Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days
Abstract
1. Introduction
1.1. Functions
1.2. Choline, the Brain, and Neurons
1.3. Accruement
1.4. Choline Requirements and Dietary Intakes
1.5. Food Sources
1.6. Choline Knowledge Gaps
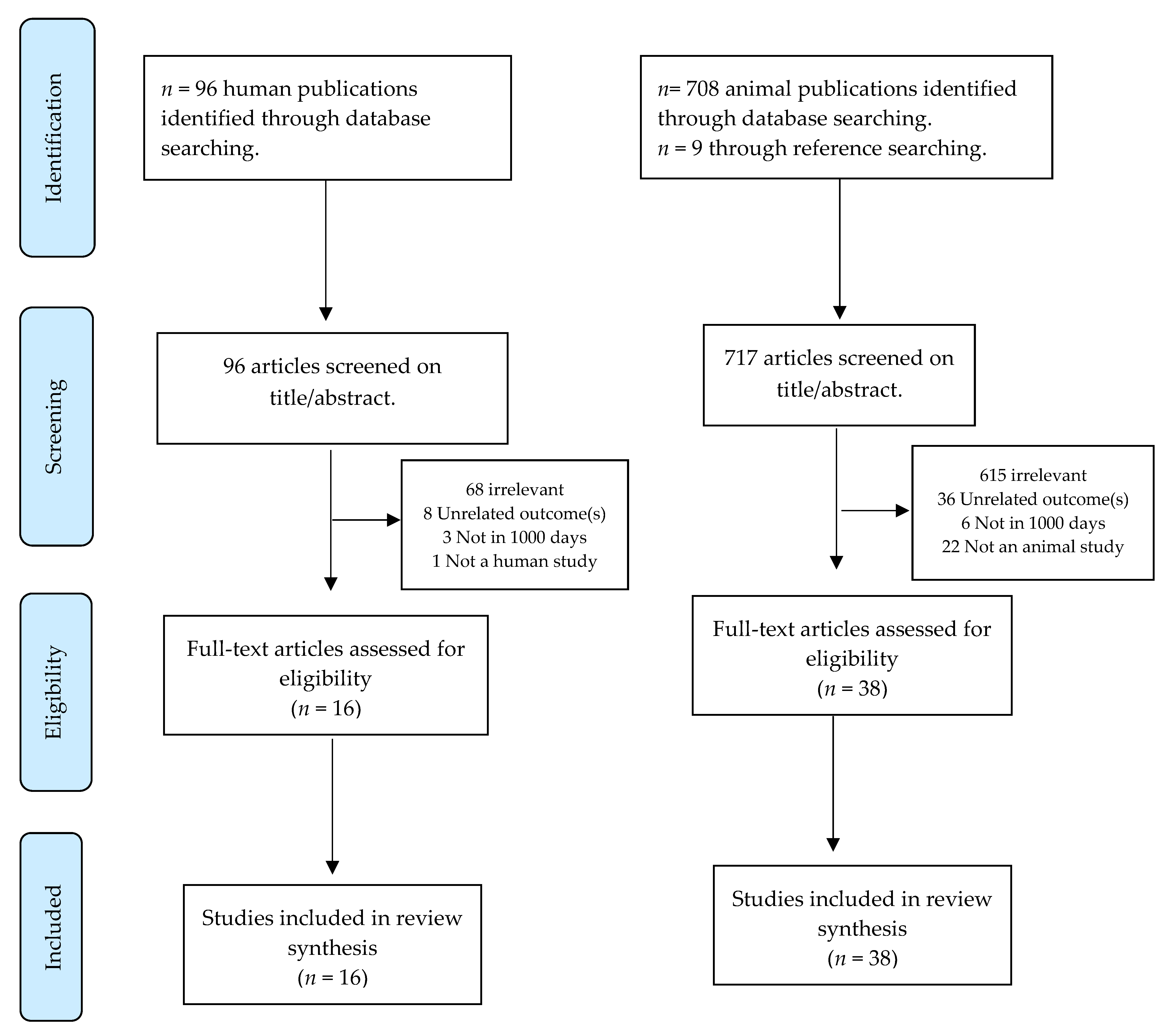
2. Materials and Methods
2.1. Approach
2.2. Defining 1000 Days
2.3. Search Strategy
2.4. Study Quality & Data Extraction
3. Results
3.1. Animal Studies
3.1.1. Brain Development
3.1.2. Cognition and Memory
3.1.3. Protection from Environmental Exposures
3.1.4. Choline Supply in Animal Models of Neurodegeneration
| Study (Author, Year, Country) | Animal Study | Stage of 1000 Days | Study Methodology | Study Outcomes | Main Findings |
|---|---|---|---|---|---|
| Alldred et al. (2019) [82] USA | Mouse model of DS and AD | Pregnancy | Perinatal choline supplementation. | Neuron gene expression | Maternal choline supplementation increased offspring gene expression. |
| Chin et al. (2019) [110] Singapore | Knockout mice | Postnatal | Fed 13 mg/day (1.7 × required daily intake) of choline | Motor co-ordination, behavioral deficits, anxiety | Choline modulated neuronal plasticity, leading to behavioral changes and showing potential to treat RTT. |
| Kelley et al. (2019) [83] USA | Mouse model of DS and AD | Pregnancy | Dams on an MCS diet or a normal choline diet from mating until weaning, | Neuron gene expression | Significant downregulation was seen in select transcripts that were normalized with MCS. |
| Moreno & Brugada et al. (2019) [96] Spain | Rat model | Pregnancy | Fed with 1.1 g choline/Kg food or 5 g choline/Kg food between embryonic days (E) 12 and E18. | Long-term memory | Prenatal supplementation with choline accelerates the development of long-term memory in rats. |
| Sawant et al. (2019) [98] USA | Sheep model | Pregnancy (1st Trimester) | Randomized to six difference ethanol/choline groups. | Alcohol-induced fetal cranio-facial abnormalities | Maternal choline supplementation mitigated most alcohol-induced effects. |
| Velazquez et al. (2019) [107] USA | Mouse model of AD | Transgenerational effects of MCS | Exposed mice to MCS and bred for two generations. | Alzheimer’s disease, brain homocysteine | Providing MCS reduced AD pathology across two generations. |
| Alldred et al. (2018) [106] USA | Mouse model of DS and AD | Pregnancy | Provided with MCS. | Neuron gene expression | MCS reprogrammed transcripts involved in neuronal signaling. |
| Kennedy et al. (2018) [89] USA | Male rat pups with ID | Pregnancy, nursing and early life | Choline (5 ppm) was given to half the nursing dams and weanlings | Cognitive performance, novel object recognition | Recognition memory deficits induced by early-life iron deficiency was prevented by postnatal choline supplementation. |
| Mudd et al. (2018) [78] USA | Pigs (Yorkshire sows) | Pregnancy and nursing | Choline-sufficient or choline-deficient diet or milk supply | Brain white and grey matter | Prenatal choline deficiency greatly alters grey and white matter development in pigs. No postnatal effects were observed. |
| Balaraman et al. (2017) [100] USA | Sprague-Dawley rats | Postnatal (+ethanol exposure) | Treated with choline chloride (100 mg/kg/day) or saline | Hippocampal microRNA alterations | Choline supplementation can normalize disturbances in miRNA expression following developmental alcohol exposure. |
| Idrus et al. (2017) [99] USA | Rat model | Pregnant | Received diets containing 40, 70, or 100% recommended choline levels | Motor development, co-ordination | Subjects exposed to ethanol and fed the low 40% choline diet had delayed eye openings, poor hind limb coordination, and were overactive compared to all other groups. |
| Mellott et al. (2017) [111] USA | Rat model | Pregnancy and nursing | Fed a diet containing 1.1 g/kg of choline or a choline-supplemented (5 g/kg) diet. | Amyloidosis, hippocampal choline acetyltransferase expression | The choline group had reduced levels of solubilized amyloid peptides and plaques; preserved levels of choline acetyltransferase protein and absence of astrogliosis indicating a role in AD prevention. |
| Birch et al. (2016) [105] USA | Sheep model | First trimester (alcohol binge) | Randomly assigned to: HBA HBC (2.5 g/kg ethanol and 10 mg/kg/day choline), saline control, saline control plus choline (10 mg/kg/day choline), and normal control. | Brain volume | Maternal choline supplementation comparable to doses in human studies fails to prevent brain volume reductions typically induced by first-trimester binge alcohol exposure. |
| Kelley et al. (2016) [108] USA | Mouse model of DS | Pregnancy | Provided with MCS. | Hippocampal function | Maternal choline supplementation increased innervation and improved hippocampal function. |
| Mudd et al. (2016) [77] USA | Piglets from Sows | Perinatal period | Choline-sufficient or choline-deficient diet or milk supply | Brain development | Prenatal choline deficiency had profound effects by delaying neurodevelopment as evidenced by reduced concentrations of glycerophosphocholine-phosphocholine, brain volumes and region-specific volumes. |
| Tran et al. (2016) [97] USA | Rat model | Pregnant and nursing dams fed an ID diet | Choline (5 g/kg) was given to half the pregnant dams in each group | Hippocampal function | Choline supplementation reduced the effects of ID, including those on gene networks associated with autism and schizophrenia. |
| Wang et al. (2016) [80] USA | Mouse model | Pregnancy | Fed either control or low-choline diets | Cortical development | Low choline supply reduced the number of 2 types of cortical neural progenitor cells, radial glial cells and intermediate progenitor cells in fetal brains (p < 0.01). |
| Zhu et al. (2016) [81] China | Rat model | Pregnant | Fed a normal or low-protein diet containing sufficient choline (1.1 g/kg choline chloride) or supplemented choline (5.0 g/kg choline chloride) until delivery | Spatial learning deficits | Prenatal choline supplementation reversed the increased width of the synaptic cleft (p < 0.05) and decreased the curvature of the synaptic interface (p < 0.05) induced by a low-protein diet. |
| Bearer et al. (2015) [102] USA | Mouse model | Pregnant | Maintained a choline-deficient diet and the 1 of 8 treatments | Balance and co-ordination | Choline alleviated ethanol-induced effects on balance and co-ordination. |
| Getty et al. (2015) [79] USA | Sows | Pregnancy and nursing | Fed a choline deficient or sufficient diet and milk supply | Brain development | The brains of piglets exposed to prenatal choline deficiency were significantly smaller than those of choline sufficient piglets. |
