Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial
Abstract
1. Introduction
2. Methods
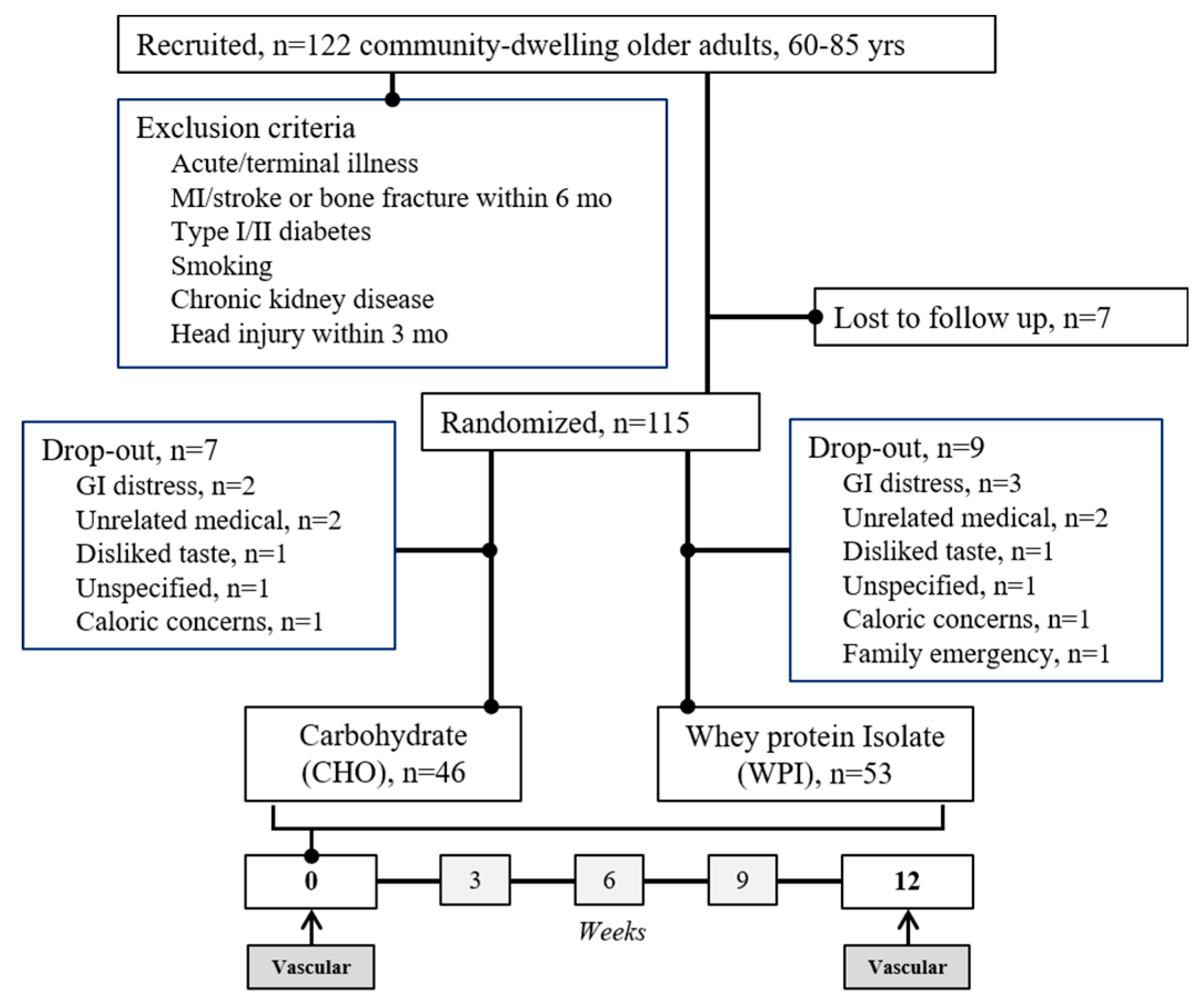
2.1. Study Design
2.2. Brachial Blood Pressure
2.3. Aortic Stiffness and Blood Pressure
2.4. Carotid Blood Flow and Stiffness
2.5. Cerebral Blood Flow Velocity
2.6. Cerebrovascular Response to Cognitive Activity
2.7. Computerized Cognitive Function Battery
2.8. Physical Activity
2.9. Statistical Approach
2.9.1. Sample Size Estimation
2.9.2. Statistical Analyses
3. Results
3.1. Intervention Effect on Anthropometrics and Secondary Outcomes
3.2. Intervention Effect on Blood Pressure and Central Hemodynamics
3.3. Intervention Effect on Cerebrovascular Hemodynamics
3.4. Intervention Effect on Cerebrovascular Response to Cognitive Activity
3.5. Intervention Effect on Cognitive Function
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Najjar, S.S.; Scuteri, A.; Lakatta, E.G. Arterial aging: Is it an immutable cardiovascular risk factor? Hypertension 2005, 46, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Townsend, R.R.; Wilkinson, I.B.; Schiffrin, E.L.; Avolio, A.P.; Chirinos, J.A.; Cockcroft, J.R.; Heffernan, K.S.; Lakatta, E.G.; McEniery, C.M.; Mitchell, G.F.; et al. Recommendations for Improving and Standardizing Vascular Research on Arterial Stiffness: A Scientific Statement from the American Heart Association. Hypertension 2015, 66, 698–722. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Aznaouridis, K.; Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2010, 55, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Aznaouridis, K.; O’Rourke, M.F.; Safar, M.E.; Baou, K.; Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: A systematic review and meta-analysis. Eur. Heart J. 2010, 31, 1865–1871. [Google Scholar] [CrossRef]
- Katz, S.; Branch, L.G.; Branson, M.H.; Papsidero, J.A.; Beck, J.C.; Greer, D.S. Active life expectancy. N. Engl. J. Med. 1983, 309, 1218–1224. [Google Scholar] [CrossRef]
- Castro-Lionard, K.; Thomas-Anterion, C.; Crawford-Achour, E.; Rouch, I.; Trombert-Paviot, B.; Barthelemy, J.C.; Laurent, B.; Roche, F.; Gonthier, R. Can maintaining cognitive function at 65 years old predict successful ageing 6 years later? The PROOF study. Age Ageing 2011, 40, 259–265. [Google Scholar] [CrossRef]
- Gaugler, J.E.; Yu, F.; Krichbaum, K.; Wyman, J.F. Predictors of nursing home admission for persons with dementia. Med. Care 2009, 47, 191–198. [Google Scholar] [CrossRef]
- Pase, M.P.; Herbert, A.; Grima, N.A.; Pipingas, A.; O’Rourke, M.F. Arterial stiffness as a cause of cognitive decline and dementia: A systematic review and meta-analysis. Intern. Med. J. 2011. [Google Scholar] [CrossRef]
- Van Sloten, T.T.; Protogerou, A.D.; Henry, R.M.; Schram, M.T.; Launer, L.J.; Stehouwer, C.D. Association between arterial stiffness, cerebral small vessel disease and cognitive impairment: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2015, 53, 121–130. [Google Scholar] [CrossRef]
