Efficacy of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study
Abstract
1. Introduction
2. Materials and Methods
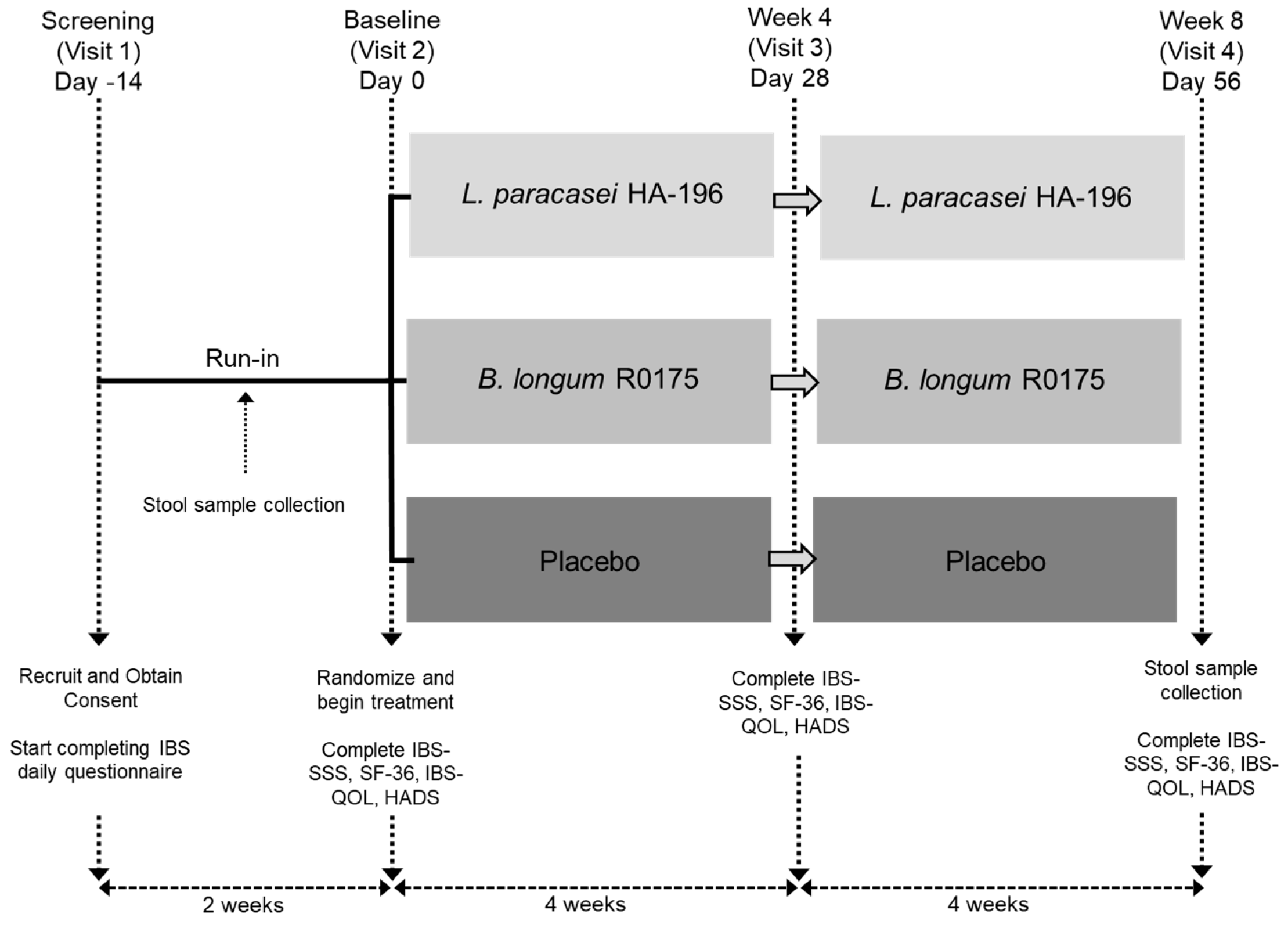
2.1. Study Design
2.2. Participants
2.3. Interventions
2.4. Randomization and Blinding
2.5. Assessments of IBS Symptom Severity, General Health and Psychological Well-Being
2.6. Fecal Microbe Composition Analysis
2.7. Compliance
2.8. Statistical Analyses
3. Results
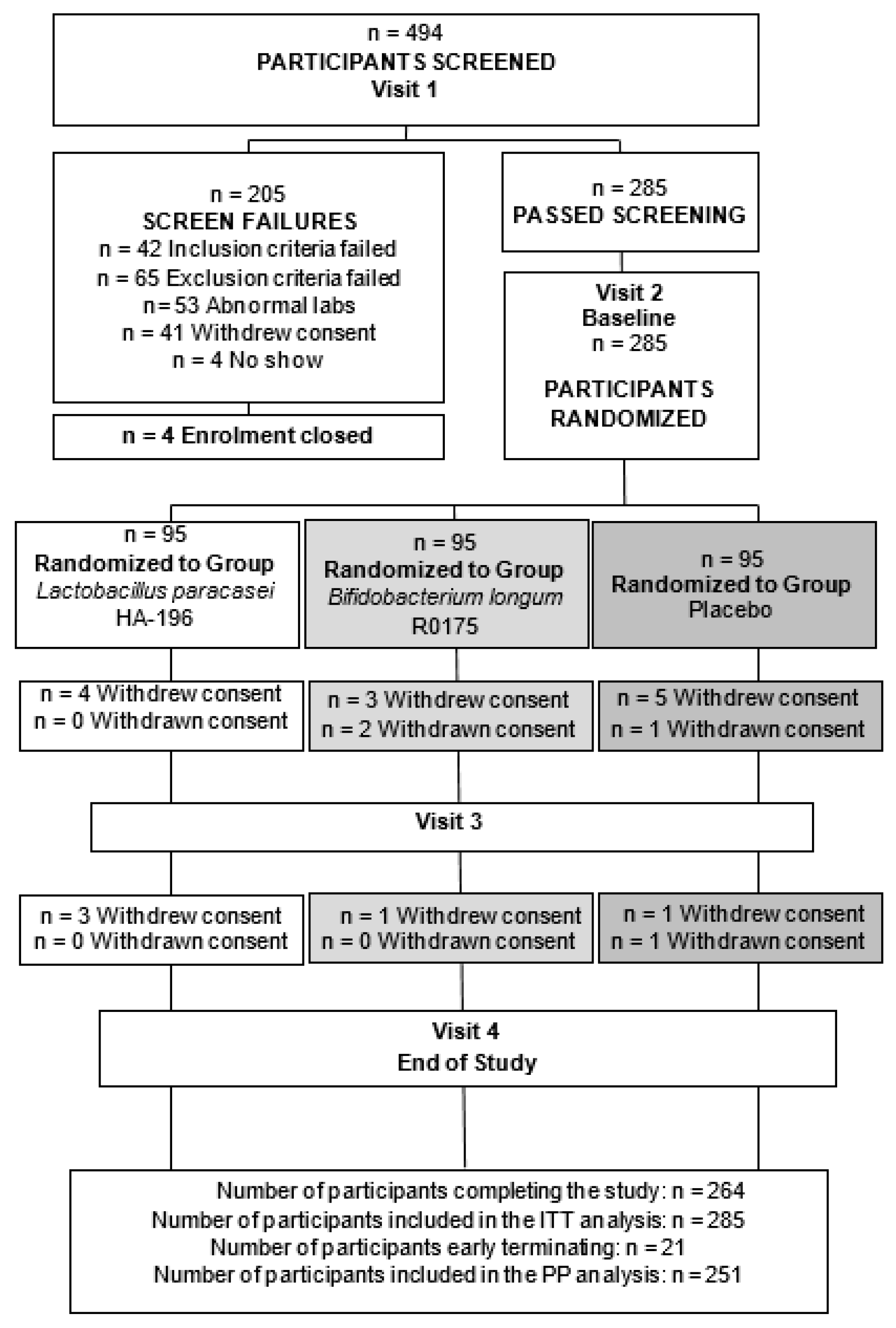
3.1. Study Participant Dispositions
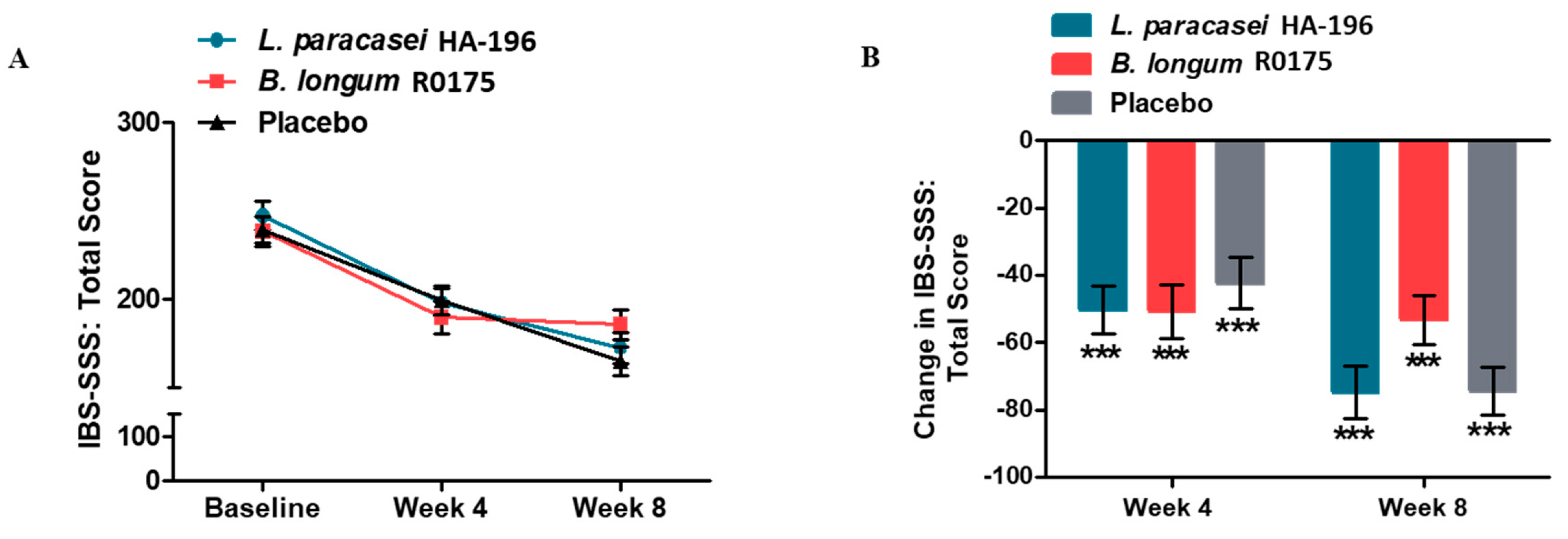
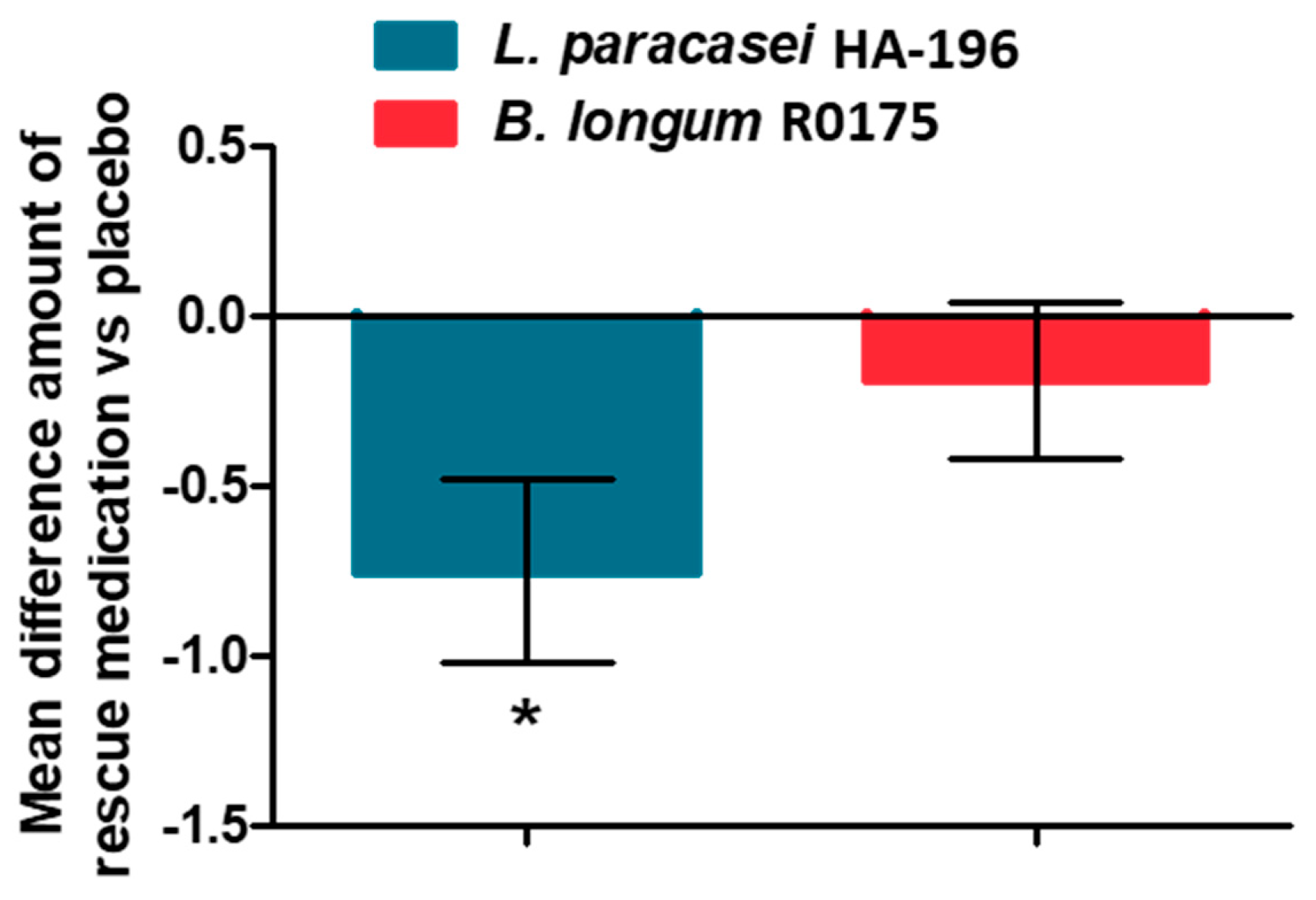
3.2. Improvement in the IBS Symptom Severity Score and Rescue Medication Use
3.3. Increase in Bowel Movement Frequency in Participants with IBS-C
3.4. Decrease in the Bowel Movement Frequency in Participants with IBS-D
3.5. No Significant Changes in the Bowel Movement Frequency in Participants with IBS-M
3.6. Improvements in Mental Health Measures in Participants Given Probiotics
3.7. L. paracasei and B. longum are Safe to Consume
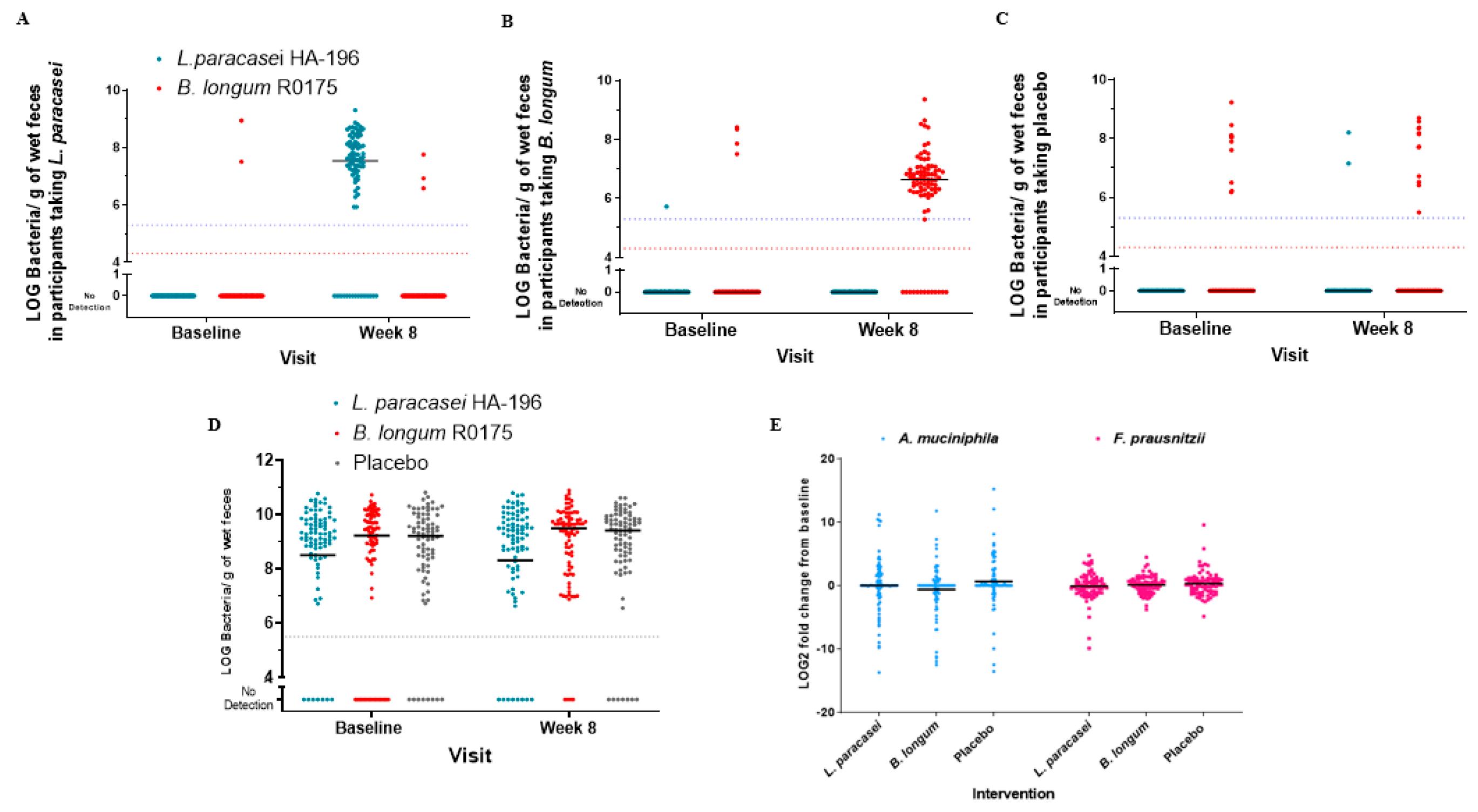
3.8. High Compliance and Increased Number of Participants with Bifidobacterium Species
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Card, T.; Canavan, C.; West, J. The epidemiology of irritable bowel syndrome. Clin. Epidemiol. 2014, 6, 71. [Google Scholar] [CrossRef] [PubMed]
- Sultan, S.; Malhotra, A. Irritable Bowel Syndrome. Ann. Intern. Med. 2017, 166, Itc81–Itc96. [Google Scholar] [CrossRef] [PubMed]
- Occhipinti, K.; Smith, J. Irritable Bowel Syndrome: A Review and Update. Clin. Colon Rectal Surg. 2012, 25, 46–52. [Google Scholar] [CrossRef]
- Lewis, J.H. The risk of ischaemic colitis in irritable bowel syndrome patients treated with serotonergic therapies. Drug Saf. 2011, 34, 545–565. [Google Scholar] [CrossRef]
- Munjal, A.; Dedania, B.; Cash, B. Update on Pharmacotherapy for Irritable Bowel Syndrome. Curr. Gastroenterol. Rep. 2019, 21, 25. [Google Scholar] [CrossRef] [PubMed]
