Patients with Community Acquired Pneumonia Exhibit Depleted Vitamin C Status and Elevated Oxidative Stress
Abstract
1. Introduction
2. Methods
2.1. Setting and Study Participants
2.2. Inclusion and Exclusion Criteria
2.3. Disease Severity Scores
2.4. Blood Sampling and Processing
2.5. Analysis of Blood Analytes
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
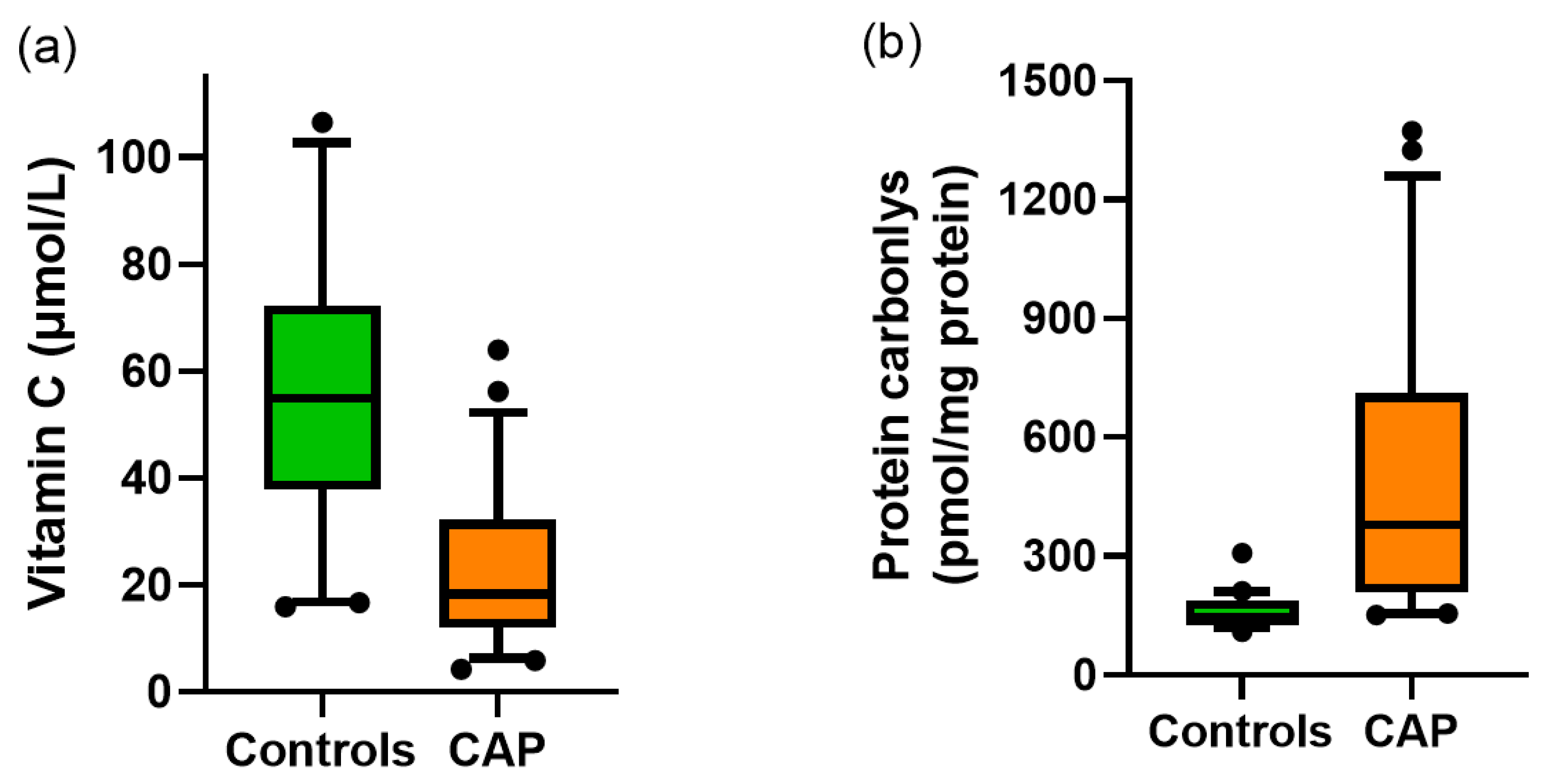
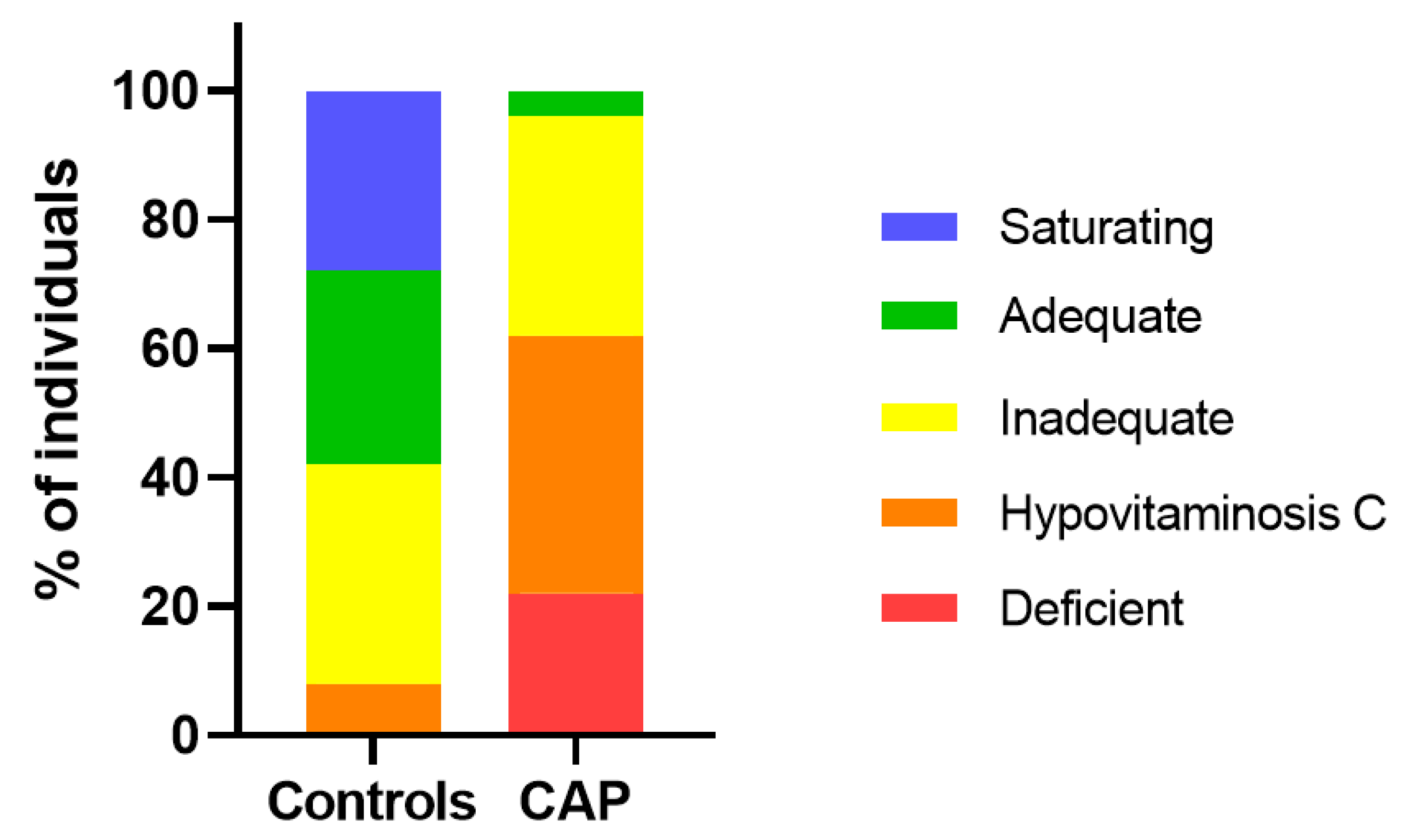
3.2. Vitamin C and Protein Carbonyls
3.3. Biomarker Correlations with Clinical and Physiological Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Musher, D.M.; Thorner, A.R. Community-acquired pneumonia. N. Engl. J. Med. 2014, 371, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.C.; Liu, Y.H.; Wang, C.Y.; Wang, Y.H.; Hsueh, S.C.; Yen, M.Y.; Ko, W.C.; Hsueh, P.R. Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): Facts and myths. J. Microbiol. Immunol. Infect 2020. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Health Estimates 2016: Deaths by Cause, Age, Sex, by Country and by Region, 2000–2016; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- GBD 2016 Lower Respiratory Infections Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis. 2018, 18, 1191–1210. [Google Scholar] [CrossRef]
- Burton, D.C.; Flannery, B.; Bennett, N.M.; Farley, M.M.; Gershman, K.; Harrison, L.H.; Lynfield, R.; Petit, S.; Reingold, A.L.; Schaffner, W.; et al. Socioeconomic and racial/ethnic disparities in the incidence of bacteremic pneumonia among US adults. Am. J. Public Health 2010, 100, 1904–1911. [Google Scholar] [CrossRef]
- Chambers, S.; Laing, R.; Murdoch, D.; Frampton, C.; Jennings, L.; Karalus, N.; Mills, G.; Town, I. Maori have a much higher incidence of community-acquired pneumonia and pneumococcal pneumonia than non-Maori. N. Z. Med. J. 2006, 119, U1978. [Google Scholar]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef]
- Myint, P.K.; Wilson, A.M.; Clark, A.B.; Luben, R.N.; Wareham, N.J.; Khaw, K.T. Plasma vitamin C concentrations and risk of incident respiratory diseases and mortality in the European Prospective Investigation into Cancer-Norfolk population-based cohort study. Eur. J. Clin. Nutr. 2019, 73, 1492–1500. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Carr, A.C. Vitamin C in pneumonia and sepsis. In Vitamin C: New Biochemical and Functional Insights. Oxidative Stress and Disease; Chen, Q., Vissers, M., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2020; pp. 115–135. [Google Scholar]
