Abstract
Vitamin D (VD) plays an essential role in mineral homeostasis and bone remodeling. A number of different VD-related genes (VDRG) are required for the metabolic activation of VD and the subsequent induction of its target genes. They include a set of genes that encode for VD-binding protein, metabolic enzymes, and the VD receptor. In addition to its well-characterized skeletal function, the immunoregulatory activities of VD and the related polymorphisms of VDRG have been reported and linked to its therapeutic and preventive actions for the control of several viral diseases. However, in regards to their roles in the progression of viral diseases, inconsistent and, in some cases, contradictory results also exist. To resolve this discrepancy, I conducted an extensive literature search by using relevant keywords on the PubMed website. Based on the volume of hit papers related to a certain viral infection, I summarized and compared the effects of VD and VDRG polymorphism on the infection, pathogenesis, and treatment outcomes of clinically important viral diseases. They include viral hepatitis, respiratory viral infections, acquired immunodeficiency syndrome (AIDS), and other viral diseases, which are caused by herpesviruses, dengue virus, rotavirus, and human papillomavirus. This review will provide the most current information on the nutritional and clinical utilization of VD and VDRG in the management of the key viral diseases. This information should be valuable not only to nutritionists but also to clinicians who wish to provide evidence-based recommendations on the use of VD to virally infected patients.
1. Vitamin D and Vitamin D-Related Genes
Vitamin D (VD) is an essential micronutrient required for the intestinal absorption of minerals such as calcium, magnesium, and phosphate and the maintenance of bone strength. This fat-soluble secosteroid hormone is a key regulator necessary for overall mineral homeostasis and bone remodeling [1]. The classical calcium-related skeletal functions of VD are well established from a historical perspective due to the clear causal association of VD deficiency with the development of rickets, a form of osteomalacia in young children [2]. Due to the critical role of VD in the prevention of bone-related diseases, VD supplementation has been recommended for populations who are at high risk for the development of VD deficiency. The beneficial effects of VD supplementation on other medical conditions such as cancer, cardiovascular, and infectious diseases have been well documented. However, despite these encouraging results, numerous studies have also reported unclear and sometimes conflicting data on the effects of VD supplementation on the progression or prevention of certain diseases [3,4,5,6]. In particular, with regard to its non-skeletal effects on general mortality, cancer, and infectious diseases, the nutritional and therapeutic benefits of VD supplementation have been controversial and inconclusive [4,7,8].
VD3 (cholecalciferol) is generated via endogenous UVB-dependent photochemical synthesis from pro-VD3 in the skin or through the dietary intake [9,10] (Figure 1). It is then converted into 25-hydroxy VD3 (25(OH)D3) (calcifediol) by a hepatic VD3 25-hydroxylase (CYP2R1) [10]. 25(OH)D3 levels are frequently measured in serum to determine VD status since it is the major circulating form of VD. The second round of 25(OH)D3 hydroxylation is further facilitated by a renal 25-hydroxy VD3-1α-hydroxylase (CYP27B1) and results in the production of 1,25-dihydroxy VD3 (1,25(OH)2D3) (calcitriol), which is an active form of VD [11]. 1,25(OH)2D3 is then transported to local tissues with the help of an α-globulin carrier protein called VD-binding protein (VDBP). After its intracellular uptake, 1,25(OH)2D3 binds to its cognate VD receptor (VDR) in the cytoplasm [12]. This 1,25(OH)2D3-bound VDR creates a complex with a retinoic acid receptor (RXR) and p62 in the nucleus. Subsequent association of this trimeric 1,25(OH)2D3-bound VDR/RXR/p62 complex with VD-responsive elements (VDRE) in the promoter regions of target genes drives the transcriptional activation of a set of VD-responsive genes. For the neutralization of VD, 1,25(OH)2D3 is further converted into an inactive metabolite (1,24,25(OH)3D3) by another hydroxylase enzyme, CYP24A1. In this paper, all genes involved in VD metabolism, activation, and degradation are collectively designated as VD-related genes (VDRG). As shown in Figure 1, they include 7-dehydrocholesterol reductase (DHCR7), VDBP, CYP2R1, CYP27A1, CYP27B1, CYP24A1, and VDR.
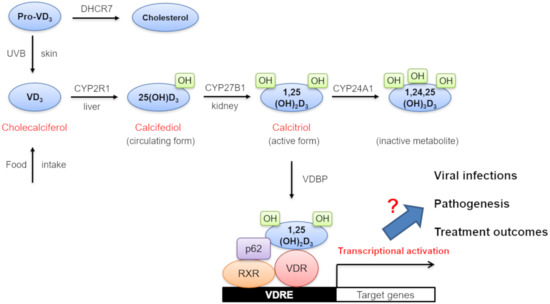
Figure 1.
Diagram of vitamin D (VD) synthesis, metabolism, biological action, and its potential effects on viral infections, pathogenesis, and treatment outcomes. Abbreviations: VD3, vitamin D3; DHCR7, 7-dehydrocholesterol reductase (DHCR7); UVB, ultraviolet B; 25(OH)D3, 25-hydroxy vitamin D3; 1, 25(OH)D3, 1, 25-dihydroxy vitamin D3; RXR, retinoic acid receptor; VDR, vitamin D receptor; VDRE, vitamin D responsive elements.
2. VD Deficiency and the Controversial Roles of VD in the Development of Viral Diseases
Plasma VD levels are closely linked to changing seasons due to the dependence of VD activation on UV radiation [9,10]. Besides, numerous epidemiological studies have confirmed the impacts of geographical location on plasma VD levels due to sun exposure variation among different latitudes. Under normal conditions, the calcium-parathyroid hormone-VD axis is involved in the maintenance of optimal plasma calcium levels. VDBP also plays an important role in the regulation of serum VD levels through its plasma transportation capability [13]. In 2011, an Institute of Medicine committee concluded that a 25(OH)D3 serum level of 20 ng/mL (50 nmol/L) is necessary for overall bone health [14]. Based on this recommendation, VD deficiency is generally defined as a plasma 25(OH)D3 level less than 20 ng/mL (50 nmol/L).
As mentioned previously, the cause and effect relationship of VD levels with certain diseases has been inconsistent [3,4,5,6,7,8,15]. In some instances, VD deficiency is believed to be a consequence of ill health rather than its cause. Nevertheless, numerous observational studies have suggested a potential role of VD in the development of various infectious diseases [16,17,18,19,20]. A plausible mechanism for the causal relationship between VD status and the progression of certain infectious diseases involves either direct or indirect interactions of the components of the VD pathway with the host immune system [18,19,20]. In support of this view, VDRs’ expression was confirmed in several white blood cells, such as monocytes and activated T and B lymphocytes [20]. Besides, the production of 1,25(OH)2D3 by local immune cells in response to infection has also been reported [21]. This paracrine and intracrine effect of VD on the immune system appears to be distinct from its classical endocrine effects on calcium regulation [18]. In line with this, the VD-induced production of the antimicrobial peptide, cathelicidin, plays a critical role in the innate defense system against several bacterial and viral pathogens [17,20]. Furthermore, the anti-proliferative effects of VD and its ability to induce autophagy and apoptosis were implicated in the active control of certain infections [18,19]. Likewise, the immunoregulatory functions of VD included the suppression of innate immune responses through the downregulation of inflammatory cytokines and the polarization of the adaptive immune system toward Th-2 responses [18]. However, these immune-related properties of VD seem to be very dependent on the nature of interactions between hosts and pathogens. Therefore, the true roles of VD and VDRG in the development and control of infectious diseases are still elusive [16,17].
To gain a better understanding and resolve the current controversy regarding the relationship between VD and the development and control of viral infections, I conducted an extensive literature search by using relevant keywords on the PubMed website. Based on the volume of hit papers related to a certain viral infection, I summarized and compared the effects of VD and VDRG polymorphism on the infection, pathogenesis, and treatment outcomes of key viral diseases (Figure 1). They are caused by the viruses of clinical importance, such as hepatitis C virus (HCV), respiratory viruses (influenza virus, respiratory syncytial virus (RSV), and rhinovirus), hepatitis B virus (HBV), human immunodeficiency virus (HIV), herpesviruses (herpes simplex virus type 1 (HSV 1), Epstein Barr virus (EBV), varicella-zoster virus (VZV), and the human cytomegalovirus (HCMV)), dengue virus (DENV), rotavirus, and human papillomavirus (HPV). Based on this information, the potential reasons for data inconsistency regarding the effects of VD on viral infections were analyzed. The nutritional and therapeutic values of VD in the management of these viral diseases were also reassessed. Finally, I proposed the future research direction to resolve the controversy on the role of VD and VDRG in the pathogenesis of viral diseases.
3. HCV
3.1. Effect of VD on HCV Infection
Hepatitis C is an inflammatory liver disease characterized by infection of hepatocytes with a hepatotropic, single-stranded RNA virus known as HCV. Approximately 170 million people are estimated to be infected with HCV worldwide [22]. The development of many effective, direct-acting antiviral agents that target the viral replication complex renders most HCV infections from different genotypes curable via pharmacological intervention.
Three research groups demonstrated the inhibition of HCV production by treatment of 1,25(OH)2D3 by using Huh 7.5 cells infected with HJ3-5 and JFH-1 HCV (Table 1) [23,24,25]. Matsumura et al. also observed a reduction in HCV core antigen production with 25(OH)D3 treatment at the extra- and intracellular levels in an in vitro system [26]. Gutierrez et al. and Lin et al. reported the anti-HCV replication activities of VD derivatives including VD2, VD3, and 1,25(OH)2D3 at 1–5 µM concentrations in vitro [27,28]. Structurally related VD analogs, such as calcipotriol and tacalcitol, also suppressed HCV replication in vitro [29]. Mechanistically, the activation of the interferon (IFN) signaling pathway, the blockade of peroxisome proliferator-activated receptor (PPAR), and the inhibition of endoplasmic reticulum-associated degradation (ERAD) pathways by VD were proposed as their modes of action [23,28]. Besides, a causal linkage between reduced apolipoprotein expression caused by 25(OH)D3 treatment and reduced HCV particle production was documented [24]. Ravid et al. also provided mechanistic evidence for the VDR-independent antiviral action by 25(OH)D3 [25]. Upon cotreatment with IFN, an enhanced inhibitory effect of 1,25(OH)2D3 on HCV replication was observed [30]. Mechanistically, this improvement in the antiviral activity of IFNα by 1,25(OH)2D3 seems to be mediated by the non-genomic action of VD since 1,25(OH)2D3 was shown to increase the binding of Stat1 to its DNA target and the subsequent upregulation of IFNα target gene expression [30]. This could be a plausible explanation for the association of VD-deficiency to poor responsiveness to PEG-IFNα treatment [31]. In clinical settings, VD deficiency (plasma 25(OH)D3 <20 ng/mL) was one of the most common symptoms among HCV patients, and a negative correlation between VD levels and viral loads in HCV patients was also frequently reported [32,33]. Thus, VD deficiency has been regarded as a risk factor for HCV infection and subsequent chronic progression [34]. In line with this observation, an inverse correlation between VD plasma levels and the expression of the VDR was also reported in HCV patients [35]. In contrast, one clinical study found no significant association of VD deficiency with poor virological characteristics in HCV patients [36].

Table 1.
Effects of VD on hepatitis C virus (HCV) infection, pathogenesis, and treatment outcomes.
3.2. Effect of VD on HCV Pathogenesis
Chronic liver inflammation and the subsequent progression to liver cirrhosis and hepatocellular carcinoma (HCC) are typical clinical symptoms of chronic HCV infection. A significant inverse relationship between the severity of inflammatory fibrosis and cirrhosis and serum 25(OH)D3 levels was observed in HCV patients [32,33,37,40,62,63,64]. This implies VD deficiency as an independent predictor and risk factor for inflammatory liver complications in HCV patients [38,39,65,66,67]. VD deficiency was also associated with reduced hepcidin expression, mixed cryoglobulinemia, and hepatic encephalopathy in HCV patients [41,42,43]. Therefore, VD supplementation has been suggested as a potential therapeutic option to reverse the hepatic complications caused by HCV infection [53]. Indeed, VD supplementation was able to suppress hepatic fibrosis induced by HCV infection [53]. One of the mechanisms for VD-dependent alleviation of hepatic fibrosis was via the VDR-dependent downregulation of the transforming growth factor (TGF) β-1/small mothers against decapentaplegic (SMAD) 3 pathway [68]. One clinical study showed a significantly higher mortality rate in HCV patients with severe VD deficiency (25(OH)D3 levels <5 ng/mL) [43]. Despite these positive effects by VD on HCV pathogenesis, several research groups also reported seemingly contradictory results. For example, treatment-naive Chinese HCV patients failed to show any correlation between serum levels of 25(OH)D3 and 24,25(OH)2D3 and liver fibrosis [54]. Oliveira et al. and Esmat et al. also reported no significant association between VD serum levels and inflammatory activity or the degree of liver fibrosis [55,59]. Another clinical study also suggested that VD deficiency was not responsible for poor virological characteristics in HCV patients [36]. In support of this view, VD supplementation had no immediate impact on the viral RNA loads of HCV patients [61].
