Schisandra Extract and Ascorbic Acid Synergistically Enhance Cognition in Mice through Modulation of Mitochondrial Respiration
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Plant Material and General Procedures of Natural Products
2.3. Isolation and Determination of Schisandrin from Schisandra Chinensis Extract
2.4. Oxygen Consumption Rate (OCR) Measurement
2.5. Animal Experiments
2.6. Behavioral Tests
2.7. Protein Extraction and Western Blot
2.8. Statistical Analysis
3. Results
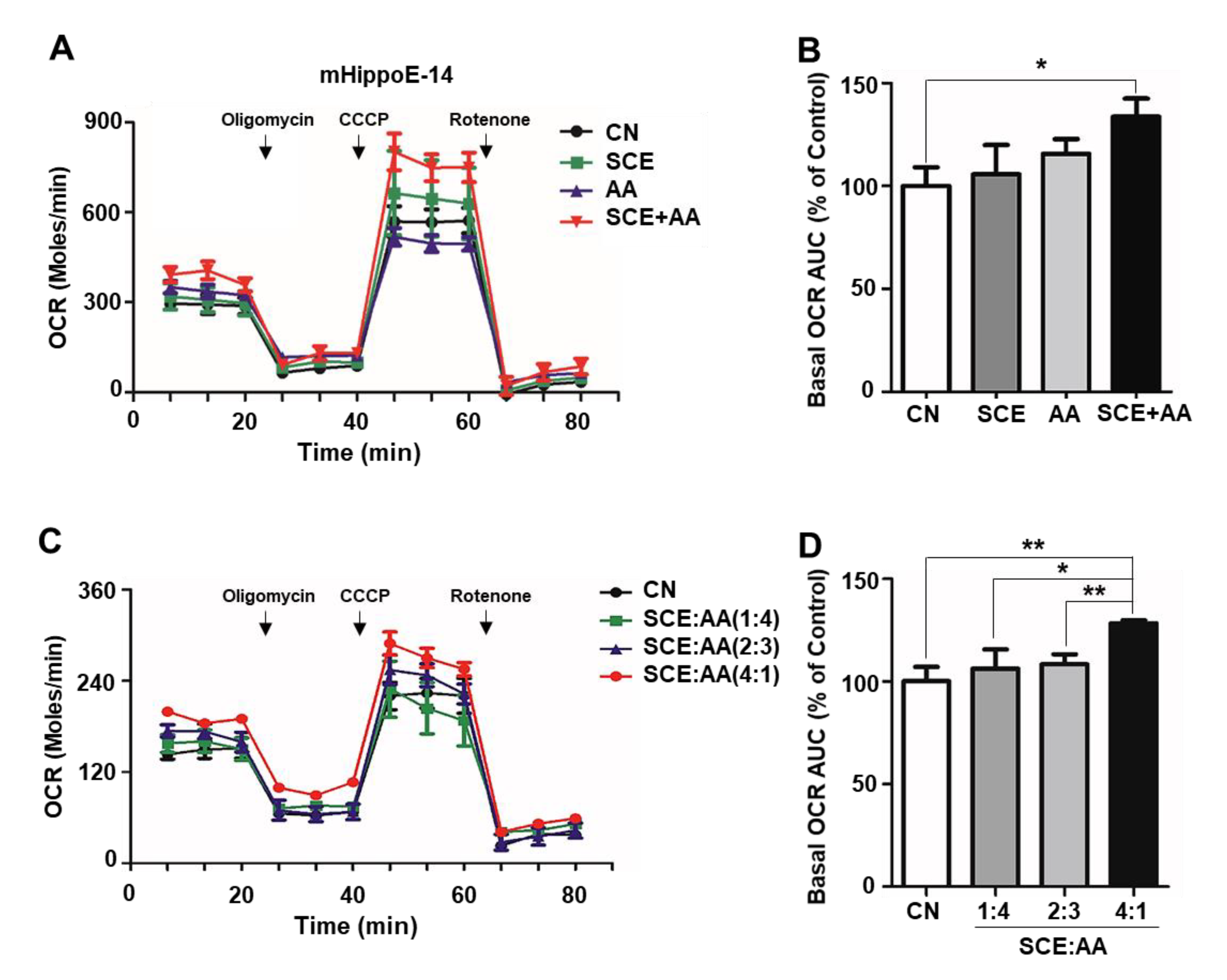
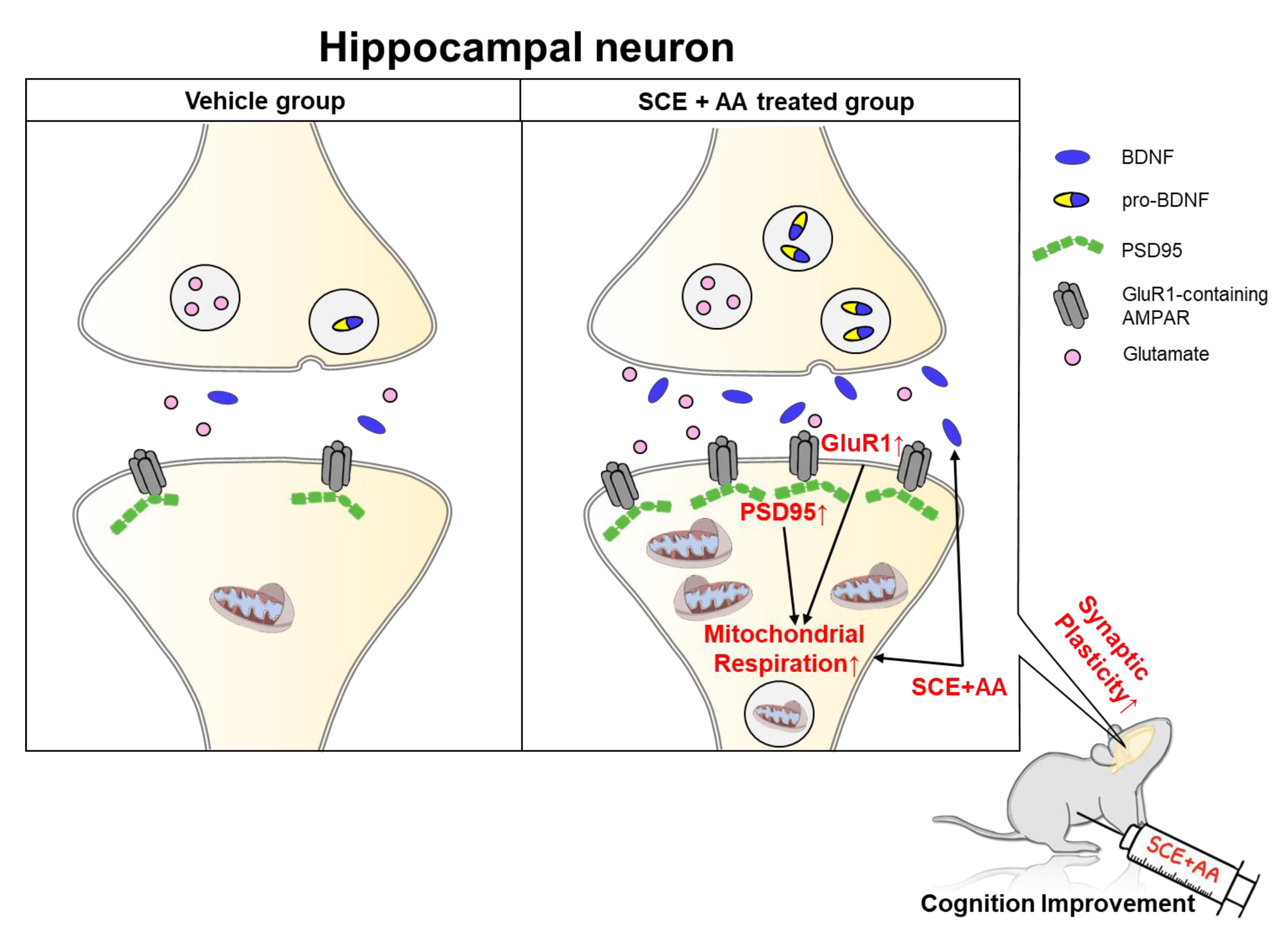
3.1. SCE-AA Mixture Enhances Mitochondrial Respiration in Hippocampal Neurons
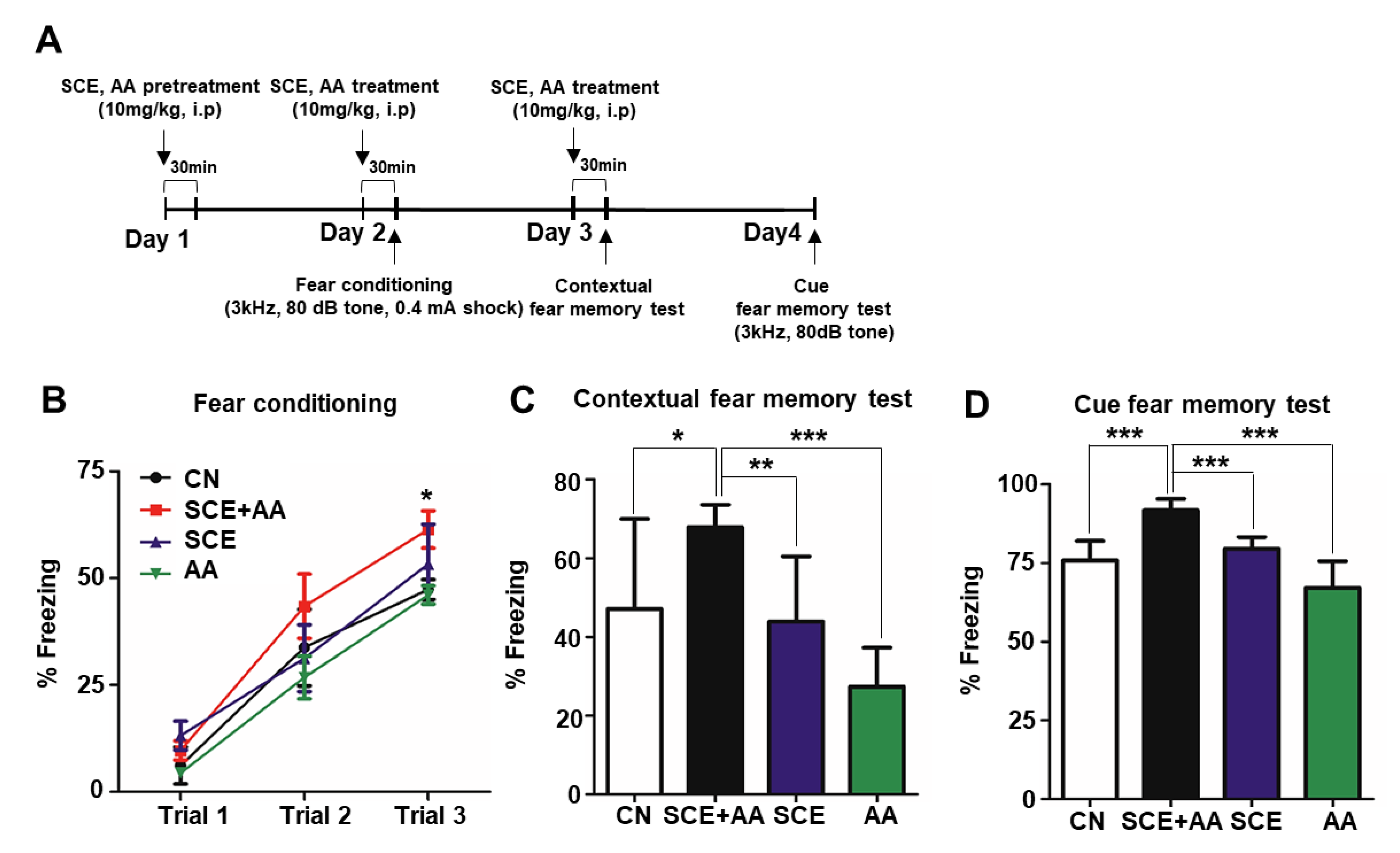
3.2. SCE-AA Mixture Improves Fear Learning and Memory in Mice
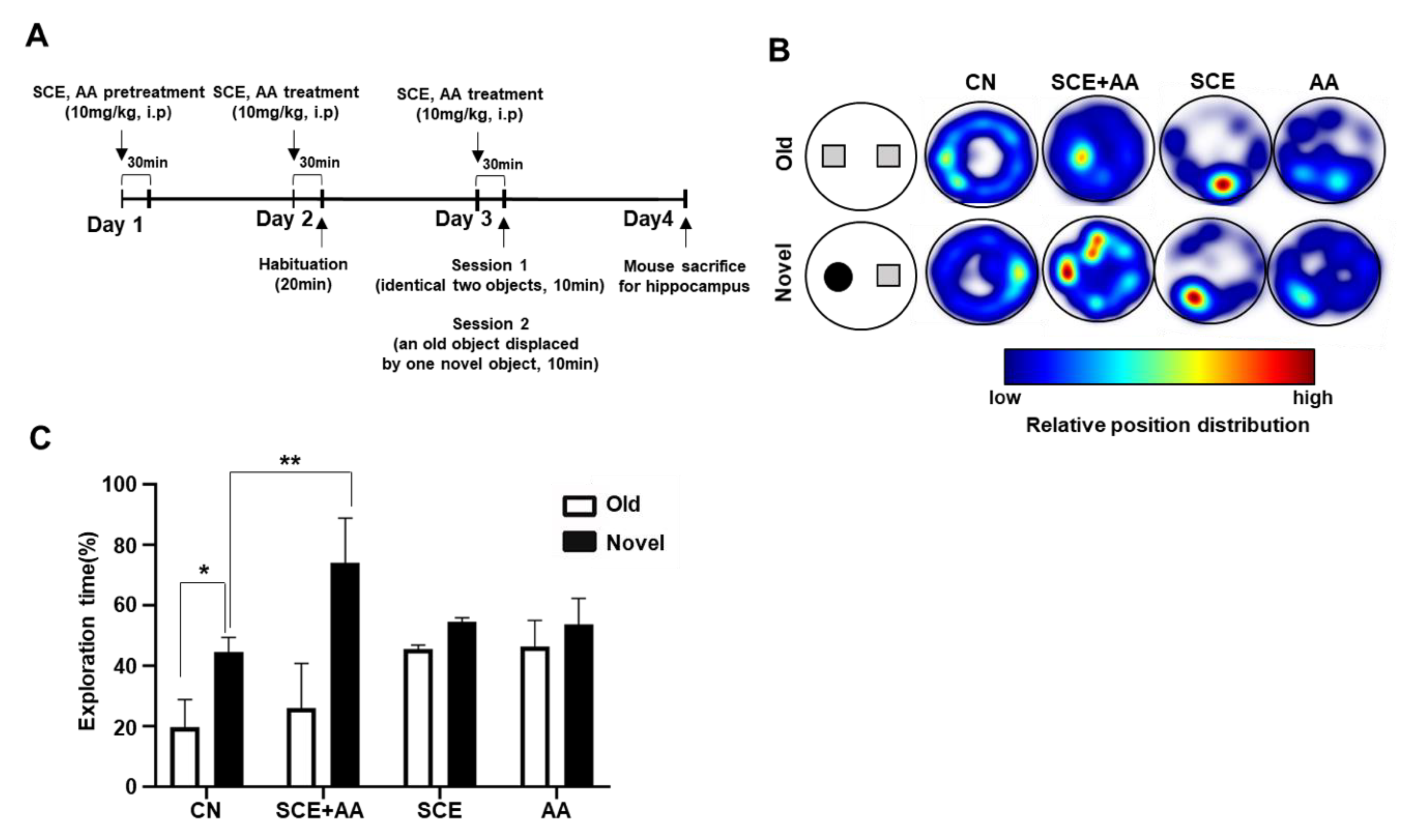
3.3. Administration of SCE-AA Mixture Enhances Recognition Memory in Mice
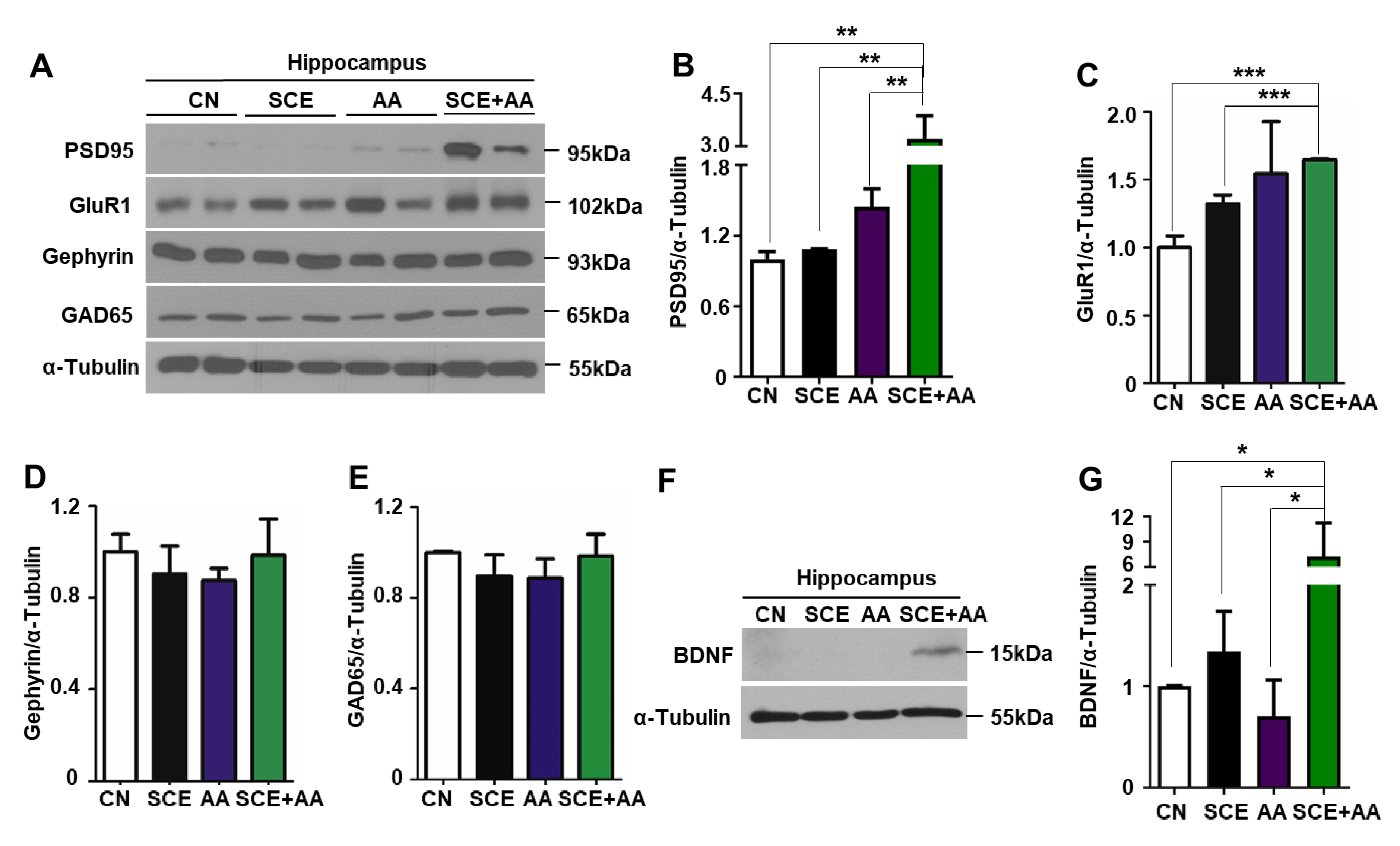
3.4. An SCE-AA Mixture Induces PSD95 Expression in the Hippocampus
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Todorova, V.; Blokland, A. Mitochondria and Synaptic Plasticity in the Mature and Aging Nervous System. Curr. Neuropharmacol. 2017, 15, 166–173. [Google Scholar] [CrossRef]
- Li, Z.; Okamoto, K.; Hayashi, Y.; Sheng, M. The Importance of Dendritic Mitochondria in the Morphogenesis and Plasticity of Spines and Synapses. Cell 2004, 119, 873–887. [Google Scholar] [CrossRef]
- Mancuso, M.; Calsolaro, V.; Orsucci, D.; Carlesi, C.; Choub, A.; Piazza, S.; Siciliano, G. Mitochondria, Cognitive Impairment, and Alzheimer’s Disease. Int. J. Alzheimer’s Dis. 2009, 2009, 1–8. [Google Scholar] [CrossRef][Green Version]