- Tap, J.; Derrien, M.; Törnblom, H.; Brazeilles, R.; Cools-Portier, S.; Doré, J.; Störsrud, S.; Le Nevé, B.; Öhman, L.; Simrén, M. Identification of an Intestinal Microbiota Signature Associated With Severity of Irritable Bowel Syndrome. Gastroenterology 2017, 152, 111–123.e8. [Google Scholar] [CrossRef]
- Lee, B.J.; Bak, Y.-T. Irritable Bowel Syndrome, Gut Microbiota and Probiotics. J. Neurogastroenterol. Motil. 2011, 17, 252–266. [Google Scholar] [CrossRef]
- Kerckhoffs, A.P.; Samsom, M.; van der Rest, M.E.; de Vogel, J.; Knol, J.; Ben-Amor, K.; Akkermans, L.M. Lower Bifidobacteria counts in both duodenal mucosa-associated and fecal microbiota in irritable bowel syndrome patients. World J. Gastroenterol. 2009, 15, 2887–2892. [Google Scholar] [CrossRef]
- Sinn, D.H.; Song, J.H.; Kim, H.J.; Lee, J.H.; Son, H.J.; Chang, D.K.; Kim, Y.H.; Kim, J.J.; Rhee, J.C.; Rhee, P.L. Therapeutic effect of Lactobacillus acidophilus-SDC 2012, 2013 in patients with irritable bowel syndrome. Dig. Dis. Sci. 2008, 53, 2714–2718. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Invernici, M.M.; Furlaneto, F.A.C.; Messora, M.R. Effectiveness of Multi-strain Versus Single-strain Probiotics: Current Status and Recommendations for the Future. J. Clin. Gastroenterol. 2018, 52, S35–S40. [Google Scholar] [CrossRef]
- Barbara, G.; Cremon, C.; Azpiroz, F. Probiotics in irritable bowel syndrome: Where are we? Neurogastroenterol. Motil. 2018, 30, e13513. [Google Scholar] [CrossRef]
- Fanigliulo, L.; Comparato, G.; Aragona, G.; Cavallaro, L.; Iori, V.; Maino, M.; Cavestro, G.M.; Soliani, P.; Sianesi, M.; Franze, A.; et al. Role of gut microflora and probiotic effects in the irritable bowel syndrome. Acta Biomed. 2006, 77, 85–89. [Google Scholar]
- Lombardo, L.; Vernetto, A.; Blanco, I. Clinical evaluation of Lactobacillus paracasei subsp. paracasei F19 with gluco-oligosaccharides in the short-term treatment of irritable bowel syndrome. Microb. Ecol. Health Dis. 2009, 21, 28–32. [Google Scholar] [CrossRef]