- Bakaev, V.V.; Duntau, A.P. Ascorbic acid in blood serum of patients with pulmonary tuberculosis and pneumonia. Int. J. Tuberc. Lung Dis. 2004, 8, 263–266. [Google Scholar]
- Chakrabarti, B.; Banerjee, S. Dehydroascorbic acid level in blood of patients suffering from various infectious diseases. Proc. Soc. Exp. Biol. Med. 1955, 88, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Hunt, C.; Chakravorty, N.K.; Annan, G.; Habibzadeh, N.; Schorah, C.J. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int. J. Vitam. Nutr. Res. 1994, 64, 212–219. [Google Scholar] [PubMed]
- Mochalkin, N.I. Ascorbic acid in the complex therapy of acute pneumonia. Voenno-Meditsinskii Zhurnal 1970, 9, 17–21, (English translation: http://www.mv.helsinki.fi/home/hemila/T5.pdf). [Google Scholar] [PubMed]
- Halm, E.A.; Fine, M.J.; Marrie, T.J.; Coley, C.M.; Kapoor, W.N.; Obrosky, D.S.; Singer, D.E. Time to clinical stability in patients hospitalized with community-acquired pneumonia: Implications for practice guidelines. JAMA 1998, 279, 1452–1457. [Google Scholar] [CrossRef] [PubMed]
- Niederman, M.S.; Mandell, L.A.; Anzueto, A.; Bass, J.B.; Broughton, W.A.; Campbell, G.D.; Dean, N.; File, T.; Fine, M.J.; Gross, P.A.; et al. Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am. J. Respir. Crit. Care Med. 2001, 163, 1730–1754. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.S.; van der Eerden, M.M.; Laing, R.; Boersma, W.G.; Karalus, N.; Town, G.I.; Lewis, S.A.; Macfarlane, J.T. Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study. Thorax 2003, 58, 377–382. [Google Scholar] [CrossRef]
- Carr, A.C.; Pullar, J.M.; Moran, S.; Vissers, M.C. Bioavailability of vitamin C from kiwifruit in non-smoking males: Determination of ‘healthy’ and ‘optimal’ intakes. J. Nutr. Sci. 2012, 1, e14. [Google Scholar] [CrossRef]
- Buss, H.; Chan, T.P.; Sluis, K.B.; Domigan, N.M.; Winterbourn, C.C. Protein carbonyl measurement by a sensitive ELISA method. Free Radic. Biol. Med. 1997, 23, 361–366. [Google Scholar] [CrossRef]
