Circulating Ionized Magnesium as a Measure of Supplement Bioavailability: Results from a Pilot Study for Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Magnesium Dosing
2.3. Specimen Collection and Measurements
2.4. Statistical Analyses
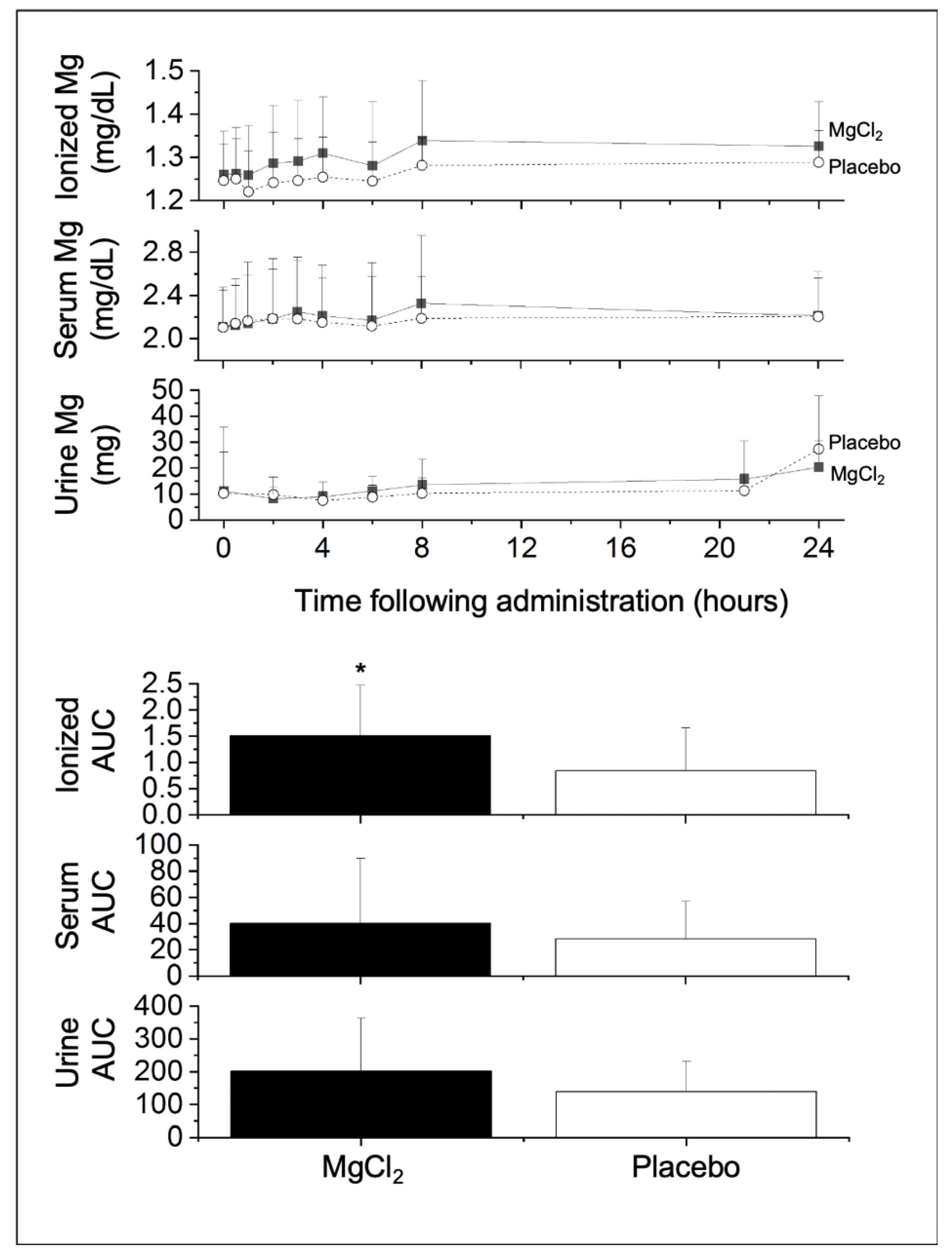
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Newhouse, I.J.; Finstad, E.W. The effects of magnesium supplementation on exercise performance. Clin. J. Sport Med. 2000, 10, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, W.J.; Haxby, E.J.; Male, D.A. Magnesium: Physiology and pharmacology. Br. J. Anaesth. 1999, 83, 302–320. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Mohammed, A. Magnesium: The forgotten electrolyte—A review on hypomagnesemia. Med. Sci. 2019, 7, E56. [Google Scholar] [CrossRef] [PubMed]
- De Baaij, J.H.; Hoenderop, J.G.; Bindels, R.J. Magnesium in man: Implications for health and disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Bairoch, A. The ENZYME database in 2000. Nucleic Acids Res. 2000, 28, 304–305. [Google Scholar] [CrossRef]
- Caspi, R.; Altman, T.; Dreher, K.; Fulcher, C.A.; Subhraveti, P.; Keseler, I.M.; Kothari, A.; Krummenacker, M.; Latendresse, M.; Mueller, L.A.; et al. The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of pathway/genome databases. Nucleic Acids Res. 2012, 40, D742–D753. [Google Scholar] [CrossRef]
- Bara, M.; Guiet-Bara, A.; Durlach, J. Regulation of sodium and potassium pathways by magnesium in cell membranes. Magnes. Res. 1993, 6, 167–177. [Google Scholar]
- Laban, E.; Charbon, G.A. Magnesium and cardiac arrhythmias: Nutrient or drug? J. Am. Coll. Nutr. 1986, 5, 521–532. [Google Scholar] [CrossRef]
- Zwillinger, L. Uber die magnesiumwirking auf das. Herz Klin Wochenschr. 1935, 14, 1429–1433. [Google Scholar] [CrossRef]
- Ford, E.S.; Mokdad, A.H. Dietary magnesium intake in a national sample of US adults. J. Nutr. 2003, 133, 2879–2882. [Google Scholar] [CrossRef]
- Fulgoni, V.L., III; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, Y.; Fulgoni, V.L. Grains contribute shortfall nutrients and nutrient density to older US adults: Data from the National Health and Nutrition Examination Survey, 2011–2014. Nutrients 2018, 10, E534. [Google Scholar] [CrossRef] [PubMed]
- McNair, P.; Christensen, M.S.; Christiansen, C.; Madsbad, S.; Transbol, I. Renal hypomagnesaemia in human diabetes mellitus: Its relation to glucose homeostasis. Eur. J. Clin. Invest. 1982, 12, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Pham, P.C.; Pham, P.M.; Pham, P.A.; Pham, S.V.; Pham, H.V.; Miller, J.M.; Yanagawa, N.; Pham, P.T. Lower serum magnesium levels are associated with more rapid decline of renal function in patients with diabetes mellitus type 2. Clin. Nephrol. 2005, 63, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Gommers, L.M.; Hoenderop, J.G.; Bindels, R.J.; de Baaij, J.H. Hypomagnesemia in type 2 diabetes: A vicious circle? Diabetes 2016, 65, 3–13. [Google Scholar] [CrossRef]
