The Effect of Caffeine on the Risk and Progression of Parkinson’s Disease: A Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search Strategy
2.2. Inclusion and Exclusion of the Literature
2.3. Data Extraction
2.4. Statistical Analysis
2.5. Data Availability
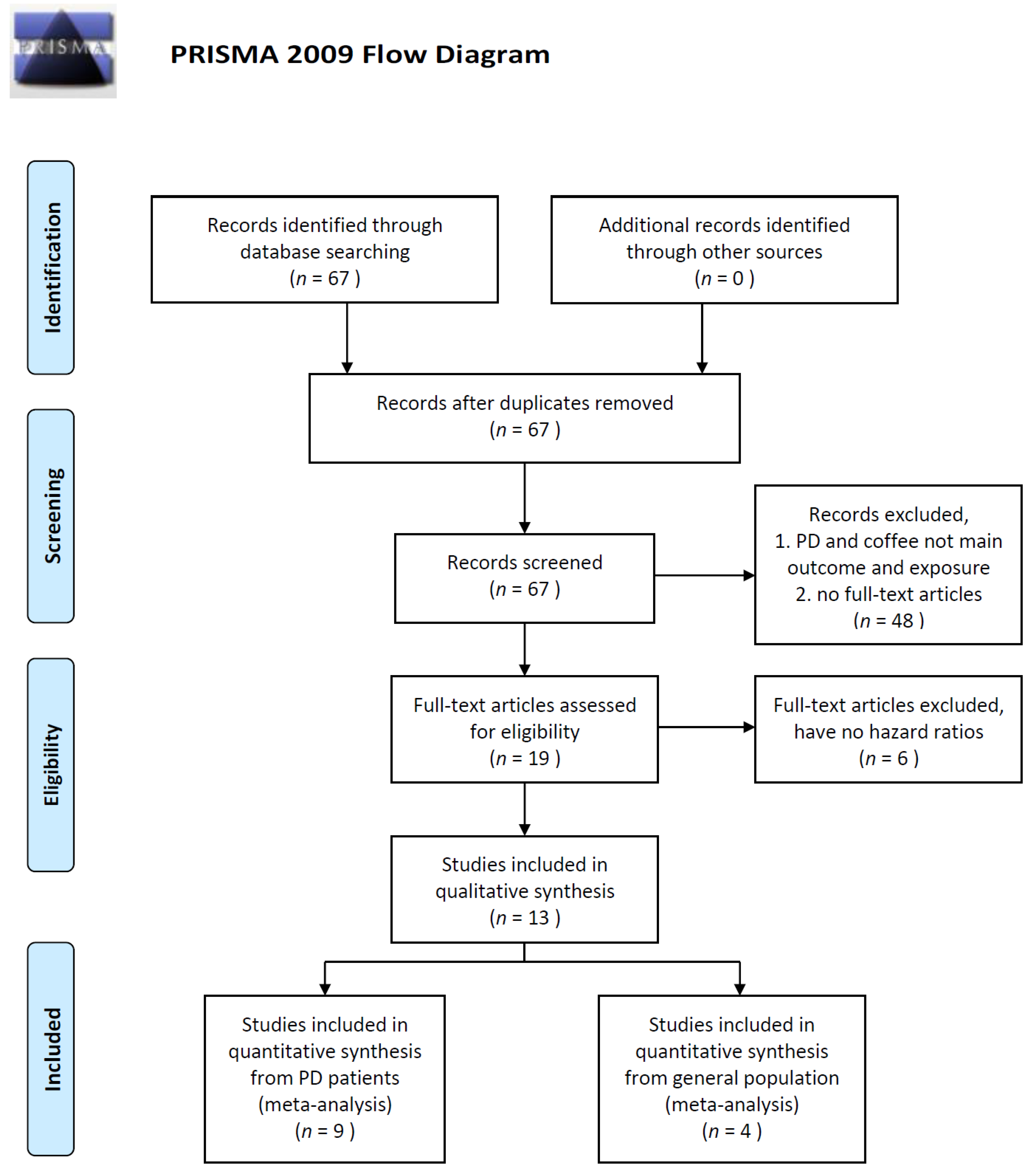
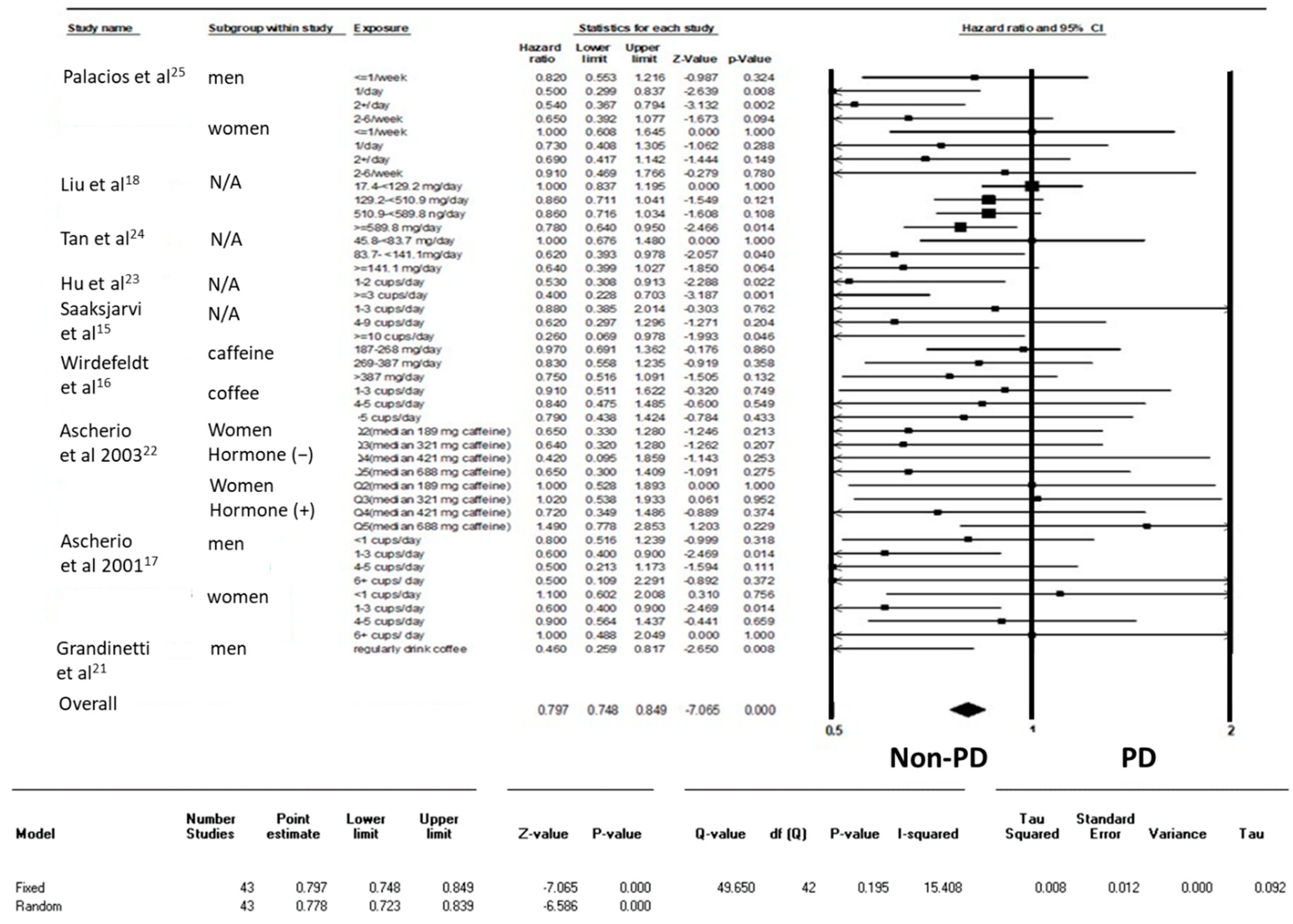
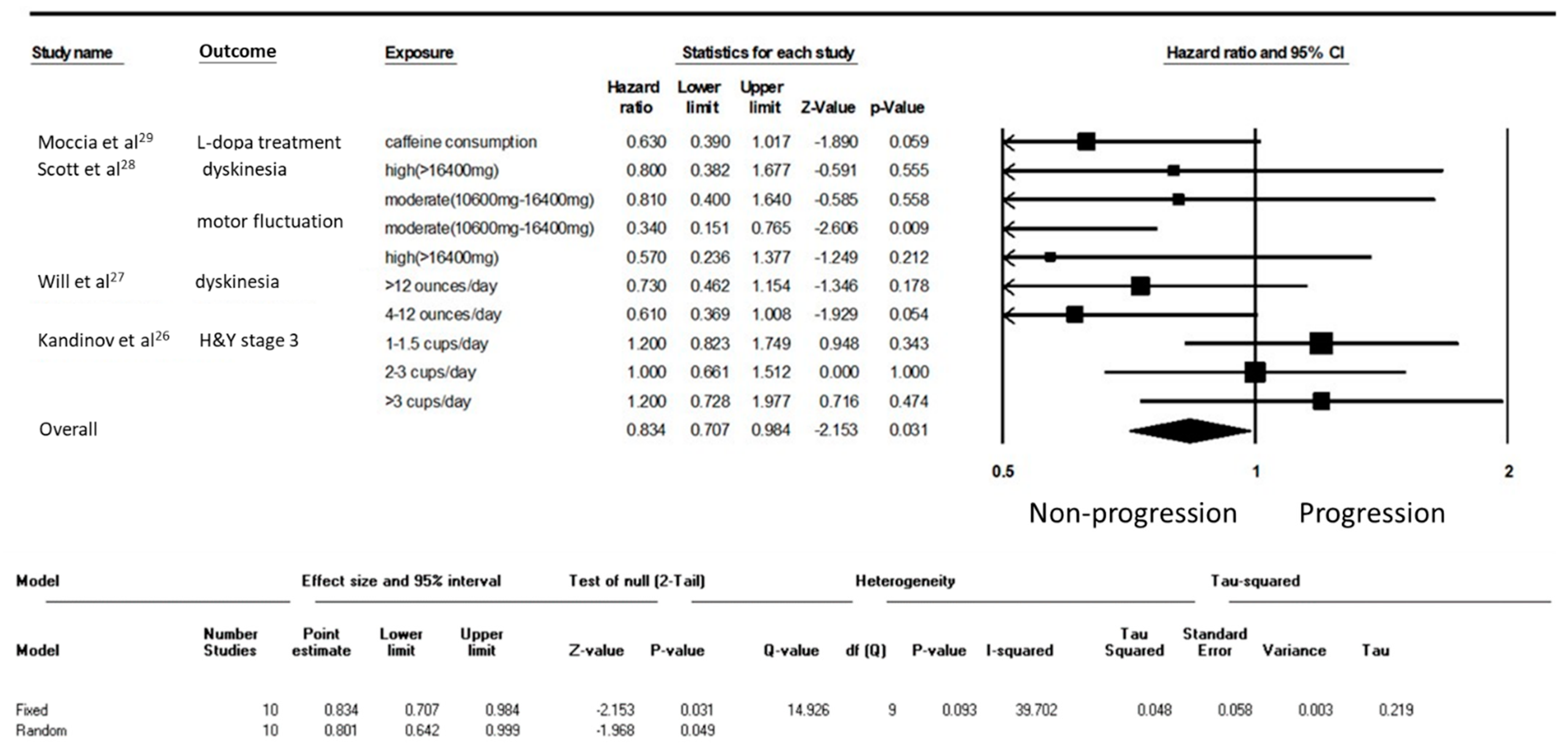
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PD | Parkinson’s disease |
| HR | hazard ratio |
| CI | confidence interval |
References
- Wirdefeldt, K.; Adami, H.-O.; Cole, P.; Trichopoulos, D.; Mandel, J. Epidemiology and etiology of Parkinson’s disease: A review of the evidence. Eur. J. Epidemiol. 2011, 26, 1. [Google Scholar] [CrossRef]
- Kieburtz, K.; Wunderle, K.B. Parkinson’s disease: Evidence for environmental risk factors. Mov. Disord. 2013, 28, 8–13. [Google Scholar] [CrossRef]
- Jiméanez-Jiméanez, F.J.; Mateo, D.; Giméanez-Roldan, S. Premorbid smoking, alcohol consumption, and coffee drinking habits in Parkinson’s disease: A case-control study. Mov. Disord. 1992, 7, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Hellenbrand, W.; Boeing, H.; Robra, B.-P.; Seidler, A.; Vieregge, P.; Nischan, P.; Joerg, J.; Oertel, W.H.; Schneider, E.; Ulm, G. Diet and Parkinson’s disease II, A possible role for the past intake of specific nutrients: Results from a self-administered food-frequency questionnaire in a case-control study. Neurology 1996, 47, 644–650. [Google Scholar] [CrossRef]
- Tan, E.K.; Tan, C.; Fook-Chong, S.M.; Lum, S.Y.; Chai, A.; Chung, H.; Shen, H.; Zhao, Y.; Teoh, M.L.; Yih, Y.; et al. Dose-dependent protective effect of coffee, tea, and smoking in Parkinson’s disease: A study in ethnic Chinese. J. Neurol. Sci. 2003, 216, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Kivipelto, M. Caffeine as a protective factor in dementia and Alzheimer’s disease. J. Alzheimers Dis. 2010, 20 (Suppl. S1), S167–S174. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Mori, T.; Cao, C.; Mamcarz, M.; Runfeldt, M.; Dickson, A.; Rezai-Zadeh, K.; Tane, J.; Citron, B.A.; Lin, X.; et al. Caffeine reverses cognitive impairment and decreases brain amyloid-beta levels in aged Alzheimer’s disease mice. J. Alzheimers Dis. 2009, 17, 661–680. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; Battig, K.; Holmen, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar]
