Acute and Chronic Effects of Green Oat (Avena sativa) Extract on Cognitive Function and Mood during a Laboratory Stressor in Healthy Adults: A Randomised, Double-Blind, Placebo-Controlled Study in Healthy Humans
Abstract
1. Introduction
2. Methods
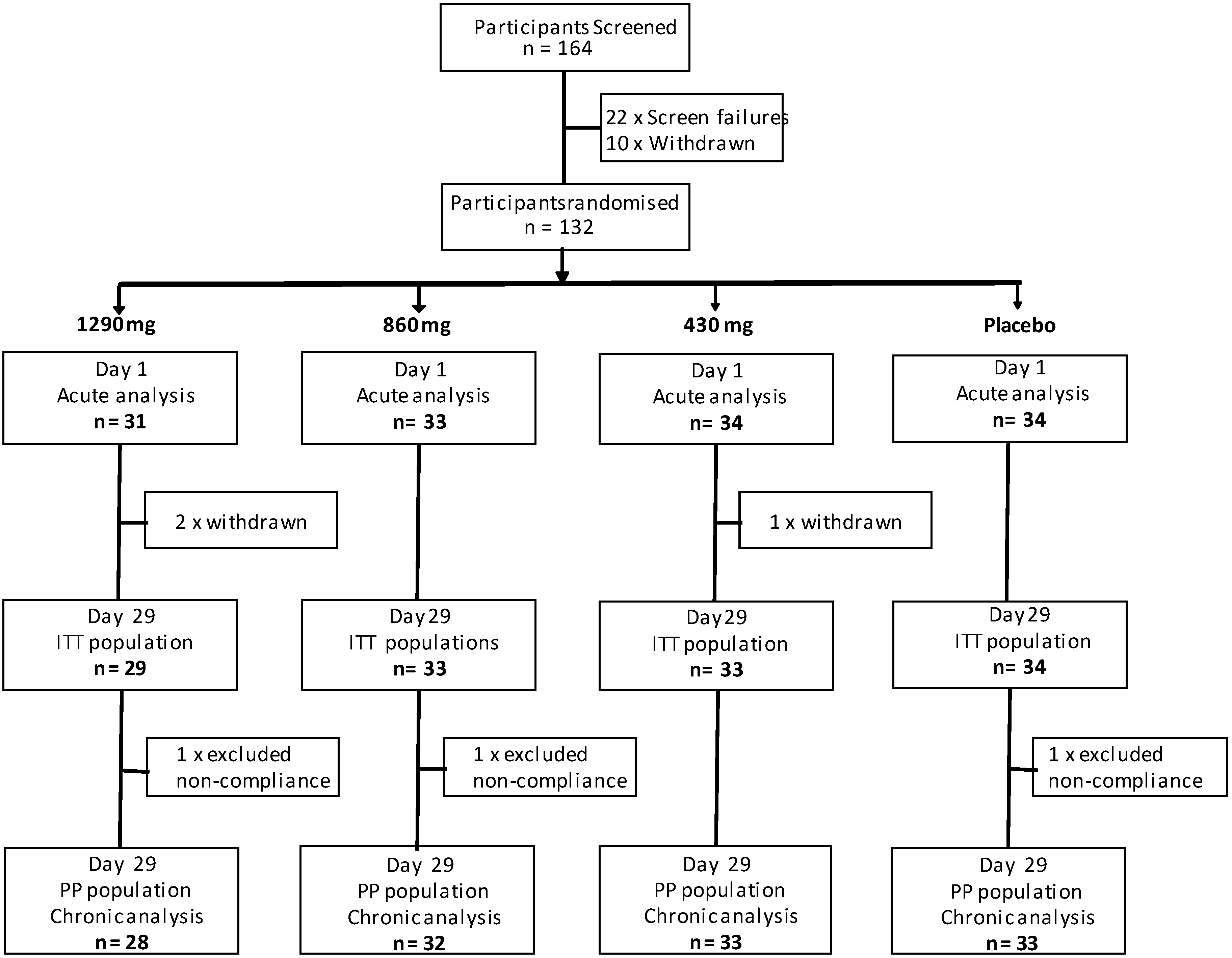
2.1. Design
2.2. Participants
2.3. Treatments
- Placebo
- 430 mg cognitaven® (equivalent to 300 mg native green oat extract)
- 860 mg cognitaven® (600 mg native extract)
- 1290 mg cognitaven® (900 mg native extract)
2.4. Psychological Measures
2.4.1. Mood
2.4.2. Cognitive Tasks
2.4.3. Multitasking Task
2.4.4. Observed Multi-Tasking Stressor (OMS)
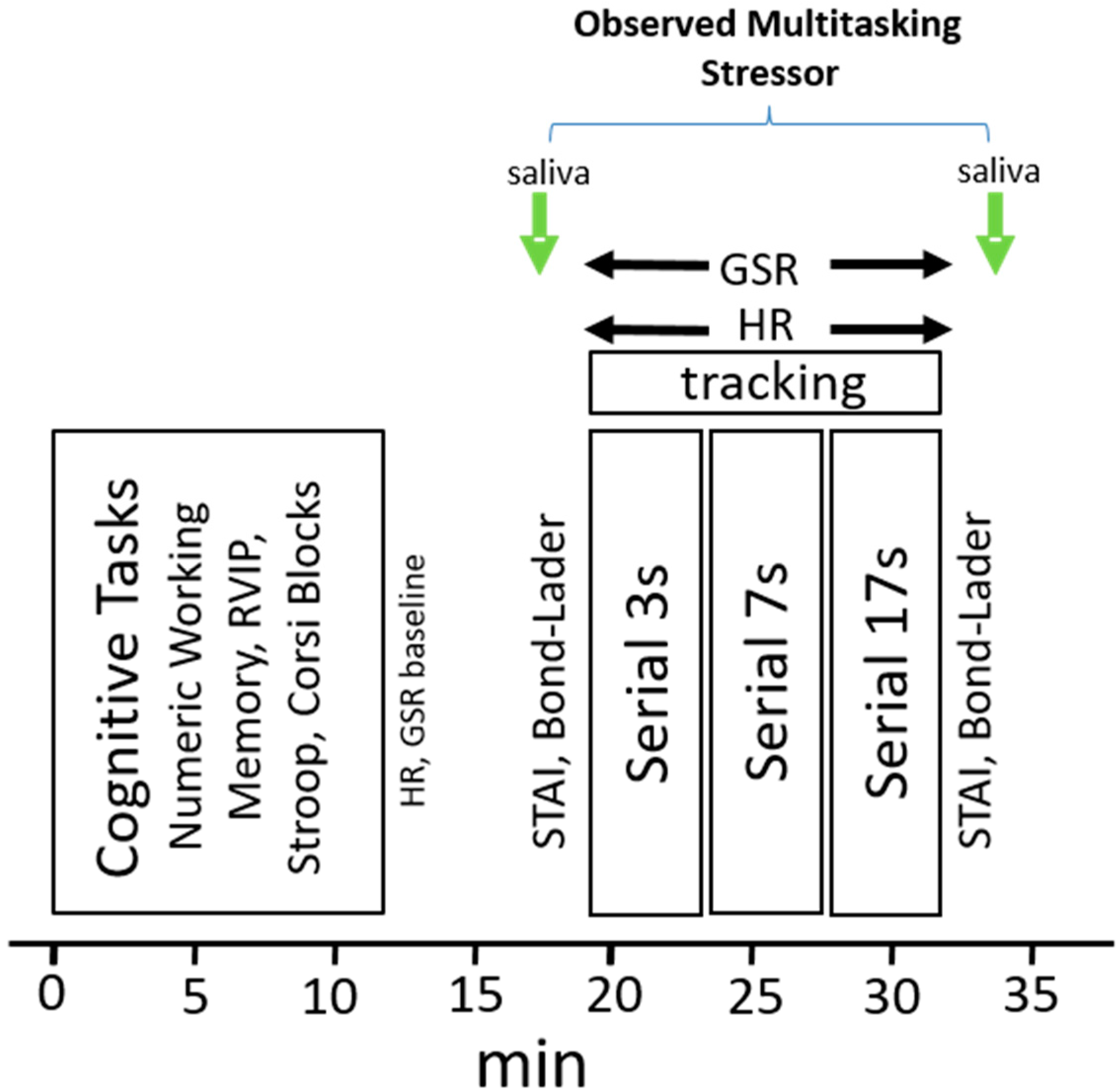
2.5. Procedure
2.6. Analysis
3. Results
3.1. Baseline Differences
3.2. Cognitive Function
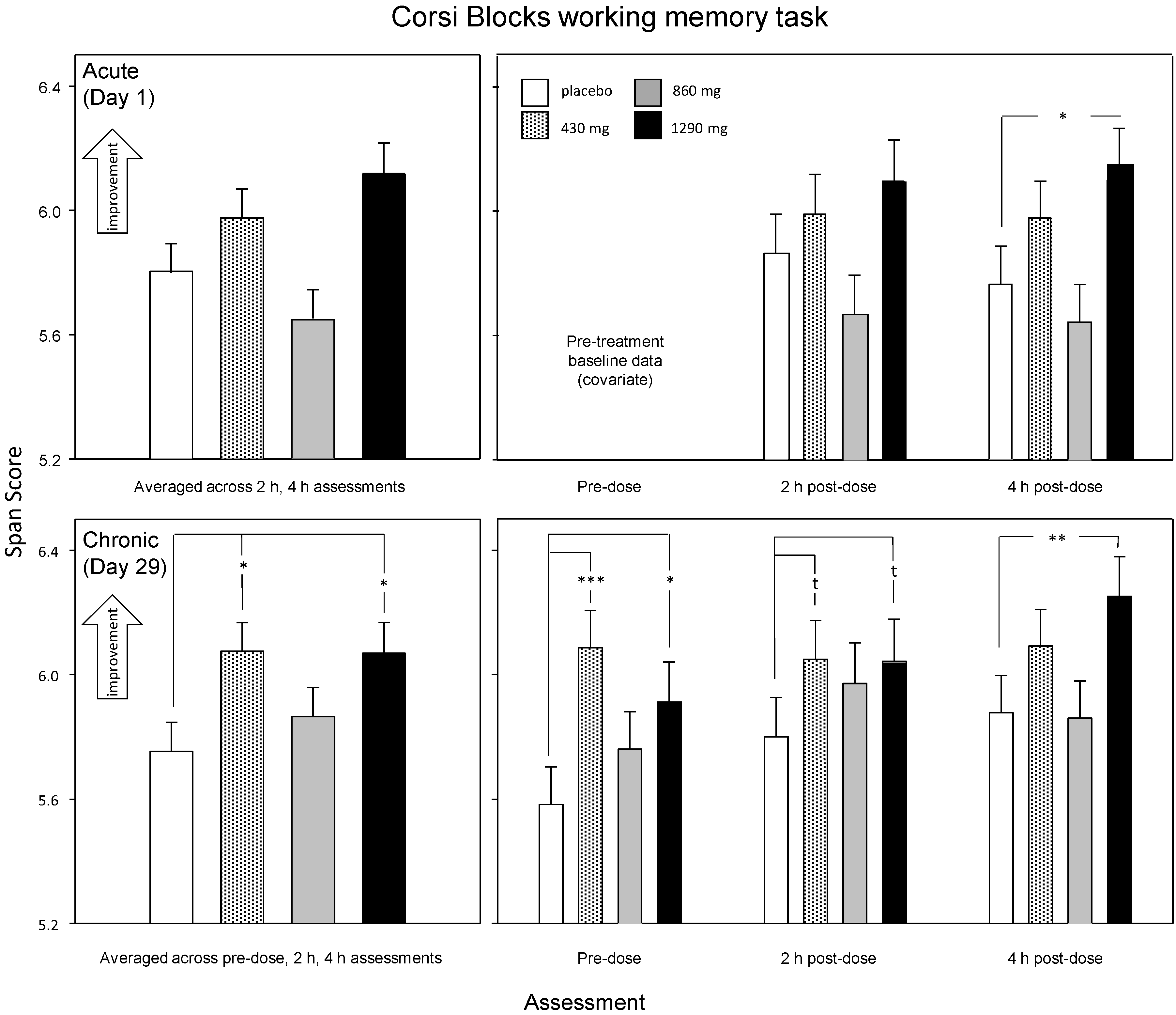
3.2.1. Corsi Blocks Working Memory Task
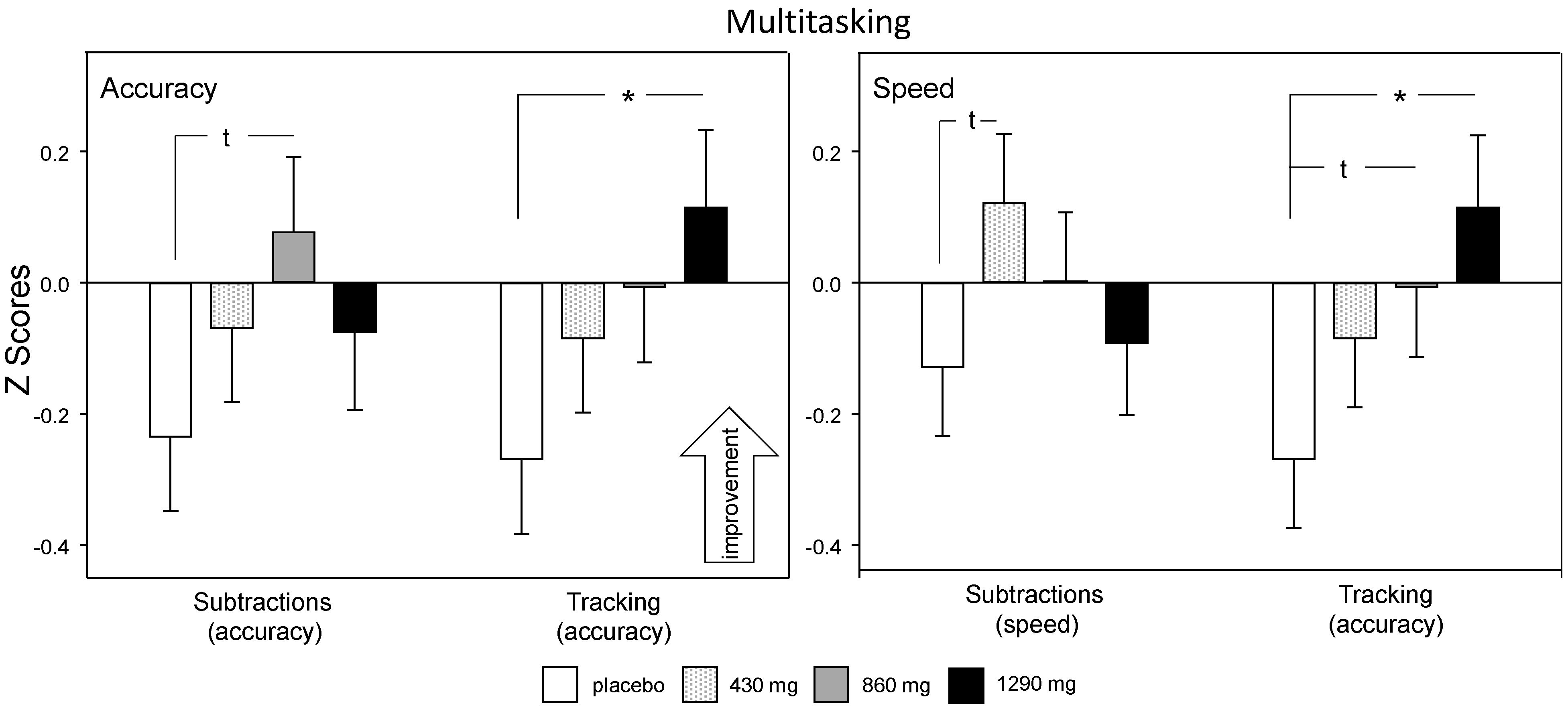
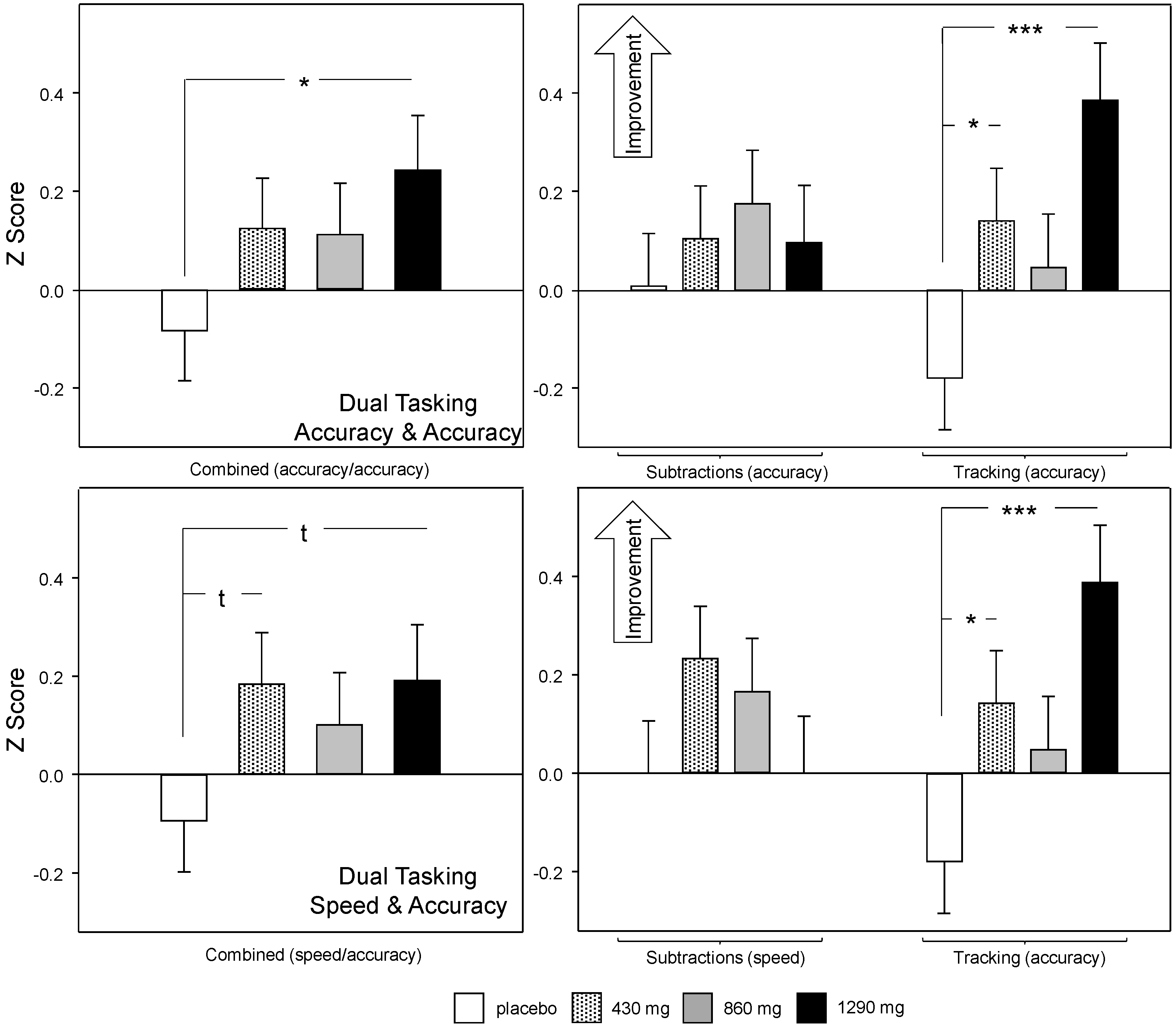
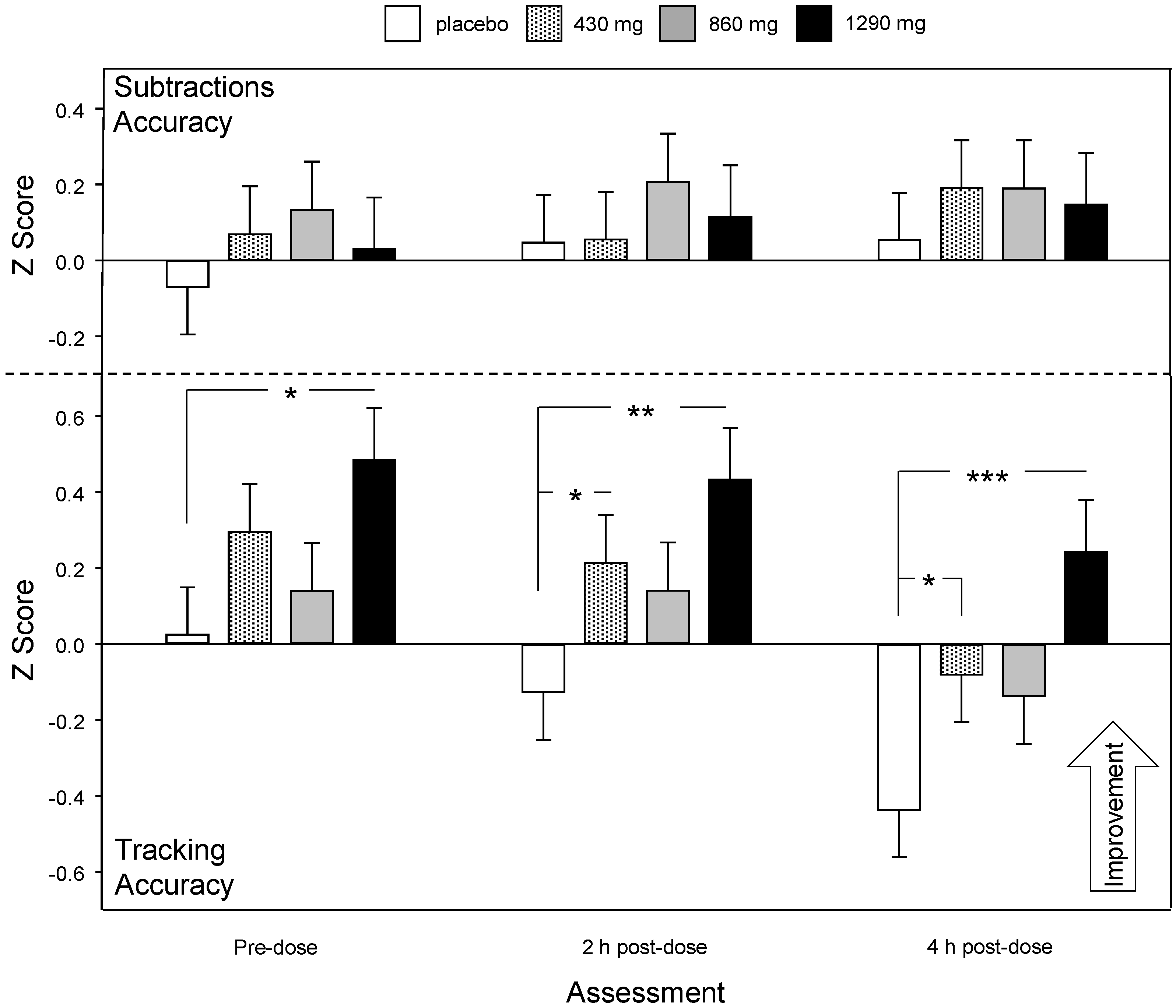
3.2.2. Multitasking Task
3.2.3. Other Cognitive Tasks
3.3. Mood
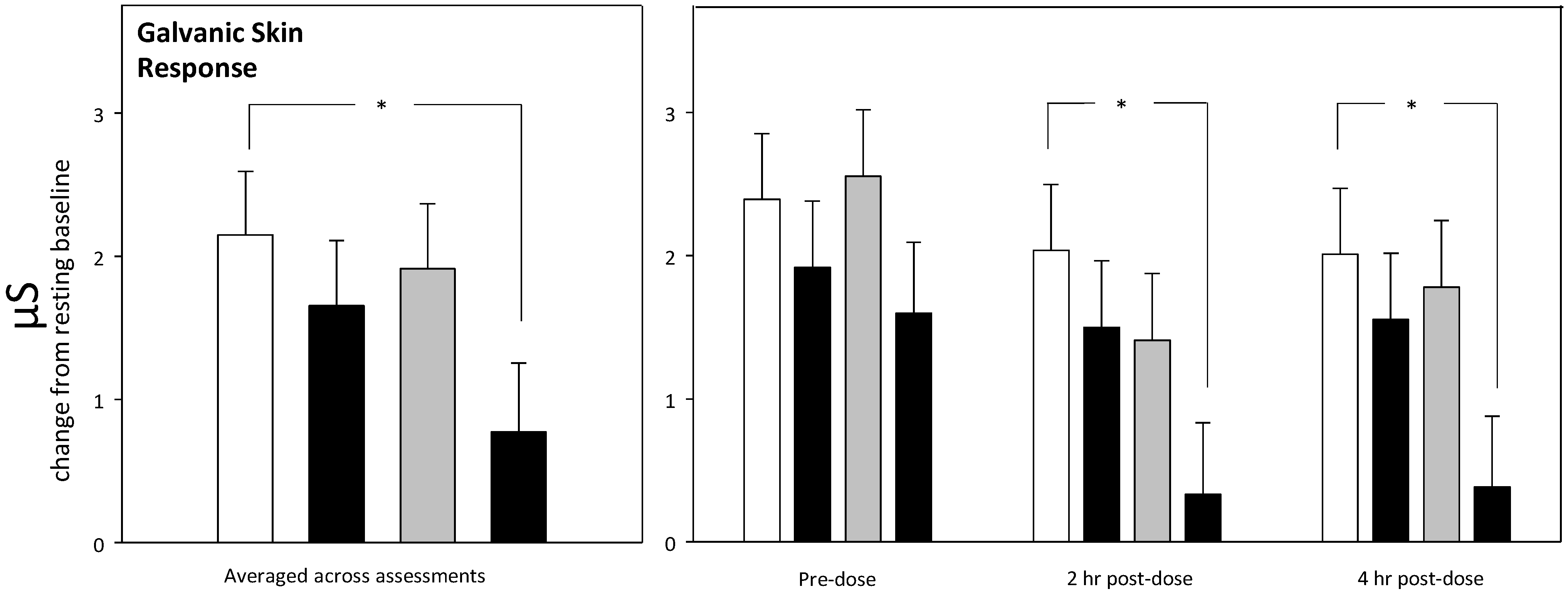
3.4. Physiological Measures
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Abascal, K.; Yarnell, E. Nervine herbs for treating anxiety. Altern. Complementary Ther. 2004, 10, 309–315. [Google Scholar] [CrossRef]
- Blumenthal, M.; Goldberg, A.; Brinckmann, J. Herbal Medicine. Expanded Commission e Monographs; Integrative Medicine Communications: Newton, MA, USA, 2000. [Google Scholar]
- Kaur, D.; Kamboj, A.; Shri, R. Comparative evaluation of anxiolytic effects of various extracts of oats (avena sativa), rice bran (oryza sativa) and spinach (spinacia oleracea) in experimental animals. Int. J. Pharm. Sci. Res. 2016, 7, 4110. [Google Scholar]
- Osbourn, A.E. Saponins in cereals. Phytochemistry 2003, 62, 1–4. [Google Scholar] [CrossRef]
- Meydani, M. Potential health benefits of avenanthramides of oats. Nutr. Rev. 2009, 67, 731–735. [Google Scholar] [CrossRef] [PubMed]
- Mylona, P.; Owatworakit, A.; Papadopoulou, K.; Jenner, H.; Qin, B.; Findlay, K.; Hill, L.; Qi, X.; Bakht, S.; Melton, R. Sad3 and sad4 are required for saponin biosynthesis and root development in oat. Plant Cell 2008, 20, 201–212. [Google Scholar] [CrossRef]