- McFarland, L.V.; Evans, C.T.; Goldstein, E.J.C. Strain-Specificity and Disease-Specificity of Probiotic Efficacy: A Systematic Review and Meta-Analysis. Front. Med. (Lausanne) 2018, 5, 124. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, M.; Lalonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.-F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M.; et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, A.; Noorbala, A.A.; Azam, K.; Eskandari, M.H.; Djafarian, K. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin. Nutr. 2019, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gotzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. Int. J. Surg. 2012, 10, 28–55. [Google Scholar] [CrossRef] [PubMed]
- Blake, M.R.; Raker, J.M.; Whelan, K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharm. Ther. 2016, 44, 693–703. [Google Scholar] [CrossRef]
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional bowel disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef]
- Brazier, J.E.; Harper, R.; Jones, N.M.; O’Cathain, A.; Thomas, K.J.; Usherwood, T.; Westlake, L. Validating the SF-36 health survey questionnaire: New outcome measure for primary care. BMJ 1992, 305, 160–164. [Google Scholar] [CrossRef]
- Williams, V.S.; Nelson, L.M.; Fehnel, S.E.; MacDougall, J.; Carson, R.T.; Tourkodimitris, S.; Kurtz, C.; Baird, M.J.; Johnston, J.M. Psychometric validation of symptom severity measures in irritable bowel syndrome with constipation. Aliment. Pharm. Ther. 2014, 40, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Derrien, M.; Collado, M.C.; Ben-Amor, K.; Salminen, S.; de Vos, W.M. The Mucin degrader Akkermansia muciniphila is an abundant resident of the human intestinal tract. Appl. Environ. Microbiol. 2008, 74, 1646–1648. [Google Scholar] [CrossRef]