- Carpenter, B.; Lebon, G. Human Adenosine A(2A) Receptor: Molecular Mechanism of Ligand Binding and Activation. Front. Pharmacol. 2017, 8, 898. [Google Scholar] [CrossRef]
- Kolahdouzan, M.; Hamadeh, M.J. The neuroprotective effects of caffeine in neurodegenerative diseases. CNS Neurosci. Ther. 2017, 23, 272–290. [Google Scholar] [CrossRef]
- Chen, J.F.; Xu, K.; Petzer, J.P.; Staal, R.; Xu, Y.H.; Beilstein, M.; Sonsalla, P.K.; Castagnoli, K.; Castagnoli, N., Jr.; Schwarzschild, M.A. Neuroprotection by caffeine and A(2A) adenosine receptor inactivation in a model of Parkinson’s disease. J. Neurosci. 2001, 21, RC143. [Google Scholar] [CrossRef] [PubMed]
- Bove, J.; Serrats, J.; Mengod, G.; Cortes, R.; Tolosa, E.; Marin, C. Neuroprotection induced by the adenosine A2A antagonist CSC in the 6-OHDA rat model of parkinsonism: Effect on the activity of striatal output pathways. Exp. Brain Res. 2005, 165, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Kelsey, J.E.; Langelier, N.A.; Oriel, B.S.; Reedy, C. The effects of systemic, intrastriatal, and intrapallidal injections of caffeine and systemic injections of A2A and A1 antagonists on forepaw stepping in the unilateral 6-OHDA-lesioned rat. Psychopharmacology 2009, 201, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Reyhani-Rad, S.; Mahmoudi, J. Effect of adenosine A2A receptor antagonists on motor disorders induced by 6-hydroxydopamine in rat. Acta Cir. Bras. 2016, 31, 133–137. [Google Scholar] [CrossRef]
- Saaksjarvi, K.; Knekt, P.; Rissanen, H.; Laaksonen, M.A.; Reunanen, A.; Mannisto, S. Prospective study of coffee consumption and risk of Parkinson’s disease. Eur. J. Clin. Nutr. 2008, 62, 908–915. [Google Scholar] [CrossRef]
- Wirdefeldt, K.; Gatz, M.; Pawitan, Y.; Pedersen, N.L. Risk and protective factors for Parkinson’s disease: A study in Swedish twins. Ann. Neurol. 2005, 57, 27–33. [Google Scholar] [CrossRef]
- Ascherio, A.; Zhang, S.M.; Hernan, M.A.; Kawachi, I.; Colditz, G.A.; Speizer, F.E.; Willett, W.C. Prospective study of caffeine consumption and risk of Parkinson’s disease in men and women. Ann. Neurol. 2001, 50, 56–63. [Google Scholar] [CrossRef]
