Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/mTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Animals
2.3. Experimental Protocols
2.4. BALF Collection and Total Cell Counts from Mice
2.5. Nitrite Assay
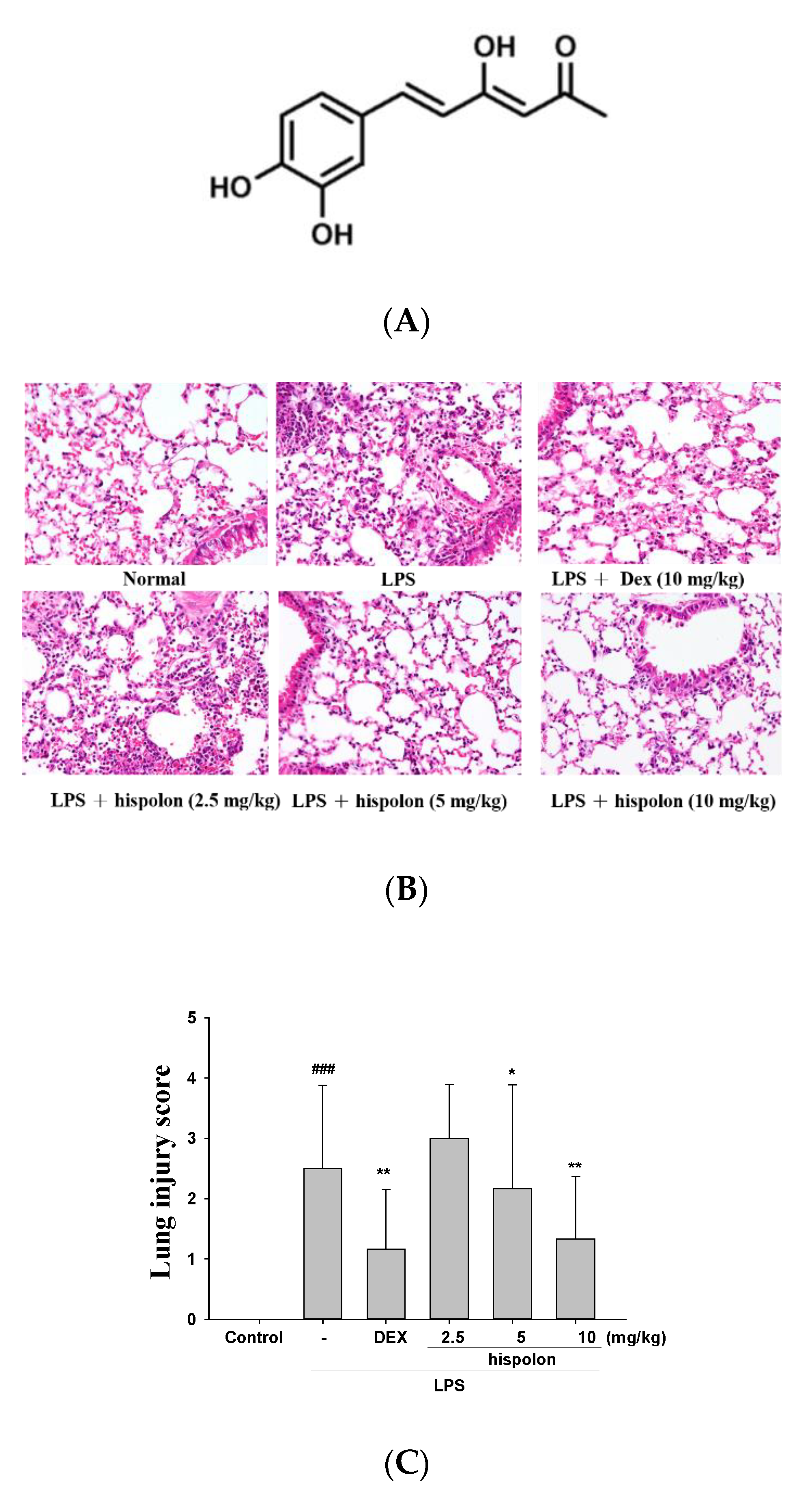
2.6. Histopathological Analysis
2.7. ELISA Analysis in the BALF
2.8. Pulmonary Tissue Wet/Dry Weight Ratio
2.9. Myeloperoxidase Activity
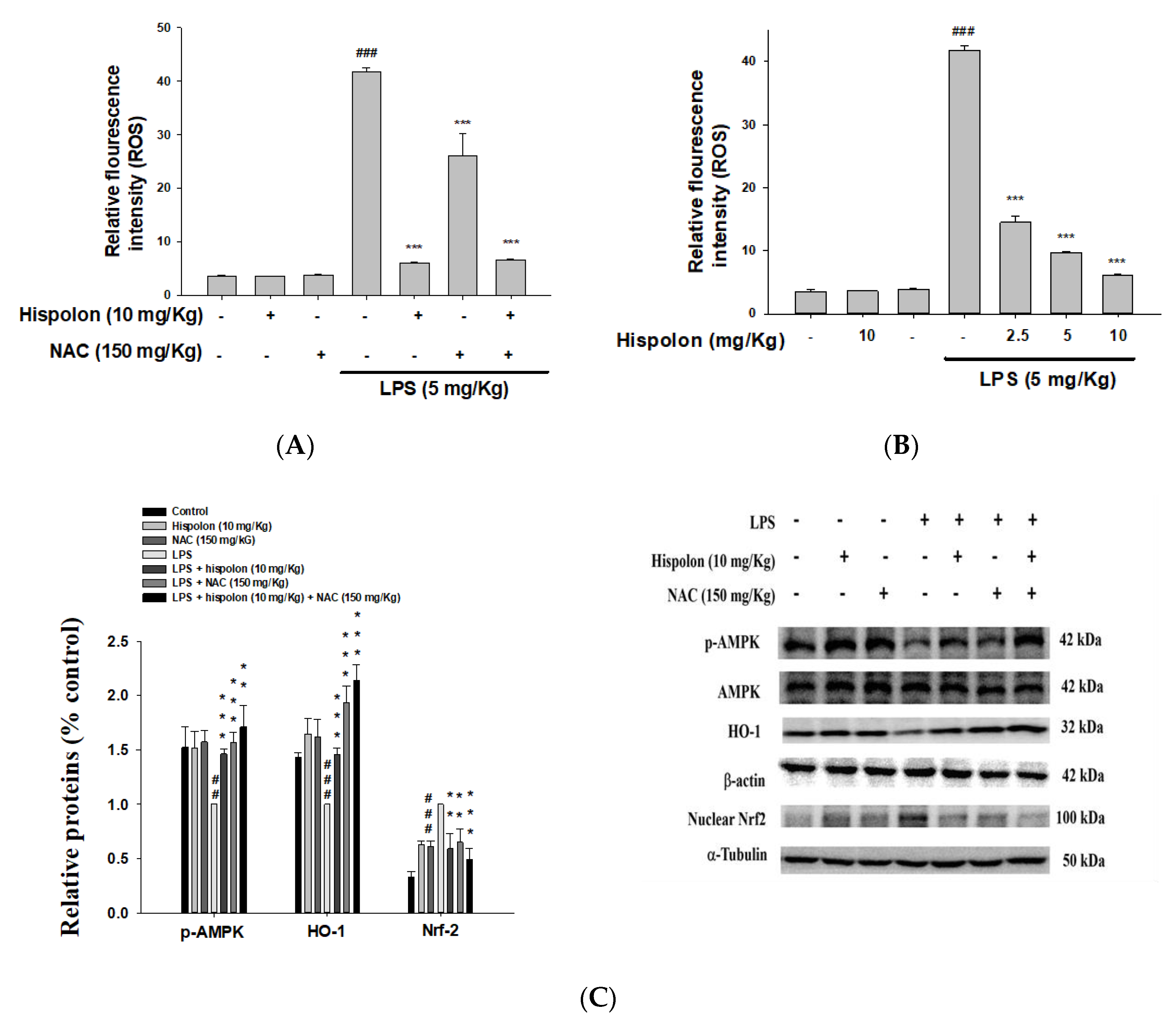
2.10. Determination of ROS
2.11. Western Blot Analysis
2.12. Statistical Analyses
3. Results
3.1. Hispolon Decreases LPS-Induced Histopathological Changes in the Mouse Lung
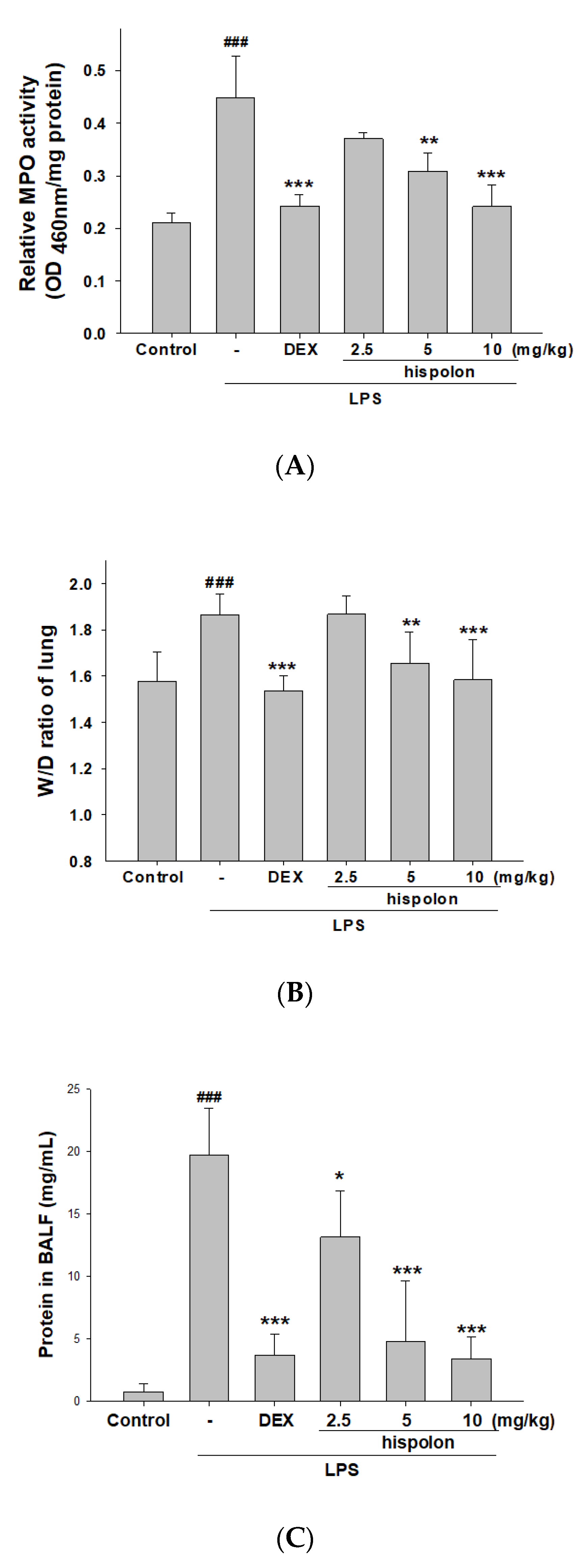
3.2. Decreased Pulmonary W/D Weight Ratio and MPO Activity
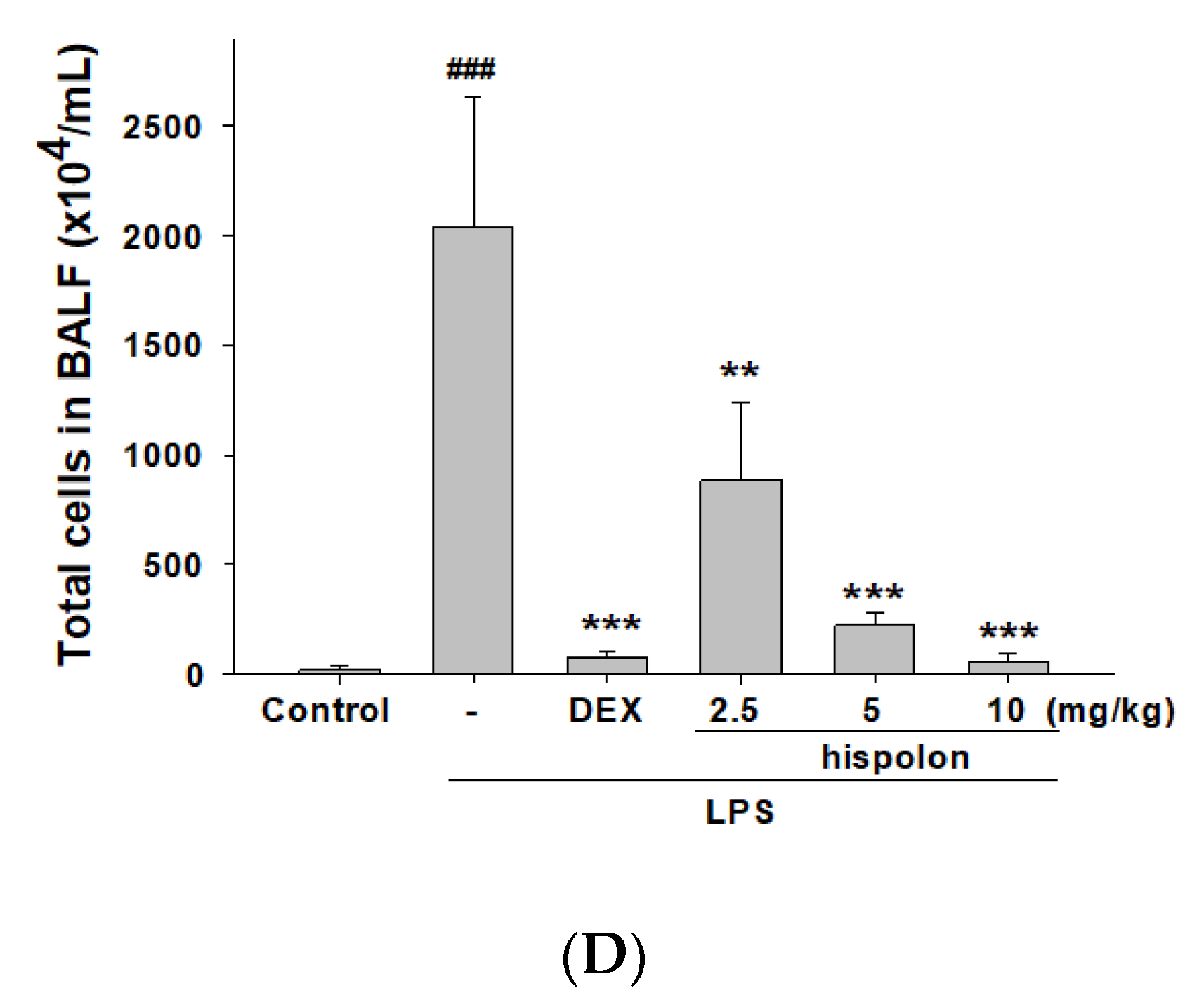
3.3. Decreased Total Cell Numbers and Protein Concentration
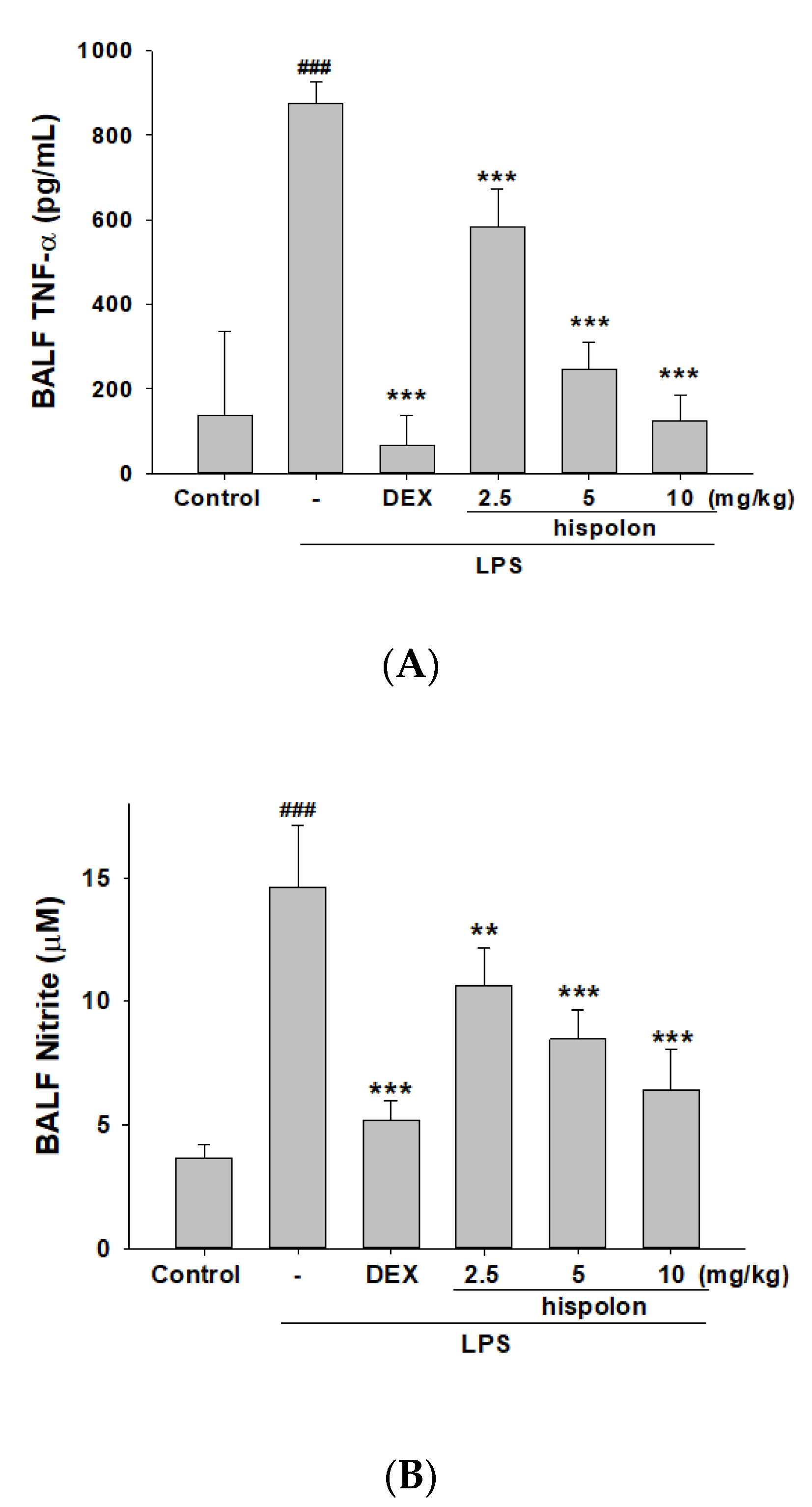
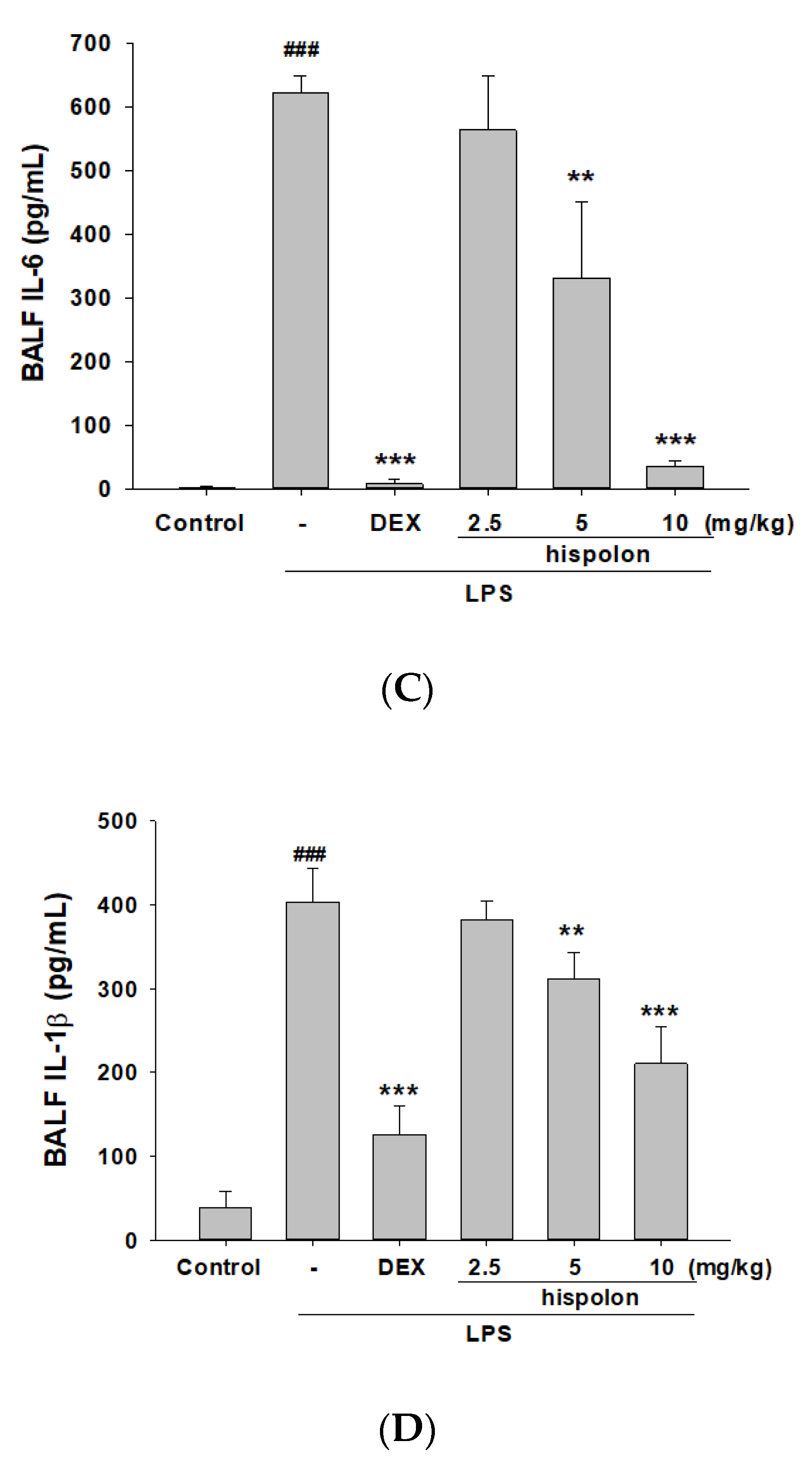
3.4. Decreased Pro-Inflammatory Cytokine Levels
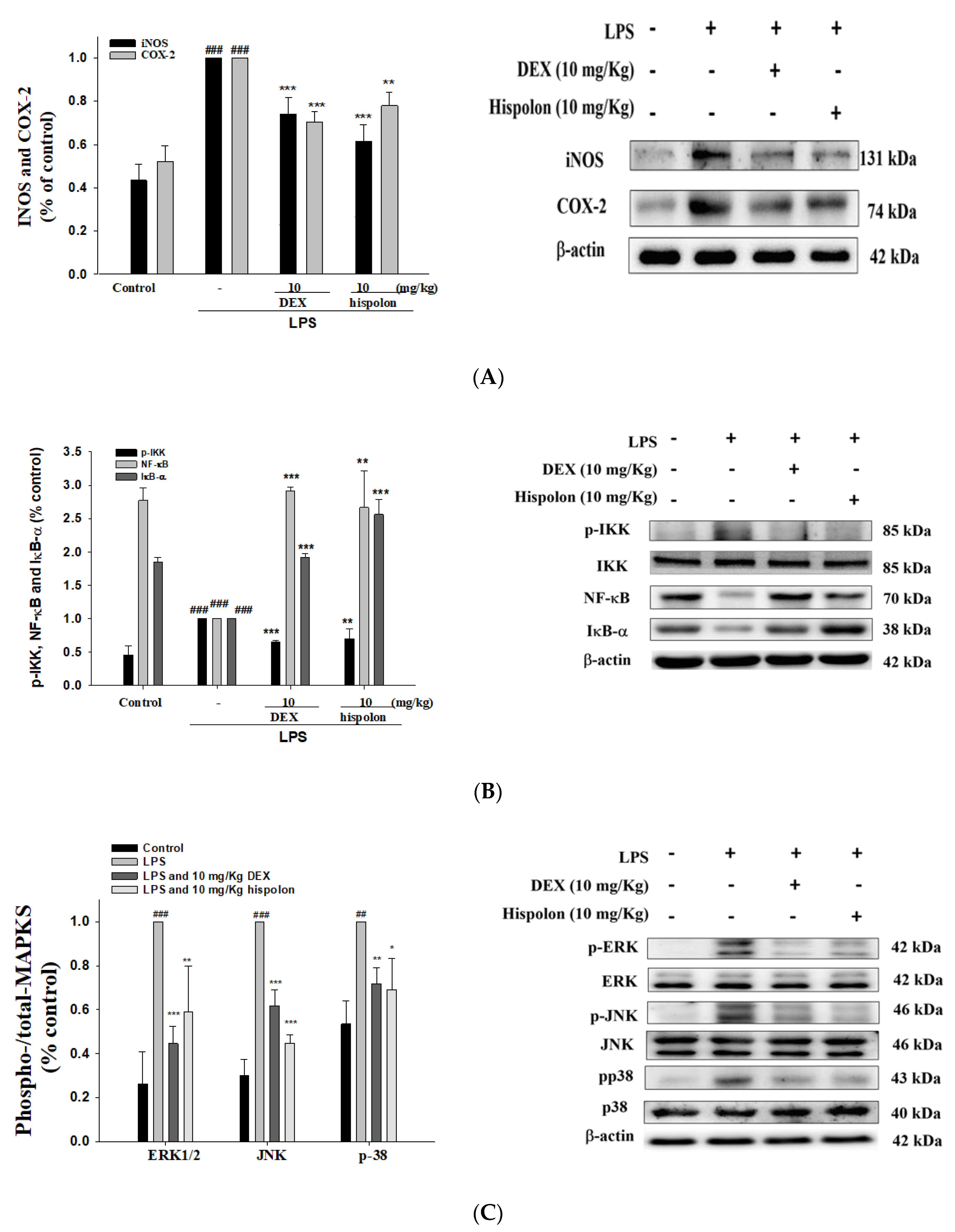
3.5. Suppression of LPS-challenged ALI iNOS, COX-2 and IKK/IκBα/NF-κB signaling in the Lung Tissues
3.6. Suppression of MAPK Pathway Activation
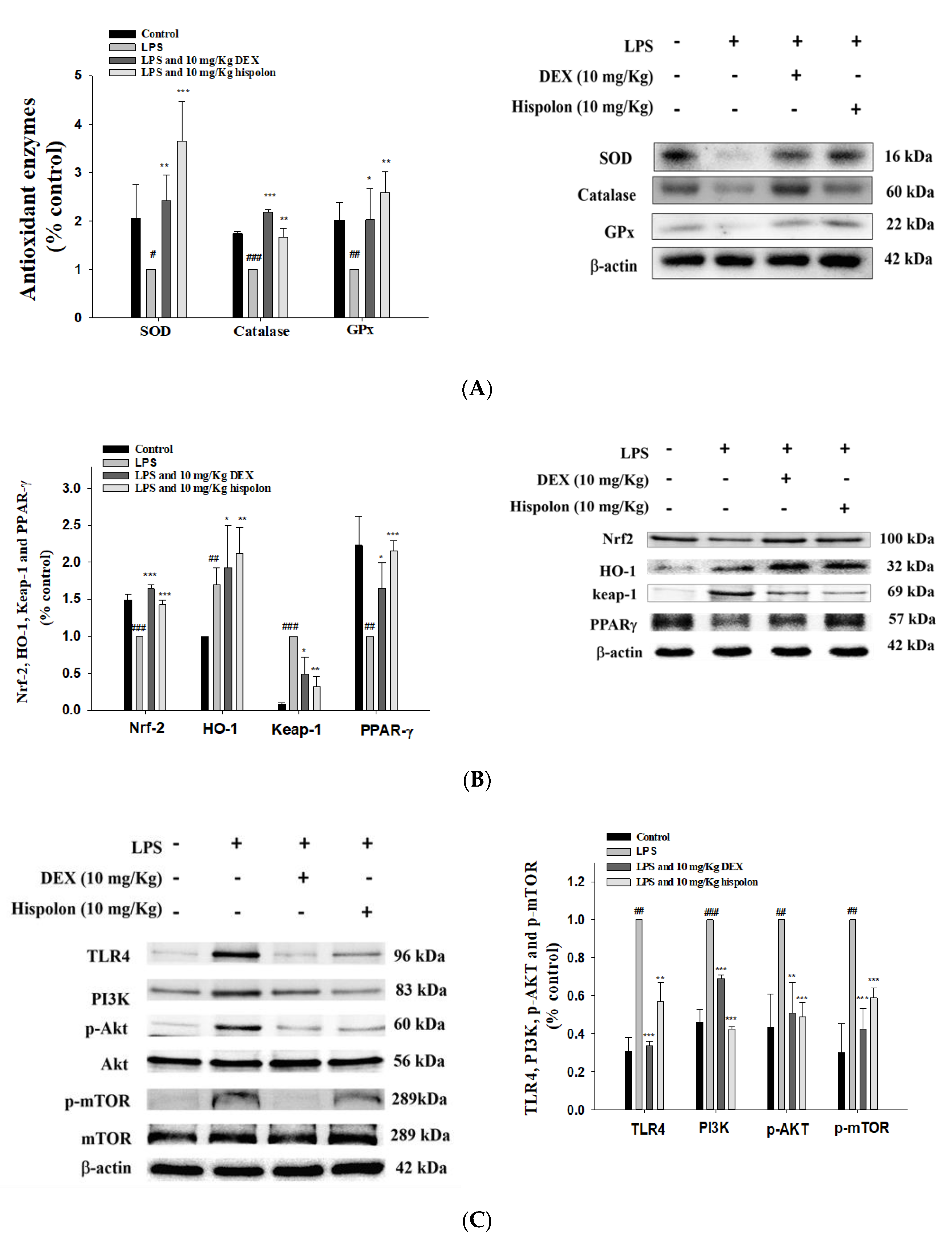
3.7. The Activation of LPS-Induced Antioxidant Enzymes and PPARγ Signaling Pathways
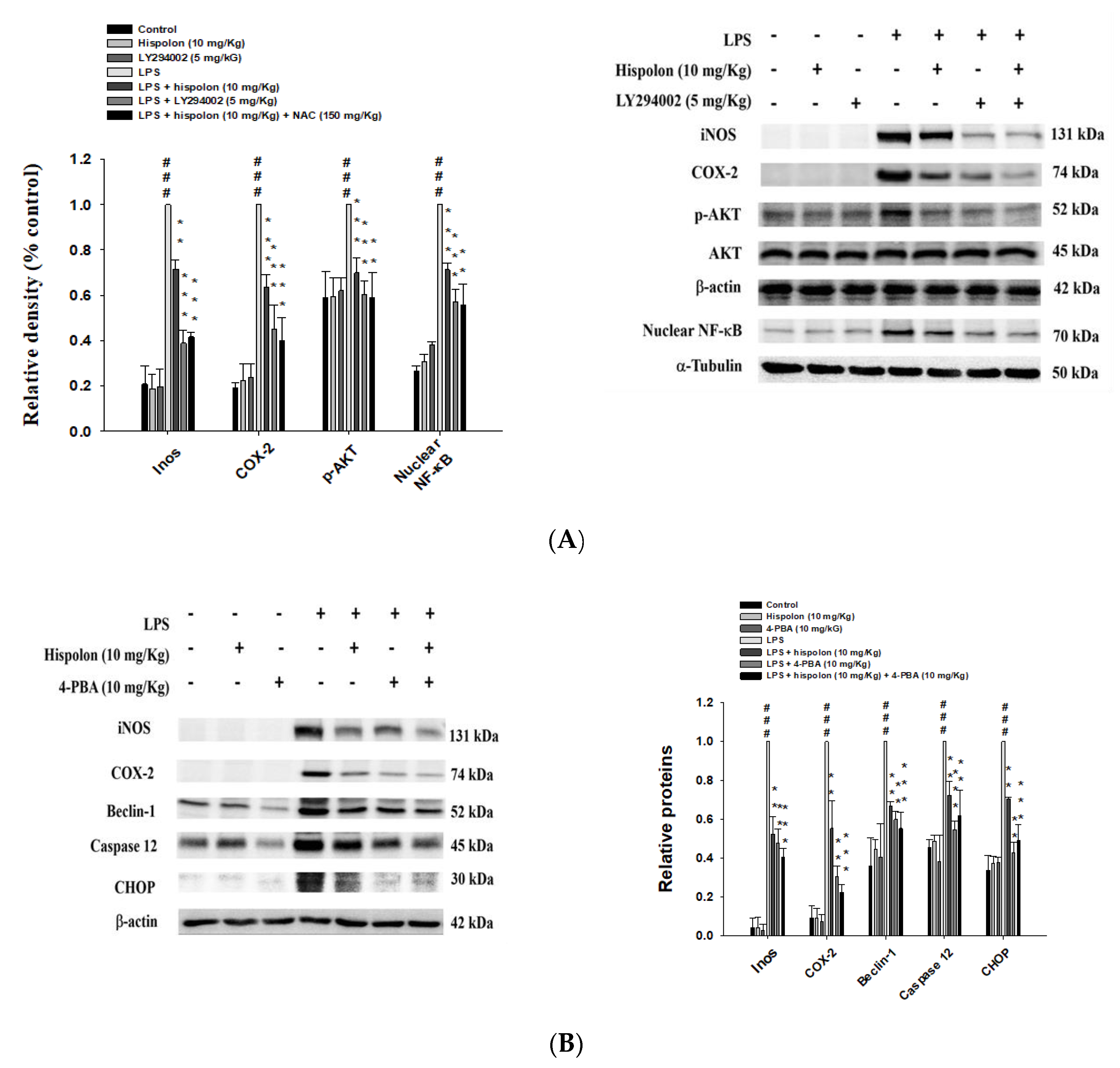
3.8. Suppression of TLR4/PI3K/Akt/mTOR Axis
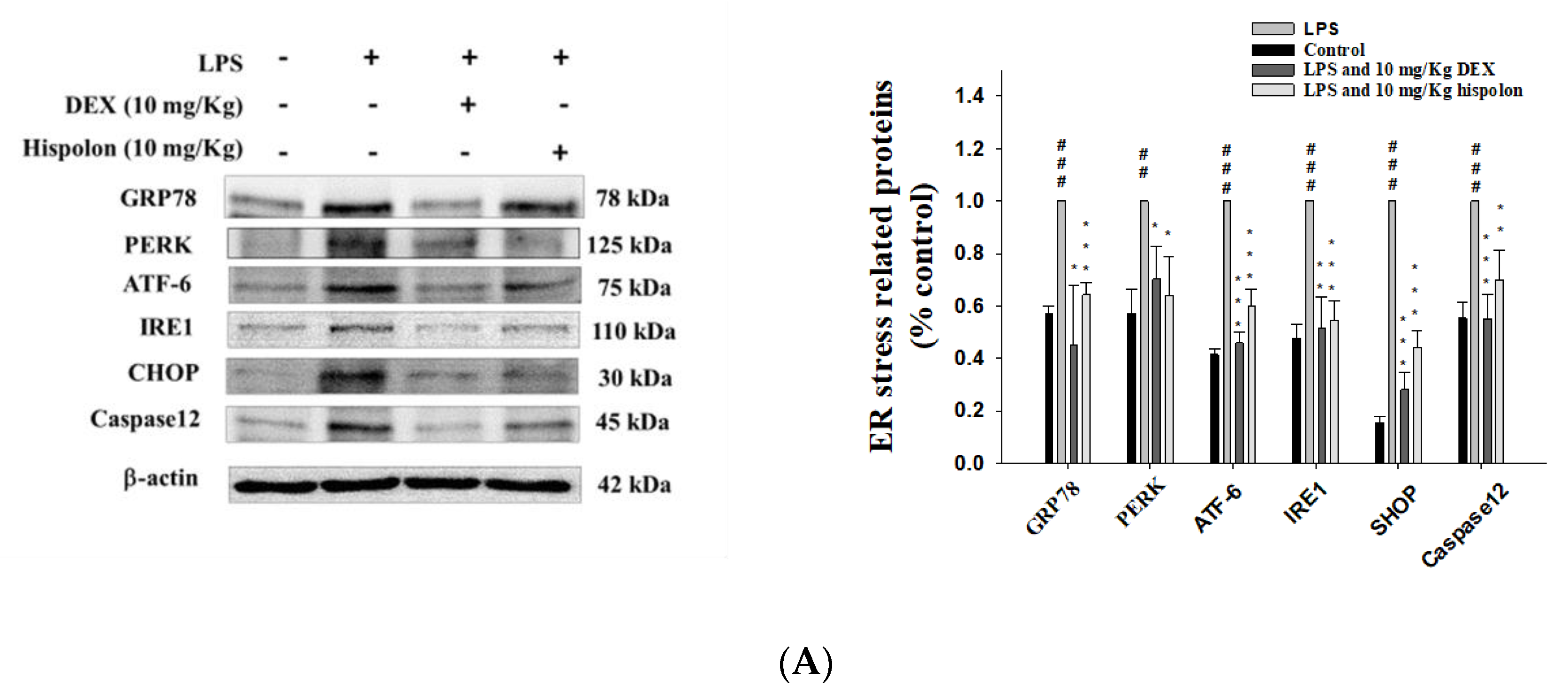
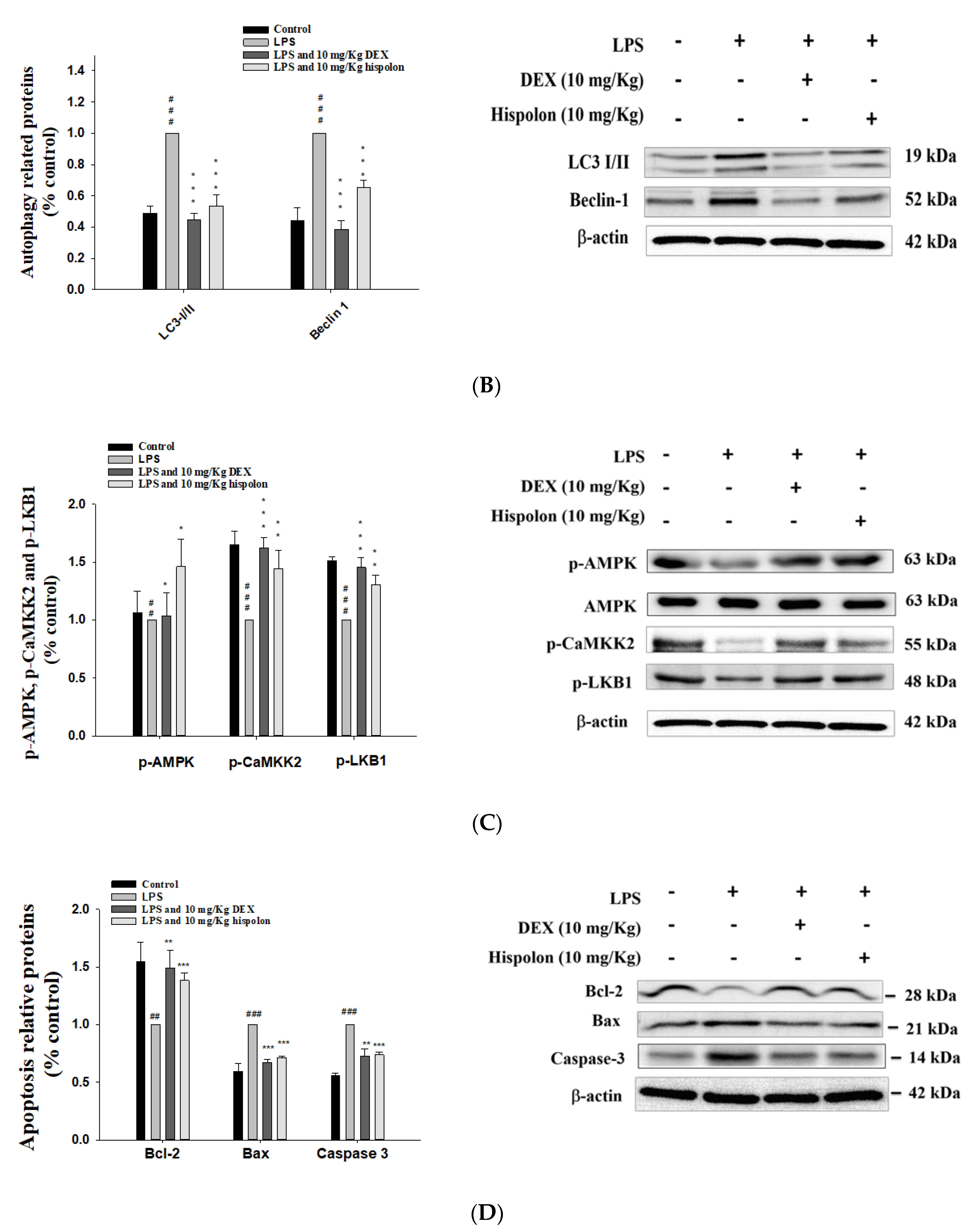
3.9. Hispolon Reduces ER Stress responses and autophagy inhibition
3.10. Hispolon Decreases LKB1/CaMKK–AMPK Signaling
3.11. Downregulation of Apoptosis-Related Proteins
3.12. Hispolon and NAC Reduce the Expression of the AMPK and Nrf-2 Proteins
3.13. Hispolon and an AKT inhibitor (LY294002) Reduce the Inflammatory Related Proteins
3.14. Hispolon and an ER Stress Inhibitor Reduces the Expression of Inflammatory and ER Stress Related Proteins
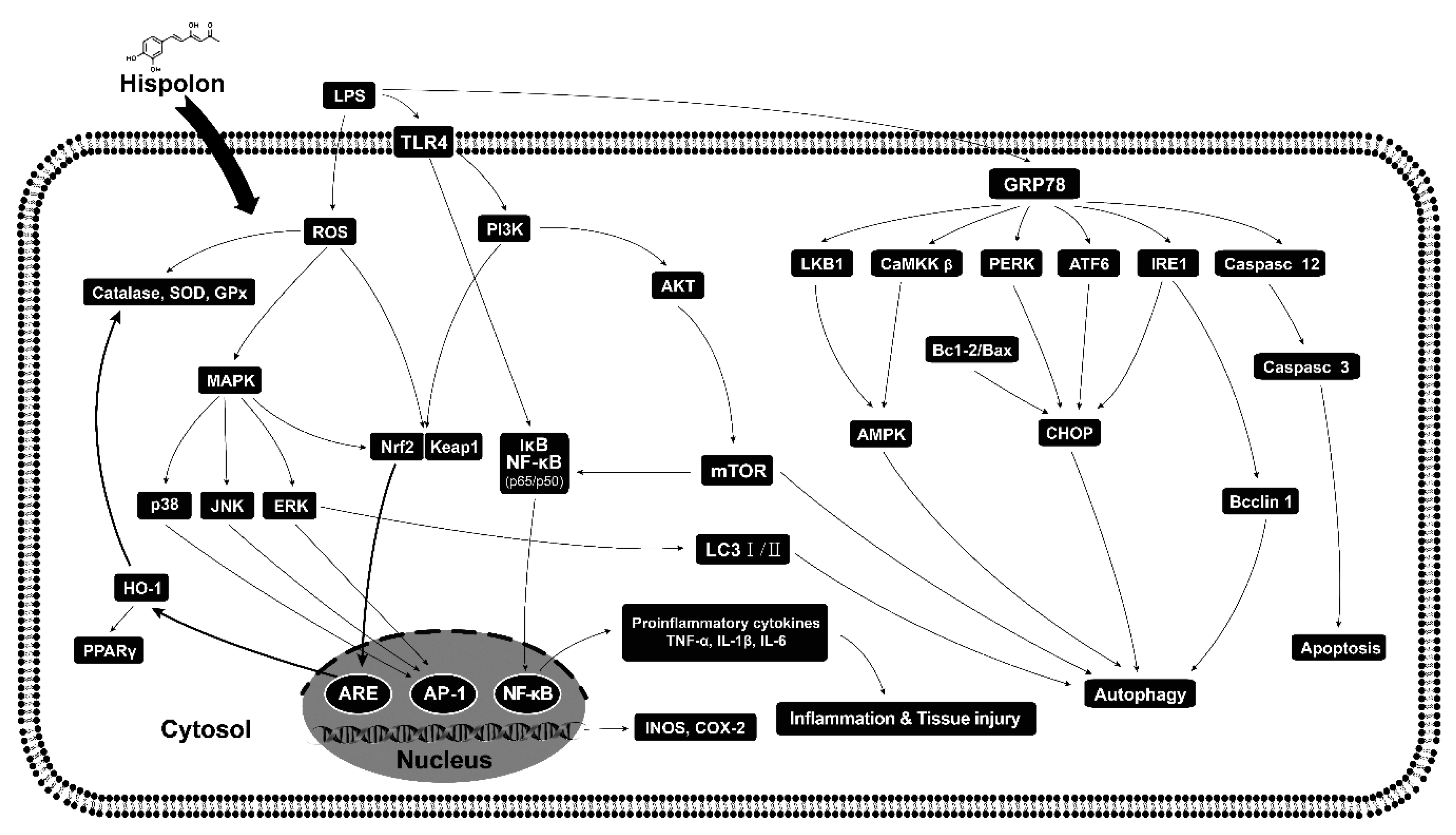
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Park, J.; Chen, Y.; Zheng, M.; Ryu, J.; Cho, G.J.; Surh, Y.J.; Sato, D.; Hamada, H.; Ryter, S.W.; Kim, U.H.; et al. Pterostilbene 4′-β-glucoside attenuates LPS-induced acute lung injury via induction of heme oxygenase-1. Oxid. Med. Cell. Longev. 2018, 2018, 2747018. [Google Scholar] [CrossRef] [PubMed]
