Effects of a Ketogenic Diet Containing Medium-Chain Triglycerides and Endurance Training on Metabolic Enzyme Adaptations in Rat Skeletal Muscle
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Tissue Sampling
2.3. Analytical Procedure
2.3.1. Energy Substrates in Plasma
2.3.2. Muscle and Liver Glycogen Concentration
2.3.3. Muscle and Liver Homogenization
2.3.4. Western Blotting
2.3.5. Stripping and Reproving Membranes
2.3.6. Statistical Analysis
3. Results
3.1. Final Body Weight, Total Energy Intake, Intra-Abdominal Fat Weight and Food Efficiency
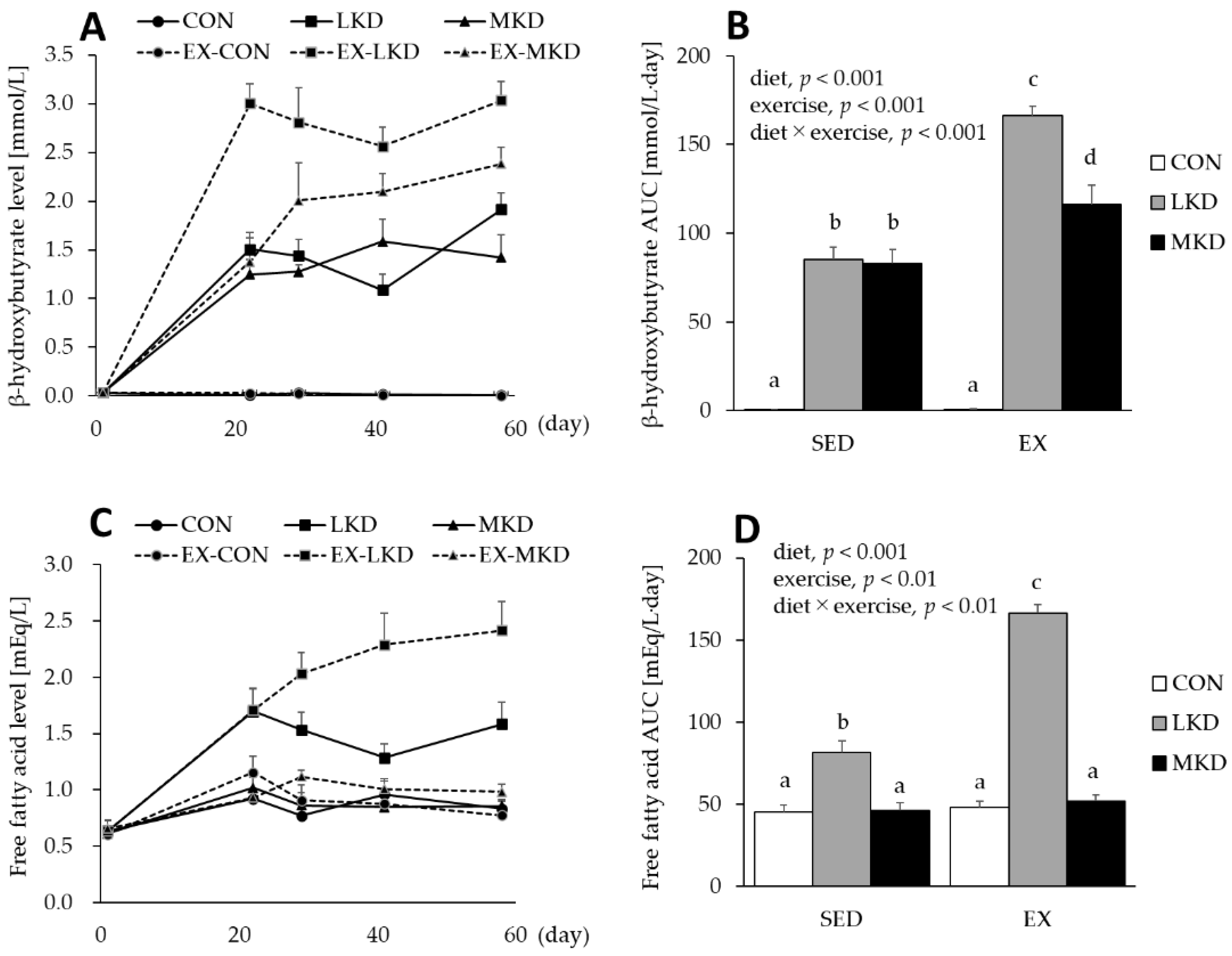
3.2. Plasma β-Hydroxybutyrate and Free Fatty Acid Concentrations
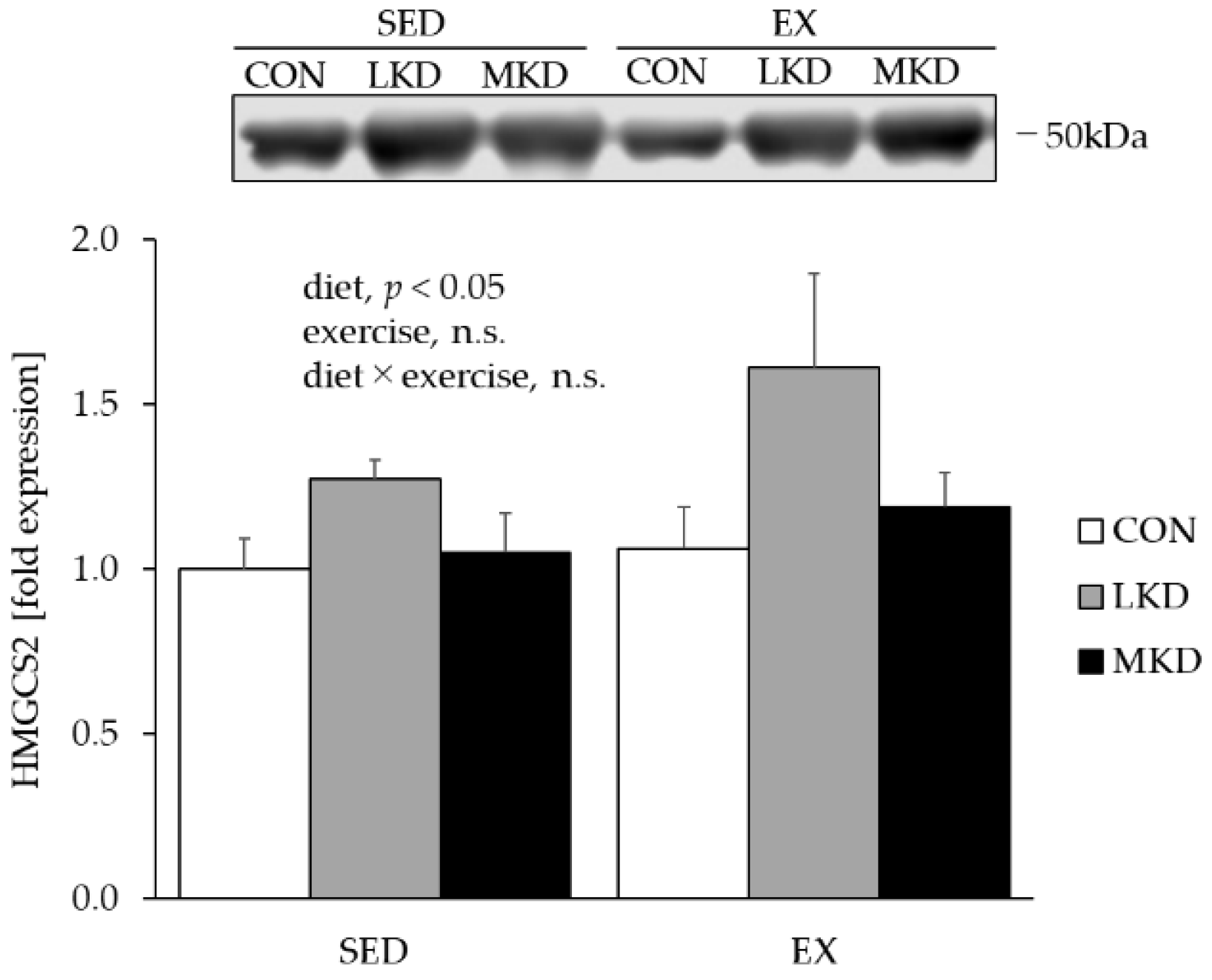
3.3. HMGCS2 Protein Content in the Liver
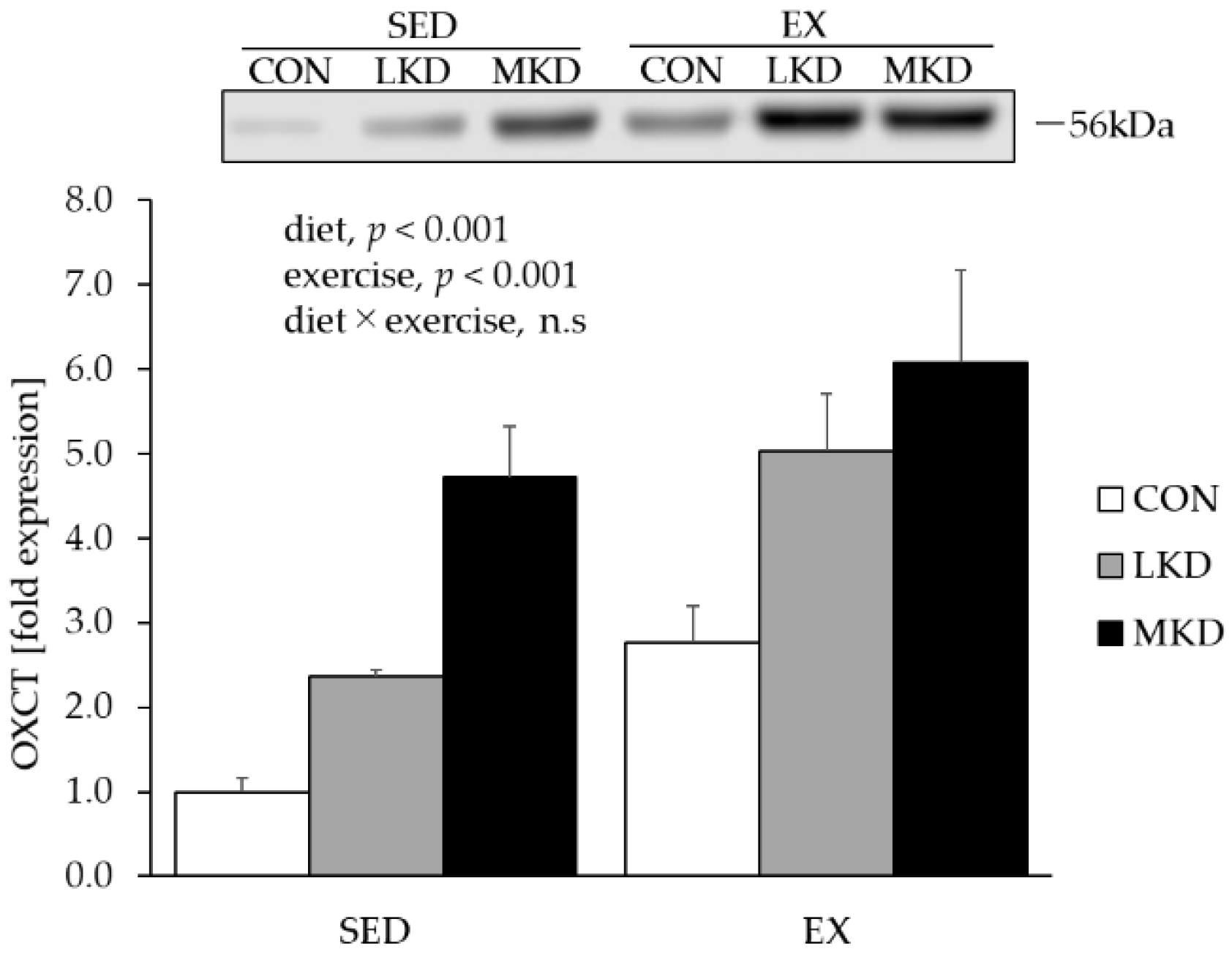
3.4. OXCT Protein Content in Epitrochlearis Muscle
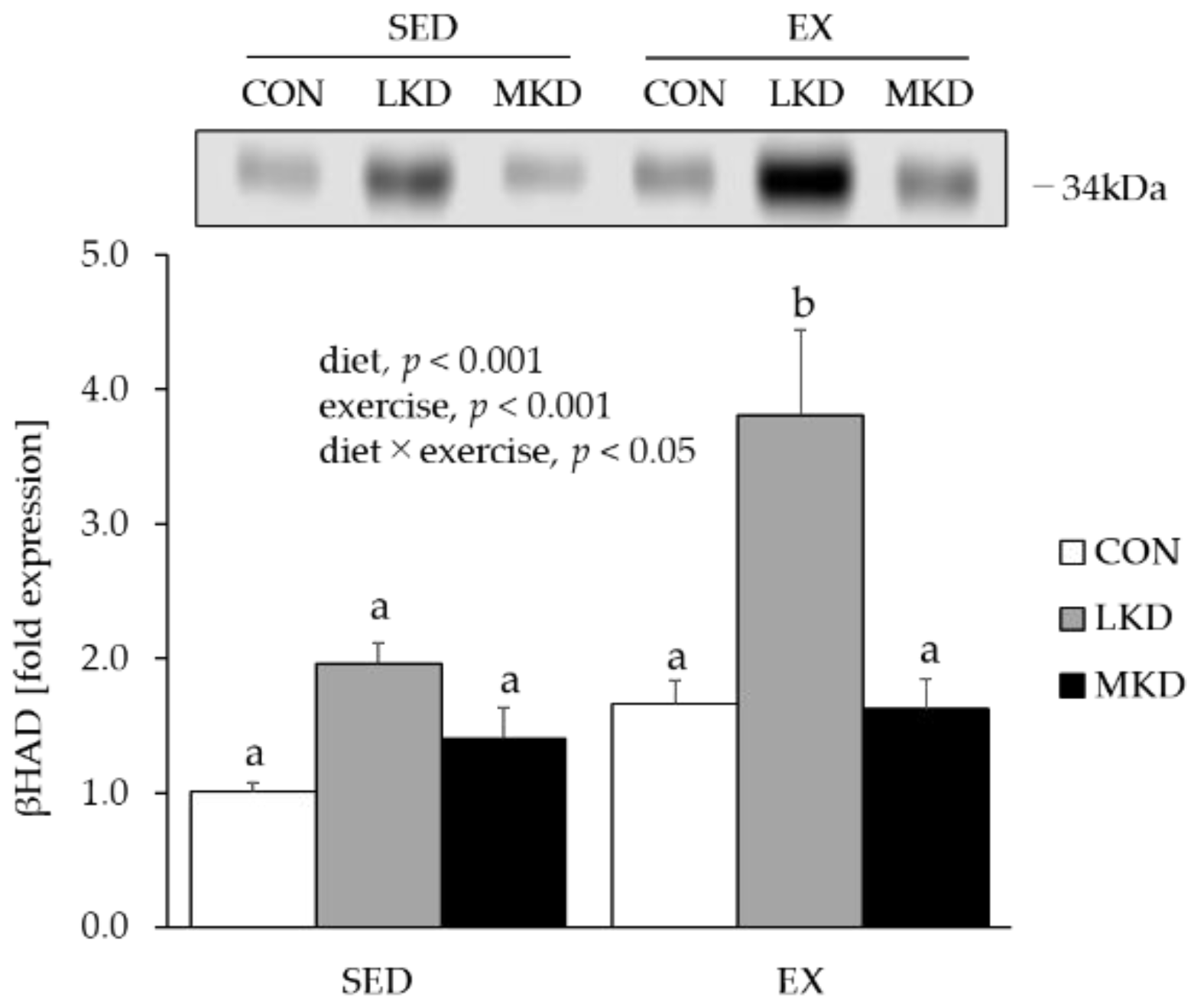
3.5. βHAD Protein Content in Epitrochlearis Muscle Tissue
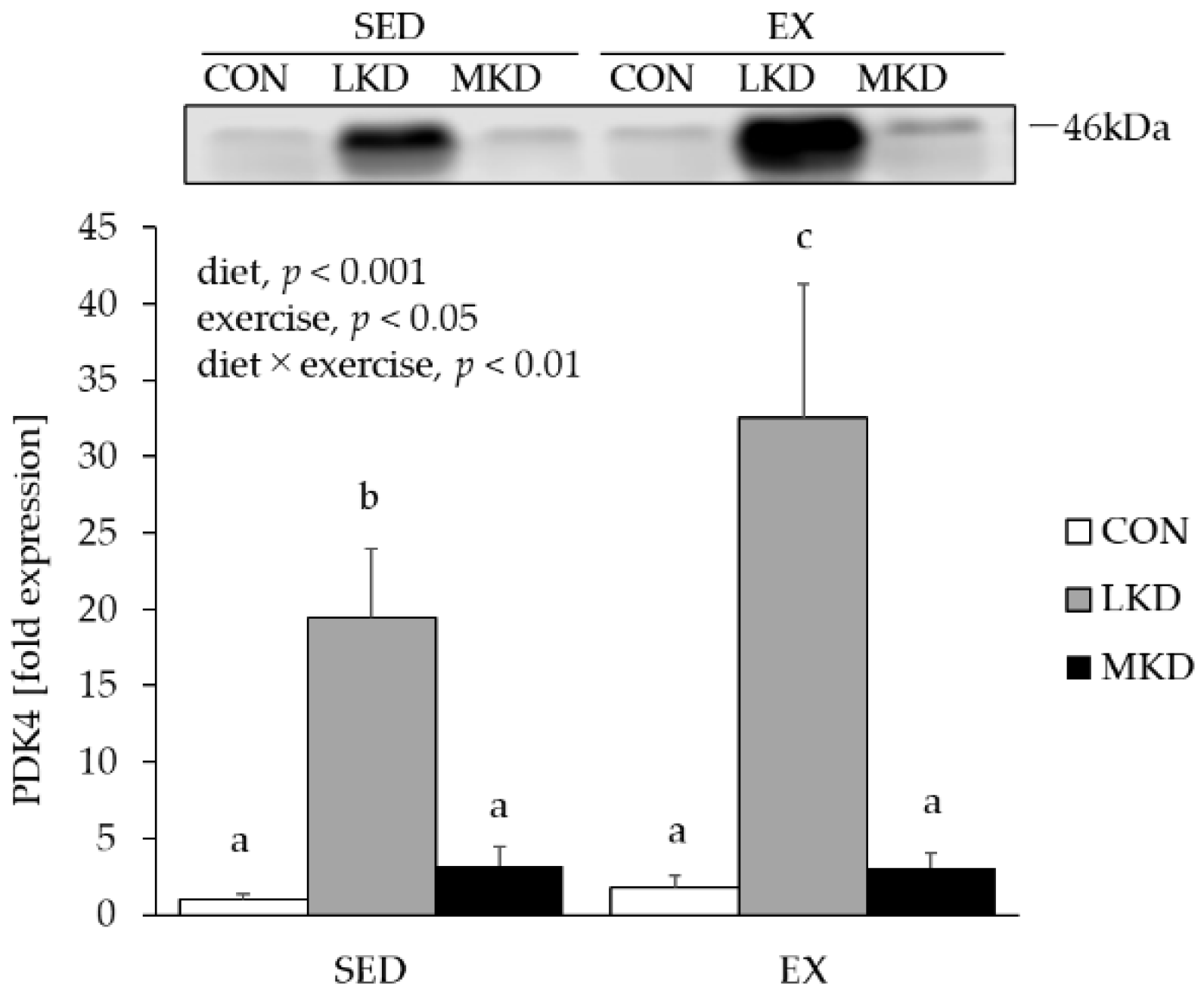
3.6. PDK4 Protein Content in Epitrochlearis Muscle Tissue
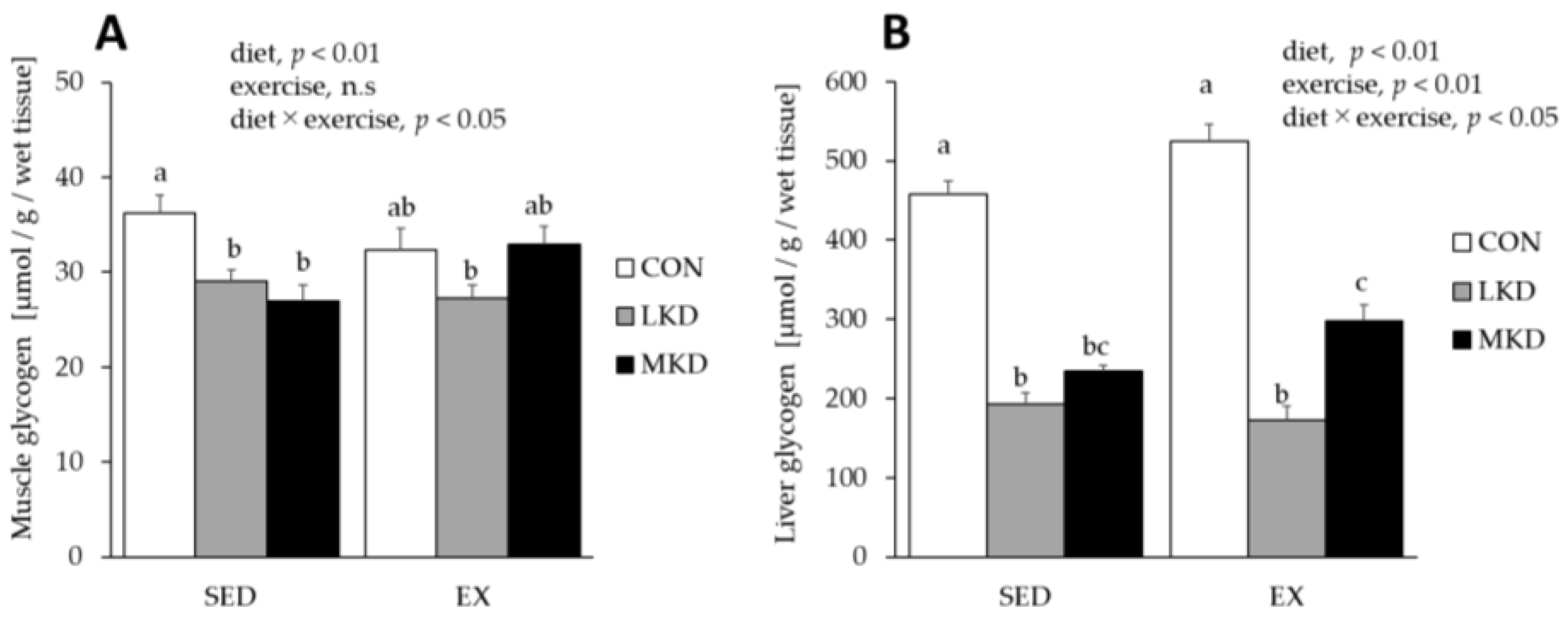
3.7. Glycogen Concentrations in Triceps Muscle and Liver Tissues
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Holloszy, J.O.; Kohrt, W.M.; Hansen, P.A. The regulation of carbohydrate and fat metabolism during and after exercise. Front. Biosci. 1998, 3, D1011–D1027. [Google Scholar] [CrossRef] [PubMed]
- Goedecke, J.H.; Christie, C.; Wilson, G.; Dennis, S.C.; Noakes, T.D.; Hopkins, W.G.; Lambert, E.V. Metabolic adaptations to a high-fat diet in endurance cyclists. Metabolism 1999, 48, 1509–1517. [Google Scholar] [CrossRef]
- Bergman, B.C.; Butterfield, G.E.; Wolfel, E.E.; Casazza, G.A.; Lopaschuk, G.D.; Brooks, G.A. Evaluation of exercise and training on muscle lipid metabolism. Am. J. Physiol. 1999, 276, E106–E117. [Google Scholar] [CrossRef] [PubMed]
- Hearris, M.A.; Hammond, K.M.; Fell, J.M.; Morton, J.P. Regulation of muscle glycogen metabolism during exercise: Implications for endurance performance and training adaptations. Nutrients 2018, 10, 298. [Google Scholar] [CrossRef]