- Sawda, C.; Moussa, C.; Turner, R.S. Resveratrol for Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2017, 1403, 142–149. [Google Scholar] [CrossRef]
- Andrade, S.; Ramalho, M.J.; Loureiro, J.A.; Pereira, M.D.C. Natural Compounds for Alzheimer’s Disease Therapy: A Systematic Review of Preclinical and Clinical Studies. Int. J. Mol. Sci. 2019, 20, 2313. [Google Scholar] [CrossRef]
- Arab, H.; Mahjoub, S.; Hajian-Tilaki, K.; Moghadasi, M. The effect of green tea consumption on oxidative stress markers and cognitive function in patients with Alzheimer’s disease: A prospective intervention study. Casp. J. Intern. Med. 2016, 7, 188–194. [Google Scholar]
- Hassan, M.; Raza, H.; Abbasi, M.A.; Moustafa, A.; Seo, S.-Y. The exploration of novel Alzheimer’s therapeutic agents from the pool of FDA approved medicines using drug repositioning, enzyme inhibition and kinetic mechanism approaches. Biomed. Pharmacother. 2019, 109, 2513–2526. [Google Scholar] [CrossRef]
- Fu, W.-Y.; Wang, X.; Ip, N.Y.-Y. Targeting Neuroinflammation as a Therapeutic Strategy for Alzheimer’s Disease: Mechanisms, Drug Candidates, and New Opportunities. ACS Chem. Neurosci. 2018, 10, 872–879. [Google Scholar] [CrossRef]
- Sestito, S.; Daniele, S.; Pietrobono, D.; Citi, V.; Bellusci, L.; Chiellini, G.; Calderone, V.; Martini, C.; Rapposelli, S. Memantine prodrug as a new agent for Alzheimer’s Disease. Sci. Rep. 2019, 9, 4612. [Google Scholar] [CrossRef]
- Nowak, A.; Zakłos-Szyda, M.; Blasiak, J.; Nowak, A.; Zhang, Z.; Zhang, B. Potential of Schisandra chinensis (Turcz.) Baill. in Human Health and Nutrition: A Review of Current Knowledge and Therapeutic Perspectives. Nutrients 2019, 11, 333. [Google Scholar] [CrossRef]
