Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study
Abstract
1. Introduction
2. Methods
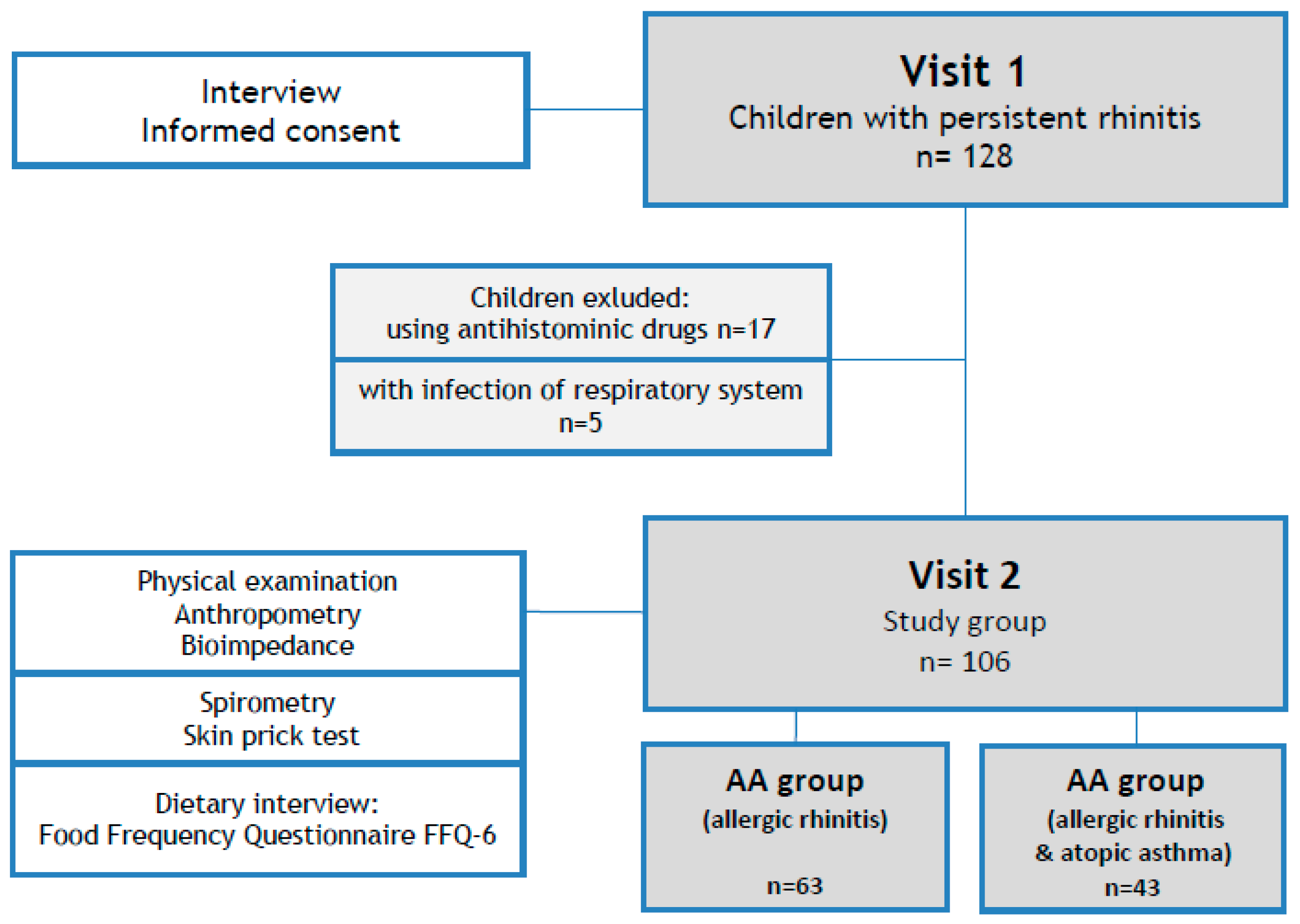
2.1. Study Design
2.2. Patients
2.3. Nutritional Habits
2.4. Physical Activity
2.5. Nutritional Status
2.6. Statistical Analysis
3. Results
3.1. Patients
3.2. Eating Habits and Physical Activity
3.2.1. Number of Meals
3.2.2. Sweets
3.2.3. Snacks
3.2.4. Fast Food
3.2.5. Meals before Bedtime
3.3. Physical Activity
3.4. Nutritional Status and Body Composition
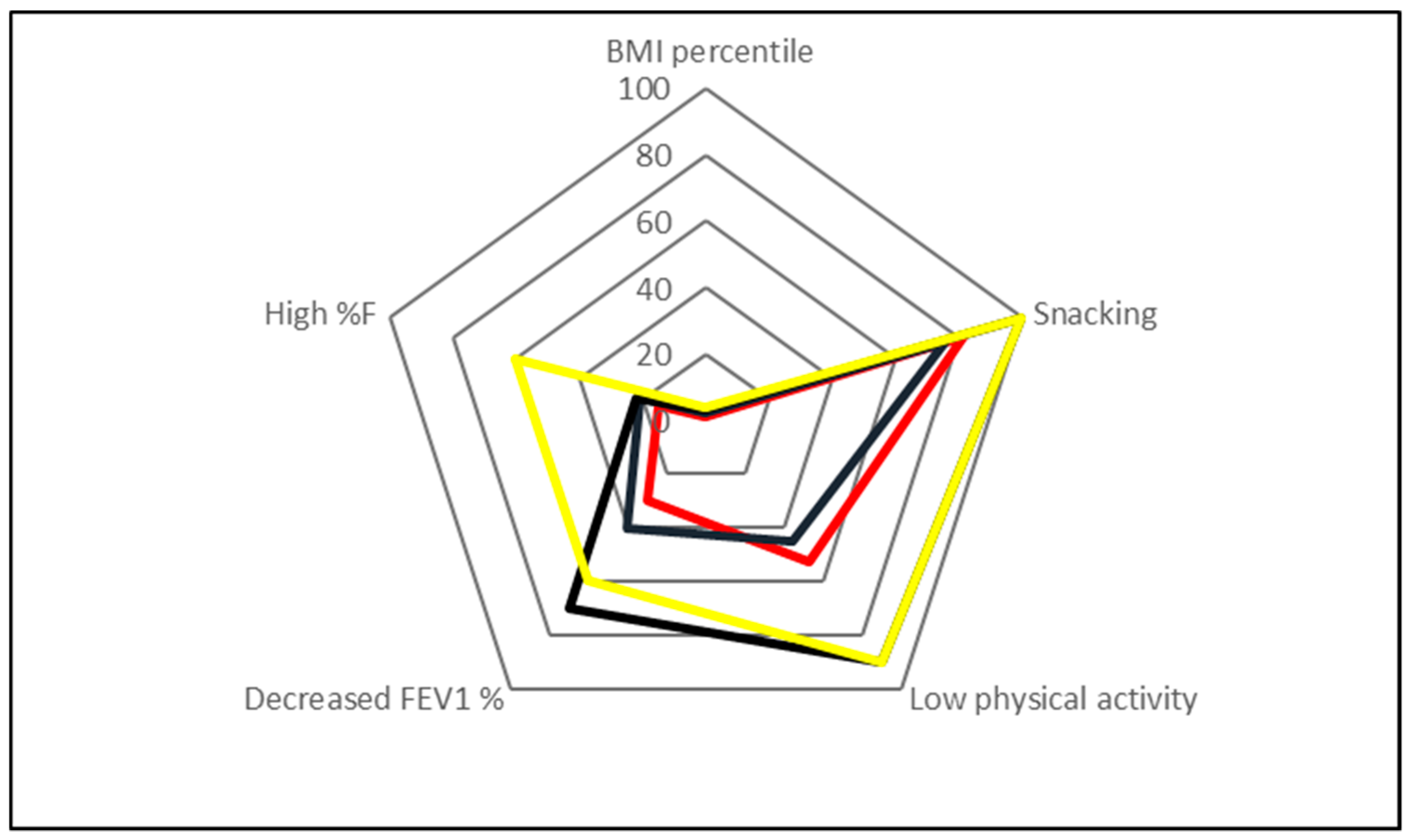
3.5. The Multifactorial Linear Regression Analysis
4. Discussion
4.1. Nutritional Status
4.2. Dietary Habits
4.3. Physical Activity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- McNeill, G.; Tagiyeva, N.; Aucott, L.; Russell, G.; Helms, P.J. Changes in the prevalence of asthma, eczema and hay fever in pre-pubertal children: A 40-year perspective. Paediatr. Perinatal. Epidemiol. 2009, 23, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. Global Initiative for Asthma (GINA) Program. Global burden of asthma developed for the Global Initiative for Asthma. