Phytosterols Supplementation Reduces Endothelin-1 Plasma Concentration in Moderately Hypercholesterolemic Individuals Independently of Their Cholesterol-Lowering Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Subject Recruitment
2.2. Blood Sampling
2.3. Lipids Profile
2.4. Inflammatory and Endothelial Dysfunction Biomarkers
2.5. Serum Phytosterols Analyses
2.6. RNA Isolation and Quantitative PCR
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ross, R. Atherosclerosis: An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, J.; van Puijvelde, G.H.; van Wanrooij, E.J.; van Es, T.; Habets, K.; Hauer, A.D.; van den Berkel, T.J. Immunomodulation of the inflammatory response in atherosclerosis. Curr. Opin. Lipidol. 2007, 18, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Shah, R. Endothelins in health and disease. Eur. J. Intern. Med. 2007, 18, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Rocha, V.Z.; Libby, P. Obesity, inflammation, and atherosclerosis. Nat. Rev. Cardiol. 2009, 6, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Böhm, F.; Pernow, J. The importance of endothelin-1 for vascular dysfunction in cardiovascular disease. Cardiovasc. Res. 2007, 76, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Agapitov, A.V.; Haynes, W.G. Role of endothelin in cardiovascular disease. J. Renin Angiotensin Aldosterone Syst. 2002, 3, 1–15. [Google Scholar] [CrossRef]
- Jing, Q.; Shen, Q.; Zhang, G.Y.; Wu, Z.G.; Qin, Y.W.; Zheng, X. Involvement of endothelin subtype A receptor (ETA) in vascular smooth muscle cells proliferation evoked by oxidized low-density lipoprotein. J. Am. Coll. Cardiol. 1998, 31 (Suppl. A), 461. [Google Scholar] [CrossRef][Green Version]
- Cardillo, C.; Kilcoyne, C.M.; Cannon, R.O.; Panza, J.A. Increased activity of endogenous endothelin in patients with hypercholesterolemia. J. Am. Coll. Cardiol. 2000, 36, 1483–1488. [Google Scholar] [CrossRef]
- Lerman, A.; Holmes, D.R., Jr.; Bell, M.R.; Garratt, K.N.; Nishimura, R.A.; Burnett, J.C., Jr. Endothelin in coronary endothelial dysfunction and early atherosclerosis in humans. Circulation 1995, 92, 2426–2431. [Google Scholar] [CrossRef]
- Lerman, A.; Zeiher, A.M. Endothelial function: Cardiac events. Circulation 2005, 111, 363–368. [Google Scholar] [CrossRef]
