Supplementation with the Leucine Metabolite β-hydroxy-β-methylbutyrate (HMB) does not Improve Resistance Exercise-Induced Changes in Body Composition or Strength in Young Subjects: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Systematic Search Strategy
2.3. Data Extraction and Outcome Measures
2.4. Risk of Bias Analysis and Sensitivity Analysis
2.5. Data Syntheses and Meta-Analysis
3. Results
3.1. Included Studies
3.2. Risk of Bias and Asymmetry Analysis
3.3. Study Characteristics
3.4. Body Mass and Composition
3.5. Gains in Muscle Strength
3.6. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Van Koevering, M.; Nissen, S. Oxidation of leucine and alpha-ketoisocaproate to beta-hydroxy-beta-methylbutyrate in vivo. Am. J. Physiol. 1992, 262, E27–E31. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.; Sharp, R.; Ray, M.; Rathmacher, J.A.; Rice, D.; Fuller, J.C., Jr.; Connelly, A.S.; Abumrad, N. Effect of leucine metabolite beta-hydroxy-beta-methylbutyrate on muscle metabolism during resistance-exercise training. J. Appl. Physiol. 1996, 81, 2095–2104. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Ferreira, M.; Wilson, M.; Almada, A.L. Effects of calcium beta-hydroxy-beta-methylbutyrate (HMB) supplementation during resistance-training on markers of catabolism, body composition and strength. Int. J. Sports Med. 1999, 20, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Panton, L.B.; Rathmacher, J.A.; Baier, S.; Nissen, S. Nutritional supplementation of the leucine metabolite beta-hydroxy-beta-methylbutyrate (hmb) during resistance training. Nutrition 2000, 16, 734–739. [Google Scholar] [CrossRef]
- Kreider, R.B.; Ferreira, M.; Greenwood, M.; Wilson, M.; Grindstaff, P.; Plisk, J.; Reinardy, J.; Cantler, E.; Almada, A.L. Effects of Calcium β-HMB Supplementation During Training on Markers of Catabolism, Body Composition, Strength and Sprint Performance. J. Exerc. Physiol. Online 2000, 3, 11. [Google Scholar]
- Slater, G.; Jenkins, D.; Logan, P.; Lee, H.; Vukovich, M.; Rathmacher, J.A.; Hahn, A.G. Beta-hydroxy-beta-methylbutyrate (HMB) supplementation does not affect changes in strength or body composition during resistance training in trained men. Int. J. Sport Nutr. Exerc. Metab. 2001, 11, 384–396. [Google Scholar] [CrossRef]
- Rowlands, D.S.; Thomson, J.S. Effects of beta-hydroxy-beta-methylbutyrate supplementation during resistance training on strength, body composition, and muscle damage in trained and untrained young men: A meta-analysis. J. Strength Cond. Res. 2009, 23, 836–846. [Google Scholar] [CrossRef]
- Teixeira, F.J.; Matias, C.N.; Monteiro, C.P.; Valamatos, M.J.; Reis, J.F.; Batista, A.; Oliveira, A.C.; Alves, F.; Sardinha, L.B.; Phillips, S.M. No effect of HMB or alpha-HICA supplementation on training-induced changes in body composition. Eur. J. Sport Sci. 2019, 19, 802–810. [Google Scholar] [CrossRef]
- Jakubowski, J.S.; Wong, E.P.T.; Nunes, E.A.; Noguchi, K.S.; Vandeweerd, J.K.; Murphy, K.T.; Morton, R.W.; McGlory, C.; Phillips, S.M. Equivalent Hypertrophy and Strength Gains in beta-Hydroxy-beta-Methylbutyrate- or Leucine-supplemented Men. Med. Sci. Sports Exerc. 2019, 51, 65–74. [Google Scholar] [CrossRef]
- Asadi, A.; Arazi, H.; Suzuki, K. Effects of beta-Hydroxy-beta-methylbutyrate-free Acid Supplementation on Strength, Power and Hormonal Adaptations Following Resistance Training. Nutrients 2017, 9, 1316. [Google Scholar] [CrossRef]
- Sanchez-Martinez, J.; Santos-Lozano, A.; Garcia-Hermoso, A.; Sadarangani, K.P.; Cristi-Montero, C. Effects of beta-hydroxy-beta-methylbutyrate supplementation on strength and body composition in trained and competitive athletes: A meta-analysis of randomized controlled trials. J. Sci. Med. Sport 2018, 21, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, F.J.; Matias, C.N.; Monteiro, C.P.; Valamatos, M.J.; Reis, J.F.; Tavares, F.; Batista, A.; Domingos, C.; Alves, F.; Sardinha, L.B.; et al. Leucine Metabolites Do Not Enhance Training-induced Performance or Muscle Thickness. Med. Sci. Sports Exerc. 2019, 51, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, G.D.; Rosa, J.C.; Lira, F.S.; Zanchi, N.E.; Ropelle, E.R.; Oyama, L.M.; Oller do Nascimento, C.M.; de Mello, M.T.; Tufik, S.; Santos, R.V. Beta-Hydroxy-beta-methylbutyrate (HMbeta) supplementation stimulates skeletal muscle hypertrophy in rats via the mTOR pathway. Nutr. Metab. (Lond.) 2011, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, C.H.; Gerlinger-Romero, F.; Guimaraes-Ferreira, L.; de Souza, A.L., Jr.; Vitzel, K.F.; Nachbar, R.T.; Nunes, M.T.; Curi, R. Metabolic and functional effects of beta-hydroxy-beta-methylbutyrate (HMB) supplementation in skeletal muscle. Eur. J. Appl. Physiol. 2012, 112, 2531–2537. [Google Scholar] [CrossRef]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite beta-hydroxy-beta-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef]