3.3. Effect of VD on HCV Infection Treatment Outcomes
Before the development of direct-acting antiviral therapy that targets viral structural proteins, a pegylated (PEG)-IFNα/ribavirin combination therapy was the standard of care for HCV patients. Several researchers reported a positive correlation between VD status and sustained virological response (SVR) rates to this standard PEG-IFNα/ribavirin therapy for HCV patients [34,48,51,52,66]. Therefore, VD deficiency was regarded as a potential predictor of an unfavorable response to IFNα-based HCV treatment [45,46,50,64]. In line with this, VD supplementation improved the probability of achieving an SVR following antiviral treatment [37,45,46,49,51,69,70,71]. HIV–HCV coinfected patients also demonstrated a strong correlation between VD levels and SVR [47]. These data strongly emphasized the utility of VD supplementation for the management of HCV infection in conjunction with standard IFNα-based therapy [35]. However, contrary to these results, many reports failed to observe a significant association between VD levels and SVR to IFNα-based therapy in chronic HCV infections [56,57,58,59,60,72]. In particular, a specific ethnic group such as African American HCV patients also did not show any relationship between 25(OH)D3 serum concentration and SVR [73]. Furthermore, two other studies also failed to establish a relationship between 25(OH)D3 levels, biochemical liver markers, and fibrosis stages in HCV patients [56,61].
3.4. Effect of VDRG Polymorphisms on HCV Infection
Three studies examined the potential effects of VDRG polymorphisms on HCV infection. Wu et al. demonstrated the protective roles of VDR variants including rs7975232-C, rs2239185-T, rs11574129-T, rs757343-A, rs739837 A, and CYP24A1 variant rs6068816-T against HCV infection [34]. On the other hand, VDBP variants (rs7041-G and rs3733359-T), a CYP24A1 variant (rs6013897-A), and CYP2R1 variants (rs12794714-G, rs10741657-A, rs1562902-C, and rs10766197-G) were associated with increased susceptibility to HCV infection [13]. Besides, HCV patients with VDR variants such as bAt [CCA]-haplotype, ApaI CC genotype, and TaqI AA genotype had higher viral loads [74].
3.5. Effect of VDRG Polymorphisms on HCV Pathogenesis
Since VDRG plays a key role in the maintenance of proper plasma VD concentration, many studies aimed to identify the potential effects of VDRG polymorphisms on viral diseases. Seven studies have reported potential VDRG polymorphism effects on HCV pathogenesis. Among HCV patients, VDBP protein levels were significantly higher for normal/mild fibrosis when compared to those for advanced fibrosis [75]. VDR BsmI and TaqI variants affected the progression of fibrosis in HCV patients [76]. When Barooach et al. determined whether VDR, VDBP, and CYP2R1 gene polymorphisms are risk factors for clinical complications in HCV-related HCC patients, they identified the VDR ApaI CC genotype and the VDR bAt haplotype as independent predictors for cirrhosis and HCC development in HCV patients [77]. When Langer et al. performed a similar study to investigate the potential link between CYP2R1, VDBP, and DHCR7 genotypes and the risk of HCV-related HCC development by using 1279 HCV-related HCC patients, they found a functionally relevant role for VD in the prevention of HCV-related HCC [44]. This further implies that defective signaling in the VD pathway may contribute to hepatocarcinogenesis in HCV-infected patients [44]. Although the detailed mechanistic explanation for this linkage of VDR, VDBP, and CYP2R1 polymorphisms to the development of hepatocellular carcinoma was not given, the hepatic conversion of VD to 23(OH)D3, the plasma transportation of VD by VDBP, and the intracellular binding of VD with VDR might be affected by polymorphic changes in VDR, VDBP, and CYP2R1 genes. These changes might, in turn, alter the overall strength of VD signaling, ultimately affecting the development of HCC in HCV patients. VDR FokI rs2228570 TT/TC genotypes were also suggested as risk factors for advanced liver fibrosis in HCV patients [78]. In addition, the expression levels of VDR in cholangiocytes were inversely correlated with pathological progression markers of HCV infection [79]. Moreover, CYP2R1 expression in hepatocytes showed a strong correlation with VDR levels. In line with this, VDR expression levels demonstrated a negative correlation with the severity of liver histology in both non-alcoholic steatohepatitis and HCV patients [79]. Interestingly, rs7041 and rs4588 VDBP polymorphisms did not play a direct role in liver fibrosis despite their strong association with VD levels in HCV patients [80].
3.6. Effect of VDRG Polymorphisms on HCV Infection Treatment Outcomes
As critical determinants of VD levels, the polymorphic effects of VDRG on the responsiveness of HCV treatment in HCV patients were also studied. Six studies examined the potential effects of VDRG polymorphisms on HCV infection treatment outcomes. Cusato et al. showed that CYP27B1 rs10741657, CYP24A1, VDR rs2228570, FokI, and TaqI polymorphisms, in combination with the IL28B polymorphism (rs12979860), impacted HCV infection treatment outcomes [81]. DHCR7-TT and rs12785878 polymorphisms were also significantly associated with an SVR to IFN-based therapy [57]. A polymorphism near the CYP27B1-1260 promoter region (rs10877012) reduced 1,25(OH)2D3 serum levels, which, in turn, worsened an SVR in HCV patients [82]. Falleti et al. showed that a combination of a basal VD level >20 ng/mL and VDBP wild type 1 was an independent predictor of a higher SVR to IFN-based therapy [83]. Petta et al. also demonstrated an association between 25(OH)D3 serum levels, IL28B status, and the probability of achieving an SVR [84]. On the other hand, the VDR bAt (CCA) haplotype, consisting of the BsmI rs1544410 C, ApaI rs7975232 C, and TaqI rs731236 A alleles, was linked with a poor response to IFNα-based therapy [78]. However, there are conflicting results regarding the roles of VDRG polymorphisms on HCV infection treatment outcomes. For example, VDR gene polymorphism did not correlate with rapid virological response and SVR achievement in HCV patients [74]. In addition, the VDBP, CYP2R1, and CYP27B1 polymorphisms did not play any role in the treatment outcomes of HCV infection [85].
4. Respiratory Viruses Such as Influenza Virus, RSV, and Rhinovirus
4.1. Effect of Vd on Respiratory Viral Infections, Immune Response, and Pathogenesis
Respiratory viruses are defined as viruses responsible for the development of either upper or lower respiratory tract infections. Typically, they include influenza virus, parainfluenza virus, adenovirus, respiratory syncytial virus (RSV), and rhinovirus. With regards to the role of VD in control of viral respiratory infections, VD inhibited rhinovirus replication in primary cystic fibrosis bronchial cells through the induction of cathelicidin [86] (Table 2). Besides, VD also decreased rhinovirus replication and release and increased the expression of IFN-stimulated genes and cathelicidin in human primary brain endothelial cells (HPBEC) infected with rhinovirus 1B [87]. In particular, cathelicidin exhibited direct anti-rhinovirus activity in in vitro experiments using HPBEC cells infected with rhinovirus 1B [87]. In line with this, the pretreatment of A549 respiratory epithelial cells with 25(OH)D3 induced transient resistance to rhinovirus infection [88]. It also attenuated the rhinovirus-induced expression of genes encoding intercellular adhesion molecule 1 and platelet-activating factor receptor in A549 cells infected with rhinovirus 16 [88]. The induction of IkBa, an NF-kB inhibitor, via VD treatment attenuated the expression of NF-kB-driven proinflammatory genes in human tracheobronchial epithelial cells (hTBE) infected with RSV strain 2A [89]. This, in turn, decreased the inflammatory response to rhinovirus infection in airway epithelium without affecting viral clearance [89]. Another research group demonstrated VD treatment-induced inhibition of NFκB and STAT1-regulated gene expression in RSV-infected A549 cells [90]. However, despite the positive effects displayed by VD on respiratory viruses, in vitro VD treatment had no direct effect on rhinovirus replication in experiments using primary human bronchial epithelial cells (hBEC) [91]. Moreover, the VD-induced production of cathelicidin had no impact on the burden of influenza virus infection [92]. Fitch et al. also failed to observe any VD-induced anti-inflammatory effects during RSV infection in experiments using fresh peripheral blood mononuclear cells (PBMC) or CD4+ monocytes [93].

Table 2.
Effects of VD on respiratory viral infection, pathogenesis, and treatment outcomes.
VD deficiency has been associated with an increased risk of lower respiratory viral infections caused by the influenza virus, RSV, and rhinovirus [100]. In particular, preschool females with low 25(OH)D3 levels were more susceptible to respiratory viral infections than males [94]. Moreover, low VD levels were associated with a significantly elevated risk of intensive care unit admission and invasive mechanical ventilation after respiratory viral infections [95]. In line with this, a higher 25(OH)D3 cord serum level reduced the risk of virally induced wheezing in a clinical study of 190 affected children [96]. As expected, VD supplementation protected against respiratory viral infections in both healthy patients and patients with respiratory diseases through the positive modulation of innate immune responses [101]. The meta-analysis of several clinical studies by Martineau et al. also showed the reduced risk of acute respiratory tract infection by VD supplementation [102]. In support of this, higher VD levels also correlated with increased immunogenicity, which was demonstrated by the more efficient production of influenza virus-specific antibodies [97]. However, despite numerous evidence supporting the positive role of VD in the prevention of respiratory viral infections, several studies reported seemingly conflicting results. For example, no significant association between VD levels and immunogenic responses to influenza vaccination was found by one clinical study [103]. Besides, no evidence of improved influenza vaccine immunogenicity with VD supplementation was reported in HIV-positive populations [98]. VD supplementation also failed to induce consistent protective effects on respiratory viral infections caused by the influenza virus, RSV, or rhinovirus [104]. VD supplementation was even found to be associated with an increase in repeat episodes of infectious pneumonia [105]. Finally, VD deficiency did not play any role in the development of acute bronchiolitis caused by RSV infection in 145 infants [99].
4.2. Effect of VDRG Polymorphisms on Respiratory Viral Infections
A significant association between VDR FokI polymorphism and respiratory viral infections has been observed [106]. In line with this, VDR FokI variants also showed enhanced immunopathology and exacerbated bronchiolitis after RSV infection [90]. This effect was mediated by blocking STAT1-mediated antiviral immune reactions to RSV infection [90]. Three single nucleotide polymorphisms (SNP) in VDR (rs4334089, rs11568820, and rs7970314) and one SNP in CYP3A4 (rs2740574) were also suggested as risk factors for upper respiratory infection by rhinovirus and RSV [107]. This is supported by McNally et al. who reported an association between the VDR FokI polymorphism and the severity of RSV infection [108]. The VDBP’s haplotype also played a positive role in RSV bronchiolitis in infancy and subsequent asthma development [109]. Besides, the VDBP’s haplotype was associated with higher VDBP levels, which might have detrimental effects on RSV bronchiolitis due to reduced levels of VD [109].
5. HBV
5.1. Effect of VD on HBV Infection, Immune Response, Pathogenesis, and Treatment Outcomes
More than 240 million people around the world live with chronic HBV infection [110,111]. Reverse transcriptase inhibitors along with nucleoside and nucleotide structures are the main treatments against HBV infection [112]. However, similar to HIV patients, these anti-HBV therapeutics are not able to provide a complete cure for HBV infection because of their inability to remove stable, nuclear, and covalently closed circular DNA (cccDNA), which serves as a transcription template for the continuous production of viral mRNA and pre-genomic RNA [113].
