Modifications to Infant Formula Instructions Improve the Accuracy of Formula Dispensing
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Methods
2.2. First Phase—Participatory Research
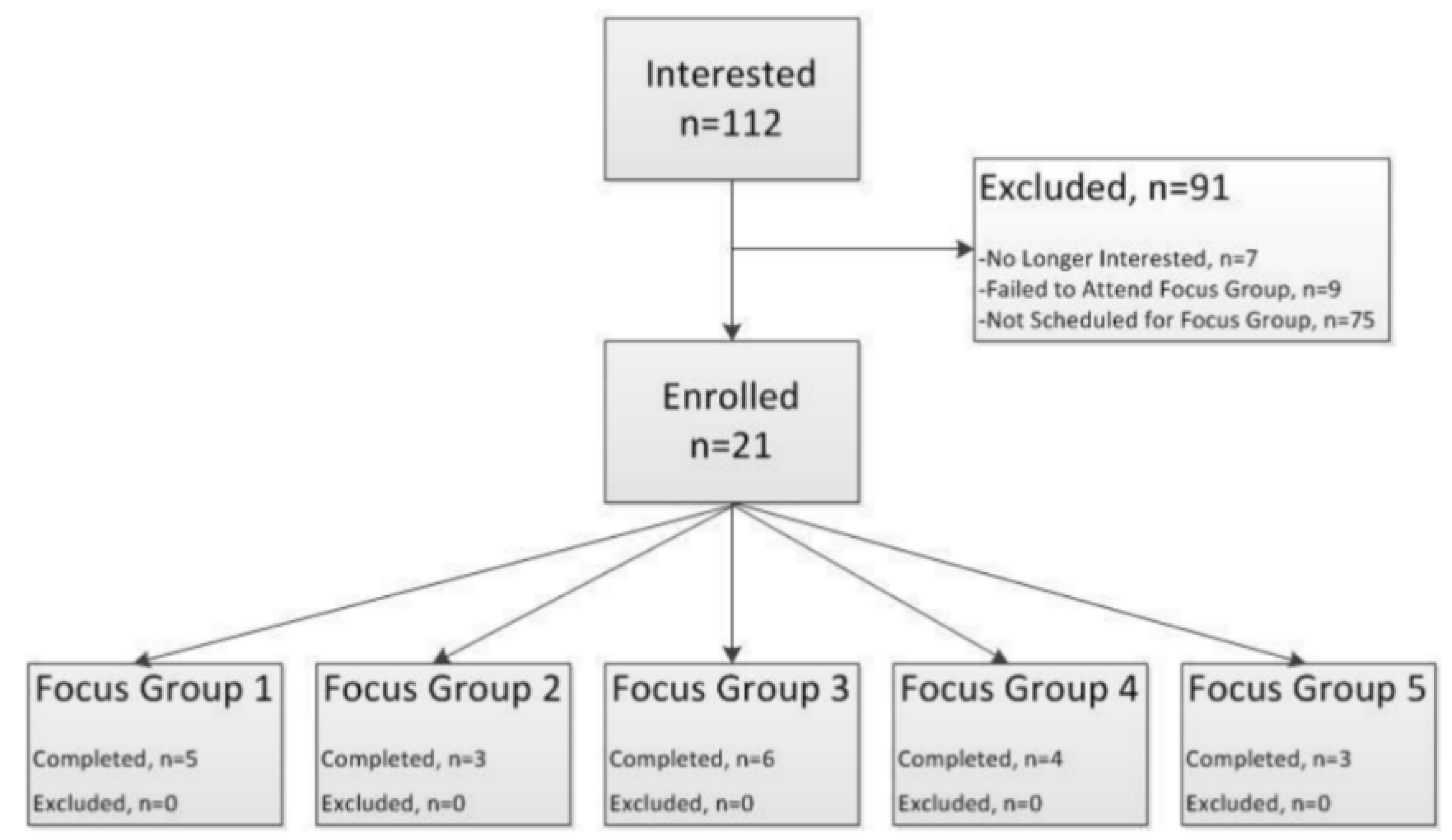
2.2.1. Focus Group Participants
2.2.2. Design
2.2.3. Measures
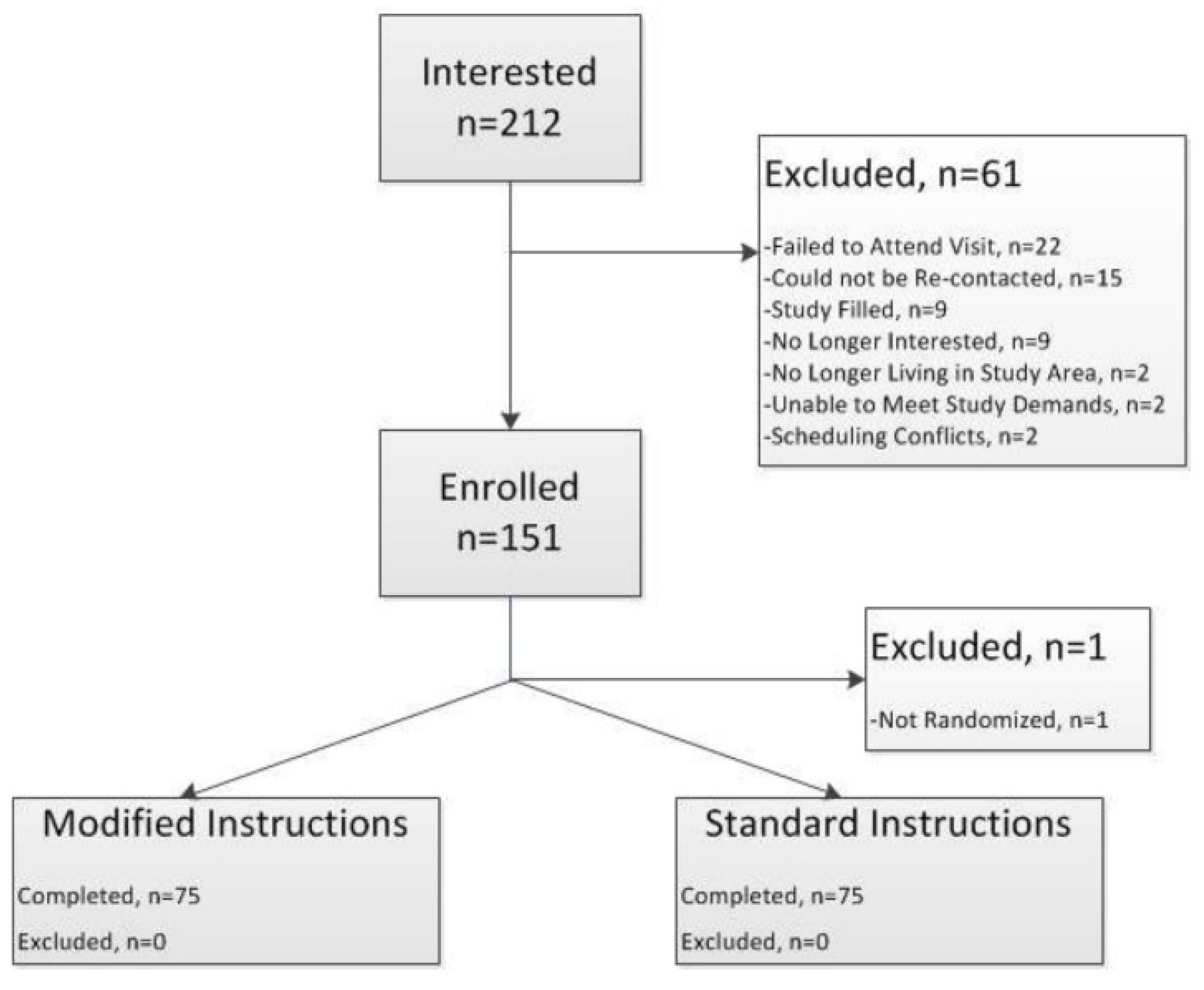