- Liu, R.; Guo, X.; Park, Y.; Huang, X.; Sinha, R.; Freedman, N.D.; Hollenbeck, A.R.; Blair, A.; Chen, H. Caffeine intake, smoking, and risk of Parkinson disease in men and women. Am. J. Epidemiol. 2012, 175, 1200–1207. [Google Scholar] [CrossRef]
- Postuma, R.B.; Lang, A.E.; Munhoz, R.P.; Charland, K.; Pelletier, A.; Moscovich, M.; Filla, L.; Zanatta, D.; Romenets, S.R.; Altman, R.; et al. Caffeine for treatment of Parkinson disease: A randomized controlled trial. Neurology 2012, 79, 651–658. [Google Scholar] [CrossRef]
- Fujimaki, M.; Saiki, S.; Li, Y.; Kaga, N.; Taka, H.; Hatano, T.; Ishikawa, K.-I.; Oji, Y.; Mori, A.; Okuzumi, A.; et al. Serum caffeine and metabolites are reliable biomarkers of early Parkinson disease. Neurology 2018, 90, e404–e411. [Google Scholar] [CrossRef] [PubMed]
- Grandinetti, A.; Morens, D.M.; Reed, D.; MacEachern, D. Prospective study of cigarette smoking and the risk of developing idiopathic Parkinson’s disease. Am. J. Epidemiol. 1994, 139, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Chen, H.; Schwarzschild, M.A.; Zhang, S.M.; Colditz, G.A.; Speizer, F.E. Caffeine, postmenopausal estrogen, and risk of Parkinson’s disease. Neurology 2003, 60, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Bidel, S.; Jousilahti, P.; Antikainen, R.; Tuomilehto, J. Coffee and tea consumption and the risk of Parkinson’s disease. Mov. Disord. 2007, 22, 2242–2248. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.C.; Koh, W.P.; Yuan, J.M.; Wang, R.; Au, W.L.; Tan, J.H.; Tan, E.K.; Yu, M.C. Differential effects of black versus green tea on risk of Parkinson’s disease in the Singapore Chinese Health Study. Am. J. Epidemiol. 2008, 167, 553–560. [Google Scholar] [CrossRef]
- Palacios, N.; Gao, X.; McCullough, M.L.; Schwarzschild, M.A.; Shah, R.; Gapstur, S.; Ascherio, A. Caffeine and risk of Parkinson’s disease in a large cohort of men and women. Mov. Disord. 2012, 27, 1276–1282. [Google Scholar] [CrossRef]
- Kandinov, B.; Giladi, N.; Korczyn, A.D. Smoking and tea consumption delay onset of Parkinson’s disease. Parkinsonism Relat. Disord. 2009, 15, 41–46. [Google Scholar] [CrossRef]
- Wills, A.-M.A.; Eberly, S.; Tennis, M.; Lang, A.E.; Messing, S.; Togasaki, D.; Tanner, C.M.; Kamp, C.; Chen, J.-F.; Oakes, D.; et al. Study, Caffeine consumption and risk of dyskinesia in CALM-PD. Mov. Disord. Off. J. Mov. Disord. Soc. 2013, 28, 380–383. [Google Scholar] [CrossRef]