- Lopez-Siles, M.; Martinez-Medina, M.; Busquets, D.; Sabat-Mir, M.; Duncan, S.H.; Flint, H.J.; Aldeguer, X.; Garcia-Gil, L.J. Mucosa-associated Faecalibacterium prausnitzii and Escherichia coli co-abundance can distinguish Irritable Bowel Syndrome and Inflammatory Bowel Disease phenotypes. Int. J. Med. Microbiol. 2014, 304, 464–475. [Google Scholar] [CrossRef] [PubMed]
- Hartman, A.L.; Lough, D.M.; Barupal, D.K.; Fiehn, O.; Fishbein, T.; Zasloff, M.; Eisen, J.A. Human gut microbiome adopts an alternative state following small bowel transplantation. Proc. Natl. Acad. Sci. USA 2009, 106, 17187–17192. [Google Scholar] [CrossRef]
- Dapoigny, M.; Piche, T.; Ducrotte, P.; Lunaud, B.; Cardot, J.M.; Bernalier-Donadille, A. Efficacy and safety profile of LCR35 complete freeze-dried culture in irritable bowel syndrome: A randomized, double-blind study. World J. Gastroenterol. 2012, 18, 2067–2075. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.A.; Stimpson, J.; Wang, D.; Plummer, S.; Garaiova, I.; Barker, M.E.; Corfe, B.M. Clinical trial: A multistrain probiotic preparation significantly reduces symptoms of irritable bowel syndrome in a double-blind placebo-controlled study. Aliment. Pharm. Ther. 2009, 29, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Saha, L. Irritable bowel syndrome: Pathogenesis, diagnosis, treatment, and evidence-based medicine. World J. Gastroenterol. 2014, 20, 6759–6773. [Google Scholar] [CrossRef]
- Lee, Y.T.; Hu, L.Y.; Shen, C.C.; Huang, M.W.; Tsai, S.J.; Yang, A.C.; Hu, C.K.; Perng, C.L.; Huang, Y.S.; Hung, J.H. Risk of Psychiatric Disorders following Irritable Bowel Syndrome: A Nationwide Population-Based Cohort Study. PLoS ONE 2015, 10, e0133283. [Google Scholar] [CrossRef]
- Bastiaanssen, T.F.S.; Cowan, C.S.M.; Claesson, M.J.; Dinan, T.G.; Cryan, J.F. Making Sense of … the Microbiome in Psychiatry. Int. J. Neuropsychopharmacol. 2019, 22, 37–52. [Google Scholar] [CrossRef]
- Nadeem, I.; Rahman, M.Z.; Ad-Dab’bagh, Y.; Akhtar, M. Effect of probiotic interventions on depressive symptoms: A narrative review evaluating systematic reviews. Psychiatry Clin. Neurosci. 2019, 73, 154–162. [Google Scholar] [CrossRef]