- Alvarez-Bueno, C.; Cunha, P.G.; Martinez-Vizcaino, V.; Pozuelo-Carrascosa, D.P.; Visier-Alfonso, M.E.; Jimenez-Lopez, E.; Cavero-Redondo, I. Arterial Stiffness and Cognition Among Adults: A Systematic Review and Meta-Analysis of Observational and Longitudinal Studies. J. Am. Heart Assoc. 2020, 9, e014621. [Google Scholar] [CrossRef]
- Scuteri, A.; Tesauro, M.; Appolloni, S.; Preziosi, F.; Brancati, A.M.; Volpe, M. Arterial stiffness as an independent predictor of longitudinal changes in cognitive function in the older individual. J. Hypertens 2007, 25, 1035–1040. [Google Scholar] [CrossRef]
- Chung, C.P.; Lee, H.Y.; Lin, P.C.; Wang, P.N. Cerebral Artery Pulsatility is Associated with Cognitive Impairment and Predicts Dementia in Individuals with Subjective Memory Decline or Mild Cognitive Impairment. J. Alzheimer’s Dis. JAD 2017, 60, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, P.B.; Scuteri, A.; Black, S.E.; Decarli, C.; Greenberg, S.M.; Iadecola, C.; Launer, L.J.; Laurent, S.; Lopez, O.L.; Nyenhuis, D.; et al. Vascular contributions to cognitive impairment and dementia: A statement for healthcare professionals from the american heart association/american stroke association. Stroke 2011, 42, 2672–2713. [Google Scholar] [CrossRef] [PubMed]
- Camfield, D.A.; Owen, L.; Scholey, A.B.; Pipingas, A.; Stough, C. Dairy constituents and neurocognitive health in ageing. Br. J. Nutr. 2011, 106, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Patel, S. Emerging trends in nutraceutical applications of whey protein and its derivatives. J. Food Sci. Technol. 2015, 52, 6847–6858. [Google Scholar] [CrossRef] [PubMed]
- Soedamah-Muthu, S.S.; Verberne, L.D.; Ding, E.L.; Engberink, M.F.; Geleijnse, J.M. Dairy consumption and incidence of hypertension: A dose-response meta-analysis of prospective cohort studies. Hypertension 2012, 60, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Crichton, G.E.; Elias, M.F.; Dore, G.A.; Abhayaratna, W.P.; Robbins, M.A. Relations between dairy food intake and arterial stiffness: Pulse wave velocity and pulse pressure. Hypertension 2012, 59, 1044–1051. [Google Scholar] [CrossRef]
- Crichton, G.E.; Elias, M.F.; Dore, G.A.; Robbins, M.A. Relation between dairy food intake and cognitive function: The Maine-Syracuse Longitudinal Study. Int. Dairy J. 2012, 22, 15–23. [Google Scholar] [CrossRef]
- FitzGerald, R.J.; Murray, B.A.; Walsh, D.J. Hypotensive peptides from milk proteins. J. Nutr. 2004, 134, 980S–988S. [Google Scholar] [CrossRef]
- Ballard, K.D.; Kupchak, B.R.; Volk, B.M.; Mah, E.; Shkreta, A.; Liptak, C.; Ptolemy, A.S.; Kellogg, M.S.; Bruno, R.S.; Seip, R.L.; et al. Acute effects of ingestion of a novel whey-derived extract on vascular endothelial function in overweight, middle-aged men and women. Br. J. Nutr. 2012, 1–12. [Google Scholar] [CrossRef]
- Markus, C.R.; Olivier, B.; de Haan, E.H. Whey protein rich in alpha-lactalbumin increases the ratio of plasma tryptophan to the sum of the other large neutral amino acids and improves cognitive performance in stress-vulnerable subjects. Am. J. Clin. Nutr. 2002, 75, 1051–1056. [Google Scholar] [CrossRef] [PubMed]
