Effects of Diet on Sleep: A Narrative Review
Abstract
1. Introduction
2. Materials and Methods
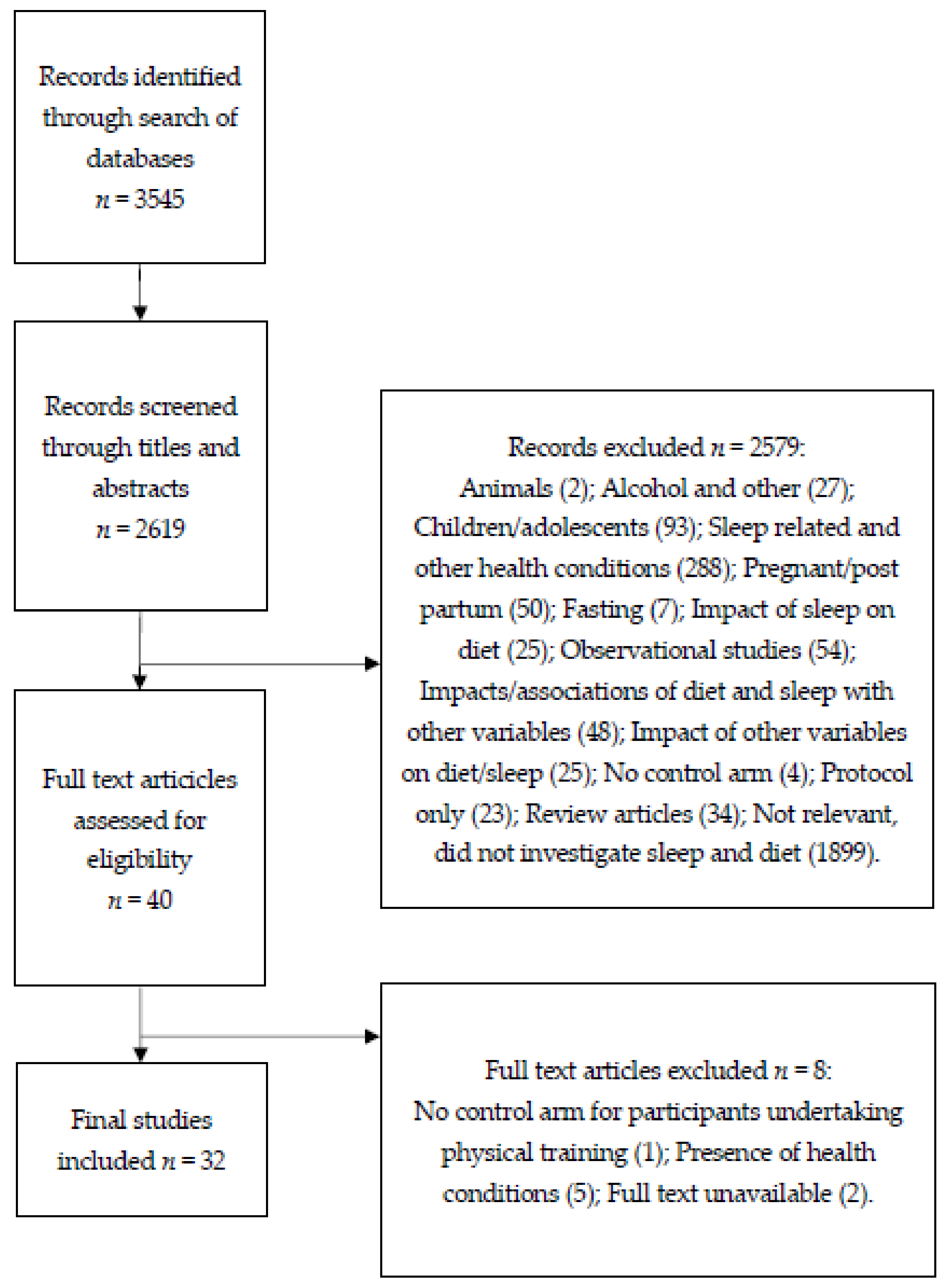
2.1. Literature Search
2.2. Study Eligibility and Sselection
2.3. Data Extraction and Synthesis
3. Results
3.1. A Summary of the Included Studies
3.1.1. Tryptophan Consumption and Depletion
3.1.2. Dietary Supplements
Zinc
B Vitamins
Polyphenols
Crocetin
Chlorogenic Acids
Chlorophytum bovivilianum (Root) and Velvet Bean
γ-Aminobutyric Acid and Apocynum venetum Leaf Extract
3.1.3. Food Items
3.1.4. Macronutrients
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gómez-González, B.; Domínguez-Salazar, E.; Hurtado-Alvarado, G.; Esqueda-Leon, E.; Santana-Miranda, R.; Rojas-Zamorano, J.A.; Velázquez-Moctezuma, J. Role of sleep in the regulation of the immune system and the pituitary hormones. Ann. N. Y. Acad. Sci. 2012, 1261, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Benington, J.H.; Craig Heller, H. Restoration of brain energy metabolism as the function of sleep. Prog. Neurobiol. 1995, 45, 347–360. [Google Scholar] [CrossRef]
- Schönauer, M. Sleep spindles: Timed for memory consolidation. Curr. Biol. 2018, 28, R656–R658. [Google Scholar] [CrossRef]
- Ohayon, M.; Wickwire, E.M.; Hirshkowitz, M.; Albert, S.M.; Avidan, A.; Daly, F.J.; Dauvilliers, Y.; Ferri, R.; Fung, C.H.; Gozal, D.; et al. National Sleep Foundation’s sleep quality recommendations: First report. Sleep Health 2017, 3, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Watson, N.F.; Badr, M.S.; Belenky, G.; Bliwise, D.L.; Buxton, O.M.; Buysse, D.; Dinges, D.F.; Gangwisch, J.; Grandner, M.A.; Kushida, C.; et al. Joint consensus statement of the american academy of sleep medicine and sleep research society on the recommended amount of sleep for a healthy adult: Methodology and discussion. J. Clin. Sleep Med. 2015, 11, 931–952. [Google Scholar] [CrossRef]
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Hazen, N.; Herman, J.; Katz, E.S.; Kheirandish-Gozal, L.; et al. National Sleep Foundation’s sleep time duration recommendations: methodology and results summary. Sleep health 2015, 1, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.; Appleton, S.T.; Mcevoy, D.; Antic, N. Report to the Sleep Health Foundation, 2016 Survey of Australian Adults. Available online: https://www.sleephealthfoundation.org.au/pdfs/surveys/SleepHealthFoundation-Survey.pdf (accessed on 14 January 2020).