- Yan, T.; He, B.; Wan, S.; Xu, M.; Yang, H.; Xiao, F.; Bi, K.; Jia, Y. Antidepressant-like effects and cognitive enhancement of Schisandra chinensis in chronic unpredictable mild stress mice and its related mechanism. Sci. Rep. 2017, 7, 6903. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Cai, Z.; Xiao, P.; Li, C. Schisandra N-butanol extract improves synaptic morphology and plasticity in ovarectomized mice. Neural. Regen. Res. 2012, 7, 1365–1369. [Google Scholar] [PubMed]
- Chen, Q.; Zhan, Q.; Li, Y.; Sun, S.; Zhao, L.; Zhang, H.; Zhang, G. Schisandra Lignan Extract Protects against Carbon Tetrachloride-Induced Liver Injury in Mice by Inhibiting Oxidative Stress and Regulating the NF-kappaB and JNK Signaling Pathways. Evid. Based Complement Altern. Med. 2017, 2017, 5140297. [Google Scholar] [CrossRef]
- Karamian, R.; Komaki, A.; Salehi, I.; Tahmasebi, L.; Komaki, H.; Shahidi, S.; Sarihi, A. Vitamin C reverses lead-induced deficits in hippocampal synaptic plasticity in rats. Brain Res. Bull. 2015, 116, 7–15. [Google Scholar] [CrossRef]
- Caesar, L.K.; Cech, N.B. Synergy and antagonism in natural product extracts: When 1 + 1 does not equal 2. Nat. Prod. Rep. 2019, 36, 869–888. [Google Scholar] [CrossRef]
- Zhou, X.; Seto, S.W.; Chang, D.; Kiat, H.; Razmovski-Naumovski, V.; Chan, K.; Bensoussan, A. Synergistic Effects of Chinese Herbal Medicine: A Comprehensive Review of Methodology and Current Research. Front. Pharmacol. 2016, 7, 117. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, J.H.; Park, H.S.; Lee, G.S.; Kim, H.W.; Ha, K.-T.; Kim, B.J. Schizandra chinensisextracts induce apoptosis in human gastric cancer cells via JNK/p38 MAPK activation and the ROS-mediated/mitochondria-dependent pathway. Pharm. Boil. 2014, 53, 212–219. [Google Scholar] [CrossRef]
