Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update
Abstract
1. Introduction
1.1. Background
1.2. Acetyl-L-Carnitine
1.3. ALC And Dementia
1.4. Aim And Rationale
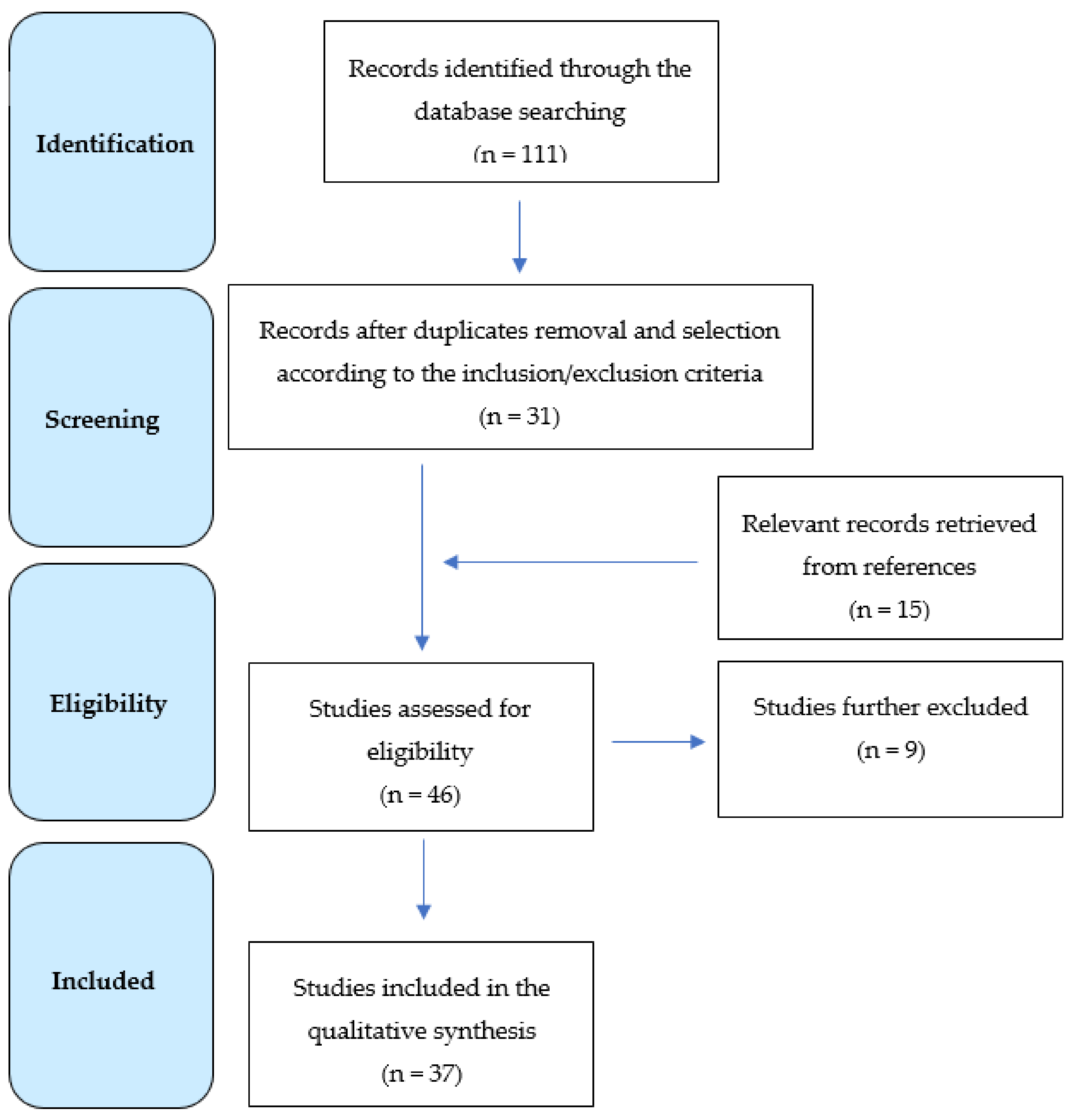
2. Data Source and Selection
3. Results
3.1. Alzheimer’s Disease and Mild Cognitive Impairment
3.2. Vascular Cognitive Impairment
3.3. Hepatic Encephalopathy
3.4. Other Secondary Dementias
3.5. Unspecified Cognitive Disorder
4. Discussion
4.1. Summary of the Evidences and Proposed Mechanisms
4.2. Potential Organ Cross-Talk Actions of ALC and Hepatic Detoxifying Mechanisms
4.3. Current Limitations and Research Agenda
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Prince, M.; Wimo, A.; Guerchet, M.; Ali, G.; Wu, Y.-T.; Prina, M. World Alzheimer Report 2015, The Global Impact of Dementia: An analysis of prevalence, incidence, cost and trends. Alzheimers Dis. Int. 2015, 87, 1–82. [Google Scholar]
- Whitehouse, P.J.; Price, D.L.; Struble, R.G.; Clark, A.W.; Coyle, J.T.; Delon, M.R. Alzheimer’s disease and senile dementia: Loss of neurons in the basal forebrain. Science 1982, 215, 1237–1239. [Google Scholar] [CrossRef]
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004, 256, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Langa, K.M.; Levine, D.A. The diagnosis and management of mild cognitive impairment: A clinical review. JAMA 2014, 312, 2551–2561. [Google Scholar] [CrossRef] [PubMed]
- Buss, S.S.; Fried, P.J.; Pascual-Leone, A. Therapeutic noninvasive brain stimulation in Alzheimer’s disease and related dementias. Curr. Opin. Neurol. 2019, 32, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Bordet, R.; Ihl, R.; Korczyn, A.D.; Lanza, G.; Jansa, J.; Hoerr, R.; Guekht, A. Towards the concept of disease-modifier in post-stroke or vascular cognitive impairment: A consensus report. BMC Med. 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.; Centonze, S.S.; Destro, G.; Vella, V.; Bellomo, M.; Pennisi, M.; Bella, R.; Ciavardelli, D. Shiatsu as an adjuvant therapy for depression in patients with Alzheimer’s disease: A pilot study. Complement. Ther. Med. 2018, 38, 74–78. [Google Scholar] [CrossRef]
- Yiannopoulou, K.G.; Papageorgiou, S.G. Current and Future Treatments in Alzheimer Disease: An Update. J. Cent. Nerv. Syst. Dis. 2020, 12, 1179573520907397. [Google Scholar] [CrossRef]
- Pennisi, G.; Ferri, R.; Lanza, G.; Cantone, M.; Pennisi, M.; Puglisi, V.; Malaguarnera, G.; Bella, R. Transcranial magnetic stimulation in Alzheimer’s disease: A neurophysiological marker of cortical hyperexcitability. J. Neural Transm. Vienna Austria 1996 2011, 118, 587–598. [Google Scholar] [CrossRef]
- Pennisi, G.; Bella, R.; Lanza, G. Motor cortex plasticity in subcortical ischemic vascular dementia: What can TMS say? Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2015, 126, 851–852. [Google Scholar] [CrossRef]
- Lanza, G.; Bella, R.; Cantone, M.; Pennisi, G.; Ferri, R.; Pennisi, M. Cognitive Impairment and Celiac Disease: Is Transcranial Magnetic Stimulation a Trait d’Union between Gut and Brain? Int. J. Mol. Sci. 2018, 19, 2243. [Google Scholar] [CrossRef] [PubMed]
- Cantone, M.; Catalano, M.A.; Lanza, G.; La Delfa, G.; Ferri, R.; Pennisi, M.; Bella, R.; Pennisi, G.; Bramanti, A. Motor and Perceptual Recovery in Adult Patients with Mild Intellectual Disability. Neural Plast. 2018. [Google Scholar] [CrossRef] [PubMed]
- Fisicaro, F.; Lanza, G.; Grasso, A.A.; Pennisi, G.; Bella, R.; Paulus, W.; Pennisi, M. Repetitive transcranial magnetic stimulation in stroke rehabilitation: Review of the current evidence and pitfalls. Ther. Adv. Neurol. Disord. 2019, 12, 175628641987831. [Google Scholar] [CrossRef] [PubMed]
- Fisicaro, F.; Lanza, G.; Bella, R.; Pennisi, M. “Self-Neuroenhancement”: The Last Frontier of Noninvasive Brain Stimulation? J. Clin. Neurol. 2020, 16, 158–159. [Google Scholar] [CrossRef]
- Pettegrew, J.W.; Levine, J.; McClure, R.J. Acetyl-L-carnitine physical-chemical, metabolic, and therapeutic properties: Relevance for its mode of action in Alzheimer’s disease and geriatric depression. Mol. Psychiatry 2000, 5, 616–632. [Google Scholar] [CrossRef]
- Spagnoli, A.; Lucca, U.; Menasce, G.; Bandera, L.; Cizza, G.; Forloni, G.; Tettamanti, M.; Frattura, L.; Tiraboschi, P.; Comelli, M. Long-term acetyl-L-carnitine treatment in Alzheimer’s disease. Neurology 1991, 41, 1726–1732. [Google Scholar] [CrossRef]
- Furlong, J. Acetyl-L-Carnitine: Metabolism and Applications in Clinical Practice. Altern. Med. Rev. 1996, 1, 9. [Google Scholar]
- Carta, A.; Calvani, M.; Bravi, D.; Bhuachalla, S.N. Acetyl-L-carnitine and Alzheimer’s disease: Pharmacological considerations beyond the cholinergic sphere. Ann. N. Y. Acad. Sci. 1993, 695, 324–326. [Google Scholar] [CrossRef]
- Kwon, O.-S.; Chung, Y.B. HPLC determination and pharmacokinetics of endogenous acetyl-L-carnitine (ALC) in human volunteers orally administered a single dose of ALC. Arch. Pharm. Res. 2004, 27, 676–681. [Google Scholar] [CrossRef]
- Parnetti, L.; Gaiti, A.; Mecocci, P.; Cadini, D.; Senin, U. Pharmacokinetics of IV and oral acetyl-L-carnitine in a multiple dose regimen in patients with senile dementia of Alzheimer type. Eur. J. Clin. Pharmacol. 1992, 42, 89–93. [Google Scholar] [CrossRef]
- Acetyl-L-Carnitine Monograph. Altern. Med. Rev. 2010, 8, 76–83.
- Purpura, D.P.; Girado, M.; Smith, T.G.; Callan, D.A.; Grundfest, H. Structure-Activity Determinants of Pharmacological Effects of Amino Acids and Related Compounds on Central Synapses. J. Neurochem. 1959, 3, 238–268. [Google Scholar] [CrossRef] [PubMed]
- Pettegrew, J.W.; Klunk, W.E.; Panchalingam, K.; Kanfer, J.N.; McClure, R.J. Clinical and neurochemical effects of acetyl-L-carnitine in Alzheimer’s disease. Neurobiol. Aging 1995, 16, 1–4. [Google Scholar] [CrossRef]
- Traina, G. The neurobiology of acetyl-L-carnitine. Front. Biosci. Landmark Ed. 2016, 21, 1314–1329. [Google Scholar] [CrossRef] [PubMed]
- Imperato, A.; Ramacci, M.T.; Angelucci, L. Acetyl-L-carnitine enhances acetylcholine release in the striatum and hippocampus of awake freely moving rats. Neurosci. Lett. 1989, 107, 251–255. [Google Scholar] [CrossRef]