- Scott, N.W.; Macleod, A.D.; Counsell, C.E. Motor complications in an incident Parkinson’s disease cohort. Eur. J. Neurol. 2016, 23, 304–312. [Google Scholar] [CrossRef]
- Moccia, M.; Erro, R.; Picillo, M.; Vitale, C.; Longo, K.; Amboni, M.; Pellecchia, M.T.; Barone, P. Caffeine consumption and the 4-year progression of de novo Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 32, 116–119. [Google Scholar] [CrossRef]
- Vingerhoets, F.J.; Snow, B.J.; Lee, C.S.; Schulzer, M.; Mak, E.; Calne, D.B. Longitudinal fluorodopa positron emission tomographic studies of the evolution of idiopathic parkinsonism. Ann. Neurol. 1994, 36, 759–764. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, J.; Park, H.J.; Park, E.S.; Oh, S.; Zheng, H.; Junn, E.; Voronkov, M.; Stock, J.B.; Mouradian, M.M. Synergistic neuroprotection by coffee components eicosanoyl-5-hydroxytryptamide and caffeine in models of Parkinson’s disease and DLB. Proc. Natl. Acad. Sci. USA 2018, 115, E12053–E12062. [Google Scholar] [CrossRef] [PubMed]
- Li, F.-J.; Ji, H.-F.; Shen, L. A meta-analysis of tea drinking and risk of Parkinson’s disease. Sci. World J. 2012, 2012, 923464. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.J.; Li, W.; Chen, J.-F. Normal and abnormal functions of adenosine receptors in the central nervous system revealed by genetic knockout studies. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 1358–1379. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, G.; Cerri, S.; Blandini, F. A further update on the role of excitotoxicity in the pathogenesis of Parkinson’s disease. J. Neural. Transm. 2014, 121, 849–859. [Google Scholar] [CrossRef]
- Machado-Filho, J.A.; Correia, A.O.; Montenegro, A.B.; Nobre, M.E.; Cerqueira, G.S.; Neves, K.R.; Mda, G.N.; Cavalheiro, E.A.; Brito, G.A.; Viana, G.S. Caffeine neuroprotective effects on 6-OHDA-lesioned rats are mediated by several factors, including pro-inflammatory cytokines and histone deacetylase inhibitions. Behav. Brain Res. 2014, 264, 116–125. [Google Scholar] [CrossRef]
- Rascol, O.; Perez-Lloret, S.; Ferreira, J.J. New treatments for levodopa-induced motor complications. Mov. Disord. 2015, 30, 1451–1460. [Google Scholar] [CrossRef]