2.3. Second Phase—Double-Blind Randomized, Controlled Trial
2.3.1. Design
2.3.2. Measures
2.3.3. Statistical Analysis
3. Results
3.1. First Phase—Participatory Research
3.2. Second Phase—Randomized Controlled Trial
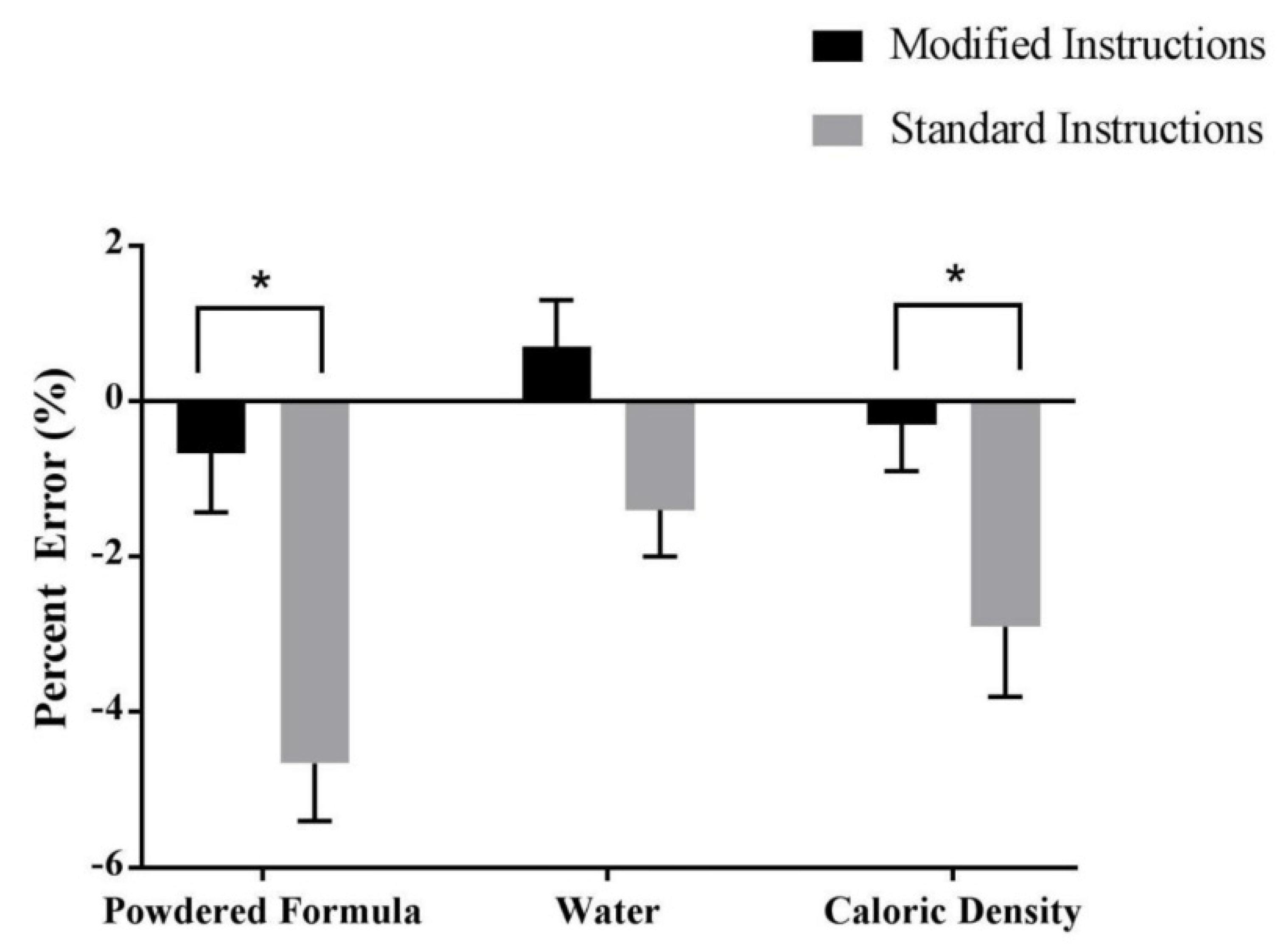
3.2.1. Powered Formula Dispensing
3.2.2. Water Dispensing
3.2.3. Caloric Density
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Rossen, L.M.; Simon, A.E.; Herrick, K.A. Types of Infant Formulas Consumed in the United States. Clin. Pediatr. 2016, 55, 278–285. [Google Scholar] [CrossRef] [PubMed]
- CDC. National Immunization Surveys 2012 and 2013; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2014.