- Santos, Í.M.S.; Tomé, A.D.R.; Saldanha, G.B.; Ferreira, P.M.P.; Militão, G.C.G.; De Freitas, R.M. Oxidative stress in the hippocampus during experimental seizures can be ameliorated with the antioxidant ascorbic acid. Oxidative Med. Cell. Longev. 2009, 2, 214–221. [Google Scholar] [CrossRef]
- De Assis, S.A.; Vellosa, J.C.R.; Brunetti, I.L.; Khalil, N.M.; Leite, K.M.D.S.C.; Martins, A.B.G.; Oliveira, O.M.M.D.F. Antioxidant activity, ascorbic acid and total phenol of exotic fruits occurring in Brazil. Int. J. Food Sci. Nutr. 2009, 60, 439–448. [Google Scholar] [CrossRef]
- Takanche, J.S.; Kim, J.-S.; Kim, J.-E.; Han, S.-H.; Yi, H.-K. Schisandrin C enhances odontoblastic differentiation through autophagy and mitochondrial biogenesis in human dental pulp cells. Arch. Oral Boil. 2018, 88, 60–66. [Google Scholar] [CrossRef]
- Gonzalez-Lima, F.; Barksdale, B.R.; Rojas, J.C. Mitochondrial respiration as a target for neuroprotection and cognitive enhancement. Biochem. Pharmacol. 2014, 88, 584–593. [Google Scholar] [CrossRef]
- Gewirtz, J.C.; McNish, K.A.; Davis, M. Is the hippocampus necessary for contextual fear conditioning? Behav. Brain Res. 2000, 110, 83–95. [Google Scholar] [CrossRef]
- Broadbent, N.J.; Gaskin, S.; Squire, L.R.; Clark, R.E. Object recognition memory and the rodent hippocampus. Learn. Mem. 2009, 17, 5–11. [Google Scholar] [CrossRef]