- Wilkinson, D.J.; Hossain, T.; Limb, M.C.; Phillips, B.E.; Lund, J.; Williams, J.P.; Brook, M.S.; Cegielski, J.; Philp, A.; Ashcroft, S.; et al. Impact of the calcium form of beta-hydroxy-beta-methylbutyrate upon human skeletal muscle protein metabolism. Clin. Nutr. 2018, 37, 2068–2075. [Google Scholar] [CrossRef]
- Giron, M.D.; Vilchez, J.D.; Salto, R.; Manzano, M.; Sevillano, N.; Campos, N.; Argiles, J.M.; Rueda, R.; Lopez-Pedrosa, J.M. Conversion of leucine to beta-hydroxy-beta-methylbutyrate by alpha-keto isocaproate dioxygenase is required for a potent stimulation of protein synthesis in L6 rat myotubes. J. Cachexia Sarcopenia Muscle 2016, 7, 68–78. [Google Scholar] [CrossRef]
- Kimura, K.; Cheng, X.W.; Inoue, A.; Hu, L.; Koike, T.; Kuzuya, M. Beta-Hydroxy-beta-methylbutyrate facilitates PI3K/Akt-dependent mammalian target of rapamycin and FoxO1/3a phosphorylations and alleviates tumor necrosis factor alpha/interferon gamma-induced MuRF-1 expression in C2C12 cells. Nutr. Res. 2014, 34, 368–374. [Google Scholar] [CrossRef]
- Gerlinger-Romero, F.; Guimaraes-Ferreira, L.; Yonamine, C.Y.; Salgueiro, R.B.; Nunes, M.T. Effects of beta-hydroxy-beta-methylbutyrate (HMB) on the expression of ubiquitin ligases, protein synthesis pathways and contractile function in extensor digitorum longus (EDL) of fed and fasting rats. J. Physiol. Sci. 2018, 68, 165–174. [Google Scholar] [CrossRef]
- Zanchi, N.E.; Gerlinger-Romero, F.; Guimaraes-Ferreira, L.; de Siqueira Filho, M.A.; Felitti, V.; Lira, F.S.; Seelaender, M.; Lancha, A.H., Jr. HMB supplementation: Clinical and athletic performance-related effects and mechanisms of action. Amino Acids 2011, 40, 1015–1025. [Google Scholar] [CrossRef]
- Holecek, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopenia Muscle 2017, 8, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Gepner, Y.; Varanoske, A.N.; Boffey, D.; Hoffman, J.R. Benefits of beta-hydroxy-beta-methylbutyrate supplementation in trained and untrained individuals. Res. Sports Med. 2019, 27, 204–218. [Google Scholar] [CrossRef] [PubMed]
- Portal, S.; Eliakim, A.; Nemet, D.; Halevy, O.; Zadik, Z. Effect of HMB supplementation on body composition, fitness, hormonal profile and muscle damage indices. J. Pediatr. Endocrinol. Metab. 2010, 23, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.R.; Belozo, F.L.; Micheletti, T.O.; Conrado, M.; Stout, J.R.; Pimentel, G.D.; Gonzalez, A.M. Beta-hydroxy-beta-methylbutyrate free acid supplementation may improve recovery and muscle adaptations after resistance training: A systematic review. Nutr. Res. 2017, 45, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Landa, J.; Calleja-Gonzalez, J.; Leon-Guereno, P.; Caballero-Garcia, A.; Cordova, A.; Mielgo-Ayuso, J. Effect of the Combination of Creatine Monohydrate Plus HMB Supplementation on Sports Performance, Body Composition, Markers of Muscle Damage and Hormone Status: A Systematic Review. Nutrients 2019, 11, 2528. [Google Scholar] [CrossRef]
- Chilibeck, P.D.; Kaviani, M.; Candow, D.G.; Zello, G.A. Effect of creatine supplementation during resistance training on lean tissue mass and muscular strength in older adults: A meta-analysis. Open Access J. Sports Med. 2017, 8, 213–226. [Google Scholar] [CrossRef]
- Nissen, S.L.; Sharp, R.L. Effect of dietary supplements on lean mass and strength gains with resistance exercise: A meta-analysis. J. Appl. Physiol. (1985) 2003, 94, 651–659. [Google Scholar] [CrossRef]
- Holland, B.M.; Roberts, B.M.; Krieger, J.W.; Schoenfeld, B.J. Does HMB Enhance Body Composition in Athletes? A Systematic Review and Meta-analysis. J. Strength Cond. Res. 2019, 20, 1–8. [Google Scholar] [CrossRef]
- Fuller, J.C.; Sharp, R.L.; Angus, H.F.; Khoo, P.Y.; Rathmacher, J.A. Comparison of availability and plasma clearance rates of beta-hydroxy-beta-methylbutyrate delivery in the free acid and calcium salt forms. Br. J. Nutr. 2015, 114, 1403–1409. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019); Cochrane: London, UK, 2019; Available online: www.training.cochrane.org/handbook (accessed on 1 February 2020).