| Jadavji et al. (2015) [90] USA | Mouse model | Pregnancy and nursing | Effect of maternal choline deficiency | Short-term memory, apoptosis | There were short-term memory deficits in the offspring of dams with dietary deficiencies of critical methyl donors, i.e., choline. |
| Langley et al. (2015) [91] USA | Mouse model of autism | Pregnancy and nursing | Fed a control or choline-supplemented diet from mating | Social interaction and anxiety | High choline intake during early development reduced deficits in social behavior and anxiety in an autistic mouse model. |
| Ash et al. (2014) [84] USA | Mouse model of DS | Pregnancy and nursing | Assigned to a choline sufficient (1.1 g/kg choline chloride) or choline supplemented (5.0 g/kg choline chloride) diet. | Basal forebrain cholinergic neuron number and size | Maternal choline supplementation significantly improved spatial mapping and increased number, density, and size of MS BFCNs in DS offspring. |
| Kelley et al. (2014) USA | Mouse model of DS | Pregnancy | Studied effects of MCS | Basal forebrain cholinergic system | MCS partially normalized the BCFN system. |
| Kennedy et al. (2014) [92] USA | Male rat pups with ID | Pregnancy | Provided with choline supplementation (5 g/kg choline chloride, E11-18) or control | Neurobehavioral effects | Prenatal choline supplementation in formerly ID rats restored novel object recognition and increased hippocampal gene expression. |
| Yan et al. (2014) [109] USA | Mouse model of DS and AD | Pregnancy | Previously mothers had been choline-supplemented | PEMT pathway | Maternal choline supplementation upregulates PEMT pathway and d9 choline metabolites in the brain. |
| Schulz et al. (2014) [112] USA | Rat model | Pregnancy and nursing | Control and stressed dams were fed choline-supplemented or control chow | Anxiety | Perinatal choline supplementation mitigated prenatal stress-induced social behavioral deficits in males. |
| Bekdash et al. (2013) [103] USA | Rat model | Pregnancy | Fed an alcohol-containing liquid diet or control diet with or without choline | Neuronal function | Gestational choline supplementation prevents the adverse effects of ethanol on neurons. |
| Moreno et al. (2013) [93] Spain | Rat model | Pregnancy | Fed choline-deficient (0 g/kg choline chloride), standard (1.1 g/kg choline chloride), or choline-supplemented (5 g/kg choline chloride) diets | Memory | The supplemented group exhibited improved memory compared with both the standard and the deficient group. |
| Velazquez et al. (2013) [86] USA | Mouse model of DS and AD | Pregnancy | Fed additional choline (4.5x more than normal) | Hippocampal neurogenesis | MCS partially normalized adult hippocampal neurogenesis. |
| Corriveau & Glenn (2012) [94] USA | Rat model | Postnatal | Rats fed a choline-supplemented, -deficient, or standard diet | Cognitive functioning | Choline deficiency impaired memory in rats that were stressed prenatally. |
| Monk et al. (2012) [104] USA | Rat model | Perinatal | Injected with choline chloride (100 mg/kg/day) or saline vehicle | Hippocampal cholinergic development | Perinatal choline supplementation can attenuate alcohol-related behavioral changes by influencing cholinergic systems. |
| Otero et al. (2012) [113] USA | Rat model | Neonatal period | Choline or saline administered subcutaneously | DNA methylation in the hippocampus and prefrontal cortex | Alcohol exposure induced hypermethylation in these regions with was significantly reduced after choline supplementation. |
| Wong-Goodrich et al. (2011) [114] USA | Rat model | Prenatal | Received either a control or choline supplemented diet | Long-term cognitive and neuropathological effects | Prenatal choline supplementation promoted long-term hippocampal recovery from seizures in adulthood. |
| Mehedint et al. (2010a) [88] USA | Mouse model | Pregnancy | C57BL/6 mice were fed either a choline-deficient, control or choline-supplemented diet. | Angiogenesis | Maternal dietary choline intake altered angiogenesis in the developing fetal hippocampus. |
| Mehedint et al. (2010b) [87] USA | Mouse model | Pregnancy | C57BL/6 mice were fed either a choline-deficient, control or choline-supplemented diet. | Methylation and epigenetic marking | Choline deficiency altered histone methylation in neural progenitor cells which appears to underlie the observed changes in neurogenesis. |
| Moon et al. (2010) [95] USA | Mouse model of DS and AD | Perinatal | Choline-supplemented Ts65Dn dams | Cognitive functioning | Perinatal choline supplementation may lessen cognitive dysfunction in DS and reduce cognitive decline in related disorders such as AD. |
| Thomas et al. (2010) [101] USA | Rat model | Pregnant | Intubated with either 250 mg/kg/day choline chloride or vehicle | Working memory, behavior | Choline supplementation during prenatal alcohol exposure may reduce the severity of fetal alcohol effects, particularly on alterations in tasks that require behavioral flexibility. |
3.2. Human Studies
3.2.1. Fetal and Infant Neurodevelopment
3.2.2. Protection from Infections and Alcohol Exposure
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s Disease |
| AI | Adequate Intake |
| BFCN | Basal Forebrain Cholinergic Neuron |
| CHRNA7 | Cholinergic Receptor Nicotinic Alpha 7 Subunit |
| DMG | Dimethylglycine |
| DNA | Deoxyribonucleic acid |
| DS | Down Syndrome |
| EFSA | European Food Safety Authority |
| FAS | Fetal Alcohol Syndrome |
| ID | Iron Deficiency |
| MCS | Maternal Choline Supplementation |
| MTHFD | Methylenetetrahydrofolate Dehydrogenase |
| NHANES | National Health and Nutrition Examination Survey |
| NPTDCHOs | Neural Progenitor Cells |
| NTDs | Neural Tube Defects |
| PEMT | Phosphatidylethanolamine N-Methyltransferase. |
| PtdCho | Phosphatidylcholine |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| RCTs | Randomized Controlled Trials. |
| SNP | Single nucleotide polymorphisms |
References
- González, H.F.; Visentin, S. Micronutrients and neurodevelopment: An update. Arch Argent. Pediatr. 2016, 114, 570–575. [Google Scholar] [PubMed]