- Wallace, L.S.; Rosenstein, P.F.; Gal, N. Readability and Content Characteristics of Powdered Infant Formula Instructions in the United States. Matern. Child Health J. 2016, 20, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Labiner-Wolfe, J.; Fein, S.B.; Shealy, K.R. Infant formula-handling education and safety. Pediatrics 2008, 122 (Suppl. 2), S85–S90. [Google Scholar] [CrossRef] [PubMed]
- Renfrew, M.J.; Ansell, P.; Macleod, K.L. Formula feed preparation: Helping reduce the risks; a systematic review. Arch. Dis. Child. 2003, 88, 855–858. [Google Scholar] [CrossRef] [PubMed]
- Jeffs, S.G. Hazards of scoop measurements in infant feeding. J. R. Coll. Gen. Pract. 1989, 39, 113. [Google Scholar] [PubMed]
- Lucas, A.; Lockton, S.; Davies, P.S. Randomised trial of a ready-to-feed compared with powdered formula. Arch. Dis. Child. 1992, 67, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Altazan, A.D.; Gilmore, L.A.; Guo, J.; Rosenberg, D.M.; Toupo, D.; Gowins, A.; Burton, J.H.; Beyl, R.A.; Chow, C.C.; Hall, K.D.; et al. Unintentional error in formula preparation and its simulated impact on infant weight and adiposity. Pediatr. Obes. 2019, e12564. [Google Scholar] [CrossRef] [PubMed]
- Butte, N.F.; Wong, W.W.; Hopkinson, J.M.; Smith, E.O.; Ellis, K.J. Infant feeding mode affects early growth and body composition. Pediatrics 2000, 106, 1355–1366. [Google Scholar] [CrossRef] [PubMed]
- Baird, J.; Poole, J.; Robinson, S.; Marriott, L.; Godfrey, K.; Cooper, C.; Inskip, H.; Law, C. Milk feeding and dietary patterns predict weight and fat gains in infancy. Paediatr. Perinat. Epidemiol. 2008, 22, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Sauder, K.A.; Kaar, J.L.; Starling, A.P.; Ringham, B.M.; Glueck, D.H.; Dabelea, D. Predictors of Infant Body Composition at 5 Months of Age: The Healthy Start Study. J. Pediatr. 2017, 183, 94–99.e1. [Google Scholar] [CrossRef] [PubMed]
- Ventura, A.K. Developmental Trajectories of Bottle-Feeding During Infancy and Their Association with Weight Gain. J. Dev. Behav. Pediatr. 2017, 38, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.A.; Wagner, C.L.; Feldman, H.A.; Shypailo, R.J.; Belfort, M.B. Associations of infant feeding with trajectories of body composition and growth. Am. J. Clin. Nutr. 2017, 106, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.D.; Sharp, S. Health literacy and prescribing: It is time to change old habits. Am. Fam. Physician 2013, 87, 755–756. [Google Scholar] [PubMed]
- Yin, H.S.; Dreyer, B.P.; Moreira, H.A.; van Schaick, L.; Rodriguez, L.; Boettger, S.; Mendelsohn, A.L. Liquid medication dosing errors in children: Role of provider counseling strategies. Acad. Pediatr. 2014, 14, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.S.; Parker, R.M.; Sanders, L.M.; Dreyer, B.P.; Mendelsohn, A.L.; Bailey, S.; Patel, D.A.; Jimenez, J.J.; Kim, K.A.; Jacobson, K.; et al. Liquid Medication Errors and Dosing Tools: A Randomized Controlled Experiment. Pediatrics 2016, 138, e20160357. [Google Scholar] [CrossRef] [PubMed]
- Altazan, A.D.; Gilmore, L.A.; Burton, J.H.; Ragusa, S.A.; Apolzan, J.W.; Martin, C.K.; Redman, L.M. Development and Application of the Remote Food Photography Method to Measure Food Intake in Exclusively Milk Fed Infants: A Laboratory-Based Study. PLoS ONE 2016, 11, e0163833. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.S.; Parker, R.M.; Wolf, M.S.; Mendelsohn, A.L.; Sanders, L.M.; Vivar, K.L.; Carney, K.; Cerra, M.E.; Dreyer, B.P. Health literacy assessment of labeling of pediatric nonprescription medications: Examination of characteristics that may impair parent understanding. Acad. Pediatr. 2012, 12, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Code of Federal Regulations. Available online: https://www.ecfr.gov/cgi-bin/text-idx?SID=660302655edbcdcb7b37ba2c9b9bd852&mc=true&node=pt21.2.107&rgn=div5#se21.2.107_120 (accessed on 15 January 2019).