- Ware, L.B.; Matthay, M.A. The acute respiratory distress syndrome. N. Eng. J. Med. 2000, 342, 1334–1349. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. Toll-like receptors in innate immunity. Int. Immunol. 2005, 17, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.C.; Deng, J.S.; Huang, S.S.; Wu, S.H.; Chen, C.C.; Lin, W.R.; Lin, H.Y.; Huang, G.J. Anti-Inflammatory Activity of Sanghuangporus Sanghuang Mycelium. Int. J. Mol. Sci. 2017, 18, 347. [Google Scholar] [CrossRef] [PubMed]
- Keppler-Noreuil, K.M.; Parker, V.E.R.; Darling, T.N.; Martinez-Agosto, J.A. Somatic overgrowth disorders of the PI3K/AKT/mTOR pathway & therapeutic strategies. Am. J. Med. Genet. C Semin. Med. Genet. 2016, 172, 402–421. [Google Scholar] [PubMed]
- Thimmulappa, R.K.; Lee, H.; Rangasamy, T.; Reddy, S.P.; Yamamoto, M.; Kensler, T.W.; Biswal, S. Nrf2 is a critical regulator of the innate immune response and survival during experimental sepsis. J. Clin. Investig. 2006, 116, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Zhang, W.; Wang, J.; Yang, H.; Zhao, X.; Zhou, Q.; Wang, H.; Li, L.; Du, G. Activation of Nrf2 signaling by salvianolic acid C attenuates NF-κB mediated inflammatory response both in vivo and in vitro. Int. Immunopharmacol. 2018, 63, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.Y.; Chen, Y.C.; Lin, J.G.; Lin, I.H.; Huang, H.F.; Yeh, C.C.; Chen, J.J.; Huang, G.J. Asatone prevents acute lung injury by reducing expressions of NF-κB, MAPK and inflammatory cytokines. Am. J. Chin. Med. 2018, 46, 651–671. [Google Scholar] [CrossRef] [PubMed]
- Jadeja, R.N.; Upadhyay, K.K.; Devkar, R.V.; Khurana, S. Naturally occurring Nrf2 activators: Potential in treatment of liver injury. Oxid. Med. Cell. Longev. 2016, 2016, 3453926. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef] [PubMed]