- Kiens, B.; Essen-Gustavsson, B.; Christensen, N.J.; Saltin, B. Skeletal muscle substrate utilization during submaximal exercise in man: Effect of endurance training. J. Physiol. 1993, 469, 459–478. [Google Scholar] [CrossRef]
- Klein, S.; Coyle, E.F.; Wolfe, R.R. Fat metabolism during low-intensity exercise in endurance-trained and untrained men. Am. J. Physiol. 1994, 267, E934–E940. [Google Scholar] [CrossRef]
- Burke, L.M.; Ross, M.L.; Garvican-Lewis, L.A.; Welvaert, M.; Heikura, I.A.; Forbes, S.G.; Mirtschin, J.G.; Cato, L.E.; Strobel, N.; Sharma, A.P.; et al. Low carbohydrate, high fat diet impairs exercise economy and negates the performance benefit from intensified training in elite race walkers. J. Physiol. 2017, 595, 2785–2807. [Google Scholar] [CrossRef]
- Hall, K.D.; Chen, K.Y.; Guo, J.; Lam, Y.Y.; Leibel, R.L.; Mayer, L.E.; Reitman, M.L.; Rosenbaum, M.; Smith, S.R.; Walsh, B.T.; et al. Energy expenditure and body composition changes after an isocaloric ketogenic diet in overweight and obese men. Am. J. Clin. Nutr. 2016, 104, 324–333. [Google Scholar] [CrossRef]
- McSwiney, F.T.; Wardrop, B.; Hyde, P.N.; Lafountain, R.A.; Volek, J.S.; Doyle, L. Keto-adaptation enhances exercise performance and body composition responses to training in endurance athletes. Metabolism 2018, 81, 25–34. [Google Scholar] [CrossRef]
- Badman, M.K.; Kennedy, A.R.; Adams, A.C.; Pissios, P.; Maratos-Flier, E. A very low carbohydrate ketogenic diet improves glucose tolerance in ob/ob mice independently of weight loss. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E1197–E1204. [Google Scholar] [CrossRef] [PubMed]