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Sterne, J.A.; Sutton, A.J.; Ioannidis, J.P.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rucker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, d4002. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.; Brozek, J.; Guyatt, G.; Oxman, A. GRADE Handbook for Grading Quality of Evidence and Strength of Recommendations. 2013; Available online: https://training.cochrane.org/resource/grade-handbook (accessed on 1 February 2020).
- The Nordic Cochrane Centre. RevMan Review Manager (RevMan) Version 5.3; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2014. [Google Scholar]
- Kraemer, W.J.; Hatfield, D.L.; Volek, J.S.; Fragala, M.S.; Vingren, J.L.; Anderson, J.M.; Spiering, B.A.; Thomas, G.A.; Ho, J.Y.; Quann, E.E.; et al. Effects of amino acids supplement on physiological adaptations to resistance training. Med. Sci. Sports Exerc. 2009, 41, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Lowery, R.P.; Joy, J.M.; Andersen, J.C.; Wilson, S.M.; Stout, J.R.; Duncan, N.; Fuller, J.C.; Baier, S.M.; Naimo, M.A.; et al. The effects of 12 weeks of beta-hydroxy-beta-methylbutyrate free acid supplementation on muscle mass, strength, and power in resistance-trained individuals: A randomized, double-blind, placebo-controlled study. Eur. J. Appl. Physiol. 2014, 114, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.S.; Watson, P.E.; Rowlands, D.S. Effects of nine weeks of beta-hydroxy-beta- methylbutyrate supplementation on strength and body composition in resistance trained men. J. Strength Cond. Res. 2009, 23, 827–835. [Google Scholar] [CrossRef]
- Tritto, A.C.; Bueno, S.; Rodrigues, R.M.P.; Gualano, B.; Roschel, H.; Artioli, G.G. Negligible Effects of beta-Hydroxy-beta-Methylbutyrate Free Acid and Calcium Salt on Strength and Hypertrophic Responses to Resistance Training: A Randomized, Placebo-Controlled Study. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 505–511. [Google Scholar] [CrossRef]
- Stahn, A.C.; Maggioni, M.A.; Gunga, H.C.; Terblanche, E. Combined protein and calcium beta-hydroxy-beta-methylbutyrate induced gains in leg fat free mass: A double-blinded, placebo-controlled study. J. Int. Soc. Sports Nutr. 2020, 17, 16. [Google Scholar] [CrossRef]
- Damas, F.; Phillips, S.M.; Libardi, C.A.; Vechin, F.C.; Lixandrao, M.E.; Jannig, P.R.; Costa, L.A.; Bacurau, A.V.; Snijders, T.; Parise, G.; et al. Resistance training-induced changes in integrated myofibrillar protein synthesis are related to hypertrophy only after attenuation of muscle damage. J. Physiol. 2016, 594, 5209–5222. [Google Scholar] [CrossRef]