- Centers for Disease Control Prevention, Unhealthy sleep-related behaviors 12 States, 2009. MMWR Morb. Mortal. Wkly. Rep. 2011, 60, 233.
- Banks, S.; Dinges, D.F. Behavioral and physiological consequences of sleep restriction. J. Clin. Sleep Med. 2007, 3, 519–528. [Google Scholar] [CrossRef]
- Brunner, D.P.; Dijk, D.J.; Tobler, I.; Borbely, A.A. Effect of partial sleep deprivation on sleep stages and EEG power spectra: evidence for non-REM and REM sleep homeostasis. Electroencephalogr. Clin. Neurophysiol. 1990, 75, 492–499. [Google Scholar] [CrossRef]
- Ancoli-Israel, S.; Cole, R.; Alessi, C.; Chambers, M.; Moorcroft, W.; Pollak, C.P. The role of actigraphy in the study of sleep and circadian rhythms. Sleep 2003, 26, 342–392. [Google Scholar] [CrossRef]
- De Souza, L.; Benedito-Silva, A.A.; Pires, M.L.N.; Poyares, D.; Tufik, S.; Calil, H.M.J.S. Further validation of actigraphy for sleep studies. Sleep 2003, 26, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Dashti, H.S.; Scheer, F.A.; Jacques, P.F.; Lamon-Fava, S.; Ordovas, J.M. Short sleep duration and dietary intake: epidemiologic evidence, mechanisms, and health implications. Adv. Nutr. 2015, 6, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Bravo, R.; Matito, S.; Cubero, J.; Paredes, S.; Franco, L.; Rivero, M.; Rodríguez, A.; Barriga, C. Tryptophan-enriched cereal intake improves nocturnal sleep, melatonin, serotonin, and total antioxidant capacity levels and mood in elderly humans. J. Am. Aging Assoc. 2013, 35, 1277–1285. [Google Scholar] [CrossRef]
- Layman, D.K.; Lönnerdal, B.; Fernstrom, J.D. Applications for α-lactalbumin in human nutrition. Nutr. Rev. 2018, 76, 444–460. [Google Scholar] [CrossRef]
- Bhatti, T.; Gillin, J.C.; Seifritz, E.; Moore, P.; Clark, C.; Golshan, S.; Stahl, S.; Rapaport, M.; Kelsoe, J. Effects of a Tryptophan-Free Amino Acid Drink Challenge on Normal Human Sleep Electroencephalogram and Mood. Biol. Psychiatry 1998, 43, 52–59. [Google Scholar] [CrossRef]
- Duan, K.M.; Ma, J.H.; Wang, S.Y.; Huang, Z.D.; Zhou, Y.Y.; Yu, H.Y. The role of tryptophan metabolism in postpartum depression. Metab. Brain Dis. 2018, 33, 647–660. [Google Scholar] [CrossRef]
- González-Gómez, D.; Lozano, M.; Fernández-León, M.; Ayuso, M.; Bernalte, M.; Rodríguez, A. Detection and quantification of melatonin and serotonin in eight Sweet Cherry cultivars (Prunus avium L.). Eur. Food Res. Technol. 2009, 229, 223–229. [Google Scholar]
- Garrido, M.; González-Gómez, D.; Lozano, M.; Barriga, C.; Paredes, S.D.; Rodríguez, A.B. A Jerte valley cherry product provides beneficial effects on sleep quality. Influence on aging. J. Nutr., Health Aging 2013, 17, 553–560. [Google Scholar] [CrossRef]
- Chaput, J.P. Sleep patterns, diet quality and energy balance. Physiol. Behav. 2014, 134, 86. [Google Scholar] [CrossRef]
- Peuhkuri, K.; Sihvola, N.; Korpela, R. Diet promotes sleep duration and quality. Nutr Res 2012, 32, 309–319. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Mikic, A.; Pietrolungo, C.E. Effects of Diet on Sleep Quality. Adv. Nutr. 2016, 7, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Steiger, A.; Pawlowski, M. Depression and sleep. Int. J. Mol. Sci. 2019, 20, 607. [Google Scholar] [CrossRef]
- Benham, G. Sleep: an important factor in stress-health models. Stress Health 2010, 26, 204–214. [Google Scholar] [CrossRef]
- Gradisar, M.; Wolfson, A.R.; Harvey, A.G.; Hale, L.; Rosenberg, R.; Czeisler, C.A. The sleep and technology use of Americans: findings from the National Sleep Foundation’s 2011 Sleep in America poll. J. Clin. Sleep Med. 2013, 9, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.H.; Wittwer, J.; Vargas, K.; Hogan, E.; Holmes, A.; Rogers, P.J.; Goralczyk, R.; Gibson, E.L. Chronic treatment with a tryptophan-rich protein hydrolysate improves emotional processing, mental energy levels and reaction time in middle-aged women. Br. J. Nutr. 2015, 113, 350–365. [Google Scholar] [CrossRef]
- Voderholzer, U.; Hornyak, M.; Thiel, B.; Huwig-Poppe, C.; Kiemen, A.; Konig, A.; Backhaus, J.; Riemann, D.; Berger, M.; Hohagen, F. Impact of experimentally induced serotonin deficiency by tryptophan depletion on sleep EEG in healthy subjects. Neuropsychopharmacology 1998, 18, 112–124. [Google Scholar] [CrossRef]