- Fekete, A.A.; Giromini, C.; Chatzidiakou, Y.; Givens, D.I.; Lovegrove, J.A. Whey protein lowers blood pressure and improves endothelial function and lipid biomarkers in adults with prehypertension and mild hypertension: Results from the chronic Whey2Go randomized controlled trial. Am. J. Clin. Nutr. 2016, 104, 1534–1544. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Ellis, V. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity 2010, 18, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Bleasdale, R.A.; Mumford, C.E.; Campbell, R.I.; Fraser, A.G.; Jones, C.J.; Frenneaux, M.P. Wave intensity analysis from the common carotid artery: A new noninvasive index of cerebral vasomotor tone. Heart Vessel. 2003, 18, 202–206. [Google Scholar] [CrossRef]
- Chiesa, S.T.; Masi, S.; Shipley, M.J.; Ellins, E.A.; Fraser, A.G.; Hughes, A.D.; Patel, R.S.; Khir, A.W.; Halcox, J.P.; Singh-Manoux, A.; et al. Carotid artery wave intensity in mid- to late-life predicts cognitive decline: The Whitehall II study. Eur. Heart J. 2019, 40, 2300–2309. [Google Scholar] [CrossRef]
- Heffernan, K.S.; Spartano, N.L.; Augustine, J.A.; Lefferts, W.K.; Hughes, W.E.; Mitchell, G.F.; Jorgensen, R.S.; Gump, B.B. Carotid artery stiffness and hemodynamic pulsatility during cognitive engagement in healthy adults: A pilot investigation. Am. J. Hypertens. 2015, 28, 615–622. [Google Scholar] [CrossRef]
- Lefferts, W.K.; DeBlois, J.P.; Barreira, T.V.; Heffernan, K.S. Neurovascular Coupling during Cognitive Activity in Adults with Controlled Hypertension. J. Appl. Physiol. 2018. [Google Scholar] [CrossRef]
- Sheu, L.K.; Jennings, J.R.; Gianaros, P.J. Test-retest reliability of an fMRI paradigm for studies of cardiovascular reactivity. Psychophysiology 2012, 49, 873–884. [Google Scholar] [CrossRef]
- Naqvi, T.Z.; Hyuhn, H.K. Cerebrovascular mental stress reactivity is impaired in hypertension. Cardiovasc. Ultrasound 2009, 7, 32. [Google Scholar] [CrossRef]
- Heffernan, K.S.; Augustine, J.A.; Lefferts, W.K.; Spartano, N.L.; Hughes, W.E.; Jorgensen, R.S.; Gump, B.B. Arterial stiffness and cerebral hemodynamic pulsatility during cognitive engagement in younger and older adults. Exp. Gerontol. 2018, 101, 54–62. [Google Scholar] [CrossRef]
- Lefferts, W.K.; Hughes, W.E.; White, C.N.; Brutsaert, T.D.; Heffernan, K.S. Effect of acute nitrate supplementation on neurovascular coupling and cognitive performance in hypoxia. Appl. Physiol. Nutr. Metab 2016, 41, 133–141. [Google Scholar] [CrossRef]
- Troiano, R.P.; Berrigan, D.; Dodd, K.W.; Masse, L.C.; Tilert, T.; McDowell, M. Physical activity in the United States measured by accelerometer. Med. Sci. Sports Exerc. 2008, 40, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Ratchford, E.V.; Gutierrez, J.; Lorenzo, D.; McClendon, M.S.; Della-Morte, D.; DeRosa, J.T.; Elkind, M.S.; Sacco, R.L.; Rundek, T. Short-term effect of atorvastatin on carotid artery elasticity: A pilot study. Stroke 2011, 42, 3460–3464. [Google Scholar] [CrossRef] [PubMed]
- Lunder, M.; Janic, M.; Habjan, S.; Sabovic, M. Subtherapeutic, low-dose fluvastatin improves functional and morphological arterial wall properties in apparently healthy, middle-aged males—A pilot study. Atherosclerosis 2011, 215, 446–451. [Google Scholar] [CrossRef]