According to epidemiological studies, significantly decreased VD levels were consistently found in chronic HBV patients [114]. The 25(OH)D3 plasma level showed a significant inverse correlation with plasma HBV DNA loads and a positive correlation with the seroclearance of the hepatitis B surface antigen (HBsAg) [115,116] (Table 3). In turn, reduced VD levels were associated with the clinical progression of liver cirrhosis and adverse clinical outcomes [117]. In line with this clinical evidence, the downregulation of VDR expression in HBV-transfected cells caused an increase in HBV transcription and translation [118]. Effective antiviral therapy, in contrast, restored VD to normal levels in HBV patients [119]. Among 560 HBV patients, those with sufficient VD achieved a higher SVR than those with VD deficiency [120]. Besides, the addition of VD to standard IFNα therapy achieved higher efficacy than IFNα alone in an HBV transgenic mouse model [121]. However, several studies also reported contradictory results regarding the effects of VD on HBV infection. Berkan-Kawinska et al. reported no association between 25(OH)D3 deficiency and poor virological characteristics in 35 HBV patients [36]. In support of this, HBV DNA levels were not associated with VD levels in 84 HBV patients [122]. In addition, serum 25(OH)D3 concentration did not correlate with histological or biochemical markers of liver inflammation in 58 HBV patients [123]. Serum 25(OH)D3 also failed to have any impact on the immune control of HBV infection [123]. 25(OH)D3 levels did not affect the immunogenicity of hepatitis B e antigen (HBeAg), HBV viral loads, or fibrosis stage in two studies examining 560 and 242 HBV patients, respectively [120,124]. Finally, baseline VD levels were not associated with HBV infection treatment outcomes with a tenofovir plus PEG-IFNα combination therapy in 737 HBV patients [125].

Table 3.
Effects of VD on hepatitis B virus (HBV) infection, pathogenesis, and treatment outcomes.
5.2. Effect of Vdrg Polymorphisms on Hbv Infection, Pathogenesis, and Treatment Outcomes
Eight studies examined the potential effects of VDRG polymorphisms on HBV infection, pathogenesis, and treatment outcomes. A VDR gene polymorphism at codon 352 (genotype tt) was significantly underrepresented among HBV patients [127]. In particular, the VDR FokI FF gene was shown to be a risk factor for HBV infection [128]. In addition, VDR gene polymorphisms were associated with distinct clinical phenotypes in Taiwanese HBV patients [129]. In line with this, VDR ApaI was also associated with the clinical outcome of and liver disease progression in HBV patients [130]. From a therapeutic perspective, VDR rs7975232/ApaI was identified as a pretreatment predictor of sustained HBsAg seroclearance in HBeAg-positive HBV patients when treated with PEG-IFNα [131]. In support of this, VDR ApaI SNP was associated with viral loads and the presence of HBsAg in response to PEG-IFNα treatment [132]. VDR FokI SNP and the bAt haplotype were also suggested as independent factors that can predict PEG-IFNα treatment responses in HBV patients [133]. Finally, the VDBP rs222020 TT genotype independently predicted the sustained HBsAg seroclearance and the normalization of aspartate aminotransferase after PEG-IFNα treatment [134].
6. HIV
6.1. Effect of VD on HI Infection, Immune Response, Pathogenesis, and Treatment Outcomes
HIV is an etiological agent responsible for the development of acquired immunodeficiency syndrome (AIDS). Concerning the effects of VD on HIV biology, 1,25(OH)2D3 inhibited HIV replication through the induction of autophagy and phagosomal maturation in PBMC infected with HIVBa-L [135,136] (Table 4). Mechanistically, the induction of cathelicidin by VD played an essential role in 1,25D3-induced autophagic flux and inhibition of HIV replication [135]. In particular, high levels of VD and its receptor are related to natural resistance to HIV-1 infection in the PBMC from HIV-1-exposed seronegative individuals [137]. In line with this, VD deficiency was suggested as a predictor for short-term mortality in 250 HIV patients [138]. The importance of cathelicidin in the suppression of HIV infection by VD was further supported by elevated cathelicidin mRNA levels in PBMCs and oral mucosa of HIV-1-exposed seronegative individuals [139]. Among 90 patients with HIV/Kaposi’s sarcoma, HIV RNA levels were significantly higher among those with VD deficiency [140]. VD deficiency was also associated with reduced recovery of CD4+ T-cell count in 398 HIV patients by anti-retroviral therapy [141]. In support of this, VD supplementation attenuated HIV-1 replication and increased the number of circulating leukocytes from 100 VD3-fed healthy adults [142]. In in vitro experiments using PBMC infected with HIVBa-L, VD treatment reduced HIV-1 infection in T cells by inducing antiviral gene expression and reducing the levels of viral co-receptor CCR5 [143]. In ex vivo experiments using PBMC from seronegative individuals infected with HIV-1, VD treatment was able to reduce HIV-1 transmission by specifically modulating the activation of T cells and the production of antiviral factors [144]. VD supplementation also reduced TB/HIV co-infection and its progression [145]. Cathelicidin levels were significantly lower in HIV patients when compared to healthy controls [146]. Besides, decreased 25(OH)D3 levels were also linked to reduced responsiveness to retroviral therapy [147]. However, despite all these positive effects of VD on HIV infection, several research groups reported opposing results. VD3 and 1,25(OH)2D3 administered over 16 weeks failed to change T cell numbers in HIV patients [148]. In addition, no HIV-dependent variables were found to be associated with 25(OH)D3 levels in HIV patients [149]. Besides, no improvements in HIV viral loads, CD4+ T cell counts, or CD8+ T cell counts were detected after VD supplementation in HIV patients [150]. Moreover, routine multiple micronutrient supplementation, including VD, showed no significant beneficial effects on mortality in HIV patients [151]. In the therapeutical perspective, VD deficiency was not associated with the outcomes of antiretroviral therapy in HIV patients [140].

Table 4.
Effects of VD human immunodeficiency virus (HIV) infection, pathogenesis, and treatment outcome.
6.2. Effect of VDRG Polymorphisms on HIV Infection and Pathogenesis
Three studies reported results regarding the potential effects of VDRG polymorphisms on HIV infection, pathogenesis, and treatment outcome. VDR diplotypes in 5′ untranslated region (UTR) and 3′UTR locus combinations were associated with the pathogenesis of AIDS [154]. The VDR variant rs1544410_AA was also associated with the progression to AIDS and resistance to HIV-1 [155]. Moreover, VDR haplotypes influenced the risk of HIV-1 acquisition [156].
7. Herpesviruses
More than 90% of the adult population is infected with one or more forms of herpes virus [157]. Herpesviruses, including herpes simplex virus type (HSV) 1, Epstein Barr virus (EBV), varicella-zoster virus (VZV), and human cytomegalovirus (HCMV), are responsible for a wide variety of recurrent diseases, such as cold sores, shingles, congenital defects, and several malignancies. Concerning the effects of VD on herpesvirus infections, a significant downregulation of HSV-1 titers was observed in both 25(OH)D3- and 1,25(OH)2D3-treated HeLa cells infected with HSV-1 [158] (Table 5). VDR expression was downregulated in human foreskin fibroblasts infected with CMV AD169 [159]. In line with this, among 547 HCMV patients, VD deficiency was independently associated with an increased incidence of opportunistic viral infection by HCMV [160]. A total of 88 chronic hemodialysis patients with VD deficiency had significantly reduced immunogenicity against VZV infection [161]. A significant correlation between VD deficiency and the incidence of HCMV disease was also reported by a clinical study examining the 139 HCMV patients [162]. In addition, VD deficiency was also suggested as a risk factor for acute rejection caused by HCMV infection after kidney transplantation [163]. VD deficiency was also frequently observed in infectious mononucleosis patients caused by EBV infection [164]. Along with an inverse correlation between 25(OH)D3 levels and EBV infection [165], a significant association between low VD serum levels and the presence of recurrent herpes labialis was also reported [166]. However, in spite of these positive results, VD metabolites failed to inhibit HCMV replication in human foreskin fibroblasts infected with CMV AD169 [159].

Table 5.
Effects of VD on infection, pathogenesis, and treatment outcome by other viruses.
8. Dengue Virus
Dengue virus (DENV) is responsible for the development of dengue fever, a mosquito-borne tropical disease. A high dose of 1,25(OH)2D3 had an immunoregulatory role in reducing inflammation during DENV infections in U937-DC-SIGN and THP1 macrophages infected with DENV-2 [167]. Mechanistically, the suppression of inflammatory cytokine response by VD treatment during DENV infection was mediated via the toll-like receptor (TLR) 4/NF-κB/miR-155-5p/suppressor of cytokine signaling (SOCS)-1 axis [179]. Mouse monocyte-derived macrophages differentiated in the presence of VD restrict DENV infection and moderate the classical inflammatory cytokine response caused by DENV infection [168] (Table 5). This VD-driven differentiation led to the reduced surface expression of C-type lectins, including the mannose receptor that acts as a primary receptor for the DENV attachment on macrophages [168]. Besides, macrophages, which were derived from VD-supplemented healthy donors, exhibited higher resistance to DENV infection, significantly decreased levels of pro-inflammatory cytokines, and increased levels of immunosuppressive cytokines like IL-10 [169]. High-dose VD administration also decreased DENV infection in monocyte-derived dendritic cells from 30 VD3-fed individuals [170]. However, paradoxically, low serum 25(OH)D3 concentrations in dengue fever patients reduced the pathological progress into dengue hemorrhagic fever/dengue shock syndrome [171]. In line with this, circulating VD is much higher during an acute dengue episode than during disease-free periods. [171]. Regarding the effects of VDRG polymorphisms on DENV infection, 3′ UTR haplotypes of the VDR gene were differentially associated with the risk of clinical symptoms caused by DENV infection [180].
9. Rotavirus
Rotaviruses are one of the most common causes of virus-induced diarrheal diseases among infants and young children. With respect to the effects of VD on rotavirus, physiological concentrations of 25(OH)D3 decreased porcine rotavirus replication in intestinal porcine enterocytes (IPEC-J2) cells [172] (Table 5). In addition, a study of 70 rotavirus patients suggested an association of VD deficiency with rotaviral diarrhea [173]. VD3 was also able to attenuate rotavirus infection by regulating autophagic maturation and cathelicidin gene expression in IPEC-J2 cells infected with porcine rotavirus [174]. 1,25(OH)2D3 also alleviated rotavirus infection through the miRNA-155-5p-mediated regulation of the TANK-binding kinase (TBK)1/interferon-responsive factor (IRF) 3 signaling pathway [175]. In line with this, VD supplementation alleviated intestinal damage and protected against inflammation in pigs infected with porcine epidemic diarrhea virus (PEDV) [176].
10. Human Papillomavirus (HPV)
High-risk type HPV infection is responsible for the development of cervical cancers in women. According to a study examining 2353 sexually active women, HPV prevalence was associated with VD deficiency [177] (Table 5). However, in contrast to this, Garcia-Carrasco et al. found no association between VD deficiency and the development of cervical cancers in 67 HPV-positive women with systemic lupus erythematosus [178].
11. Reasons for the Controversies and Future Research Direction
For the successful establishment of viral infection and subsequent pathogenesis in the host, a virus needs to overcome numerous hurdles that are imposed by a host immune system. Therefore, the evolution of the immune evasion strategy by a virus is an absolute requirement for its maximal survival inside the host. As shown previously, the biological functions of VD seem to be in intricate association with the many aspects of the host physiology. In particular, the non-skeletal, immune-related functions of VD begin to be explored in the context of different pathological conditions induced by viral infections [18,19,20]. The discovery of VDRs’ expression in several white blood cells [20] and the production of 1,25(OH)2D3 by local immune cells in response to infection further support the potential role of VD in the control of pathological viral infections [21]. In particular, the VD-induced production of cathelicidin is one of the best characterized immune-related functions of VD for the control of bacterial and viral infections [17,20]. Furthermore, the direct regulation of autophagy and the apoptosis of immune cells by VD seems to be one of the immune-regulatory actions of VD required for the clearance of the viral infections [18,19]. However, in spite of all this progress, our knowledge in this filed is still lagging far behind compared to the already known immunological functions of other essential nutrients, further complicating the deciphering of the cause and effect relationship of the VD biology in the development of many viral diseases [3,4,5,6,7,8,15]. On top of this, many different variables seem to serve as contributing factors to this added inconsistency and discrepancy surrounding the biological roles of VD in the context of viral infections. They include in vitro and in vivo variability, differences in experimental systems, differences in the criteria used for defining patients with a certain viral infection, and differences in clinical trial design. This might be one of the ultimate reasons for all these controversial effects of VD on the infection, pathogenesis, and treatment outcomes of several viral diseases. Therefore, we need a more complete picture of the diverse biological activities of VD in the context of different viral infections. For this goal, more systematic and thorough in vitro as well as in vivo preclinical studies should be performed before attempting the clinical application of VD. More validated markers for the VD-dependent immunological modulation will be necessary for a more accurate application of the beneficial effects of VD to virally infected patients. In particular, due to the seasonal and dietary fluctuation of the plasma VD concentrations among patients, more careful attention needs to be paid to determine the optimal VD concentrations required for the maximal beneficial effects on either the prevention or control of viral infections. For a successful translation of in vitro experimental data into in vivo, the only physiologically achievable concentrations of VD should be tested in an in vitro system. Due to the lack of coexisting immune systems in most in vitro cell experiments, more relevant immune-related functions of VD need to be explored by using a variety of animal infection models with an intact immune system. Instead of the random empirical approaches, which formed the basis for most VD clinical trials so far, the nutritional and clinical application of VD in the management of viral infections should be based on a more solid scientific rationale that will be only formed by more hypothesis-driven research in the future.