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Samoliński, B.; Sybilski, A.J.; Raciborski, F.; Tomaszewska, A.; Samelkowalik, P.; Walkiewicz, A.; Lusawa, A.; Borowicz, J.; Gutowskaślesik, J.; Trzpil, L.; et al. Prevalence of rhinitis in Polish population according to the ECAP (Epidemiology of Allergic Disorders in Poland) study. Otolaryngol. Pol. 2009, 63, 324–330. [Google Scholar] [CrossRef]
- Bousquet, P.J.; Demoly, P.; Devillier, P.; Mesbah, K.; Bousquet, J. Impact of allergic rhinitis symptoms on quality of life in primary care. Int. Arch. Allergy. Immunol. 2013, 160, 393–400. [Google Scholar] [CrossRef]
- Guerra, S.; Sherrill, D.L.; Martinez, F.D.; Barbee, R.A. Rhinitis is an independent risk factor for adult-onset asthma. J. Allergy Clin. Immunol. 2002, 109, 419–425. [Google Scholar] [CrossRef]
- Bosquet, J.; Van Cauwenberge, P.; Khaltaev, N. ARIA Workshop Group. Allergic rhinitis and its impact on asthma. J. Allergy Clin. Immunol. 2001, 108, 147–334. [Google Scholar] [CrossRef]
- Samoliński, B.; Raciborski, F.; Lipiec, A.; Tomaszewska, A.; Krzych-Fałta, E.; Nowicka, A. Epidemiology of allergic diseases in Poland. Pol. J. Allergol. 2014, 1, 10–18. [Google Scholar]
