Mitochondrial Dysfunction as a Novel Target for Neuroprotective Nutraceuticals in Ocular Diseases
Abstract
1. Introduction
1.1. The High-Energy Demands of the Retina
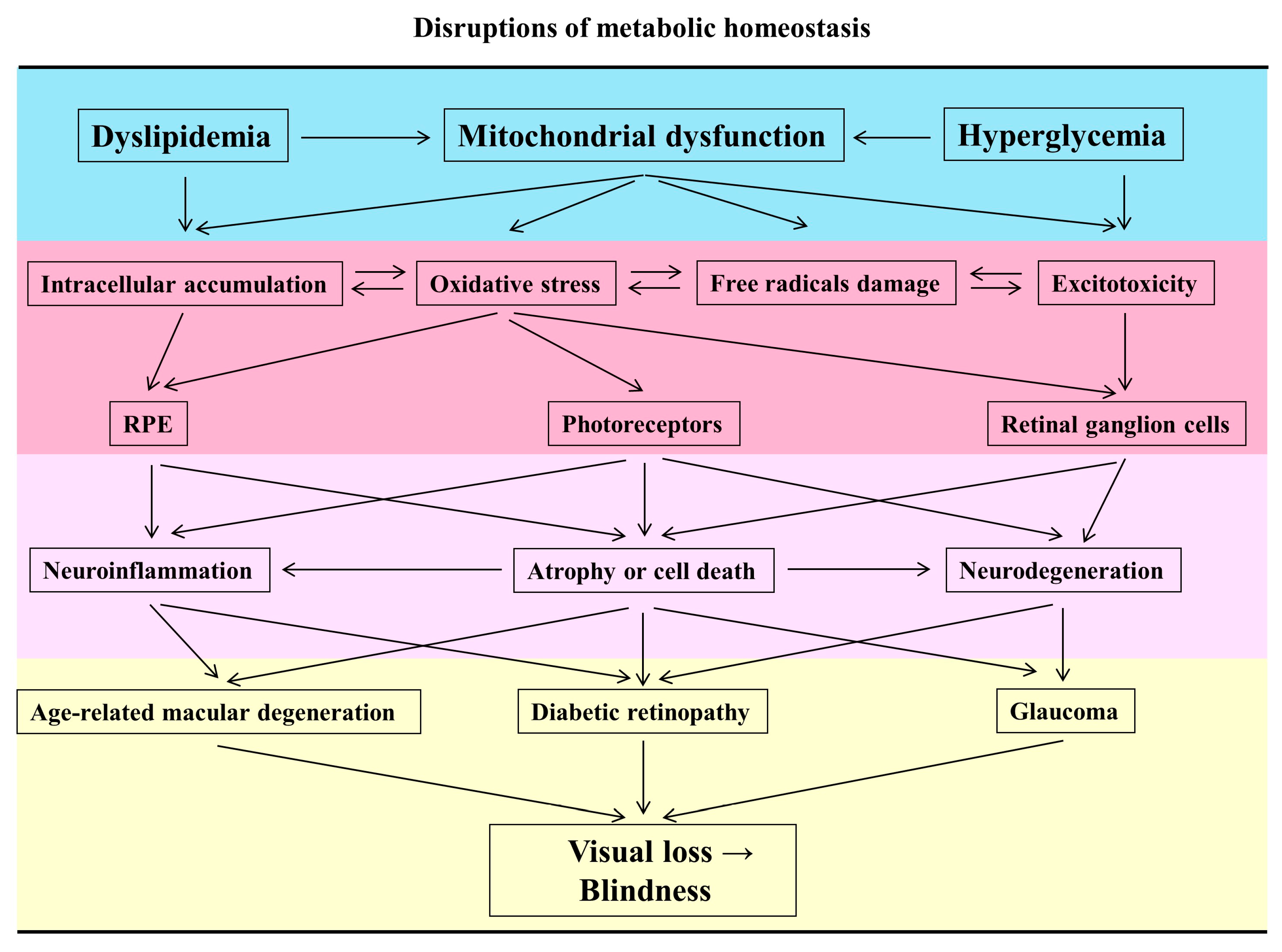
1.2. Disruptions of the Metabolic Homeostasis Cause Mitochondrial Dysfunction in the Retina
1.3. The Characteristics of Mitochondrial Dysfunction in Retinal Neurodegeneration
1.4. Nutraceuticals as Neuroprotectants for Retinal Neurodegeneration
2. Mitochondrial Dysfunction in the Pathogenesis of Ocular Diseases
2.1. Glaucoma
2.2. Age-Related Macular Degeneration
2.3. Diabetic Retinopathy
3. Neuroprotective Effects of Nutraceuticals in Animal Studies and Clinical Trails
3.1. Resveratrol
3.2. Quercetin
3.3. Xanthophylls (Lutein and Zeaxanthin)
3.4. Omega-3 Fatty Acid
3.5. Curcumin
3.6. Crocetin
3.7. Other Potential Nutraceuticals
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schmidt, K.G.; Bergert, H.; Funk, R.H. Neurodegenerative diseases of the retina and potential for protection and recovery. Curr. Neuropharmacol. 2008, 6, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Wong-Riley, M.T. Energy metabolism of the visual system. Eye Brain 2010, 2, 99–116. [Google Scholar] [CrossRef]
- Seung, H.S.; Sumbul, U. Neuronal cell types and connectivity: Lessons from the retina. Neuron 2014, 83, 1262–1272. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.A.; Di Polo, A. Mitochondrial dynamics, transport, and quality control: A bottleneck for retinal ganglion cell viability in optic neuropathies. Mitochondrion 2017, 36, 186–192. [Google Scholar] [CrossRef]
- Roy, S.; Kim, D.; Sankaramoorthy, A. Mitochondrial structural changes in the pathogenesis of diabetic retinopathy. J. Clin. Med. 2019, 8, 1363. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A. Mitochondrial stability in diabetic retinopathy: Lessons learned from epigenetics. Diabetes 2019, 68, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Federico, A.; Cardaioli, E.; Da Pozzo, P.; Formichi, P.; Gallus, G.N.; Radi, E. Mitochondria, oxidative stress and neurodegeneration. J. Neurol. Sci. 2012, 322, 254–262. [Google Scholar] [CrossRef]
- Wang, Y.; Branicky, R.; Noe, A.; Hekimi, S. Superoxide dismutases: Dual roles in controlling ros damage and regulating ros signaling. J. Cell Biol. 2018, 217, 1915–1928. [Google Scholar] [CrossRef]
- Raha, S.; Robinson, B.H. Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem. Sci. 2000, 25, 502–508. [Google Scholar] [CrossRef]
- Schinder, A.F.; Olson, E.C.; Spitzer, N.C.; Montal, M. Mitochondrial dysfunction is a primary event in glutamate neurotoxicity. J. Neurosci. 1996, 16, 6125–6133. [Google Scholar] [CrossRef]
- Nguyen, D.; Alavi, M.V.; Kim, K.Y.; Kang, T.; Scott, R.T.; Noh, Y.H.; Lindsey, J.D.; Wissinger, B.; Ellisman, M.H.; Weinreb, R.N.; et al. A new vicious cycle involving glutamate excitotoxicity, oxidative stress and mitochondrial dynamics. Cell Death Dis. 2011, 2, e240. [Google Scholar] [CrossRef]
- Rego, A.C.; Oliveira, C.R. Mitochondrial dysfunction and reactive oxygen species in excitotoxicity and apoptosis: Implications for the pathogenesis of neurodegenerative diseases. Neurochem. Res. 2003, 28, 1563–1574. [Google Scholar] [CrossRef]
- Abramov, A.Y.; Scorziello, A.; Duchen, M.R. Three distinct mechanisms generate oxygen free radicals in neurons and contribute to cell death during anoxia and reoxygenation. J. Neurosci. 2007, 27, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, E.M.; Emri, E.; Merle, B.M.J.; Colijn, J.M.; Kersten, E.; Cougnard-Gregoire, A.; Dammeier, S.; Meester-Smoor, M.; Pool, F.M.; De Jong, E.K.; et al. A new perspective on lipid research in age-related macular degeneration. Prog. Retin. Eye Res. 2018, 67, 56–86. [Google Scholar] [CrossRef] [PubMed]
- Pilz, Y.L.; Bass, S.J.; Sherman, J. A review of mitochondrial optic neuropathies: From inherited to acquired forms. J. Optom. 2017, 10, 205–214. [Google Scholar] [CrossRef]
- Chrysostomou, V.; Rezania, F.; Trounce, I.A.; Crowston, J.G. Oxidative stress and mitochondrial dysfunction in glaucoma. Curr. Opin. Pharmacol. 2013, 13, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Barot, M.; Gokulgandhi, M.R.; Mitra, A.K. Mitochondrial dysfunction in retinal diseases. Curr. Eye Res. 2011, 36, 1069–1077. [Google Scholar] [CrossRef]
- Joyal, J.S.; Gantner, M.L.; Smith, L.E.H. Retinal energy demands control vascular supply of the retina in development and disease: The role of neuronal lipid and glucose metabolism. Prog. Retin. Eye Res. 2018, 64, 131–156. [Google Scholar] [CrossRef]
- Wu, S.; Zhou, F.; Zhang, Z.; Xing, D. Mitochondrial oxidative stress causes mitochondrial fragmentation via differential modulation of mitochondrial fission-fusion proteins. FEBS J. 2011, 278, 941–954. [Google Scholar] [CrossRef]
- Willems, P.H.; Rossignol, R.; Dieteren, C.E.; Murphy, M.P.; Koopman, W.J. Redox homeostasis and mitochondrial dynamics. Cell Metab. 2015, 22, 207–218. [Google Scholar] [CrossRef]
- Yu-Wai-Man, P.; Griffiths, P.G.; Chinnery, P.F. Mitochondrial optic neuropathies—disease mechanisms and therapeutic strategies. Prog. Retin. Eye Res. 2011, 30, 81–114. [Google Scholar] [CrossRef] [PubMed]
- Gaens, K.H.; Stehouwer, C.D.; Schalkwijk, C.G. Advanced glycation endproducts and its receptor for advanced glycation endproducts in obesity. Curr. Opin. Lipidol. 2013, 24, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Geraldes, P.; King, G.L. Activation of protein kinase c isoforms and its impact on diabetic complications. Circ. Res. 2010, 106, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Bristow, E.A.; Griffiths, P.G.; Andrews, R.M.; Johnson, M.A.; Turnbull, D.M. The distribution of mitochondrial activity in relation to optic nerve structure. Arch. Ophthalmol. 2002, 120, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.; van Driel, D.; Valter, K.; Rees, S.; Provis, J. The locations of mitochondria in mammalian photoreceptors: Relation to retinal vasculature. Brain Res. 2008, 1189, 58–69. [Google Scholar] [CrossRef]
- Kanow, M.A.; Giarmarco, M.M.; Jankowski, C.S.; Tsantilas, K.; Engel, A.L.; Du, J.; Linton, J.D.; Farnsworth, C.C.; Sloat, S.R.; Rountree, A.; et al. Biochemical adaptations of the retina and retinal pigment epithelium support a metabolic ecosystem in the vertebrate eye. Elife 2017, 6, e28899. [Google Scholar] [CrossRef]
- Brown, E.E.; DeWeerd, A.J.; Ildefonso, C.J.; Lewin, A.S.; Ash, J.D. Mitochondrial oxidative stress in the retinal pigment epithelium (rpe) led to metabolic dysfunction in both the rpe and retinal photoreceptors. Redox Biol. 2019, 24, 101201. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.Y.; Cringle, S.J. Oxygen distribution and consumption within the retina in vascularised and avascular retinas and in animal models of retinal disease. Prog. Retin. Eye Res. 2001, 20, 175–208. [Google Scholar] [CrossRef]