- Zanelli, S.A.; Solenski, N.J.; Rosenthal, R.E.; Fiskum, G. Mechanisms of ischemic neuroprotection by acetyl-L-carnitine. Ann. N. Y. Acad. Sci. 2005, 1053, 153–161. [Google Scholar] [CrossRef]
- Bagetta, V.; Barone, I.; Ghiglieri, V.; Di Filippo, M.; Sgobio, C.; Bernardi, G.; Calabresi, P.; Picconi, B. Acetyl-L-Carnitine selectively prevents post-ischemic LTP via a possible action on mitochondrial energy metabolism. Neuropharmacology 2008, 55, 223–229. [Google Scholar] [CrossRef]
- Di Stefano, G.; Di Lionardo, A.; Galosi, E.; Truini, A.; Cruccu, G. Acetyl-L-carnitine in painful peripheral neuropathy: A systematic review. J. Pain Res. 2019, 12, 1341–1351. [Google Scholar] [CrossRef]
- Sano, M.; Bell, K.; Cote, L.; Dooneief, G.; Lawton, A.; Legler, L.; Marder, K.; Naini, A.; Stern, Y.; Mayeux, R. Double-blind Parallel Design Pilot Study of Acetyl Levocarnitine in Patients With Alzheimer’s Disease. Arch. Neurol. 1992, 49, 1137–1141. [Google Scholar] [CrossRef]
- Bella, R.; Biondi, R.; Raffaele, R.; Pennisi, G. Effect of acetyl-L-carnitine on geriatric patients suffering from dysthymic disorders. Int. J. Clin. Pharmacol. Res. 1990, 10, 355–360. [Google Scholar]
- Fulgente, T.; Onofrj, M.; DELRE, M.; Ferracci, F.; Bazzano, S.; Ghilardi, M.F.; Malatesta, G. Levoacetylcarnitine (nicetile) treatment of senile depression. Clin. Trials J. 1990, 27, 155–163. [Google Scholar]
- Garzya, G.; Corallo, D.; Fiore, A.; Lecciso, G.; Petrelli, G.; Zotti, C. Evaluation of the effects of L-acetylcarnitine on senile patients suffering from depression. Drugs Exp. Clin. Res. 1990, 16, 101–106. [Google Scholar] [PubMed]
- Tomassini, V.; Pozzilli, C.; Onesti, E.; Pasqualetti, P.; Marinelli, F.; Pisani, A.; Fieschi, C. Comparison of the effects of acetyl L-carnitine and amantadine for the treatment of fatigue in multiple sclerosis: Results of a pilot, randomised, double-blind, crossover trial. J. Neurol. Sci. 2004, 218, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Cauli, O. Effects of l-Carnitine in Patients with Autism Spectrum Disorders: Review of Clinical Studies. Mol. Basel Switz. 2019, 24, 4262. [Google Scholar] [CrossRef]
- Malaguarnera, M. Carnitine derivatives: clinical usefulness. Curr. Opin. Gastroenterol. 2012, 28, 166–176. [Google Scholar] [CrossRef]
- Brooks, J.O.; Yesavage, J.A.; Carta, A.; Bravi, D. Acetyl L-Carnitine Slows Decline in Younger Patients With Alzheimer’s Disease: A Reanalysis of a Double-Blind, Placebo-Controlled Study Using the Trilinear Approach. Int. Psychogeriatr. 1998, 10, 193–203. [Google Scholar] [CrossRef]
- Hudson, S.A.; Tabet, N. Acetyl-l-carnitine for dementia. Cochrane Database Syst. Rev. 2003. [Google Scholar] [CrossRef]
- Montgomery, S.A.; Thal, L.J.; Amrein, R. Meta-analysis of double blind randomized controlled clinical trials of acetyl-L-carnitine versus placebo in the treatment of mild cognitive impairment and mild Alzheimers disease? Int. Clin. Psychopharmacol. 2003, 18, 61–71. [Google Scholar] [CrossRef]
- Battistin, L.; Pizzolato, G.; Dam, M.; Ponza, I.; Perlotto, N.; Bergamo, L.C.; Furlanut, M.; Ziliotto, R.; Bardin, G.; Chinaglia, L. Effects of acetyl-L-carnitine (ALC) treatment in dementia: A multicentric, randomized, double-blind study. New Trends Clin. Neuropharmacol. 1989, 3, 131–132. [Google Scholar]
- Battistin, L.; Pizzolato, G.; Dam, M.; Da Col, C.; Perlotto, N.; Saitta, B.; Borsato, N.; Calvani, M.; Ferlin, G. Single-photon emission computed tomography studies with 99mTc-hexamethylpropyleneamine oxime in dementia: Effects of acute administration of L-acetylcarnitine. Eur. Neurol. 1989, 29, 261–265. [Google Scholar] [CrossRef]
- Bellagamba, G.; Postacchini, D.; Moretti, V.; Pennacchietti, L. Acetyl-l-carnitine activity in senile dementia Alzheimer type. Neurobiol. Aging 1990. [Google Scholar] [CrossRef]
- Campi, N.; Todeschini, G.P.; Scarzella, L. Selegiline versus L-acetylcarnitine in the treatment of Alzheimer-type dementia. Clin. Ther. 1990, 12, 306–314. [Google Scholar] [PubMed]
- Passeri, M.; Cucinotta, D.; Bonati, P.A.; Iannuccelli, M.; Parnetti, L.; Senin, U. Acetyl-L-carnitine in the treatment of mildly demented elderly patients. Int. J. Clin. Pharmacol. Res. 1990, 10, 75–79. [Google Scholar] [PubMed]
- Rai, G.; Wright, G.; Scott, L.; Beston, B.; Rest, J.; Exton-Smith, A.N. Double-blind, placebo controlled study of acetyl-l-carnitine in patients with Alzheimer’s dementia. Curr. Med. Res. Opin. 1990, 11, 638–647. [Google Scholar] [CrossRef]
- Costa, A.; Martignoni, E.; Bono, G.; Sinforiani, E.; Petraglia, F.; Genazzani, A.R.; Nappi, G. Pituitary-adrenal function and cognitive performance in demented patients on acetyl-L-carnitine treatment. Med. Sci. Res. 1993, 21, 589–591. [Google Scholar]
- Bayer The Effect of Acetyl-L-Carnitine in Senile Dementia of the Alzheimer Type Clinical Report, Study Protocol No.: S104/1 CT4. On file Sigma-Tau. 1994.
- Mullin The Effect of Acetyl-l-carnitine in Senile Dementia of the Alzheimer Type. Clinical Report, Study Protocol No.: S104/1 CT3. On file Sigma-Tau. 1994.
- Bruno, G.; Scaccianoce, S.; Bonamini, M.; Patacchioli, F.R.; Cesarino, F.; Grassini, P.; Sorrentino, E.; Angelucci, L.; Lenzi, G.L. Acetyl-L-carnitine in Alzheimer disease: A short-term study on CSF neurotransmitters and neuropeptides. Alzheimer Dis. Assoc. Disord. 1995, 9, 128–131. [Google Scholar] [CrossRef]
- Thal, L.J.; Carta, A.; Clarke, W.R.; Ferris, S.H.; Friedland, R.P.; Petersen, R.C.; Pettegrew, J.W.; Pfeiffer, E.; Raskind, M.A.; Sano, M.; et al. A 1-year multicenter placebo-controlled study of acetyl-L-carnitine in patients with Alzheimer’s disease. Neurology 1996, 47, 705–711. [Google Scholar] [CrossRef]
- Thal, L.J.; Calvani, M.; Amato, A.; Carta, A. A 1-year controlled trial of acetyl-l-carnitine in early-onset AD. Neurology 2000, 55, 805–810. [Google Scholar] [CrossRef]
- Bianchetti, A.; Rozzini, R.; Trabucchi, M. Effects of acetyl-L-carnitine in Alzheimer’s disease patients unresponsive to acetylcholinesterase inhibitors. Curr. Med. Res. Opin. 2003, 19, 350–353. [Google Scholar] [CrossRef]
- Jeong, H.S.; Park, J.-S.; Yang, Y.; Na, S.-H.; Chung, Y.-A.; Song, I.-U. Cerebral Perfusion Changes after Acetyl-L-Carnitine Treatment in Early Alzheimer’s Disease Using Single Photon Emission Computed Tomography. Dement. Neurocognitive Disord. 2017, 16, 26. [Google Scholar] [CrossRef]