- Nagel, G.; Weinmayr, G.; Kleiner, A.; Garcia-Marcos, L.; Strachan, D.P. Effect of diet on asthma and allergic sensitisation in the International Study on Allergies and Asthma in Childhood (ISAAC) phase two. Thorax. 2010, 65, 516–522. [Google Scholar] [CrossRef]
- Rosenheck, R. Fast food consumption and increased caloric intake: A systematic review of a trajectory towards weight gain and obesity risk. Obes. Rev. 2008, 9, 535–547. [Google Scholar] [CrossRef]
- Leynaert, B.; Neukirch, C.; Liard, R.; Bousquet, J.; Neukirch, F. Quality of life in allergic rhinitis and asthma: A population-based study of young adults. Am. J. Respir. Crit. Care. Med. 2000, 162, 1391–1396. [Google Scholar] [CrossRef]
- Bernstein, J.A. Allergic and mixed rhinitis: Epidemiology and natural history. Allergy Asthma Proc. 2010, 31, 365–369. [Google Scholar] [CrossRef] [PubMed]
- International Obesity Task Force. Prevalence of overweight and obesity in a number of countries (global prevalence database) [in:] European Union Public Health Information System 2007. Available online: http://www.euro.who.int/__data/assets/pdf_file/0005/96980/2.3.-Prevalence-of-overweight-and-obesity-EDITED_layouted_V3.pdf (accessed on 12 June 2018).