- Cusick, S.E.; Georgieff, M.K. The role of nutrition in brain development: The golden opportunity of the “first 1000 days”. J. Pediatr. 2016, 175, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Natker, E.; Zeisel, S.H.; Zelman, K.M. Choline: The underconsumed and underappreciated essential nutrient. Nutr. Today 2018, 53, 240–253. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Blusztajn, J.K.; Mellott, T.J. Choline nutrition programs brain development via DNA and histone methylation. Cent. Nerv. Syst. Agents Med. Chem. 2012, 12, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Muthukrishna, M.; Doebeli, M.; Chudek, M.; Henrich, J. The cultural brain hypothesis: How culture drives brain expansion, sociality, and life history. PLoS Comput. Biol. 2018, 14, e1006504. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.S.; Iannotti, L.L.; Raichle, M.E. Brain nutrition: A life span approach. Annu. Rev. Nutr. 2018, 38, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. The supply of choline is important for fetal progenitor cells. Semin. Cell Dev. Biol. 2011, 22, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Zeisel, S.H. Choline: The neurocognitive essential nutrient of interest to obstetricians and gynecologists. J. Diet Suppl. 2019, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Giri, S. Importance of choline as essential nutrient and its role in prevention of various toxicities. Prague Med. Rep. 2015, 116, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Caudill, M.A. Pre-and postnatal health: Evidence of increased choline needs. J. Am. Diet Assoc. 2010, 110, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary choline intake: Current state of knowledge across the life cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Fletcher, C. Trimethylamine N-oxide: Breathe new life. Br. J. Pharm. 2018, 175, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Filipcev, B.; Kojic, J.; Krulj, J.; Bodroza-Solarov, M.; Ilic, N. Betaine in cereal grains and grain-based products. Foods 2018, 7, 49. [Google Scholar] [CrossRef]
- Melnyk, S.; Fuchs, G.J.; Schulz, E.; Lopez, M.; Kahler, S.G.; Fussell, J.J.; Bellando, J.; Pavliv, O.; Rose, S.; Seidel, L.; et al. Metabolic imbalance associated with methylation dysregulation and oxidative damage in children with autism. J. Autism Dev. Disord. 2012, 42, 367–377. [Google Scholar] [CrossRef] [PubMed]
- James, S.J.; Melnyk, S.; Jernigan, S.; Cleves, M.A.; Halsted, C.H.; Wong, D.H.; Cutler, P.; Bock, K.; Boris, M.; Bradstreet, J.J.; et al. Metabolic endophenotype and related genotypes are associated with oxidative stress in children with autism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2006, 141, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Ikonomidou, C.; Bittigau, P.; Koch, C.; Genz, K.; Hoerster, F.; Felderhoff-Mueser, U.; Tenkova, T.; Dikranian, K.; Olney, J.W. Neurotransmitters and apoptosis in the developing brain. Biochem. Pharm. 2001, 62, 401–405. [Google Scholar] [CrossRef]
- Bernhard, W.; Poets, C.F.; Franz, A.R. Choline and choline-related nutrients in regular and preterm infant growth. Eur. J. Nutr. 2019, 58, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Van Echten-Deckert, G.; Alam, S. Sphingolipid metabolism-an ambiguous regulator of autophagy in the brain. Biol. Chem. 2018, 399, 837–850. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Dietary reference values for choline. EFSA J. 2016, 14, 4484. [Google Scholar]
- Zeisel, S.H.; Niculescu, M.D. Perinatal choline influences brain structure and function. Nutr. Rev. 2006, 64, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.L.; Wurtman, R.J. Brain acetylcholine: Control by dietary choline. Science 1976, 191, 561–562. [Google Scholar] [CrossRef] [PubMed]
- Freeman, J.J.; Jenden, D.J. The source of choline for acetylcholine synthesis in brain. Life Sci. 1976, 19, 949–961. [Google Scholar] [CrossRef]
- Blusztajn, J.K.; Wurtman, R.J. Choline and cholinergic neurons. Science 1983, 221, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Ganz, A.B.; Klatt, K.C.; Caudill, M.A. Common genetic variants alter metabolism and influence dietary choline requirements. Nutrients 2017, 9, 837. [Google Scholar] [CrossRef] [PubMed]
- Ganz, A.B.; Cohen, V.V.; Swersky, C.C.; Stover, J.; Vitiello, G.A.; Lovesky, J.; Chuang, J.C.; Shields, K.; Fomin, V.G.; Lopez, Y.S.; et al. Genetic variation in choline-metabolizing enzymes alters choline metabolism in young women consuming choline intakes meeting current recommendations. Int. J. Mol. Sci. 2017, 18, 252. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Jiang, X.; West, A.A.; Perry, C.A.; Malysheva, O.V.; Brenna, J.T.; Stabler, S.P.; Allen, R.H.; Gregory, J.F.; Caudill, M.A. Pregnancy alters choline dynamics: Results of a randomized trial using stable isotope methodology in pregnant and nonpregnant women. Am. J. Clin. Nutr. 2013, 98, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Bekdash, R.A. Choline, the brain and neurodegeneration: Insights from epigenetics. Front. Biosci. 2018, 23, 1113–1143. [Google Scholar] [CrossRef] [PubMed]
- Blusztajn, J.K.; Mellott, T.J. Neuroprotective actions of perinatal choline nutrition. Clin. Chem. Lab. Med. 2013, 51, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Akison, L.K.; Kuo, J.; Reid, N.; Boyd, R.N.; Moritz, K.M. Effect of choline supplementation on neurological, cognitive, and behavioral outcomes in offspring arising from alcohol exposure during development: A quantitative systematic review of clinical and preclinical studies. Alcohol Clin. Exp. Res. 2018, 42, 1591–1611. [Google Scholar] [CrossRef] [PubMed]
- Dubois, J.; Dehaene-Lambertz, G.; Kulikova, S.; Poupon, C.; Huppi, P.S.; Hertz-Pannier, L. The early development of brain white matter: A review of imaging studies in fetuses, newborns and infants. Neuroscience 2014, 276, 48–71. [Google Scholar] [CrossRef] [PubMed]
- Bekdash, R.A. Choline and the brain: An epigenetic perspective. Adv. Neurobiol. 2016, 12, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Growdon, J.H.; Wurtman, R.J.; Magil, S.G.; Logue, M. Normal plasma choline responses to ingested lecithin. Neurology 1980, 30, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Svennerholm, L.; Vanier, M.T. The distribution of lipids in the human nervous system. II. Lipid composition of human fetal and infant brain. Brain Res. 1972, 47, 457–468. [Google Scholar] [CrossRef]
- Meck, W.H.; Williams, C.L. Metabolic imprinting of choline by its availability during gestation: Implications for memory and attentional processing across the lifespan. Neurosci. Biobehav. Rev. 2003, 27, 385–399. [Google Scholar] [CrossRef]
- Zeisel, S.H. The fetal origins of memory: The role of dietary choline in optimal brain development. J. Pediatr. 2006, 149, S131–S136. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline: Essential for brain development and function. Adv. Pediatr. 1997, 44, 263–295. [Google Scholar]
- Yen, C.L.; Mar, M.H.; Meeker, R.B.; Fernandes, A.; Zeisel, S.H. Choline deficiency induces apoptosis in primary cultures of fetal neurons. FASEB J. 2001, 15, 1704–1710. [Google Scholar] [CrossRef]
- Holmes-McNary, M.Q.; Loy, R.; Mar, M.H.; Albright, C.D.; Zeisel, S.H. Apoptosis is induced by choline deficiency in fetal brain and in PC12 cells. Brain Res. Dev. Brain Res. 1997, 101, 9–16. [Google Scholar] [CrossRef]
- Albright, C.D.; Friedrich, C.B.; Brown, E.C.; Mar, M.H.; Zeisel, S.H. Maternal dietary choline availability alters mitosis, apoptosis and the localization of TOAD-64 protein in the developing fetal rat septum. Brain Res. Dev. Brain. Res. 1999, 115, 123–129. [Google Scholar] [CrossRef]