- Arrigo, A.; Clano, E.; Casale, R.; Buonocore, M. The effects of L-acetylcarnitine on reaction times in patients with cerebrovascular insufficiency. A double blind cross-over study. Clin. Trials J. 1988, 25, 47–56. [Google Scholar]
- Arrigo, A.; Casale, R.; Buonocore, M.; Ciano, C. Effects of acetyl-L-carnitine on reaction times in patients with cerebrovascular insufficiency. Int. J. Clin. Pharmacol. Res. 1990, 10, 133–137. [Google Scholar] [PubMed]
- Yang, Y.; Choi, H.; Lee, C.-N.; Kim, Y.B.; Kwak, Y.T. A Multicenter, Randomized, Double-blind, Placebo-controlled Clinical Trial for Efficacy of Acetyl-L-carnitine in Patients with Dementia Associated with Cerebrovascular Disease. Dement. Neurocognitive Disord. 2018, 17, 1. [Google Scholar] [CrossRef]
- Siciliano, M.; Annicchiarico, B.E.; Lucchese, F.; Bombardieri, G. Effects of a single, short intravenous dose of acetyl-L-carnitine on pattern-reversal visual-evoked potentials in cirrhotic patients with hepatic encephalopathy. Clin. Exp. Pharmacol. Physiol. 2006, 33, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Gargante, M.P.; Cristaldi, E.; Vacante, M.; Risino, C.; Cammalleri, L.; Pennisi, G.; Rampello, L. Acetyl-l-Carnitine Treatment in Minimal Hepatic Encephalopathy. Dig. Dis. Sci. 2008, 53, 3018–3025. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Bella, R.; Vacante, M.; Giordano, M.; Malaguarnera, G.; Gargante, M.P.; Motta, M.; Mistretta, A.; Rampello, L.; Pennisi, G. Acetyl-l-carnitine reduces depression and improves quality of life in patients with minimal hepatic encephalopathy. Scand. J. Gastroenterol. 2011, 46, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Vacante, M.; Giordano, M.; Pennisi, G.; Bella, R.; Rampello, L.; Malaguarnera, M.; Li Volti, G.; Galvano, F. Oral acetyl-l-carnitine therapy reduces fatigue in overt hepatic encephalopathy: A randomized, double-blind, placebo-controlled study. Am. J. Clin. Nutr. 2011, 93, 799–808. [Google Scholar] [CrossRef]
- Malaguarnera, M.; Vacante, M.; Motta, M.; Giordano, M.; Malaguarnera, G.; Bella, R.; Nunnari, G.; Rampello, L.; Pennisi, G. Acetyl-L-carnitine improves cognitive functions in severe hepatic encephalopathy: A randomized and controlled clinical trial. Metab. Brain Dis. 2011, 26, 281–289. [Google Scholar] [CrossRef]
- Goetz, C.G.; Tanner, C.M.; Cohen, J.A.; Thelen, J.A.; Carroll, V.S.; Klawans, H.L.; Fariello, R.G. L-acetyl-carnitine in Huntington’s disease: Double-blind placebo controlled crossover study of drug effects on movement disorder and dementia. Mov. Disord. Off. J. Mov. Disord. Soc. 1990, 5, 263–265. [Google Scholar] [CrossRef]
- Tempesta, E.; Troncon, R.; Janiri, L.; Colusso, L.; Riscica, P.; Saraceni, G.; Gesmundo, E.; Calvani, M.; Benedetti, N.; Pola, P. Role of acetyl-L-carnitine in the treatment of cognitive deficit in chronic alcoholism. Int. J. Clin. Pharmacol. Res. 1990, 10, 101–107. [Google Scholar]
- Famularo, G.; Moretti, S.; Alesse, E.; Trinchieri, V.; Angelucci, A.; Santini, G.; Cifone, G.; De Simone, C. Reduction of glutamate levels in HIV-infected subjects treated with acetylcarnitine. J. Neuro AIDS 1999, 2, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Pueschel, S. The effect of acetyl-l-carnitine administration on persons with Down syndrome. Res. Dev. Disabil. 2006, 27, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Passeri, M.; Iannuccelli, M.; Ciotti, G.; Bonati, P.A.; Nolfe, G.; Cucinotta, D. Mental impairment in aging: Selection of patients, methods of evaluation and therapeutic possibilities of acetyl-L-carnitine. Int. J. Clin. Pharmacol. Res. 1988, 8, 367–376. [Google Scholar] [PubMed]
- Mantero, M.A.; Barbero, M.; Giannini, R.; Grosso, V.G.; Tomasina, C.; Iannuccelli, M. Acetyl-L-carnitine as a therapeutic agent for mental deterioration in geriatric patients.(Double-blind placebo controlled study). New Trends Clin. Neuropharmacol. 1989, 3, 17–24. [Google Scholar]
- Herrmann, W.M.; Dietrich, B.; Hiersemenzel, R. Pharmaco-electroencephalographic and clinical effects of the cholinergic substance--acetyl-L-carnitine--in patients with organic brain syndrome. Int. J. Clin. Pharmacol. Res. 1990, 10, 81–84. [Google Scholar] [PubMed]
- Sinforiani, E.; Iannuccelli, M.; Mauri, M.; Costa, A.; Merlo, P.; Bono, G.; Nappi, G. Neuropsychological changes in demented patients treated with acetyl-L-carnitine. Int. J. Clin. Pharmacol. Res. 1990, 10, 69–74. [Google Scholar]
- Livingston, G.A.; Sax, K.B.; McClenahan, Z.; Blumenthal, E.; Foley, K.; Willison, J.; Mann, A.H.; James, I.M. Acetyl-L-Carnitine in dementia. Int. J. Geriatr. Psychiatry 1991, 6, 853–860. [Google Scholar] [CrossRef]
- Salvioli, G.; Neri, M. L-acetylcarnitine treatment of mental decline in the elderly. Drugs Exp. Clin. Res. 1994, 20, 169–176. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6. [Google Scholar] [CrossRef]
- McDaniel, M.A.; Maier, S.F.; Einstein, G.O. “Brain-specific” nutrients: A memory cure? Nutr. Burbank Los Angel. Cty. Calif 2003, 19, 957–975. [Google Scholar] [CrossRef]
- Rebouche, C.J. Kinetics, pharmacokinetics, and regulation of L-carnitine and acetyl-L-carnitine metabolism. Ann. N. Y. Acad. Sci. 2004, 1033, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Cavanna, A.E.; Trimble, M.R. The precuneus: A review of its functional anatomy and behavioural correlates. Brain J. Neurol. 2006, 129, 564–583. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J.T.; Erkinjuntti, T.; Reisberg, B.; Roman, G.; Sawada, T.; Pantoni, L.; Bowler, J.V.; Ballard, C.; DeCarli, C.; Gorelick, P.B.; et al. Vascular cognitive impairment. Lancet Neurol. 2003, 2, 89–98. [Google Scholar] [CrossRef]
- Gorelick, P.B.; Scuteri, A.; Black, S.E.; Decarli, C.; Greenberg, S.M.; Iadecola, C.; Launer, L.J.; Laurent, S.; Lopez, O.L.; Nyenhuis, D.; et al. Vascular contributions to cognitive impairment and dementia: A statement for healthcare professionals from the american heart association/american stroke association. Stroke 2011, 42, 2672–2713. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Ferri, R.; Cantone, M.; Lanza, G.; Pennisi, M.; Vinciguerra, L.; Malaguarnera, G.; Bella, R. A review of transcranial magnetic stimulation in vascular dementia. Dement. Geriatr. Cogn. Disord. 2011, 31, 71–80. [Google Scholar] [CrossRef]