12. Conclusions
In this review, an extensive summary was generated regarding the diverse effects of VD and VDRG on the infection, pathogenesis, and treatment outcomes of several diseases caused by many clinically relevant viruses. Despite many exciting and promising results, an extensive literature review demonstrates that there is no consensus on the roles of VD and VDRG in the development and control of various diseases caused by viral infections. The full picture of the immunomodulatory functions of VD and VDRG in the context of different viral infections is still being formed. Therefore, more thorough and systematic in vitro, in vivo, and clinical studies are necessary to fully harness the potential preventive and therapeutic effects of VD and VDRG at controlling infectious diseases. Until then, it would be premature to provide recommendations on the use of VD for virally infected patients.
Author Contributions
C.L. collected all necessary references and wrote the paper. All authors have read and agree to the published version of the manuscript.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), which is funded by the Ministry of Education, Science, and Technology (NRF-2019R1F1A1058628).
References
- Bell, T.D.; Demay, M.B.; Burnett-Bowie, S.A. The biology and pathology of vitamin D control in bone. J. Cell Biochem. 2010, 111, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Wolf, G. The discovery of vitamin D: The contribution of Adolf Windaus. J. Nutr. 2004, 134, 1299–1302. [Google Scholar] [CrossRef] [PubMed]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P. Vitamin D and multiple health outcomes: umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef]
- Chung, M.; Balk, E.M.; Brendel, M.; Ip, S.; Lau, J.; Lee, J.; Lichtenstein, A.; Patel, K.; Raman, G.; Tatsioni, A.; et al. Vitamin D and calcium: A systematic review of health outcomes. Evid. Rep. Technol. Assess. (Full Rep.) 2009, 183, 1–420. [Google Scholar]
- Newberry, S.J.; Chung, M.; Shekelle, P.G.; Booth, M.S.; Liu, J.L.; Maher, A.R.; Motala, A.; Cui, M.; Perry, T.; Shanman, R.; et al. Vitamin D and Calcium: A Systematic Review of Health Outcomes (Update). Evid. Rep. Technol. Assess. (Full Rep.) 2014, 1–929. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Pittas, A.G.; Chung, M.; Trikalinos, T.; Mitri, J.; Brendel, M.; Patel, K.; Lichtenstein, A.H.; Lau, J.; Balk, E.M. Systematic review: Vitamin D and cardiometabolic outcomes. Ann. Intern. Med. 2010, 152, 307–314. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Gamble, G.D.; Reid, I.R. The effect of vitamin D supplementation on skeletal, vascular, or cancer outcomes: A trial sequential meta-analysis. Lancet Diabetes Endocrinol. 2014, 2, 307–320. [Google Scholar] [CrossRef]
- Holick, M.F.; Frommer, J.E.; McNeill, S.C.; Richtand, N.M.; Henley, J.W.; Potts, J.T., Jr. Photometabolism of 7-dehydrocholesterol to previtamin D3 in skin. Biochem. Biophys. Res. Commun. 1977, 76, 107–114. [Google Scholar] [CrossRef]
- Deluca, H.F. History of the discovery of vitamin D and its active metabolites. Bonekey Rep. 2014, 3, 479. [Google Scholar] [CrossRef]
- Fraser, D.R.; Kodicek, E. Unique biosynthesis by kidney of a biological active vitamin D metabolite. Nature 1970, 228, 764–766. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.D.; Kato, S.; Xie, W.; Mangelsdorf, D.J.; Schmidt, D.R.; Xiao, R.; Kliewer, S.A. International Union of Pharmacology. LXII. The NR1H and NR1I receptors: Constitutive androstane receptor, pregnene X receptor, farnesoid X receptor alpha, farnesoid X receptor beta, liver X receptor alpha, liver X receptor beta, and vitamin D receptor. Pharmacol. Rev. 2006, 58, 742–759. [Google Scholar] [CrossRef]
- Xie, C.N.; Yue, M.; Huang, P.; Tian, T.; Fan, H.Z.; Wu, M.P.; Yu, R.B.; Yi, H.G.; Xia, X.S.; Feng, Y.; et al. Vitamin D binding protein polymorphisms influence susceptibility to hepatitis C virus infection in a high-risk Chinese population. Gene 2018, 679, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.N.; Ahmad, S.N.; Ahmad, N. HIV Infection and Bone Abnormalities. Open Orthop. J. 2017, 11, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.A.; Bearden, A.; Striker, R. Vitamin D and the anti-viral state. J. Clin. Virol. 2011, 50, 194–200. [Google Scholar] [CrossRef]
- Gois, P.H.F.; Ferreira, D.; Olenski, S.; Seguro, A.C. Vitamin D and Infectious Diseases: Simple Bystander or Contributing Factor? Nutrients 2017, 9. [Google Scholar] [CrossRef]
- Lang, P.O.; Samaras, N.; Samaras, D.; Aspinall, R. How important is vitamin D in preventing infections? Osteoporos. Int. 2013, 24, 1537–1553. [Google Scholar] [CrossRef]
- Teymoori-Rad, M.; Shokri, F.; Salimi, V.; Marashi, S.M. The interplay between vitamin D and viral infections. Rev. Med. Virol. 2019, 29, e2032. [Google Scholar] [CrossRef]
- Watkins, R.R.; Lemonovich, T.L.; Salata, R.A. An update on the association of vitamin D deficiency with common infectious diseases. Can. J. Physiol. Pharmacol. 2015, 93, 363–368. [Google Scholar] [CrossRef]
- Khoo, A.L.; Chai, L.; Koenen, H.; Joosten, I.; Netea, M.; van der Ven, A. Translating the role of vitamin D3 in infectious diseases. Crit. Rev. Microbiol. 2012, 38, 122–135. [Google Scholar] [CrossRef] [PubMed]
- Shepard, C.W.; Finelli, L.; Alter, M.J. Global epidemiology of hepatitis C virus infection. Lancet Infect. Dis. 2005, 5, 558–567. [Google Scholar] [CrossRef]
- Gal-Tanamy, M.; Bachmetov, L.; Ravid, A.; Koren, R.; Erman, A.; Tur-Kaspa, R.; Zemel, R. Vitamin D: An innate antiviral agent suppressing hepatitis C virus in human hepatocytes. Hepatology 2011, 54, 1570–1579. [Google Scholar] [CrossRef] [PubMed]
- Murayama, A.; Saitoh, H.; Takeuchi, A.; Yamada, N.; Matsumura, T.; Shiina, M.; Muramatsu, M.; Wakita, T.; Imawari, M.; Kato, T. Vitamin D derivatives inhibit hepatitis C virus production through the suppression of apolipoprotein. Antiviral. Res. 2018, 160, 55–63. [Google Scholar] [CrossRef]
- Ravid, A.; Rapaport, N.; Issachar, A.; Erman, A.; Bachmetov, L.; Tur-Kaspa, R.; Zemel, R. 25-Hydroxyvitamin D Inhibits Hepatitis C Virus Production in Hepatocellular Carcinoma Cell Line by a Vitamin D Receptor-Independent Mechanism. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Matsumura, T.; Kato, T.; Sugiyama, N.; Tasaka-Fujita, M.; Murayama, A.; Masaki, T.; Wakita, T.; Imawari, M. 25-Hydroxyvitamin D3 suppresses hepatitis C virus production. Hepatology 2012, 56, 1231–1239. [Google Scholar] [CrossRef]
- Gutierrez, J.A.; Jones, K.A.; Flores, R.; Singhania, A.; Woelk, C.H.; Schooley, R.T.; Wyles, D.L. Vitamin D Metabolites Inhibit Hepatitis C Virus and Modulate Cellular Gene Expression. J. Virol. Antivir. Res. 2014, 3. [Google Scholar] [CrossRef]
- Lin, Y.M.; Sun, H.Y.; Chiu, W.T.; Su, H.C.; Chien, Y.C.; Chong, L.W.; Chang, H.C.; Bai, C.H.; Young, K.C.; Tsao, C.W. Calcitriol Inhibits HCV Infection via Blockade of Activation of PPAR and Interference with Endoplasmic Reticulum-Associated Degradation. Viruses 2018, 10. [Google Scholar] [CrossRef]
- Saleh, M.; Welsch, C.; Cai, C.; Doring, C.; Gouttenoire, J.; Friedrich, J.; Haselow, K.; Sarrazin, C.; Badenhoop, K.; Moradpour, D.; et al. Differential modulation of hepatitis C virus replication and innate immune pathways by synthetic calcitriol-analogs. J. Steroid. Biochem. Mol. Biol. 2018, 183, 142–151. [Google Scholar] [CrossRef]
- Lange, C.M.; Gouttenoire, J.; Duong, F.H.; Morikawa, K.; Heim, M.H.; Moradpour, D. Vitamin D receptor and Jak-STAT signaling crosstalk results in calcitriol-mediated increase of hepatocellular response to IFN-alpha. J. Immunol. 2014, 192, 6037–6044. [Google Scholar] [CrossRef]
- Hii, C.S.; Ferrante, A. The Non-Genomic Actions of Vitamin D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- El Husseiny, N.M.; Fahmy, H.M.; Mohamed, W.A.; Amin, H.H. Relationship between vitamin D and IL-23, IL-17 and macrophage chemoattractant protein-1 as markers of fibrosis in hepatitis C virus Egyptians. World J. Hepatol. 2012, 4, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Schaalan, M.F.; Mohamed, W.A.; Amin, H.H. Vitamin D deficiency: Correlation to interleukin-17, interleukin-23 and PIIINP in hepatitis C virus genotype 4. World J. Gastroenterol. 2012, 18, 3738–3744. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Yue, M.; Huang, P.; Zhang, Y.; Xie, C.; Yu, R.; Li, J.; Wang, J. Vitamin D level and vitamin D receptor genetic variations contribute to HCV infection susceptibility and chronicity in a Chinese population. Infect. Genet. Evol. 2016, 41, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mohsen, M.A.; El-Braky, A.A.; Ghazal, A.A.E.; Shamseya, M.M. Autophagy, apoptosis, vitamin D, and vitamin D receptor in hepatocellular carcinoma associated with hepatitis C virus. Medicine (Baltimore) 2018, 97, e0172. [Google Scholar] [CrossRef]
- Berkan-Kawinska, A.; Koslinska-Berkan, E.; Piekarska, A. The prevalence and severity of 25-(OH)-vitamin D insufficiency in HCV infected and in HBV infected patients: A prospective study. Clin. Exp. Hepatol. 2015, 1, 5–11. [Google Scholar] [CrossRef]
- Amanzada, A.; Goralczyk, A.D.; Moriconi, F.; van Thiel, D.H.; Ramadori, G.; Mihm, S. Vitamin D status and serum ferritin concentration in chronic hepatitis C virus type 1 infection. J. Med. Virol. 2013, 85, 1534–1541. [Google Scholar] [CrossRef]
- Avihingsanon, A.; Jitmitraparp, S.; Tangkijvanich, P.; Ramautarsing, R.A.; Apornpong, T.; Jirajariyavej, S.; Putcharoen, O.; Treeprasertsuk, S.; Akkarathamrongsin, S.; Poovorawan, Y.; et al. Advanced liver fibrosis by transient elastography, fibrosis 4, and alanine aminotransferase/platelet ratio index among Asian hepatitis C with and without human immunodeficiency virus infection: Role of vitamin D levels. J. Gastroenterol. Hepatol. 2014, 29, 1706–1714. [Google Scholar] [CrossRef]
- Mandorfer, M.; Payer, B.A.; Schwabl, P.; Steiner, S.; Ferlitsch, A.; Aichelburg, M.C.; Stattermayer, A.F.; Ferenci, P.; Obermayer-Pietsch, B.; Grabmeier-Pfistershammer, K.; et al. Revisiting liver disease progression in HIV/HCV-coinfected patients: The influence of vitamin D, insulin resistance, immune status, IL28B and PNPLA3. Liver Int. 2015, 35, 876–885. [Google Scholar] [CrossRef]