- Goldman, L.; Schafer, A.I. Goldman-Cecil Medicine, 26th ed.; Elsevier Health Sciences: Philadelphia, PA, USA, 2015. [Google Scholar]
- Dasgupta, A.; Sarma, D.; Saikia, U.K. Hypomagnesemia in type 2 diabetes mellitus. Indian J. Endocrinol. Metab. 2012, 16, 1000–1003. [Google Scholar] [CrossRef]
- Cheungpasitporn, W.; Thongprayoon, C.; Qian, Q. Dysmagnesemia in hospitalized patients: Prevalence and prognostic importance. Mayo Clin. Proc. 2015, 90, 1001–1010. [Google Scholar] [CrossRef]
- Hayes, J.P.; Ryan, M.F.; Brazil, N.; Riordan, T.O.; Walsh, J.B.; Coakley, D. Serum hypomagnesaemia in an elderly day-hospital population. Ir. Med. J. 1989, 82, 117–119. [Google Scholar]
- Wong, E.T.; Rude, R.K.; Singer, F.R.; Shaw, S.T., Jr. A high prevalence of hypomagnesemia and hypermagnesemia in hospitalized patients. Am. J. Clin. Pathol. 1983, 79, 348–352. [Google Scholar] [CrossRef]
- Whang, R.; Oei, T.O.; Aikawa, J.K.; Watanabe, A.; Vannatta, J.; Fryer, A.; Markanich, M. Predictors of clinical hypomagnesemia. Hypokalemia, hypophosphatemia, hyponatremia, and hypocalcemia. Arch. Intern. Med. 1984, 144, 1794–1796. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, R.A.; Desbiens, N.A. Hypomagnesemia in patients entering the ICU. Crit. Care Med. 1985, 13, 506–507. [Google Scholar] [CrossRef] [PubMed]
- Ryzen, E.; Wagers, P.W.; Singer, F.R.; Rude, R.K. Magnesium deficiency in a medical ICU population. Crit. Care Med. 1985, 13, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Altura, B.M.; Altura, B.T. Role of magnesium in patho-physiological processes and the clinical utility of magnesium ion selective electrodes. Scand. J. Clin. Lab. Invest. 1996, 224, 211–234. [Google Scholar] [CrossRef] [PubMed]
- Thode, J.; Juul-Jorgensen, B.; Seibaek, M.; Elming, H.; Borresen, E.; Jordal, R. Evaluation of an ionized magnesium-pH analyzer—NOVA 8. Scand. J. Clin. Lab. Invest. 1998, 58, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Yeh, D.D.; Chokengarmwong, N.; Chang, Y.; Yu, L.; Arsenault, C.; Rudolf, J.; Lee-Lewandrowski, E.; Lewandrowski, K. Total and ionized magnesium testing in the surgical intensive care unit-Opportunities for improved laboratory and pharmacy utilization. J. Crit. Care 2017, 42, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Altura, B.T.; Wilimzig, C.; Trnovec, T.; Nyulassy, S.; Altura, B.M. Comparative effects of a Mg-enriched diet and different orally administered magnesium oxide preparations on ionized Mg, Mg metabolism and electrolytes in serum of human volunteers. J. Am. Coll. Nutr. 1994, 13, 447–454. [Google Scholar] [CrossRef]
- Rooney, M.R.; Rudser, K.D.; Alonso, A.; Harnack, L.; Saenger, A.K.; Lutsey, P.L. Circulating ionized magnesium: Comparisons with circulating total magnesium and the response to magnesium supplementation in a randomized controlled trial. Nutrients 2020, 12, E263. [Google Scholar] [CrossRef]