- Boulanger, C.M.; Tanner, F.C.; Béa, M.L.; Hahn, A.W.; Werner, A.; Lüscher, T.F. Oxidized low density lipoproteins induce mRNA expression and release of endothelin from human and porcine endothelium. Circ. Res. 1992, 70, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Ascer, E.; Bertolami, M.C.; Venturinelli, M.L.; Buccheri, V.; Souza, J.; Nicolau, J.C.; Ramires, J.A.; Serrano, C.V., Jr. Atorvastatin reduces proinflammatory markers in hypercholesterolemic patients. Atherosclerosis 2004, 177, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Danielson, E.; Fonseca, F.A.; Genest, J.; Gotto, A.M., Jr.; Kastelein, J.J.; Koenig, W.; Libby, P.; Lorenzatti, A.J.; MacFadyen, J.G.; et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 2008, 359, 2195–2207. [Google Scholar] [CrossRef]
- Lottenberg, A.M.; Nunes, V.S.; Nakandakare, E.R.; Neves, M.; Bernik, M.; Santos, J.E.; Quintao, E.C. Food phytosterol ester efficiency on the plasma lipid reduction in moderate hypercholesterolemic subjects. Arq. Bras. Cardiol. 2002, 79, 139–142. [Google Scholar] [PubMed]
- Acuff, R.V.; Cai, D.J.; Dong, Z.P.; Bell, D. The lipid lowering effect of plant sterol ester capsules in hypercholesterolemic subjects. Lipids Health Dis. 2007, 6, 11. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Padro, T.; Vilahur, G.; Sánchez-Hernández, J.; Hernández, M.; Antonijoan, R.M.; Perez, A.; Badimon, L. Lipidomic changes of LDL in overweight and moderately hypercholesterolemic subjects taking phytosterol- and omega-3-supplemented milk. J. Lipid Res. 2015, 56, 1043–1056. [Google Scholar] [CrossRef]
- Vanstone, C.A.; Raeini-Sarjaz, M.; Jones, P.J. Injected phytosterols/stanol suppress plasma cholesterol levels in hamsters. J. Nutr. Biochem. 2001, 12, 565–574. [Google Scholar] [CrossRef]
- Ikeda, I.; Tanaka, K.; Sugano, M.; Vahouny, G.V.; Gallo, L.L. Inhibition of cholesterol absorption in rats by plant sterols. J. Lipid Res. 1988, 29, 1573–1582. [Google Scholar]
- Field, J.F.; Born, E.; Mathur, S.N. Effect of micellar beta-sitosterol on cholesterol metabolism in CaCo-2 cells. J. Lipid Res. 1997, 38, 348–360. [Google Scholar]
- Carden, T.J.; Hang, J.; Dussault, P.H.; Carr, T.P. Dietary plant sterol esters must be hydrolyzed to reduce intestinal cholesterol absorption in hamsters. J. Nutr. 2015, 145, 1402–1407. [Google Scholar] [CrossRef]
- Moghadasian, M.H.; Alsaif, M.; Le, K.; Gangadaran, S.; Masisi, K.; Beta, T.; Shen, G.X. Combination effects of wild rice and phytosterols on prevention of atherosclerosis in LDL receptor knockout mice. J. Nutr. Biochem. 2016, 33, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Plat, J.; Mensink, R.P. Effects of plant stanol esters on LDL receptor protein expression and on LDL receptor and HMG-CoA reductase mRNA expression in mononuclear blood cells of healthy men and women. FASEB J. 2002, 16, 258–260. [Google Scholar] [CrossRef] [PubMed]