- Pullar, J.M.; Bayer, S.; Carr, A.C. Appropriate handling, processing and analysis of blood samples is essential to avoid oxidation of vitamin C to dehydroascorbic acid. Antioxidants 2018, 7, 29. [Google Scholar] [CrossRef]
- Weber, D.; Davies, M.J.; Grune, T. Determination of protein carbonyls in plasma, cell extracts, tissue homogenates, isolated proteins: Focus on sample preparation and derivatization conditions. Redox Biol. 2015, 5, 367–380. [Google Scholar] [CrossRef]
- Winterbourn, C.C.; Buss, I.H.; Chan, T.P.; Plank, L.D.; Clark, M.A.; Windsor, J.A. Protein carbonyl measurements show evidence of early oxidative stress in critically ill patients. Crit. Care Med. 2000, 28, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Arnaout, L.; Remick, D. Hydrocortisone, ascorbic acid and thiamine (HAT) therapy decreases oxidative stress, improves cardiovascular function and improves survival in murine sepsis. Shock 2020, 53, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Vissers, M.C.; Wilkie, R.P. Ascorbate deficiency results in impaired neutrophil apoptosis and clearance and is associated with up-regulation of hypoxia-inducible factor 1alpha. J. Leukoc. Biol. 2007, 81, 1236–1244. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.J.; Kraskauskas, D.; Martin, E.J.; Farkas, D.; Wegelin, J.A.; Brophy, D.; Ward, K.R.; Voelkel, N.F.; Fowler, A.A., 3rd; Natarajan, R. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L20–L32. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, B.M.; Fisher, B.J.; Kraskauskas, D.; Farkas, D.; Brophy, D.F.; Fowler, A.A.; Natarajan, R. Vitamin C: A novel regulator of neutrophil extracellular trap formation. Nutrients 2013, 5, 3131–3151. [Google Scholar] [CrossRef]
- Bozonet, S.M.; Carr, A.C. The role of physiological vitamin C concentrations on key functions of neutrophils isolated from healthy individuals. Nutrients 2019, 11, 1363. [Google Scholar] [CrossRef]
- Hume, R.; Weyers, E. Changes in leucocyte ascorbic acid during the common cold. Scott Med. J. 1973, 18, 3–7. [Google Scholar] [CrossRef]
- Wilson, C.W. Ascorbic acid function and metabolism during colds. Ann. N. Y. Acad. Sci. 1975, 258, 529–539. [Google Scholar] [CrossRef]
- Hemilä, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2013, 8, Cd005532. [Google Scholar]