- Arnulf, I.; Quintin, P.; Alvarez, J.C.; Vigil, L.; Touitou, Y.; Lebre, A.S.; Bellenger, A.; Varoquaux, O.; Derenne, J.P.; Allilaire, J.F.; et al. Mid-morning tryptophan depletion delays REM sleep onset in healthy subjects. Neuropsychopharmacology 2002, 27, 843–851. [Google Scholar] [CrossRef]
- Moore, P.; Landolt, H.; Seifritz, E.; Clark, C.; Bhatti, T.; Kelsoe, J.; Rapaport, M.; Gillin, J.C. Clinical and physiological consequences of rapid tryptophan depletion. Neuropsychopharmacology 2000, 23, 601. [Google Scholar] [CrossRef]
- Aspy, D.J.; Madden, N.A.; Delfabbro, P. Effects of Vitamin B6 (Pyridoxine) and a B Complex Preparation on Dreaming and Sleep. Percept. Mot. Ski 2018, 125, 451–462. [Google Scholar] [CrossRef]
- Gholipour Baradari, A.; Alipour, A.; Mahdavi, A.; Sharifi, H.; Nouraei, S.M.; Emami Zeydi, A. The effect of zinc supplementation on sleep quality of ICU nurses: a double blinded randomized controlled trial. Workplace Health Saf. 2018, 66, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Romain, C.; Alcaraz, P.E.; Chung, L.H.; Cases, J. Regular consumption of HolisFiit, a polyphenol-rich extract-based food supplement, improves mind and body well-being of overweight and slightly obese volunteers: a randomized, double-blind, parallel trial. Int. J. Food Sci. Nutr. 2017, 68, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Cox, K.H.; Camfield, D.A.; Scholey, A.; Stough, C.; Fogg, E.; Kras, M.; White, D.J.; Sali, A.; Pipingas, A. Participant experiences from chronic administration of a multivitamin versus placebo on subjective health and wellbeing: a double-blind qualitative analysis of a randomised controlled trial. Nutr. J. 2012, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Wouters-Wesseling, W.; Van Hooijdonk, C.; Wagenaar, L.; Bindels, J.; de Groot, L.; Van Staveren, W. The effect of a liquid nutrition supplement on body composition and physical functioning in elderly people. Clin. Nutr. 2003, 22, 371–377. [Google Scholar] [CrossRef]
- Wightman, E.L.; Haskell-Ramsay, C.F.; Reay, J.L.; Williamson, G.; Dew, T.; Zhang, W.; Kennedy, D.O. The effects of chronic trans-resveratrol supplementation on aspects of cognitive function, mood, sleep, health and cerebral blood flow in healthy, young humans. Br. J. Nutr. 2015, 114, 1427–1437. [Google Scholar] [CrossRef]
- McCarthy, C.G.; Alleman, R.J.; Bell, Z.W.; Bloomer, R.J. A dietary supplement containing chlorophytum borivilianum and velvet bean improves sleep quality in men and women. Integr. Med. Insights 2012, 7. [Google Scholar] [CrossRef]
- Park, I.; Ochiai, R.; Ogata, H.; Kayaba, M.; Hari, S.; Hibi, M.; Katsuragi, Y.; Satoh, M.; Tokuyama, K. Effects of subacute ingestion of chlorogenic acids on sleep architecture and energy metabolism through activity of the autonomic nervous system: a randomised, placebo-controlled, double-blinded cross-over trial. Br. J. Nutr. 2017, 117, 979–984. [Google Scholar] [CrossRef]
- Yamatsu, A.; Yamashita, Y.; Maru, I.; Yang, J.; Tatsuzaki, J.; Kim, M. The Improvement of Sleep by Oral Intake of GABA and Apocynum venetum Leaf Extract. J. Nutr. Sci. Vitaminol. 2015, 61, 182–187. [Google Scholar] [CrossRef]
- Kuratsune, H.; Umigai, N.; Takeno, R.; Kajimoto, Y.; Nakano, T. Effect of crocetin from Gardenia jasminoides Ellis on sleep: a pilot study. Phytomedicine 2010, 17, 840–843. [Google Scholar] [CrossRef]
- Um, M.Y.; Kim, J.Y.; Han, J.K.; Kim, J.; Yang, H.; Yoon, M.; Kim, J.; Kang, S.W.; Cho, S. Phlorotannin supplement decreases wake after sleep onset in adults with self-reported sleep disturbance: a randomized, controlled, double-blind clinical and polysomnographic study. Phytother. Res. 2018, 32, 698–704. [Google Scholar] [CrossRef]
- Umigai, N.; Takeda, R.; Mori, A. Effect of crocetin on quality of sleep: a randomized, double-blind, placebo-controlled, crossover study. Complement. Ther. Med. 2018, 41, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Cherasse, Y.; Suzuki, R.; Mitarai, M.; Ueda, F.; Urade, Y. Zinc-rich oysters as well as zinc-yeast- and astaxanthin-enriched food improved sleep efficiency and sleep onset in a randomized controlled trial of healthy individuals. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Lentjes, M.A.H. The balance between food and dietary supplements in the general population. Proc. Nutr. Soc. 2019, 78, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Garrido, M.; Paredes, S.D.; Cubero, J.; Lozano, M.; Toribio-Delgado, A.F.; Muñoz, J.L.; Reiter, R.J.; Barriga, C.; Rodríguez, A.B. Jerte valley cherry-enriched diets improve nocturnal rest and increase 6-sulfatoxymelatonin and total antioxidant capacity in the urine of middle-aged and elderly humans. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 909–914. [Google Scholar] [CrossRef]