- Askew, E.W.; Dohm, G.L.; Huston, R.L. Fatty acid and ketone body metabolism in the rat: Response to diet and exercise. J. Nutr. 1975, 105, 1422–1432. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Kashiwaya, Y.; Keon, C.A.; Tsuchiya, N.; King, M.T.; Radda, G.K.; Chance, B.; Clarke, K.; Veech, R.L. Insulin, ketone bodies, and mitochondrial energy transduction. FASEB J. 1995, 9, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.J.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.J.; Stubbs, B.; West, J.; McLure, S.W.; et al. Nutritional ketosis alters fuel preference and thereby endurance performance in athletes. Cell Metab. 2016, 24, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Cotter, D.G.; Schugar, R.C.; Crawford, P.A. Ketone body metabolism and cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H1060–H1076. [Google Scholar] [CrossRef]
- Chang, C.K.; Borer, K.; Lin, P.J. Low-carbohydrate-high-fat diet: Can it help exercise performance? J. Hum. Kinet. 2017, 56, 81–92. [Google Scholar] [CrossRef]
- Volek, J.S.; Freidenreich, D.J.; Saenz, C.; Kunces, L.J.; Creighton, B.C.; Bartley, J.M.; Davitt, P.M.; Munoz, C.X.; Anderson, J.M.; Maresh, C.M.; et al. Metabolic characteristics of keto-adapted ultra-endurance runners. Metabolism 2016, 65, 100–110. [Google Scholar] [CrossRef]
- Vargas, S.; Romance, R.; Petro, J.L.; Bonilla, D.A.; Galancho, I.; Espinar, S.; Kreider, R.B.; Benitez-Porres, J. Efficacy of ketogenic diet on body composition during resistance training in trained men: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2018, 15, 31. [Google Scholar] [CrossRef]
- Rinnankoski-Tuikka, R.; Silvennoinen, M.; Torvinen, S.; Hulmi, J.J.; Lehti, M.; Kivela, R.; Reunanen, H.; Kainulainen, H. Effects of high-fat diet and physical activity on pyruvate dehydrogenase kinase-4 in mouse skeletal muscle. Nutr. Metab. (Lond.) 2012, 9, 53. [Google Scholar] [CrossRef]
- Wende, A.R.; Huss, J.M.; Schaeffer, P.J.; Giguere, V.; Kelly, D.P. PGC-1alpha coactivates PDK4 gene expression via the orphan nuclear receptor ERRalpha: A mechanism for transcriptional control of muscle glucose metabolism. Mol. Cell Biol. 2005, 25, 10684–10694. [Google Scholar] [CrossRef]