- Oblacinska, A.; Wroclawska, M.; Woynarowska, B. Frequency of overweight and obesity in the school-age population in Poland and health care for pupils with these disorders. Ped. Pol. 1997, 72, 241–245. [Google Scholar]
- Mazur, A.; Malecka-Tendera, E.; Lewin-Kowalik, J. Overweight and obesity in primary school children from the Podkarpatian Region. Ped. Pol. 2001, 76, 743–748. [Google Scholar]
- Lipowicz, A.; Opuszalska, M.; Kolodziej, H.; Szklarska, A.; Bielicki, T. Secular trends in BMI and the prevalence of obesity in young Polish males from 1965 to 2010. Eur. J. Public Health 2015, 25, 279–282. [Google Scholar] [CrossRef][Green Version]
- De, A.; Rastogi, D. Association of pediatric obesity and asthma, pulmonary physiology, metabolic dysregulation, and atopy; and the role of weight management. Expert Rev. Endocrinol. Metab. 2019, 14, 335–349. [Google Scholar] [CrossRef]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2020. Available online: www.ginasthma.org (accessed on 3 April 2018).
- Miller, M.R.; Crapo, R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Enright, P.; van der Grinten, C.P.M.; Gustafsson, P.; et al. General consideration for lung function testing. Eur. Respir. J. 2005, 26, 153–161. [Google Scholar] [CrossRef]
- Niedzwiedzka, E.; Wadolowska, L.; Kowalkowska, J. Reproducibility of a Non-Quantitative Food Frequency Questionnaire (62-Item FFQ-6) and PCA-Driven Dietary Pattern Identification in 13–21-Year-Old Females. Nutrients 2019, 11, 2183. [Google Scholar] [CrossRef]
- Kułaga, Z.; Litwin, M.; Tkaczyk, M.; Palczewska, I.; Zajączkowska, M.; Zwolińska, D.; Krynicki, T.; Wasilewska, A.; Moczulska, A.; Morawiec-Knysak, A.; et al. Pan HPolish 2010 growth references for school-a-ged children and adolescents. Eur. J. Pediatr. 2011, 170, 599–609. [Google Scholar]
- Stankiewicz, M.; Pieszko, M.; Sliwińska, A.; Małgorzewicz, S.; Wierucki, Ł.; Zdrojewski, T.; Wyrzykowski, B.; Łysiak-Szydlowska, W. Obesity and diet awareness among Polish children and adolescents in small towns and villages. Cent. Eur. J. Public Health. 2014, 22, 12–16. [Google Scholar] [CrossRef]
- Chrzanowska, M.; Suder, A. Changes in central fatness and abdominal obesity in children and adolescents from Cracow, Poland 1983–2000. Ann. Hum. Biol. 2010, 37, 242–252. [Google Scholar] [CrossRef]
- Forno, E.; Acosta-Pérez, E.; Brehm, J.; Han, YY.; Alvarez, M.; Colón-Semidey, A.; Canino, G.; Celedón, J. Obesity and adiposity indicators, asthma, and atopy in Puerto Rican children. J. Aller. Clin. Immunol. 2014, 133, 1308–1314. [Google Scholar] [CrossRef]
- Mitchell, E.A.; Stewart, A.W.; Braithwaite, I.; Murphy, R.; Hancox, R.J.; Wall, C.; Beasley, R. ISAAC Phase Three Study Group. Factors associated with body mass index in children and adolescents: An international cross-sectional study. PLoS ONE 2018, 2, e0196221. [Google Scholar] [CrossRef]
- Lang, J.E. Obesity, Nutrition, and Asthma in Children. Ped. Allerg. Immunol. Pulmonol. 2012, 25, 64–75. [Google Scholar] [CrossRef]