- Howatson, G.; Bell, P.G.; Tallent, J.; Middleton, B.; McHugh, M.P.; Ellis, J. Effect of tart cherry juice (Prunus cerasus) on melatonin levels and enhanced sleep quality. Eur. J. Nutr. 2012, 51, 909–916. [Google Scholar] [CrossRef]
- Driver, H.S.; Shulman, I.; Baker, F.C.; Buffenstein, R. Energy content of the evening meal alters nocturnal body temperature but not sleep. Physiol. Behav. 1999, 68, 17–23. [Google Scholar] [CrossRef]
- Afaghi, A.; O’Connor, H.; Chow, C.M. High-glycemic-index carbohydrate meals shorten sleep onset. Am. J. Clin. Nutr. 2007, 85, 426–430. [Google Scholar] [CrossRef]
- Afaghi, A.; O’Connor, H.; Chow, C.M. Acute effects of the very low carbohydrate diet on sleep indices. Nutr. Neurosci. 2008, 11, 146–154. [Google Scholar] [CrossRef]
- Lindseth, G.; Lindseth, P.; Thompson, M. Nutritional effects on sleep. West. J. Nurs. Res. 2011, 35, 497–513. [Google Scholar] [CrossRef]
- Yajima, K.; Seya, T.; Iwayama, K.; Hibi, M.; Hari, S.; Nakashima, Y.; Ogata, H.; Omi, N.; Satoh, M.; Tokuyama, K. Effects of nutrient composition of dinner on sleep architecture and energy metabolism during sleep. J. Nutr. Sci. Vitaminol. 2014, 60, 114–121. [Google Scholar] [CrossRef]
- Lindseth, G.; Murray, A. Dietary Macronutrients and Sleep. West. J. Nurs. Res. 2016, 38, 938–958. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Roberts, A.; Shechter, A.; Choudhury, A.R. Fiber and saturated fat are associated with sleep arousals and slow wave sleep. J. Clin. Sleep Med. 2016, 12, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Kim, J.E.; Armstrong, C.L.; Chen, N.; Campbell, W.W. Higher-protein diets improve indexes of sleep in energy-restricted overweight and obese adults: results from 2 randomized controlled trials. Am. J. Clin. Nutr. 2016, 103, 766–774. [Google Scholar] [CrossRef]
- Castro, A.I.; Gomez-Arbelaez, D.; Crujeiras, A.B.; Granero, R.; Aguera, Z.; Jimenez-Murcia, S.; Sajoux, I.; Lopez-Jaramillo, P.; Fernandez-Aranda, F.; Casanueva, F.F. Effect of a very low-calorie ketogenic diet on food and alcohol cravings, physical and sexual activity, sleep disturbances, and quality of life in obese patients. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Richard, D.M.; Dawes, M.A.; Mathias, C.W.; Acheson, A.; Hill-Kapturczak, N.; Dougherty, D.M. L-Tryptophan: Basic metabolic functions, behavioral research and therapeutic indications. Int. J. Tryptophan Res. 2009, 2, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, R.C.; McDougle, C.J.; Schumacher, M.; Olcese, J.; Heninger, G.R.; Price, L.H. Urinary 6-hydroxymelatonin sulfate as a measure of melatonin secretion during acute tryptophan depletion. Psychoneuroendocrinology 1993, 18, 567–578. [Google Scholar] [CrossRef]
- Kim, D.O.; Heo, H.J.; Kim, Y.J.; Yang, H.S.; Lee, C.Y. Sweet and sour cherry phenolics and their protective effects on neuronal cells. J. Agric. Food Chem. 2005, 53, 9921–9927. [Google Scholar] [CrossRef]
- Pigeon, W.R.; Carr, M.; Gorman, C.; Perlis, M.L. Effects of a tart cherry juice beverage on the sleep of older adults with insomnia: a pilot study. J. Med. Food 2010, 13, 579. [Google Scholar] [CrossRef]
- Meng, J.F.; Shi, T.C.; Song, S.; Zhang, Z.W.; Fang, Y.L. Melatonin in grapes and grape-related foodstuffs: A review. Food Chem. 2017, 231, 185–191. [Google Scholar] [CrossRef]
- Cho, S.; Yoon, M.; Pae, A.; Jin, Y.H.; Cho, N.C.; Takata, Y.; Urade, Y.; Kim, S.; Kim, J.S.; Yang, H.; et al. Marine polyphenol phlorotannins promote non-rapid eye movement sleep in mice via the benzodiazepine site of the GABA A receptor. Psychopharmacology 2014, 231, 2825–2837. [Google Scholar] [CrossRef]
- Cherasse, Y.; Urade, Y. Dietary zinc acts as a sleep modulator. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Grandner, M.A.; Jackson, N.; Gerstner, J.R.; Knutson, K.L. Dietary nutrients associated with short and long sleep duration. Data from a nationally representative sample. Appetite 2013, 64, 71–80. [Google Scholar] [CrossRef]
