Large Variability of Iodine Content in Retail Cow’s Milk in the U.S.
Abstract
1. Introduction
2. Materials and Methods
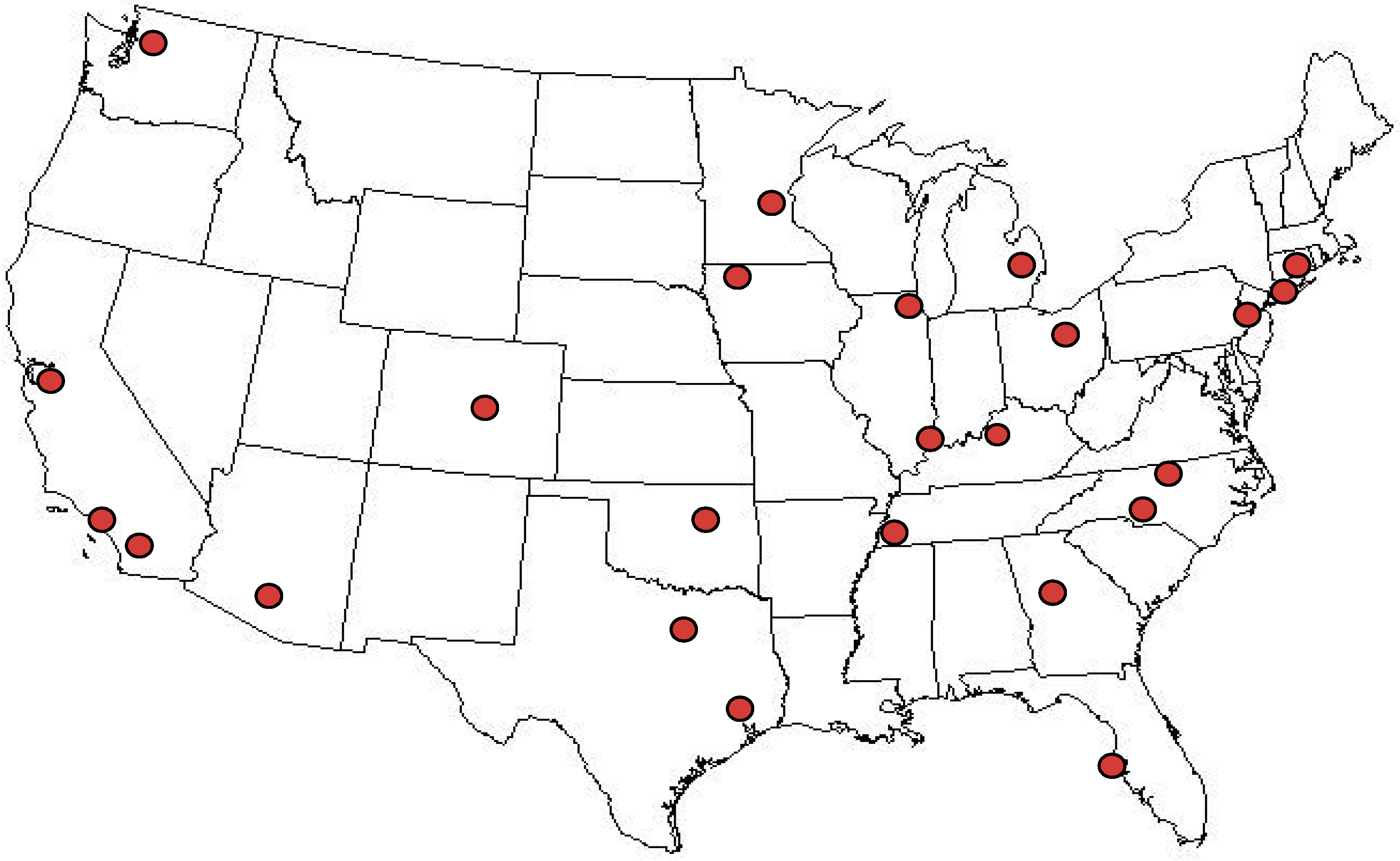
2.1. Sampling Plan
2.2. Sample Procurement
2.3. Sample Preparation
2.4. Iodine Analysis
2.5. Quality Control
2.6. Data Analysis
3. Results
3.1. Sample Descriptive Information
3.2. Quality Control
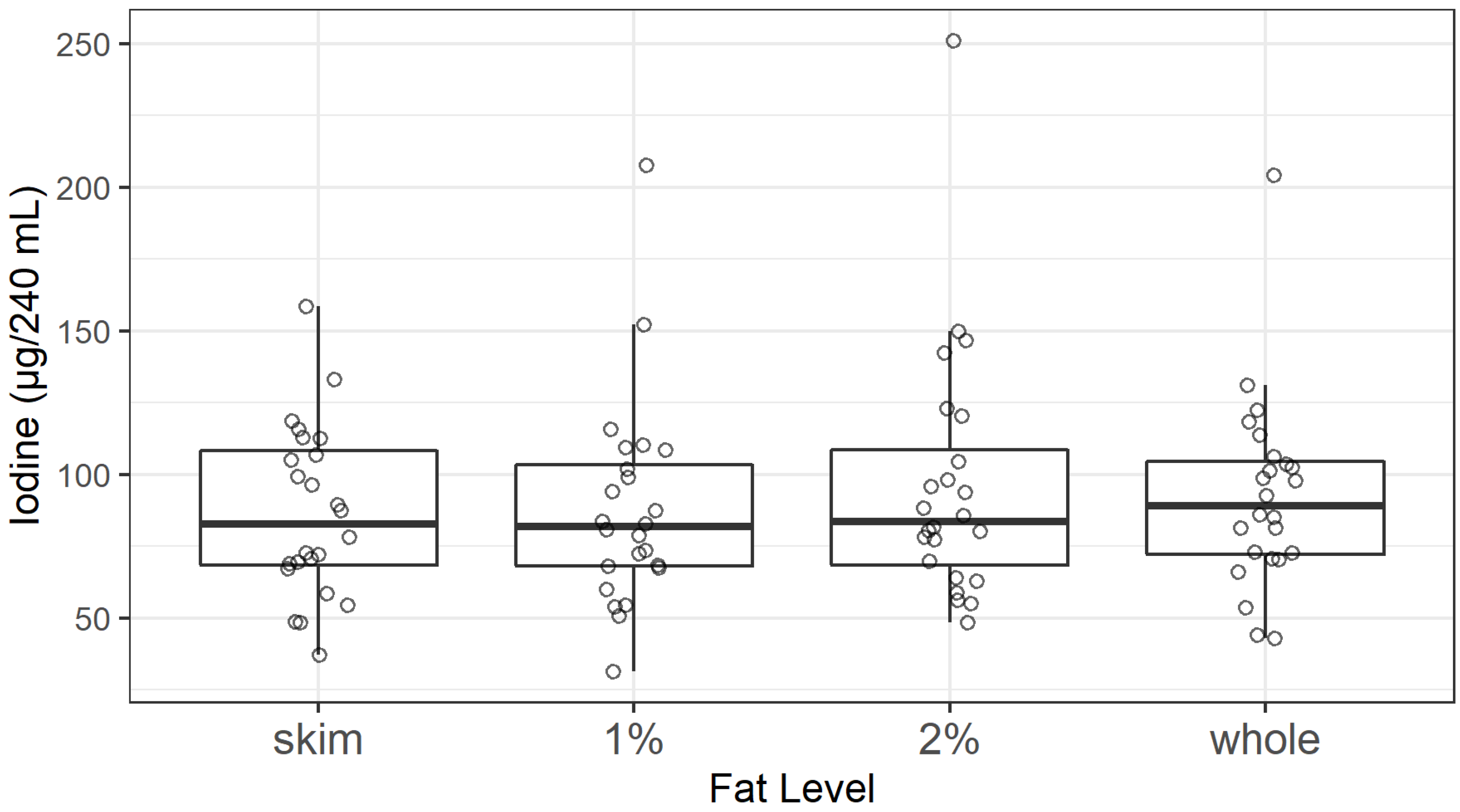
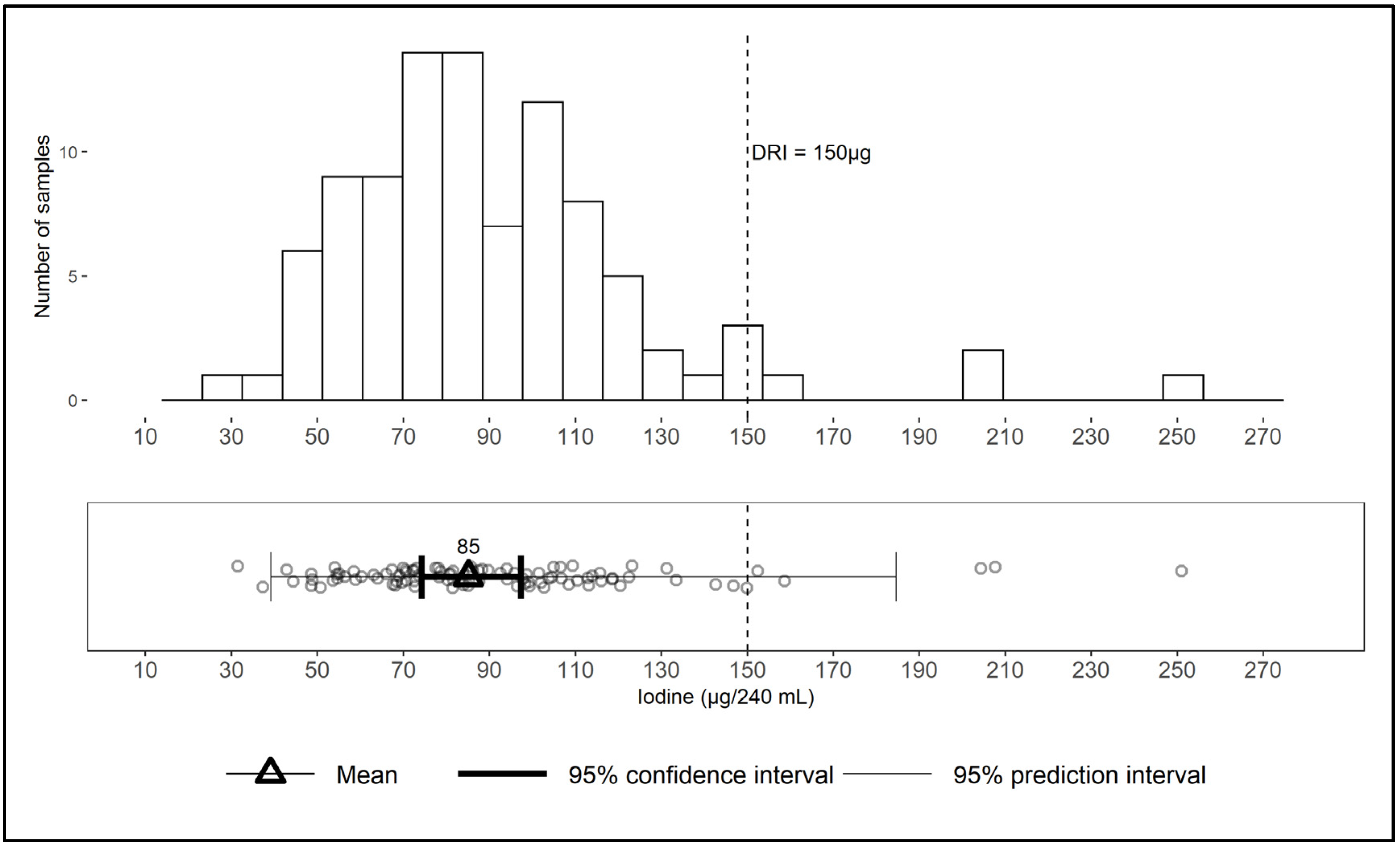
3.3. Iodine Content of Retail Milk
4. Discussion
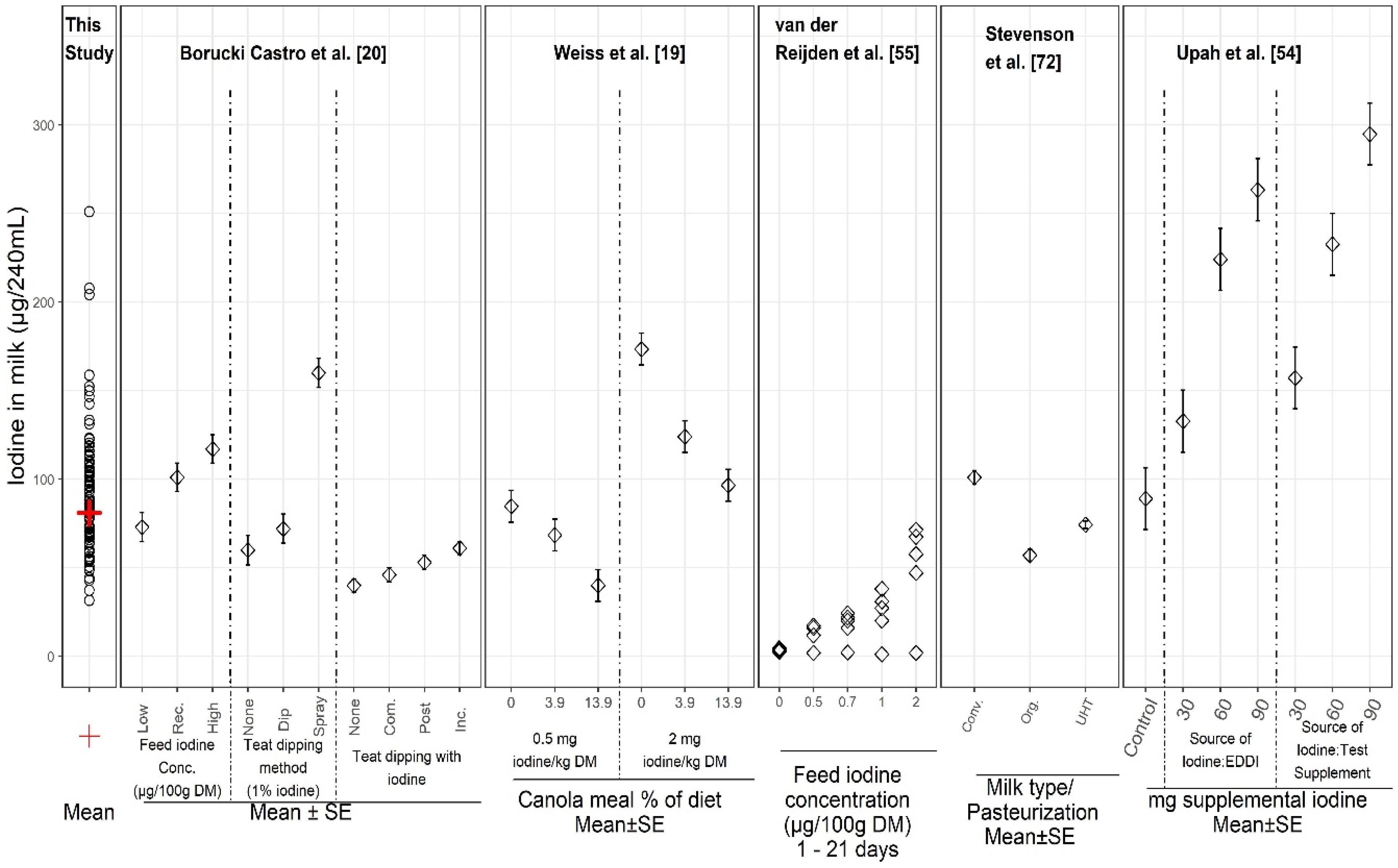
4.1. Other Reports on Iodine in Milk
4.2. Factors Influencing Iodine Levels in Milk
4.3. Importance of Quality Control and Reference Materials
4.4. Implications for Nutrition Research and Use of Food Composition Data