- Stellingwerff, T.; Spriet, L.L.; Watt, M.J.; Kimber, N.E.; Hargreaves, M.; Hawley, J.A.; Burke, L.M. Decreased PDH activation and glycogenolysis during exercise following fat adaptation with carbohydrate restoration. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E380–E388. [Google Scholar] [CrossRef]
- Walker, J.L.; Heigenhauser, G.J.; Hultman, E.; Spriet, L.L. Dietary carbohydrate, muscle glycogen content, and endurance performance in well-trained women. J. Appl. Physiol. (1985) 2000, 88, 2151–2158. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.M. Re-Examining high-fat diets for sports performance: Did we call the ‘nail in the coffin’ too soon? Sports Med. 2015, 45 (Suppl. 1), S33–S49. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, T.; Nosaka, N.; Kasai, M. Research on the nutritional characteristics of medium-chain fatty acids. J. Med. Investig. 2007, 54, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Bach, A.C.; Babayan, V.K. Medium-chain triglycerides: An update. Am. J. Clin. Nutr. 1982, 36, 950–962. [Google Scholar] [CrossRef]
- Blazquez, C.; Sanchez, C.; Velasco, G.; Guzman, M. Role of carnitine palmitoyltransferase I in the control of ketogenesis in primary cultures of rat astrocytes. J. Neurochem. 1998, 71, 1597–1606. [Google Scholar] [CrossRef]
- Terada, S.; Tabata, I. Effects of acute bouts of running and swimming exercise on PGC-1alpha protein expression in rat epitrochlearis and soleus muscle. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E208–E216. [Google Scholar] [CrossRef]
- Lowry, O.H.; Passonneau, J.V. A Flexible System of Enzymatic Analysis; Academic Press: Cambridge, MA, USA, 1972; pp. 189–193. [Google Scholar]
- Grabacka, M.; Pierzchalska, M.; Dean, M.; Reiss, K. Regulation of ketone body metabolism and the role of PPARalpha. Int. J. Mol. Sci. 2016, 17, 2093. [Google Scholar] [CrossRef]
- Abdurrachim, D.; Woo, C.C.; Teo, X.Q.; Chan, W.X.; Radda, G.K.; Lee, P.T.H. A new hyperpolarized (13)C ketone body probe reveals an increase in acetoacetate utilization in the diabetic rat heart. Sci. Rep. 2019, 9, 5532. [Google Scholar] [CrossRef]