- Padhani, Z.A.; Moazzam, Z.; Ashraf, A.; Bilal, H.; Salam, R.A.; Das, J.K.; Bhutta, Z.A. Vitamin C supplementation for prevention and treatment of pneumonia. Cochrane Database Syst. Rev. 2020, 4, 1–39. [Google Scholar]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef] [PubMed]
- Long, C.L.; Maull, K.I.; Krishnan, R.S.; Laws, H.L.; Geiger, J.W.; Borghesi, L.; Franks, W.; Lawson, T.C.; Sauberlich, H.E. Ascorbic acid dynamics in the seriously ill and injured. J. Surg. Res. 2003, 109, 144–148. [Google Scholar] [CrossRef]
- de Grooth, H.J.; Manubulu-Choo, W.P.; Zandvliet, A.S.; Spoelstra-de Man, A.M.E.; Girbes, A.R.; Swart, E.L.; Oudemans-van Straaten, H.M. Vitamin-C pharmacokinetics in critically ill patients: A randomized trial of four intravenous regimens. Chest 2018, 153, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.Y.; Jo, E.J.; Eom, J.S.; Mok, J.; Kim, M.H.; Kim, K.U.; Park, H.K.; Lee, M.K.; Lee, K. Combined vitamin C, hydrocortisone, and thiamine therapy for patients with severe pneumonia who were admitted to the intensive care unit: Propensity score-based analysis of a before-after cohort study. J. Crit. Care 2018, 47, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, e418. [Google Scholar] [CrossRef]
- Klenner, F.R. Virus pneumonia and its treatment with vitamin C. South Med. Surg. 1948, 110, 36–38. [Google Scholar]
| Characteristic | Total Cohort (n = 50) | AMAU (n = 44) | ICU (n = 6) |
|---|---|---|---|
| Age, years 1 | 68 (17) | 66 (22) | 58 (17) |
| Male sex, n (%) | 27 (54) | 23 (52) | 4 (67) |
| Comorbidities, n (%) | COPD 6 (14) Asthma 11 (25) Chronic heart failure 9 (20) Cardiovascular disease 19 (43) Diabetes 5 (11) Cerebrovascular disease 6 (14) Renal disease 3 (7) Solid organ malignancy 2 (5) Hematological malignancy 1 (2) Immune suppression 2 (5) | COPD 1 (17) Cardiovascular disease 1 (17) Renal failure 1 (17) Hematological malignancy 1 (17) Immune compromised 1 (17) | |
| Temperature, °C | 38 (1.2) | ||
| Hypothermia, n (%) | 2 (4.5) | ||
| Systolic BP | 139 (38) | 147 (33) | 80 (11) |
| Diastolic BP | 75 (20) | 79 (18) | 46 (10) |
| Heart rate, beats/min | 106 (22) | ||
| Respiratory rate, breaths/min | 36 (11) | ||
| Radiological confirmation, n (%) | 37 (84) | ||
| CURB-65 score (0–5) | 1.9 (1.3) | 1.8 (1.3) | 3.5 (0.6) 2 |