- Kennedy, D.O. Plants and the Human Brain; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Sur, R.; Nigam, A.; Grote, D.; Liebel, F.; Southall, M.D. Avenanthramides, polyphenols from oats, exhibit anti-inflammatory and anti-itch activity. Arch. Dermatol. Res. 2008, 300, 569. [Google Scholar] [CrossRef]
- Bahraminejad, S.; Asenstorfer, R.; Riley, I.; Schultz, C. Analysis of the antimicrobial activity of flavonoids and saponins isolated from the shoots of oats (avena sativa l.). J. Phytopathol. 2008, 156, 1–7. [Google Scholar] [CrossRef]
- Günther-Jordanland, K.; Dawid, C.; Dietz, M.; Hofmann, T. Key phytochemicals contributing to the bitter off-taste of oat (avena sativa l.). J. Agric. Food Chem. 2016, 64, 9639–9652. [Google Scholar]
- Kennedy, D.O. Polyphenols and the human brain: Plant “secondary metabolite” ecologic roles and endogenous signaling functions drive benefits. Adv. Nutr. 2014, 5, 515–533. [Google Scholar] [CrossRef]
- Spencer, J.P. Flavonoids and brain health: Multiple effects underpinned by common mechanisms. Genes Nutr. 2009, 4, 243–250. [Google Scholar] [CrossRef]
- Williams, R.J.; Spencer, J.P. Flavonoids, cognition, and dementia: Actions, mechanisms, and potential therapeutic utility for alzheimer disease. Free Radic. Biol. Med. 2012, 52, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Baptista, F.I.; Henriques, A.G.; Silva, A.M.; Wiltfang, J.; Da Cruz e Silva, O.A. Flavonoids as therapeutic compounds targeting key proteins involved in alzheimer’ s disease. ACS Chem. Neurosci. 2014, 5, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Chin, S.P.; Sukumaran, S.D.; Buckle, M.J.C.; Kiew, L.V.; Chung, L.Y. In silico and in vitro analysis of bacoside a aglycones and its derivatives as the constituents responsible for the cognitive effects of bacopa monnieri. PLoS ONE 2015, 10, e0126565. [Google Scholar] [CrossRef] [PubMed]
- Ryoo, N.; Rahman, M.A.; Hwang, H.; Ko, S.K.; Nah, S.-Y.; Kim, H.-C.; Rhim, H. Ginsenoside rk1 is a novel inhibitor of nmda receptors in cultured rat hippocampal neurons. J. Ginseng Res. 2019, 44, 490–495. [Google Scholar] [CrossRef]