- Kern, T.S.; Berkowitz, B.A. Photoreceptors in diabetic retinopathy. J. Diabetes Investig. 2015, 6, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Silva, K.C.; Rosales, M.A.; Biswas, S.K.; Lopes de Faria, J.B.; Lopes de Faria, J.M. Diabetic retinal neurodegeneration is associated with mitochondrial oxidative stress and is improved by an angiotensin receptor blocker in a model combining hypertension and diabetes. Diabetes 2009, 58, 1382–1390. [Google Scholar] [CrossRef]
- Kong, G.Y.; Van Bergen, N.J.; Trounce, I.A.; Crowston, J.G. Mitochondrial dysfunction and glaucoma. J. Glaucoma 2009, 18, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Dadhania, V.P.; Trivedi, P.P.; Vikram, A.; Tripathi, D.N. Nutraceuticals against neurodegeneration: A mechanistic insight. Curr. Neuropharmacol. 2016, 14, 627–640. [Google Scholar] [CrossRef] [PubMed]
- Kelsey, N.A.; Wilkins, H.M.; Linseman, D.A. Nutraceutical antioxidants as novel neuroprotective agents. Molecules 2010, 15, 7792–7814. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The pathophysiology and treatment of glaucoma: A review. Jama 2014, 311, 1901–1911. [Google Scholar] [CrossRef] [PubMed]
- Sacca, S.C.; Izzotti, A. Oxidative stress and glaucoma: Injury in the anterior segment of the eye. Prog. Brain Res. 2008, 173, 385–407. [Google Scholar]
- Ju, W.K.; Kim, K.Y.; Lindsey, J.D.; Angert, M.; Duong-Polk, K.X.; Scott, R.T.; Kim, J.J.; Kukhmazov, I.; Ellisman, M.H.; Perkins, G.A.; et al. Intraocular pressure elevation induces mitochondrial fission and triggers opa1 release in glaucomatous optic nerve. Investig. Ophthalmol. Vis. Sci. 2008, 49, 4903–4911. [Google Scholar] [CrossRef]
- Chen, Y.; Hughes, G.; Chen, X.; Qian, S.; Cao, W.; Wang, L.; Wang, M.; Sun, X. Genetic variants associated with different risks for high tension glaucoma and normal tension glaucoma in a chinese population. Investig. Ophthalmol. Vis. Sci. 2015, 56, 2595–2600. [Google Scholar] [CrossRef]
- Marek, B.; Harris, A.; Kanakamedala, P.; Lee, E.; Amireskandari, A.; Carichino, L.; Guidoboni, G.; Tobe, L.A.; Siesky, B. Cerebrospinal fluid pressure and glaucoma: Regulation of trans-lamina cribrosa pressure. Br. J. Ophthalmol. 2014, 98, 721–725. [Google Scholar] [CrossRef]
- Pinazo-Duran, M.D.; Shoaie-Nia, K.; Zanon-Moreno, V.; Sanz-Gonzalez, S.M.; Del Castillo, J.B.; Garcia-Medina, J.J. Strategies to reduce oxidative stress in glaucoma patients. Curr. Neuropharmacol. 2018, 16, 903–918. [Google Scholar] [CrossRef]
- Tezel, G. Oxidative stress in glaucomatous neurodegeneration: Mechanisms and consequences. Prog. Retin. Eye Res. 2006, 25, 490–513. [Google Scholar] [CrossRef]
- Mitchell, P.; Liew, G.; Gopinath, B.; Wong, T.Y. Age-related macular degeneration. Lancet 2018, 392, 1147–1159. [Google Scholar] [CrossRef]
- Donoso, L.A.; Vrabec, T.; Kuivaniemi, H. The role of complement factor h in age-related macular degeneration: A review. Surv. Ophthalmol. 2010, 55, 227–246. [Google Scholar] [CrossRef]
- Wu, Z.; Lauer, T.W.; Sick, A.; Hackett, S.F.; Campochiaro, P.A. Oxidative stress modulates complement factor h expression in retinal pigmented epithelial cells by acetylation of foxo3. J. Biol. Chem. 2007, 282, 22414–22425. [Google Scholar] [CrossRef]
- Thurman, J.M.; Renner, B.; Kunchithapautham, K.; Ferreira, V.P.; Pangburn, M.K.; Ablonczy, Z.; Tomlinson, S.; Holers, V.M.; Rohrer, B. Oxidative stress renders retinal pigment epithelial cells susceptible to complement-mediated injury. J. Biol. Chem. 2009, 284, 16939–16947. [Google Scholar] [CrossRef] [PubMed]
- Weismann, D.; Hartvigsen, K.; Lauer, N.; Bennett, K.L.; Scholl, H.P.; Charbel Issa, P.; Cano, M.; Brandstatter, H.; Tsimikas, S.; Skerka, C.; et al. Complement factor h binds malondialdehyde epitopes and protects from oxidative stress. Nature 2011, 478, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.; Wang, J.J.; Smith, W.; Leeder, S.R. Smoking and the 5-year incidence of age-related maculopathy: The blue mountains eye study. Arch. Ophthalmol. 2002, 120, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Kunchithapautham, K.; Atkinson, C.; Rohrer, B. Smoke exposure causes endoplasmic reticulum stress and lipid accumulation in retinal pigment epithelium through oxidative stress and complement activation. J. Biol. Chem. 2014, 289, 14534–14546. [Google Scholar] [CrossRef]
- Feher, J.; Kovacs, I.; Artico, M.; Cavallotti, C.; Papale, A.; Balacco Gabrieli, C. Mitochondrial alterations of retinal pigment epithelium in age-related macular degeneration. Neurobiol. Aging 2006, 27, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Terluk, M.R.; Kapphahn, R.J.; Soukup, L.M.; Gong, H.; Gallardo, C.; Montezuma, S.R.; Ferrington, D.A. Investigating mitochondria as a target for treating age-related macular degeneration. J. Neurosci. 2015, 35, 7304–7311. [Google Scholar] [CrossRef] [PubMed]
- Kaarniranta, K.; Pawlowska, E.; Szczepanska, J.; Jablkowska, A.; Blasiak, J. Role of mitochondrial DNA damage in ros-mediated pathogenesis of age-related macular degeneration (amd). Int. J. Mol. Sci. 2019, 20, 2374. [Google Scholar] [CrossRef] [PubMed]
- Collaboration, N.C.D.R.F. Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 2016, 387, 1513–1530. [Google Scholar]
- Mohamed, Q.; Gillies, M.C.; Wong, T.Y. Management of diabetic retinopathy: A systematic review. JAMA 2007, 298, 902–916. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; Chew, E.; Duh, E.J.; Sobrin, L.; Sun, J.K.; VanderBeek, B.L.; Wykoff, C.C.; Gardner, T.W. Diabetic retinopathy: A position statement by the american diabetes association. Diabetes Care 2017, 40, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Simo, R.; Stitt, A.W.; Gardner, T.W. Neurodegeneration in diabetic retinopathy: Does it really matter? Diabetologia 2018, 61, 1902–1912. [Google Scholar] [CrossRef]
- Diabetes, C.; Nathan, D.M.; Genuth, S.; Lachin, J.; Cleary, P.; Crofford, O.; Davis, M.; Rand, L.; Siebert, C. Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 1993, 329, 977–986. [Google Scholar]
- Nathan, D.M.; Group, D.E.R. The diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: Overview. Diabetes Care 2014, 37, 9–16. [Google Scholar] [CrossRef]
- Carrasco, E.; Hernandez, C.; Miralles, A.; Huguet, P.; Farres, J.; Simo, R. Lower somatostatin expression is an early event in diabetic retinopathy and is associated with retinal neurodegeneration. Diabetes Care 2007, 30, 2902–2908. [Google Scholar] [CrossRef]
- Garcia-Ramirez, M.; Hernandez, C.; Villarroel, M.; Canals, F.; Alonso, M.A.; Fortuny, R.; Masmiquel, L.; Navarro, A.; Garcia-Arumi, J.; Simo, R. Interphotoreceptor retinoid-binding protein (irbp) is downregulated at early stages of diabetic retinopathy. Diabetologia 2009, 52, 2633–2641. [Google Scholar] [CrossRef]
- Barber, A.J.; Lieth, E.; Khin, S.A.; Antonetti, D.A.; Buchanan, A.G.; Gardner, T.W. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J. Clin. Investig. 1998, 102, 783–791. [Google Scholar] [CrossRef]
