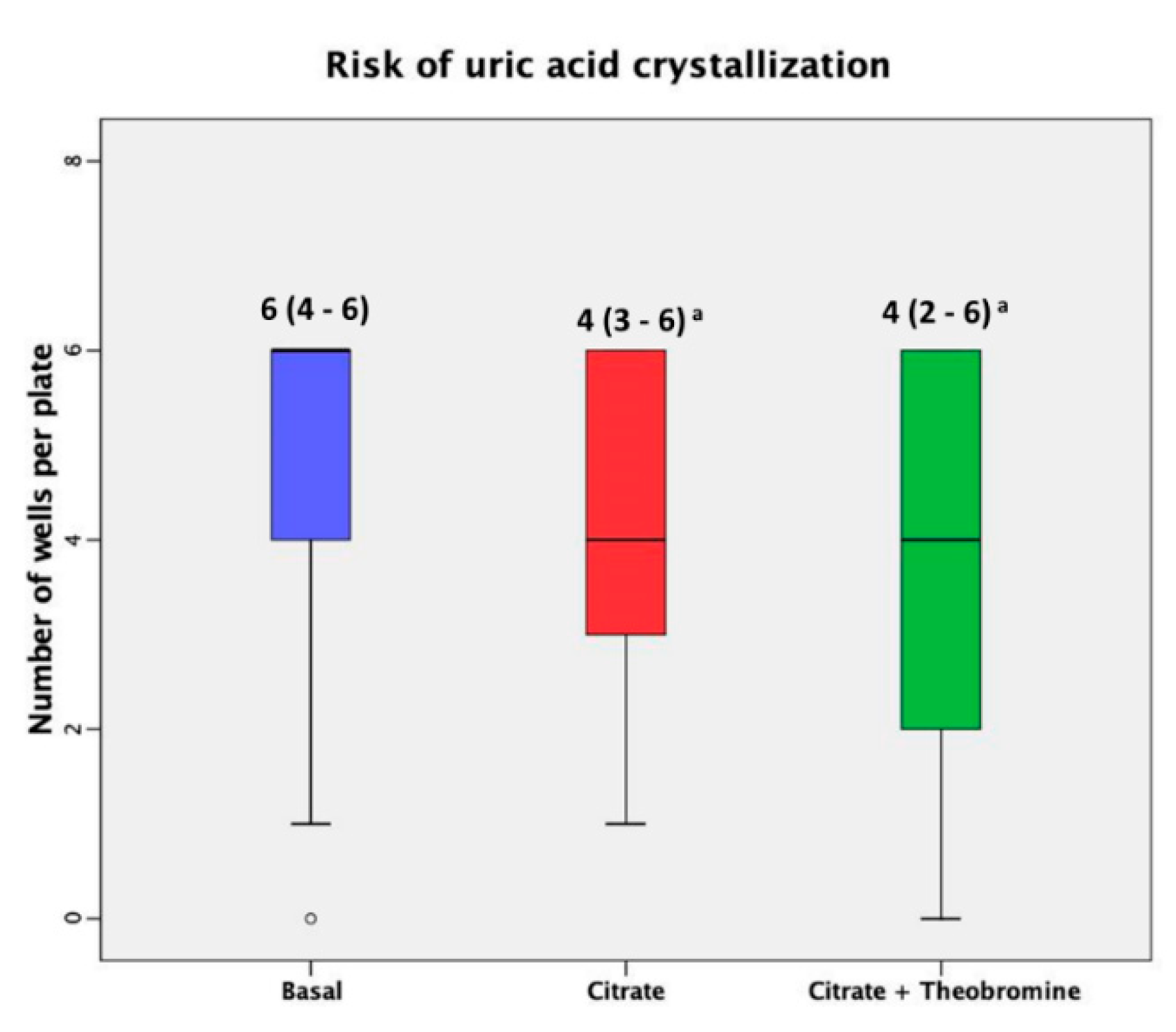
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Daudon, M.; Jungers, P.; Bazin, D.; Williams, J.C. Recurrence rates of urinary calculi according to stone composition and morphology. Urolithiasis 2018, 46, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Canales, B.K.; Sharma, N.; Yuzhakov, S.V.; Bozorgmehri, S.; Otto, B.J.; Bird, V.G. Long-term Recurrence Rates in Uric Acid Stone Formers With or Without Medical Management. Urology 2019, 131, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Gentle, D.L.; Stoller, M.L.; Bruce, J.E.; Leslie, S.W. Geriatric urolithiasis. J. Urol. 1997, 158, 2221–2224. [Google Scholar] [CrossRef]
- Costa-Bauzá, A.; Ramis, M.; Montesinos, V.; Grases, F.; Conte, A.; Pizá, P.; Pieras, E. Type of renal calculi: Variation with age and sex. World J. Urol. 2007, 25, 415–421. [Google Scholar] [CrossRef]
- Trinchieri, A.; Montanari, E. Prevalence of renal uric acid stones in the adult. Urolithiasis 2017, 45, 553–562. [Google Scholar] [CrossRef]
- Cicerello, E. Uric acid nephrolithiasis: An update. Urologia 2018, 85, 93–98. [Google Scholar] [CrossRef]
- Bell, D.S.H. Beware the low urine pH-the major cause of the increased prevalence of nephrolithiasis in the patient with type 2 diabetes. Diabetes Obes. Metab. 2012, 14, 299–303. [Google Scholar] [CrossRef]
- Wagner, C.A.; Mohebbi, N. Urinary pH and stone formation. J. Nephrol. 2010, 23 (Suppl. 1), S165–S169. [Google Scholar]
- Ngo, T.C.; Assimos, D.G. Uric Acid nephrolithiasis: Recent progress and future directions. Rev. Urol. 2007, 9, 17–27. [Google Scholar]