- Lowery, R.P.; Joy, J.M.; Rathmacher, J.A.; Baier, S.M.; Fuller, J.C., Jr.; Shelley, M.C., 2nd; Jager, R.; Purpura, M.; Wilson, S.M.; Wilson, J.M. Interaction of Beta-Hydroxy-Beta-Methylbutyrate Free Acid and Adenosine Triphosphate on Muscle Mass, Strength, and Power in Resistance Trained Individuals. J. Strength Cond. Res. 2016, 30, 1843–1854. [Google Scholar] [CrossRef]
- Bhasin, S.; Storer, T.W.; Berman, N.; Callegari, C.; Clevenger, B.; Phillips, J.; Bunnell, T.J.; Tricker, R.; Shirazi, A.; Casaburi, R. The effects of supraphysiologic doses of testosterone on muscle size and strength in normal men. N. Engl. J. Med. 1996, 335, 1–7. [Google Scholar] [CrossRef]
- Shirato, M.; Tsuchiya, Y.; Sato, T.; Hamano, S.; Gushiken, T.; Kimura, N.; Ochi, E. Effects of combined beta-hydroxy-beta-methylbutyrate (HMB) and whey protein ingestion on symptoms of eccentric exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2016, 13, 7. [Google Scholar] [CrossRef]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Bear, D.E.; Langan, A.; Dimidi, E.; Wandrag, L.; Harridge, S.D.R.; Hart, N.; Connolly, B.; Whelan, K. β-Hydroxy-β-methylbutyrate and its impact on skeletal muscle mass and physical function in clinical practice: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2019, 109, 1119–1132. [Google Scholar] [CrossRef] [PubMed]
- Courel-Ibáñez, J.; Vetrovsky, T.; Dadova, K.; Pallarés, J.G.; Steffl, M. Health Benefits of β-Hydroxy-β-Methylbutyrate (HMB) Supplementation in Addition to Physical Exercise in Older Adults: A Systematic Review with Meta-Analysis. Nutrients 2019, 11, 2082. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.H.; Stout, J.R.; Miramonti, A.A.; Fukuda, D.H.; Wang, R.; Townsend, J.R.; Mangine, G.T.; Fragala, M.S.; Hoffman, J.R. High-intensity interval training and β-hydroxy-β-methylbutyric free acid improves aerobic power and metabolic thresholds. J. Int. Soc. Sports Nutr. 2014, 11, 16. [Google Scholar] [CrossRef]
- Durkalec-Michalski, K.; Jeszka, J.; Podgórski, T. The Effect of a 12-Week Beta-hydroxy-beta-methylbutyrate (HMB) Supplementation on Highly-Trained Combat Sports Athletes: A Randomised, Double-Blind, Placebo-Controlled Crossover Study. Nutrients 2017, 9, 753. [Google Scholar] [CrossRef]
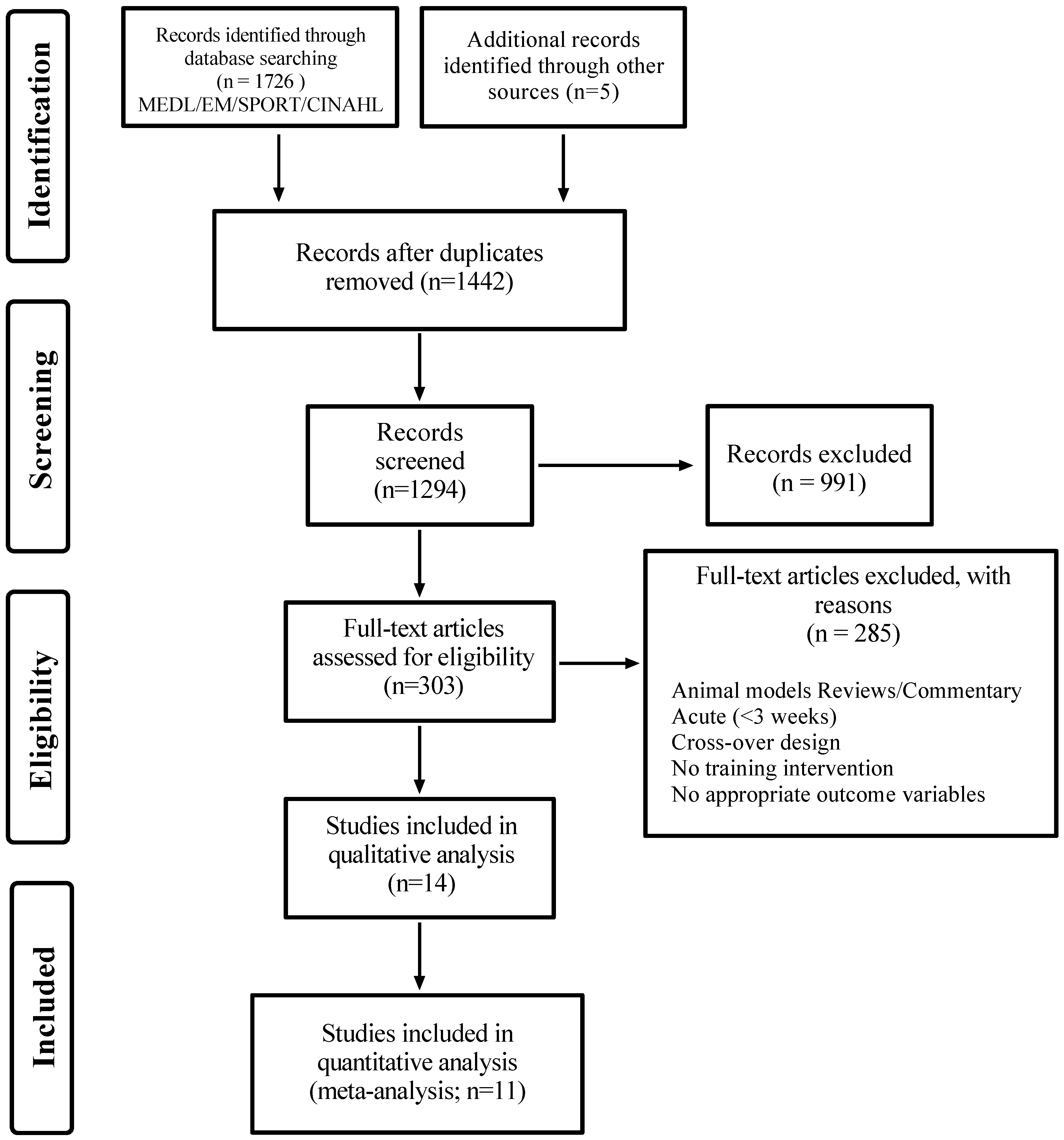
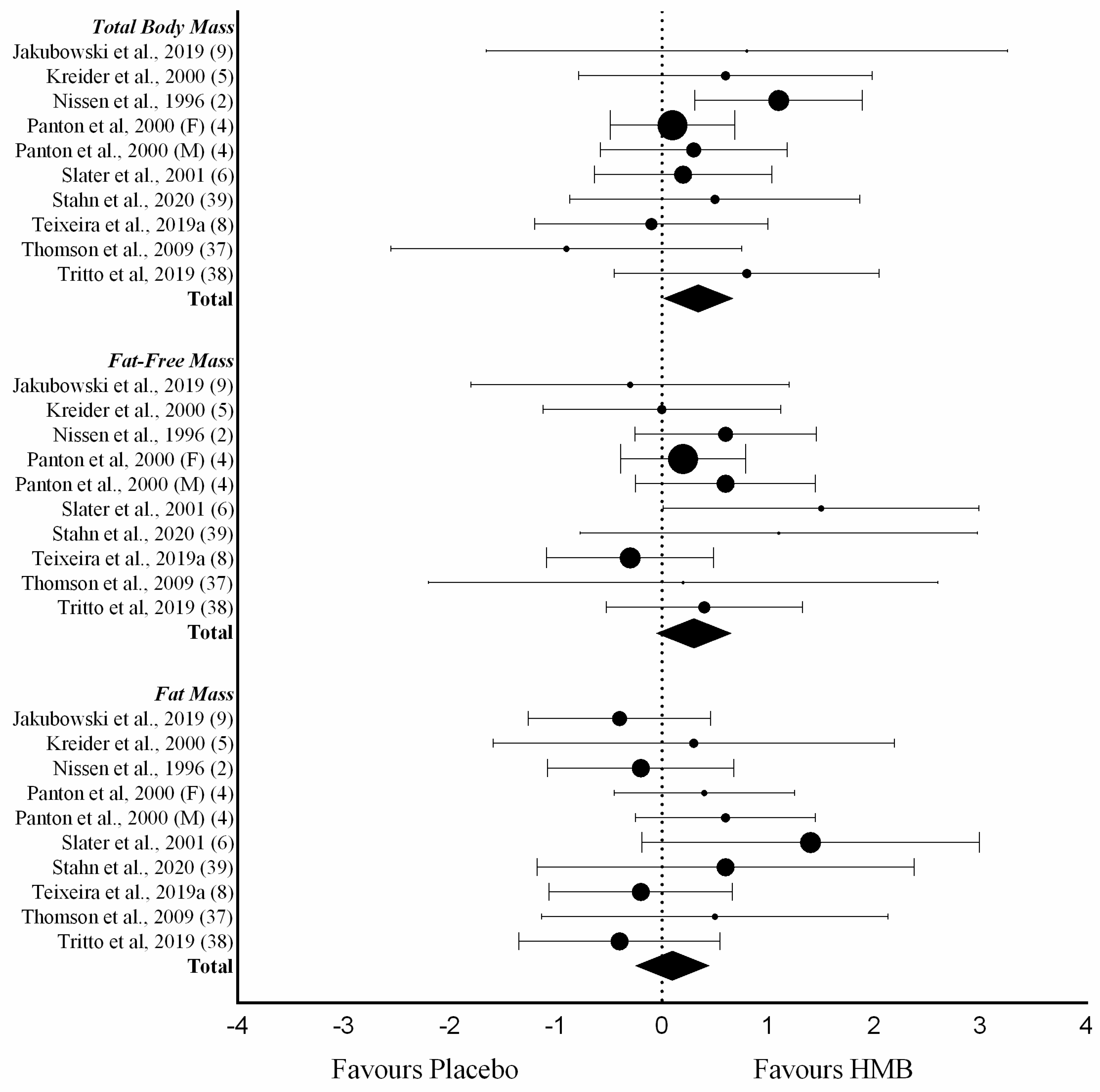