- Sanchez, C.O.; Amin, E.; Warburton, E.C.; Aggleton, J.P. Perirhinal cortex lesions impair tests of object recognition memory but spare novelty detection. Eur. J. Neurosci. 2015, 42, 3117–3127. [Google Scholar] [CrossRef]
- Cohen, S.J.; Robert, W.S., Jr. Assessing rodent hippocampal involvement in the novel object recognition task. A review. Behav. Brain Res. 2014, 285, 105–117. [Google Scholar] [CrossRef]
- Licznerski, P.; Jonas, E.A. BDNF signaling: Harnessing stress to battle mood disorder. Proc. Natl. Acad. Sci. USA 2018, 115, 3742–3744. [Google Scholar] [CrossRef]
- Yoshii, A.; Constantine-Paton, M. BDNF induces transport of PSD-95 to dendrites through PI3K-AKT signaling after NMDA receptor activation. Nat. Neurosci. 2007, 10, 702–711. [Google Scholar] [CrossRef]
- Benarroch, E.E. Glutamatergic synaptic plasticity and dysfunction in Alzheimer disease: Emerging mechanisms. Neurology 2018, 91, 125–132. [Google Scholar] [CrossRef]
- Sowndhararajan, K.; Deepa, P.; Kim, M.; Park, S.J.; Kim, S. An overview of neuroprotective and cognitive enhancement properties of lignans from Schisandra chinensis. Biomed. Pharmacother. 2018, 97, 958–968. [Google Scholar] [CrossRef]
- Dixit, S.; Bernardo, A.; Walker, J.M.; Kennard, J.A.; Kim, G.Y.; Kessler, E.S.; Harrison, F.; Walker, M.J. Vitamin C Deficiency in the Brain Impairs Cognition, Increases Amyloid Accumulation and Deposition, and Oxidative Stress in APP/PSEN1 and Normally Aging Mice. ACS Chem. Neurosci. 2015, 6, 570–581. [Google Scholar] [CrossRef]
- Tamari, Y.; Nawata, H.; Inoue, E.; Yoshimura, A.; Yoshii, H.; Kashino, G.; Seki, M.; Enomoto, T.; Watanabe, M.; Tano, K. Protective roles of ascorbic acid in oxidative stress induced by depletion of superoxide dismutase in vertebrate cells. Free. Radic. Res. 2012, 47, 1–7. [Google Scholar] [CrossRef]