- Kiens, B. Skeletal muscle lipid metabolism in exercise and insulin resistance. Physiol. Rev. 2006, 86, 205–243. [Google Scholar] [CrossRef]
- Johnstone, A.M.; Horgan, G.W.; Murison, S.D.; Bremner, D.M.; Lobley, G.E. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 2008, 87, 44–55. [Google Scholar] [CrossRef]
- Jornayvaz, F.R.; Jurczak, M.J.; Lee, H.Y.; Birkenfeld, A.L.; Frederick, D.W.; Zhang, D.; Zhang, X.M.; Samuel, V.T.; Shulman, G.I. A high-fat, ketogenic diet causes hepatic insulin resistance in mice, despite increasing energy expenditure and preventing weight gain. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E808–E815. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Bourque, C.; Jones, P.J.; Ross, R.; Parsons, W.E. Medium- versus long-chain triglycerides for 27 days increases fat oxidation and energy expenditure without resulting in changes in body composition in overweight women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 95–102. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Bosarge, A. Weight-loss diet that includes consumption of medium-chain triacylglycerol oil leads to a greater rate of weight and fat mass loss than does olive oil. Am. J. Clin. Nutr. 2008, 87, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Ooyama, K.; Kojima, K.; Aoyama, T.; Takeuchi, H. Decrease of food intake in rats after ingestion of medium-chain triacylglycerol. J. Nutr. Sci. Vitaminol. (Tokyo) 2009, 55, 423–427. [Google Scholar] [CrossRef][Green Version]
- Elamin, M.; Ruskin, D.N.; Masino, S.A.; Sacchetti, P. Ketone-based metabolic therapy: Is increased NAD(+) a primary mechanism? Front. Mol. Neurosci. 2017, 10, 377. [Google Scholar] [CrossRef]
- Veech, R.L. Ketone ester effects on metabolism and transcription. J. Lipid Res. 2014, 55, 2004–2006. [Google Scholar] [CrossRef]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef]