- Alleman, R.J.; Canale, R.E.; McCarthy, C.G.; Bloomer, R.J. A blend of chlorophytum borivilianum and velvet bean increases serum growth hormone in exercise-trained men. Nutr. Metab. Insights 2011, 4. [Google Scholar]
- Peterfi, Z.; McGinty, D.; Sarai, E.; Szymusiak, R. Growth hormone-releasing hormone activates sleep regulatory neurons of the rat preoptic hypothalamus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R147–R156. [Google Scholar] [CrossRef] [PubMed]
- Kerkhofs, M.; Van Cauter, E.; Van Onderbergen, A.; Caufriez, A.; Thorner, M.O.; Copinschi, G. Sleep-promoting effects of growth hormone-releasing hormone in normal men. Am. J. Physiol. 1993, 264, E594–E598. [Google Scholar] [CrossRef]
- Rutenfranz, J.; Colquhoun, W.P.; Knauth, P.; Ghata, J.N. Biomedical and psychosocial aspects of shift work. A review. Scand. J. Work Environ. Health. 1977, 165–182. [Google Scholar] [CrossRef]
- Gracia, P.; Kalmijn, M. Parents’ family time and work schedules: The split-shift schedule in Spain. J. Marraige 2016, 78, 401–415. [Google Scholar] [CrossRef]
- Campanini, M.Z.; Lopez-Garcia, E.; Rodríguez-Artalejo, F.; González, A.D.; Andrade, S.M.; Mesas, A.E. Agreement between sleep diary and actigraphy in a highly educated Brazilian population. Sleep Med. 2017, 35, 27–34. [Google Scholar] [CrossRef]
- Oda, H. Chrononutrition. J. Nutr. Sci. Vitaminol. 2015, 61 Suppl., S92–S94. [Google Scholar] [CrossRef]
- Almoosawi, S.; Vingeliene, S.; Karagounis, L.; Pot, G.K. Chrono-nutrition: a review of current evidence from observational studies on global trends in time-of-day of energy intake and its association with obesity. 2016, 75, 487–500. [Google Scholar] [CrossRef]
- Aparecida Crispim, C.; Carliana Mota, M. New perspectives on chrononutrition. Biol. Rhythm Res. 2019, 50, 63–77. [Google Scholar] [CrossRef]
- Oike, H.; Oishi, K.; Kobori, M. Nutrients, clock genes, and chrononutrition. Curr. Nutr. Rep. 2014, 3, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Wurtman, R.J.; Wurtman, J.J.; Regan, M.M.; McDermott, J.M.; Tsay, R.H.; Breu, J.J. Effects of normal meals rich in carbohydrates or proteins on plasma tryptophan and tyrosine ratios. Am. J. Clin. Nutr. 2003, 77, 128. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Design; Location | Duration; Age; Participants (m/f) | Intervention/Control | Sleep Measurements | Outcomes |
|---|---|---|---|---|---|
| Bravo (2013) [15] | SB, P; Spain | 3 wk; 55–75 y; 35 (9/26) | 30 g tryptophan-enriched cereals (containing 60 mg tryptophan)/control Subjects consumed their habitual diet posttreatment week (third week) with no cereals. | Actigraphy | Increase in actual sleep time, SE and immobile time and decrease in SOL, wake bouts, total activity and fragmentation index for intervention compared with control and habitual diet. |
| Bhatti (1998) [17] | RC, DB, CO | 6 dy; 21–53 y; 11 (11/0) | 100% tryptophan-free amino acid drink/control | PSG | Significant reduction in REM latency for both intervention and control compared to baseline. Significant increase in REM sleep time and REM% for control, non-significant increase in REM% for intervention compared to baseline. Significant increase in SOL for intervention compared to baseline. Significant decrease in TST, SE and stage 2 min for intervention compared to control. Significant increase in WASO for control compared to intervention. No significant difference in NREM measures. |
| Mohajeri (2015) [27] | RC, DB, P; UK | 19 dy; 45–65 y; 59 (0/59) | lumiVida™ (egg-white protein hydrolysate formulation containing ~70 mg tryptophan)/placebo | Subjective (sleep diaries) | Non-significant improvement in sleep quality for intervention compared with control. Non-significant increase in difficulty in getting up out of bed during trial for intervention. |
| Voderholzer (1998) [28] | RC, DB, CO, P; Germany | 2 × 4 dy; 23–55 y; 12 (6/6) | Low protein diet + one capsule of 0.5 g L-tryptophan (day 3 and 4), tryptophan-free amino acid drink (day 4)/placebo | PSG | Significant increase in amount of wake periods, wake times and REM density for tryptophan depletion compared with baseline. Significant decrease in stage 2 sleep for tryptophan depletion compared with baseline. |
| Arnulf (2002) [29] | RC, DB, CO, P; France | 3 dy; 18–39 y; 18 (7/11) | 100% tryptophan-free amino acid drink + capsules containing methionine, arginine and cysteine/placebo | PSG | Significant increase in arousal index, REM sleep onset latency and REM density for intervention. Non-significant increase in sleep N1 and N2. No significant difference in SOL, TST, total sleep period, duration of WASO, and 3–4 of NREM sleep or REM sleep for intervention. |