- McCann, J.C.; Hudes, M.; Ames, B.N. An overview of evidence for a causal relationship between dietary availability of choline during development and cognitive function in offspring. Neurosci. Biobehav. Rev. 2006, 30, 696–712. [Google Scholar] [CrossRef] [PubMed]
- Blusztajn, J.K.; Slack, B.E.; Mellott, T.J. Neuroprotective actions of dietary choline. Nutrients 2017, 9, 815. [Google Scholar] [CrossRef] [PubMed]
- Craciunescu, C.N.; Albright, C.D.; Mar, M.H.; Song, J.; Zeisel, S.H. Choline availability during embryonic development alters progenitor cell mitosis in developing mouse hippocampus. J. Nutr. 2003, 133, 3614–3618. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Nutritional importance of choline for brain development. J. Am. Coll. Nutr. 2004, 23, 621S–626S. [Google Scholar] [CrossRef] [PubMed]
- Radziejewska, A.; Chmurzynska, A. Folate and choline absorption and uptake: Their role in fetal development. Biochimie 2019, 158, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Wurtman, R.J. Developmental changes in rat blood choline concentration. Biochem. J. 1981, 198, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Ilcol, O.Y.; Uncu, G.; Ulus, I.H. Free and phospholipid-bound choline concentrations in serum during pregnancy, after delivery and in newborns. Arch. Physiol. Biochem. 2002, 110, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Intrauterine fatty acid accretion rates in human brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 121–129. [Google Scholar] [CrossRef]
- Evangelou, I.E.; du Plessis, A.J.; Vezina, G.; Noeske, R.; Limperopoulos, C. Elucidating metabolic maturation in the healthy fetal brain using 1H-MR spectroscopy. Ajnr. Am. J. Neuroradiol. 2016, 37, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Van der Aa, E.M.; Wouterse, A.C.; Peereboom-Stegeman, J.H.; Russel, F.G. Uptake of choline into syncytial microvillus membrane vesicles of human term placenta. Biochem. Pharm. 1994, 47, 453–456. [Google Scholar] [CrossRef]
- Sweiry, J.H.; Page, K.R.; Dacke, C.G.; Abramovich, D.R.; Yudilevich, D.L. Evidence of saturable uptake mechanisms at maternal and fetal sides of the perfused human placenta by rapid paired-tracer dilution: Studies with calcium and choline. J. Dev. Physiol. 1986, 8, 435–445. [Google Scholar] [PubMed]
- Bernhard, W.; Raith, M.; Kunze, R.; Koch, V.; Heni, M.; Maas, C.; Abele, H.; Poets, C.F.; Franz, A.R. Choline concentrations are lower in postnatal plasma of preterm infants than in cord plasma. Eur. J. Nutr. 2015, 54, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Ramel, S.E.; Georgieff, M.K. Preterm nutrition and the brain. World Rev. Nutr. Diet 2014, 110, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; Deoni, S. The development of brain white matter microstructure. Neuroimage 2018, 182, 207–218. [Google Scholar] [CrossRef] [PubMed]
- AMA. AMA Backs Global Health Experts in Calling Infertility a Disease; American Medical Association: Chicago, IL, USA, 2017. [Google Scholar]
- Schwarzenberg, S.J.; Georgieff, M.K.; Committee on Nutrition. Advocacy for improving nutrition in the first 1000 days to support childhood development and adult health. Pediatrics 2018, 141. [Google Scholar] [CrossRef] [PubMed]
- AAP. Food for Thought: AAP Aims to Ensure Kids Get Key Nutrients for Brain Development; Association of American Publishers, Inc.: Washington, DC, USA, 2018. [Google Scholar]
- Vennemann, F.B.; Ioannidou, S.; Valsta, L.M.; Dumas, C.; Ocke, M.C.; Mensink, G.B.; Lindtner, O.; Virtanen, S.M.; Tlustos, C.; D’Addezio, L.; et al. Dietary intake and food sources of choline in European populations. Br. J. Nutr. 2015, 114, 2046–2055. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Use of the EFSA comprehensive European food consumption database in exposure assessment. EFSA J. 2011, 9, 2097. [Google Scholar] [CrossRef]
- Wallace, T.C.; Fulgoni, V.L. Assessment of total choline intakes in the United States. J. Am. Coll. Nutr. 2016, 35, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Subhan, F.B.; Bell, R.C.; McCargar, L.J.; Curtis, J.M.; Jacobs, R.L.; Field, C.J.; Team, A.P. Estimation of choline intake from 24 h dietary intake recalls and contribution of egg and milk consumption to intake among pregnant and lactating women in Alberta. Br. J. Nutr. 2014, 112, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Patterson, K.Y.; Bhagwat, S.A.; Williams, J.R.; Howe, J.C.; Holden, J.M. USD Database for The Choline Content of Common Foods, Release 2. Agricultural Research Service; USDA: Montgomery, AL, USA, 2008.
- Derbyshire, E. Could we be overlooking a potential choline crisis in the United Kingdom? BMJ Nutr. Prev. Health 2019, 1–4. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Mar, M.H.; Howe, J.C.; Holden, J.M. Concentrations of choline-containing compounds and betaine in common foods. J. Nutr. 2003, 133, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Fenech, M.F.; Kim, P.J. Nutritionally recommended food for semi-to strict vegetarian diets based on large-scale nutrient composition data. Sci. Rep. 2018, 8, 4344. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Kosik, S.J.; Zhao, Y.Y.; Jacobs, R.L.; Curtis, J.M.; Field, C.J. Total choline and choline-containing moieties of commercially available pulses. Plant Foods Hum. Nutr. 2014, 69, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline: Critical role during fetal development and dietary requirements in adults. Annu. Rev. Nutr. 2006, 26, 229–250. [Google Scholar] [CrossRef] [PubMed]
- Korsmo, H.W.; Jiang, X.; Caudill, M.A. Choline: Exploring the growing science on its benefits for moms and babies. Nutrients 2019, 11, 1823. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific opinion on the substantiation of health claims related to choline and contribution to normal lipid metabolism (ID 3186), maintenance of normal liver function (ID 1501), contribution to normal homocysteine metabolism (ID 3090), maintenance of normal neurological function (ID 1502), contribution to normal cognitive function (ID 1502), and brain and neurological development (ID 1503) pursuant to article 13(1) of regulation (EC) no 1924/20061. EFSA panel on dietetic products, nutrition and allergies (NDA). EFSA J. 2011, 9, 2056. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Beluska-Turkan, K.; Korczak, R.; Hartell, B.; Moskal, K.; Maukonen, J.; Alexander, D.E.; Salem, N.; Harkness, L.; Ayad, W.; Szaro, J.; et al. Nutritional gaps and supplementation in the first 1000 days. Nutrients 2019, 11, 2891. [Google Scholar] [CrossRef] [PubMed]
- Cochrane. Cochrane Linked Data. PICO Ontology. 2019. Available online: https://linkeddata.cochrane.org/pico-ontology (accessed on 1 June 2020).