- Bella, R.; Ferri, R.; Pennisi, M.; Cantone, M.; Lanza, G.; Malaguarnera, G.; Spampinato, C.; Giordano, D.; Alagona, G.; Pennisi, G. Enhanced motor cortex facilitation in patients with vascular cognitive impairment-no dementia. Neurosci. Lett. 2011, 503, 171–175. [Google Scholar] [CrossRef]
- Malaguarnera, M.; Pistone, G.; Astuto, M.; Vecchio, I.; Raffaele, R.; Lo Giudice, E.; Rampello, L. Effects of L-acetylcarnitine on cirrhotic patients with hepatic coma: Randomized double-blind, placebo-controlled trial. Dig. Dis. Sci. 2006, 51, 2242–2247. [Google Scholar] [CrossRef]
- Ilias, I.; Manoli, I.; Blackman, M.R.; Gold, P.W.; Alesci, S. L-Carnitine and acetyl-L-carnitine in the treatment of complications associated with HIV infection and antiretroviral therapy. Mitochondrion 2004, 4, 163–168. [Google Scholar] [CrossRef]
- Cristofano, A.; Sapere, N.; La Marca, G.; Angiolillo, A.; Vitale, M.; Corbi, G.; Scapagnini, G.; Intrieri, M.; Russo, C.; Corso, G.; et al. Serum Levels of Acyl-Carnitines along the Continuum from Normal to Alzheimer’s Dementia. PLoS ONE 2016, 11, e0155694. [Google Scholar] [CrossRef]
- Jones, L.L.; McDonald, D.A.; Borum, P.R. Acylcarnitines: Role in brain. Prog. Lipid Res. 2010, 49, 61–75. [Google Scholar] [CrossRef]
- Onofrj, M.; Ciccocioppo, F.; Varanese, S.; di Muzio, A.; Calvani, M.; Chiechio, S.; Osio, M.; Thomas, A. Acetyl-L-carnitine: From a biological curiosity to a drug for the peripheral nervous system and beyond. Expert Rev. Neurother. 2013, 13, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, C.; Bates, T.E.; Butterfield, D.A.; Calafato, S.; Cornelius, C.; De Lorenzo, A.; Dinkova Kostova, A.T.; Calabrese, V. Natural antioxidants in Alzheimer’s disease. Expert Opin. Investig. Drugs 2007, 16, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Aureli, T.; Capuani, G.; Micchelli, O.; Ghirardi, O.; Ramacci, M.; Conti, E. Changes in brain memebrane phospholipid composition during aging: Effect of Acetyl-l-carnitine. J. Neurochem. 1991, 57, S115. [Google Scholar]
- Rosenthal, R.E.; Williams, R.; Bogaert, Y.E.; Getson, P.R.; Fiskum, G. Prevention of postischemic canine neurological injury through potentiation of brain energy metabolism by acetyl-L-carnitine. Stroke 1992, 23, 1312–1317. [Google Scholar] [CrossRef]
- Forloni, G.; Angeretti, N.; Smiroldo, S. Neuroprotective activity of acetyl-L-carnitine: Studies in vitro. J. Neurosci. Res. 1994, 37, 92–96. [Google Scholar] [CrossRef]
- Ghirardi, O.; Caprioli, A.; Milano, S.; Giuliani, A.; Ramacci, M.T.; Angelucci, L. Active avoidance learning in old rats chronically treated with levocarnitine acetyl. Physiol. Behav. 1992, 52, 185–187. [Google Scholar] [CrossRef]
- Ames, B.N.; Liu, J. Delaying the mitochondrial decay of aging with acetylcarnitine. Ann. N. Y. Acad. Sci. 2004, 1033, 108–116. [Google Scholar] [CrossRef]
- Paradies, G.; Ruggiero, F.M.; Petrosillo, G.; Gadaleta, M.N.; Quagliariello, E. Effect of aging and acetyl-L-carnitine on the activity of cytochrome oxidase and adenine nucleotide translocase in rat heart mitochondria. FEBS Lett. 1994, 350, 213–215. [Google Scholar] [CrossRef]
- Pascale, A.; Milano, S.; Corsico, N.; Lucchi, L.; Battaini, F.; Martelli, E.A.; Trabucchi, M.; Govoni, S. Protein kinase C activation and anti-amnesic effect of acetyl-L-carnitine: In vitro and in vivo studies. Eur. J. Pharmacol. 1994, 265, 1–7. [Google Scholar] [CrossRef]
- Piovesan, P.; Pacifici, L.; Taglialatela, G.; Ramacci, M.T.; Angelucci, L. Acetyl-l-carnitine treatment increases choline acetyltransferase activity and NGF levels in the CNS of adult rats following total fimbria-fornix transection. Brain Res. 1994, 633, 77–82. [Google Scholar] [CrossRef]
- Taglialatela, G.; Navarra, D.; Cruciani, R.; Ramacci, M.T.; Alemà, G.S.; Angelucci, L. Acetyl-L-carnitine treatment increases nerve growth factor levels and choline acetyltransferase activity in the central nervous system of aged rats. Exp. Gerontol. 1994, 29, 55–66. [Google Scholar] [CrossRef]
- Castorina, M.; Ambrosini, A.M.; Pacific, L.; Ramacci, M.T.; Angelucci, L. Age-dependent loss of NMDA receptors in hippocampus, striatum, and frontal cortex of the rat: Prevention by acetyl-L-carnitine. Neurochem. Res. 1994, 19, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Abdul, H.M.; Calabrese, V.; Calvani, M.; Butterfield, D.A. Acetyl-L-carnitine-induced up-regulation of heat shock proteins protects cortical neurons against amyloid-beta peptide 1-42-mediated oxidative stress and neurotoxicity: Implications for Alzheimer’s disease. J. Neurosci. Res. 2006, 84, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Colombrita, C.; Sultana, R.; Scapagnini, G.; Calvani, M.; Butterfield, D.A.; Stella, A.M.G. Redox modulation of heat shock protein expression by acetylcarnitine in aging brain: Relationship to antioxidant status and mitochondrial function. Antioxid. Redox Signal. 2006, 8, 404–416. [Google Scholar] [CrossRef]
- Mazère, J.; Prunier, C.; Barret, O.; Guyot, M.; Hommet, C.; Guilloteau, D.; Dartigues, J.F.; Auriacombe, S.; Fabrigoule, C.; Allard, M. In vivo SPECT imaging of vesicular acetylcholine transporter using [(123)I]-IBVM in early Alzheimer’s disease. NeuroImage 2008, 40, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Kendziorra, K.; Wolf, H.; Meyer, P.M.; Barthel, H.; Hesse, S.; Becker, G.A.; Luthardt, J.; Schildan, A.; Patt, M.; Sorger, D.; et al. Decreased cerebral α4β2* nicotinic acetylcholine receptor availability in patients with mild cognitive impairment and Alzheimer’s disease assessed with positron emission tomography. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 515–525. [Google Scholar] [CrossRef]
- Minoshima, S.; Giordani, B.; Berent, S.; Frey, K.A.; Foster, N.L.; Kuhl, D.E. Metabolic reduction in the posterior cingulate cortex in very early Alzheimer’s disease. Ann. Neurol. 1997, 42, 85–94. [Google Scholar] [CrossRef]
- Schneider, J.A.; Arvanitakis, Z.; Bang, W.; Bennett, D.A. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology 2007, 69, 2197–2204. [Google Scholar] [CrossRef]