| Author (Year) | Design; Location | Duration; Age; Participants (m/f/t) | Intervention/Control | Sleep Measurement | Outcomes/Results | Notes |
|---|---|---|---|---|---|---|
| Aspy (2018) [31] | RC, DB; Australia | 5 dy; 18–40 y; 100 (31/68/1) | B6 (pyridoxine hydrochloride) only (240 mg) OR B complex (range of doses for each B vitamin)/placebo | Subjective (Sleep diary) | Significantly lower sleep quality for B complex compared with B6 only and placebo. Significantly higher tiredness upon waking for B complex compared with B6 only. No significant differences found for vitamin B6. | |
| Gholipour Baradari (2017) [32] | RC, DB; Iran | 1 m; 31.20 ± 5.42 y; 53 (4/49/0) | Zn gelatin capsule/placebo | Subjective (PSQI) | Significant improvement of global sleep score and subjective sleep quality for intervention compared with placebo. Significant improvements within-group for intervention in global sleep score, subjective sleep quality and SOL. Increased zinc serum levels associated with decreased odds of poor sleep. | Intensive care unit nurses, poor sleepers |
| Romain (2017) [33] | RC, DB, P; Spain | 16 wk; 30–50 y; 33 (17/16/0) | HolisFiit supplement (blend of polyphenol compounds; 500 mg)/placebo | Subjective (Athens Insomnia Scale) | Significantly reduced awakening during the night for intervention. Significantly increased total sleep duration, sleep quality, wellbeing during the day and functioning capacity during the day for intervention. Significant improvement in total score for both groups. | Overweight to slightly obese participants (BMI = 25–35 kg/m2) |
| Sarris (2012) [34] | RC, DB, P; Australia | 16 wk; 20–50 y; 114 (59/55/0) | Swisse Men’s Ultivite F1W multivitamin or Swisse Women’s Ultivite F1 W multivitamin/placebo | Subjective ratings of sleep-quality | Non-significant increase in SQ Reports of impaired sleep similar between groups | |
| Wouters-Wesseling (2003) [35] | RC, DB, P; The Netherlands | 6 mo; ≥ 65 y; 68 (39/29/0) | Liquid nutrition supplement/placebo | Subjective (Nottingham Health Profile) | Improved sleep quality | |
| Wightman (2015) [36] | RC, DB, P; UK | 28 dy; 18–25 y; 15 (5/10/0) | Pure trans-resveratrol capsule (500 mg; TransmaxTM by BiotiviaTM)/placebo | Subjective (PSQI) | No significant findings for global sleep quality score or individual sleep domains. | |
| McCarthy (2012) [37] | Single arm trial; US | 28 dy; 27.2 ± 1.7 y; 18 (9/9) | Capsule containing Chlorophytum borivilianum and Velvet bean | Subjective (PSQI) | Significant improvements in global sleep quality score and all sleep domains. | |
| Park (2017) [38] | RC, DB, P, CO; Japan | 2 × 5 dy; 25.7 y (mean); 9 (4/5/0) | 100ml can containing 600 mg of chlorogenic acid (CGA)/placebo | PSG | Significantly lower SOL for intervention. No significant difference in length of N1 or N2, N3, REM and wakefulness after sleep onset between groups. Significantly higher delta power during first hour of sleep for CGA compared with placebo. | |
| Yamatsu (2015) [39] | SB, P; Japan | 2 × 1 wk; 36.8 ± 8.9; 16 (7/9/0) | Capsules containing GABA OR AVLE OR combination of GABA and AVLE/placebo | PSG | Decrease in SOL for GABA Non-significant decreases in SOL for AVLE and GABA+AVLE. Non-significant decrease in NREM sleep latency for all intervention groups Significant increase in NREM time for AVLE. Non-significant decrease in REM time for all intervention groups. | Poor sleepers |
| Subjective (PSQI) | Improved subjective SQ for AVLE | |||||
| Kuratsune (2010) [40] | RC, DB, P, CO; Japan | 2 × 2 wk; 25–59 yo; 21 (21/0) | Crocetin and dextrin capsule/placebo | Actigraphy | Significantly less wake episodes for intervention compared with placebo No significant difference in SOL or SE. | |
| Subjective (St Mary’s Hospital Sleep Questionnaire) | No significant difference in subjective measures. | |||||
| Um (2017) [41] | RC, DB, P; South Korea | 7 dy; 20 (13/7/0) | Phlorotannin supplement/placebo | PSG | Significant decrease in WASO, difference for TST No significant difference for SOL, SE, TST, REM latency, REM, N1-N3, apnea-hypopnea index or total arousal index between groups. | Participants with self-reported sleep disturbances |