- Golembiowska, K.; Wardas, J.; Noworyta-Sokolowska, K.; Kaminska, K.; Gorska, A. Effects of adenosine receptor antagonists on the in vivo LPS-induced inflammation model of Parkinson’s disease. Neurotox. Res. 2013, 24, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Bibbiani, F.; Oh, J.D.; Petzer, J.P.; Castagnoli, N., Jr.; Chen, J.F.; Schwarzschild, M.A.; Chase, T.N. A2A antagonist prevents dopamine agonist-induced motor complications in animal models of Parkinson’s disease. Exp. Neurol. 2003, 184, 285–294. [Google Scholar] [CrossRef]
- Grondin, R.; Bedard, P.J.; Tahar, A.H.; Gregoire, L.; Mori, A.; Kase, H. Antiparkinsonian effect of a new selective adenosine A2A receptor antagonist in MPTP-treated monkeys. Neurology 1999, 52, 1673–1677. [Google Scholar] [CrossRef]
- Arab, L.; Biggs, M.L.; O’Meara, E.S.; Longstreth, W.T.; Crane, P.K.; Fitzpatrick, A.L. Gender differences in tea, coffee, and cognitive decline in the elderly: The Cardiovascular Health Study. J. Alzheimers Dis. 2011, 27, 553–566. [Google Scholar] [CrossRef]
- Yang, A.; Palmer, A.A.; de Wit, H. Genetics of caffeine consumption and responses to caffeine. Psychopharmacology 2010, 211, 245–257. [Google Scholar] [CrossRef]
- Breen, D.P.; Evans, J.R.; Farrell, K.; Brayne, C.; Barker, R.A. Determinants of delayed diagnosis in Parkinson’s disease. J. Neurol. 2013, 260, 1978–1981. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S.; Oakes, D.; Shoulson, I.; Kieburtz, K.; Rudolph, A.; Lang, A.; Olanow, C.W.; Tanner, C.; Marek, K. Levodopa and the progression of Parkinson’s disease. N. Engl. J. Med. 2004, 351, 2498–2508. [Google Scholar] [PubMed]
- Olanow, C.W.; Rascol, O.; Hauser, R.; Feigin, P.D.; Jankovic, J.; Lang, A.; Langston, W.; Melamed, E.; Poewe, W.; Stocchi, F.; et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. N. Engl. J. Med. 2009, 361, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.J.; Lawton, M.A.; Baig, F.; Ruffmann, C.; Barber, T.R.; Lo, C.; Klein, J.C.; Ben-Shlomo, Y.; Hu, M.T. Predictors of motor complications in early Parkinson’s disease: A prospective cohort study. Mov. Disord. 2019, 34, 1174–1183. [Google Scholar] [CrossRef]
- Wu, T.L.; Wang, C.C.; Lin, F.J.; Wu, R.M. The association between early treatment with amantadine and delayed onset of levodopa-induced dyskinesia in patients with Parkinson’s disease. Parkinsonism Relat. Disord. 2018, 46, e14–e15. [Google Scholar] [CrossRef]