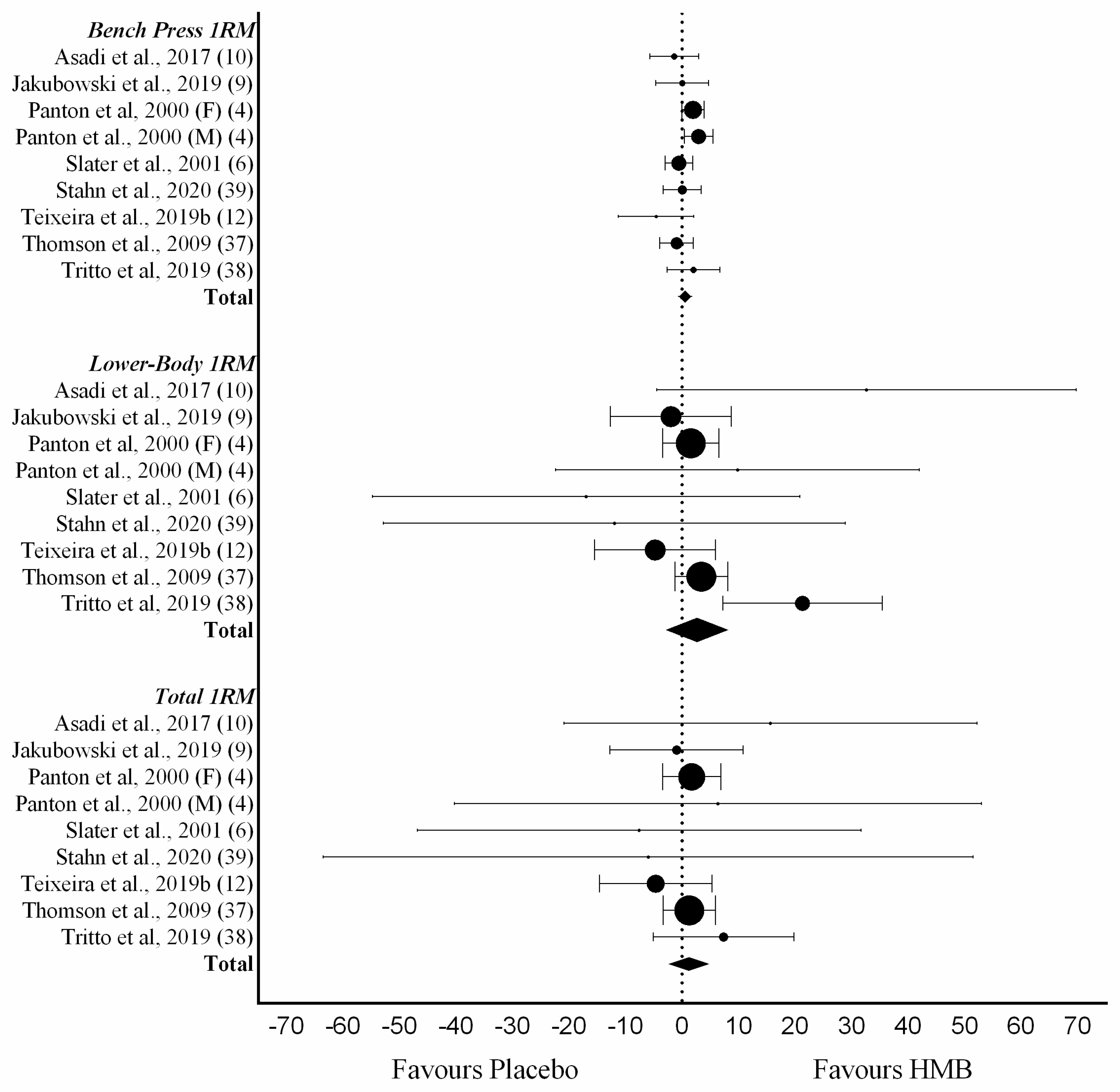
| Study | Objective | Criteria | Included Studies | Supplement | Outcome | Conclusion | Lean Mass | Strength | Effect Size | |
|---|---|---|---|---|---|---|---|---|---|---|
| Lean Mass | Strength | |||||||||
| Nissen and Sharp (2003) [27] | To quantify dietary supplements to augment lean mass and strength gains during Resistance Exercise Training (RET) | Randomized clinical trial (RCT) Duration: Full Body RET ≥ 3 wk Frequency: ≥ 2 x/wk | 9 studies - Nissen et al. (1996) [2] included as 2 studies | 3 g HMB-Ca | Lean Mass Total Strength | There is sufficient data to support the use of HMB to augment lean mass and strength gains with RET | HMB results in an increase of 0.28%/wk confidence interval (CI): 0.13% to 0.42% | HMB results in an increase of 1.40%/wk CI: 0.41% to 2.39% | Trivial effect size (ES): 0.15 CI: 0.06 to 0.24 p < 0.005 | Trivial ES: 0.19 CI: 0.09–0.29 p < 0.01 |
| Rowlands and Thomson (2007) [7] | To meta-analyze the effectiveness of HMB on strength, body composition, and muscle damage in trained (TR) and untrained (UT) participants during RET | RCT 8/9 Crossover 1/9 TR or UT men No criteria for duration | 9 studies N = 394 | 1/9 HMB + drink (whey protein-carbohydrate, vitamins, minerals, glutamine, and chromium picolinate) 1/9 HMB+ glucose+ taurine+ disodium phosphate+ potassium phosphate 2/9 HMB + 50 mg of potassium phosphate | Lean Mass Strength Creatine kinase (CK) | HMB supplementation results in a small beneficial increase to overall strength in UT lifters but has a negligible effect on TR lifters. In UT participants HMB results in a small increase in lower-body strength, In both trained and untrained lifters, the effect of HMB supplementation on body composition is negligible. | Fat-free mass (FFM) increases and changes in fat mass (FM) in UT and TR lifters were negligible | UT: Small benefit Lower-body: 9.9% ± 5.9% Average strength: 6.6% ± 5.7% Negligible gains Upper-body strength: 2.1% ± 5.5% TR: All outcomes Trivial UT and TR combined: Trivial Average strength: 3.7% ± 2.4% | Trivial 3-unit increase of HMB daily dose ES: −0.07 ± 0.17 | Trivial 3-unit increase of HMB daily dose ES: 0.02 ± 0.2 |