- Vujosevic, S.; Midena, E. Retinal layers changes in human preclinical and early clinical diabetic retinopathy support early retinal neuronal and muller cells alterations. J. Diabetes Res. 2013, 2013, 905058. [Google Scholar] [CrossRef]
- Sohn, E.H.; Van Dijk, H.W.; Jiao, C.; Kok, P.H.; Jeong, W.; Demirkaya, N.; Garmager, A.; Wit, F.; Kucukevcilioglu, M.; Van Velthoven, M.E.; et al. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc. Natl. Acad. Sci. USA 2016, 113, E2655–E2664. [Google Scholar] [CrossRef] [PubMed]
- Wanek, J.; Blair, N.P.; Chau, F.Y.; Lim, J.I.; Leiderman, Y.I.; Shahidi, M. Alterations in retinal layer thickness and reflectance at different stages of diabetic retinopathy by en face optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 2016, 57, OCT341–OCT347. [Google Scholar] [CrossRef]
- Chen, S.Y.; Hsu, Y.M.; Lin, Y.J.; Huang, Y.C.; Chen, C.J.; Lin, W.D.; Liao, W.L.; Chen, Y.T.; Lin, W.Y.; Liu, Y.H.; et al. Current concepts regarding developmental mechanisms in diabetic retinopathy in Taiwan. BioMedicine 2016, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Bek, T. Mitochondrial dysfunction and diabetic retinopathy. Mitochondrion 2017, 36, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Mishra, M. Therapeutic targets for altering mitochondrial dysfunction associated with diabetic retinopathy. Expert. Opin. Ther. Targets 2018, 22, 233–245. [Google Scholar] [CrossRef]
- Durham, J.T.; Herman, I.M. Microvascular modifications in diabetic retinopathy. Curr. Diabetes Rep. 2011, 11, 253–264. [Google Scholar] [CrossRef]
- Simo, R.; Hernandez, C. European Consortium for the Early Treatment of Diabetic, R. Neurodegeneration in the diabetic eye: New insights and therapeutic perspectives. Trends Endocrinol. Metab. 2014, 25, 23–33. [Google Scholar] [CrossRef]
- Lekli, I.; Ray, D.; Das, D.K. Longevity nutrients resveratrol, wines and grapes. Genes Nutr. 2010, 5, 55–60. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating sirt1 and pgc-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Abu-Amero, K.K.; Kondkar, A.A.; Chalam, K.V. Resveratrol and ophthalmic diseases. Nutrients 2016, 8, 200. [Google Scholar] [CrossRef]
- Pirhan, D.; Yuksel, N.; Emre, E.; Cengiz, A.; Kursat Yildiz, D. Riluzole-and resveratrol-induced delay of retinal ganglion cell death in an experimental model of glaucoma. Curr. Eye Res. 2016, 41, 59–69. [Google Scholar] [CrossRef]
- Lindsey, J.D.; Duong-Polk, K.X.; Hammond, D.; Leung, C.K.; Weinreb, R.N. Protection of injured retinal ganglion cell dendrites and unfolded protein response resolution after long-term dietary resveratrol. Neurobiol. Aging 2015, 36, 1969–1981. [Google Scholar] [CrossRef] [PubMed]
- Razali, N.; Agarwal, R.; Agarwal, P.; Kumar, S.; Tripathy, M.; Vasudevan, S.; Crowston, J.G.; Ismail, N.M. Role of adenosine receptors in resveratrol-induced intraocular pressure lowering in rats with steroid-induced ocular hypertension. Clin. Exp. Ophthalmol. 2015, 43, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Natesan, S.; Pandian, S.; Ponnusamy, C.; Palanichamy, R.; Muthusamy, S.; Kandasamy, R. Co-encapsulated resveratrol and quercetin in chitosan and peg modified chitosan nanoparticles: For efficient intra ocular pressure reduction. Int. J. Biol. Macromol. 2017, 104, 1837–1845. [Google Scholar] [CrossRef]
- Kubota, S.; Kurihara, T.; Ebinuma, M.; Kubota, M.; Yuki, K.; Sasaki, M.; Noda, K.; Ozawa, Y.; Oike, Y.; Ishida, S.; et al. Resveratrol prevents light-induced retinal degeneration via suppressing activator protein-1 activation. Am. J. Pathol. 2010, 177, 1725–1731. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wu, Z.; Li, J.; Marmalidou, A.; Zhang, R.; Yu, M. Protective effect of resveratrol against light-induced retinal degeneration in aged samp8 mice. Oncotarget 2017, 8, 65778–65788. [Google Scholar] [CrossRef] [PubMed]
- Soufi, F.G.; Mohammad-Nejad, D.; Ahmadieh, H. Resveratrol improves diabetic retinopathy possibly through oxidative stress - nuclear factor kappab - apoptosis pathway. Pharmacol. Rep. 2012, 64, 1505–1514. [Google Scholar] [CrossRef]
- Sedlak, L.; Wojnar, W.; Zych, M.; Wygledowska-Promienska, D.; Mrukwa-Kominek, E.; Kaczmarczyk-Sedlak, I. Effect of resveratrol, a dietary-derived polyphenol, on the oxidative stress and polyol pathway in the lens of rats with streptozotocin-induced diabetes. Nutrients 2018, 10, 1423. [Google Scholar] [CrossRef]
- Chen, Y.; Meng, J.; Li, H.; Wei, H.; Bi, F.; Liu, S.; Tang, K.; Guo, H.; Liu, W. Resveratrol exhibits an effect on attenuating retina inflammatory condition and damage of diabetic retinopathy via pon1. Exp. Eye Res. 2019, 181, 356–366. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, Y.S.; Roh, G.S.; Choi, W.S.; Cho, G.J. Resveratrol blocks diabetes-induced early vascular lesions and vascular endothelial growth factor induction in mouse retinas. Acta Ophthalmol. 2012, 90, e31–e37. [Google Scholar] [CrossRef]
- Berman, A.Y.; Motechin, R.A.; Wiesenfeld, M.Y.; Holz, M.K. The therapeutic potential of resveratrol: A review of clinical trials. NPJ Precis. Oncol. 2017, 1, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Singh, R.; Verma, S.S.; Rai, V.; Kaschula, C.H.; Maiti, P.; Gupta, S.C. Health benefits of resveratrol: Evidence from clinical studies. Med. Res. Rev. 2019, 39, 1851–1891. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Munoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdu-Queralt, A.; Lamuela-Raventos, R.M. Health effects of resveratrol: Results from human intervention trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef] [PubMed]
- Bola, C.; Bartlett, H.; Eperjesi, F. Resveratrol and the eye: Activity and molecular mechanisms. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albrecht Graefes Arch. Klin. Exp. Ophthalmol. 2014, 252, 699–713. [Google Scholar] [CrossRef]
- Richer, S.; Stiles, W.; Ulanski, L.; Carroll, D.; Podella, C. Observation of human retinal remodeling in octogenarians with a resveratrol based nutritional supplement. Nutrients 2013, 5, 1989–2005. [Google Scholar] [CrossRef]
- Timmers, S.; De Ligt, M.; Phielix, E.; Van de Weijer, T.; Hansen, J.; Moonen-Kornips, E.; Schaart, G.; Kunz, I.; Hesselink, M.K.; Schrauwen-Hinderling, V.B.; et al. Resveratrol as add-on therapy in subjects with well-controlled type 2 diabetes: A randomized controlled trial. Diabetes Care 2016, 39, 2211–2217. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Boots, A.W.; Haenen, G.R.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef]
- Qiu, L.; Luo, Y.; Chen, X. Quercetin attenuates mitochondrial dysfunction and biogenesis via upregulated ampk/sirt1 signaling pathway in oa rats. Biomed. Pharmacother. 2018, 103, 1585–1591. [Google Scholar] [CrossRef]
- Wang, D.M.; Li, S.Q.; Wu, W.L.; Zhu, X.Y.; Wang, Y.; Yuan, H.Y. Effects of long-term treatment with quercetin on cognition and mitochondrial function in a mouse model of alzheimer’s disease. Neurochem. Res. 2014, 39, 1533–1543. [Google Scholar] [CrossRef]