- SYRCLE. SYstematic Review Center for Laboratory Animal Experimentation; SYRCLE: Sentrum, Norway, 2020. [Google Scholar]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Mudd, A.T.; Getty, C.M.; Sutton, B.P.; Dilger, R.N. Perinatal choline deficiency delays brain development and alters metabolite concentrations in the young pig. Nutr. Neurosci. 2016, 19, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Mudd, A.T.; Getty, C.M.; Dilger, R.N. Maternal dietary choline status influences brain gray and white matter development in young pigs. Curr. Dev. Nutr. 2018, 2. [Google Scholar] [CrossRef] [PubMed]
- Getty, C.M.; Dilger, R.N. Moderate perinatal choline deficiency elicits altered physiology and metabolomic profiles in the piglet. PLoS ONE 2015, 10, e0133500. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Surzenko, N.; Friday, W.B.; Zeisel, S.H. Maternal dietary intake of choline in mice regulates development of the cerebral cortex in the offspring. FASEB J. 2016, 30, 1566–1578. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.H.; Wu, T.; Jin, Y.; Huang, B.X.; Zhou, R.F.; Wang, Y.Q.; Luo, X.L.; Zhu, H.L. Prenatal choline supplementation attenuates spatial learning deficits of offspring rats exposed to low-protein diet during fetal period. J. Nutr. Biochem. 2016, 32, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Alldred, M.J.; Chao, H.M.; Lee, S.H.; Beilin, J.; Powers, B.E.; Petkova, E.; Strupp, B.J.; Ginsberg, S.D. Long-term effects of maternal choline supplementation on CA1 pyramidal neuron gene expression in the Ts65Dn mouse model of Down Syndrome and Alzheimer’s disease. FASEB J. 2019, 33, 9871–9884. [Google Scholar] [CrossRef] [PubMed]
- Kelley, C.M.; Ginsberg, S.D.; Alldred, M.J.; Strupp, B.J.; Mufson, E.J. Maternal choline supplementation alters basal forebrain cholinergic neuron gene expression in the Ts65Dn mouse model of Down Syndrome. Dev. Neurobiol. 2019, 79, 664–683. [Google Scholar] [CrossRef] [PubMed]
- Ash, J.A.; Velazquez, R.; Kelley, C.M.; Powers, B.E.; Ginsberg, S.D.; Mufson, E.J.; Strupp, B.J. Maternal choline supplementation improves spatial mapping and increases basal forebrain cholinergic neuron number and size in aged Ts65Dn mice. Neurobiol. Dis. 2014, 70, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Kelley, C.M.; Powers, B.E.; Velazquez, R.; Ash, J.A.; Ginsberg, S.D.; Strupp, B.J.; Mufson, E.J. Maternal choline supplementation differentially alters the basal forebrain cholinergic system of young-adult Ts65Dn and disomic mice. J. Comp. Neurol. 2014, 522, 1390–1410. [Google Scholar] [CrossRef] [PubMed]
- Velazquez, R.; Ash, J.A.; Powers, B.E.; Kelley, C.M.; Strawderman, M.; Luscher, Z.I.; Ginsberg, S.D.; Mufson, E.J.; Strupp, B.J. Maternal choline supplementation improves spatial learning and adult hippocampal neurogenesis in the Ts65Dn mouse model of Down Syndrome. Neurobiol. Dis. 2013, 58, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Mehedint, M.G.; Niculescu, M.D.; Craciunescu, C.N.; Zeisel, S.H. Choline deficiency alters global histone methylation and epigenetic marking at the Re1 site of the calbindin 1 gene. FASEB J. 2010, 24, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Mehedint, M.G.; Craciunescu, C.N.; Zeisel, S.H. Maternal dietary choline deficiency alters angiogenesis in fetal mouse hippocampus. Proc. Natl. Acad. Sci. USA 2010, 107, 12834–12839. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.C.; Tran, P.V.; Kohli, M.; Maertens, J.J.; Gewirtz, J.C.; Georgieff, M.K. Beneficial effects of postnatal choline supplementation on long-term neurocognitive deficit resulting from fetal-neonatal iron deficiency. Behav. Brain Res. 2018, 336, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Jadavji, N.M.; Deng, L.; Malysheva, O.; Caudill, M.A.; Rozen, R. MTHFR deficiency or reduced intake of folate or choline in pregnant mice results in impaired short-term memory and increased apoptosis in the hippocampus of wild-type offspring. Neuroscience 2015, 300, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Langley, E.A.; Krykbaeva, M.; Blusztajn, J.K.; Mellott, T.J. High maternal choline consumption during pregnancy and nursing alleviates deficits in social interaction and improves anxiety-like behaviors in the BTBR T+Itpr3tf/J mouse model of autism. Behav. Brain Res. 2015, 278, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.C.; Dimova, J.G.; Siddappa, A.J.; Tran, P.V.; Gewirtz, J.C.; Georgieff, M.K. Prenatal choline supplementation ameliorates the long-term neurobehavioral effects of fetal-neonatal iron deficiency in rats. J. Nutr. 2014, 144, 1858–1865. [Google Scholar] [CrossRef] [PubMed]
- Moreno, H.C.; de Brugada, I.; Carias, D.; Gallo, M. Long-lasting effects of prenatal dietary choline availability on object recognition memory ability in adult rats. Nutr. Neurosci. 2013, 16, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Corriveau, J.A.; Glenn, M.J. Postnatal choline levels mediate cognitive deficits in a rat model of schizophrenia. Pharm. Biochem. Behav. 2012, 103, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.; Chen, M.; Gandhy, S.U.; Strawderman, M.; Levitsky, D.A.; Maclean, K.N.; Strupp, B.J. Perinatal choline supplementation improves cognitive functioning and emotion regulation in the Ts65Dn mouse model of Down Syndrome. Behav. Neurosci. 2010, 124, 346–361. [Google Scholar] [CrossRef] [PubMed]
- Moreno, H.; de Brugada, I. Prenatal dietary choline supplementation modulates long-term memory development in rat offspring. Nutr. Neurosci. 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.V.; Kennedy, B.C.; Pisansky, M.T.; Won, K.J.; Gewirtz, J.C.; Simmons, R.A.; Georgieff, M.K. Prenatal choline supplementation diminishes early-life iron deficiency-induced reprogramming of molecular networks associated with behavioral abnormalities in the adult rat hippocampus. J. Nutr. 2016, 146, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Sawant, O.B.; Birch, S.M.; Goodlett, C.R.; Cudd, T.A.; Washburn, S.E. Maternal choline supplementation mitigates alcohol-induced fetal cranio-facial abnormalities detected using an ultrasonographic examination in a sheep model. Alcohol 2019, 81, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Idrus, N.M.; Breit, K.R.; Thomas, J.D. Dietary choline levels modify the effects of prenatal alcohol exposure in rats. Neurotoxicol. Teratol. 2017, 59, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Balaraman, S.; Idrus, N.M.; Miranda, R.C.; Thomas, J.D. Postnatal choline supplementation selectively attenuates hippocampal microRNA alterations associated with developmental alcohol exposure. Alcohol 2017, 60, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.D.; Idrus, N.M.; Monk, B.R.; Dominguez, H.D. Prenatal choline supplementation mitigates behavioral alterations associated with prenatal alcohol exposure in rats. Birth Defects Res. A Clin. Mol. Teratol. 2010, 88, 827–837. [Google Scholar] [CrossRef] [PubMed]