| Sanchez-Martinez et al. (2018) [11] | Examine the effectiveness of HMB supplementation on strength and body composition in trained athletes | RCT or cross-over TR (≥ 1 RET) or competitive athletes | 6 studies - Slater et al. (2001) [6] included as 4 studies - Kreider et al. (1999) [3] included as 2 studies | 5/6 RCT 1/6 crossover 4/6 3 g HMB-Ca 2/6 did not specify HMB-FA/HMB-Ca 1/6 HMB + drink (proteins, carbohydrates, vitamins, minerals) 1/7 HMB+ L-Carnitine, Choline, Boron and Garcinia Cambogia | Body mass Fat-free mass Fat mass Bench Leg press | No effect of HMB on strength and body composition in competitive athletes | HMB has a negligible effect on body composition and strength in trained and competitive athletes | Body mass ES = −0.01, CI: −0.29 to 0.27 Fat free mass ES = 0.16, CI: −0.5 to 0.46 | Bench press ES = 0.0, CI: −0.32 to 0.32 Leg press ES = −0.09, CI: 0.46 to 0.28 | |
| Holland et al. 2019 [28] | Determine the effects of HMB on body composition in athletes | RCT Minimum supplement period of 4 wk | Body Mass: 7 studies, N = 208 Fat Free and Fat Mass 5 studies, N = 161 and N = 128, respectively | HMB * Does not differentiate HMB-Ca vs. HMB-FA | Body Mass Fat Free Mass Fat Mass | HMB may have a small, positive impact on FFM in athletes when protein intake is suboptimal (<1.6 g/kg/day) | There was no significant effect of HMB on FFM, although the CI was skewed in favor of a small effect There was no significant effect of HMB on BM | ES = −0.30 ± 0.13; 95% CI: 20.07 to 0.68 (p = 0.00) | Body Mass ES = 20.02 ± 0.04; 95% CI: 20.14 to 0.10 (p = 0.70) | |
| Study | Country | Design | Participants | Intervention | Outcome Measure | Dietary Assessment and Protein Ingestion | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | Age | Training Status | Dose or Placebo | Duration (Weeks) | Training | HMB n | Control n | Strength | Body Composition | ||||||
| Upper Body | Lower Body | Fat-free mass | Fat mass | ||||||||||||
| Asadi et al., 2017 [10] | Japan | Randomized controlled trial (RCT) Double blinded (DB) | Male(M) | 21.4 ± 0.7 | Not Described | 3 g HMB-FA or polydextrose | 6 | 2 x/w 3 sets of 8–12 repetitions per set (rep) at 75–85% of 1 repetition maximum (RM) | 8 | 8 | Bench press | Leg Press | --- | --- | 3-day diet records week 0 and week 6 Protein ingestion ~1.45 g/kg/d |
| Jakubowski et al., 2019 [9] | Canada | RCT DB | M | 22.5 ± 2.2 | Trained (TR) (Recreationally trained—2x/week) | 3 g HMB-Ca + 50 g Whey Protein or 50 g Whey Protein | 12 | 3–5 x/w Phase 1: 8 w Undulating periodized resistance-training (UPRT) Phase 2: 2 w overreaching Phase 3: 2 w | 13 | 13 | Bench press | Squat | Dual X-ray absorptiometry (DXA) | DXA | 3-day diet records weeks 0,8 and 12. Protein ingestion ~1.8–1.9 g/kg/d |