- Nakayama, M.; Aihara, M.; Chen, Y.N.; Araie, M.; Tomita-Yokotani, K.; Iwashina, T. Neuroprotective effects of flavonoids on hypoxia-, glutamate-, and oxidative stress-induced retinal ganglion cell death. Mol. Vis. 2011, 17, 1784–1793. [Google Scholar]
- Maher, P.; Hanneken, A. Flavonoids protect retinal ganglion cells from oxidative stress-induced death. Investig. Ophthalmol. Vis. Sci. 2005, 46, 4796–4803. [Google Scholar] [CrossRef]
- Gao, F.J.; Zhang, S.H.; Xu, P.; Yang, B.Q.; Zhang, R.; Cheng, Y.; Zhou, X.J.; Huang, W.J.; Wang, M.; Chen, J.Y.; et al. Quercetin declines apoptosis, ameliorates mitochondrial function and improves retinal ganglion cell survival and function in in vivo model of glaucoma in rat and retinal ganglion cell culture in vitro. Front. Mol. Neurosci. 2017, 10, 285. [Google Scholar] [CrossRef]
- Zhou, X.; Li, G.; Yang, B.; Wu, J. Quercetin enhances inhibitory synaptic inputs and reduces excitatory synaptic inputs to off- and on-type retinal ganglion cells in a chronic glaucoma rat model. Front. Neurosci. 2019, 13, 672. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Cozier, F.; Ong, O.C.; Caprioli, J. Induction of heat shock protein 72 protects retinal ganglion cells in a rat glaucoma model. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1522–1530. [Google Scholar]
- Caprioli, J.; Ishii, Y.; Kwong, J.M. Retinal ganglion cell protection with geranylgeranylacetone, a heat shock protein inducer, in a rat glaucoma model. Trans. Am. Ophthalmol. Soc. 2003, 101, 39–50. [Google Scholar]
- Li, N.; Li, Y.; Duan, X. Heat shock protein 72 confers protection in retinal ganglion cells and lateral geniculate nucleus neurons via blockade of the sapk/jnk pathway in a chronic ocular-hypertensive rat model. Neural Regen. Res. 2014, 9, 1395–1401. [Google Scholar] [PubMed]
- Kim, J.; Jin, H.L.; Jang, D.S.; Jeong, K.W.; Choung, S.Y. Quercetin-3-o-alpha-l-arabinopyranoside protects against retinal cell death via blue light-induced damage in human rpe cells and balb-c mice. Food Funct. 2018, 9, 2171–2183. [Google Scholar] [CrossRef] [PubMed]
- Koyama, Y.; Kaidzu, S.; Kim, Y.C.; Matsuoka, Y.; Ishihara, T.; Ohira, A.; Tanito, M. Suppression of light-induced retinal degeneration by quercetin via the ap-1 pathway in rats. Antioxidants 2019, 8, 79. [Google Scholar] [CrossRef]
- Kim, J.H.; Kang, M.J.; Choi, H.N.; Jeong, S.M.; Lee, Y.M.; Kim, J.I. Quercetin attenuates fasting and postprandial hyperglycemia in animal models of diabetes mellitus. Nutr. Res. Pract. 2011, 5, 107–111. [Google Scholar] [CrossRef]
- Jeong, S.M.; Kang, M.J.; Choi, H.N.; Kim, J.H.; Kim, J.I. Quercetin ameliorates hyperglycemia and dyslipidemia and improves antioxidant status in type 2 diabetic db/db mice. Nutr. Res. Pract. 2012, 6, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Gupta, S.K.; Nag, T.C.; Srivastava, S.; Saxena, R.; Jha, K.A.; Srinivasan, B.P. Retinal neuroprotective effects of quercetin in streptozotocin-induced diabetic rats. Exp. Eye Res. 2014, 125, 193–202. [Google Scholar] [CrossRef]
- Mohammadi-Sartang, M.; Mazloom, Z.; Sherafatmanesh, S.; Ghorbani, M.; Firoozi, D. Effects of supplementation with quercetin on plasma c-reactive protein concentrations: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2017, 71, 1033–1039. [Google Scholar] [CrossRef]
- Boots, A.W.; Drent, M.; De Boer, V.C.; Bast, A.; Haenen, G.R. Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis. Clin. Nutr. 2011, 30, 506–512. [Google Scholar] [CrossRef]
- Ferry, D.R.; Smith, A.; Malkhandi, J.; Fyfe, D.W.; De Takats, P.G.; Anderson, D.; Baker, J.; Kerr, D.J. Phase i clinical trial of the flavonoid quercetin: Pharmacokinetics and evidence for in vivo tyrosine kinase inhibition. Clin. Cancer Res. 1996, 2, 659–668. [Google Scholar]
- Ramos, S. Cancer chemoprevention and chemotherapy: Dietary polyphenols and signalling pathways. Mol. Nutr. Food Res. 2008, 52, 507–526. [Google Scholar] [CrossRef]
- Kooshyar, M.M.; Mozafari, P.M.; Amirchaghmaghi, M.; Pakfetrat, A.; Karoos, P.; Mohasel, M.R.; Orafai, H.; Azarian, A.A. A randomized placebo- controlled double blind clinical trial of quercetin in the prevention and treatment of chemotherapy-induced oral mucositis. J. Clin. Diagn. Res. 2017, 11, ZC4–ZC50. [Google Scholar] [CrossRef]
- Gopinath, B.; Liew, G.; Kifley, A.; Flood, V.M.; Joachim, N.; Lewis, J.R.; Hodgson, J.M.; Mitchell, P. Dietary flavonoids and the prevalence and 15-y incidence of age-related macular degeneration. Am. J. Clin. Nutr. 2018, 108, 381–387. [Google Scholar] [CrossRef]
- Sommerburg, O.G.; Siems, W.G.; Hurst, J.S.; Lewis, J.W.; Kliger, D.S.; Van Kuijk, F.J. Lutein and zeaxanthin are associated with photoreceptors in the human retina. Curr. Eye Res. 1999, 19, 491–495. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E. Carotenoids: How effective are they to prevent age-related diseases? Molecules 2019, 24, 1801. [Google Scholar] [CrossRef]
- Sasaki, M.; Ozawa, Y.; Kurihara, T.; Noda, K.; Imamura, Y.; Kobayashi, S.; Ishida, S.; Tsubota, K. Neuroprotective effect of an antioxidant, lutein, during retinal inflammation. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Arnal, E.; Miranda, M.; Johnsen-Soriano, S.; Alvarez-Nolting, R.; Diaz-Llopis, M.; Araiz, J.; Cervera, E.; Bosch-Morell, F.; Romero, F.J. Beneficial effect of docosahexanoic acid and lutein on retinal structural, metabolic, and functional abnormalities in diabetic rats. Curr. Eye Res. 2009, 34, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Vachali, P.P.; Gorusupudi, A.; Shen, Z.; Sharifzadeh, H.; Besch, B.M.; Nelson, K.; Horvath, M.M.; Frederick, J.M.; Baehr, W.; et al. Inactivity of human beta,beta-carotene-9′,10′-dioxygenase (bco2) underlies retinal accumulation of the human macular carotenoid pigment. Proc. Natl. Acad. Sci. USA 2014, 111, 10173–10178. [Google Scholar] [CrossRef]
- Widjaja-Adhi, M.A.K.; Ramkumar, S.; Von Lintig, J. Protective role of carotenoids in the visual cycle. FASEB J. 2018, 32, 6305–6315. [Google Scholar] [CrossRef]
- Woo, T.T.; Li, S.Y.; Lai, W.W.; Wong, D.; Lo, A.C. Neuroprotective effects of lutein in a rat model of retinal detachment. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albrecht Graefes Arch. Klin. Exp. Ophthalmol. 2013, 251, 41–51. [Google Scholar] [CrossRef]
- Kamoshita, M.; Toda, E.; Osada, H.; Narimatsu, T.; Kobayashi, S.; Tsubota, K.; Ozawa, Y. Lutein acts via multiple antioxidant pathways in the photo-stressed retina. Sci. Rep. 2016, 6, 30226. [Google Scholar] [CrossRef]
- Yu, M.; Yan, W.; Beight, C. Lutein and zeaxanthin isomers protect against light-induced retinopathy via decreasing oxidative and endoplasmic reticulum stress in balb/cj mice. Nutrients 2018, 10, 842. [Google Scholar] [CrossRef]