| Subjective (PSQI; ESS; Stanford Sleepiness Scale) | Significant decrease in global sleep qual score and daytime functioning for intervention, no difference between groups.Increase in sleep duration. | |||||
| Umigai (2018) [42] | RC, DB, CO, P; Japan | 2 × 14 dy; 35–60 y; 24 (14/10/0) | Gardenia Yellow capsule with 7.5mg of crocetin/placebo | PSG | Significantly increased delta power for crocetin compared with placebo. No significant differences for SOL, SE, TST, WASO or REM. | Participants with mild sleep complaints All female participants postmenopausal |
| Subjective (OSA-MA) | Significant improvements for sleepiness upon rising and feeling refreshed No differences found for sleep duration | |||||
| Saito (2017) [43] | RC, DB, P; Japan | 12 wk; 20–84 y; 94 (45/74/0) | Placebo (scallop) supplemented with zinc and astaxanthin supplements/placebo | Actigraphy | Significant improvement for SOL compared with placebo. No significant difference in TST, sleep efficiency or body positional changes. | |
| Subjective (PSQI; sleep diary) | Significant improvement for global sleep score but not significantly different from placebo. |
| Author (Year) | Design; Location | Duration; Age; Participants (M/F/T) | Intervention/C | Sleep Measurement | Outcomes/Results | Notes |
|---|---|---|---|---|---|---|
| Garrido (2013) [20] | RC, P, SB, CO | 2 × 5 dy; 20–85 y; 30 (15/15/0) | Combination of four cultivars of Jerte Valley cherries, plus 7.5 g maltodextrin and 1.5 g ascorbic acid, diluted | Actigraphy | Significant increase in actual sleep time and immobility for young, middle-aged and elderly groups for intervention compared to baseline. Significant increase in sleep efficiency for elderly groups for intervention with baseline. Significant decrease in number of awakenings and nocturnal activity for young, middle-aged and elderly groups for intervention compared to baseline. Significant decrease in SOL in middle-aged and elderly groups for intervention compared with baseline. | Young, middle-aged and elderly participants |
| Saito (2017) [43] | RC, DB, P; Japan | 12 wk; 20–84 y; 94 (45/74/0) | Zinc-rich food (oysters) OR Zinc- and astaxanthin-rich food (oysters and krill)/placebo | Actigraphy | No significant difference in TST. Significant increase in SE for oysters compared with placebo. Significant improvement in SOL for oysters compared with placebo. | |
| Subjective (PSQI) | Significant improvement for sleep scores for all groups, not significantly different from placebo. | |||||
| Garrido (2010) [45] | CO; Spain | 7 × 3 dy; 35–85 y; 12 | Seven cultivars of Jerte Valley cherries 1 | Actigraphy | Significant increase in actual sleep time for six cultivars in middle-aged group. Significant increase in SE for Van cherries in middle-aged group. Significant decrease in number of awakenings for Pico Limón cherries in middle-aged group and Pico Colorado cherries in elderly group. Significant decrease in total nocturnal activity for Bourlat, Pico Limón, Pico Negro and Pico Colarado cherries for elderly group, and Bourlat cherries for middle-aged group. Significant decrease in SOL for Navalinda cherries in both groups and for Pico Negro cherries in elderly group. Significant increase in assumed sleep for Bourlat, Navalinda and Pico Negro cherries for elderly group, and for Van cherries in middle-aged group. Significant increase in immobility for Ambrunés, Pico Negro and Pico Colorado cherries for elderly group. | Middle-aged and elderly participants |
| Howatson (2011) [46] | RC, DB, P, CO; UK | 7 dy; 18–40 y; 20 (10/10/0) | Tart Montmorency cherry juice (Prunus cerasus) concentrate/placebo | Actigraphy | Significantly greater time in bed, TST and SE total for intervention compared with placebo and baseline. Non-significant decrease in SOL and fragmentation index and non-significant increase in SE for intervention. | |
| Subjective (Sleep diaries) | Significantly less napping time for intervention Non-significant increase in SET and TST and non-significant decrease in SOL and WASO for intervention. |
| Author (Year) | Design; Location | Duration; Age; Participants (M/F) | Intervention/Control | Sleep Measurements | Outcomes/Results | Notes |
|---|---|---|---|---|---|---|