- Bearer, C.F.; Wellmann, K.A.; Tang, N.; He, M.; Mooney, S.M. Choline ameliorates deficits in balance caused by acute neonatal ethanol exposure. Cerebellum 2015, 14, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Bekdash, R.A.; Zhang, C.; Sarkar, D.K. Gestational choline supplementation normalized fetal alcohol-induced alterations in histone modifications, DNA methylation, and proopiomelanocortin (POMC) gene expression in beta-endorphin-producing POMC neurons of the hypothalamus. Alcohol Clin. Exp. Res. 2013, 37, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.R.; Leslie, F.M.; Thomas, J.D. The effects of perinatal choline supplementation on hippocampal cholinergic development in rats exposed to alcohol during the brain growth spurt. Hippocampus 2012, 22, 1750–1757. [Google Scholar] [CrossRef] [PubMed]
- Birch, S.M.; Lenox, M.W.; Kornegay, J.N.; Paniagua, B.; Styner, M.A.; Goodlett, C.R.; Cudd, T.A.; Washburn, S.E. Maternal choline supplementation in a sheep model of first trimester binge alcohol fails to protect against brain volume reductions in peripubertal lambs. Alcohol 2016, 55, 1–8. [Google Scholar] [CrossRef]
- Alldred, M.J.; Chao, H.M.; Lee, S.H.; Beilin, J.; Powers, B.E.; Petkova, E.; Strupp, B.J.; Ginsberg, S.D. CA1 pyramidal neuron gene expression mosaics in the Ts65Dn murine model of Down Syndrome and Alzheimer’s disease following maternal choline supplementation. Hippocampus 2018, 28, 251–268. [Google Scholar] [CrossRef] [PubMed]
- Velazquez, R.; Ferreira, E.; Winslow, W.; Dave, N.; Piras, I.S.; Naymik, M.; Huentelman, M.J.; Tran, A.; Caccamo, A.; Oddo, S. Maternal choline supplementation ameliorates Alzheimer’s disease pathology by reducing brain homocysteine levels across multiple generations. Mol. Psychiatry 2019. [Google Scholar] [CrossRef] [PubMed]
- Kelley, C.M.; Ash, J.A.; Powers, B.E.; Velazquez, R.; Alldred, M.J.; Ikonomovic, M.D.; Ginsberg, S.D.; Strupp, B.J.; Mufson, E.J. Effects of maternal choline supplementation on the septohippocampal cholinergic system in the Ts65Dn mouse model of Down Syndrome. Curr. Alzheimer Res. 2016, 13, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Ginsberg, S.D.; Powers, B.; Alldred, M.J.; Saltzman, A.; Strupp, B.J.; Caudill, M.A. Maternal choline supplementation programs greater activity of the phosphatidylethanolamine N-methyltransferase (PEMT) pathway in adult Ts65Dn trisomic mice. FASEB J. 2014, 28, 4312–4323. [Google Scholar] [CrossRef] [PubMed]
- Chin, E.W.M.; Lim, W.M.; Ma, D.; Rosales, F.J.; Goh, E.L.K. Choline rescues behavioural deficits in a mouse model of rett syndrome by modulating neuronal plasticity. Mol. Neurobiol. 2019, 56, 3882–3896. [Google Scholar] [CrossRef] [PubMed]
- Mellott, T.J.; Huleatt, O.M.; Shade, B.N.; Pender, S.M.; Liu, Y.B.; Slack, B.E.; Blusztajn, J.K. Perinatal choline supplementation reduces amyloidosis and increases choline acetyltransferase expression in the hippocampus of the APPswePS1dE9 Alzheimer’s disease model mice. PLoS ONE 2017, 12, e0170450. [Google Scholar] [CrossRef]
- Schulz, K.M.; Pearson, J.N.; Gasparrini, M.E.; Brooks, K.F.; Drake-Frazier, C.; Zajkowski, M.E.; Kreisler, A.D.; Adams, C.E.; Leonard, S.; Stevens, K.E. Dietary choline supplementation to dams during pregnancy and lactation mitigates the effects of in utero stress exposure on adult anxiety-related behaviors. Behav. Brain Res. 2014, 268, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Otero, N.K.; Thomas, J.D.; Saski, C.A.; Xia, X.; Kelly, S.J. Choline supplementation and DNA methylation in the hippocampus and prefrontal cortex of rats exposed to alcohol during development. Alcohol Clin. Exp. Res. 2012, 36, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Wong-Goodrich, S.J.; Glenn, M.J.; Mellott, T.J.; Liu, Y.B.; Blusztajn, J.K.; Williams, C.L. Water maze experience and prenatal choline supplementation differentially promote long-term hippocampal recovery from seizures in adulthood. Hippocampus 2011, 21, 584–608. [Google Scholar] [CrossRef] [PubMed]
- Bahnfleth, C.; Canfield, R.; Nevins, J.; Caudill, M.; Strupp, B. Prenatal choline supplementation improves child color-location memory task performance at 7 y of age (FS05-01-19). Curr. Dev. Nutr. 2019, 3. [Google Scholar] [CrossRef]
- Andrew, M.J.; Parr, J.R.; Montague-Johnson, C.; Laler, K.; Qi, C.; Baker, B.; Sullivan, P.B. Nutritional intervention and neurodevelopmental outcome in infants with suspected cerebral palsy: The Dolphin infant double-blind randomized controlled trial. Dev. Med. Child Neurol. 2018, 60, 906–913. [Google Scholar] [CrossRef] [PubMed]
- Caudill, M.A.; Strupp, B.J.; Muscalu, L.; Nevins, J.E.H.; Canfield, R.L. Maternal choline supplementation during the third trimester of pregnancy improves infant information processing speed: A randomized, double-blind, controlled feeding study. FASEB J. 2018, 32, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.G.; Hunter, S.K.; Hoffman, M.C.; McCarthy, L.; Chambers, B.M.; Law, A.J.; Leonard, S.; Zerbe, G.O.; Freedman, R. Perinatal Phosphatidylcholine supplementation and early childhood behavior problems: Evidence for CHRNA7 moderation. Am. J. Psychiatry 2016, 173, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Kable, J.A.; Coles, C.D.; Keen, C.L.; Uriu-Adams, J.Y.; Jones, K.L.; Yevtushok, L.; Kulikovsky, Y.; Wertelecki, W.; Pedersen, T.L.; Chambers, C.D.; et al. The impact of micronutrient supplementation in alcohol-exposed pregnancies on information processing skills in Ukrainian infants. Alcohol 2015, 49, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Boeke, C.E.; Gillman, M.W.; Hughes, M.D.; Rifas-Shiman, S.L.; Villamor, E.; Oken, E. Choline intake during pregnancy and child cognition at age 7 years. Am. J. Epidemiol. 2013, 177, 1338–1347. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.G.; Hunter, S.K.; McCarthy, L.; Beuler, J.; Hutchison, A.K.; Wagner, B.D.; Leonard, S.; Stevens, K.E.; Freedman, R. Perinatal choline effects on neonatal pathophysiology related to later schizophrenia risk. Am. J. Psychiatry 2013, 170, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Cheatham, C.L.; Goldman, B.D.; Fischer, L.M.; da Costa, K.A.; Reznick, J.S.; Zeisel, S.H. Phosphatidylcholine supplementation in pregnant women consuming moderate-choline diets does not enhance infant cognitive function: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2012, 96, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Villamor, E.; Rifas-Shiman, S.L.; Gillman, M.W.; Oken, E. Maternal intake of methyl-donor nutrients and child cognition at 3 years of age. Paediatr. Perinat. Epidemiol. 2012, 26, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.T.; Dyer, R.A.; King, D.J.; Richardson, K.J.; Innis, S.M. Early second trimester maternal plasma choline and betaine are related to measures of early cognitive development in term infants. PLoS ONE 2012, 7, e43448. [Google Scholar] [CrossRef]
- Shaw, G.M.; Finnell, R.H.; Blom, H.J.; Carmichael, S.L.; Vollset, S.E.; Yang, W.; Ueland, P.M. Choline and risk of neural tube defects in a folate-fortified population. Epidemiology 2009, 20, 714–719. [Google Scholar] [CrossRef] [PubMed]
- Signore, C.; Ueland, P.M.; Troendle, J.; Mills, J.L. Choline concentrations in human maternal and cord blood and intelligence at 5 y of age. Am. J. Clin. Nutr. 2008, 87, 896–902. [Google Scholar] [CrossRef] [PubMed]