- Yu, H.; Wark, L.; Ji, H.; Willard, L.; Jaing, Y.; Han, J.; He, H.; Ortiz, E.; Zhang, Y.; Medeiros, D.M.; et al. Dietary wolfberry upregulates carotenoid metabolic genes and enhances mitochondrial biogenesis in the retina of db/db diabetic mice. Mol. Nutr. Food Res. 2013, 57, 1158–1169. [Google Scholar] [CrossRef]
- Mohn, E.S.; Erdman, J.W., Jr.; Neuringer, M.; Kuchan, M.J.; Johnson, E.J. Brain xanthophyll content and exploratory gene expression analysis: Subspecies differences in rhesus macaque. Genes Nutr. 2017, 12, 9. [Google Scholar] [CrossRef]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef]
- Moeller, S.M.; Parekh, N.; Tinker, L.; Ritenbaugh, C.; Blodi, B.; Wallace, R.B.; Mares, J.A.; Group, C.R.S. Associations between intermediate age-related macular degeneration and lutein and zeaxanthin in the carotenoids in age-related eye disease study (careds): Ancillary study of the women’s health initiative. Arch. Ophthalmol. 2006, 124, 1151–1162. [Google Scholar] [CrossRef]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Lutein and zeaxanthin status and risk of age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2461–2465. [Google Scholar] [CrossRef]
- Ma, L.; Dou, H.L.; Wu, Y.Q.; Huang, Y.M.; Huang, Y.B.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Lutein and zeaxanthin intake and the risk of age-related macular degeneration: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, 350–359. [Google Scholar] [CrossRef]
- Raman, G.; Haslam, D.; Avendano, E.; Johnson, E.J. Lutein/zeaxanthin intake and visual outcomes in adults with healthy eyes: Qualitative gap analysis. Cogent Med. 2019, 6, 1683939. [Google Scholar] [CrossRef]
- Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.; Ferris, F.L.; Elman, M.; Antoszyk, A.; Ruby, A.; Orth, D.; Bressler, S.; et al. Age-Related Eye Disease Study 2 Research, G. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The age-related eye disease study 2 (areds2) randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar]
- Bazan, N.G.; Molina, M.F.; Gordon, W.C. Docosahexaenoic acid signalolipidomics in nutrition: Significance in aging, neuroinflammation, macular degeneration, alzheimer’s, and other neurodegenerative diseases. Annu. Rev. Nutr. 2011, 31, 321–351. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.H.; Fukushima, A.; Sakuma, K.; Kagawa, Y. Chronic docosahexaenoic acid intake enhances expression of the gene for uncoupling protein 3 and affects pleiotropic mrna levels in skeletal muscle of aged c57bl/6njcl mice. J. Nutr. 2001, 131, 2636–2642. [Google Scholar] [CrossRef] [PubMed]
- Flachs, P.; Horakova, O.; Brauner, P.; Rossmeisl, M.; Pecina, P.; Franssen-van Hal, N.; Ruzickova, J.; Sponarova, J.; Drahota, Z.; Vlcek, C.; et al. Polyunsaturated fatty acids of marine origin upregulate mitochondrial biogenesis and induce beta-oxidation in white fat. Diabetologia 2005, 48, 2365–2375. [Google Scholar] [CrossRef]
- Afshordel, S.; Hagl, S.; Werner, D.; Rohner, N.; Kogel, D.; Bazan, N.G.; Eckert, G.P. Omega-3 polyunsaturated fatty acids improve mitochondrial dysfunction in brain aging--impact of bcl-2 and npd-1 like metabolites. Prostaglandins Leukot. Essent. Fat. Acids 2015, 92, 23–31. [Google Scholar] [CrossRef]
- Bazan, N.G.; Birkle, D.L.; Reddy, T.S. Docosahexaenoic acid (22:6, n-3) is metabolized to lipoxygenase reaction products in the retina. Biochem. Biophys. Res. Commun. 1984, 125, 741–747. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Marcheselli, V.L.; De Rivero Vaccari, J.C.; Gordon, W.C.; Jackson, F.E.; Bazan, N.G. Photoreceptor outer segment phagocytosis attenuates oxidative stress-induced apoptosis with concomitant neuroprotectin d1 synthesis. Proc. Natl. Acad. Sci. USA 2007, 104, 13158–13163. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.T.; Bui, B.V.; Sinclair, A.J.; Vingrys, A.J. Dietary omega 3 fatty acids decrease intraocular pressure with age by increasing aqueous outflow. Investig. Ophthalmol. Vis. Sci. 2007, 48, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Schnebelen, C.; Pasquis, B.; Salinas-Navarro, M.; Joffre, C.; Creuzot-Garcher, C.P.; Vidal-Sanz, M.; Bron, A.M.; Bretillon, L.; Acar, N. A dietary combination of omega-3 and omega-6 polyunsaturated fatty acids is more efficient than single supplementations in the prevention of retinal damage induced by elevation of intraocular pressure in rats. Graefe’s Arch. Clin. Exp. Ophthalmol. = Albrecht Graefes Arch. Klin. Exp. Ophthalmol. 2009, 247, 1191–1203. [Google Scholar] [CrossRef] [PubMed]
- Tuo, J.; Ross, R.J.; Herzlich, A.A.; Shen, D.; Ding, X.; Zhou, M.; Coon, S.L.; Hussein, N.; Salem, N., Jr.; Chan, C.C. A high omega-3 fatty acid diet reduces retinal lesions in a murine model of macular degeneration. Am. J. Pathol. 2009, 175, 799–807. [Google Scholar] [CrossRef]
- Sapieha, P.; Chen, J.; Stahl, A.; Seaward, M.R.; Favazza, T.L.; Juan, A.M.; Hatton, C.J.; Joyal, J.S.; Krah, N.M.; Dennison, R.J.; et al. Omega-3 polyunsaturated fatty acids preserve retinal function in type 2 diabetic mice. Nutr. Diabetes 2012, 2, e36. [Google Scholar] [CrossRef]
- Prokopiou, E.; Kolovos, P.; Georgiou, C.; Kalogerou, M.; Potamiti, L.; Sokratous, K.; Kyriacou, K.; Georgiou, T. Omega-3 fatty acids supplementation protects the retina from age-associated degeneration in aged c57bl/6j mice. BMJ Open Ophthalmol. 2019, 4, e000326. [Google Scholar] [CrossRef]
- Sangiovanni, J.P.; Agron, E.; Meleth, A.D.; Reed, G.F.; Sperduto, R.D.; Clemons, T.E.; Chew, E.Y.; Age-Related Eye Disease Study Research Group. {omega}-3 long-chain polyunsaturated fatty acid intake and 12-y incidence of neovascular age-related macular degeneration and central geographic atrophy: Areds report 30, a prospective cohort study from the age-related eye disease study. Am. J. Clin. Nutr. 2009, 90, 1601–1607. [Google Scholar]
- Gerstenblith, A.T.; Baskin, D.E.; Shah, C.P.; Wolfe, J.D.; Fineman, M.S.; Kaiser, R.S.; Ho, A.C. Electroretinographic effects of omega-3 fatty acid supplementation on dry age-related macular degeneration. JAMA Ophthalmol. 2013, 131, 365–369. [Google Scholar] [CrossRef]
- Van Asten, F.; Chiu, C.Y.; Agron, E.; Clemons, T.E.; Ratnapriya, R.; Swaroop, A.; Klein, M.L.; Fan, R.; Chew, E.Y. Age-Related Eye Disease Study 2 Research, G. No cfh or arms2 interaction with omega-3 fatty acids, low versus high zinc, or beta-carotene versus lutein and zeaxanthin on progression of age-related macular degeneration in the age-related eye disease study 2: Age-related eye disease study 2 report no. 18. Ophthalmology 2019, 126, 1541–1548. [Google Scholar]