- Group, P.S. Pramipexole vs Levodopa as Initial Treatment for Parkinson DiseaseA Randomized Controlled Trial. JAMA 2000, 284, 1931–1938. [Google Scholar] [CrossRef]
- Olanow, C.W.; Kieburtz, K.; Rascol, O.; Poewe, W.; Schapira, A.H.; Emre, M.; Nissinen, H.; Leinonen, M.; Stocchi, F. Factors predictive of the development of Levodopa-induced dyskinesia and wearing-off in Parkinson’s disease. Mov. Disord. 2013, 28, 1064–1071. [Google Scholar] [CrossRef]
- Stocchi, F.; Vacca, L.; Radicati, F.G. How to optimize the treatment of early stage Parkinson’s disease. Transl. Neurodegener. 2015, 4, 4. [Google Scholar] [CrossRef]
- Oñatibia-Astibia, A.; Franco, R.; Martínez-Pinilla, E. Health benefits of methylxanthines in neurodegenerative diseases. Mol. Nutr. Food Res. 2017, 61, 1600670. [Google Scholar] [CrossRef]
- Martino, R.; Candundo, H.; Lieshout, P.v.; Shin, S.; Crispo, J.A.G.; Barakat-Haddad, C. Onset and progression factors in Parkinson’s disease: A systematic review. NeuroToxicology 2017, 61, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Pinares-Garcia, P.; Stratikopoulos, M.; Zagato, A.; Loke, H.; Lee, J. Sex: A Significant Risk Factor for Neurodevelopmental and Neurodegenerative Disorders. Brain Sci. 2018, 8, 154. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.K.; Wu, C.; Tilley, B.C.; Lohmann, K.; Klein, C.; Payami, H.; Wills, A.M.; Aminoff, M.J.; Bainbridge, J.; Dewey, R.; et al. Caffeine, creatine, GRIN2A and Parkinson’s disease progression. J. Neurol. Sci. 2017, 375, 355–359. [Google Scholar] [CrossRef]
- Chuang, Y.H.; Lill, C.M.; Lee, P.C.; Hansen, J.; Lassen, C.F.; Bertram, L.; Greene, N.; Sinsheimer, J.S.; Ritz, B. Gene-Environment Interaction in Parkinson’s Disease: Coffee, ADORA2A, and CYP1A2. Neuroepidemiology 2016, 47, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.Y.; O’Reilly É, J.; Hughes, K.C.; Gao, X.; Schwarzschild, M.A.; McCullough, M.L.; Hannan, M.T.; Betensky, R.A.; Ascherio, A. Interaction between caffeine and polymorphisms of glutamate ionotropic receptor NMDA type subunit 2A (GRIN2A) and cytochrome P450 1A2 (CYP1A2) on Parkinson’s disease risk. Mov. Disord. 2018, 33, 414–420. [Google Scholar] [CrossRef]