- Armijos, C.; Gilardoni, G.; Amay, L.; Lozano, A.; Bracco, F.; Ramirez, J.; Bec, N.; Larroque, C.; Finzi, P.V.; Vidari, G. Phytochemical and ethnomedicinal study of huperzia species used in the traditional medicine of saraguros in southern ecuador; ache and mao inhibitory activity. J. Ethnopharmacol. 2016, 193, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Bahadori, M.B.; Dinparast, L.; Valizadeh, H.; Farimani, M.M.; Ebrahimi, S.N. Bioactive constituents from roots of salvia syriaca l.: Acetylcholinesterase inhibitory activity and molecular docking studies. S. Afr. J. Bot. 2016, 106, 1–4. [Google Scholar] [CrossRef]
- Singh, R.; Ramakrishna, R.; Bhateria, M.; Bhatta, R.S. In vitro evaluation of bacopa monniera extract and individual constituents on human recombinant monoamine oxidase enzymes. Phytother. Res. 2014, 28, 1419–1422. [Google Scholar] [CrossRef]
- Francis, G.; Kerem, Z.; Makkar, H.P.; Becker, K. The biological action of saponins in animal systems: A review. Br. J. Nutr. 2002, 88, 587–605. [Google Scholar] [CrossRef]
- Gao, X.-Q.; Du, Z.-R.; Yuan, L.-J.; Zhang, W.-D.; Chen, L.; Teng, J.-J.; Wong, M.-S.; Xie, J.-X.; Chen, W.-F. Ginsenoside rg1 exerts anti-inflammatory effects via g protein-coupled estrogen receptor in lipopolysaccharide-induced microglia activation. Front. Neurosci. 2019, 13, 1168. [Google Scholar] [CrossRef]
- Scholey, A.B.; French, S.J.; Morris, P.J.; Kennedy, D.O.; Milne, A.L.; Haskell, C.F. Consumption of cocoa flavanols results in acute improvements in mood and cognitive performance during sustained mental effort. J. Psychopharmacol. 2010, 24, 1505–1514. [Google Scholar] [CrossRef]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C. Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The cocoa, cognition, and aging (cocoa) study—A randomized controlled trial. Am. J. Clin. Nutr. 2014, 101, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Desideri, G.