- Jellinger, K.A. Pathology and pathogenesis of vascular cognitive impairment-a critical update. Front. Aging Neurosci. 2013, 5, 17. [Google Scholar] [CrossRef]
- Bella, R.; Pennisi, G.; Cantone, M.; Palermo, F.; Pennisi, M.; Lanza, G.; Zappia, M.; Paolucci, S. Clinical presentation and outcome of geriatric depression in subcortical ischemic vascular disease. Gerontology 2010, 56, 298–302. [Google Scholar] [CrossRef]
- Nardone, R.; Golaszewski, S.; Ladurner, G.; Tezzon, F.; Trinka, E. A review of transcranial magnetic stimulation in the in vivo functional evaluation of central cholinergic circuits in dementia. Dement. Geriatr. Cogn. Disord. 2011, 32, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Bella, R.; Cantone, M.; Lanza, G.; Ferri, R.; Vinciguerra, L.; Puglisi, V.; Pennisi, M.; Ricceri, R.; Di Lazzaro, V.; Pennisi, G. Cholinergic circuitry functioning in patients with vascular cognitive impairment--no dementia. Brain Stimulat. 2016, 9, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Kavirajan, H.; Schneider, L.S. Efficacy and adverse effects of cholinesterase inhibitors and memantine in vascular dementia: A meta-analysis of randomised controlled trials. Lancet Neurol. 2007, 6, 782–792. [Google Scholar] [CrossRef]
- Kumaran, D.; Udayabanu, M.; Kumar, M.; Aneja, R.; Katyal, A. Involvement of angiotensin converting enzyme in cerebral hypoperfusion induced anterograde memory impairment and cholinergic dysfunction in rats. Neuroscience 2008, 155, 626–639. [Google Scholar] [CrossRef]
- Moghaddas, A.; Dashti-Khavidaki, S. L-Carnitine and Potential Protective Effects Against Ischemia-Reperfusion Injury in Noncardiac Organs: From Experimental Data to Potential Clinical Applications. J. Diet. Suppl. 2018, 15, 740–756. [Google Scholar] [CrossRef]
- Malaguarnera, M. Acetyl-L-carnitine in hepatic encephalopathy. Metab. Brain Dis. 2013, 28, 193–199. [Google Scholar] [CrossRef]
- Rose, C.; Felipo, V. Limited capacity for ammonia removal by brain in chronic liver failure: Potential role of nitric oxide. Metab. Brain Dis. 2005, 20, 275–283. [Google Scholar] [CrossRef]
- Rodrigo, R.; Cauli, O.; Boix, J.; ElMlili, N.; Agusti, A.; Felipo, V. Role of NMDA receptors in acute liver failure and ammonia toxicity: Therapeutical implications. Neurochem. Int. 2009, 55, 113–118. [Google Scholar] [CrossRef]
- Cauli, O.; Rodrigo, R.; Llansola, M.; Montoliu, C.; Monfort, P.; Piedrafita, B.; El Mlili, N.; Boix, J.; Agustí, A.; Felipo, V. Glutamatergic and gabaergic neurotransmission and neuronal circuits in hepatic encephalopathy. Metab. Brain Dis. 2009, 24, 69–80. [Google Scholar] [CrossRef]
- Calabrese, V.; Ravagna, A.; Colombrita, C.; Scapagnini, G.; Guagliano, E.; Calvani, M.; Butterfield, D.A.; Giuffrida Stella, A.M. Acetylcarnitine induces heme oxygenase in rat astrocytes and protects against oxidative stress: Involvement of the transcription factor Nrf2. J. Neurosci. Res. 2005, 79, 509–521. [Google Scholar] [CrossRef]
- Szabó, K.; Nagy, Z.; Juhász, V.; Zolnerciks, J.K.; Csorba, A.; Tímár, Z.; Molnár, É.; Pádár, P.; Johnson, W.; Beéry, E.; et al. Species specificity profiling of rat and human organic cation/carnitine transporter Slc22a5/SLC22A5 (Octn2/OCTN2). Drug Metab. Pharmacokinet. 2017, 32, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.E.; Evans, A.M. Carnitine and acylcarnitines: Pharmacokinetic, pharmacological and clinical aspects. Clin. Pharmacokinet. 2012, 51, 553–572. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, L.; Zampino, M. A mitochondrial root to accelerated ageing and frailty. Nat. Rev. Endocrinol. 2020, 16, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Madiraju, P.; Pande, S.V.; Prentki, M.; Madiraju, S.R.M. Mitochondrial acetylcarnitine provides acetyl groups for nuclear histone acetylation. Epigenetics 2009, 4, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Nasca, C.; Xenos, D.; Barone, Y.; Caruso, A.; Scaccianoce, S.; Matrisciano, F.; Battaglia, G.; Mathé, A.A.; Pittaluga, A.; Lionetto, L.; et al. L-acetylcarnitine causes rapid antidepressant effects through the epigenetic induction of mGlu2 receptors. Proc. Natl. Acad. Sci. USA 2013, 110, 4804–4809. [Google Scholar] [CrossRef]
- Chiechio, S.; Canonico, P.L.; Grilli, M. l-Acetylcarnitine: A Mechanistically Distinctive and Potentially Rapid-Acting Antidepressant Drug. Int. J. Mol. Sci. 2017, 19, 11. [Google Scholar] [CrossRef]
- Ferreira, G.C.; McKenna, M.C. L-Carnitine and Acetyl-L-carnitine Roles and Neuroprotection in Developing Brain. Neurochem. Res. 2017, 42, 1661–1675. [Google Scholar] [CrossRef]
- Mancuso, C.; Siciliano, R.; Barone, E.; Preziosi, P. Natural substances and Alzheimer’s disease: From preclinical studies to evidence based medicine. Biochim. Biophys. Acta 2012, 1822, 616–624. [Google Scholar] [CrossRef]
- Freidl, W.; Schmidt, R.; Stronegger, W.J.; Irmler, A.; Reinhart, B.; Koch, M. Mini mental state examination: Influence of sociodemographic, environmental and behavioral factors and vascular risk factors. J. Clin. Epidemiol. 1996, 49, 73–78. [Google Scholar] [CrossRef]
| Disorder | Reference | Study Characteristics | Main Results |
|---|---|---|---|
| Alzheimer’s Disease/Mild Cognitive Impairment | Battistin et al., 1989 [39] | Type of study: randomized, double blind placebo controlled Subjects: 137 AD patients Treatment: ALC 2 g die orally for 180 days Outcome measures: Digit span, block tapping, verbal fluency, copying test, MMSE, BDS, BIMC, Raven’s matrices, digit symbol, Rey’s test | Improvement in verbal fluency in the treated group. |
| Battistin et al., 1989 [40] | Type of study: prospective Subjects: 30 demented patients (21 AD, 9 with mixed dementia) randomly assigned to different groups Treatment: ALC 0.5 g, 1 g, 1.5 g, or 2 g die IV Outcome measures: regional cerebral blood flow measured with SPECT with 99mTc-HM-PAO | The lowest dose of ALC did not produce changes. Uptake increases in the parietal cortex after higher doses intake; significant elevations in the frontal cortex after 1.5 g and in the temporal region after 2 g. When side-to-side cortical asymmetries were present, they significantly reduced after ALC. 99mTc-HM-PAO uptake increased in thalamus after 1 and 1.5 g. | |