| Study Name | Country | Original Cohort (Established-Last Outcome Assessment) | n | Assessment Caffeine Consumption | Amount of Caffeine Consumption | The Diagnosis of PD |
|---|---|---|---|---|---|---|
| Ascherio et al. [17] | USA | Health Professionals’ Follow-Up Study and Nurses’ Health Study (1976 and 1986/1994) | 135,916 | Semiquantitative food-frequency questionnaire (SFFQ) | Caffeine was 137 mg per cup of coffee, 47 mg per cup of tea, 46 mg per can or bottle of cola beverage, and 7 mg per serving of chocolate candy. | Self-report and medical records |
| Ascherio et al. [22] | USA | Nurses’ Health Study (1976/1998) | 121,700 women | Semiquantitative food-frequency questionnaire (SFFQ) | Caffeine was 137 mg per cup of coffee, 47 mg per cup of tea, 46 mg per can or bottle of cola beverage, and 7 mg per serving of chocolate candy. | Medical records |
| Grandinetti et al. [21] | USA | Honolulu Heart Program-Japanese and Okinawan ancestry (1965/1991) | 8006 men | Questionnaires | NA | Medical records |
| Hu et al. [23] | FIN | Four independent cross-sectional population surveys were carried out in five geographic areas of Finland in 1982, 1987, 1992, and 1997 (1982/2002) | 29,335 | Self-administered questionnaire | Cups of coffee | National Social Insurance Institution’s Register |
| Liu et al. [18] | USA | NIH-AARP Diet and Health Study (1995/2010) | 566,401 | Diet History Questionnaire | Nutrient calculation: 1994–1996 US Department of Agriculture’s Continuing Survey of Food Intakes by Individuals. | Interview and copy of medical records |
| Palacios et al. [25] | USA | CPS II–Nutrition cohort (1992/2007) | 184,190 | Food Frequency Questionnaire | 137 and 47 mg per cup of coffee and tea, respectively, 46 mg per can or bottle of cola; and 7 mg per serving of chocolate. | Interview and copy of medical records |
| Sääksjärvi et al. [15] | FIN | Finnish Mobile Clinic Health Examination Survey (1973/1994) | 7246 | Self-administered, health questionnaire | Cups of coffee | National Social Insurance Institution’s Register |
| Tan et al. [24] | SG | Singapore Chinese Health Study (1993/2005) | 63,257 | A validated, semiquantitative food frequency section questionnaire | Singapore Food Composition Table, a food-nutrient database that lists the levels of 96 Nutritive/nonnutritive components (including caffeine) per 100 g of cooked food and beverages | Interview and linkage database to medical record |