- De Almeida, J.P.; Liberatti, L.S.; Barros, F.E.; Kallaur, A.P.; Lozovoy, M.A.; Scavuzzi, B.M.; Panis, C.; Reiche, E.M.; Dichi, I.; Simao, A.N. Profile of oxidative stress markers is dependent on vitamin D levels in patients with chronic hepatitis C. Nutrition 2016, 32, 362–367. [Google Scholar] [CrossRef]
- Mohamed, A.A.; Abd Almonaem, E.R.; Mansour, A.I.; Algebaly, H.F.; Khattab, R.A.; El Abd, Y.S. Importance of Studying the Levels of Hepcidin and Vitamin D in Egyptian Children with Chronic Hepatitis C. J. Transl. Int. Med. 2019, 7, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Gentile, I.; Scarano, F.; Celotti, A.; De Iuliis, E.; Scarano, R.; Granata, V.; Pinchera, B.; Meola, M.; D’Ambra, A.; Piccirillo, M.; et al. Low vitamin D levels are associated with the presence of serum cryoglobulins in patients with chronic HCV infection. In Vivo 2015, 29, 399–404. [Google Scholar] [PubMed]
- Yousif, M.M.; Sadek, A.; Farrag, H.A.; Selim, F.O.; Hamed, E.F.; Salama, R.I. Associated vitamin D deficiency is a risk factor for the complication of HCV-related liver cirrhosis including hepatic encephalopathy and spontaneous bacterial peritonitis. Intern. Emerg. Med. 2019, 14, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.M.; Miki, D.; Ochi, H.; Nischalke, H.D.; Bojunga, J.; Bibert, S.; Morikawa, K.; Gouttenoire, J.; Cerny, A.; Dufour, J.F.; et al. Genetic analyses reveal a role for vitamin D insufficiency in HCV-associated hepatocellular carcinoma development. PLoS ONE 2013, 8, e64053. [Google Scholar] [CrossRef]
- Bitetto, D.; Fabris, C.; Fornasiere, E.; Pipan, C.; Fumolo, E.; Cussigh, A.; Bignulin, S.; Cmet, S.; Fontanini, E.; Falleti, E.; et al. Vitamin D supplementation improves response to antiviral treatment for recurrent hepatitis C. Transpl. Int. 2011, 24, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Nimer, A.; Mouch, A. Vitamin D improves viral response in hepatitis C genotype 2-3 naive patients. World J. Gastroenterol. 2012, 18, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Mandorfer, M.; Reiberger, T.; Payer, B.A.; Ferlitsch, A.; Breitenecker, F.; Aichelburg, M.C.; Obermayer-Pietsch, B.; Rieger, A.; Trauner, M.; Peck-Radosavljevic, M.; et al. Low vitamin D levels are associated with impaired virologic response to PEGIFN + RBV therapy in HIV-hepatitis C virus coinfected patients. AIDS 2013, 27, 227–232. [Google Scholar] [CrossRef]
- Atsukawa, M.; Tsubota, A.; Shimada, N.; Abe, H.; Kondo, C.; Itokawa, N.; Nakagawa, A.; Iwakiri, K.; Kawamoto, C.; Aizawa, Y.; et al. Serum 25(OH)D3 levels affect treatment outcomes for telaprevir/peg-interferon/ribavirin combination therapy in genotype 1b chronic hepatitis C. Dig. Liver Dis. 2014, 46, 738–743. [Google Scholar] [CrossRef]
- Abu-Mouch, S.; Fireman, Z.; Jarchovsky, J.; Zeina, A.R.; Assy, N. Vitamin D supplementation improves sustained virologic response in chronic hepatitis C (genotype 1)-naive patients. World J. Gastroenterol. 2011, 17, 5184–5190. [Google Scholar] [CrossRef]
- Yokoyama, S.; Takahashi, S.; Kawakami, Y.; Hayes, C.N.; Kohno, H.; Kohno, H.; Tsuji, K.; Aisaka, Y.; Kira, S.; Yamashina, K.; et al. Effect of vitamin D supplementation on pegylated interferon/ribavirin therapy for chronic hepatitis C genotype 1b: A randomized controlled trial. J. Viral Hepat. 2014, 21, 348–356. [Google Scholar] [CrossRef]
- Eltayeb, A.A.; Abdou, M.A.; Abdel-Aal, A.M.; Othman, M.H. Vitamin D status and viral response to therapy in hepatitis C infected children. World J. Gastroenterol. 2015, 21, 1284–1291. [Google Scholar] [CrossRef]
- Omori-Mizuno, Y.; Nakayama, N.; Inao, M.; Funyu, J.; Asabe, S.; Tomita, K.; Nishikawa, K.; Hosoda, Y.; Tanaka, M.; Hashimoto, Y.; et al. Randomized study comparing vitamin D3 and 1alpha-Hydroxyvitamin D3 in combination with pegylated interferon/ribavirin therapy for chronic hepatitis C. J. Gastroenterol. Hepatol. 2015, 30, 1384–1390. [Google Scholar] [CrossRef]
- Komolmit, P.; Kimtrakool, S.; Suksawatamnuay, S.; Thanapirom, K.; Chattrasophon, K.; Thaimai, P.; Chirathaworn, C.; Poovorawan, Y. Vitamin D supplementation improves serum markers associated with hepatic fibrogenesis in chronic hepatitis C patients: A randomized, double-blind, placebo-controlled study. Sci. Rep. 2017, 7, 8905. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Liu, M.; Zhao, J.; Ren, F.; Chen, Y.; Li, J.F.; Zhang, J.Y.; Qu, F.; Zhang, J.L.; Duan, Z.P.; et al. Serum vitamin D(3) does not correlate with liver fibrosis in chronic hepatitis C. World J. Gastroenterol. 2015, 21, 11152–11159. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, K.S.; Buss, C.; Tovo, C.V. Is there an association between vitamin D and liver fibrosis in patients with chronic hepatitis C? Arq. Gastroenterol. 2017, 54, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Kitson, M.T.; Dore, G.J.; George, J.; Button, P.; McCaughan, G.W.; Crawford, D.H.; Sievert, W.; Weltman, M.D.; Cheng, W.S.; Roberts, S.K. Vitamin D status does not predict sustained virologic response or fibrosis stage in chronic hepatitis C genotype 1 infection. J. Hepatol. 2013, 58, 467–472. [Google Scholar] [CrossRef]
- Grammatikos, G.; Lange, C.; Susser, S.; Schwendy, S.; Dikopoulos, N.; Buggisch, P.; Encke, J.; Teuber, G.; Goeser, T.; Thimme, R.; et al. Vitamin D levels vary during antiviral treatment but are unable to predict treatment outcome in HCV genotype 1 infected patients. PLoS ONE 2014, 9, e87974. [Google Scholar] [CrossRef]
- Loftfield, E.; O’Brien, T.R.; Pfeiffer, R.M.; Howell, C.D.; Horst, R.; Prokunina-Olsson, L.; Weinstein, S.J.; Albanes, D.; Morgan, T.R.; Freedman, N.D. Vitamin D Status and Virologic Response to HCV Therapy in the HALT-C and VIRAHEP-C Trials. PLoS ONE 2016, 11, e0166036. [Google Scholar] [CrossRef]
- Esmat, G.; El Raziky, M.; Elsharkawy, A.; Sabry, D.; Hassany, M.; Ahmed, A.; Assem, N.; El Kassas, M.; Doss, W. Impact of vitamin D supplementation on sustained virological response in chronic hepatitis C genotype 4 patients treated by pegylated interferon/ribavirin. J. Interferon Cytokine Res. 2015, 35, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Terrier, B.; Lapidus, N.; Pol, S.; Serfaty, L.; Ratziu, V.; Asselah, T.; Thibault, V.; Souberbielle, J.C.; Carrat, F.; Cacoub, P. Vitamin D in addition to peg-interferon-alpha/ribavirin in chronic hepatitis C virus infection: ANRS-HC25-VITAVIC study. World J. Gastroenterol. 2015, 21, 5647–5653. [Google Scholar] [CrossRef]
- Ladero, J.M.; Torrejón, M.J.; Sánchez-Pobre, P.; Suárez, A.; Cuenca, F.; de la Orden, V.; Devesa, M.J.; Rodrigo, M.; Estrada, V.; López-Alonso, G.; et al. Vitamin D deficiency and vitamin D therapy in chronic hepatitis C. Ann. Hepatol. 2013, 12, 199–204. [Google Scholar] [CrossRef]
- Gutierrez, J.A.; Parikh, N.; Branch, A.D. Classical and emerging roles of vitamin D in hepatitis C virus infection. Semin. Liver Dis. 2011, 31, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Gabr, S.A.; Alghadir, A.H.; Allam, A.A.; Ajarem, J.; Al-Basher, G.; Abdel-Maksoud, M.A.; Ghfar, A.A.; Aboud, A. Correlation between vitamin D levels and apoptosis in geriatric patients infected with hepatitis C virus genotype 4. Clin. Interv. Aging 2016, 11, 523–533. [Google Scholar] [CrossRef]
- Behera, M.K.; Shukla, S.K.; Dixit, V.K.; Nath, P.; Abhilash, V.B.; Asati, P.K.; Jain, A.K. Effect of vitamin D supplementation on sustained virological response in genotype 1/4 chronic hepatitis C treatment-naive patients from India. Indian J. Med. Res. 2018, 148, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.Q.; Wu, X.X.; Ling, Z.X.; Cheng, Y.W.; Yuan, L.; Xiang, C. Association between serum vitamin D and severity of liver fibrosis in chronic hepatitis C patients: A systematic meta-analysis. J. Zhejiang Univ. Sci. B 2014, 15, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Alvarez, M.; Pineda-Tenor, D.; Jimenez-Sousa, M.A.; Fernandez-Rodriguez, A.; Guzman-Fulgencio, M.; Resino, S. Relationship of vitamin D status with advanced liver fibrosis and response to hepatitis C virus therapy: A meta-analysis. Hepatology 2014, 60, 1541–1550. [Google Scholar] [CrossRef]
- Dadabhai, A.S.; Saberi, B.; Lobner, K.; Shinohara, R.T.; Mullin, G.E. Influence of vitamin D on liver fibrosis in chronic hepatitis C: A systematic review and meta-analysis of the pooled clinical trials data. World J. Hepatol. 2017, 9, 278–287. [Google Scholar] [CrossRef]
- De Azevedo, L.A.; Matte, U.; da Silveira, T.R.; Alvares-da-Silva, M.R. Genetic variants underlying vitamin D metabolism and VDR-TGFbeta-1-SMAD3 interaction may impact on HCV progression: A study based on dbGaP data from the HALT-C study. J. Hum. Genet. 2017, 62, 969–977. [Google Scholar] [CrossRef]
- Villar, L.M.; Del Campo, J.A.; Ranchal, I.; Lampe, E.; Romero-Gomez, M. Association between vitamin D and hepatitis C virus infection: A meta-analysis. World J. Gastroenterol. 2013, 19, 5917–5924. [Google Scholar] [CrossRef]
- Huang, J.F.; Ko, Y.M.; Huang, C.F.; Yeh, M.L.; Dai, C.Y.; Hsieh, M.H.; Huang, C.I.; Yang, H.L.; Wang, S.C.; Lin, Z.Y.; et al. 25-Hydroxy vitamin D suppresses hepatitis C virus replication and contributes to rapid virological response of treatment efficacy. Hepatol. Res. 2017, 47, 1383–1389. [Google Scholar] [CrossRef]
- Kim, H.B.; Myung, S.K.; Lee, Y.J.; Park, B.J.; Korean Meta-Analysis Study Group. Efficacy of vitamin D supplementation in combination with conventional antiviral therapy in patients with chronic hepatitis C infection: A meta-analysis of randomised controlled trials. J. Hum. Nutr. Diet. 2018, 31, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Kitson, M.T.; Sarrazin, C.; Toniutto, P.; Eslick, G.D.; Roberts, S.K. Vitamin D level and sustained virologic response to interferon-based antiviral therapy in chronic hepatitis C: A systematic review and meta-analysis. J. Hepatol. 2014, 61, 1247–1252. [Google Scholar] [CrossRef]