- Schönthal, A.H. Endoplasmic reticulum stress: Its role in disease and novel prospects for therapy. Scientifica 2012, 2012, 857516. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.F.; Li, X.H.; Yuan, Z.P.; Li, C.Y.; Tian, R.B.; Jia, W.; Xiao, Z.P. Allicin improves endoplasmic reticulum stress-related cognitive deficits via PERK/Nrf2 antioxidative signaling pathway. Eur. J. Pharmacol. 2015, 762, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Wang, L.; Wang, B.; Wang, T.; Yang, G.; Shen, L.; Wang, T.; Guo, X.; Liu, Y.; Xia, Y.; et al. Activation of volume-sensitive outwardly rectifying chloride channel by ROS contributes to ER stress and cardiac contractile dysfunction: Involvement of CHOP through Wnt. Cell Death Dis. 2014, 5, e1528. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Lee, A.S. Role of the unfolded protein response, GRP78 and GRP94 in organ homeostasis. J. Cell. Physiol. 2015, 230, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Cullinan, S.B.; Zhang, D.; Hannink, M.; Arvisais, E.; Kaufman, R.J.; Diehl, J.A. Nrf2 is a direct PERK substrate and effector of PERK-dependent cell survival. Mol. Cell. Biol. 2003, 23, 7198–7209. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Guo, L.; Qian, P.; Zhao, Y.; Liu, A.; Ji, F.; Chen, L.; Wu, X.; Qian, G. Lipopolysaccharide induces autophagic cell death through the PERK-dependent branch of the unfolded protein response in human alveolar epithelial A549 cells. Cell. Physiol. Biochem. 2015, 36, 2403–2417. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yan, H.; Dou, C.; Su, Y. Human leptin triggers proliferation of A549 cells via blocking endoplasmic reticulum stress-related apoptosis. Biochemistry (Moscow) 2013, 78, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, E.L.; Saftig, P. Autophagy: A lysosomal degradation pathway with a central role in health and disease. Biochim. Biophys. Acta 2009, 1793, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.H.; Ro, S.H.; Cao, J.; Otto, N.M.; Kim, D.H. mTOR regulation of autophagy. FEBS Lett. 2010, 584, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Jagannath, C.; Liu, X.D.; Sharafkhaneh, A.; Kolodziejska, K.E.; Eissa, N.T. Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 2007, 27, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011, 18, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Qiao, J.Y.; Wang, B.Y.; Bai, M.; Shen, J.D.; Cheng, Y.X. Paeoniflorin ameliorates fructose-induced insulin resistance and hepatic steatosis by activating LKB1/AMPK and AKT pathways. Nutrients 2018, 10, 1024. [Google Scholar] [CrossRef] [PubMed]