- Pinto-Sanchez, M.I.; Hall, G.B.; Ghajar, K.; Nardelli, A.; Bolino, C.; Lau, J.T.; Martin, F.P.; Cominetti, O.; Welsh, C.; Rieder, A.; et al. Probiotic Bifidobacterium longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients With Irritable Bowel Syndrome. Gastroenterology 2017, 153, 448–459.e8. [Google Scholar] [CrossRef] [PubMed]
- Aragon, G.; Graham, D.B.; Borum, M.; Doman, D.B. Probiotic therapy for irritable bowel syndrome. Gastroenterol. Hepatol. (N Y) 2010, 6, 39–44. [Google Scholar]
- Cruz-Aguliar, R.M.; Wantia, N.; Clavel, T.; Vehreschild, M.; Buch, T.; Bajbouj, M.; Haller, D.; Busch, D.; Schmid, R.M.; Stein-Thoeringer, C.K. An Open-Labeled Study on Fecal Microbiota Transfer in Irritable Bowel Syndrome Patients Reveals Improvement in Abdominal Pain Associated with the Relative Abundance of Akkermansia Muciniphila. Digestion 2019, 100, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Miquel, S.; Martin, R.; Lashermes, A.; Gillet, M.; Meleine, M.; Gelot, A.; Eschalier, A.; Ardid, D.; Bermudez-Humaran, L.G.; Sokol, H.; et al. Anti-nociceptive effect of Faecalibacterium prausnitzii in non-inflammatory IBS-like models. Sci. Rep. 2016, 6, 19399. [Google Scholar] [CrossRef]
- Dorn, S.D.; Kaptchuk, T.J.; Park, J.B.; Nguyen, L.T.; Canenguez, K.; Nam, B.H.; Woods, K.B.; Conboy, L.A.; Stason, W.B.; Lembo, A.J. A meta-analysis of the placebo response in complementary and alternative medicine trials of irritable bowel syndrome. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2007, 19, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.M.; Stason, W.B.; Legedza, A.; Ock, S.M.; Kaptchuk, T.J.; Conboy, L.; Canenguez, K.; Park, J.K.; Kelly, E.; Jacobson, E.; et al. The placebo effect in irritable bowel syndrome trials: A meta-analysis. Neurogastroenterol. Motil. 2005, 17, 332–340. [Google Scholar] [CrossRef]