- Garcia-Roves, P.; Huss, J.M.; Han, D.H.; Hancock, C.R.; Iglesias-Gutierrez, E.; Chen, M.; Holloszy, J.O. Raising plasma fatty acid concentration induces increased biogenesis of mitochondria in skeletal muscle. Proc. Natl. Acad. Sci. USA 2007, 104, 10709–10713. [Google Scholar] [CrossRef]
- Holloszy, J.O.; Booth, F.W. Biochemical adaptations to endurance exercise in muscle. Annu. Rev. Physiol. 1976, 38, 273–291. [Google Scholar] [CrossRef]
- Forman, B.M.; Chen, J.; Evans, R.M. Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors alpha and delta. Proc. Natl. Acad. Sci. USA 1997, 94, 4312–4317. [Google Scholar] [CrossRef]
- Simi, B.; Sempore, B.; Mayet, M.H.; Favier, R.J. Additive effects of training and high-fat diet on energy metabolism during exercise. J. Appl. Physiol. (1985) 1991, 71, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Higashida, K.; Kawamura, T.; Higuchi, M. Alternate-day high-fat diet induces an increase in mitochondrial enzyme activities and protein content in rat skeletal muscle. Nutrients 2016, 8, 203. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, Y.; Takagi, T.; Inai, M.; Nishimura, S.; Urashima, S.; Honda, K.; Aoyama, T.; Terada, S. Lauric acid stimulates ketone body production in the KT-5 astrocyte cell line. J. Oleo Sci. 2016, 65, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Challoner, D.R.; Steinberg, D. Oxidative metabolism of myocardium as influenced by fatty acids and epinephrine. Am. J. Physiol. 1966, 211, 97–902. [Google Scholar] [CrossRef]
- Durkalec-Michalski, K.; Nowaczyk, P.M.; Siedzik, K. Effect of a four-week ketogenic diet on exercise metabolism in CrossFit-trained athletes. J. Int. Soc. Sports Nutr. 2019, 16, 16. [Google Scholar] [CrossRef]
- Peters, S.J.; Harris, R.A.; Wu, P.; Pehleman, T.L.; Heigenhauser, G.J.; Spriet, L.L. Human skeletal muscle PDH kinase activity and isoform expression during a 3-day high-fat/low-carbohydrate diet. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E1151–E1158. [Google Scholar] [CrossRef]