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Ghiadoni, L.; Mastroiacovo, D.; Raffaele, A.; Ferri, L.; Bocale, R.; Lechiara, M.C. Benefits in cognitive function, blood pressure, and insulin resistance through cocoa flavanol consumption in elderly subjects with mild cognitive impairmentnovelty and significance the cocoa, cognition, and aging (cocoa) study. Hypertension 2012, 60, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Kongkeaw, C.; Dilokthornsakul, P.; Thanarangsarit, P.; Limpeanchob, N.; Norman Scholfield, C. Meta-analysis of randomized controlled trials on cognitive effects of bacopa monnieri extract. J. Ethnopharmacol. 2014, 151, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Reay, J.L.; Kennedy, D.O.; Scholey, A.B. Single doses of panax ginseng (g115) reduce blood glucose levels and improve cognitive performance during sustained mental activity. J. Psychopharmacol. 2005, 19, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Reay, J.L.; Kennedy, D.O.; Scholey, A.B. Effects of panax ginseng, consumed with and without glucose, on blood glucose levels and cognitive performance during sustained ‘mentally demanding’ tasks. J. Psychopharmacol. 2006, 20, 771–781. [Google Scholar] [CrossRef]
- Ossoukhova, A.; Owen, L.; Savage, K.; Meyer, M.; Ibarra, A.; Roller, M.; Pipingas, A.; Wesnes, K.; Scholey, A. Improved working memory performance following administration of a single dose of american ginseng (panax quinquefolius l.) to healthy middle-age adults. Hum. Psychopharmacol. Clin. Exp. 2015, 30, 108–122. [Google Scholar] [CrossRef]
- Lang, S.C. Avena sativa: A natural supporter of cognition and mental fitness. In Wellness Foods & Supplements; Dr. Harnisch Verlags GmbH: Nürnberg, Germany, 2017. [Google Scholar]
- Moccetti, T.; Wullschleger, C.; Schmidt, A.; Aydogan, C.; Kreuter, M. Bioactivity-based development of a wild green oat (avena sativa l.) extract in support of mental health disorders. Z. Für Phytother. 2006, 27, P24. [Google Scholar] [CrossRef]
- Schellekens, C.; Perrinjaquet-Moccetti, T.; Wullschleger, C.; Heyne, A. An extract from wild green oat improves rat behaviour. Phytother. Res. 2009, 23, 1371–1377. [Google Scholar] [CrossRef]
- Dimpfel, W.; Storni, C.; Verbruggen, M. Ingested oat herb extract (avena sativa) changes eeg spectral frequencies in healthy subjects. J. Altern. Complementary Med. 2011, 17, 427–434. [Google Scholar] [CrossRef]