- Shaw, G.M.; Carmichael, S.L.; Yang, W.; Selvin, S.; Schaffer, D.M. Periconceptional dietary intake of choline and betaine and neural tube defects in offspring. Am. J. Epidemiol. 2004, 160, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.; Hunter, S.K.; Law, A.J.; Wagner, B.D.; D’Alessandro, A.; Christians, U.; Noonan, K.; Wyrwa, A.; Hoffman, M.C. Higher gestational choline levels in maternal infection are protective for infant brain development. J. Pediatr. 2019, 208, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, S.W.; Carter, R.C.; Molteno, C.D.; Stanton, M.E.; Herbert, J.S.; Lindinger, N.M.; Lewis, C.E.; Dodge, N.C.; Hoyme, H.E.; Zeisel, S.H.; et al. Efficacy of maternal choline supplementation during pregnancy in mitigating adverse effects of prenatal alcohol exposure on growth and cognitive function: A randomized, double-blind, placebo-controlled clinical trial. Alcohol Clin. Exp. Res. 2018, 42, 1327–1341. [Google Scholar] [CrossRef] [PubMed]
- Coles, C.D.; Kable, J.A.; Keen, C.L.; Jones, K.L.; Wertelecki, W.; Granovska, I.V.; Pashtepa, A.O.; Chambers, C.D. Cifasd. dose and timing of prenatal alcohol exposure and maternal nutritional supplements: Developmental effects on 6-month-old infants. Matern. Child Health J. 2015, 19, 2605–2614. [Google Scholar] [CrossRef] [PubMed]
- Avila, A.; Nguyen, L.; Rigo, J.M. Glycine receptors and brain development. Front. Cell Neurosci. 2013, 7, 184. [Google Scholar] [CrossRef] [PubMed]
- Bekdash, R.A. Neuroprotective effects of choline and other methyl donors. Nutrients 2019, 11, 2995. [Google Scholar] [CrossRef] [PubMed]
- Bearak, J.; Popinchalk, A.; Alkema, L.; Sedgh, G. Global, regional, and subregional trends in unintended pregnancy and its outcomes from 1990 to 2014: Estimates from a bayesian hierarchical model. Lancet Glob. Health 2018, 6, e380–e389. [Google Scholar] [CrossRef]
- Gupta, K.K.; Gupta, V.K.; Shirasaka, T. An update on fetal alcohol syndrome-pathogenesis, risks, and treatment. Alcohol Clin. Exp. Res. 2016, 40, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Denny, C.H.; Cheal, N.E.; Sniezek, J.E.; Kanny, D. Alcohol use and binge drinking among women of childbearing age-United States, 2011–2013. Morb. Mortal Wkly. Rep. 2015, 1042–1046. [Google Scholar] [CrossRef] [PubMed]
- Ganz, A.B.; Shields, K.; Fomin, V.G.; Lopez, Y.S.; Mohan, S.; Lovesky, J.; Chuang, J.C.; Ganti, A.; Carrier, B.; Yan, J.; et al. Genetic impairments in folate enzymes increase dependence on dietary choline for phosphatidylcholine production at the expense of betaine synthesis. FASEB J. 2016, 30, 3321–3333. [Google Scholar] [CrossRef] [PubMed]
- Strupp, B.J.; Powers, B.E.; Velazquez, R.; Ash, J.A.; Kelley, C.M.; Alldred, M.J.; Strawderman, M.; Caudill, M.A.; Mufson, E.J.; Ginsberg, S.D. Maternal choline supplementation: A potential prenatal treatment for Down Syndrome and Alzheimer’s disease. Curr. Alzheimer Res. 2016, 13, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Powers, B.E.; Kelley, C.M.; Velazquez, R.; Ash, J.A.; Strawderman, M.S.; Alldred, M.J.; Ginsberg, S.D.; Mufson, E.J.; Strupp, B.J. Maternal choline supplementation in a mouse model of Down Syndrome: Effects on attention and nucleus basalis/substantia innominata neuron morphology in adult offspring. Neuroscience 2017, 340, 501–514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Han, X.; Bao, J.; Yang, J.; Shi, S.Q.; Garfield, R.E.; Liu, H. Choline supplementation during pregnancy protects against gestational lipopolysaccharide-induced inflammatory responses. Reprod. Sci. 2018, 25, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.T.C.; King, J.H.; Yan, J.; Wang, Z.; Jiang, X.; Hutzler, J.S.; Klein, H.R.; Brenna, J.T.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation modulates placental nutrient transport and metabolism in late gestation of mouse pregnancy. J. Nutr. 2017, 147, 2083–2092. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.T.C.; King, J.H.; Grenier, J.K.; Yan, J.; Jiang, X.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation during normal murine pregnancy alters the placental epigenome: Results of an exploratory study. Nutrients 2018, 10, 417. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.T.C.; King, J.H.; Yan, J.; Jiang, X.; Wei, E.; Fomin, V.G.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation during murine pregnancy modulates placental markers of inflammation, apoptosis and vascularization in a fetal sex-dependent manner. Placenta 2017, 53, 57–65. [Google Scholar] [CrossRef] [PubMed]
- King, J.H.; Kwan, S.T.C.; Bae, S.; Klatt, K.C.; Yan, J.; Malysheva, O.V.; Jiang, X.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation alters vitamin B-12 status in human and murine pregnancy. J. Nutr. Biochem. 2019, 72, 108210. [Google Scholar] [CrossRef] [PubMed]
- Joselit, Y.; Nanobashvili, K.; Jack-Roberts, C.; Greenwald, E.; Malysheva, O.V.; Caudill, M.A.; Saxena, A.; Jiang, X. Maternal betaine supplementation affects fetal growth and lipid metabolism of high-fat fed mice in a temporal-specific manner. Nutr. Diabetes 2018, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.; Greenwald, E.; Jack-Roberts, C.; Ajeeb, T.T.; Malysheva, O.V.; Caudill, M.A.; Axen, K.; Saxena, A.; Semernina, E.; Nanobashvili, K.; et al. Choline prevents fetal overgrowth and normalizes placental fatty acid and glucose metabolism in a mouse model of maternal obesity. J. Nutr. Biochem. 2017, 49, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Bar, H.Y.; Yan, J.; Jones, S.; Brannon, P.M.; West, A.A.; Perry, C.A.; Ganti, A.; Pressman, E.; Devapatla, S.; et al. A higher maternal choline intake among third-trimester pregnant women lowers placental and circulating concentrations of the antiangiogenic factor fms-like tyrosine kinase-1 (sFLT1). FASEB J. 2013, 27, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Jones, S.; Andrew, B.Y.; Ganti, A.; Malysheva, O.V.; Giallourou, N.; Brannon, P.M.; Roberson, M.S.; Caudill, M.A. Choline inadequacy impairs trophoblast function and vascularization in cultured human placental trophoblasts. J. Cell Physiol. 2014, 229, 1016–1027. [Google Scholar] [CrossRef] [PubMed]
- King, J.H.; Kwan, S.T.C.; Yan, J.; Jiang, X.; Fomin, V.G.; Levine, S.P.; Wei, E.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation modulates placental markers of inflammation, angiogenesis, and apoptosis in a mouse model of placental insufficiency. Nutrients 2019, 11, 374. [Google Scholar] [CrossRef] [PubMed]
- Faraoni, D.; Schaefer, S.T. Randomized controlled trials vs. observational studies: Why not just live together? BMC Anesth. 2016, 16, 102. [Google Scholar] [CrossRef] [PubMed]
- Meuli, L.; Dick, F. Understanding confounding in observational studies. Eur. J. Vasc. Endovasc. Surg. 2018, 55, 737. [Google Scholar] [CrossRef] [PubMed]
| Study (Author, Year, Country) | Stage of 1000 Days | Study Design | Dose of Choline/Choline Measurements | Study Outcomes | Main Findings |
|---|---|---|---|---|---|
| Bahnfleth et al. (2019) [115] USA | 3rd T (from 27 weeks gestation for 12 weeks) (n = 20) | DB randomized parallel-group controlled feeding intervention. | Randomized to consume 480 mg or 930 mg choline/d from gestational week 27 until delivery. | Computer-based color-location memory task | Children whose mothers consumed 930 vs.480 mg choline/day performed significantly better on a task of color-location memory at age 7 years, suggesting a long-term beneficial effect of prenatal choline. |