- Bombo, R.P.; Afonso, M.S.; Machado, R.M.; Lavrador, M.S.; Nunes, V.S.; Quintão, E.R.; Koike, M.; Catanozi, S.; Lin, C.J.; Nakandakare, E.R.; et al. Dietary phytosterol does not accumulate in the arterial wall and prevents atherosclerosis of LDLr-KO mice. Atherosclerosis 2013, 231, 442–447. [Google Scholar] [CrossRef] [PubMed]
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421. [Google Scholar] [CrossRef]
- Weingärtner, O.; Lütjohann, D.; Ji, S.; Weisshoff, N.; List, F.; Sudhop, T.; von Bergmann, K.; Gertz, K.; König, J.; Schäfers, H.J.; et al. Vascular effects of diet supplementation with plant sterols. J. Am. Coll. Cardiol. 2008, 51, 1553–1561. [Google Scholar] [CrossRef]
- Weingärtner, O.; Böhm, M.; Laufs, U. Controversial role of plant sterol esters in the management of hypercholesterolaemia. Eur. Heart J. 2009, 30, 404–409. [Google Scholar] [CrossRef]
- Bachorik, P.S.; Albers, J.J. Precipitation methods for quantification of lipoproteins. Methods Enzymol. 1986, 129, 78–100. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Clauss, A. Rapid physiological coagulation method in determination of fibrinogen. Acta Haematol. 1957, 17, 237–246. [Google Scholar] [CrossRef]
- Phillips, K.M.; Ruggio, D.M.; Bailey, J.A. Precise quantitative determination of phytosterols, stanols, and cholesterol metabolites in human serum by capillary gas-liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 17–29. [Google Scholar] [CrossRef]
- Plat, J.; Brufau, G.; Dallinga-Thie, G.M.; Dasselaar, M.; Mensink, R.P. A plant stanol yogurt drink alone or combined with a low-dose statin lowers serum triacylglycerol and non-HDL cholesterol in metabolic syndrome patients. J. Nutr. 2009, 139, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, A.C.; Maranhão, R.C.; de Sousa, H.P.; Schaefer, E.J.; Santos, R.D. Effects of margarines and butter consumption on lipid profiles, inflammation markers and lipid transfer to HDL particles in free-living subjects with the metabolic syndrome. Eur. J. Clin. Nutr. 2010, 64, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- De Jong, A.; Plat, J.; Bast, A.; Godschalk, R.W.; Basu, S.; Mensink, R.P. Effects of plant sterol and stanol ester consumption on lipid metabolism, antioxidant status and markers of oxidative stress, endothelial function and low-grade inflammation in patients on current statin treatment. Eur. J. Clin. Nutr. 2008, 62, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Rosticci, M.; Parini, A.; Giovannini, M.; Veronesi, M.; D’Addato, S.; Borghi, C. Effect of a short-term dietary supplementation with phytosterols, red yeast rice or both on lipid. pattern in moderately hypercholesterolemic subjects: A three-arm, double-blind, randomized clinical trial. Nutr. Metab. (Lond.) 2017, 25, 61. [Google Scholar] [CrossRef]