- Berry, N.M.; Robinson, M.J.; Bryan, J.; Buckley, J.D.; Murphy, K.J.; Howe, P.R. Acute effects of an avena sativa herb extract on responses to the stroop color–word test. J. Altern. Complementary Med. 2011, 17, 635–637. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Jackson, P.A.; Forster, J.; Khan, J.; Grothe, T.; Perrinjaquet-Moccetti, T.; Haskell-Ramsay, C.F. Acute effects of a wild green-oat (avena sativa) extract on cognitive function in middle-aged adults: A double-blind, placebo-controlled, within-subjects trial. Nutr. Neurosci. 2017, 20, 135–151. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.; Howe, P.R.; Coates, A.M.; Buckley, J.D.; Berry, N.M. Chronic consumption of a wild green oat extract (neuravena) improves brachial flow-mediated dilatation and cerebrovascular responsiveness in older adults. J. Hypertens. 2013, 31, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.; Howe, P.R.; Bryan, J.; Coates, A.M.; Buckley, J.D.; Berry, N.M. Chronic effects of a wild green oat extract supplementation on cognitive performance in older adults: A randomised, double-blind, placebo-controlled, crossover trial. Nutrients 2012, 4, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Bond, A.; Lader, M. Use of analog scales in rating subjective feelings. Br. J. Med Psychol. 1974, 47, 211–218. [Google Scholar] [CrossRef]
- McNair, P.M.; Lorr, M.; Droppleman, L. Poms Manual: Profile of Mood States; Educational and Industrial Testing Service: San Diego, CA, USA, 1992. [Google Scholar]
- Spielberger, C.; Gorsuch, R.; Lushene, R. The State Trait Anxiety Inventory Manual; Consulting Psychologists Press: Palo Alto, CA, USA, 1969. [Google Scholar]
- Haskell, C.F.; Robertson, B.; Jones, E.; Forster, J.; Jones, R.; Wilde, A.; Maggini, S.; Kennedy, D.O. Effects of a multi-vitamin/mineral supplement on cognitive function and fatigue during extended multi-tasking. Hum. Psychopharmacol. 2010, 25, 448–461. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, D.P.; Williams, P. A User’s Guide to the General Health Questionnaire; NFER-Nelson: Slough, UK, 1988. [Google Scholar]