- Fan, W.; Waizenegger, W.; Lin, C.S.; Sorrentino, V.; He, M.X.; Wall, C.E.; Li, H.; Liddle, C.; Yu, R.T.; Atkins, A.R.; et al. PPARdelta promotes running endurance by preserving glucose. Cell Metab. 2017, 25, 1186–1193.e4. [Google Scholar] [CrossRef]
- St Amand, T.A.; Spriet, L.L.; Jones, N.L.; Heigenhauser, G.J. Pyruvate overrides inhibition of PDH during exercise after a low-carbohydrate diet. Am. J. Physiol. Endocrinol. Metab. 2000, 279, E275–E283. [Google Scholar] [CrossRef]
- Takahashi, Y.; Matsunaga, Y.; Tamura, Y.; Terada, S.; Hatta, H. Pre-exercise high-fat diet for 3 days affects post-exercise skeletal muscle glycogen repletion. J. Nutr. Sci. Vitaminol. (Tokyo) 2017, 63, 323–330. [Google Scholar] [CrossRef]
| Ingredient | (g/kg Diet) | ||
|---|---|---|---|
| CON | LKD | MKD | |
| Cornstarch | 520.80 | − | 189.00 |
| Casein (>85% protein) | 206.60 | 174.00 | 174.00 |
| Sucrose | 101.80 | − | − |
| LCT | 71.30 | 539.00 | 35.10 |
| MCT | − | − | 315.25 |
| Cellulose fiber | 51.00 | 238.00 | 238.00 |
| Mineral mix (AIN-93-M-MX) | 35.00 | 35.00 | 35.00 |
| Vitamin mix (AIN-93-VX) | 10.00 | 10.00 | 10.00 |
| l-Cystine | 2.50 | 2.50 | 2.50 |
| Choline bitartrate (41.1% choline) | 1.80 | 1.80 | 1.80 |
| tert-Butylhydroquinone | 0.08 | 0.08 | 0.08 |
| Energy density (kcal/g) | 4.00 | 5.60 | 4.50 |
| Protein:Fat:Carbohydrate Ratio (% total energy) | 21:16:63 | 12:87:1 | 16:66:18 |
| Fatty Acid | (g/100 g Fatty Acids) | |
|---|---|---|
| CON and LKD | MKD | |
| C8:0 * | ND | 64.7 |
| C10:0 | ND | 23.7 |
| C16:0 | 10.6 | 1.2 |
| C18:0 | 4.0 | 0.5 |
| C18:1 | 24.9 | 2.9 |
| C18:2 | 52.1 | 6.1 |
| C18:3 | 6.7 | 0.8 |
| C20:0 | 0.4 | ND |
| C20:1 | 0.2 | ND |
| C22:0 | 0.4 | ND |
| C24:0 | 0.2 | ND |
| Others | 0.6 | ND |
| p-Values | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CON | LKD | MKD | Diet | Exercise | Interaction | ||||||||
| Initial body weight (g) | SED | 274 | ± | 7 | 274 | ± | 4 | 274 | ± | 4 | n.s. | n.s. | n.s. |
| EX | 273 | ± | 5 | 273 | ± | 4 | 273 | ± | 3 | ||||