- Drossman, D.A. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features, and Rome IV. Gastroenterology 2016, 150, 1262–1279. [Google Scholar] [CrossRef]
- Black, C.J.; Yiannakou, Y.; Houghton, L.A.; Ford, A.C. Epidemiological, Clinical, and Psychological Characteristics of Individuals with Self-reported Irritable Bowel Syndrome Based on the Rome IV vs Rome III Criteria. Clin. Gastroenterol. Hepatol. 2020, 18, 392–398. [Google Scholar] [CrossRef]
- Zhang, L.; Song, J.; Hou, X. Mast Cells and Irritable Bowel Syndrome: From the Bench to the Bedside. J. Neurogastroenterol. Motil. 2016, 22, 181–192. [Google Scholar] [CrossRef]
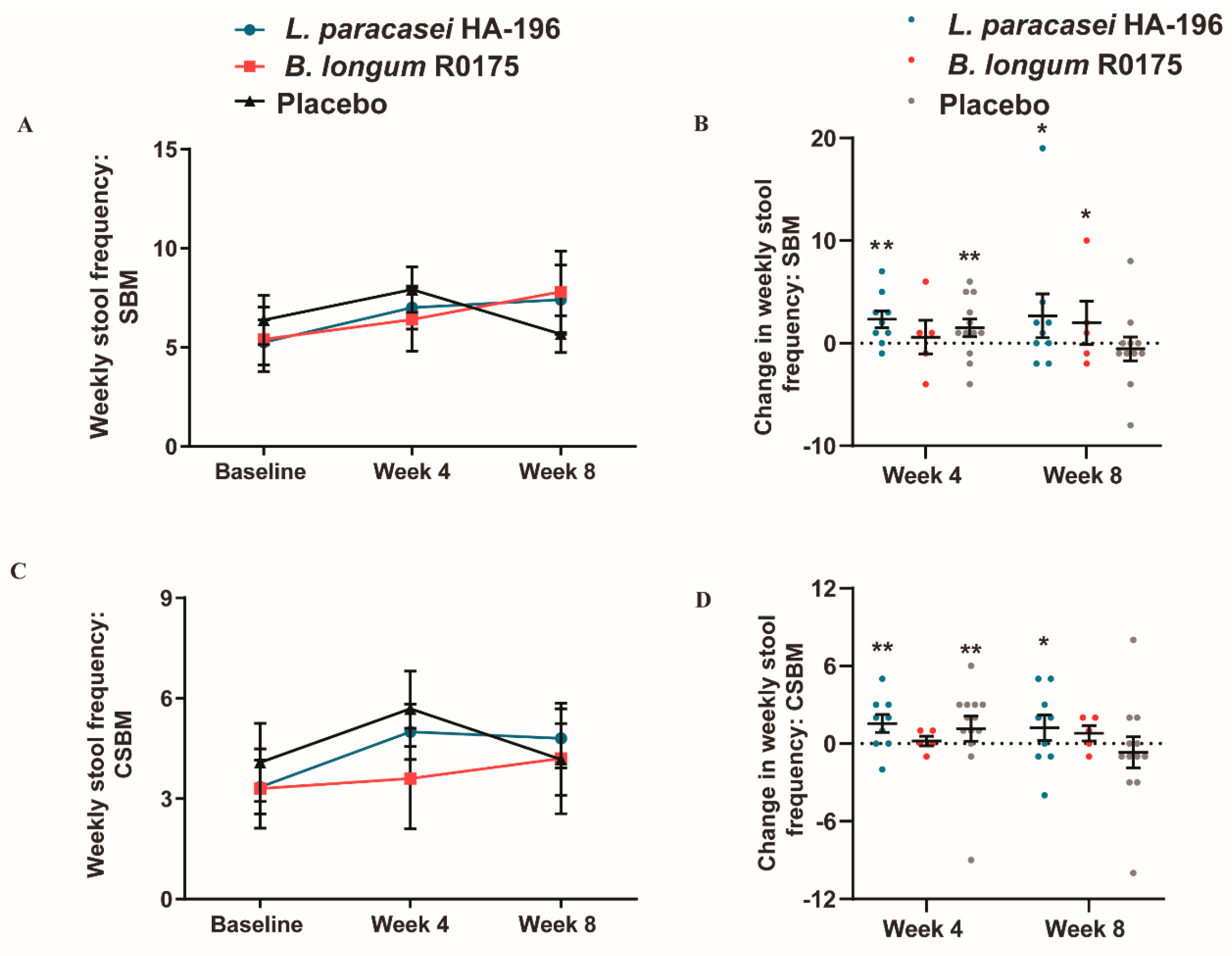
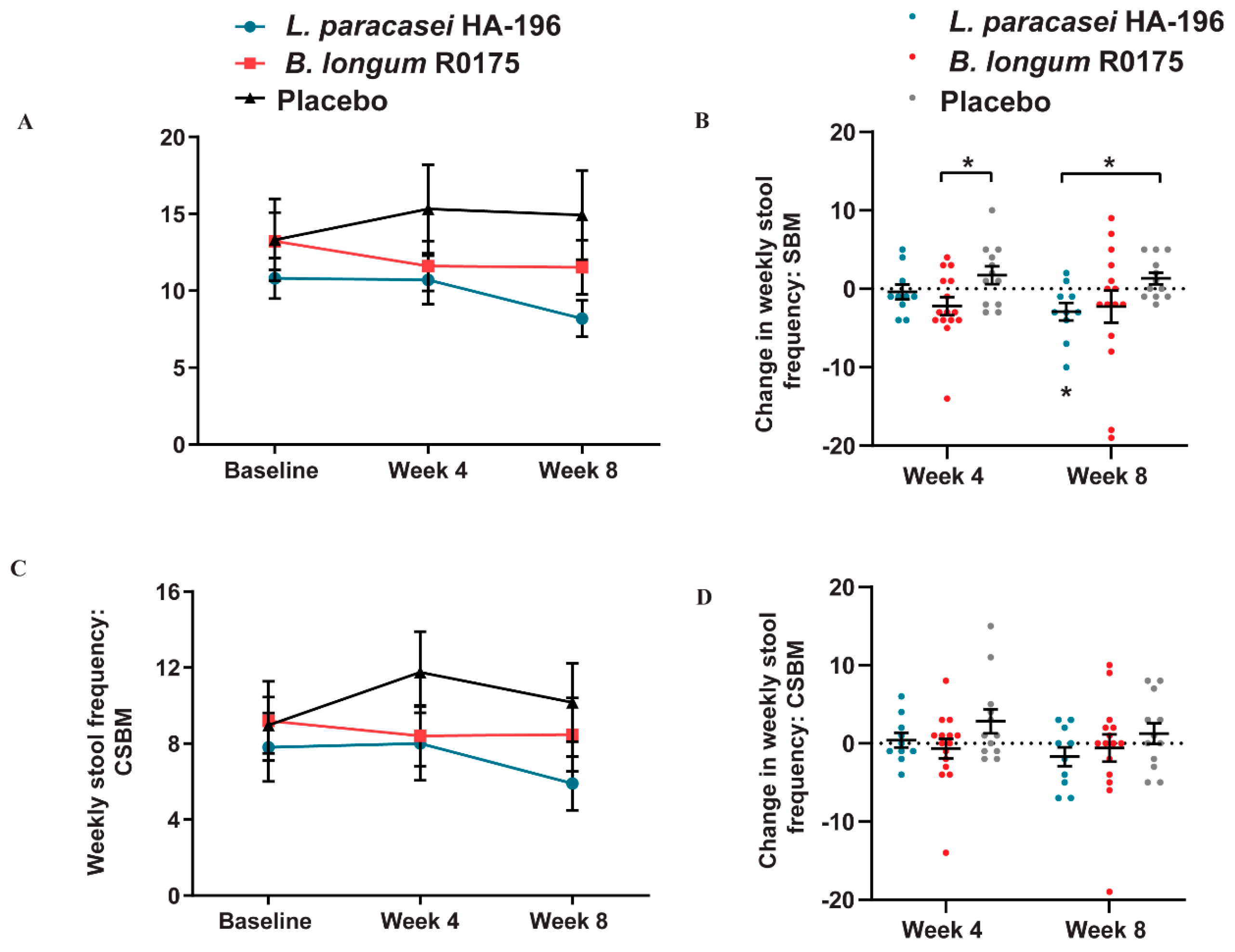
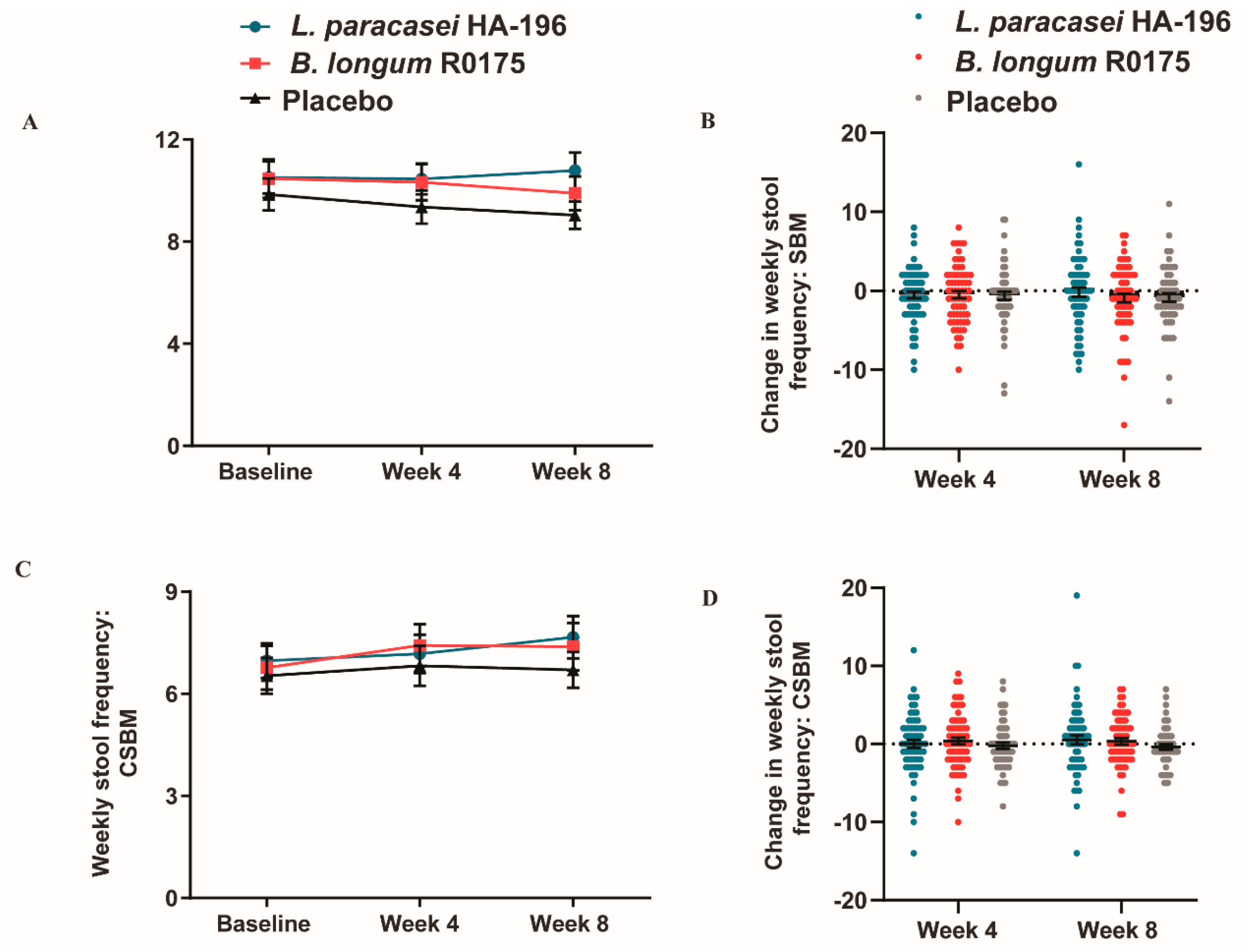
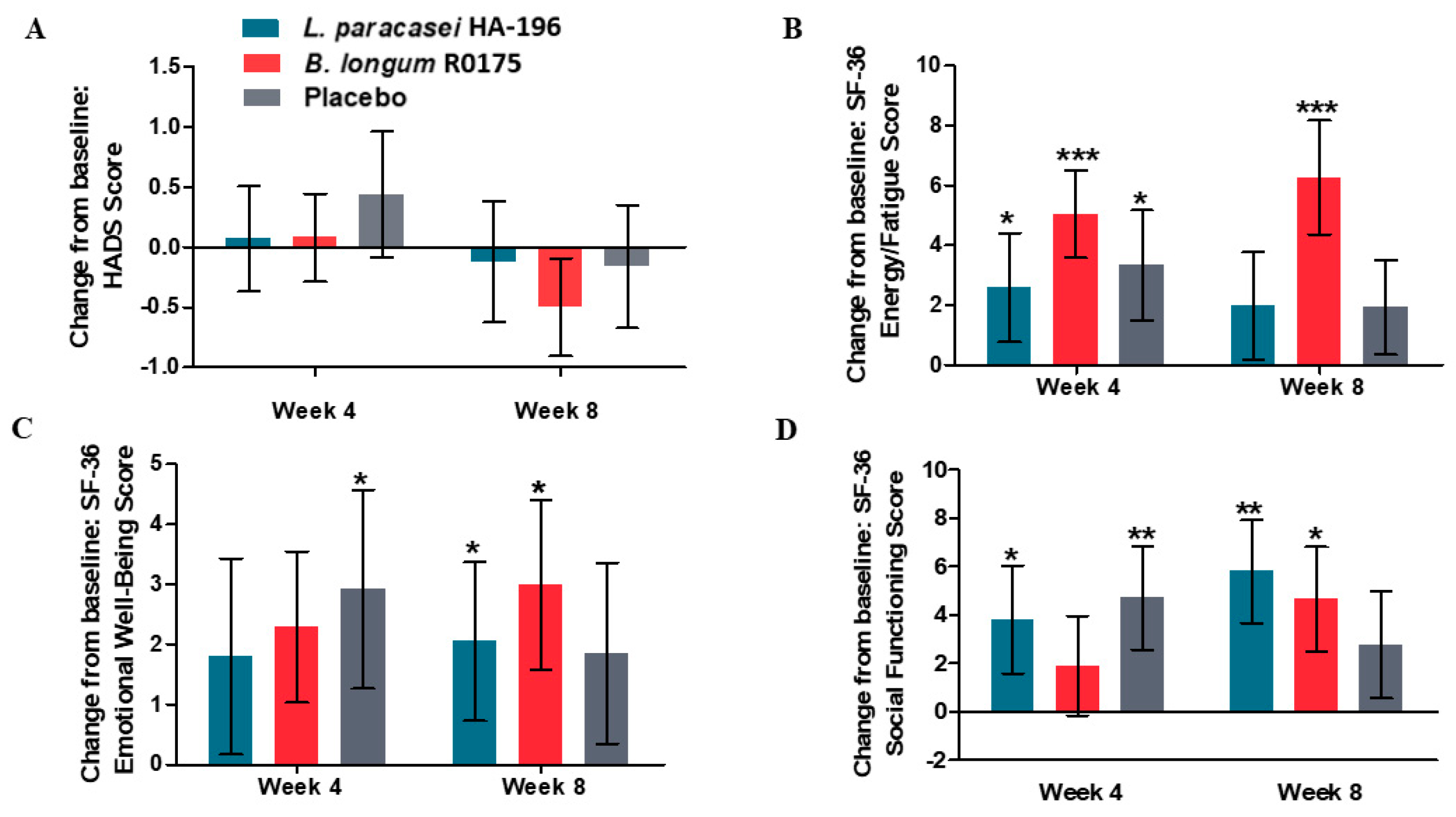
| Parameter | L. paracasei HA-196 (n) | B. longum R0175 (n) | Placebo (n) |
|---|---|---|---|
| Age (years) | |||
| Mean+/−SD | 42.42 ± 12.30 (84) | 42.31 ± 16.88 (86) | 41.84 ± 16.14 (81) |
| Median (Min–Max) | 43.00 (21.00–72.00) | 41.00 (19.00–87.00) | 40.00 (18.00–75.00) |
| p-value * | 0.9573 | 0.9706 | |
| Gender [n (%)] | |||
| Female | 67 (79.8%) | 64 (74.4%) | 64 (79.0%) |
| Male | 17 (20.2%) | 22 (25.6%) | 17 (21.0%) |
| p-value * | 1.0000 | 0.5836 | |
| Ethnicity [n (%)] | |||
| Hispanic or Latino | 7 (8.3%) | 5 (5.8%) | 1 (1.2%) |
| Not Hispanic or Latino | 77 (91.7%) | 81 (94.2%) | 80 (98.8%) |
| p-value * | 0.0641 | 0.2115 | |
| Race [n (%)] | |||
| Black or African American | 2 (2.3%) | 2 (2.5%) | |
| Central American | 1 (1.2%) | 1 (1.2%) | |
| East Asian | 2 (2.5%) | ||
| Eastern European White | 10 (11.9%) | 9 (10.5%) | 8 (9.9%) |
| Middle Eastern | 8 (9.5%) | 2 (2.3%) | 3 (3.7%) |
| North American Indian | 1 (1.2%) | 1 (1.2%) | |
| South American | 5 (6.0%) | 3 (3.5%) | 1 (1.2%) |
| South Asian | 2 (2.3%) | 1 (1.2%) | |
| South East Asian | 1 (1.2%) | 1 (1.2%) | |
| Western European White | 59 (70.2%) | 66 (76.7%) | 62 (76.5%) |
| p-value * | 0.1463 | 0.8926 | |