- Grases, F.; Costa-Bauzá, A.; Gomila, I.; Ramis, M.; García-Raja, A.; Prieto, R.M. Urinary pH and renal lithiasis. Urol. Res. 2012, 40, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Hess, B.; Jaeger, P. Physiopathology, etiology and medical treatment of non-calcium lithiasis. Rev. Prat. 1991, 41, 2037–2042. [Google Scholar]
- Heilberg, I.P. Treatment of patients with uric acid stones. Urolithiasis 2016, 44, 57–63. [Google Scholar] [CrossRef]
- Grases, F.; Rodriguez, A.; Costa-Bauza, A. Theobromine inhibits uric acid crystallization. A potential application in the treatment of uric acid nephrolithiasis. PLoS ONE 2014, 9, e111184. [Google Scholar] [CrossRef] [PubMed]
- Costa-Bauza, A.; Grases, F.; Calvó, P.; Rodriguez, A.; Prieto, R. Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers. Nutrients 2018, 10, 1516. [Google Scholar] [CrossRef]
- Craig, W.J.; Nguyen, T.T. Caffeine and Theobromine Levels in Cocoa and Carob Products. J. Food Sci. 2006, 49, 302–303. [Google Scholar] [CrossRef]
- Gates, S.; Miners, J.O. Cytochrome P450 isoform selectivity in human hepatic theobromine metabolism. Br. J. Clin. Pharmacol. 1999, 47, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Rodopoulos, N.; Höjvall, L.; Norman, A. Elimination of theobromine metabolites in healthy adults. Scand. J. Clin. Lab. Investig. 1996, 56, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Shively, C.A.; Tarka, S.M.; Arnaud, M.J.; Dvorchik, B.H.; Passananti, G.T.; Vesell, E.S. High levels of methylxanthines in chocolate do not alter theobromine disposition. Clin. Pharmacol. Ther. 1985, 37, 415–424. [Google Scholar] [CrossRef]