- Gates, P.E.; Tanaka, H.; Hiatt, W.R.; Seals, D.R. Dietary sodium restriction rapidly improves large elastic artery compliance in older adults with systolic hypertension. Hypertension 2004, 44, 35–41. [Google Scholar] [CrossRef]
- Moreau, K.L.; Donato, A.J.; Seals, D.R.; DeSouza, C.A.; Tanaka, H. Regular exercise, hormone replacement therapy and the age-related decline in carotid arterial compliance in healthy women. Cardiovasc. Res. 2003, 57, 861–868. [Google Scholar] [CrossRef]
- Balkestein, E.J.; van Aggel-Leijssen, D.P.; van Baak, M.A.; Struijker-Boudier, H.A.; Van Bortel, L.M. The effect of weight loss with or without exercise training on large artery compliance in healthy obese men. J. Hypertens. 1999, 17, 1831–1835. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.P.; Tong, X.; Li, Z.N.; Xu, J.Y.; Zhou, L.; Zhou, B.Y.; Qin, L.Q. Effect of whey protein on blood pressure in pre- and mildly hypertensive adults: A randomized controlled study. Food Sci. Nutr. 2019, 7, 1857–1864. [Google Scholar] [CrossRef]
- Niiranen, T.J.; Kalesan, B.; Hamburg, N.M.; Benjamin, E.J.; Mitchell, G.F.; Vasan, R.S. Relative Contributions of Arterial Stiffness and Hypertension to Cardiovascular Disease: The Framingham Heart Study. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef]
- Westerhof, N. Cardiac work and efficiency. Cardiovasc. Res. 2000, 48, 4–7. [Google Scholar] [CrossRef]
- O’Rourke, M.F.; Hashimoto, J. Mechanical factors in arterial aging: A clinical perspective. J. Am. Coll. Cardiol. 2007, 50, 1–13. [Google Scholar] [CrossRef]
- Custodis, F.; Schirmer, S.H.; Baumhakel, M.; Heusch, G.; Bohm, M.; Laufs, U. Vascular pathophysiology in response to increased heart rate. J. Am. Coll. Cardiol. 2010, 56, 1973–1983. [Google Scholar] [CrossRef]
- Benetos, A.; Adamopoulos, C.; Bureau, J.M.; Temmar, M.; Labat, C.; Bean, K.; Thomas, F.; Pannier, B.; Asmar, R.; Zureik, M.; et al. Determinants of accelerated progression of arterial stiffness in normotensive subjects and in treated hypertensive subjects over a 6-year period. Circulation 2002, 105, 1202–1207. [Google Scholar] [CrossRef]
- Aune, D.; Sen, A.; o’Hartaigh, B.; Janszky, I.; Romundstad, P.R.; Tonstad, S.; Vatten, L.J. Resting heart rate and the risk of cardiovascular disease, total cancer, and all-cause mortality—A systematic review and dose-response meta-analysis of prospective studies. Nutr. Metab. Cardiovasc. Dis. NMCD 2017, 27, 504–517. [Google Scholar] [CrossRef]
- Bohm, M.; Reil, J.C.; Deedwania, P.; Kim, J.B.; Borer, J.S. Resting heart rate: Risk indicator and emerging risk factor in cardiovascular disease. Am. J. Med. 2015, 128, 219–228. [Google Scholar] [CrossRef]
- Oliva, F.; Sormani, P.; Contri, R.; Campana, C.; Carubelli, V.; Ciro, A.; Morandi, F.; Di Tano, G.; Mortara, A.; Senni, M.; et al. Heart rate as a prognostic marker and therapeutic target in acute and chronic heart failure. Int. J. Cardiol. 2018, 253, 97–104. [Google Scholar] [CrossRef]