- Jain, D.P.; Pancholi, S.S.; Patel, R. Synergistic antioxidant activity of green tea with some herbs. J. Adv. Pharm. Technol. Res. 2011, 2, 177–183. [Google Scholar] [CrossRef]
- Manap, A.S.A.; Tan, A.C.W.; Leong, W.H.; Chia, A.Y.Y.; Vijayabalan, S.; Arya, A.; Wong, E.H.; Rizwan, F.; Bindal, U.; Koshy, S.; et al. Synergistic Effects of Curcumin and Piperine as Potent Acetylcholine and Amyloidogenic Inhibitors With Significant Neuroprotective Activity in SH-SY5Y Cells via Computational Molecular Modeling and in vitro Assay. Front. Aging Neurosci. 2019, 11, 206. [Google Scholar] [CrossRef]
- Pei, L.; Mu, Y.; Leblanc, M.; Alaynick, W.; Barish, G.D.; Pankratz, M.; Tseng, T.W.; Kaufman, S.; Liddle, C.; Yu, R.T.; et al. Dependence of hippocampal function on ERRgamma-regulated mitochondrial metabolism. Cell Metab. 2015, 21, 628–636. [Google Scholar] [CrossRef]
- Pintana, H.; Apaijai, N.; Chattipakorn, N.; Chattipakorn, S. DPP-4 inhibitors improve cognition and brain mitochondrial function of insulin-resistant rats. J. Endocrinol. 2013, 218, 1–11. [Google Scholar] [CrossRef]
- Cousins, S.L.; Stephenson, F.A. Identification of N-Methyl-d-aspartic Acid (NMDA) Receptor Subtype-specific Binding Sites That Mediate Direct Interactions with Scaffold Protein PSD-95. J. Boil. Chem. 2012, 287, 13465–13476. [Google Scholar] [CrossRef]
- Sheng, M. The postsynaptic NMDA-receptor--PSD-95 signaling complex in excitatory synapses of the brain. J. Cell Sci. 2001, 114, 1251. [Google Scholar]