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rohner, F.; Zimmermann, M.; Jooste, P.; Pandav, C.; Caldwell, K.; Raghavan, R.; Raiten, D.J. Biomarkers of nutrition for development—iodine review. J. Nutr. 2014, 144, 1322S–1342S. [Google Scholar] [CrossRef] [PubMed]
- Swanson, C.A.; Zimmermann, M.B.; Skeaff, S.; Pearce, E.N.; Dwyer, J.T.; Trumbo, P.R.; Zehaluk, C.; Andrews, K.W.; Carriquiry, A.; Caldwell, K.L. Summary of an NIH workshop to identify research needs to improve the monitoring of iodine status in the United States and to inform the DRI. J. Nutr. 2012, 142, 1175s–1185s. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.N. Is iodine deficiency reemerging in the United States? AACE Clin. Case Rep. 2015, 1, e81–e82. [Google Scholar] [CrossRef]
- Lee, K.W.; Shin, D.; Cho, M.S.; Song, W.O. Food group intakes as determinants of iodine status among US adult population. Nutrients 2016, 8, 325. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Panel on Micronutrients. In Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Luo, Y.; Kawashima, A.; Ishido, Y.; Yoshihara, A.; Oda, K.; Hiroi, N.; Ito, T.; Ishii, N.; Suzuki, K. Iodine excess as an environmental risk factor for autoimmune thyroid disease. Int. J. Mol. Sci. 2014, 15, 12895–12912. [Google Scholar] [CrossRef] [PubMed]
- Ershow, A.G.; Goodman, G.; Coates, P.M.; Swanson, C.A. Research needs for assessing iodine intake, iodine status, and the effects of maternal iodine supplementation. Am. J. Clin. Nutr. 2016, 104, 941S–949S. [Google Scholar] [CrossRef]
- Pehrsson, P.R.; Patterson, K.Y.; Spungen, J.H.; Wirtz, M.S.; Andrews, K.W.; Dwyer, J.T.; Swanson, C.A. Iodine in food- and dietary supplement–composition databases. Am. J. Clin. Nutr. 2016, 104, 868S–876S. [Google Scholar] [CrossRef]
- Dasgupta, P.K.; Lui, Y.; Dyke, J.V. Iodine nutrition: Iodine content of iodized salt in the United States. Environ. Sci. Technol. 2008, 42, 1315–1323. [Google Scholar] [CrossRef]
- Winger, R.J.; Konig, J.; House, D.A. Technological issues associated with iodine fortification of foods. Trends Food Sci. Technol. 2008, 19, 94–101. [Google Scholar] [CrossRef]
- Perrine, C.G.; Herrick, K.; Serdula, M.K.; Sullivan, K.M. Some subgroups of reproductive age women in the United States may be at risk for iodine deficiency. J. Nutr. 2010, 140, 1489–1494. [Google Scholar] [CrossRef]
- Maalouf, J.; Barron, J.; Gunn, J.P.; Yuan, K.; Perrine, C.G.; Cogswell, M.E. Iodized salt sales in the United States. Nutrients 2015, 7, 1691–1695. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture (USDA), Agricultural Research Service. FoodData Central. 2019. Available online: https://fdc.nal.usda.gov/ (accessed on 11 June 2019).