| SAPS II score (0–163) | 40 (11) | ||
| APACHE II score (0–79) | 20 (6) | ||
| APACHE III score (0–299) | 72 (24) | ||
| SOFA score (0–24) | 10 (3) | ||
| Vasopressors, n (%) | 0 | 6 (100) | |
| Mechanical ventilation, n (%) | 0 | 4 (67) | |
| FiO2 | 0.38 (0.07) | ||
| Hospital LOS, days 3 | 3 (0–99) | 3 (0–91) | 24 (5–99) |
| Mortality, n (%) | 2 (4.0) | 2 (4.5) | 0 |
| Parameter | Total Cohort (n = 50) | AMAU (n = 44) | ICU (n = 6) |
|---|---|---|---|
| White cell count (×109/L) 1 | 13 (6) | 13 (5) | 18 (13) |
| Neutrophils (×109/L) | 11 (5) | 11 (5) | 15 (10) |
| Hemoglobin (g/L) | 130 (17) | 130 (17) | 125 (26) |
| Platelets (×109/L) | 241 (117) | 244 (91) | 218 (227) |
| Urea (mmol/L) | 8.4 (6.5) | 7.4 (4.1) | 15 (13) |
| Creatinine (µmol/L) | 107 (61) | 98 (29) | 168 (138) |
| Bilirubin (µmol/L) | 19 (11) | 18 (9) | 25 (13) |
| C-reactive protein (mg/L) | 165 (127) | 152 (127) | 261 (78) 2 |
| Alanine transaminase (U/L) | 34 (21) | 33 (21) | 35 (23) |
| Alkaline phosphatase (U/L) | 112 (53) | ||
| Lactate (mmol/L) | 1.5 (0.8) | ||
| PaO2 (mmHg) | 79 (19) | ||
| PaO2/FiO2 | 212 (59) |
| Biomarker | Baseline Sample 1 | Second Sample 2 | p Value |
|---|---|---|---|
| Vitamin C (µmol/L) | 20 (7) | 22 (7) | 0.6 |
| Protein carbonyls (pmol/mg protein) | 541 (157) | 625 (252) | 0.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carr, A.C.; Spencer, E.; Dixon, L.; Chambers, S.T. Patients with Community Acquired Pneumonia Exhibit Depleted Vitamin C Status and Elevated Oxidative Stress. Nutrients 2020, 12, 1318. https://doi.org/10.3390/nu12051318
Carr AC, Spencer E, Dixon L, Chambers ST. Patients with Community Acquired Pneumonia Exhibit Depleted Vitamin C Status and Elevated Oxidative Stress. Nutrients. 2020; 12(5):1318. https://doi.org/10.3390/nu12051318
Chicago/Turabian StyleCarr, Anitra C., Emma Spencer, Liane Dixon, and Stephen T. Chambers. 2020. "Patients with Community Acquired Pneumonia Exhibit Depleted Vitamin C Status and Elevated Oxidative Stress" Nutrients 12, no. 5: 1318. https://doi.org/10.3390/nu12051318
APA StyleCarr, A. C., Spencer, E., Dixon, L., & Chambers, S. T. (2020). Patients with Community Acquired Pneumonia Exhibit Depleted Vitamin C Status and Elevated Oxidative Stress. Nutrients, 12(5), 1318. https://doi.org/10.3390/nu12051318