| Driver (1999) [47] | RC; South Africa | 4 dy; 20–24 y; 7 (7/0) | High energy meal (fat 37%, protein 21%, CHO 42%) OR no meal/control | PSG | No significant difference for TST, SOL, TRT or ROL, or spent in each sleep stage. | |
| Subjective (Questionnaire) | No significant difference for subjective sleep quality. | |||||
| Afaghi (2007) [48] | RC, CO; Australia | 3 × 1 dy; 18–35 y; 12 (12/0) | High GI meals consumed either 4h or 1h before bedtime/control | PSG | Significant shortening of SOL for high GI meal consumed 4 h before bedtime compared with high GI meal consumed 1 hour before bedtime and low GI meal. No significant differences for other sleep parameters between meals or timing of meals. | |
| Afaghi (2008) [49] | CO; Australia | 5 dy; 18–35 y; 14 (14/0) | Very low CHO, high fat, high protein diet (acute and ketosis phases)/control | PSG | Arousal index significantly increased for NREM N1 and N2 during very low CHO phases. No significant difference for N 3 and N4 or REM. Significant increase in total sum of NREM N1-4, N3 and N4 and proportion to TST in very low CHO phases compared with control (due to increases in stage 4). Significant reduction in proportion of REM sleep to TST for very low CHO phases compared with control. | |
| Lindseth (2011) [50] | RC, DB, CO; US | 4 × 4 dy; 19–22 y; 44 | High protein diet OR high fat diet OR high CHO diet/control | Actigraphy | Significantly less wake episodes for high protein diet Significantly reduced SOL for high CHO diet No significant differences for high fat diet. | |
| Yajima (2014) [51] | CO; Japan | 2 dy; 24.6 ± 0.7 y; 10 (10/0) | High CHO meal OR high fat meal | PSG | No significant differences when sleep stages were averaged. Significant increase in N3 in first sleep stage for high CHO meal compared with high fat meal. | |
| Lindseth (2016) [52] | RC, CO; US | 4 × 4 dy; 20.9 ± 1.9; 36 | High protein OR high fat OR high carbohydrate meals/control | Actigraphy | Significantly shorter wake times after consuming high CHO diet. | |
| Subjective (PSQI) | Significantly lower global sleep quality score for high fat diet | |||||
| St Onge (2016) [53] | RC, CO; US | 5 dy; 30–45 yo; 26 (13/13) | Ad libitum/control | PSG | No significant difference between conditions for TST, absolute time in N1 and N2, and REM sleep. Significant increase of SOL for ad libitum compared with control. Significant reduction in N1 sleep and significant increase in N3 for fibre intake. Significant reduction in N3 for saturated fat. Significant increase in arousals for sugar and other non-sugar/non-fibre CHO. | |
| Zhou (2016) [54] | RC; CO; US | 3 × 4 wk; 14 (3/11) | Energy restricted diets with protein source either beef/pork (BP) or soy/legumes (SL) and either 10% (control), 20% or 30% of intake as protein | Subjective(PSQI) | Improved global sleep quality score for 20% protein intake. No difference in individual sleep domains. No difference in sleep quality between protein source. | Overweight or obese participants (BMI range 27 to 37.9) |
| Zhou (2016) [54] | RC; DB, P; US | 16 wk; 44 (12/32) | High protein energy-restricted diet/control | Subjective (PSQI) | Improvement in global sleep quality score for intervention at week 12 and 16. Greater use of sleep medication for control. No other difference in individual sleep domains. | Overweight or obese participants (BMI range 25 to 38) |
| Castro (2018) [55] | SA - Spain | 4 mo; 18–58 y; 20 (8/12) | High protein diet | Subjective (PSQI; ESS) | Significant improvement in sleepiness mid-intervention (reduced ketosis) No significant difference in sleep quality or sleep duration. | Obese participants (BMI ≥ 30) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binks, H.; E. Vincent, G.; Gupta, C.; Irwin, C.; Khalesi, S. Effects of Diet on Sleep: A Narrative Review. Nutrients 2020, 12, 936. https://doi.org/10.3390/nu12040936
Binks H, E. Vincent G, Gupta C, Irwin C, Khalesi S. Effects of Diet on Sleep: A Narrative Review. Nutrients. 2020; 12(4):936. https://doi.org/10.3390/nu12040936
Chicago/Turabian StyleBinks, Hannah, Grace E. Vincent, Charlotte Gupta, Christopher Irwin, and Saman Khalesi. 2020. "Effects of Diet on Sleep: A Narrative Review" Nutrients 12, no. 4: 936. https://doi.org/10.3390/nu12040936
APA StyleBinks, H., E. Vincent, G., Gupta, C., Irwin, C., & Khalesi, S. (2020). Effects of Diet on Sleep: A Narrative Review. Nutrients, 12(4), 936. https://doi.org/10.3390/nu12040936