- Ershow, A.G.; Skeaff, S.A.; Merkel, J.M.; Pehrsson, P.R. Development of databases on iodine in foods and dietary supplements. Nutrients 2018, 10, 100. [Google Scholar] [CrossRef] [PubMed]
- Abt, E.; Spungen, J.; Pouillot, R.; Gamalo-Siebers, M.; Wirtz, M. Update on dietary intake of perchlorate and iodine from U.S. Food and Drug Administration’s Total Diet Study: 2008–2012. J. Expo. Sci. Environ. Epidemiol. 2018, 28, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E. National trends in iodine nutrition: Are we getting enough? Thyroid 2007, 17, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.W.; Egan, S.K.; Kim, H.; Beru, N.; Bolger, P.M. US Food and Drug Administration’s Total Diet Study: Dietary perchlorate and iodine. J. Expo. Sci. Environ. Epidemiol. 2008, 18, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Borucki Castro, S.I.; Lacasse, P.; Fouquet, A.; Beraldin, F.; Robichaud, A.; Berthiaume, R. Feed iodine concentrations on farms with contrasting levels of iodine in milk. J. Dairy Sci. 2011, 94, 4684–4689. [Google Scholar] [CrossRef]
- Weiss, W.P.; Wyatt, D.J.; Kleinschmit, D.H.; Socha, M.T. Effect of including canola meal and supplemental iodine in diets of dairy cows on short-term changes in iodine concentrations in milk. J. Dairy Sci. 2015, 98, 4841–4849. [Google Scholar] [CrossRef]
- Borucki Castro, S.I.; Berthiaume, R.; Robichaud, A.; Lacasse, P. Effects of iodine intake and teat-dipping practices on milk iodine concentrations in dairy cows. J. Dairy Sci. 2012, 95, 213–220. [Google Scholar] [CrossRef]
- Franke, K.; Meyer, U.; Wagner, H.; Flachowsky, G. Influence of various iodine supplementation levels and two different iodine species on the iodine content of the milk of cows fed rapeseed meal or distillers dried grains with solubles as the protein source. J. Dairy Sci. 2009, 92, 4514–4523. [Google Scholar] [CrossRef]
- Flachowsky, G.; Franke, K.; Meyer, U.; Leiterer, M.; Schone, F. Influencing factors on iodine content of cow milk. Eur. J. Nutr. 2014, 53, 351–365. [Google Scholar] [CrossRef]
- Van der Reijden, O.L.; Zimmermann, M.B.; Galetti, V. Iodine in dairy milk: Sources, concentrations and importance to human health. Best Pract. Res. Clin. Endocrinol. Metab. 2017, 31, 385–395. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture (USDA), Agricultural Marketing Service. Estimated Fluid Milk Products Sales Report. EFMS-0417; 2017. Available online: https://www.ams.usda.gov/sites/default/files/media/Estimated_Fluid_Products_Milk_Sales.pdf (accessed on 10 September 2018).
- Pehrsson, P.R.; Perry, C.; Daniel, M. ARS, USDA updates food sampling strategies to keep pace with demographic shifts. Procedia Food Sci. 2013, 2, 52–59. [Google Scholar] [CrossRef]
- Trainer, D.; Pehrsson, P.; Haytowitz, D.; Holden, J.M.; Phillips, K.M.; Rasor, A.S.; Conley, N.A. Development of sample handling procedures for foods under USDA’s National Food and Nutrient Analysis Program. J. Food Compost. Anal. 2010, 23, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Patterson, K.Y.; Phillips, K.M.; Horst, R.L.; Byrdwell, W.C.; Exler, J.; Lemar, L.E.; Holden, J.M. Vitamin D content and variability in fluid milks from a US Department of Agriculture nationwide sampling to update values in the National Nutrient Database for Standard Reference. J. Dairy Sci. 2010, 93, 5082–5090. [Google Scholar] [CrossRef] [PubMed]
- Haytowitz, D.B.; Pehrsson, P.R. USDA’s National Food and Nutrient Analysis Program (NFNAP) produces high-quality data for USDA foods composition databases: Two decades of collaboration. Food Chem. 2018, 238, 134–135. [Google Scholar] [CrossRef] [PubMed]
- AOAC International. Official Methods of Analysis, 20th ed.; AOAC International: Gaithersburg, MD, USA, 2016. [Google Scholar]
- Phillips, K.M.; Patterson, K.Y.; Rasor, A.S.; Exler, J.; Haytowitz, D.B.; Holden, J.M.; Pehrsson, P.R. Quality control materials in the USDA National Food and Nutrient Analysis Program (NFNAP). Anal. Bioanal. Chem. 2006, 384, 1341–1355. [Google Scholar] [CrossRef]
- Phillips, K.M.; Byrdwell, W.C.; Exler, J.; Harnly, J.M.; Holden, J.M.; Holick, M.F.; Hollis, B.W.; Horst, R.L.; Lemar, L.E.; Patterson, K.Y.; et al. Development and validation of control materials for the measurement of vitamin D3 in selected US foods. J. Food Compost. Anal. 2008, 21, 527–534. [Google Scholar] [CrossRef]
- U.S. Department of Commerce (USDC); National Institute of Standards & Technology (NIST). Certificate of Analysis, Standard Reference Material 1849a, Infant/Adult Nutritional Formula 1. 2018. Available online: https://www-s.nist.gov/srmors/certificates/1849a.pdf (accessed on 5 August 2019).
- Horwitz, W.; Albert, R. The Horwitz ratio (HorRat): A useful index of method performance with respect to precision. J. AOAC Int. 2006, 89, 1095–1109. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008; Available online: https://www.R-project.org/ (accessed on 12 June 2019).