- Kennedy, D.; Wightman, E.; Khan, J.; Grothe, T.; Jackson, P. The acute and chronic cognitive and cerebral blood-flow effects of nepalese pepper (zanthoxylum armatum dc.) extract—A randomized, double-blind, placebo-controlled study in healthy humans. Nutrients 2019, 11, 3022. [Google Scholar] [CrossRef] [PubMed]
- Kirschbaum, C.; Pirke, K.-M.; Hellhammer, D.H. The ‘trier social stress test’—A tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 1993, 28, 76–81. [Google Scholar] [CrossRef]
- Scholey, A.; Haskell, C.; Robertson, B.; Kennedy, D.; Milne, A.; Wetherell, M. Chewing gum alleviates negative mood and reduces cortisol during acute laboratory psychological stress. Physiol. Behav. 2009, 97, 304–312. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Little, W.; Haskell, C.F.; Scholey, A.B. Anxiolytic effects of a combination of melissa officinalis and valeriana officinalis during laboratory induced stress. Phytother. Res. 2006, 20, 96–102. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Little, W.; Scholey, A.B. Attenuation of laboratory-induced stress in humans after acute administration of melissa officinalis (lemon balm). Psychosom. Med. 2004, 66, 607–613. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Baldwin, L.A. U-shaped dose-responses in biology, toxicology, and public health. Annu. Rev. Public Health 2001, 22, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.D.; Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cassidy, A. Relative impact of flavonoid composition, dose and structure on vascular function: A systematic review of randomised controlled trials of flavonoid-rich food products. Mol. Nutr. Food Res. 2012, 56, 1605–1616. [Google Scholar] [CrossRef] [PubMed]
- Fiebich, B.L.; Knörle, R.; Appel, K.; Kammler, T.; Weiss, G. Pharmacological studies in an herbal drug combination of st. John’s wort (hypericum perforatum) and passion flower (passiflora incarnata): In vitro and in vivo evidence of synergy between hypericum and passiflora in antidepressant pharmacological models. Fitoterapia 2011, 82, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Hellum, B.H.; Hu, Z.; Nilsen, O.G. The induction of cyp1a2, cyp2d6 and cyp3a4 by six trade herbal products in cultured primary human hepatocytes. Basic Clin. Pharmacol. Toxicol. 