- Blancquaert, L.; Vervaet, C.; Derave, W. Predicting and Testing Bioavailability of Magnesium Supplements. Nutrients 2019, 11, 1663. [Google Scholar] [CrossRef]
- Wilimzig, C.; Latz, R.; Vierling, W.; Mutschler, E.; Trnovec, T.; Nyulassy, S. Increase in magnesium plasma level after orally administered trimagnesium dicitrate. European J. Clin. Pharmacol. 1996, 49, 317–323. [Google Scholar] [CrossRef]
- Malon, A.; Brockmann, C.; Fijalkowska-Morawska, J.; Rob, P.; Maj-Zurawska, M. Ionized magnesium in erythrocytes--the best magnesium parameter to observe hypo- or hypermagnesemia. Clin. Chim. Acta 2004, 349, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Greenway, D.C.; Hindmarsh, J.T.; Wang, J.; Khodadeen, J.A.; Hebert, P.C. Reference interval for whole blood ionized magnesium in a healthy population and the stability of ionized magnesium under varied laboratory conditions. Clin. Biochem. 1996, 29, 515–520. [Google Scholar] [CrossRef]
- Palacios, C.; Wigertz, K.; Braun, M.; Martin, B.R.; McCabe, G.P.; McCabe, L.; Pratt, J.H.; Peacock, M.; Weaver, C.M. Magnesium retention from metabolic-balance studies in female adolescents: Impact of race, dietary salt, and calcium. Am. J. Clin. Nutr. 2013, 97, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Lowenstein, F.W.; Stanton, M.F. Serum magnesium levels in the United States, 1971–1974. J. Am. Coll. Nutr. 1986, 5, 399–414. [Google Scholar] [CrossRef]
- Norbert Wolfgang Tietz. Clinical Guide to Laboratory Tests; W.B. Saunders Company: Philadelphia, PA, USA, 1990. [Google Scholar]
- Firoz, M.; Graber, M. Bioavailability of US commercial magnesium preparations. Magnes. Res. 2001, 14, 257–262. [Google Scholar]
- Von Ehrlich, B.; Barbagallo, M.; Classen, H.G.; Guerrero-Romero, F.; Mooren, F.C.; Rodriguez-Moran, M.; Vierling, W.; Vormann, J.; Kisters, K. Significance of magnesium in insulin resistance, metabolic syndrome, and diabetes–recommendations of the Association of Magnesium Research e.V. Trace Elem. Electrolytes 2017, 34, 124. [Google Scholar] [CrossRef]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The case for an evidence-based reference interval for serum magnesium: The time has come. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef]
- Chan, K.H.; Chacko, S.A.; Song, Y.; Cho, M.; Eaton, C.B.; Wu, W.C.; Liu, S. Genetic variations in magnesium-related ion channels may affect diabetes risk among African American and Hispanic American women. J. Nutr. 2015, 145, 418–424. [Google Scholar] [CrossRef]
- McHale, J. Thiocyanate interference with Nova’s ionized magnesium electrode. Clin. Chem. 1997, 43, 1672. [Google Scholar] [CrossRef]
- Wong, S.L.; Epperson, A.E.; Rogers, J.; Castro, R.J.; Jackler, R.K.; Prochaska, J.J. A multimodal assessment of tobacco use on a university campus and support for adopting a comprehensive tobacco-free policy. Prev. Med. 2020, 133, 106008. [Google Scholar] [CrossRef]