| Type of IBS: ROME III [n (%)] | |||
| IBS-C | 10 (11.9%) | 5 (5.8%) | 13 (16.0%) |
| IBS-D | 10 (11.9%) | 15 (17.4%) | 13 (16.0%) |
| IBS-M | 64 (76.2%) | 66 (76.7%) | 55 (67.9%) |
| p-value * | 0.5434 | 0.1041 |
| IBS-SSS | ||||
|---|---|---|---|---|
| Study Day | Statistic | L. paracasei HA-196 | B. longum R0175 | Placebo |
| Baseline (Day 0) | Mean ± SD (n) | 246.94 ± 75.53 (84) | 238.13 ± 76.59 (83) | 238.96 ± 64.55 (81) |
| p-value * | 0.7011 | 0.9960 | ||
| Week 4 | Mean ± SD (n) | 198.22 ± 69.07 (82) | 189.57 ± 82.55 (83) | 199.39 ± 72.13 (77) |
| Week 8 | Mean ± SD (n) | 172.23 ± 80.90 (84) | 185.71 ± 77.74 (84) | 164.56 ± 73.25 (81) |
| Change from Baseline to Week 4 | Mean ± SD (n) | −50.20 ± 64.42 (82) | −50.78 ± 72.12 (82) | −42.32 ± 66.93 (77) |
| Within Group p-Value + | p < 0.001 (r) | p < 0.001 (r) | p < 0.001 (r) | |
| Between Group p-value ** | 0.4246 (r) | 0.1390 (r) | ||
| Change from Baseline to Week 8 | Mean ± SD (n) | −74.71 ± 71.52 (84) | −53.25 ± 65.65 (83) | −74.41 ± 62.83 (81) |
| Within Group p-Value + | p < 0.001 (r) | p < 0.001 (r) | p < 0.001 (r) | |
| Between Group p-value ** | 0.9632 (r) | 0.0763 (r) | ||
| Study Day | Statistic | L. paracasei HA-196 | B. longum R0175 | Placebo |
|---|---|---|---|---|
| Baseline (average of Weeks −2 and −1) | Mean ± SD (n) | 2.84 ± 1.43 (10) | 2.55 ± 1.57 (5) | 2.69 ± 0.78 (12) |
| p-value * | 0.9467 | 0.9656 | ||
| Week 4 | Mean ± SD (n) | 3.14 ± 1.05 (10) | 3.21 ± 1.15 (5) | 2.91 ± 0.82 (13) |
| Week 8 | Mean ± SD (n) | 3.21 ± 1.32 (10) | 2.44 ± 0.74 (5) | 2.49 ± 0.95 (12) |
| Change from Baseline to Week 4 | Mean ± SD (n) | 0.44 ± 1.09 (9) | 0.49 ± 1.74 (5) | −0.05 ± 0.80 (12) |
| Within Group p-Value + | p = 0.221 (r) | p = 0.210 (r) | p = 0.295 (r) | |
| Between Group p-value ** | 0.2585 (r) | 0.9940 (r) | ||
| Change from Baseline to Week 8 | Mean ± SD (n) | 0.62 ± 1.12 (9) | −0.28 ± 1.54 (5) | −0.41 ± 1.00 (11) |
| Within Group p-Value + | p = 0.186 (r) | p = 0.789 (r) | p = 0.858 (r) | |