- Proctor, D.T.; Coulson, E.J.; Dodd, P.R. Reduction in Post-Synaptic Scaffolding PSD-95 and SAP-102 Protein Levels in the Alzheimer Inferior Temporal Cortex is Correlated with Disease Pathology. J. Alzheimer’s Dis. 2010, 21, 795–811. [Google Scholar] [CrossRef]
- Savioz, A.; Leuba, G.; Vallet, P.G. A framework to understand the variations of PSD-95 expression in brain aging and in Alzheimer’s disease. Ageing Res. Rev. 2014, 18, 86–94. [Google Scholar] [CrossRef]
- Lee, H.-K.; Takamiya, K.; He, K.; Song, L.; Huganir, R.L. Specific roles of AMPA receptor subunit GluR1 (GluA1) phosphorylation sites in regulating synaptic plasticity in the CA1 region of hippocampus. J. Neurophysiol. 2009, 103, 479–489. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Narisawa-Saito, M.; Iwakura, Y.; Arai, T.; Ikeda, K.; Takahashi, H.; Nawa, H. Phenotypic down-regulation of glutamate receptor subunit GluR1 in Alzheimer’s disease. Neurobiol. Aging 1999, 20, 287–295. [Google Scholar] [CrossRef]
- Chen, F.; Danladi, J.; Ardalan, M.; Elfving, B.; Müller, H.K.; Wegener, G.; Sánchez, C.; Nyengaard, J.R. A Critical Role of Mitochondria in BDNF-Associated Synaptic Plasticity after One-Week Vortioxetine Treatment. Int. J. Neuropsychopharmacol. 2018, 21, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Ardalan, M.; Elfving, B.; Wegener, G.; Madsen, T.M.; Nyengaard, J.R. Mitochondria Are Critical for BDNF-Mediated Synaptic and Vascular Plasticity of Hippocampus following Repeated Electroconvulsive Seizures. Int. J. Neuropsychopharmacol. 2017, 21, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.A.; Roberts, K.N.; Scheff, S.W. Oxidative stress and modification of synaptic proteins in hippocampus after traumatic brain injury. Free. Radic. Boil. Med. 2008, 45, 443–452. [Google Scholar] [CrossRef]