- Sasaki, M.; Kawasaki, R.; Rogers, S.; Man, R.E.; Itakura, K.; Xie, J.; Flood, V.; Tsubota, K.; Lamoureux, E.; Wang, J.J. The associations of dietary intake of polyunsaturated fatty acids with diabetic retinopathy in well-controlled diabetes. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7473–7479. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Diaz-Lopez, A.; Valls-Pedret, C.; Cofan, M.; Garcia-Layana, A.; Lamuela-Raventos, R.M.; Castaner, O.; Zanon-Moreno, V.; Martinez-Gonzalez, M.A.; Toledo, E.; et al. Dietary marine omega-3 fatty acids and incident sight-threatening retinopathy in middle-aged and older individuals with type 2 diabetes: Prospective investigation from the predimed trial. JAMA Ophthalmol. 2016, 134, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, J.; Granados-Castro, L.F.; Zazueta, C.; Anderica-Romero, A.C.; Chirino, Y.I.; Pedraza-Chaverri, J. Mitochondria as a target in the therapeutic properties of curcumin. Arch. Pharm. 2014, 347, 873–884. [Google Scholar] [CrossRef]
- Lopez-Malo, D.; Villaron-Casares, C.A.; Alarcon-Jimenez, J.; Miranda, M.; Diaz-Llopis, M.; Romero, F.J.; Villar, V.M. Curcumin as a therapeutic option in retinal diseases. Antioxidants 2020, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.M.; Pahlitzsch, M.; Guo, L.; Balendra, S.; Shah, P.; Ravindran, N.; Malaguarnera, G.; Sisa, C.; Shamsher, E.; Hamze, H.; et al. Topical curcumin nanocarriers are neuroprotective in eye disease. Sci. Rep. 2018, 8, 11066. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.H.; Ko, Y.C.; Chang, Y.F.; Huang, S.H.; Liu, C.J. Thermosensitive chitosan-gelatin-based hydrogel containing curcumin-loaded nanoparticles and latanoprost as a dual-drug delivery system for glaucoma treatment. Exp. Eye Res. 2019, 179, 179–187. [Google Scholar] [CrossRef]
- Salehi, B.; Stojanovic-Radic, Z.; Matejic, J.; Sharifi-Rad, M.; Anil Kumar, N.V.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of curcumin: A review of clinical trials. Eur. J. Med. Chem. 2019, 163, 527–545. [Google Scholar] [CrossRef]
- Mazzolani, F.; Togni, S. Oral administration of a curcumin-phospholipid delivery system for the treatment of central serous chorioretinopathy: A 12-month follow-up study. Clin. Ophthalmol. 2013, 7, 939–945. [Google Scholar]
- Stohs, S.J.; Chen, O.; Ray, S.D.; Ji, J.; Bucci, L.R.; Preuss, H.G. Highly bioavailable forms of curcumin and promising avenues for curcumin-based research and application: A review. Molecules 2020, 25, 1397. [Google Scholar] [CrossRef]
- Ichi, T.; Higashimura, Y.; Katayama, T.; Koda, T.; Shimizu, T.; Tada, M. Analysis of crocetin derivatives from gardenia fruits. Nippon Shokuhin Kagaku Kogaku Kaishi 1995, 42, 776–783. [Google Scholar] [CrossRef][Green Version]
- Li, N.; Lin, G.; Kwan, Y.W.; Min, Z.D. Simultaneous quantification of five major biologically active ingredients of saffron by high-performance liquid chromatography. J. Chromatogr. A 1999, 849, 349–355. [Google Scholar] [CrossRef]
- Khazdair, M.R.; Boskabady, M.H.; Hosseini, M.; Rezaee, R.A.; Tsatsakis, A.M. The effects of crocus sativus (saffron) and its constituents on nervous system: A review. Avicenna J. Phytomed. 2015, 5, 376–391. [Google Scholar]
- Jose Bagur, M.; Alonso Salinas, G.L.; Jimenez-Monreal, A.M.; Chaouqi, S.; Llorens, S.; Martinez-Tome, M.; Alonso, G.L. Saffron: An old medicinal plant and a potential novel functional food. Molecules 2017, 23, 30. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, M.; Tsuruma, K.; Imai, S.; Nakanishi, T.; Umigai, N.; Shimazawa, M.; Hara, H. Crocetin prevents retinal degeneration induced by oxidative and endoplasmic reticulum stresses via inhibition of caspase activity. Eur. J. Pharmacol. 2011, 650, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Ishizuka, F.; Shimazawa, M.; Umigai, N.; Ogishima, H.; Nakamura, S.; Tsuruma, K.; Hara, H. Crocetin, a carotenoid derivative, inhibits retinal ischemic damage in mice. Eur. J. Pharmacol. 2013, 703, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Albarral, J.A.; Ramirez, A.I.; De Hoz, R.; Lopez-Villarin, N.; Salobrar-Garcia, E.; Lopez-Cuenca, I.; Licastro, E.; Inarejos-Garcia, A.M.; Almodovar, P.; Pinazo-Duran, M.D.; et al. Neuroprotective and anti-inflammatory effects of a hydrophilic saffron extract in a model of glaucoma. Int. J. Mol. Sci. 2019, 20, 4110. [Google Scholar] [CrossRef]
- Heitmar, R.; Brown, J.; Kyrou, I. Saffron (Crocus sativus L.) in ocular diseases: A narrative review of the existing evidence from clinical studies. Nutrients 2019, 11, 649. [Google Scholar] [CrossRef]
- Piccardi, M.; Fadda, A.; Martelli, F.; Marangoni, D.; Magli, A.; Minnella, A.M.; Bertelli, M.; Di Marco, S.; Bisti, S.; Falsini, B. Antioxidant saffron and central retinal function in abca4-related stargardt macular dystrophy. Nutrients 2019, 11, 2461. [Google Scholar] [CrossRef]
- Eckert, A.; Keil, U.; Scherping, I.; Hauptmann, S.; Muller, W.E. Stabilization of mitochondrial membrane potential and improvement of neuronal energy metabolism by ginkgo biloba extract egb 761. Ann. N.Y. Acad. Sci. 2005, 1056, 474–485. [Google Scholar] [CrossRef]
- Hirooka, K.; Tokuda, M.; Miyamoto, O.; Itano, T.; Baba, T.; Shiraga, F. The ginkgo biloba extract (egb 761) provides a neuroprotective effect on retinal ganglion cells in a rat model of chronic glaucoma. Curr. Eye Res. 2004, 28, 153–157. [Google Scholar] [CrossRef]
- Chung, H.S.; Harris, A.; Kristinsson, J.K.; Ciulla, T.A.; Kagemann, C.; Ritch, R. Ginkgo biloba extract increases ocular blood flow velocity. J. Ocul. Pharmacol. Ther. J. Assoc. Ocul. Pharmacol. Ther. 1999, 15, 233–240. [Google Scholar] [CrossRef]
- Jia, L.Y.; Sun, L.; Fan, D.S.; Lam, D.S.; Pang, C.P.; Yam, G.H. Effect of topical ginkgo biloba extract on steroid-induced changes in the trabecular meshwork and intraocular pressure. Arch. Ophthalmol. 2008, 126, 1700–1706. [Google Scholar] [CrossRef] [PubMed]
- Quaranta, L.; Bettelli, S.; Uva, M.G.; Semeraro, F.; Turano, R.; Gandolfo, E. Effect of ginkgo biloba extract on preexisting visual field damage in normal tension glaucoma. Ophthalmology 2003, 110, 359–362. [Google Scholar] [CrossRef]
- Shim, S.H.; Kim, J.M.; Choi, C.Y.; Kim, C.Y.; Park, K.H. Ginkgo biloba extract and bilberry anthocyanins improve visual function in patients with normal tension glaucoma. J. Med. Food 2012, 15, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Kong, X.; Huang, R.; Jin, L.; Ding, X.; He, M.; Liu, X.; Patel, M.C.; Congdon, N.G. Effect of ginkgo biloba on visual field and contrast sensitivity in chinese patients with normal tension glaucoma: A randomized, crossover clinical trial. Investig. Ophthalmol. Vis. Sci. 2014, 55, 110–116. [Google Scholar] [CrossRef]
- Deng, Y.; Ng, E.S.; Kwan, Y.W.; Lau, C.B.; Cheung, D.W.; Koon, J.C.; Zhang, Z.; Zuo, Z.; Leung, P.C.; Fung, K.P.; et al. Cerebral vasodilator properties of danshen and gegen: A study of their combined efficacy and mechanisms of actions. Phytomed. Int. J. Phytother. Phytopharm. 2014, 21, 391–399. [Google Scholar] [CrossRef]
- Yue, K.K.; Lee, K.W.; Chan, K.K.; Leung, K.S.; Leung, A.W.; Cheng, C.H. Danshen prevents the occurrence of oxidative stress in the eye and aorta of diabetic rats without affecting the hyperglycemic state. J. Ethnopharmacol. 2006, 106, 136–141. [Google Scholar] [CrossRef]