- U.S. Department of Health and Human Services (USHHS); U.S. Food and Drug Administration (FDA); Center for Food Safety and Applied Nutrition. Food Labeling: Serving Sizes of Foods that can Reasonably be Consumed at One Eating Occasion; Dual-Column Labeling; Updating, Modifying, and Establishing Certain Reference Amounts Customarily Consumed; Serving Size for Breath Mints; and Technical Amendments: Guidance for Industry Small Entity Compliance Guide. Page 26; 2018. Available online: https://www.fda.gov/media/111144/download (accessed on 24 September 2019).
- Bates, D.; Machler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Ishiguro, M.; Kitagawa, G. Akaike Information Criterion Statistics; Springer Publishing Company: New York, NY, USA, 1986; ISBN 978-90-277-2253-9. [Google Scholar]
- Harville, D.A. Bayesian inference for variance components using only error contrasts. Biometrika 1974, 61, 383–385. [Google Scholar] [CrossRef]
- Cook, R.D.; Weisberg, S. Residuals and Influence in Regression; Chapman and Hall: London, UK, 1982. [Google Scholar]
- Lenth, R. Emmeans: Estimated Marginal Means, aka Least-Squares Means, R package version 1.3.4.; Vienna, Austria; 2019; Available online: https://CRAN.R-project.org/package=emmeans (accessed on 1 August 2019).
- U.S. Department of Health and Human Services (USHHS); U.S. Food and Drug Administration (FDA). Interstate Milk Shippers List. April 2017. Available online: https://www.fda.gov/media/104934/download (accessed on 22 March 2019).
- Komorowski, E.S. Saturated Fat Reduction in Milk and Dairy Products in “Reducing Saturated Fats in Foods”; Talbot, G., Ed.; Woodhead Publishing Ltd.: Sawston, Cambridge, UK, 2011; pp. 179–194. ISBN 978-1-84569-740-2. [Google Scholar] [CrossRef]
- Mohan, M.S.; Hopkinson, J.; Harte, F. Milk and Ice Cream Processing. In Food Processing: Principles and Applications; Clark, S., Jung, S., Lamsal, B., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 387–391. [Google Scholar]
- U.S. Department of Health and Human Services (USHHS); U.S. Food and Drug Administration (FDA). Total Diet Study statistics on element results—2006–2011. Version current 27 March 2014 [cited 15 July 2014]; 2014. Available online: http://www.fda.gov/downloads/Food/FoodScienceResearch/TotalDietStudy/UCM184301.pdf (accessed on 7 August 2019).
- Pennington, J.A.T. Iodine concentrations in US milk: Variation due to time, season, and region. J. Dairy Sci. 1990, 73, 3421–3427. [Google Scholar] [CrossRef]
- Pearce, E.; Pino, S.; He, X.; Bazrafshan, H.R.; Lee, S.L.; Braverman, L.E. Sources of dietary iodine: Bread, cows’ milk, and infant formula in the Boston area. J. Clin. Endocrinol. Metab. 2004, 89, 3421–3424. [Google Scholar] [CrossRef] [PubMed]
- Kirk, A.B.; Martinelango, P.K.; Tian, K.; Dutta, A.; Smith, E.E.; Dasgupta, P.K. Perchlorate and iodine in dairy and breast milk. Environ. Sci. Technol. 2005, 39, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Frida Food Data. Version 2. National Food Institute, Technical University of Denmark, 2016. Available online: http://frida.fooddata.dk (accessed on 26 March 2019).
- U.S. Department of Health and Human Services (USHHS); Public Health Services, Food and Drug Administration. Grade “A” Pasteurized Milk Ordinance, p. 51 and 211. 2017. Available online: http://ncims.org/wp-content/uploads/2018/06/2017-PMO-FINAL.pdf (accessed on 22 March 2019).
- Rasmussen, L.B.; Carle, A.; Jorgensen, T.; Knuthsen, P.; Krejbjerg, A.; Perrild, H.; Bjergved, L.; Sloth, J.J.; Laurberg, P.; Ovesen, L. Iodine excretion has decreased in Denmark between 2004 and 2010—The importance of iodine content in milk. Br. J. Nutr. 2014, 112, 1993–2001. [Google Scholar] [CrossRef] [PubMed]
- Payling, L.M.; Juniper, D.T.; Drake, C.; Rymer, C.; Givens, D.I. Effect of milk type and processing on iodine concentration of organic and conventional winter milk at retail: Implications for nutrition. Food Chem. 2015, 178, 327–330. [Google Scholar] [CrossRef] [PubMed]