- Rowe, S.; Alexander, N.; Clydesdale, F.M.; Applebaum, R.S.; Atkinson, S.; Black, R.M.; Dwyer, J.T.; Hentges, E.; Higley, N.A.; Lefevre, M.; et al. Funding food science and nutrition research: Financial conflicts and scientific integrity. J. Nutr. 2009, 139, 1051–1053. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Value 1 | Reference Range |
|---|---|---|
| Age (y) | 25.0 (18–44) | |
| Male (%) | 41.2 | |
| Female (%) | 58.8 | |
| Race/ethnicity (%) | ||
| White | 58.8 | |
| Black | 11.8 | |
| Hispanic | 5.9 | |
| Asian | 23.5 | |
| Body mass index (kg/m2) | 24.7 ± 3.7 | |
| iMg2+ (mmol/L) | 0.52 ± 0.03 | 0.44–0.59 |
| Serum total magnesium (mmol/L) | 0.84 ± 0.01 | 0.75–0.95 |
| Serum creatinine (mmol/L) | ||
| Female (mmol/L) | 0.30 ± 0.07 | 0.23–0.42 |
| Male (mmol/L) | 0.37 ± 0.05 | 0.30–0.49 |
| Parameter | Placebo | MgCl2 | p Value |
|---|---|---|---|
| iMg2+ (n = 14) | |||
| Baseline (mg/dL) | 1.25 ± 0.08 | 1.26 ± 0.01 | 0.242 |
| AUC (mg/dL•24h) | 0.84 ± 0.82 | 1.51 ± 0.96 | 0.029 |
| Cmax (mg/dL) | 1.32 ± 0.07 | 1.38 ± 0.13 | 0.034 |
| Tmax (h) | 9.54 ± 9.85 | 10.36 ± 9.30 | 0.396 |
| Serum magnesium (n = 17) | |||
| Baseline (mg/dL) | 1.95 (1.78–3.30) | 2.08 (1.76–3.19) | 0.491 |
| AUC (mg/dL•24h) | 14.55 (0–91.18) | 27.00 (0–172.93) | 0.097 |
| Cmax (mg/dL) | 2.24 (1.98–4.31) | 2.38 (1.97–4.01) | 0.221 |
| Tmax (h) | 11.38 ± 9.93 | 10.82 ± 9.11 | 0.396 |
| Urine magnesium (n = 12) | |||
| Baseline (mg) | 2.90 (0.81–47.15) | 2.95 (0.35–88.63) | 0.267 |
| AUC (mg•24h) | 139.30 ± 92.84 | 201.74 ± 161.63 | 0.118 |
| Cmax (mg) | 24.38 (13.51–81.51) | 26.12 (12.91–88.63) | 0.469 |
| Tmax (h) | 24.00 (0–24.00) | 24.00 (0–24.00) | 0.363 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhan, J.; Wallace, T.C.; Butts, S.J.; Cao, S.; Ansu, V.; Spence, L.A.; Weaver, C.M.; Gletsu-Miller, N. Circulating Ionized Magnesium as a Measure of Supplement Bioavailability: Results from a Pilot Study for Randomized Clinical Trial. Nutrients 2020, 12, 1245. https://doi.org/10.3390/nu12051245
Zhan J, Wallace TC, Butts SJ, Cao S, Ansu V, Spence LA, Weaver CM, Gletsu-Miller N. Circulating Ionized Magnesium as a Measure of Supplement Bioavailability: Results from a Pilot Study for Randomized Clinical Trial. Nutrients. 2020; 12(5):1245. https://doi.org/10.3390/nu12051245
Chicago/Turabian StyleZhan, Jiada, Taylor C. Wallace, Sarah J. Butts, Sisi Cao, Velarie Ansu, Lisa A. Spence, Connie M. Weaver, and Nana Gletsu-Miller. 2020. "Circulating Ionized Magnesium as a Measure of Supplement Bioavailability: Results from a Pilot Study for Randomized Clinical Trial" Nutrients 12, no. 5: 1245. https://doi.org/10.3390/nu12051245
APA StyleZhan, J., Wallace, T. C., Butts, S. J., Cao, S., Ansu, V., Spence, L. A., Weaver, C. M., & Gletsu-Miller, N. (2020). Circulating Ionized Magnesium as a Measure of Supplement Bioavailability: Results from a Pilot Study for Randomized Clinical Trial. Nutrients, 12(5), 1245. https://doi.org/10.3390/nu12051245