- Papoutsakis, C.; Priftis, K.N.; Drakouli, M.; Prifti, S.; Konstantaki, E.; Chondronikola, M.; Antonogeorgos, G.; Matziou, V. Childhood overweight/obesity and asthma: Is there a link? A systematic review of recent epidemiologic evidence. J. Acad. Nutr. Diet. 2013, 113, 77–105. [Google Scholar] [CrossRef]
- Han, Y.Y.; Forno, E.; Gogna, M.; Celedón, J. FAAAAI Obesity and rhinitis in a nationwide study of children and adults in the United States. J. Allerg. Clin. Immunol. 2016, 137, 1460–1465. [Google Scholar] [CrossRef]
- Evans, E.W.; Koinis-Mitchell, D.; Kopel, S.J.; Jelalian, E. Lung Function, Dietary Intake, and Weight Status in Children with Persistent Asthma from Low-Income, Urban Communities. Nutrients 2019, 3, 2943. [Google Scholar] [CrossRef]
- Wang, C.S.; Wang, J.; Zhang, X.; Zhang, L.; Zhang, H.P.; Wang, L.; Wood, L.G.; Wang, G. Systematic Review Free Access Is the consumption of fast foods associated with asthma or other allergic diseases? Respirology 2018, 23, 901–913. [Google Scholar] [CrossRef]
- Wickens, K.; Barry, D.; Friezema, A.; Rhodius, R.; Bone, N.; Purdie, G.; Crane, J. Fast foods—Are they a risk factor for asthma? Allergy 2005, 60, 1537–1541. [Google Scholar] [CrossRef]
- Braithwaite, I.; Stewart, A.; Hancox, R.J.; Beasley, R.; Murphy, R.; Mitchell, E.A. ISAAC Phase Three Study Group. Fast-food consumption and body mass index in children and adolescents: An international cross-sectional study. BMJ Open 2014, 8, 4. [Google Scholar]
- Lawson, J.A.; Rennie, D.C.; Dosman, J.A.; Cammer, A.L.; Senthilselvan, A. Obesity, diet, and activity in relation to asthma and wheeze among rural dwelling children and adolescents. J. Obes. 2013, 2013, 315096. [Google Scholar] [CrossRef]
- Mai, X.M.; Becker, A.B.; Liem, J.J.; Kozyrskyj, A.L. Fast foods consumption counters the protective effect of breastfeeding on asthma in children? Clin. Exp. Allergy 2009, 39, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Hijazi, N.; Abalkhail, B.; Seaton, A. Diet and childhood asthma in a society in transition a study in urban and rural Saudi Arabia. Thorax 2000, 55, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.L.; Elfman, L.; Mi, Y.; Johansson, M.; Smedje, G.; Norbäck, D. Current asthma and respiratory symptoms among pupils in relation to dietary factors and allergens in the school environment. Indoor Air. 2005, 15, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Marcos, L.; Canflanca, I.M.; Garrido, J.B.; Varela, A.L.; Garcia-Hernandez, G.; Grima, F.G.; Gonzalez-Diaz, C.; Carvajal-Urueña, I.; Arnedo-Pena, A.; Busquets-Monge, R.M.; et al. Relationship of asthma and rhinoconjunctivitis with obesity, exercise and Mediterranean diet in Spanish school children. Thorax 2007, 62, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.L.; Lin, K.C.; Pan, W.H. Dietary factors associated with physician-diagnosed asthma and allergic rhinitis in teenagers: Analyses of the first nutrition and health survey in Taiwan. Clin. Exp. Allergy 2001, 31, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, M.; Norback, D. Diet among Japanese female university students and asthmatic symptoms, infections, pollen and furry pet allergy. Respir. Med. 2008, 102, 1045–1054. [Google Scholar] [CrossRef]