| Bellagamba et al., 1990 [41] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 35 AD patients randomized in real (16) and placebo (19) groups Treatment: ALC 3 g die orally for 3 months Outcome measures: SCAG, Rey’s Auditory Verbal Learning Test, Raven PM 47 and TP Barrage Test, CGI | Significant improvement in behavioral profile, memory, attention, intellective function, and CGI for ALC compared to placebo. | |
| Campi et al., 1990 [42] | Type of study: single-blind, randomized, parallel Subjects: 40 AD patients randomized in ALC and selegiline groups Treatment: ALC 0.5 g bis in die orally for 90 days Outcome measures: psychometric examination, at baseline and every 30 days | Selegiline led to a global improvement in processing, storage, retrieval of given information, verbal fluency, and visuospatial abilities. Excellent tolerability of both drugs. | |
| Passeri et al., 1990 [43] | Type of study: randomized, double-blind, placebo controlled Subjects: 60 MCI patients randomized in real (30) and placebo (30) groups Treatment: ALC 2 g die orally for 3 months Outcome measures: MMSE, SHGRS, BDS, BIMC, Rey’s test, Corsi’s test, verbal fluency, TP, digit span, HDRS, Gibson’s test | Statistically significant improvement in behavioral scales, memory tests, attention barrage test, and verbal fluency test for ALC compared to placebo. | |
| Rai et al., 1990 [44] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 20 AD patients randomized in real (7) and placebo (13) groups Treatment: ALC 1 g bis in die orally for 24 weeks Outcome measures: P300 latency of the auditory evoked potentials, GDS - short form; Kendrick Battery Tests, OLT and Digit Copying Test, NLT, Word Fluency Test-modification of Set Test, ADL | No changes for P300 latency, ADL, GDS, Digit Copying Test and Word Fluency Test. Trend for more improvement in relation to the NLT, computerized Digit Recall Test and reaction time in the ALC group compared to placebo. | |
| Spagnoli et al., 1991 [16] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 130 AD patients Treatment: ALC 2 g die orally for one year Outcome measures: SBI, BDS, BIMC, Raven’s matrices, prose memory, apraxia, finger agnosia | Compared to controls, significant improvement in BDS, ideomotor and bucco-facial apraxia, logical intelligence, and selective attention for ALC. Better performance at BDS, logical intelligence, verbal critical abilities, long-term verbal memory, and selective attention at the analysis of covariance. | |
| Parnetti et al., 1992 [20] | Type of study: prospective Subjects: 11 AD patients Treatment: ALC 30 mg/kg bis in die IV for 10 days; ALC 2 g die orally in 3 daily doses for 50 days Outcome measures: ALC concentration in plasma and CSF | IV and oral administration of multiple doses of ALC increased both plasma and CSF concentration of ALC. | |
| Sano et al., 1992 [29] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 27 AD patients randomized in real (13) and placebo (14) groups Treatment: ALC 2.5 g die for 3 months, followed by 3 g die for 3 months Outcome measures: SRT; modified MMS, which included: Digit Span (forward-backward); the Logical Memory, Paired Associate, and Visual Reproduction subtests from the Wechsler Memory Scale; the Benton Visual Retention Test-Multiple Choice Version; verbal fluency test for letter; the category naming test; cancellation test scoring time and errors; SIP, SMQ, CGI; CSF ALC | Significantly less deterioration in timed cancellation tasks and Digit Span (forward) and a trend toward less deterioration in a timed verbal fluency task for ALC group compared to placebo; no difference in any other neuropsychological test. A subgroup with the lowest baseline scores had significantly less deterioration on the verbal memory test and a significant increase in CSF ALC levels compared to placebo. | |
| Costa et al., 1993 [45] | Type of study: randomized, parallel, double blind, placebo controlled Treatment: ALC 2 g die for 16 weeks Outcome measures: Reys test, Digit span, Corsi’s test, digit symbol test, Gibson’s spiral, verbal fluency, GBS | ALC normalized cortisol and ACTH level in response to a CRF stimulation test and reduced the number of non-suppressants in a dexamethasone test. Improvement at Digit Symbol Substitution test, verbal fluency test, and Rey’s verbal test. | |
| Bayer 1994 [46] | Type of study: randomized, double blind, placebo controlled Subjects: 30 AD patients Treatment: ALC 2 g die for 24 weeks Outcome measures: MMSE, HRSD, BDS, GBS, KOLT, KDCT, CGI-S, CGI-C, CGI-E, Relatives Assessment | Improvement in CGI for the treatment group compared to placebo. | |
| Mullin 1994 [47] | Type of study: randomized, parallel, double blind, placebo controlled Subjects: 62 AD patients Treatment: ALC 2 g die orally for 180 days Outcome measures: MMSE, CGI, GDS, NART, KOLT, KDGT, NLT, ADL, Word Fluency | No significant difference between treatment and control group. | |
| Bruno et al., 1995 [48] | Type of study: open label Subjects: AD patients Treatment: ALC at highly dose IV Outcome measures: CSF and plasma ALC concentrations | CSF levels under ALC treatment were significantly higher compare to baseline. Beta-endorphins significantly decreased after treatment, with plasma cortisol levels matching this reduction. | |
| Pettegrew et al., 1995 [23] | Type of study: double-blind, placebo controlled Subjects: 12 AD patients randomized in real (7) and placebo (5) groups Treatment: ALC 3 g die orally for one year Outcome measures: MMS, ADAS, membrane phospholipid and high-energy phosphate metabolism measured by 31P-MRS | Patients showed significantly less deterioration in MMS and ADAS scores. The decrease in phosphomonoester and high-energy phosphate levels observed in both ALC and placebo groups at baseline was normalized in the real group but not in the placebo group. | |
| Thal et al., 1996 [49] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 419 AD patients randomized in real (207) and placebo (212) groups Treatment: ALC 3 g die orally for one year Outcome measures: primary: ADAS, CDR; secondary: ADAS-non-cognitive subscale, MMSE, ADL, IADL, CGI-S and CGI-C | Both groups declined on primary and most of secondary measures during the trial. A trend for early-onset patients on ALC to decline more slowly than placebo on both primary endpoints was found; conversely, late-onset AD patients on ALC tended to progress more rapidly than early-onset. | |