- O’Kane, S.M.; Pourshahidi, L.K.; Mulhern, M.S.; Weir, R.R.; Hill, S.; O’Reilly, J.; Kmiotek, D.; Deitrich, C.; Mackle, E.M.; Fitzgerald, E.; et al. The effect of processing and seasonality on the iodine and selenium concentration of cow’s milk produced in northern Ireland (NI): Implications for population dietary intake. Nutrients 2018, 10, 287. [Google Scholar] [CrossRef] [PubMed]
- Soriguer, F.; Gutierrez-Repiso, C.; Gonzalez-Romero, S.; Olveira, G.; Garriga, M.J.; Velasco, I.; Santiago, P.; de Escobar, G.M.; Garcia-Fuentes, E. Iodine concentration in cow’s milk and its relation with urinary iodine concentrations in the population. Clin. Nutr. ESPEN 2011, 30, 44–48. [Google Scholar] [CrossRef]
- Upah, N.; Pearce, S.; Baumgard, L. Effects of Iodine and Dose in Lactating Dairy Cows; Animal Industry Report: AS 657, ASL R2602; Iowa State University: Ames, IA, USA, 2011. [Google Scholar] [CrossRef]
- Van der Reijden, O.L.; Galetti, V.; Herter-Aeberli, I.; Zimmermann, M.B.; Zeder, C.; Krzystek, A.; Haldimann, M.; Barmaz, A.; Kreuzer, M.; Berard, J.; et al. Effects of feed iodine concentrations and milk processing on iodine concentrations of cows’ milk and dairy products, and potential impact on iodine intake in Swiss adults. Brit. J. Nutr. 2019, 122, 172–185. [Google Scholar] [CrossRef]
- Fuge, R.; Johnson, C.C. Iodine and human health, the role of environmental geochemistry and diet, a review. Appl. Geochem. 2015, 63, 282–302. [Google Scholar] [CrossRef]
- Cardenas, Q.H.; Gomez, B.C.; Pretell, E.A. Iodine content of cattle milk from mountain and coast from Peru. Arch. Latinoam. Nutr. 2003, 53, 409–412. [Google Scholar]
- Paulikova, I.; Seidel, H.; Nagy, O.; Kovac, G. Milk iodine content in Slovakia. Acta Vet. Brno 2008, 77, 533–538. [Google Scholar] [CrossRef]
- Suttle, N. Mineral Nutrition of Livestock, 4th ed.; Chp 12. Iodine; CAB International: Cambridge, MA, USA, 2010; p. 310. [Google Scholar] [CrossRef]
- Leung, A.M.; Braverman, L.E.; Pearce, E.N. History of U.S. iodine fortification and supplementation. Nutrients 2012, 4, 1740–1746. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Nutrient Requirements of Dairy Cattle, 7th ed.; The National Academies Press: Washington, DC, USA, 2001; p. 138. ISBN 978-0-309-06997-7. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services (USHHS); Food and Drug Administration (FDA). Compliance Policy Guide Section 651.100 Ethylenediamine Dihydroiodide (EDDI). 2019. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/cpg-sec-651100-ethylenediamine-dihydroiodide-eddi (accessed on 7 August 2019).
- European Food Safety Authority (EFSA). Panel on additives and products or substances using in animal feeds. Scientific opinion on the safety and efficacy of iodine compounds (E2) as feed additives for all animal species: Calcium iodate anhydrous and potassium iodide, based on a dossier submitted by Ajay Europe SARL. EFSA J. 2013, 11, 3099–3133. [Google Scholar]
- Dahl, L.; Opsahl, J.A.; Meltzer, H.M.; Julshamn, K. Iodine concentration in Norwegian milk and dairy products. Brit. J. Nutr. 2003, 90, 679–685. [Google Scholar] [CrossRef]
- Troan, G.; Dahl, L.; Meltzer, H.M.; Abel, M.H.; Indahl, U.G.; Haug, A.; Prestløkken, E. A model to secure a stable iodine concentration in milk. Food Nutr. Res. 2015, 59, e29829. [Google Scholar] [CrossRef]
- O’Brien, B.; Gleeson, D.; Jordan, K. Iodine concentrations in milk. Irish J. Agric. Food Res. 2013, 52, 209–216. [Google Scholar]
- U.S. Department of Agriculture (USDA); APHIS National Animal Health Monitoring System. Dairy 2014, Milk Quality, Milking Procedures, and Mastitis on U.S. Dairies. USDA-APHIS-VS-CEAH-NAHMS. Fort Collins, CO #704.0916, Pages ii, 36–38; 2016. Available online: https://www.aphis.usda.gov/animal_health/nahms/dairy/downloads/dairy14/Dairy14_dr_Mastitis.pdf (accessed on 24 October 2017).
- U.S. Environmental Protection Agency (EPA). Reregistration Eligibility Decision for Iodine and Iodophor Complexes. 739-R-06-010. Pages 19, 29, and 33; 2006. Available online: https://archive.epa.gov/pesticides/reregistration/web/pdf/iodine-red.pdf (accessed on 24 September 2019).
- Mateus, C.; (DeValal Inc., Kansas City, MO, USA). Personal communication, 2019.