| Freedman et al. (2019) [128] USA | 2nd T. Mothers with an infection from 16 weeks gestation and their infants (n = 66) | Prospective Cohort | Serum-free choline and baseline levels. | Infant brain development, cerebral inhibition, and auditory response | Development of cerebral inhibition, auditory cerebral response and behavioral regulation significantly improved in infants born to infected mothers with higher gestational choline concentrations, attenuating the effects of infections. |
| Andrew et al. (2018) [116] UK | Infants aged 1 to 18 months (n = 40) | DB RCT. | Randomized to a supplementation program (included 10.5 mg choline) or control. | Neurodevelopment | No statistically significant differences in neurodevelopmental outcome were identified between groups. |
| Caudill et al. (2018) [117] USA | Entering 3rd T. Infants assessed up to 13 months (n = 24). | DB randomized parallel-group controlled feeding intervention. | Choline supplement of either 100 or 550 mg/day. Diet provided 380 mg choline/day. Total choline received was 480 or 930 mg/day. | Infant processing speed, visuospatial memory | Mean reaction time was significantly faster for infants born to mothers in the 930 (vs. 480) mg choline/day group. Maternal consumption of approximately twice the recommended amount of choline during the last trimester improved infant information processing speed. |
| Jacobson et al. (2018) [129] South Africa | Mid-pregnancy—heavy drinkers (n = 69). | DB PC RCT. | 2 g of choline daily (choline bitartrate) or placebo from enrolment until delivery. | Infant cognitive function, eyeblink conditioning | At 6.5 months infants in the choline arm had better eyeblink conditioning and at 12 months higher novelty preference scores, indicating better visual recognition memory. |
| Ross et al. (2016) [118] USA | 1st T. Data for n = 49 infants. | PC RCT. | Received 900 mg choline/day. After birth, infants received 100 mg of phosphatidylcholine in an oral suspension once daily or placebo. | Childhood behavior, attention problems | At 40 months, parent ratings of children in the phosphatidylcholine group indicated fewer attention problems and less social withdrawal compared with the control group. |
| Coles et al. (2015) [130] USA | 2nd T (from 19 weeks gestation). Moderate to heavy drinking (n = 301), and low/unexposed comparison women (n = 313). | Prospective cohort study (and RCT within) | Randomly assigned to receive: (1) daily MVM supplement, (2) “standard of care” or (3) MVM-supplement providing 750 mg choline/day. | Infant development (Bayley Scales) | Developmental improvement in infants associated with choline seen not observed in this study. |
| Kable et al. (2015) [119] USA | 1st T. Studied from first prenatal visit (n = 372) | RCT. | Randomly assigned to receive: (1) daily MVM supplement, (2) “standard of care” or (3) MVM-supplement providing 750 mg choline/day. | Information processing skills | Choline supplementation +routine MVM supplements resulted in a more significant difference in visual habituation, indicating a beneficial impact on learning mechanisms involved in encoding/memory in alcohol-exposed and non/low alcohol-exposed pregnancies. This process may be mediated by the breakdown of choline to betaine and then to DMG. |
| Boeke et al. (2013) [120] USA | 1st/2nd T. n = 895 | Prospective cohort | Maternal choline intakes observed. | Offspring visual memory | Mean choline intake in the 2nd trimester was 328 mg and associated with modestly better child visual memory at age 7 years. |
| Ross et al. (2013) [121] USA | 3rd T (from 17 weeks) (n = 100) | DB PC trial. | Received 900 mg choline/day. After birth, infants received 100 mg of phosphatidylcholine in an oral suspension once daily or placebo. | Electroencephalographic recordings | More choline-treated infants (76%) suppressed the P50 response, compared to placebo-treated infants (43%) at the fifth postnatal week (effect size 0.7). A CHRNA7 genotype associated with schizophrenia diminished P50 inhibition in the placebo-treated infants, but not in the choline-treated infants. |
| Cheatham et al. (2012) [122] USA | Pregnancy and postpartum (n = 140). Infants assessed up to 13 months. | DB RCT. | Receive supplemental phosphatidylcholine (750 mg) or a placebo (corn oil) from 18 week gestation through 90 day postpartum | Infant cognitive function | Phosphatidylcholine supplementation of pregnant women eating diets containing moderate amounts of choline did not enhance their infants’ brain function. It is possible that a longer follow-up period would reveal late-emerging effects |
| Villamor et al. (2012) [123] USA | 1st/2nd T. n = 1210 | Prospective cohort | Maternal choline intakes observed. | Child cognition | No associations observed between choline or cognitive outcomes at 3 years. |
| Wu et al. (2012) [124] Canada | 2nd T. n = 154 mother-infant pairs. | Prospective cohort | Measured maternal plasma free choline. | Early cognitive development | Significant positive associations were found between infant cognitive test scores at 18 months of age and maternal plasma free choline at 16 weeks of gestation. |
| Shaw et al. (2009) [125] USA | 2nd T. n = 180,000 pregnant women | Prospective cohort. | Serum total choline concentrations measured between 15–18 weeks gestation. | Neural tube defects | NTD risk was elevated with lower levels of total choline and reduced with higher levels of choline. |
| Signore et al. (2008) [126] USA | 2nd/3rd T. n = 404 maternal-child pairs | Prospective cohort. | Serum concentrations of total and free choline measured at 16–18 week, 24–26 week, 30–32 week, and 36-38 week and in cord blood. | Intelligence | Gestational and newborn choline concentrations in the physiologic range showed no associations with childhood intelligence at 5 years. |
| Shaw et al. (2004) [127] USA | 3 months before conception | Case -control. | Maternal dietary choline intake recorded 3 months prior to conception | Neural tube defects | NTD risk estimates were lowest amongst women whose diets were rich in choline. |
| Publication | Randomized | Method of Randomization Described and Appropriate | Blinding Mentioned | Method of Blinding | Withdrawal and Dropout of Subjects | Total Score |
|---|---|---|---|---|---|---|
| Bahnfleth et al. (2019) [115] | 1 | * | 1 | * | 1 | 3 |
| Andrew et al. (2018) [116] | 1 | 1 | 1 | 1 | 1 | 5 |
| Caudill et al. (2018) [117] | 1 | 1 | 1 | 1 | 1 | 5 |
| Jacobson et al. (2018) [129] | 1 | 1 | 1 | 1 | 1 | 5 |
| Ross et al. (2016) [118] | 0 | 0 | 1 | 1 | 1 | 3 |
| Coles et al. (2015) [130] | 1 | 0 | 0 | 0 | 0 | 1 |
| Kable et al. (2015) [119] | 1 | 1 | 0 | 0 | 1 | 3 |
| Ross et al. (2013) [121] | 1 | 0 | 1 | 0 | 1 | 3 |
| Cheatham et al. (2012) [122] | 1 | 1 | 1 | 1 | 1 | 5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Derbyshire, E.; Obeid, R. Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days. Nutrients 2020, 12, 1731. https://doi.org/10.3390/nu12061731
Derbyshire E, Obeid R. Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days. Nutrients. 2020; 12(6):1731. https://doi.org/10.3390/nu12061731
Chicago/Turabian StyleDerbyshire, Emma, and Rima Obeid. 2020. "Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days" Nutrients 12, no. 6: 1731. https://doi.org/10.3390/nu12061731
APA StyleDerbyshire, E., & Obeid, R. (2020). Choline, Neurological Development and Brain Function: A Systematic Review Focusing on the First 1000 Days. Nutrients, 12(6), 1731. https://doi.org/10.3390/nu12061731