| Kreider et al. (2000) [5] | USA | RCT DB | M | 20.0 ± 1.5 | TR | 3 g HMB-Ca, 99 g/d of glucose, 1.1 g Na2HPO4, 1.2 K2PO4 and 3 g/d of Taurine or a placebo without HMB-Ca | 4 | 4 x/week, 1 to 3 sets of 2–8 rep, 60 to 95% of 1RM (+ 3x week of agility /sprint training) | 14 | 14 | Bench Press | Squat | DXA | DXA | 4-day nutritional intake assessment day 0 and day 28. Protein ingestion 1.5–2.4 g/kg/d |
| Nissen et al., 1996 [2] | USA | RCT DB? (not clear) | M | 19–29 | Untrained (UT) (at least 3 months) | 3 g HMB-Ca + MET-Rx (37 g milk protein) or MET-Rx | 7 | 3 x/w 3 sets of 3–5 rep at 90% of 1RM. | 14 | 14 | Several upper body exercises | Several lower body exercises | Total body electrical conductivity (TOBEC) | TOBEC | No dietary assessment for study 2. Estimated protein intake ~1.8–2 g/kg/d |
| Panton et al., 2000 [4] | USA | RCT DB | M/Female(F) | 25 ± 1.2 (M) 23 ± 0.6 (F) | TR (> 6 months experience)/UT (not training for at least 6 months) | 3 g HMB-Ca or rice flour | 4 | 3 x/w. 3–6 rep 90% 1RM. | 39 (21M/18F) | 36 (18M/18F) | Bench press | Leg Press (M) Leg Extension (F) | UWW | UWW | Not described |
| Slater et al. (2001) [6] | Australia | RCT DB | M | 24.5 ± 1.7 | TR | 3 g HMB-Ca (Standard encapsulation vs. Time Release) or rice flour | 6 | 2–3 x/w. 4–6 repetitions for 3–5 sets (24 to 32 sets per session) | 7 | 7 | Bench press | Leg Press | DXA | DXA | Regularly dietary logs Pre 1.7 g/kg/d Post 2.4 g/kg/d |
| Stahn et al. (2020) [39] | USA | RCT DB | M | 22.1 ± 1.5 | UT (for the past 6 months) | 3 g HMB-Ca + 30 g Whey Protein (daily). +30 g carbohydrate supplement only on training days or the supplements + microcrystalline cellulose as placebo | 12 | 4x/w upper/lower body split routine. Weeks 1–6: linear periodization. Week 7 tapering. Weeks 8–12: undulating periodization | 8 | 7 | Bench Press | Leg Press | Bioelectrical impedance (BIA) | BIA | No dietary assessment |
| Teixeira et al., 2019a [8] | Portugal | RCT DB | M | 31.7 ± 7.6 | TR (>1 year experience) | 3 g HMB-Ca or 3 g HMB-FA or Mg stearate as placebo | 8 | 3 x/w Weeks 1–3, 3–4 sets 12RM Weeks 4–6, 3–4 sets 10RM Weeks 7–8, 4 sets 8RM | 20 | 10 | --- | --- | DXA | DXA | 3-day dietary logs weeks 0, 4 and 8. Dietary instructions to adjust energy and protein ingestion. Protein ingestion 3.0–3.3 g/kg/d |