- Jiao, Y.; Fan, H.; Wang, K.; Lu, S. Sevoflurane Impairs Short-Term Memory by Affecting PSD-95 and AMPA Receptor in the Hippocampus of a Mouse Model. Behav. Neurol. 2019, 2019, 1068260. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, Y.; Lee, J.H.; Lee, M.J.; Kim, S.J.; Ju, X.; Cui, J.; Zhu, J.; Lee, Y.L.; Namgung, E.; Sung, H.W.J.; et al. Schisandra Extract and Ascorbic Acid Synergistically Enhance Cognition in Mice through Modulation of Mitochondrial Respiration. Nutrients 2020, 12, 897. https://doi.org/10.3390/nu12040897
Jang Y, Lee JH, Lee MJ, Kim SJ, Ju X, Cui J, Zhu J, Lee YL, Namgung E, Sung HWJ, et al. Schisandra Extract and Ascorbic Acid Synergistically Enhance Cognition in Mice through Modulation of Mitochondrial Respiration. Nutrients. 2020; 12(4):897. https://doi.org/10.3390/nu12040897
Chicago/Turabian StyleJang, Yunseon, Jae Hyeon Lee, Min Joung Lee, Soo Jeong Kim, Xianshu Ju, Jianchen Cui, Jiebo Zhu, Yu Lim Lee, Eunji Namgung, Han Wool John Sung, and et al. 2020. "Schisandra Extract and Ascorbic Acid Synergistically Enhance Cognition in Mice through Modulation of Mitochondrial Respiration" Nutrients 12, no. 4: 897. https://doi.org/10.3390/nu12040897
APA StyleJang, Y., Lee, J. H., Lee, M. J., Kim, S. J., Ju, X., Cui, J., Zhu, J., Lee, Y. L., Namgung, E., Sung, H. W. J., Lee, H. W., Ryu, M. J., Oh, E., Chung, W., Kweon, G. R., Choi, C. W., & Heo, J. Y. (2020). Schisandra Extract and Ascorbic Acid Synergistically Enhance Cognition in Mice through Modulation of Mitochondrial Respiration. Nutrients, 12(4), 897. https://doi.org/10.3390/nu12040897