- Rodriguez, A.; Costa-Bauza, A.; Saez-Torres, C.; Rodrigo, D.; Grases, F. HPLC method for urinary theobromine determination: Effect of consumption of cocoa products on theobromine urinary excretion in children. Clin. Biochem. 2015, 48, 1138–1143. [Google Scholar] [CrossRef]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37 (Suppl. 1), S81–S90. [Google Scholar] [CrossRef]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T.; et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef] [PubMed]
- National Cholesterol Education Program (NCEP). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002. [Google Scholar] [CrossRef]
- Grases, F.; Villacampa, A.I.; Costa-Bauzá, A.; Söhnel, O. Uric acid calculi: Types, etiology and mechanisms of formation. Clin. Chim. Acta 2000, 302, 89–104. [Google Scholar] [CrossRef]
- Tonannavar, J.; Deshpande, G.; Yenagi, J.; Patil, S.B.; Patil, N.A.; Mulimani, B.G. Identification of mineral compositions in some renal calculi by FT Raman and IR spectral analysis. Spectrochim. Acta—Part A Mol. Biomol. Spectrosc. 2016. [Google Scholar] [CrossRef] [PubMed]
| Baseline Characteristics of Patients (n = 47) | ||
|---|---|---|
| Gender (male) | 42 | (89.4%) |
| Age (years) | 60.4 | ±10.1 |
| BMI (kg/m2) | 30.2 | ±4.7 |
| Systolic blood pressure (mmHg) | 143 | ±18 |
| Diastolic blood pressure (mmHg) | 87 | ±10 |
| Type of calculi | ||
| UA | 34 | (72.3%) |
| UA/COM | 12 | (25.5%) |
| UA/AU | 1 | (2.1%) |
| Type 2 diabetes mellitus | 10 | (21.3%) |
| Hypertension | 22 | (46.8%) |
| Dyslipidemia | 16 | (34.0%) |
| BMI categories | ||
| 25–30 kg/m2 | 18 | (38.3%) |
| 30–35 kg/m2 | 14 | (29.8%) |
| >35 kg/m2 | 7 | (14.9%) |
| Parameter | Baseline | After Citrate Treatment | After Citrate + Theobromine | InterG p-Value | ||