| Brooks et al., 1998 [36] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 334 AD patients randomized in real (165) and placebo (169) groups Treatment: ALC 3 g die orally for one year Outcome measures: primary: ADAS, CDR; secondary: ADAS, CDR; secondary: ADAS-non-cognitive subscale, MMSE, ADL, IADL, CGI-S and CGI-C | Reanalysis of the data by Thal et al., 1996 by using the trilinear approach, in which measurements are allowed to follow a pattern of stability-change-stability. Both groups exhibited the same mean rate of change on ADAS. Multiple regression analysis revealed younger subjects benefiting more from ALC. | |
| Thal et al., 2000 [50] | Type of study: randomized, double-blind, placebo controlled Subjects: 229 AD patients randomized in real (112) and placebo (117) groups Treatment: ALC 1 g ter in die orally for one year Outcome measures: primary: ADAS, CDR; secondary: ADAS-non-cognitive subscale, MMSE, ADL, CIBIC | No significant difference was found in the primary outcomes; less deterioration in MMSE score was observed for ALC, whereas no difference for ADL and CIBIC. | |
| Bianchetti et al., 2003 [51] | Type of study: open-label Subjects: 21 AD patients treated with donezepil or rivastigmine. Treatment: ALC 2 g die orally for 3 months Outcome measures: ADAS-cognitive subscale, MMSE | Response rate (defined as a reduction of ADAS-Cog score ≥4) increased from 38% to 50% after ALC administration. | |
| Jeong et al., 2017 [52] | Type of study: prospective Subjects: 18 AD patients Treatment: ALC 1.5 g die orally for 1.4 ± 0.3 years Outcome measures: SPECT with 99mTc-HM-PAO, MMSE, CDR, GDS, NPI | Non-significant changes in MMSE, CDR, GDS, and NPI. Cerebral perfusion significantly increased in the right precuneus, whereas it reduced in the left inferior temporal gyrus, the right middle frontal gyrus, and the right insular cortex | |
| Vascular Cognitive Impairment | Arrigo et al., 1988 [53] | Type of study: randomized, double-blind, crossover Subjects: 12 patients with cerebrovascular insufficiency (not further specified) Treatment: ALC 1.5 g die orally for 4 weeks Outcome measures: verbal and numerical memory, non-verbal performance, reaction times (simple and multiple-choice) and EEG | ALC significantly more active than placebo in memory and non-verbal tests and in simple reaction times. |
| Arrigo et al., 1990 [54] | Type of study: double-blind, cross-over Subjects: 12 elderly patients with acute brain circulatory insufficiency Treatment: ALC 1.5 g die orally for 4 weeks Outcome measures: memory, number, and word tests; responses to simple stimuli and performance of the maze test | Significant differences between drug and placebo in memory, number, and word tests, as well as in the responses to simple stimuli and the performance of the maze test. No side effects. | |
| Yang et al., 2018 [55] | Type of study: multicenter, randomized, double-blind, placebo-controlled, parallel-group trial Subjects: 56 patients with dementia and cerebrovascular disease, randomized in real (30) and placebo (26) groups Treatment: ALC 0.5 g ter in die for 28 weeks Outcome measures: primary measure: MoCA-K; secondary measures: K-MMSE, K-CWST, COWAT, K-TMT-E, K-IADL, GDS, CDR, CDR-SB | Cognitive function measured by the MoCA-K significantly improved in the ALC-treated group, in particular the attention and language sub-items. No difference in secondary outcome measures. | |
| Hepatic Encephalopathy | Siciliano et al., 2006 [56] | Type of study: prospective Subjects: 18 cirrhotic patients with HE and 6 with previous TIA as controls Treatment: ALC 0.5 g in 50 mL isotonic saline IV (infusion rate 10 mL/min) Outcome measures: pattern reversal P100 latency of visual-evoked potentials | Significant reduction in P100 latencies 30 min after ALC infusion in HE patients. The mean P100 latencies measured in HE subjects was significantly shorter after ALC infusion compared with values obtained before ALC administration. |
| Malaguarnera et al. 2008 [57] | Type of study: randomized, double-blind, placebo controlled Subjects: 115 minimal HE patients randomized in real (60) and placebo (55) Treatment: ALC 2 g bis in die orally for 90 days Outcome measures: TMT, BDT, AVL, MMSE, and EEG | Improvement of TMT-A, TMT-B, MMSE, BDT, SDMT, and AVL scores in the real group; no difference in EEG. No differences in the treated group compared to baseline and placebo in both neurophysiological and neuropsychological assessment. | |
| Malaguarnera et al., 2011 [58] | Type of study: randomized, double-blind, placebo controlled. Subjects: 67 minimal HE patients randomized in real (33) and placebo (34) Treatment: ALC 2 g bis in die orally for 90 days Outcome measures: Line Drawing Test, Serial Dotting Test, TMT, SF-36, BDI, STAI, and EEG | Significant improvements in BDI, TMT-B, STAI, and line tracing in real group compared to baseline and placebo group. No differences in EEG. | |
| Malaguarnera et al., 2011 [59] | Type of study: randomized, double-blind, placebo controlled Subjects: 121 HE patients divided in mild (HE1: 61) and moderate (HE2: 60), randomized to ALC (31 HE1 and 30 HE2) or placebo (30 HE1 and 30 HE2) Treatment: 2 g ALC bis in die orally for 90 days Outcome measures: FSS, Wessely’s test, Powell’s test, 7-d PAR, SPPB, 6-min walking test, and EEG. | The ALC-treated patients in the HE1 group showed significant improvement than placebo group in mental fatigue score, FSS, 7-d PAR score, and SPPB. The HE2 group showed significant improvement in FSS and in the 6-min walking test. | |
| Malaguarnera et al., 2011 [60] | Type of study: randomized, double-blind, placebo controlled Subjects: 60 severe HE patients randomized in real (30) and placebo (30) groups Treatment: ALC 2 g bis in die orally for 90 days Outcome measures: TMT, digit cancellation, MMSE, COWAT, JLO, Logical Memory (Paragraph Recall), EMQ, Hooper visual organization test, and EEG | Significant improvement in the real group compared to placebo for EMQ, Paragraph Recall, TMT-A, TMT-B, COWAT, Hooper test, JLO, and digit cancellation time. | |