- Fogarty, S.; Ross, F.A.; Vara Ciruelos, D.; Gray, A.; Gowans, G.J.; Hardie, D.G. AMPK causes cell cycle arrest in LKB1-deficient cells via activation of CAMKK2. Mol. Cancer Res. 2016, 14, 683–695. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.Y.; Sheu, M.J.; Yang, C.H.; Lu, T.C.; Chang, Y.S.; Peng, W.H.; Huang, S.S.; Huang, G.J. Analgesic effects and the mechanisms of anti-inflammation of hispolon in mice. Evid. Based Complement. Altern. Med. Res. 2011, 2011, 478246. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.J.; Deng, J.S.; Chiu, C.S.; Liao, J.C.; Hsieh, W.T.; Sheu, M.J.; Wu, C.H. Hispolon protects against acute liver damage in the rat by inhibiting lipid peroxidation, proinflammatory cytokine, and oxidative stress and downregulating the expressions of iNOS, COX-2, and MMP-9. Evid. Based Complement. Altern. Med. Res. 2012, 2012, 480714. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Chang, H.Y.; Deng, J.S.; Chen, J.J.; Huang, S.S.; Lin, I.H.; Kuo, W.L.; Chao, W.; Huang, G.J. Hispolon from Phellinus linteus induces G0/G1 cell cycle arrest and apoptosis in NB4 human leukaemia cells. Am. J. Chin. Med. 2013, 41, 1439–1457. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.J.; Deng, J.S.; Huang, S.S.; Hu, M.L. Hispolon induces apoptosis and cell cycle arrest of human hepatocellular carcinoma Hep3B cells by modulating ERK phosphorylation. J. Agric. Food Chem. 2011, 59, 7104–7113. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.J.; Yang, C.M.; Chang, Y.S.; Amagaya, S.; Wang, H.C.; Hou, W.C.; Huang, S.S.; Hu, M.L. Hispolon suppresses SK-Hep1 human hepatoma cell metastasis by inhibiting matrix metalloproteinase-2/9 and urokinase-plasminogen activator through the PI3K/Akt and ERK signaling pathways. J. Agric. Food Chem. 2010, 58, 9468–9475. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.S.; Chien, C.C.; Cheng, K.T.; Subbaraju, G.V.; Chen, Y.C. Hispolon Suppresses LPS- or LTA-Induced iNOS/NO Production and Apoptosis in BV-2 Microglial Cells. Am. J. Chin. Med. 2017, 45, 1649–1666. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Deng, J.S.; Chang, Y.S.; Huang, G.J. Ginsenoside Rh2 ameliorates lipopolysaccharide-induced acute lung injury by regulating the TLR4/PI3K/Akt/mTOR, Raf-1/MEK/ERK, and Keap1/Nrf2/HO-1 signaling pathways in mice. Nutrients 2018, 10, 1208. [Google Scholar]
- Li, P.Y.; Liang, Y.C.; Sheu, M.J.; Huang, S.S.; Chao, C.Y.; Kuo, Y.H.; Huang, G.J. Alpinumisoflavone attenuates lipopolysaccharide-induced acute lung injury by regulating the effects of anti-oxidation and anti-inflammation both in vitro and in vivo. RSC Advances 2018, 8, 31515–31528. [Google Scholar] [CrossRef]
- Hsieh, Y.H.; Deng, J.S.; Pan, H.P.; Liao, J.C.; Huang, S.S.; Huang, G.J. Sclareol ameliorate lipopolysaccharide-induced acute lung injury through inhibition of MAPK and induction of HO-1 signaling. Int. Immunopharmacol. 2017, 44, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, G.; Zhou, S. Protective effects of isoliquiritigenin on LPS-induced acute lung injury by activating PPAR-γ. Inflammation 2018, 41, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, X.; Ma, C.; Li, Z.; Chen, H.; Zhang, Z.; Li, T. Genipin protects rats against lipopolysaccharide-induced acute lung injury by reinforcing autophagy. Int. Immunopharmacol. 2019, 72, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Sundararaman, A.; Amirtham, U.; Rangarajan, A. Calcium-oxidant signaling network regulates AMP-activated protein kinase (AMPK) activation upon matrix deprivation. J. Biol. Chem. 2016, 291, 14410–14429. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; He, Z.; Wang, X.; Pineda, M.; Chen, R.; Liu, H.; Ma, K.; Shen, H.; Wu, C.; Huang, N.; et al. Apigenin C-glycosides of microcos paniculata protects lipopolysaccharide induced apoptosis and inflammation in acute lung injury through TLR4 signaling pathway. Free Radic. Biol. Med. 2018, 124, 163–175. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Shi, W.; Yu, M.; Li, X.; Xu, J.; Zhu, J.; Jin, L.; Xie, W.; Kong, H. Nicorandil attenuates LPS-induced acute lung injury by pulmonary endothelial cell protection via NF-κB and MAPK Pathways. Oxid. Med. Cell Longev. 