| Wirdefeldt et al. [16] | SE | Swedish Twin Registry (1961 and 1973/without clear mentioning) | 52,149 | Questionnaires | Did not provide the formula | Inpatient Discharge Register and Cause of Death Register |
| Study Name | Country | Number of PD | Stage of PD | Assessment Caffeine Consumption | Amount of Caffeine Consumption | Mean Follow-Up Period of Time | Outcome as the Progression of PD |
|---|---|---|---|---|---|---|---|
| Kandinov et al. [26] | IL | 278 | Onset of PD motor symptoms | Interview | The number of cups of coffee per day | 10.3 years | Time from onset to Hoehn and Yahr stage 3 |
| Moccia et al. [29] | IL | 79 | de novo, drug naïve | Caffeine Consumption Questionnaire | i.e., Espresso 1oz = 50 mg caffeine | 4 years | Starting L-dopa treatment |
| Scott et al. [28] | GB | 183 | Newly diagnosed | Verbal interview about the average level of exposure before baseline | Cups of tea: 47 mg caffeine Cup of coffee: 62 mg caffeine | 59 months | 1.Motor fluctuation 2.Dyskinesia |
| Wills et al. [27] | US | 228 | Early PD | questionnaire assessing both current (“in the past week”) and prior (“on average over the past 5 years”) caffeine intake | Coffee (85 mg caffeine/5 oz) Tea (36 mg caffeine/5 oz) Soda (45 mg caffeine/12 oz) | 5.5 years | Dyskinesia |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, C.T.; Chan, L.; Bai, C.-H. The Effect of Caffeine on the Risk and Progression of Parkinson’s Disease: A Meta-Analysis. Nutrients 2020, 12, 1860. https://doi.org/10.3390/nu12061860
Hong CT, Chan L, Bai C-H. The Effect of Caffeine on the Risk and Progression of Parkinson’s Disease: A Meta-Analysis. Nutrients. 2020; 12(6):1860. https://doi.org/10.3390/nu12061860
Chicago/Turabian StyleHong, Chien Tai, Lung Chan, and Chyi-Huey Bai. 2020. "The Effect of Caffeine on the Risk and Progression of Parkinson’s Disease: A Meta-Analysis" Nutrients 12, no. 6: 1860. https://doi.org/10.3390/nu12061860
APA StyleHong, C. T., Chan, L., & Bai, C.-H. (2020). The Effect of Caffeine on the Risk and Progression of Parkinson’s Disease: A Meta-Analysis. Nutrients, 12(6), 1860. https://doi.org/10.3390/nu12061860