- Ma, L.; Tang, L.; Yi, Q. Salvianolic acids: Potential source of natural drugs for the treatment of fibrosis disease and cancer. Front. Pharmacol. 2019, 10, 97. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.Y.; Jiang, Q.; Li, K.R.; Zhao, Y.X.; Cao, C.; Yao, J. Salvianolic acid a protects rpe cells against oxidative stress through activation of nrf2/ho-1 signaling. Free Radic. Biol. Med. 2014, 69, 219–228. [Google Scholar] [CrossRef]
- Mao, K.; Shu, W.; Liu, L.; Gu, Q.; Qiu, Q.; Wu, X. Salvianolic acid a inhibits ox-ldl effects on exacerbating choroidal neovascularization via downregulating cyld. Oxid. Med. Cell Longev. 2017, 2017, 6210694. [Google Scholar] [CrossRef]
- Xu, C.; Hou, B.; He, P.; Ma, P.; Yang, X.; Yang, X.; Zhang, L.; Qiang, G.; Li, W.; Du, G. Neuroprotective effect of salvianolic acid a against diabetic peripheral neuropathy through modulation of nrf2. Oxid. Med. Cell Longev. 2020, 2020, 6431459. [Google Scholar] [CrossRef]
- Liu, X.; Xavier, C.; Jann, J.; Wu, H. Salvianolic acid b (sal b) protects retinal pigment epithelial cells from oxidative stress-induced cell death by activating glutaredoxin 1 (grx1). Int. J. Mol. Sci. 2016, 17, 1835. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Qin, Y.; Yuan, W.; Deng, H.; Zhang, Y.; Jin, M. Compound danshen dripping pill for treating early diabetic retinopathy: A randomized, double-dummy, double-blind study. Evid. Based Complement. Altern. Med. Ecam. 2015, 2015, 539185. [Google Scholar] [CrossRef] [PubMed]
- Lian, F.; Wu, L.; Tian, J.; Jin, M.; Zhou, S.; Zhao, M.; Wei, L.; Zheng, Y.; Wang, Y.; Zhang, M.; et al. The effectiveness and safety of a danshen-containing chinese herbal medicine for diabetic retinopathy: A randomized, double-blind, placebo-controlled multicenter clinical trial. J. Ethnopharmacol. 2015, 164, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Wang, Z.; Huang, L.; Zheng, S.; Wang, D.; Chen, S.; Zhang, H.; Yang, S. Review of the botanical characteristics, phytochemistry, and pharmacology of astragalus membranaceus (huangqi). Phytother. Res. Ptr. 2014, 28, 1275–1283. [Google Scholar] [CrossRef]
- Aldarmaa, J.; Liu, Z.; Long, J.; Mo, X.; Ma, J.; Liu, J. Anti-convulsant effect and mechanism of astragalus mongholicus extract in vitro and in vivo: Protection against oxidative damage and mitochondrial dysfunction. Neurochem. Res. 2010, 35, 33–41. [Google Scholar] [CrossRef]
- Li, X.T.; Zhang, Y.K.; Kuang, H.X.; Jin, F.X.; Liu, D.W.; Gao, M.B.; Liu, Z.; Xin, X.J. Mitochondrial protection and anti-aging activity of astragalus polysaccharides and their potential mechanism. Int. J. Mol. Sci. 2012, 13, 1747–1761. [Google Scholar] [CrossRef]
- Huang, Y.F.; Lu, L.; Zhu, D.J.; Wang, M.; Yin, Y.; Chen, D.X.; Wei, L.B. Effects of astragalus polysaccharides on dysfunction of mitochondrial dynamics induced by oxidative stress. Oxidative Med. Cell Longev. 2016, 2016, 9573291. [Google Scholar] [CrossRef]
- Ding, Y.; Yuan, S.; Liu, X.; Mao, P.; Zhao, C.; Huang, Q.; Zhang, R.; Fang, Y.; Song, Q.; Yuan, D.; et al. Protective effects of astragaloside iv on db/db mice with diabetic retinopathy. PLoS ONE 2014, 9, e112207. [Google Scholar] [CrossRef]
- Wu, J.; Ke, X.; Ma, N.; Wang, W.; Fu, W.; Zhang, H.; Zhao, M.; Gao, X.; Hao, X.; Zhang, Z. Formononetin, an active compound of astragalus membranaceus (fisch) bunge, inhibits hypoxia-induced retinal neovascularization via the hif-1alpha/vegf signaling pathway. Drug Des. Dev. Ther. 2016, 10, 3071–3081. [Google Scholar] [CrossRef]
- Daliu, P.; Santini, A.; Novellino, E. From pharmaceuticals to nutraceuticals: Bridging disease prevention and management. Expert Rev. Clin. Pharmacol. 2019, 12, 1–7. [Google Scholar] [CrossRef]
| Diseases | Nutraceutical | Effects | Mechanisms | Animal Models | Refs. |
|---|---|---|---|---|---|
| Glaucoma | Resveratrol | Neuroprotection | Apoptosis ↓ | Intracameral injection of hyaluronic acid-induced rats | [71] |
| Resveratrol | Neuroprotection | BiP↑, CHOP↑, XBP-1↓ | Optic nerve crush experimental mice | [72] | |
| Resveratrol | IOP reduction | Binding through A1R | Steroid-induced ocular hypertension rats | [73] | |
| Resveratrol and quercetin | IOP reduction | Synergic effects | Normal normotensive rabbits | [74] | |
| AMD | Resveratrol | Prevent retinal degeneration | ONL↑, ERG↑, Apoptosis↓, AP-1↓, SIRT1↑ | Light-induced retinal degeneration mice | [75] |
| Resveratrol | Neuroprotection | ONL↑, ERG↑, LIF, BDNF, OSM, CT-1 and CLC↑ | Light-induced retinal degeneration mice | [76] | |
| DR | Resveratrol | Alleviate oxidative stress | BG↓, BW↑, SOD↑, 8-Isoprostane↓, GSSG/GSH↓, NF-κB↓, Apoptosis↓, ONL↑ | STZ-nicotinamide-induced DR rats | [77] |
| Resveratrol | Anti-oxidative stress | AOPP↓, MDA↓, TOS↓ | STZ-induced type 1 diabetes rats | [78] | |
| Resveratrol | Anti-inflammation and retinal protection | BG↓, BW↑, AGEs↓, Insulin↑, Apoptosis↓, PON1↑, Ox-LDL↓, IL-1β, IL-6, TNF-α, VEGF, IFN-γ and MCP-1↓ | STZ-induced diabetes rats | [79] | |
| Resveratrol | Retinal vascular protection | BG↓, Pericytes↑ | STZ-induced diabetes mice | [80] | |
| Glaucoma | Quercetin | Neuroprotection | ERG↑, RGC survival↑, Apoptosis↓, ΔΨm↑ | Chronic ocular hypertension rats | [93] |
| Quercetin | Neuroprotection | GABAergic inhibitory neurotransmission↑, glutamatergic excitatory neurotransmission↓, excitability of the RGCs↓ | Electrocoagulation of the superior scleral vein rats | [94] | |
| AMD | Quercetin | Neuroprotection | Thickness of whole retina↑, Apoptosis↓, Inflammation↓ | Blue light-induced damage mice | [98] |
| Quercetin | Neuroprotection | ERG↑, ONL↑, Phagosomes in RPE↑, AP-1↓ | Light-induced retinal degeneration rats | [99] | |
| DR | Quercetin | Neuroprotection | GSH↑, SOD↑, Thickness of whole retina↑, ONL↑, INL↑, TNF-α↓, IL-1β↓, Apoptosis↓, AQP4, GFAP and caspase-3↓ | STZ-induced diabetes rats | [102] |
| Lutein or DHA | Neuroprotection | Thickness of whole retina↑, ONL↑, INL↑, Apoptosis↓ | STZ-induced diabetes rats | [112] | |
| Retinal detachment (RD) | Lutein | Neuroprotection | ONL↑, GFAP↓, RHO↑, Apoptosis↓ | Subretinal injections-induced RD rats | [115] |
| AMD | Lutein | Anti-oxidative stress | RPE tight junctions↑, ROS↓, SOD↑, Macrophage-related markers↓ | Light-induced AMD-related mice | [116] |
| Lutein and zeaxanthin | Neuroprotection | ERG↑, Apoptosis↓, p-JNK↓, Nrf2↑, GRP78, p-PERK, ATF4 and ATF6↓ | Light-induced damage mice | [117] | |
| DR | Wolfberry | Retinoprotection | Levels of zeaxanthin and lutein↑, SRB1↑, GSTP1, BCO2, and AMPK-α2↑, HIF-1α, VEGF, and HSP↓, Mitochondrial copy number↑, Citrate synthase activity↑, PGC-1α, Nrf1, and TFAM↑ | Leptin receptor-deficient (db/db) type 2 diabetic mice | [118] |
| Glaucoma | Omega-3 PUFAs | IOP reduction | IOP↓, Aqueous outflow↑ | Age-induced IOP increase rats | [132] |
| Omega-3 and omega-6 PUFAs | Anti-inflammation | GFAP↓, Thickness of whole retina↑ | Photocoagulation-induced IOP increase rats | [133] | |