- Lanigan, J.A.; Wells, J.C.; Lawson, M.S.; Lucas, A. Validation of food diary method for assessment of dietary energy and macronutrient intake in infants and children aged 6-24 months. Eur. J. Clin. Nutr. 2001, 55, 124–129. [Google Scholar] [CrossRef] [PubMed]


| All Phase 1 (n = 21) | Phase 2 Modified Instructions (n = 75) | Phase 2 Standard Instructions (n = 75) | |
|---|---|---|---|
| Age, year | 32.5 ± 11.7 | 40.6 ± 17.6 | 36.0 ± 15.8 |
| BMI, kg/m2 | 26.8 ± 8.1 | 29.5 ± 7.9 | 29.6 ± 6.6 |
| BMI Classification, n (%) | |||
| Normal or Under Weight | 14 (66.7) | 24 (32.0) | 18 (24.0) |
| Overweight | 2 (9.5) | 20 (26.7) | 23 (30.7) |
| Obese | 5 (23.8) | 31 (41.3) | 34 (45.3) |
| Race, n (%) | |||
| Caucasian | 15 (71.4) | 54 (72.0) | 46 (61.3) |
| African American | 6 (28.6) | 16 (21.3) | 23 (30.7) |
| Other | 0 (0.0) | 5 (6.7) | 6 (8.0) |
| Gender, n (%) | |||
| Male | 4 (19.0) | 16 (21.3) | 10 (13.3) |
| Female | 17 (81.0) | 59 (78.7) | 65 (86.7) |
| Caregiver of Infants Status, n (%) | |||
| Primary Caregiver | 6 (28.6) | 10 (13.3) | 15 (20.0) |
| Secondary Caregiver | 5 (23.8) | 11 (14.7) | 10 (13.3) |
| Non-Caregiver | 10 (47.6) | 54 (72.0) | 49 (65.4) |
| No Answer | 0 (0.0) | 0 (0.0) | 1 (1.3) |
| Education, n (%) | |||
| High School Diploma/GED or Less | 1 (4.8) | 5 (6.7) | 8 (10.7) |
| 1–3 Years College | 5 (23.8) | 37 (49.3) | 26 (34.7) |
| College Degree | 9 (42.8) | 17 (22.7) | 25 (33.3) |
| Post-Graduate Degree | 6 (28.6) | 16 (21.3) | 16 (21.3) |
| Income, n (%) | |||
| <$30,000/year | 5 (23.8) | 21 (28.0) | 21 (28.0) |
| $30,000–$99,999/year | 9 (42.9) | 28 (37.3) | 32 (42.7) |
| >$80,000/year | 7 (33.3) | 25 (33.3) | 21 (28.0) |
| No Answer | 0 (0.0) | 1 (1.3) | 1 (1.3) |
| Employment, n (%) | |||
| Full Time | 11 (52.4) | 22 (29.3) | 33 (44.0) |
| Not Full Time | 10 (47.6) | 53 (70.7) | 42 (56.0) |
| Parent/Guardian, n (%) | |||
| Yes | 11 (52.4) | 38 (50.7) | 43 (57.3) |
| No | 10 (47.6) | 37 (49.3) | 32 (42.7) |
| Prepared Formula in Past Year, n (%) | |||
| Yes | 8 (38.1) | 21 (28.0) | 26 (34.7) |
| No | 13 (61.9) | 54 (72.0) | 49 (65.3) |
| Focus Group Order | Participant Characteristics | Primary Recommendations * |
|---|---|---|
| First |
|
|
| Second |
|
|
| Third |
|
|
| Fourth |
|
|
| Fifth |
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gilmore, L.A.; Altazan, A.D.; Flanagan, E.W.; Beyer, A.G.; Olson, K.N.; O’Connell, A.A.; Nguyen, T.H.; Beyl, R.A.; Redman, L.M. Modifications to Infant Formula Instructions Improve the Accuracy of Formula Dispensing. Nutrients 2020, 12, 1150. https://doi.org/10.3390/nu12041150
Gilmore LA, Altazan AD, Flanagan EW, Beyer AG, Olson KN, O’Connell AA, Nguyen TH, Beyl RA, Redman LM. Modifications to Infant Formula Instructions Improve the Accuracy of Formula Dispensing. Nutrients. 2020; 12(4):1150. https://doi.org/10.3390/nu12041150
Chicago/Turabian StyleGilmore, Linda A., Abby D. Altazan, Emily W. Flanagan, Alexandra G. Beyer, Kelsey N. Olson, Alexis A. O’Connell, Timothy H. Nguyen, Robbie A. Beyl, and Leanne M. Redman. 2020. "Modifications to Infant Formula Instructions Improve the Accuracy of Formula Dispensing" Nutrients 12, no. 4: 1150. https://doi.org/10.3390/nu12041150
APA StyleGilmore, L. A., Altazan, A. D., Flanagan, E. W., Beyer, A. G., Olson, K. N., O’Connell, A. A., Nguyen, T. H., Beyl, R. A., & Redman, L. M. (2020). Modifications to Infant Formula Instructions Improve the Accuracy of Formula Dispensing. Nutrients, 12(4), 1150. https://doi.org/10.3390/nu12041150