- Scolaro, B.; Nogueira, M.S.; Paiva, A.; Bertolami, A.; Barroso, L.P.; Vaisar, T.; Heffron, S.P.; Fisher, E.A.; Castro, I.A. Statin dose reduction with complementary diet therapy: A pilot study of personalized medicine. Mol. Metab. 2018, 11, 137–144. [Google Scholar] [CrossRef]
- Hernández-Perera, O.; Pérez-Sala, D.; Navarro-Antolín, J.; Sánchez-Pascuala, R.; Hernández, G.; Díaz, C.; Lamas, S. Effects of the 3-hydroxy-3-methylgutaryl-CoA reductase inhibitors, atorvastatin and simvastatin, on the expression of endothelin-1 and endothelial nitric oxide synthase in vascular endothelial cells. J. Clin. Investig. 1998, 101, 2711–2719. [Google Scholar] [CrossRef]
- Economides, P.A.; Caselli, A.; Tiani, E.; Khaodhiar, L.; Horton, E.S.; Veves, A. The effects of atorvastatin on endothelial function in diabetic patients and subjects at risk for type 2 diabetes. J. Clin. Endocrinol. Metab. 2004, 89, 740–747. [Google Scholar] [CrossRef]
- Arca, M.; Montali, A.; Pigna, G.; Antonini, R.; Antonini, T.M.; Luigi, P.; Fraioli, A.; Mastrantoni, M.; Maddaloni, M.; Letizia, C. Comparison of atorvastatin versus fenofibrate in reaching lipid targets and influencing biomarkers of endothelial damage in patients with familial combined hyperlipidemia. Metabolism 2007, 56, 1534–1541. [Google Scholar] [CrossRef]
- Liao, P.C.; Lai, M.H.; Hsu, K.P.; Kuo, Y.H.; Chen, J.; Tsai, M.C.; Li, C.X.; Yin, X.J.; Jeyashoke, N.; Chao, L.K. Identification of β-Sitosterol as in Vitro Anti-Inflammatory Constituent in Moringa oleifera. J. Agric. Food Chem. 2018, 66, 10748–10759. [Google Scholar] [CrossRef]
- Nashed, B.; Yeganeh, B.; HayGlass, K.T.; Moghadasian, M.H. Antiatherogenic effects of dietary plant sterols are associated with inhibition of proinflammatory cytokine production in Apo E-KO mice. J. Nutr. 2005, 135, 2438–2444. [Google Scholar] [CrossRef]
- Ho, X.L.; Liu, J.J.; Loke, W.M. Plant sterol-enriched soy milk consumption modulates 5-lipoxygenase, 12-lipoxygenase, and myeloperoxidase activities in healthy adults - a randomized-controlled trial. Free Radic. Res. 2016, 50, 1396–1407. [Google Scholar] [CrossRef] [PubMed]
- Kurano, M.; Hasegawa, K.; Kunimi, M.; Hara, M.; Yatomi, Y.; Teramoto, T.; Tsukamoto, K. Sitosterol prevents obesity-related chronic inflammation. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; de Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef] [PubMed]