2019, 2019, 4957646. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.S.; Zhao, Z.; Zhu, H.P. Hispolon inhibits TPA-induced invasion by reducing MMP-9 expression through the NF-κB signaling pathway in MDA-MB-231 human Breast cancer cells. Oncol. Lett. 2015, 10, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, Y.C.; Park, B. Hispolon from Phellinus linteus induces apoptosis and sensitizes human cancer cells to the tumor necrosis factor-related apoptosis-inducing ligand through upregulation of death receptors. Oncol. Rep. 2016, 35, 1020–1026. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hsiao, P.C.; Hsieh, Y.H.; Chow, J.M.; Yang, S.F.; Hsiao, M.; Hua, K.T.; Lin, C.H.; Chen, H.Y.; Chien, M.H. Hispolon induces apoptosis through JNK1/2-mediated activation of a caspase-8, -9, and -3-dependent pathway in acute myeloid leukemia (AML) cells and inhibits AML xenograft tumor growth in vivo. J. Agric. Food Chem. 2013, 61, 10063–10073. [Google Scholar] [CrossRef] [PubMed]
- Shyamsundar, M.; McKeown, S.T.; O’Kane, C.M.; Craig, T.R.; Brown, V.; Thickett, D.R.; Matthay, M.A.; Taggart, C.C.; Backman, J.T.; Elborn, J.S.; et al. Simvastatin decreases lipopolysaccharide-induced pulmonary inflammation in healthy volunteers. Am. J. Respir. Crit. Care Med. 2009, 179, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.J.; Chien, S.Y.; Chou, Y.E.; Chen, C.J.; Chen, J.; Chen, M.K. Hispolon from Phellinus linteus possesses mediate caspases activation and induces human nasopharyngeal carcinomas cells apoptosis through ERK1/2, JNK1/2 and p38 MAPK pathway. Phytomedicine 2014, 21, 1746–1752. [Google Scholar] [CrossRef] [PubMed]
- Hsin, M.C.; Hsieh, Y.H.; Wang, P.H.; Ko, J.L.; Hsin, I.L.; Yang, S.F. Hispolon suppresses metastasis via autophagic degradation of cathepsin S in cervical Cancer Cells. Cell Death Dis. 2017, 8, e3089. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wong, Y.S. Selenocystine induces S-phase arrest and apoptosis in human breast adenocarcinoma MCF-7 cells by modulating ERK and Akt phosphorylation. J. Agric. Food Chem. 2008, 56, 10574–10581. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, A.S.; Hagan, S.; Rath, O.; Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 2007, 26, 3279–3290. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Bi, D.; Zheng, R. Identification and activation of TLR4-mediated signalling pathways by alginate-derived guluronate oligosaccharide in RAW264.7 Macrophages. Sci. Rep. 2017, 7, 1663. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; Kostenko, S.; Sveinbjørnsson, B. The role of mitogen-activated protein kinase-activated protein kinases (MAPKAPKs) in inflammation. Genes (Basel) 2013, 4, 101–133. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Liu, H.; Song, X.; Ling, Y.; He, S.; Yan, Y.; Yan, J.; Wang, S.; Wang, X.; Chen, A. Honokiol post-treatment ameliorates myocardial ischemia/reperfusion injury by enhancing autophagic flux and reducing intracellular ROS production. Chem. Biol. Interact. 2019, 307, S0009–2797(19)30279-0. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Sang, W.; Chen, S.; Chen, R.; Zhang, H.; Xue, F.; Li, Z.; Liu, Y.; Gong, Y.; Zhang, H.; et al. 4-PBA inhibits LPS-induced inflammation through regulating ER stress and autophagy in acute lung injury models. Toxicol. Lett. 2017, 271, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Chiu, T.L.; Su, C.C. Tanshinone IIA increases protein expression levels of PERK, ATF6, IRE1α, CHOP, caspase-3 and caspase-12 in pancreatic cancer BxPC-3 cell-derived xenograft tumors. Mol. Med. Rep. 2017, 15, 3259–3263. [Google Scholar] [CrossRef] [PubMed]
- Maamoun, H.; Benameur, T.; Pintus, G.; Munusamy, S.; Agouni, A. Crosstalk between oxidative stress and endoplasmic reticulum (ER) stress in endothelial dysfunction and aberrant angiogenesis associated with diabetes: A focus on the protective roles of heme oxygenase (HO)-1. Front. Physiol. 2019, 11, 70. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.S.; Lee, S.M.; Lin, C.C.; Liu, C.Y. Hispolon decreases melanin production and induces apoptosis in melanoma cells through the downregulation of tyrosinase and microphthalmia-associated transcription factor (MITF) expressions and the activation of caspase-3, -8 and -9. Int. J. Mol. Sci. 2014, 15, 1201–1215. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.M.; Min, K.J.; Kwon, T.K. Involvement of up-regulation of death receptors and Bim in hispolon-mediated TNF-related apoptosis-inducing ligand sensitization in human renal carcinoma. J. Cancer Prev. 2019, 24, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhang, L.; Yu, L.; Han, L.; Ji, W.; Shen, H.; Hu, Z. Time-dependent changes of autophagy and apoptosis in lipopolysaccharide-induced rat acute lung injury. Iran. J. Basic Med. Sci. 2016, 19, 632–637. [Google Scholar] [PubMed]
- Wang, H.; Zuo, X.; Wang, Q.; Yu, Y.; Xie, L.; Wang, H.; Wu, H.; Xie, W. Nicorandil inhibits hypoxia-induced apoptosis in human pulmonary artery endothelial cells through activation of mitoKATP and regulation of eNOS and the NF-κB pathway. Int. J. Mol. Med. 2013, 32, 187–194. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bonneau, B.; Prudent, J.; Popgeorgiev, N.; Gillet, G. Non-apoptotic roles of Bcl-2 family: The calcium connection. Biochim. Biophys. Acta 2013, 1833, 1755–1765. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.-Y.; Deng, J.-S.; Huang, W.-C.; Jiang, W.-P.; Huang, G.-J. Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/mTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy. Nutrients 2020, 12, 1742. https://doi.org/10.3390/nu12061742
Huang C-Y, Deng J-S, Huang W-C, Jiang W-P, Huang G-J. Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/mTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy. Nutrients. 2020; 12(6):1742. https://doi.org/10.3390/nu12061742
Chicago/Turabian StyleHuang, Ching-Ying, Jeng-Shyan Deng, Wen-Chin Huang, Wen-Ping Jiang, and Guan-Jhong Huang. 2020. "Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/mTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy" Nutrients 12, no. 6: 1742. https://doi.org/10.3390/nu12061742
APA StyleHuang, C.-Y., Deng, J.-S., Huang, W.-C., Jiang, W.-P., & Huang, G.-J. (2020). Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Hispolon in Mice, Through Regulating the TLR4/PI3K/Akt/mTOR and Keap1/Nrf2/HO-1 Pathways, and Suppressing Oxidative Stress-Mediated ER Stress-Induced Apoptosis and Autophagy. Nutrients, 12(6), 1742. https://doi.org/10.3390/nu12061742