2007, 100, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Caird, J.K.; Simmons, S.M.; Wiley, K.; Johnston, K.A.; Horrey, W.J. Does talking on a cell phone, with a passenger, or dialing affect driving performance? An updated systematic review and meta-analysis of experimental studies. Hum. Factors 2018, 60, 101–133. [Google Scholar] [CrossRef]
- Bühner, M.; König, C.J.; Pick, M.; Krumm, S. Working memory dimensions as differential predictors of the speed and error aspect of multitasking performance. Hum. Perform. 2006, 19, 253–275. [Google Scholar] [CrossRef]
- Boucsein, W. Electrodermal Activit; Springer Science & Business Media: New York, USA, 2012. [Google Scholar]
- Christopoulos, G.I.; Uy, M.A.; Yap, W.J. The body and the brain: Measuring skin conductance responses to understand the emotional experience. Organ. Res. Methods 2019, 22, 394–420. [Google Scholar] [CrossRef]
- Kyriakou, K.; Resch, B.; Sagl, G.; Petutschnig, A.; Werner, C.; Niederseer, D.; Liedlgruber, M.; Wilhelm, F.H.; Osborne, T.; Pykett, J. Detecting moments of stress from measurements of wearable physiological sensors. Sensors 2019, 19, 3805. [Google Scholar] [CrossRef]
- Arnsten, A.F. Catecholamine modulation of prefrontal cortical cognitive function. Trends Cogn. Sci. 1998, 2, 436–447. [Google Scholar] [CrossRef]
- Robbins, T.W.; Arnsten, A.F. The neuropsychopharmacology of fronto-executive function: Monoaminergic modulation. Annu. Rev. Neurosci. 2009, 32, 267–287. [Google Scholar] [CrossRef]
- Blokland, A.; Van Duinen, M.A.; Sambeth, A.; Heckman, P.R.; Tsai, M.; Lahu, G.; Uz, T.; Prickaerts, J. Acute treatment with the pde4 inhibitor roflumilast improves verbal word memory in healthy old individuals: A double-blind placebo-controlled study. Neurobiol. Aging 2019, 77, 37–43. [Google Scholar] [CrossRef] [PubMed]

| Treatment Group | ||||
|---|---|---|---|---|
| Placebo | 430 mg | 860 mg | 1290 mg | |
| Age at Enrolment (years) | 49.39 | 49.35 | 49.52 | 47.21 |
| Gender | F25/M9 | F21/M13 | F23/M10 | F23/M8 |