- Dobre, D.; Borer, J.S.; Fox, K.; Swedberg, K.; Adams, K.F.; Cleland, J.G.; Cohen-Solal, A.; Gheorghiade, M.; Gueyffier, F.; O’Connor, C.M.; et al. Heart rate: A prognostic factor and therapeutic target in chronic heart failure. The distinct roles of drugs with heart rate-lowering properties. Eur. J. Heart Fail. 2014, 16, 76–85. [Google Scholar] [CrossRef]
- Bangalore, S.; Sawhney, S.; Messerli, F.H. Relation of beta-blocker-induced heart rate lowering and cardioprotection in hypertension. J. Am. Coll. Cardiol. 2008, 52, 1482–1489. [Google Scholar] [CrossRef]
- Olafiranye, O.; Qureshi, G.; Salciccioli, L.; Weber, M.; Lazar, J.M. Association of beta-blocker use with increased aortic wave reflection. J. Am. Soc. Hypertens. JASH 2008, 2, 64–69. [Google Scholar] [CrossRef]
- Manisty, C.H.; Zambanini, A.; Parker, K.H.; Davies, J.E.; Francis, D.P.; Mayet, J.; Mc, G.T.S.A.; Hughes, A.D. Differences in the magnitude of wave reflection account for differential effects of amlodipine- versus atenolol-based regimens on central blood pressure: An Anglo-Scandinavian Cardiac Outcome Trial substudy. Hypertension 2009, 54, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, I.B.; MacCallum, H.; Flint, L.; Cockcroft, J.R.; Newby, D.E.; Webb, D.J. The influence of heart rate on augmentation index and central arterial pressure in humans. J. Physiol. 2000, 525, 263–270. [Google Scholar] [CrossRef]
- Papaioannou, T.G.; Vlachopoulos, C.V.; Alexopoulos, N.A.; Dima, I.; Pietri, P.G.; Protogerou, A.D.; Vyssoulis, G.G.; Stefanadis, C.I. The effect of heart rate on wave reflections may be determined by the level of aortic stiffness: Clinical and technical implications. Am. J. Hypertens. 2008, 21, 334–340. [Google Scholar] [CrossRef]
- Pierdomenico, S.D.; Bucci, A.; Lapenna, D.; Cuccurullo, F.; Mezzetti, A. Heart rate in hypertensive patients treated with ACE inhibitors and long-acting dihydropyridine calcium antagonists. J. Cardiovasc. Pharmacol. 2002, 40, 288–295. [Google Scholar] [CrossRef]
- Hohneck, A.L.; Fries, P.; Stroder, J.; Schneider, G.; Wagenpfeil, S.; Schirmer, S.H.; Bohm, M.; Laufs, U.; Custodis, F. Effects of heart rate reduction with ivabradine on vascular stiffness and endothelial function in chronic stable coronary artery disease. J. Hypertens. 2019, 37, 1023–1031. [Google Scholar] [CrossRef]
- Bruno, R.M.; Cartoni, G.; Stea, F.; Armenia, S.; Bianchini, E.; Buralli, S.; Giannarelli, C.; Taddei, S.; Ghiadoni, L. Carotid and aortic stiffness in essential hypertension and their relation with target organ damage: The CATOD study. J. Hypertens. 2017, 35, 310–318. [Google Scholar] [CrossRef]
- Van Sloten, T.T.; Sedaghat, S.; Laurent, S.; London, G.M.; Pannier, B.; Ikram, M.A.; Kavousi, M.; Mattace-Raso, F.; Franco, O.H.; Boutouyrie, P.; et al. Carotid stiffness is associated with incident stroke: A systematic review and individual participant data meta-analysis. J. Am. Coll. Cardiol. 2015, 66, 2116–2125. [Google Scholar] [CrossRef]
- Paini, A.; Boutouyrie, P.; Calvet, D.; Tropeano, A.I.; Laloux, B.; Laurent, S. Carotid and aortic stiffness: Determinants of discrepancies. Hypertension 2006, 47, 371–376. [Google Scholar] [CrossRef]