| Between Group p-value ** | 0.0721 (r) | 0.9805 (r) |
| Study Day | Statistic | L. paracasei HA-196 | B. longum R0175 | Placebo |
|---|---|---|---|---|
| Baseline (average of Weeks −2 and −1) | Mean ± SD (n) | 4.34 ± 0.65 (10) | 4.46 ± 0.89 (15) | 4.79 ± 0.77 (13) |
| p-value * | 0.3106 | 0.4416 | ||
| Week 4 | Mean ± SD (n) | 3.73 ± 0.75 (10) | 4.13 ± 0.64 (15) | 4.26 ± 0.57 (12) |
| Week 8 | Mean ± SD (n) | 3.68 ± 0.95 (10) | 4.22 ± 0.97 (15) | 4.66 ± 0.76 (12) |
| Change from Baseline to Week 4 | Mean ± SD (n) | −0.39 ± 0.80 (10) | −0.36 ± 1.01 (15) | −0.60 ± 0.74 (12) |
| Within Group p-Value + | p = 0.023 | p = 0.132 | p = 0.016 | |
| Between Group p-value ** | 0.8523 | 0.9868 | ||
| Change from Baseline to Week 8 | Mean ± SD (n) | −0.45 ± 1.14 (10) | −0.26 ± 1.12 (15) | −0.20 ± 0.78 (12) |
| Within Group p-Value + | p = 0.013 | p = 0.281 | p = 0.450 | |
| Between Group p-value ** | 0.2896 | 0.5955 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewis, E.D.; Antony, J.M.; Crowley, D.C.; Piano, A.; Bhardwaj, R.; Tompkins, T.A.; Evans, M. Efficacy of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study. Nutrients 2020, 12, 1159. https://doi.org/10.3390/nu12041159
Lewis ED, Antony JM, Crowley DC, Piano A, Bhardwaj R, Tompkins TA, Evans M. Efficacy of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study. Nutrients. 2020; 12(4):1159. https://doi.org/10.3390/nu12041159
Chicago/Turabian StyleLewis, Erin D., Joseph M. Antony, David C. Crowley, Amanda Piano, Renu Bhardwaj, Thomas A. Tompkins, and Malkanthi Evans. 2020. "Efficacy of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study" Nutrients 12, no. 4: 1159. https://doi.org/10.3390/nu12041159
APA StyleLewis, E. D., Antony, J. M., Crowley, D. C., Piano, A., Bhardwaj, R., Tompkins, T. A., & Evans, M. (2020). Efficacy of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study. Nutrients, 12(4), 1159. https://doi.org/10.3390/nu12041159