| Teixeira et al., 2019b [12] | Portugal | RCT DB | M | 31.7 ± 7.6 | TR (>1 year experience) | 3 g HMB-Ca or 3 g HMB-FA or Mg stearate as placebo | 8 | 3 x/w Weeks 1–3, 3–4 sets 12RM Weeks 4–6, 3–4 sets 10RM Weeks 7–8, 4 sets 8RM | 20 | 10 | Bench Press | Squat | --- | --- | 3-day dietary logs weeks 0, 4 and 8. Dietary counselling to adjust energy and protein ingestion Protein ingestion 3.0–3.3 g/kg/d |
| Thomson et al., 2009 [37] | New Zealand | RCT DB | M | 24 ± 4 | TR (>3 years experience) | 3 g HMB-Ca or corn starch | 9 | 3 x/w | 13 | 9 | Bench press | Leg extension | BIA | BIA | Diets evaluated by 3-day records at weeks 0 and 9. Protein ingestion: not stated. |
| Tritto et al., 2019 [38] | Brazil | RCT DB | M | 25.3 ± 3.7 | TR (>6 months experience) | 3 g HMB-Ca or 3 g HMB-FA or corn starch | 12 | 2 x /w 3–4 sets 8–10 RM | 29 | 15 | Bench press | Leg Press | DXA | DXA | Diets evaluated by 3-day records at weeks 0 and 12. Protein ingestion: 1.9–2.1 g/kg/d. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakubowski, J.S.; Nunes, E.A.; Teixeira, F.J.; Vescio, V.; Morton, R.W.; Banfield, L.; Phillips, S.M. Supplementation with the Leucine Metabolite β-hydroxy-β-methylbutyrate (HMB) does not Improve Resistance Exercise-Induced Changes in Body Composition or Strength in Young Subjects: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 1523. https://doi.org/10.3390/nu12051523
Jakubowski JS, Nunes EA, Teixeira FJ, Vescio V, Morton RW, Banfield L, Phillips SM. Supplementation with the Leucine Metabolite β-hydroxy-β-methylbutyrate (HMB) does not Improve Resistance Exercise-Induced Changes in Body Composition or Strength in Young Subjects: A Systematic Review and Meta-Analysis. Nutrients. 2020; 12(5):1523. https://doi.org/10.3390/nu12051523
Chicago/Turabian StyleJakubowski, Josephine S., Everson A. Nunes, Filipe J. Teixeira, Victoria Vescio, Robert W. Morton, Laura Banfield, and Stuart M. Phillips. 2020. "Supplementation with the Leucine Metabolite β-hydroxy-β-methylbutyrate (HMB) does not Improve Resistance Exercise-Induced Changes in Body Composition or Strength in Young Subjects: A Systematic Review and Meta-Analysis" Nutrients 12, no. 5: 1523. https://doi.org/10.3390/nu12051523
APA StyleJakubowski, J. S., Nunes, E. A., Teixeira, F. J., Vescio, V., Morton, R. W., Banfield, L., & Phillips, S. M. (2020). Supplementation with the Leucine Metabolite β-hydroxy-β-methylbutyrate (HMB) does not Improve Resistance Exercise-Induced Changes in Body Composition or Strength in Young Subjects: A Systematic Review and Meta-Analysis. Nutrients, 12(5), 1523. https://doi.org/10.3390/nu12051523