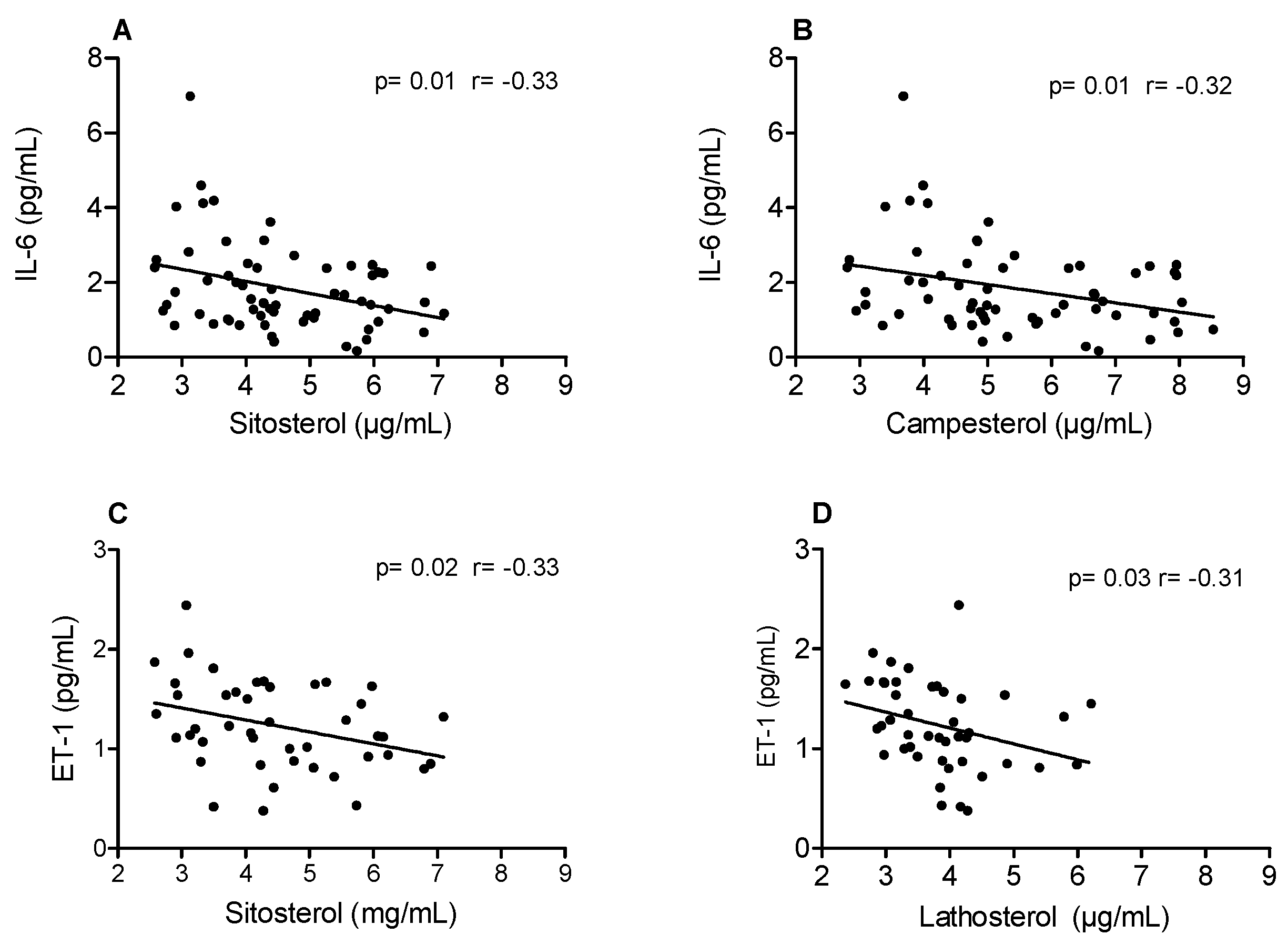
| Parameter | Screening | After Run-in Period |
|---|---|---|
| Age (years) | 58 ± 12 | 58 ± 12 |
| Weight (Kg) | 64 ± 10 | 64 ± 9 |
| BMI (kg/m2) | 25.3 ± 2.4 | 25.4 ± 2.3 |
| Total cholesterol (mg/dL) | 245 ± 34 | 249 ± 37 |
| Triglycerides (mg/dL) | 141 ± 53 | 142 ± 10 |
| LDL-c (mg/dL) | 165 ± 34 | 173 ± 31 |
| HDL-c (mg/dL) | 49 ± 12 | 47 ± 10 |
| Nutritional Composition | Soy Milk | Soy Milk + PS |
|---|---|---|
| Energy (kcal) | 138 | 144 |
| Protein (g) | 6.5 | 6.5 |
| Total fat (g) | 4.4 | 5.0 |
| Polyunsaturated fat | 2.3 | 2.5 |
| Monounsaturated fat | 1.0 | 1.1 |
| Saturated fat | 0.7 | 0.9 |
| Trans fatty acid | 0 | 0 |
| Cholesterol (mg) | 0 | 0 |
| Carbohydrates (g) | 18.2 | 18.2 |
| Total sugar | 14.1 | 14.1 |
| Lactose | 0 | 0 |
| Phytosterols (g) | 0 | 0.8 |
| β-sitosterol-ester | 0.63 | |
| Sitostanol-ester | 0.10 | |
| Campesterol-ester | 0.05 | |
| Campestanol-ester | 0.005 | |
| Sodium (g) | 0.1 | 0.1 |
| n | Placebo | Phyto | p | |
|---|---|---|---|---|
| Weight (kg) | 38 | 64.9 ± 10.2 | 65.1 ± 10.3 | 0.08 |
| BMI (kg/m2) | 38 | 25.4 ± 0.4 | 25.4 ± 0.4 | 0.89 |
| Total cholesterol (mg/dL) | 38 | 261 ± 7.1 | 244 ± 5.8 * | <0.001 |
| HDL-c (mg/dL) | 38 | 46 ± 1.7 | 48 ± 1.9 | 0.54 |
| LDL-c (mg/dL) | 38 | 183 ± 5.9 | 169 ± 5.2 * | 0.001 |
| Apo B (mg/dL) | 38 | 126 ± 3.7 | 118 ± 3.2 * | 0.006 |
| Triglycerides (mg/dL) | 38 | 154 ± 10 | 133 ± 7 * | 0.008 |
| Fibrinogen (mg/dL) | 25 | 3.6 ± 0.5 | 3.5 ± 0.5 | 0.79 |
| hs-CRP (mg/L) | 30 | 3.1 ± 0.4 | 3.1 ± 0.4 | 0.96 |
| SAA (mg/L) | 33 | 6.78 ± 0.52 | 5.94 ± 0.51 | 0.16 |
| IL-6 (pg/mL) | 33 | 2.69 ± 1.03 | 2.24 ± 1.01 | 0.25 |
| TNFα (pg/mL) | 36 | 1.35 ± 0.07 | 1.28 ± 0.06 | 0.19 |
| VCAM-1(ng/mL) | 38 | 469 ± 127 | 472 ± 118 | 0.69 |
| ET-1 (pg/mL) | 24 | 1.31 ± 0.09 | 1.13 ± 0.09 * | 0.02 |
| HMGCoAr/Actb (fold change) | 21 | 1.10 ± 0.20 | 1.05 ± 0.16 | 0.70 |
| LDLr/Actb (fold change) | 17 | 1.24 ± 0.23 | 1.26 ± 0.21 | 0.66 |
| Plasma Sterols Expressed as µg/ mg Cholesterol | ||||
| Lathosterol | 38 | 1.53 ± 0.09 | 1.69 ± 0.06 * | 0.01 |
| Campesterol | 38 | 1.96 ± 0.12 | 2.34 ± 0.11 * | 0.02 |
| β-sitosterol | 38 | 1.64 ± 0.09 | 2.02 ± 0.09 * | <0.001 |
| Lathosterol/campesterol ratio | 38 | 0.85 ± 0.05 | 0.76 ± 0.03 * | <0.001 |
| Lathosterol/β-sitosterol ratio | 38 | 1.03 ± 0.05 | 0.88 ± 0.04 * | <0.001 |
| LDL-c ≤ 166 mg/dL 1 | LDL-c > 167 mg/dL 2 | |||||
|---|---|---|---|---|---|---|