| AMD | Omega-3 PUFAs | Anti-inflammation | PGE2, LTB4, TNF-α and IL-6↓, PGD2↑ | AMD-like retinal lesions mice | [134] |
| DR | Omega-3 PUFAs | Retinoprotection | ERG↑, BG↓ | Leptin receptor-deficient (db/db) type 2 diabetic mice | [135] |
| AMD | Omega-3 PUFAs | Retinoprotection | Lipofuscin↓, ONL↑, MBP, MPP, MRFLP and GFAP↑ | Aged (24-month-old) wild-type mice | [136] |
| Glaucoma | Curcumin | Neuroprotection | IOP↓, RGC density↑ | Ocular hypertension and partial optic nerve transection rats | [144] |
| Retinal degeneration | Crocetin | Neuroprotection | ERG↑, ONL↑, Apoptosis↓ | Light-induced damage mice | [153] |
| Retinal ischemia | Crocetin | Neuroprotection | GCL, INL, ONL↑, ERG↑, p-p38, p-JNK, p- ERK 1/2, p-c-Jun, p-NF-κB↓ | I/R-induced retinal damage mice | [154] |
| Glaucoma | Saffron | Neuroprotection | IOP↓, RGC↑, Iba-1 (+) microglia↓ | Laser-induced ocular hypertension mice | [155] |
| GBE | Neuroprotection | IOP↓, RGC↑, | Ocular hypertension by cautery of three episcleral vessels rats | [159] | |
| GBE | Retinoprotection | IOP↓, Apoptotic TM cells↓ | Dexamethasone–induced ocular hypertension rabbits | [161] | |
| Diabetes | Danshen | Anti-oxidative stress | GSH↑, MDA↓ | STZ-induced diabetes rats | [166] |
| CNV | Salvianolic acid A | Anti-angiogenesis | OX-LDL↓, Fluorescein angiography↓, VEGF↓, PDGF↓, Angiostatin↑, CYLD↓ | Laser photocoagulations plus OX-LDL injection-induced CNV mice | [169] |
| DPN | Salvianolic acid A | Anti-oxidative stress | BG↓, Fructosamine↓, Myelin sheath thickness↑, Nrf2↑ | KK-Ay diabetic mice | [170] |
| DR | Astragaloside IV | Neuroprotection | ERG↑, Apoptosis of RGCs↓, AR↓, p-ERK1/2, NF-kB↓ | Leptin receptor-deficient (db/db) type 2 diabetic mice | [178] |
| Diseases | Nutraceutical | Study Population | Study Design/Follow-Up | Results/Findings | Refs. |
|---|---|---|---|---|---|
| AMD | Resveratrol | 3 cases | Case report | Restoration of structure and visual function | [85] |
| T2D | Resveratrol | 17 subjects | Double-blind randomized cross-over study | Intrahepatic lipid↓, Intramyocellular lipid↑, Mitochondrial function (ex vivo) ↑, Metformin dose↓ | [86] |
| AMD | Quercetin | 2856 adults and 2037 followed | Population-based cohort study/15-y | Quercetin was associated with reduced odds of any AMD (OR: 0.76; 95% CI: 0.58–0.99) | [108] |
| AMD | Lutein/zeaxanthin | 93,676 women | Cohort study/7-y | Lutein/zeaxanthin may protect against intermediate AMD (OR: 0.57; 95% CI: 0.34–0.95) | [121] |
| AMD | Analysis of Lutein/zeaxanthin | 380 adults | Cohort study | Risk of AMD was associated with plasma concentrations of lutein/zeaxanthin (OR: 1.9; 95% CI: 0.9–3.5) | [122] |
| AMD | Lutein/zeaxanthin | 6 publications | Meta-analysis | Dietary intake of lutein/zeaxanthin was significantly related with a reduction in risk of late AMD (RR: 0.74; 95% CI: 0.57–0.97) | [123] |
| AMD | Lutein/zeaxanthin, Omega-3, PUFAs, Zinc | 4203 participants | multicenter, randomized, double-masked, placebo-controlled phase 3 study with a 2 × 2 factorial design/12-y | Participants were limited to those with the lowest dietary intake of lutein + zeaxanthin, results of exploratory subgroup analyses showed a protective effect for progression to advanced AMD (HR: 0.74; 95% CI, 0.59–0.94; p = 0.01) | [125] |
| AMD | Omega-3 PUFAs | 1837 participants | Nested cohort study/12-y | Omega-3 PUFAs intake reduces 30% incidence to develop CGA (OR: 0.65; 95% CI: 0.45–0.92; p < 0.02) and neovascular AMD (OR: 0.68; 95% CI:0.49–0.94; p < 0.02) | [137] |
| AMD | Omega-3 PUFAs | 17 patients | Prospective, noncomparative, descriptive pilot study | No statistically significant improvement in visual acuity and ERG; Serum omega-3 index increased by a mean of 7.6% (p < 0.001) | [138] |
| AMD | Lutein/zeaxanthin, Omega-3 PUFAs, Zinc, β-carotene | 1684 participants | AREDS2/5-y | No significant interaction between supplements and genotype with improvement of progression to late AMD | [139] |
| DR | PUFAs | 379 patients | Cohort study | PUFAs was associated with a reduction in DR severity (OR: 0.18; 95% CI: 0.06–0.59) | [140] |
| DR | Omega-3 PUFAs | 3482 participants | Prospective, randomized clinical trial | Participants meeting the LCω3PUFA recommendation at baseline (≥500 mg/d) compared with those not fulfilling this recommendation (<500 mg/d) showed a 48% relatively reduced risk of incident sight-threatening DR, with a HR of 0.52 (95% CI, 0.31–0.88; p = 0.001). This association was slightly stronger for yearly updated LCω3PUFA intake (relative risk, 0.48; 95% CI, 0.28–0.82; p = 0.007) | [141] |
| CSCR | Curcuminoids and lecithin formulation | 12 patients | Follow-up study/1-y | Visual acuity improvement (p = 0.0005 by Wilcoxon signed rank test), Reduction in neuroretinal or RPE detachment (p = 0.0004 by Wilcoxon signed rank test) | [147] |
| STG/FF | Saffron | 31 patients | Randomized, double-blind, placebo-controlled study/3-y | Saffron had no detrimental effects on the visual acuity and focal ERG | [157] |
| NTG | GBE | 27 patients | Prospective, randomized, placebo-controlled, double-masked cross-over trial | Visual fields improvement in mean deviation (t = 8.86, p = 0.0001, chi-square test) and corrected pattern standard deviation (t = 9.89, p = 0.0001, chi-square test) | [162] |
| NTG | GBE | 332 patients | Retrospective study/1-y or more | HVF mean deviation improvement from −5.25 ± 6.13 to −4.31 ± 5.60 (p = 0.002) | [163] |
| NTG | GBE | 35 patients | Prospective, randomized, placebo-controlled crossover study | No effect on mean defect or contrast sensitivity | [164] |
| NPDR | CDDP | 57 patients | Randomized, double-dummy, double-blind study | Improvement of the best corrected visual acuity (p < 0.05). | [172] |
| NPDR | CDDP | 223 patients | Randomized, double-blind, placebo-controlled clinical trial | Improvement of fluorescence fundus angiography and funduscopic examination (p < 0.001) | [173] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.-P.; Lin, Y.-W.; Huang, Y.-C.; Tsai, F.-J. Mitochondrial Dysfunction as a Novel Target for Neuroprotective Nutraceuticals in Ocular Diseases. Nutrients 2020, 12, 1950. https://doi.org/10.3390/nu12071950
Huang C-P, Lin Y-W, Huang Y-C, Tsai F-J. Mitochondrial Dysfunction as a Novel Target for Neuroprotective Nutraceuticals in Ocular Diseases. Nutrients. 2020; 12(7):1950. https://doi.org/10.3390/nu12071950
Chicago/Turabian StyleHuang, Chun-Ping, Yi-Wen Lin, Yu-Chuen Huang, and Fuu-Jen Tsai. 2020. "Mitochondrial Dysfunction as a Novel Target for Neuroprotective Nutraceuticals in Ocular Diseases" Nutrients 12, no. 7: 1950. https://doi.org/10.3390/nu12071950
APA StyleHuang, C.-P., Lin, Y.-W., Huang, Y.-C., & Tsai, F.-J. (2020). Mitochondrial Dysfunction as a Novel Target for Neuroprotective Nutraceuticals in Ocular Diseases. Nutrients, 12(7), 1950. https://doi.org/10.3390/nu12071950