| Final body weight (g) | SED | 518 | ± | 21 | 478 | ± | 16 | 432 | ± | 8 | p < 0.001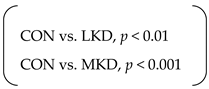 | p < 0.001 | n.s. |
| EX | 427 | ± | 10 | 367 | ± | 15 | 380 | ± | 5 | ||||
| Total energy intake (kcal) | SED | 6754 | ± | 269 | 6863 | ± | 256 | 6151 | ± | 126 | p < 0.01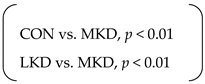 | p < 0.001 | n.s. |
| EX | 5969 | ± | 133 | 5714 | ± | 226 | 5135 | ± | 42 | ||||
| Intra-abdominal fat weight (g) | SED | 40 | ± | 3 | 36 | ± | 2 | 24 | ± | 2 | p < 0.001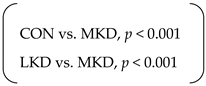 | p < 0.001 | n.s. |
| EX | 21 | ± | 1 | 18 | ± | 1 | 13 | ± | 1 | ||||
| Food efficiency (Δg/1000 kcal) * | SED | 36 | ± | 1 a | 30 | ± | 1 b | 26 | ± | 1 c | p < 0.001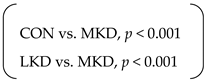 | p < 0.001 | p < 0.001 |
| EX | 26 | ± | 1 c | 16 | ± | 1 d | 21 | ± | 1 e | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukazawa, A.; Koike, A.; Karasawa, T.; Tsutsui, M.; Kondo, S.; Terada, S. Effects of a Ketogenic Diet Containing Medium-Chain Triglycerides and Endurance Training on Metabolic Enzyme Adaptations in Rat Skeletal Muscle. Nutrients 2020, 12, 1269. https://doi.org/10.3390/nu12051269
Fukazawa A, Koike A, Karasawa T, Tsutsui M, Kondo S, Terada S. Effects of a Ketogenic Diet Containing Medium-Chain Triglycerides and Endurance Training on Metabolic Enzyme Adaptations in Rat Skeletal Muscle. Nutrients. 2020; 12(5):1269. https://doi.org/10.3390/nu12051269
Chicago/Turabian StyleFukazawa, Ayumi, Atsuko Koike, Takuya Karasawa, Momoko Tsutsui, Saki Kondo, and Shin Terada. 2020. "Effects of a Ketogenic Diet Containing Medium-Chain Triglycerides and Endurance Training on Metabolic Enzyme Adaptations in Rat Skeletal Muscle" Nutrients 12, no. 5: 1269. https://doi.org/10.3390/nu12051269
APA StyleFukazawa, A., Koike, A., Karasawa, T., Tsutsui, M., Kondo, S., & Terada, S. (2020). Effects of a Ketogenic Diet Containing Medium-Chain Triglycerides and Endurance Training on Metabolic Enzyme Adaptations in Rat Skeletal Muscle. Nutrients, 12(5), 1269. https://doi.org/10.3390/nu12051269