- Weintraub, S.J.; Fleckenstein, J.F.; Marion, T.N.; Madey, M.A.; Mahmoudi, T.M.; Schechtman, K.B. Vitamin D and the racial difference in the genotype 1 chronic hepatitis C treatment response. Am. J. Clin. Nutr. 2012, 96, 1025–1031. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hung, C.H.; Hu, T.H.; Lu, S.N.; Chen, C.H.; Wang, J.H.; Lee, C.M. Association of vitamin D receptor gene polymorphisms with response to peginterferon plus ribavirin in Asian patients with chronic hepatitis C. J. Formos. Med. Assoc. 2016, 115, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Ho, A.S.; Cheng, C.C.; Lee, S.C.; Liu, M.L.; Lee, J.Y.; Wang, W.M.; Wang, C.C. Novel biomarkers predict liver fibrosis in hepatitis C patients: alpha 2 macroglobulin, vitamin D binding protein and apolipoprotein AI. J. Biomed. Sci. 2010, 17, 58. [Google Scholar] [CrossRef] [PubMed]
- Scalioni, L.P.; Santos, B.R.D.; Spritzer, P.M.; Villela-Nogueira, C.A.; Laura Lewis-Ximenez, L.; Pollo-Flores, P.; Bordalo Cathala Esberard, E.; Brandao-Mello, C.E.; Lampe, E.; Villar, L.M. Impact of vitamin D receptor and binding protein gene polymorphisms in clinical and laboratory data of HCV patients: Cross sectional study. Medicine (Baltimore) 2018, 97, e9881. [Google Scholar] [CrossRef]
- Barooah, P.; Saikia, S.; Bharadwaj, R.; Sarmah, P.; Bhattacharyya, M.; Goswami, B.; Medhi, S. Role of VDR, GC, and CYP2R1 Polymorphisms in the Development of Hepatocellular Carcinoma in Hepatitis C Virus-Infected Patients. Genet. Test. Mol. Biomarkers 2019, 23, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Thanapirom, K.; Suksawatamnuay, S.; Sukeepaisarnjaroen, W.; Tangkijvanich, P.; Thaimai, P.; Wasitthankasem, R.; Poovorawan, Y.; Komolmit, P. Genetic associations of vitamin D receptor polymorphisms with advanced liver fibrosis and response to pegylated interferon-based therapy in chronic hepatitis C. Peer J. 2019, 7, e7666. [Google Scholar] [CrossRef]
- Barchetta, I.; Carotti, S.; Labbadia, G.; Gentilucci, U.V.; Muda, A.O.; Angelico, F.; Silecchia, G.; Leonetti, F.; Fraioli, A.; Picardi, A.; et al. Liver vitamin D receptor, CYP2R1, and CYP27A1 expression: Relationship with liver histology and vitamin D3 levels in patients with nonalcoholic steatohepatitis or hepatitis C virus. Hepatology 2012, 56, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, L.A.; Matte, U.; Silveira, T.R.; Bonfanti, J.W.; Bruch, J.P.; Álvares-da-Silva, M.R. Effect of Vitamin D Serum Levels and GC Gene Polymorphisms in Liver Fibrosis Due to Chronic Hepatitis C. Ann. Hepatol. 2017, 16, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Cusato, J.; Allegra, S.; Boglione, L.; De Nicolo, A.; Baietto, L.; Cariti, G.; Di Perri, G.; D’Avolio, A. Vitamin D pathway gene variants and HCV-2/3 therapy outcomes. Antivir. Ther. 2015, 20, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.M.; Bojunga, J.; Ramos-Lopez, E.; von Wagner, M.; Hassler, A.; Vermehren, J.; Herrmann, E.; Badenhoop, K.; Zeuzem, S.; Sarrazin, C. Vitamin D deficiency and a CYP27B1-1260 promoter polymorphism are associated with chronic hepatitis C and poor response to interferon-alfa based therapy. J. Hepatol. 2011, 54, 887–893. [Google Scholar] [CrossRef]
- Falleti, E.; Bitetto, D.; Fabris, C.; Fattovich, G.; Cussigh, A.; Cmet, S.; Ceriani, E.; Fornasiere, E.; Pasino, M.; Ieluzzi, D.; et al. Vitamin D binding protein gene polymorphisms and baseline vitamin D levels as predictors of antiviral response in chronic hepatitis C. Hepatology 2012, 56, 1641–1650. [Google Scholar] [CrossRef] [PubMed]
- Petta, S.; Grimaudo, S.; Marco, V.D.; Scazzone, C.; Macaluso, F.S.; Camma, C.; Cabibi, D.; Pipitone, R.; Craxi, A. Association of vitamin D serum levels and its common genetic determinants, with severity of liver fibrosis in genotype 1 chronic hepatitis C patients. J. Viral. Hepat. 2013, 20, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Thanapirom, K.; Suksawatamnuay, S.; Sukeepaisarnjaroen, W.; Tangkijvanich, P.; Treeprasertsuk, S.; Thaimai, P.; Wasitthankasem, R.; Poovorawan, Y.; Komolmit, P. Vitamin D-related gene polymorphism predict treatment response to pegylated interferon-based therapy in Thai chronic hepatitis C patients. BMC Gastroenterol. 2017, 17, 54. [Google Scholar] [CrossRef]
- Schogler, A.; Muster, R.J.; Kieninger, E.; Casaulta, C.; Tapparel, C.; Jung, A.; Moeller, A.; Geiser, T.; Regamey, N.; Alves, M.P. Vitamin D represses rhinovirus replication in cystic fibrosis cells by inducing LL-37. Eur. Respir. J. 2016, 47, 520–530. [Google Scholar] [CrossRef]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antivir. Res. 2017, 137, 93–101. [Google Scholar] [CrossRef]
- Greiller, C.L.; Suri, R.; Jolliffe, D.A.; Kebadze, T.; Hirsman, A.G.; Griffiths, C.J.; Johnston, S.L.; Martineau, A.R. Vitamin D attenuates rhinovirus-induced expression of intercellular adhesion molecule-1 (ICAM-1) and platelet-activating factor receptor (PAFR) in respiratory epithelial cells. J. Steroid Biochem. Mol. Biol. 2019, 187, 152–159. [Google Scholar] [CrossRef]
- Hansdottir, S.; Monick, M.M.; Lovan, N.; Powers, L.; Gerke, A.; Hunninghake, G.W. Vitamin D decreases respiratory syncytial virus induction of NF-kappaB-linked chemokines and cytokines in airway epithelium while maintaining the antiviral state. J. Immunol. 2010, 184, 965–974. [Google Scholar] [CrossRef]
- Stoppelenburg, A.J.; von Hegedus, J.H.; Huis in’t Veld, R.; Bont, L.; Boes, M. Defective control of vitamin D receptor-mediated epithelial STAT1 signalling predisposes to severe respiratory syncytial virus bronchiolitis. J. Pathol. 2014, 232, 57–64. [Google Scholar] [CrossRef]
- Brockman-Schneider, R.A.; Pickles, R.J.; Gern, J.E. Effects of vitamin D on airway epithelial cell morphology and rhinovirus replication. PLoS ONE 2014, 9, e86755. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T. Vitamin D and Lung Infection. Infect. Immun. 2016, 84, 3094–3096. [Google Scholar] [CrossRef] [PubMed]
- Fitch, N.; Becker, A.B.; HayGlass, K.T. Vitamin D [1,25(OH)2D3] Differentially Regulates Human Innate Cytokine Responses to Bacterial versus Viral Pattern Recognition Receptor Stimuli. J. Immunol. 2016, 196, 2965–2972. [Google Scholar] [CrossRef] [PubMed]
- Bodin, J.; Mihret, A.; Holm-Hansen, C.; Dembinski, J.L.; Trieu, M.C.; Tessema, B.; Tarekegne, A.; Yimer, S.A.; Cox, R.; Aseffa, A.; et al. Vitamin D Deficiency is Associated with Increased Use of Antimicrobials among Preschool Girls in Ethiopia. Nutrients 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, J.L.; Jones, B.G.; Penkert, R.R.; Gansebom, S.; Sun, Y.; Tang, L.; Bramley, A.M.; Jain, S.; McCullers, J.A.; Arnold, S.R. Low Retinol-Binding Protein and Vitamin D Levels Are Associated with Severe Outcomes in Children Hospitalized with Lower Respiratory Tract Infection and Respiratory Syncytial Virus or Human Metapneumovirus Detection. J. Pediatr. 2017, 187, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Stelmach, I.; Majak, P.; Jerzynska, J.; Podlecka, D.; Stelmach, W.; Polanska, K.; Gromadzinska, J.; Wasowicz, W.; Hanke, W. Cord serum 25-hydroxyvitamin D correlates with early childhood viral-induced wheezing. Respir. Med. 2015, 109, 38–43. [Google Scholar] [CrossRef]
- Jones, B.G.; Oshansky, C.M.; Bajracharya, R.; Tang, L.; Sun, Y.; Wong, S.S.; Webby, R.; Thomas, P.G.; Hurwitz, J.L. Retinol binding protein and vitamin D associations with serum antibody isotypes, serum influenza virus-specific neutralizing activities and airway cytokine profiles. Clin. Exp. Immunol. 2016, 183, 239–247. [Google Scholar] [CrossRef]
- Cooper, C.; Thorne, A.; Canadian Hiv Trials Network Ctn Influenza Vaccine Research Group. Vitamin D supplementation does not increase immunogenicity of seasonal influenza vaccine in HIV-infected adults. HIV Clin. Trials 2011, 12, 275–276. [Google Scholar] [CrossRef]
- Beigelman, A.; Castro, M.; Schweiger, T.L.; Wilson, B.S.; Zheng, J.; Yin-DeClue, H.; Sajol, G.; Giri, T.; Sierra, O.L.; Isaacson-Schmid, M.; et al. Vitamin D Levels Are Unrelated to the Severity of Respiratory Syncytial Virus Bronchiolitis Among Hospitalized Infants. J. Pediatr. Infect. Dis. Soc. 2015, 4, 182–188. [Google Scholar] [CrossRef]
- Maxwell, C.S.; Carbone, E.T.; Wood, R.J. Better newborn vitamin D status lowers RSV-associated bronchiolitis in infants. Nutr. Rev. 2012, 70, 548–552. [Google Scholar] [CrossRef]
- Zdrenghea, M.T.; Makrinioti, H.; Bagacean, C.; Bush, A.; Johnston, S.L.; Stanciu, L.A. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev. Med. Virol. 2017, 27. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.D.; Lin, C.H.; Lei, W.T.; Chang, H.Y.; Lee, H.C.; Yeung, C.Y.; Chiu, N.C.; Chi, H.; Liu, J.M.; Hsu, R.J.; et al. Does Vitamin D Deficiency Affect the Immunogenic Responses to Influenza Vaccination? A Systematic Review and Meta-Analysis. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Bryson, K.J.; Nash, A.A.; Norval, M. Does vitamin D protect against respiratory viral infections? Epidemiol. Infect. 2014, 142, 1789–1801. [Google Scholar] [CrossRef] [PubMed]
- Yakoob, M.Y.; Salam, R.A.; Khan, F.R.; Bhutta, Z.A. Vitamin D supplementation for preventing infections in children under five years of age. Cochrane Database Syst. Rev. 2016, 11, CD008824. [Google Scholar] [CrossRef] [PubMed]
- Laplana, M.; Royo, J.L.; Fibla, J. Vitamin D Receptor polymorphisms and risk of enveloped virus infection: A meta-analysis. Gene 2018, 678, 384–394. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Greiller, C.L.; Mein, C.A.; Hoti, M.; Bakhsoliani, E.; Telcian, A.G.; Simpson, A.; Barnes, N.C.; Curtin, J.A.; Custovic, A.; et al. Vitamin D receptor genotype influences risk of upper respiratory infection. Br. J. Nutr. 2018, 120, 891–900. [Google Scholar] [CrossRef]
- McNally, J.D.; Sampson, M.; Matheson, L.A.; Hutton, B.; Little, J. Vitamin D receptor (VDR) polymorphisms and severe RSV bronchiolitis: A systematic review and meta-analysis. Pediatr. Pulmonol. 2014, 49, 790–799. [Google Scholar] [CrossRef]
- Randolph, A.G.; Yip, W.K.; Falkenstein-Hagander, K.; Weiss, S.T.; Janssen, R.; Keisling, S.; Bont, L. Vitamin D-binding protein haplotype is associated with hospitalization for RSV bronchiolitis. Clin. Exp. Allergy 2014, 44, 231–237. [Google Scholar] [CrossRef]