| Years in Education | 16.84 | 16.88 | 17.14 | 15.28 |
| Portions of Fruit & Veg | 4.23 | 4.15 | 4.59 | 4.32 |
| Alcohol consumption daily (units) | 0.99 | 0.99 | 0.66 | 0.83 |
| Caffeine Consumption (mg/day) | 207 | 194 | 219 | 243 |
| Blood Pressure—Systolic | 122 | 122 | 119 | 124 |
| Blood Pressure—Diastolic | 80 | 81 | 78 | 80 |
| Heart Rate (bpm) | 72 | 75 | 72 | 69 |
| Body Mass Index | 26.43 | 26.87 | 25.45 | 25.93 |
| Treatment guess (% placebo) | 47% | 52% | 36% | 38% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kennedy, D.O.; Bonnländer, B.; Lang, S.C.; Pischel, I.; Forster, J.; Khan, J.; Jackson, P.A.; Wightman, E.L. Acute and Chronic Effects of Green Oat (Avena sativa) Extract on Cognitive Function and Mood during a Laboratory Stressor in Healthy Adults: A Randomised, Double-Blind, Placebo-Controlled Study in Healthy Humans. Nutrients 2020, 12, 1598. https://doi.org/10.3390/nu12061598
Kennedy DO, Bonnländer B, Lang SC, Pischel I, Forster J, Khan J, Jackson PA, Wightman EL. Acute and Chronic Effects of Green Oat (Avena sativa) Extract on Cognitive Function and Mood during a Laboratory Stressor in Healthy Adults: A Randomised, Double-Blind, Placebo-Controlled Study in Healthy Humans. Nutrients. 2020; 12(6):1598. https://doi.org/10.3390/nu12061598
Chicago/Turabian StyleKennedy, David O., Bernd Bonnländer, Stefanie C. Lang, Ivo Pischel, Joanne Forster, Julie Khan, Philippa A. Jackson, and Emma L. Wightman. 2020. "Acute and Chronic Effects of Green Oat (Avena sativa) Extract on Cognitive Function and Mood during a Laboratory Stressor in Healthy Adults: A Randomised, Double-Blind, Placebo-Controlled Study in Healthy Humans" Nutrients 12, no. 6: 1598. https://doi.org/10.3390/nu12061598
APA StyleKennedy, D. O., Bonnländer, B., Lang, S. C., Pischel, I., Forster, J., Khan, J., Jackson, P. A., & Wightman, E. L. (2020). Acute and Chronic Effects of Green Oat (Avena sativa) Extract on Cognitive Function and Mood during a Laboratory Stressor in Healthy Adults: A Randomised, Double-Blind, Placebo-Controlled Study in Healthy Humans. Nutrients, 12(6), 1598. https://doi.org/10.3390/nu12061598