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | IntraG p-Value | Mean ± SD | IntraG p-Value | ||
| Volume (L) | 0.078 ± 0.023 | 0.083 ± 0.044 | 0.674 | 0.08 ± 0.03 | 0.767 | 0.311 |
| pH | 5.45 ± 0.47 | 5.45 ± 0.44 | 0.879 | 5.49 ± 0.46 | 0.620 | 0.672 |
| Creatinine (mmol/L) | 9.8 ± 3.3 | 9.5 ± 3.9 | 0.388 | 9.9 ± 3.6 | 0.532 | 0.514 |
| Uric acid (mmol/L) | 2.8 ± 1.1 | 2.7 ± 1.3 | 0.536 | 2.9 ± 1.4 | 0.400 | 0.460 |
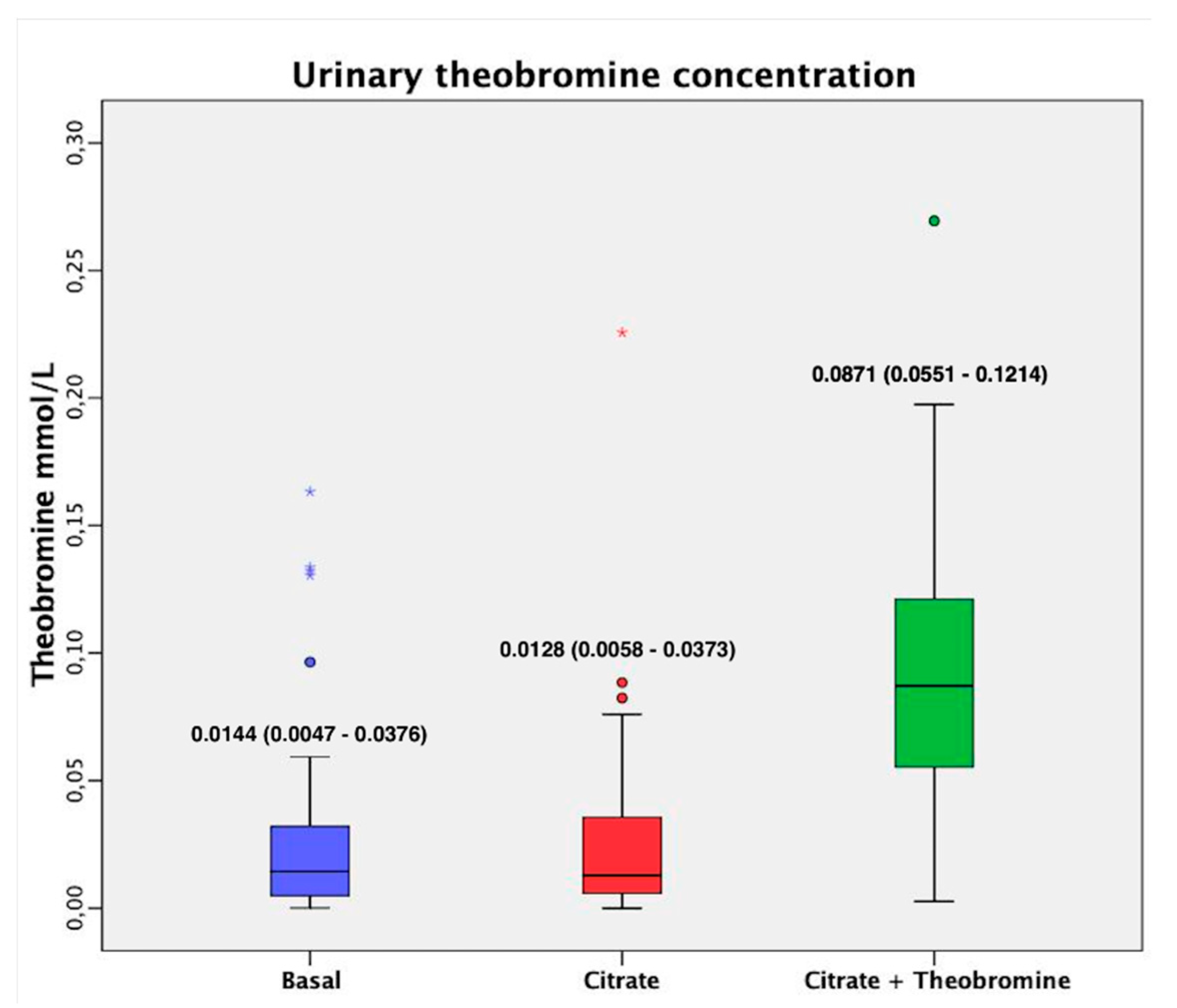
| Theobromine (mmol/L) | 0.029 ± 0.039 | 0.036 ± 0.070 | 0.604 | 0.102 ± 0.086 | <0.001 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernandez, Y.; Costa-Bauza, A.; Calvó, P.; Benejam, J.; Sanchis, P.; Grases, F. Comparison of Two Dietary Supplements for Treatment of Uric Acid Renal Lithiasis: Citrate vs. Citrate + Theobromine. Nutrients 2020, 12, 2012. https://doi.org/10.3390/nu12072012
Hernandez Y, Costa-Bauza A, Calvó P, Benejam J, Sanchis P, Grases F. Comparison of Two Dietary Supplements for Treatment of Uric Acid Renal Lithiasis: Citrate vs. Citrate + Theobromine. Nutrients. 2020; 12(7):2012. https://doi.org/10.3390/nu12072012
Chicago/Turabian StyleHernandez, Yumaira, Antonia Costa-Bauza, Paula Calvó, Joan Benejam, Pilar Sanchis, and Felix Grases. 2020. "Comparison of Two Dietary Supplements for Treatment of Uric Acid Renal Lithiasis: Citrate vs. Citrate + Theobromine" Nutrients 12, no. 7: 2012. https://doi.org/10.3390/nu12072012
APA StyleHernandez, Y., Costa-Bauza, A., Calvó, P., Benejam, J., Sanchis, P., & Grases, F. (2020). Comparison of Two Dietary Supplements for Treatment of Uric Acid Renal Lithiasis: Citrate vs. Citrate + Theobromine. Nutrients, 12(7), 2012. https://doi.org/10.3390/nu12072012