- Ott, J.J.; Stevens, G.A.; Groeger, J.; Wiersma, S.T. Global epidemiology of hepatitis B virus infection: new estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine 2012, 30, 2212–2219. [Google Scholar] [CrossRef]
- Lee, W.M. Hepatitis B virus infection. N. Engl. J. Med. 1997, 337, 1733–1745. [Google Scholar] [CrossRef]
- Block, T.M.; Rawat, S.; Brosgart, C.L. Chronic hepatitis B: A wave of new therapies on the horizon. Antivir. Res. 2015, 121, 69–81. [Google Scholar] [CrossRef]
- Shlomai, A.; Rice, C.M. Virology. Getting rid of a persistent troublemaker to cure hepatitis. Science 2014, 343, 1212–1213. [Google Scholar] [CrossRef]
- Hu, Y.C.; Wang, W.W.; Jiang, W.Y.; Li, C.Q.; Guo, J.C.; Xun, Y.H. Low vitamin D levels are associated with high viral loads in patients with chronic hepatitis B: A systematic review and meta-analysis. BMC Gastroenterol. 2019, 19, 84. [Google Scholar] [CrossRef] [PubMed]
- Mohamadkhani, A.; Bastani, F.; Khorrami, S.; Ghanbari, R.; Eghtesad, S.; Sharafkhah, M.; Montazeri, G.; Poustchi, H. Negative Association of Plasma Levels of Vitamin D and miR-378 With Viral Load in Patients With Chronic Hepatitis B Infection. Hepat. Mon. 2015, 15, e28315. [Google Scholar] [CrossRef]
- Mahamid, M.; Nseir, W.; Abu Elhija, O.; Shteingart, S.; Mahamid, A.; Smamra, M.; Koslowsky, B. Normal vitamin D levels are associated with spontaneous hepatitis B surface antigen seroclearance. World J. Hepatol. 2013, 5, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Hoan, N.X.; Khuyen, N.; Binh, M.T.; Giang, D.P.; Van Tong, H.; Hoan, P.Q.; Trung, N.T.; Anh, D.T.; Toan, N.L.; Meyer, C.G.; et al. Association of vitamin D deficiency with hepatitis B virus—Related liver diseases. BMC Infect. Dis. 2016, 16, 507. [Google Scholar] [CrossRef] [PubMed]
- Gotlieb, N.; Tachlytski, I.; Lapidot, Y.; Sultan, M.; Safran, M.; Ben-Ari, Z. Hepatitis B virus downregulates vitamin D receptor levels in hepatoma cell lines, thereby preventing vitamin D-dependent inhibition of viral transcription and production. Mol. Med. 2018, 24, 53. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Q.; Bai, L.; Zhou, T.Y.; Fe, M.; Zhang, D.M.; Tang, H. Sustained suppression of viral replication in improving vitamin D serum concentrations in patients with chronic hepatitis B. Sci. Rep. 2015, 5, 15441. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Tan, D.; Ning, Q.; Niu, J.; Bai, X.; Chen, S.; Cheng, J.; Yu, Y.; Wang, H.; Xu, M.; et al. Association of baseline vitamin D level with genetic determinants and virologic response in patients with chronic hepatitis B. Hepatol. Res. 2018, 48, E213–E221. [Google Scholar] [CrossRef]
- Bi, Y.; Zhang, W.; Sun, J.; Gao, R. Therapeutic and immune function improvement of vitamin D combined with IFN-alpha on mouse with hepatitis B infection. Int. J. Immunopathol. Pharmacol. 2018, 32. [Google Scholar] [CrossRef] [PubMed]
- Sali, S.; Tavakolpour, S.; Farkhondemehr, B. Comparison of Vitamin D Levels in Naive, Treated, and Inactive Carriers with Chronic Hepatitis B Virus. J. Clin. Transl. Hepatol. 2016, 4, 306–309. [Google Scholar] [CrossRef] [PubMed]
- Parfieniuk-Kowerda, A.; Swiderska, M.; Rogalska, M.; Maciaszek, M.; Jaroszewicz, J.; Flisiak, R. Chronic hepatitis B virus infection is associated with decreased serum 25(OH)D concentration in non-cirrhotic patients. Clin. Exp. Hepatol. 2019, 5, 75–80. [Google Scholar] [CrossRef]
- Zhao, X.Y.; Li, J.; Wang, J.H.; Habib, S.; Wei, W.; Sun, S.J.; Strobel, H.W.; Jia, J.D. Vitamin D serum level is associated with Child-Pugh score and metabolic enzyme imbalances, but not viral load in chronic hepatitis B patients. Medicine (Baltimore) 2016, 95, e3926. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.L.; Elkhashab, M.; Trinh, H.; Tak, W.Y.; Ma, X.; Chuang, W.L.; Kim, Y.J.; Martins, E.B.; Lin, L.; Dinh, P.; et al. Association of baseline vitamin D levels with clinical parameters and treatment outcomes in chronic hepatitis B. J. Hepatol. 2015, 63, 1086–1092. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Sun, J.; Zheng, Z.; Chen, J.; Fan, R.; Liang, X.; Zhu, Y.; Liu, Y.; Shen, S.; Hou, J. Association between vitamin D level and viral load or fibrosis stage in chronic hepatitis B patients from Southern China. J. Gastroenterol. Hepatol. 2015, 30, 566–574. [Google Scholar] [CrossRef]
- Bellamy, R.; Ruwende, C.; Corrah, T.; McAdam, K.P.; Thursz, M.; Whittle, H.C.; Hill, A.V. Tuberculosis and chronic hepatitis B virus infection in Africans and variation in the vitamin D receptor gene. J. Infect. Dis. 1999, 179, 721–724. [Google Scholar] [CrossRef]
- He, Q.; Huang, Y.; Zhang, L.; Yan, Y.; Liu, J.; Song, X.; Chen, W. Association between vitamin D receptor polymorphisms and hepatitis B virus infection susceptibility: A meta-analysis study. Gene 2018, 645, 105–112. [Google Scholar] [CrossRef]
- Huang, Y.W.; Liao, Y.T.; Chen, W.; Chen, C.L.; Hu, J.T.; Liu, C.J.; Lai, M.Y.; Chen, P.J.; Chen, D.S.; Yang, S.S.; et al. Vitamin D receptor gene polymorphisms and distinct clinical phenotypes of hepatitis B carriers in Taiwan. Genes Immun. 2010, 11, 87–93. [Google Scholar] [CrossRef]
- Hoan, N.X.; Khuyen, N.; Giang, D.P.; Binh, M.T.; Toan, N.L.; Anh, D.T.; Trung, N.T.; Bang, M.H.; Meyer, C.G.; Velavan, T.P.; et al. Vitamin D receptor ApaI polymorphism associated with progression of liver disease in Vietnamese patients chronically infected with hepatitis B virus. BMC Med. Genet. 2019, 20, 201. [Google Scholar] [CrossRef] [PubMed]
- Shan, B.; Wang, J.Y.; Wang, X.; Fu, J.J.; Li, L.; Pan, X.C.; Li, J.J.; Tang, X.T. VDR rs7975232/ApaI genetic variation predicts sustained HBsAg loss in HBeAg-positive chronic hepatitis B patients treated with pegylated interferon. J. Med. Virol. 2019, 91, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Cusato, J.; Boglione, L.; De Nicolo, A.; Imbornone, R.; Cardellino, C.S.; Ghisetti, V.; Carcieri, C.; Cariti, G.; Di Perri, G.; D’Avolio, A. Association of vitamin D pathway SNPs and clinical response to interferon in a cohort of HBeAg-negative patients. Pharmacogenomics 2017, 18, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Mostafa-Hedeab, G.; Sabry, D.; Abdelaziz, G.M.; Ewaiss, M.; Adli, N.; Fathy, W. Influence of Vitamin D Receptor Gene Polymorphisms on Response to Pegylated Interferon in Chronic Hepatitis B Egyptian Patients. Rep. Biochem. Mol. Biol. 2018, 6, 186–196. [Google Scholar] [PubMed]
- Thanapirom, K.; Suksawatamnuay, S.; Sukeepaisarnjaroen, W.; Treeprasertsuk, S.; Tanwandee, T.; Charatcharoenwitthaya, P.; Thongsawat, S.; Leerapun, A.; Piratvisuth, T.; Boonsirichan, R.; et al. Vitamin D-Binding protein Gene Polymorphism Predicts Pegylated Interferon-Related HBsAg Seroclearance in HBeAg-Negative Thai Chronic Hepatitis B Patients: A Multicentre Study. Asian Pac. J. Cancer Prev. 2019, 20, 1257–1264. [Google Scholar] [CrossRef]
- Campbell, G.R.; Spector, S.A. Vitamin D inhibits human immunodeficiency virus type 1 and Mycobacterium tuberculosis infection in macrophages through the induction of autophagy. PLoS Pathog. 2012, 8, e1002689. [Google Scholar] [CrossRef]
- Campbell, G.R.; Spector, S.A. Autophagy induction by vitamin D inhibits both Mycobacterium tuberculosis and human immunodeficiency virus type 1. Autophagy 2012, 8, 1523–1525. [Google Scholar] [CrossRef]
- Aguilar-Jimenez, W.; Zapata, W.; Caruz, A.; Rugeles, M.T. High transcript levels of vitamin D receptor are correlated with higher mRNA expression of human beta defensins and IL-10 in mucosa of HIV-1-exposed seronegative individuals. PLoS ONE 2013, 8, e82717. [Google Scholar] [CrossRef]
- Shepherd, L.; Souberbielle, J.C.; Bastard, J.P.; Fellahi, S.; Capeau, J.; Reekie, J.; Reiss, P.; Blaxhult, A.; Bickel, M.; Leen, C.; et al. Prognostic value of vitamin D level for all-cause mortality, and association with inflammatory markers, in HIV-infected persons. J. Infect. Dis. 2014, 210, 234–243. [Google Scholar] [CrossRef]
- Aguilar-Jimenez, W.; Saulle, I.; Trabattoni, D.; Vichi, F.; Lo Caputo, S.; Mazzotta, F.; Rugeles, M.T.; Clerici, M.; Biasin, M. High Expression of Antiviral and Vitamin D Pathway Genes Are a Natural Characteristic of a Small Cohort of HIV-1-Exposed Seronegative Individuals. Front. Immunol. 2017, 8, 136. [Google Scholar] [CrossRef]
- Erlandson, K.M.; Gudza, I.; Fiorillo, S.; Ndemera, B.; Schooley, R.T.; Gwanzura, L.; Borok, M.; Campbell, T.B. Relationship of vitamin D insufficiency to AIDS-associated Kaposi’s sarcoma outcomes: Retrospective analysis of a prospective clinical trial in Zimbabwe. Int. J. Infect. Dis. 2014, 24, 6–10. [Google Scholar] [CrossRef]
- Ezeamama, A.E.; Guwatudde, D.; Wang, M.; Bagenda, D.; Kyeyune, R.; Sudfeld, C.; Manabe, Y.C.; Fawzi, W.W. Vitamin-D deficiency impairs CD4+T-cell count recovery rate in HIV-positive adults on highly active antiretroviral therapy: A longitudinal study. Clin. Nutr. 2016, 35, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Coussens, A.K.; Naude, C.E.; Goliath, R.; Chaplin, G.; Wilkinson, R.J.; Jablonski, N.G. High-dose vitamin D3 reduces deficiency caused by low UVB exposure and limits HIV-1 replication in urban Southern Africans. Proc. Natl. Acad. Sci. USA 2015, 112, 8052–8057. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Jimenez, W.; Villegas-Ospina, S.; Gonzalez, S.; Zapata, W.; Saulle, I.; Garziano, M.; Biasin, M.; Clerici, M.; Rugeles, M.T. Precursor Forms of Vitamin D Reduce HIV-1 Infection In Vitro. J. Acquir. Immune Defic. Syndr. 2016, 73, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, S.M.; Aguilar-Jimenez, W.; Trujillo-Gil, E.; Zapata, W.; Su, R.C.; Ball, T.B.; Rugeles, M.T. Vitamin D treatment of peripheral blood mononuclear cells modulated immune activation and reduced susceptibility to HIV-1 infection of CD4+ T lymphocytes. PLoS ONE 2019, 14, e0222878. [Google Scholar] [CrossRef]
- Ayelign, B.; Workneh, M.; Molla, M.D.; Dessie, G. Role Of Vitamin-D Supplementation In TB/HIV Co-Infected Patients. Infect. Drug Resist. 2020, 13, 111–118. [Google Scholar] [CrossRef]