- Norbäck, D.; Zhao, Z.H.; Wang, Z.H.; Wieslander, G.; Mi, Y.H.; Zhang, Z. Asthma, eczema, and reports on pollen and cat allergy among pupils in Shanxi province, China. Int. Arch. Occup. Environ. Health 2007, 80, 207–216. [Google Scholar] [CrossRef]
- Awasthi, S.; Kalra, E.; Roy, S. Prevalence and risk factors of asthma and wheeze in school-going children in Lucknow, North India. Indian Pediatr. 2004, 41, 1205–1210. [Google Scholar]
- Arvaniti, F.; Priftis, K.N.; Papadimitriou, A.; Yiallouros, P.; Kapsokefalou, M.; Anthracopoulos, M.B.; Panagiotakos, D.B. Salty-Snack Eating, Television or Video-Game Viewing, and Asthma Symptoms among 10-to 12-Year-Old Children: The PANACEA Study. J. Am. Diet. Assoc. 2011, 111, 251–257. [Google Scholar] [CrossRef]
- Wood, L.G.; Garg, M.L.; Gibson, P.G. A high-fat challenge increases airway inflammation and impairs bronchodilator recovery in asthma. J. Allergy Clin. Immunol. 2011, 127, 1133–1140. [Google Scholar] [CrossRef]
- Weiland, S.K.; von Mutius, E.; Hüsing, A.; Asher, M.I. Intake of trans fatty acids and prevalence of childhood asthma and allergies in Europe. ISAAC Steering Committee. Lancet 1999, 353, 2040–2041. [Google Scholar] [CrossRef]
- Eijkemans, M.; Mommers, M.M.; Draisma, J.; Thijs, C.; Martin, H. Physical Activity and Asthma: A Systematic Review and Meta-Analysis. PLoS ONE 2012, 7, e50775. [Google Scholar] [CrossRef] [PubMed]
- Byberg, K.K.; Eide, G.E.; Forman, M.R.; Júlíusson, P.B.; Øymar, K. Body mass index and physical activity in early childhood are associated with atopic sensitization, atopic dermatitis and asthma in later childhood. Clin. Trans. Allergy 2016, 6, 33. [Google Scholar] [CrossRef] [PubMed]
| Parameters | All Children * n = 106 | AR Group n = 63 | AA Group n = 43 | p-Value AR vs. AA |
|---|---|---|---|---|
| M/F | 60/46 | 38/25 | 22/21 | 0.34 |
| Age (years) mean ± SD (range) | 12.2 ± 3.5 (7–18) | 13.3 ± 3.5 (7–18) | 11.5 ± 3.2 (7–18) | 0.01 |
| Tobacco smoking exposure n (%) | 21 (19.8%) | 15 (24%) | 6 (28%) | 0.21 |
| Animal at home n (%) | 49 (50.7%) | 32 (50.7%) | 17 (39.5%) | 0.25 |
| Family allergies n (%) | 65 (61%) | 34 (53%) | 31 (72%) | 0.09 |
| Spirometry mean % pv ± SD | ||||
| FEV1 | 95.4 ± 16.3 | 100.0 ± 11.1 | 92.1 ± 15.0 | 0.05 |
| FVC | 95.1 ± 10.0 | 97.1 ± 10.1 | 94.0 ± 10.9 | 0.13 |
| FEV1%FVC | 108.8 ± 9.9 | 102.0 ± 4.4 | 99.0 ± 9.2 | 0.16 |
| PEF | 86.7 ± 16.0 | 90.2 ± 15.4 | 84.2 ± 15.9 | 0.06 |
| Parameters | All Patients n = 106 | AR Group n = 63 | AA Group n = 43 |
|---|---|---|---|
| Meals (number per day) | |||
| 2 | 4 (3.7%) | 3 (4.7%) | 1 (2.3%) |
| 3 | 35 (33.0%) | 19 (30.1%) | 16 (37.2%) |
| 4 | 31 (29.2%) | 16 (25.6%) | 15 (34.8%) |
| 5 or more | 36 (34.1%) | 25 (39.6%) | 11 (25.7%) |
| Sweets (days per week) | |||
| 1 | 12 (11.5%) | 7 (11.1%) | 5 (11.6%) |
| 2–3 | 29 (27.3%) | 19 (30.1%) | 10 (23.2%) |
| 4–6 | 18 (16.9%) | 10 (15.8%) | 8 (18.6%) |
| every day | 47 (44.3%) | 27 (43.0%) | 20 (46.6%) |
| Fast food (days per month) | |||
| never | 17 (16.0%) | 12 (19.0%) | 5 (11.6%) |
| 1 | 52 (49.0%) | 32 (50.7%) | 20 (46.6%) |