| Placebo | Phyto | % Change | Placebo | Phyto | % Change | |
| Triglycerides (mg/dL) | 146 ± 58 | 134 ± 45 | −1.9 | 164 ± 61 | 132 ± 41 * | −16.0 |
| Total cholesterol (mg/dL) | 243 ± 34 | 230 ± 26 * | −4.7 | 283 ± 44 | 263 ± 37 * | −6.4 |
| LDL-c (mg/dL) | 166 ± 24 | 154 ± 20 * | −6.2 | 202 ± 39 | 186 ± 33 * | v6.8 |
| Apo-B (mg/dL) | 115 ± 12 | 109 ± 13 | −4.3 | 140 ± 25 | 129 ± 20 * | −6.5 |
| Response to Treatment | Placebo | Phytosterol | % Change |
|---|---|---|---|
| PS Responsive (n = 18) | 1.41 ± 0.38 | 1.21 ± 0.44 * | −12 |
| PS Non-Responsive (n = 6) | 1.00 ± 0.39 | 0.91 ± 0.35 * | −8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira Godoy Ilha, A.; Sutti Nunes, V.; Silva Afonso, M.; Regina Nakandakare, E.; da Silva Ferreira, G.; de Paula Assis Bombo, R.; Rodrigues Giorgi, R.; Marcondes Machado, R.; Carlos Rocha Quintão, E.; Lottenberg, A.M. Phytosterols Supplementation Reduces Endothelin-1 Plasma Concentration in Moderately Hypercholesterolemic Individuals Independently of Their Cholesterol-Lowering Properties. Nutrients 2020, 12, 1507. https://doi.org/10.3390/nu12051507
Oliveira Godoy Ilha A, Sutti Nunes V, Silva Afonso M, Regina Nakandakare E, da Silva Ferreira G, de Paula Assis Bombo R, Rodrigues Giorgi R, Marcondes Machado R, Carlos Rocha Quintão E, Lottenberg AM. Phytosterols Supplementation Reduces Endothelin-1 Plasma Concentration in Moderately Hypercholesterolemic Individuals Independently of Their Cholesterol-Lowering Properties. Nutrients. 2020; 12(5):1507. https://doi.org/10.3390/nu12051507
Chicago/Turabian StyleOliveira Godoy Ilha, Angela, Valeria Sutti Nunes, Milessa Silva Afonso, Edna Regina Nakandakare, Guilherme da Silva Ferreira, Renata de Paula Assis Bombo, Ricardo Rodrigues Giorgi, Roberta Marcondes Machado, Eder Carlos Rocha Quintão, and Ana Maria Lottenberg. 2020. "Phytosterols Supplementation Reduces Endothelin-1 Plasma Concentration in Moderately Hypercholesterolemic Individuals Independently of Their Cholesterol-Lowering Properties" Nutrients 12, no. 5: 1507. https://doi.org/10.3390/nu12051507
APA StyleOliveira Godoy Ilha, A., Sutti Nunes, V., Silva Afonso, M., Regina Nakandakare, E., da Silva Ferreira, G., de Paula Assis Bombo, R., Rodrigues Giorgi, R., Marcondes Machado, R., Carlos Rocha Quintão, E., & Lottenberg, A. M. (2020). Phytosterols Supplementation Reduces Endothelin-1 Plasma Concentration in Moderately Hypercholesterolemic Individuals Independently of Their Cholesterol-Lowering Properties. Nutrients, 12(5), 1507. https://doi.org/10.3390/nu12051507