- Norouzian, M.A. Iodine in raw and pasteurized milk of dairy cows fed different amounts of potassium iodide. Biol. Trace Elem. Res. 2011, 139, 160–167. [Google Scholar] [CrossRef]
- Nazeri, P.; Norouzian, M.A.; Mirmiran, P.; Hedayati, M.; Azizi, F. Heating process in pasteurization and not in sterilization decreases the iodine concentration of milk. Int. J. Endocrinol. Metab. 2015, 13, e27995. [Google Scholar] [CrossRef]
- Stevenson, M.C.; Drake, C.; Givens, D.I. Further studies on the iodine concentration of conventional, organic, and UHT semi-skimmed milk at retail in the UK. Food Chem. 2018, 551–555. [Google Scholar] [CrossRef]
- Phillips, K.M.; Rasor, A.S. A mixed mushroom control material to facilitate inter-laboratory harmonization of mushroom composition analyses. J. Food Compost. Anal. 2016, 48, 48–66. [Google Scholar] [CrossRef]
- Todorov, T.I.; Gray, P.J. Analysis of iodine in food samples by inductively coupled plasma-mass spectrometry. Food Addit. Contam. Part A 2016, 33, 282–290. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bowman, S.A.; Martin, C.L.; Carlson, J.L.; Clemens, J.C.; Lin, B.-H.; Moshfegh, A.J. Retail food commodity intakes: Mean amounts of retail commodities per individual, 2007–2008. Issued 2013. U.S. Department of Agriculture, Agricultural Research Service and U.S. Department of Agriculture, Economic Research Service. Available online: https://data.nal.usda.gov/dataset/food-intakes-converted-retail-commodities-databases-ficrcd (accessed on 11 December 2019).
- U.S. Department of Health and Human Services (USHHS); U.S. Department of Agriculture (USDA). 2015–2020 Dietary Guidelines for Americans, 8th ed.; HHS Publication #HHS-ODPHP-2015-2020-01-DGA-A; USDA Publication #Home and Garden Bulletin No. 232; U.S. Government Printing Office: Washington, DC, USA, 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 10 September 2018).
- Haug, A.; Hostmark, A.T.; Harstad, O.M. Bovine milk in human nutrition—A review. Lipids Health Dis. 2007, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Carriquiry, A.L.; Spungen, J.H.; Murphy, S.P.; Pehrsson, P.R.; Dwyer, J.T.; Juan, W.; Wirtz, M.S. Variation in the iodine concentrations of foods: Considerations for dietary assessment. Am. J. Clin. Nutr. 2016, 104, 877S–887S. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.M.; Braverman, L.E. Consequences of excess iodine. Nat. Rev. Endocrinol. 2014, 10, 136–142. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture (USDA), Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Legacy. 2018. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 23 September 2019).
- Food Standards Australia New Zealand. Food Standards Code—Standard 2.1.1—Cereals and Cereal Products. Available online: https://www.legislation.gov.au/Series/F2015L00420 (accessed on 19 April 2020).
- U.S. Department of Health and Human Services (USHHS); Public Health Service, Food and Drug Administration (FDA); National Conference on Interstate Milk Shipments. Procedures Governing the Cooperative State-Public Health Service/Food and Drug Administration Program of the National Conference on Interstate Milk Shipments (Revision). 2017. Available online: https://www.fda.gov/media/111155/download (accessed on 7 August 2019).
- Moschini, M.; Battaglia, M.; Beone, G.M.; Piva, G.; Masoero, F. Iodine and selenium carry over in milk and cheese in dairy cows: Effect of diet supplementation and milk yield. Animal 2010, 4, 147–155. [Google Scholar] [CrossRef] [PubMed]
| Control Material | n | Mean Iodine (µg/ 100 g) | SD | % RSD | HorRat a | Minimum | Maximum | Previous Mean (Range, n) b | Expected c |
|---|---|---|---|---|---|---|---|---|---|
| Chocolate Milk (Control Composite II) d,e | 6 | 41.9 | 1.03 | 2.5 | 0.3 | 40.6 | 43.1 | 40.9 (1) | n/a |
| 2% Milk (Control Composite) d | 3 | 32.2 | 0.31 | 0.9 | 0.1 | 31.9 | 32.5 | n/a | n/a |
| NIST SRM®1849a Infant/Adult Nutritional Formula f | 1 | 133 | 127 (118–134, 7) | 118–140 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roseland, J.M.; Phillips, K.M.; Patterson, K.Y.; Pehrsson, P.R.; Bahadur, R.; Ershow, A.G.; Somanchi, M. Large Variability of Iodine Content in Retail Cow’s Milk in the U.S. Nutrients 2020, 12, 1246. https://doi.org/10.3390/nu12051246
Roseland JM, Phillips KM, Patterson KY, Pehrsson PR, Bahadur R, Ershow AG, Somanchi M. Large Variability of Iodine Content in Retail Cow’s Milk in the U.S. Nutrients. 2020; 12(5):1246. https://doi.org/10.3390/nu12051246
Chicago/Turabian StyleRoseland, Janet M., Katherine M. Phillips, Kristine Y. Patterson, Pamela R. Pehrsson, Rahul Bahadur, Abby G. Ershow, and Meena Somanchi. 2020. "Large Variability of Iodine Content in Retail Cow’s Milk in the U.S." Nutrients 12, no. 5: 1246. https://doi.org/10.3390/nu12051246
APA StyleRoseland, J. M., Phillips, K. M., Patterson, K. Y., Pehrsson, P. R., Bahadur, R., Ershow, A. G., & Somanchi, M. (2020). Large Variability of Iodine Content in Retail Cow’s Milk in the U.S. Nutrients, 12(5), 1246. https://doi.org/10.3390/nu12051246