| 2–3 | 27 (25.4%) | 13 (20.6%) | 14 (32.5%) |
| 4–6 | 9 (8.7%) | 6 (9.7%) | 3 (7.0%) |
| every day | 1 (0.9%) | 0 (0.0%) | 1 (2.3%) |
| Last meal before sleep (hours to bedtime) | |||
| <1 | 52 (49.0%) | 30 (47.6%) | 22 (51.1%) |
| 1 | 20 (18.8%) | 7 (11.1%) | 13 (30.2%) |
| 2 | 13 (12.4%) | 11 (17.4%) | 2 (4.6%) |
| >2 | 21 (19.8%) | 15 (23.9%) | 6 (14.1%) |
| Snacking between meals | |||
| yes | 85 (80%) | 50 (79.3%) | 35 (81.4%) |
| no | 21 (20%) | 13 (20.7%) | 8 (18.6%) |
| Physical activity | |||
| sedentary lifestyle | 9 (8.4%) | 4 (6.3%) | 5 (11.6%) |
| low | 58 (54.7%) | 40 (63.4%) | 18 (42.0%) |
| moderate | 27 (25.4%) | 12 (19.2%) | 15 (34.8%) |
| high | 12 (11.5%) | 7 (11.1%) | 5 (11.6%) |
| Parameters | All Patients | AR Group n = 63 | AA Group n = 43 | p-Value AR vs. AA |
|---|---|---|---|---|
| Anthropometric data, mean ± SD (range) | ||||
| Weight (kg) | 47.1 ± 17.9 (18–98.2) | 51.3 ± 17.9 (21–95) | 44.5 ± 17 (18–92) | 0.06 |
| Height (cm) | 154.6 ± 19.1 (110–185) | 160.3 ± 17.1 (116–182) | 151.4 ± 19.2 (110–185) | 0.03 |
| BMI (percentile) | 45.5 ± 32.1 (1–99) | 41.6 ± 31.1 (5–99) | 47.9 ± 33.1 (1–99) | 0.63 |
| BMI, n (%) | ||||
| Underweight BMI <10th percentile | 8 (7.5%) | 5 (7.9%) | 3 (6.9%) | 0.81 |
| Normal BMI, 10th–90th percentile | 84 (79.0%) | 52 (82.5%) | 32 (74.4%) | 0.43 |
| Overweight BMI, 90th–97th percentile | 8 (7.5%) | 3 (6.9%) | 5 (11.0%) | 0.57 |
| Obesity BMI, >97th percentile | 6 (6.0%) | 3 (4.7%) | 3 (6.9%) | 0.89 |
| Body composition; mean ± SD (range) | ||||
| Body fat (%) | 29.6 ± 20.6 (1–90) | 20 ± 15.4 (3–70) | 21.5 ± 17 (1–90) | 0.14 |
| Body fat (kg) | 9.2 ± 4.8 (1–30) | 8.2 ± 4.7 (2–31) | 7.9 ± 3.9 (1–17) | 0.94 |
| LEAN (%) | 70.3 ± 20.6 (8.9–89) | 79 ± 16.5 (21–89) | 78.1 ± 17.1 (8.9–56) | 0.42 |
| LEAN (kg) | 27.9 ± 17.3 (2–24) | 39.5 ± 17.6 (6–24) | 36.8 ± 17.4 (2–24) | 0.23 |
| Regression Model | B | Standard Error | Beta | p-Value |
|---|---|---|---|---|
| Constant | 16.7 | 2.84 | <0.001 | |
| Snacking | 2.07 | 0.94 | 0.21 | 0.03 |
| Fat%pv | −0.05 | 0.02 | −0.21 | 0.058 |
| FEV1%pv | 0.03 | 0.02 | 0.11 | 0.23 |
| Physical activity | –1.02 | 0.46 | –0.21 | 0.028 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasilewska, E.; Małgorzewicz, S.; Gruchała-Niedoszytko, M.; Skotnicka, M.; Jassem, E. Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study. Nutrients 2020, 12, 1521. https://doi.org/10.3390/nu12051521
Wasilewska E, Małgorzewicz S, Gruchała-Niedoszytko M, Skotnicka M, Jassem E. Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study. Nutrients. 2020; 12(5):1521. https://doi.org/10.3390/nu12051521
Chicago/Turabian StyleWasilewska, Eliza, Sylwia Małgorzewicz, Marta Gruchała-Niedoszytko, Magdalena Skotnicka, and Ewa Jassem. 2020. "Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study" Nutrients 12, no. 5: 1521. https://doi.org/10.3390/nu12051521
APA StyleWasilewska, E., Małgorzewicz, S., Gruchała-Niedoszytko, M., Skotnicka, M., & Jassem, E. (2020). Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study. Nutrients, 12(5), 1521. https://doi.org/10.3390/nu12051521