- Honda, J.R.; Connick, E.; MaWhinney, S.; Chan, E.D.; Flores, S.C. Plasma LL-37 correlates with vitamin D and is reduced in human immunodeficiency virus-1 infected individuals not receiving antiretroviral therapy. J. Med. Microbiol. 2014, 63, 997–1003. [Google Scholar] [CrossRef]
- Gao, W.; Wang, R.; Wang, X.; Wu, H.; Wang, Y.; Lu, X.; Li, L.; Zheng, J.; Li, W. Vitamin D serum levels and receptor genetic polymorphisms are associated with hepatitis B virus and HIV infections and IFN-lambda levels. Biomark Med. 2017, 11, 733–740. [Google Scholar] [CrossRef]
- Bang, U.; Kolte, L.; Hitz, M.; Dam Nielsen, S.; Schierbeck, L.L.; Andersen, O.; Haugaard, S.B.; Mathiesen, L.; Benfield, T.; Jensen, J.E. Correlation of increases in 1,25-dihydroxyvitamin D during vitamin D therapy with activation of CD4+ T lymphocytes in HIV-1-infected males. HIV Clin. Trials 2012, 13, 162–170. [Google Scholar] [CrossRef]
- Ross, A.C.; Judd, S.; Ziegler, T.R.; Camacho-Gonzalez, A.; Fitzpatrick, A.; Hadley, G.; Grossmann, R.; Seydafkan, S.; Mulliga, M.J.; Rimann, N.; et al. 2012 Risk factors for vitamin D deficiency and relationship with cardiac biomarkers, inflammation and immune restoration in HIV-infected youth. Antivir. Ther. 2012, 17, 1069–1078. [Google Scholar] [CrossRef]
- Ashenafi, S.; Amogne, W.; Kassa, E.; Gebreselassie, N.; Bekele, A.; Aseffa, G.; Getachew, M.; Aseffa, A.; Worku, A.; Hammar, U.; et al. Daily Nutritional Supplementation with Vitamin D(3) and Phenylbutyrate to Treatment-Naive HIV Patients Tested in a Randomized Placebo-Controlled Trial. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Visser, M.E.; Durao, S.; Sinclair, D.; Irlam, J.H.; Siegfried, N. Micronutrient supplementation in adults with HIV infection. Cochrane Database Syst. Rev. 2017, 5, CD003650. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.R.; Spector, S.A. Toll-like receptor 8 ligands activate a vitamin D mediated autophagic response that inhibits human immunodeficiency virus type 1. PLoS Pathog. 2012, 8, e1003017. [Google Scholar] [CrossRef]
- Aguilar-Jimenez, W.; Zapata, W.; Rugeles, M.T. Antiviral molecules correlate with vitamin D pathway genes and are associated with natural resistance to HIV-1 infection. Microbes Infect. 2016, 18, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Laplana, M.; Sanchez-de-la-Torre, M.; Puig, T.; Caruz, A.; Fibla, J. Vitamin-D pathway genes and HIV-1 disease progression in injection drug users. Gene 2014, 545, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Torres, C.; Sanchez-de-la-Torre, M.; Garcia-Moruja, C.; Carrero, A.J.; Trujillo, M.D.M.; Fibla, J.; Caruz, A. 2010 Immunophenotype of vitamin D receptor polymorphism associated to risk of HIV-1 infection and rate of disease progression. Curr. HIV Res. 2010, 8, 487–492. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, M.S.; Torres, C.; Nieto, G.; Vergara, S.; Carrero, A.J.; Macias, J.; Pineda, J.A.; Caruz, A.; Fibla, J. Vitamin D receptor gene haplotypes and susceptibility to HIV-1 infection in injection drug users. J. Infect. Dis. 2008, 197, 405–410. [Google Scholar] [CrossRef]
- Xu, F.; Schillinger, J.A.; Sternberg, M.R.; Johnson, R.E.; Lee, F.K.; Nahmias, A.J.; Markowitz, L.E. Seroprevalence and coinfection with herpes simplex virus type 1 and type 2 in the United States, 1988-1994. J. Infect. Dis. 2002, 185, 1019–1024. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, M.P.; Kumar, R.S.; Ratho, R.K. 25-Hydroxyvitamin D3 and 1,25 Dihydroxyvitamin D3 as an Antiviral and Immunomodulator Against Herpes Simplex Virus-1 Infection in HeLa Cells. Viral Immunol. 2018, 31, 589–593. [Google Scholar] [CrossRef]
- Rieder, F.J.J.; Groschel, C.; Kastner, M.T.; Kosulin, K.; Laengle, J.; Zadnikar, R.; Marculescu, R.; Schneider, M.; Lion, T.; Bergmann, M.; et al. Human cytomegalovirus infection downregulates vitamin-D receptor in mammalian cells. J. Steroid Biochem. Mol. Biol. 2017, 165, 356–362. [Google Scholar] [CrossRef]
- Rech, M.A.; Fleming, J.N.; Moore, C.L. 25-hydroxyvitamin D deficiency and opportunistic viral infections after kidney transplant. Exp. Clin. Transplant. 2014, 12, 95–100. [Google Scholar]
- Chao, C.T.; Lee, S.Y.; Yang, W.S.; Yen, C.J.; Chiang, C.K.; Huang, J.W.; Hung, K.Y. Serum vitamin D levels are positively associated with varicella zoster immunity in chronic dialysis patients. Sci. Rep. 2014, 4, 7371. [Google Scholar] [CrossRef] [PubMed]
- Von Bahr, L.; Blennow, O.; Alm, J.; Bjorklund, A.; Malmberg, K.J.; Mougiakakos, D.; Le Blanc, A.; Oefner, P.J.; Labopin, M.; Ljungman, P.; et al. Increased incidence of chronic GvHD and CMV disease in patients with vitamin D deficiency before allogeneic stem cell transplantation. Bone Marrow Transplant. 2015, 50, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Moscarelli, L.; Antognoli, G.; Buti, E.; Dervishi, E.; Fani, F.; Caroti, L.; Tsalouchos, A.; Romoli, E.; Ghiandai, G.; Minetti, E. 1,25 Dihydroxyvitamin D circulating levels, calcitriol administration, and incidence of acute rejection, CMV infection, and polyoma virus infection in renal transplant recipients. Clin. Transplant. 2016, 30, 1347–1359. [Google Scholar] [CrossRef]
- Maghzi, H.; Ataei, B.; Khorvash, F.; Yaran, M.; Maghzi, A.H. Association Between Acute Infectious Mononucleosis and Vitamin D Deficiency. Viral Immunol. 2016, 29, 398–400. [Google Scholar] [CrossRef] [PubMed]
- Perez-Perez, S.; Dominguez-Mozo, M.I.; Garcia-Martinez, M.A.; Aladro, Y.; Martinez-Gines, M.; Garcia-Dominguez, J.M.; Lopez de Silanes, C.; Casanova, I.; Ortega-Madueno, I.; Lopez-Lozano, L.; et al. Study of the possible link of 25-hydroxyvitamin D with Epstein-Barr virus and human herpesvirus 6 in patients with multiple sclerosis. Eur. J. Neurol. 2018, 25, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Oztekin, A.; Oztekin, C. Vitamin D Levels in Patients with Recurrent Herpes Labialis. Viral Immunol. 2019, 32, 258–262. [Google Scholar] [CrossRef]
- Jadhav, N.J.; Gokhale, S.; Seervi, M.; Patil, P.S.; Alagarasu, K. Immunomodulatory effect of 1, 25 dihydroxy vitamin D3 on the expression of RNA sensing pattern recognition receptor genes and cytokine response in dengue virus infected U937-DC-SIGN cells and THP-1 macrophages. Int. Immunopharmacol. 2018, 62, 237–243. [Google Scholar] [CrossRef]
- Arboleda Alzate, J.F.; Rodenhuis-Zybert, I.A.; Hernandez, J.C.; Smit, J.M.; Urcuqui-Inchima, S. Human macrophages differentiated in the presence of vitamin D3 restrict dengue virus infection and innate responses by downregulating mannose receptor expression. PLoS Negl. Trop. Dis. 2017, 11, e0005904. [Google Scholar] [CrossRef]
- Giraldo, D.M.; Cardona, A.; Urcuqui-Inchima, S. High-dose of vitamin D supplement is associated with reduced susceptibility of monocyte-derived macrophages to dengue virus infection and pro-inflammatory cytokine production: An exploratory study. Clin. Chim. Acta 2018, 478, 140–151. [Google Scholar] [CrossRef]
- Martinez-Moreno, J.; Hernandez, J.C.; Urcuqui-Inchima, S. Effect of high doses of vitamin D supplementation on dengue virus replication, Toll-like receptor expression, and cytokine profiles on dendritic cells. Mol. Cell. Biochem. 2020, 464, 169–180. [Google Scholar] [CrossRef]
- Villamor, E.; Villar, L.A.; Lozano, A.; Herrera, V.M.; Herran, O.F. Vitamin D serostatus and dengue fever progression to dengue hemorrhagic fever/dengue shock syndrome. Epidemiol. Infect. 2017, 145, 2961–2970. [Google Scholar] [CrossRef]
- Zhao, Y.; Yu, B.; Mao, X.; He, J.; Huang, Z.; Zheng, P.; Yu, J.; Han, G.; Liang, X.; Chen, D. Effect of 25-hydroxyvitamin D3 on rotavirus replication and gene expressions of RIG-I signalling molecule in porcine rotavirus-infected IPEC-J2 cells. Arch. Anim. Nutr. 2015, 69, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Bucak, I.H.; Ozturk, A.B.; Almis, H.; Cevik, M.O.; Tekin, M.; Konca, C.; Turgut, M.; Bulbul, M. Is there a relationship between low vitamin D and rotaviral diarrhea? Pediatr. Int. 2016, 58, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.; Liang, X.; Chen, D.; Mao, X.; Yu, J.; Zheng, P.; He, J.; Huang, Z.; Yu, B. Vitamin D3 supplementation alleviates rotavirus infection in pigs and IPEC-J2 cells via regulating the autophagy signaling pathway. J. Steroid Biochem. Mol. Biol. 2016, 163, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ran, Z.; Jiang, Q.; Hu, N.; Yu, B.; Zhu, L.; Shen, L.; Zhang, S.; Chen, L.; Chen, H.; et al. Vitamin D Alleviates Rotavirus Infection through a Microrna-155-5p Mediated Regulation of the TBK1/IRF3 Signaling Pathway In Vivo and In Vitro. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Tian, G.; Chen, D.; Zheng, P.; Yu, J.; Mao, X.; He, J.; Luo, Y.; Luo, J.; Huang, Z.; et al. Dietary 25-Hydroxyvitamin D3 Supplementation Alleviates Porcine Epidemic Diarrhea Virus Infection by Improving Intestinal Structure and Immune Response in Weaned Pigs. Animals (Basel) 2019, 9. [Google Scholar] [CrossRef]
- Shim, J.; Perez, A.; Symanski, E.; Nyitray, A.G. Association Between Serum 25-Hydroxyvitamin D Level and Human Papillomavirus Cervicovaginal Infection in Women in the United States. J. Infect. Dis. 2016, 213, 1886–1892. [Google Scholar] [CrossRef]
- Garcia-Carrasco, M.; Mendoza-Pinto, C.; Munguia-Realpozo, P.; Rodriguez-Gallegos, A.; Vallejo-Ruiz, V.; Munoz-Guarneros, M.; Mendez-Martinez, S.; Soto-Santillan, P.; Pezzat-Said, E.; Reyes-Leyva, J.; et al. Lack of association between serum 25-hydroxyvitamin D levels and cervical human papillomavirus infection in systemic lupus erythematosus. Lupus 2015, 24, 606–612. [Google Scholar] [CrossRef]
- Arboleda, J.F.; Fernandez, G.J.; Urcuqui-Inchima, S. Vitamin D-mediated attenuation of miR-155 in human macrophages infected with dengue virus: Implications for the cytokine response. Infect. Genet. Evol. 2019, 69, 12–21. [Google Scholar] [CrossRef]
- Alagarasu, K.; Honap, T.; Mulay, A.P.; Bachal, R.V.; Shah, P.S.; Cecilia, D. Association of vitamin D receptor gene polymorphisms with clinical outcomes of dengue virus infection. Hum. Immunol. 2012, 73, 1194–1199. [Google Scholar] [CrossRef]
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).