| Others Secondary Dementias | Goetz et al., 1990 [61] | Type of study: randomized, double-blind, placebo controlled, crossover Subjects: 10 patients with HD randomized to real and placebo treatment and switched over after a 2-week drug-free period. Treatment: ALC 45 mg/kg/day for one week Outcome measures: Shoulson–Fahn Disability Scale for HD, AIMS, reaction time, MMSE, HDRS, Verbal Fluency using the Controlled Oral Word Association Test, global severity assessment from AIMS. | Both placebo and ALC significantly improved reaction time compared to baseline and did not significantly differed from each other. No serious side effects of ALC during the study. |
| Tempesta et al., 1990 [62] | Type of study: randomized, double-blind, placebo controlled Subjects: 55 chronic alcoholic patients randomized in real and placebo group Treatment: ALC 2 g die orally for 90 days Outcome measures: memory, constructional praxia, deductive-logical functions, and language. Testing time at baseline (T0), after 45 (T45) and 90 (T90) days | At T90, significant differences favoring the real treatment on the Rey’s 15-word memory test, the Wechsler memory scale, and the Similarities WAIS subtest were noted. On the copying drawing test, the placebo group did not show any T0-T90 variation, while improvement was observed in the ALC group. | |
| Famularo et al., 1999 [63] | Type of study: case report, followed by a prospective pilot study Subjects: one patient with AIDS-dementia complex Treatment: single course of ALC IV Outcome measures: motor and cognitive tests; blood and CSF glutamate level | Significant motor and cognitive improvement after ALC, along with a significant reduction of glutamate concentration in both blood and CSF, as confirmed by the prospective pilot study on blood levels of glutamate in AIDS patients. | |
| Pueschel, et al., 2006 [64] | Type of study: randomized, double-blind, placebo controlled Subjects: 40 Down syndrome randomized in real and placebo group Treatment: ALC 10 mg/kg/day (first month), 20 mg/kg/day (second month), 30 mg/kg/day (next 4 months) Outcome measures: SBIS, Hiskey–Nebraska VAS, MFFT, WISC-r, KABC, VABS, CBCL; assessment performed at follow-up of 3, 6, and 9 months | No significant difference between the two groups. | |
| Unspecified cognitive disorder | Passeri et al., 1988 [65] | Type of study: randomized, double-blind, placebo controlled Subjects: 30 elderly patients with “mild mental impairment” randomized in real and placebo group (15 patients each) Treatment: ALC 2 g die orally for 3 months Outcome measures: BDS, SHGRS, Rey short-term, Rey long-term, Corsi, Barrage, and Verbal Fluency | Overall, no significant difference between groups. ALC treated patients showed improvement in BSD, SHGRS, Rey short- and long-term memory tests, Corsi, Barrage test, and in the Verbal Fluency test. |
| Mantero et al., 1989 [66] | Type of study: double-blind, placebo controlled Subjects: 50 patients with dementia (not further specified) Treatment: 2 g ALC die orally for 180 days Outcome measures: MMSE, BDS, BIMC, HRSD, CGI-C, CGI-S, CGI-E, HIS | Improvement in MMSE, CGI, and BDS in the treated group. | |
| Herrmann et al., 1990 [67] | Type of study: randomized, double-blind, placebo controlled Subjects: 230 patients aged 60–80 with cognitive decline corresponding to stages 3 or 4 on GDS (80% with stage 3), randomized to real and placebo group Treatment: ALC 1.5 g die for 3 months Outcome measures: CGI-C, NGDAS, modified digit symbol substitution test | Significant effect of the ALC treatment on the physician’s CGI and the patient-rated level of ADL. | |
| Sinforiani et al., 1990 [68] | Type of study: single-blind Subjects: 24 patients with mild-to-moderate dementia according to DSM III criteria divided in ALC group and piracetam group (12 patients each) Treatment: ALC IV for 2 weeks followed by oral ALC for 10 weeks Outcome measures: cognitive, attentive, and behavioral functions | Statistically significant improvement in behavioral, attentional, and psychomotricity features of patients treated with ALC. | |
| Livingston et al., 1991 [69] | Type of study: randomized, double-blind, parallel, placebo controlled Subjects: 71 patients with dementia randomized in real (35) and placebo (36) Treatment: ALC for 24 weeks (dosage not specified) Outcome measures: MMSE, KOLT, word fluency, drawing, recognition memory for words and pictures, modified NLT, PADL, GCI | Statistically significant improvement in the recognition memory in the ALC group. | |
| Salvioli and Neri 1994 [70] | Type of study: single-blind Subjects: 481 elderly patients with “mental decline” (not further specified) Treatment: phase T0: placebo treatment for 30 days; T1 and T2: ALC 1.5 g die orally for 90 days; T3: further 30 days of placebo treatment Outcome measures: MMSE, Randt Memory Test, GDS, HDRS, FSS | Significant improvement observed during and after ALC treatment. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pennisi, M.; Lanza, G.; Cantone, M.; D’Amico, E.; Fisicaro, F.; Puglisi, V.; Vinciguerra, L.; Bella, R.; Vicari, E.; Malaguarnera, G. Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients 2020, 12, 1389. https://doi.org/10.3390/nu12051389
Pennisi M, Lanza G, Cantone M, D’Amico E, Fisicaro F, Puglisi V, Vinciguerra L, Bella R, Vicari E, Malaguarnera G. Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients. 2020; 12(5):1389. https://doi.org/10.3390/nu12051389
Chicago/Turabian StylePennisi, Manuela, Giuseppe Lanza, Mariagiovanna Cantone, Emanuele D’Amico, Francesco Fisicaro, Valentina Puglisi, Luisa Vinciguerra, Rita Bella, Enzo Vicari, and Giulia Malaguarnera. 2020. "Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update" Nutrients 12, no. 5: 1389. https://doi.org/10.3390/nu12051389
APA StylePennisi, M., Lanza, G., Cantone, M., D’Amico, E., Fisicaro, F., Puglisi, V., Vinciguerra, L., Bella, R., Vicari, E., & Malaguarnera, G. (2020). Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients, 12(5), 1389. https://doi.org/10.3390/nu12051389