- Mathersul, D.; Palmer, D.M.; Gur, R.C.; Gur, R.E.; Cooper, N.; Gordon, E.; Williams, L.M. Explicit identification and implicit recognition of facial emotions: II. Core domains and relationships with general cognition. J. Clin. Exp. Neuropsychol. 2009, 31, 278–291. [Google Scholar] [CrossRef]
- Lee, J.; Fu, Z.; Chung, M.; Jang, D.J.; Lee, H.J. Role of milk and dairy intake in cognitive function in older adults: A systematic review and meta-analysis. Nutr. J. 2018, 17, 82. [Google Scholar] [CrossRef]
- Zajac, I.T.; Herreen, D.; Bastiaans, K.; Dhillon, V.S.; Fenech, M. The Effect of Whey and Soy Protein Isolates on Cognitive Function in Older Australians with Low Vitamin B12: A Randomised Controlled Crossover Trial. Nutrients 2018, 11, 19. [Google Scholar] [CrossRef]
- Heffernan, K.S. Carotid artery stiffness and cognitive function in adults with and without type 2 diabetes: Extracranial contribution to an intracranial problem? Atherosclerosis 2016, 253, 268–269. [Google Scholar] [CrossRef] [PubMed]
- Rigamonti, A.E.; Leoncini, R.; Casnici, C.; Marelli, O.; Col, A.; Tamini, S.; Lucchetti, E.; Cicolini, S.; Abbruzzese, L.; Cella, S.G.; et al. Whey Proteins Reduce Appetite, Stimulate Anorexigenic Gastrointestinal Peptides and Improve Glucometabolic Homeostasis in Young Obese Women. Nutrients 2019, 11, 247. [Google Scholar] [CrossRef] [PubMed]
- Stojkovic, V.; Simpson, C.A.; Sullivan, R.R.; Cusano, A.M.; Kerstetter, J.E.; Kenny, A.M.; Insogna, K.L.; Bihuniak, J.D. The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial. Nutrients 2017, 9, 484. [Google Scholar] [CrossRef] [PubMed]
- Tomiyama, H.; Hashimoto, H.; Tanaka, H.; Matsumoto, C.; Odaira, M.; Yamada, J.; Yoshida, M.; Shiina, K.; Nagata, M.; Yamashina, A. Synergistic relationship between changes in the pulse wave velocity and changes in the heart rate in middle-aged Japanese adults: A prospective study. J. Hypertens. 2010, 28, 687–694. [Google Scholar] [CrossRef]
- Jae, S.Y.; Heffernan, K.S.; Yoon, E.S.; Park, S.H.; Choi, Y.H.; Fernhall, B.; Park, J.B. Pulsatile stress, inflammation and change in arterial stiffness. J. Atheroscler. Thromb. 2012, 19, 1035–1042. [Google Scholar] [CrossRef][Green Version]
- McEniery, C.M.; Wilkinson, I.B.; Johansen, N.B.; Witte, D.R.; Singh-Manoux, A.; Kivimaki, M.; Tabak, A.G.; Brunner, E.J.; Shipley, M.J. Nondiabetic Glucometabolic Status and Progression of Aortic Stiffness: The Whitehall II Study. Diabetes Care 2017, 40, 599–606. [Google Scholar] [CrossRef]
- Zhu, K.; Meng, X.; Kerr, D.A.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. The effects of a two-year randomized, controlled trial of whey protein supplementation on bone structure, IGF-1, and urinary calcium excretion in older postmenopausal women. J. Bone Miner. Res. 2011, 26, 2298–2306. [Google Scholar] [CrossRef]
- Chale, A.; Cloutier, G.J.; Hau, C.; Phillips, E.M.; Dallal, G.E.; Fielding, R.A. Efficacy of whey protein supplementation on resistance exercise-induced changes in lean mass, muscle strength, and physical function in mobility-limited older adults. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2013, 68, 682–690. [Google Scholar] [CrossRef]
- Sattler, F.R.; Rajicic, N.; Mulligan, K.; Yarasheski, K.E.; Koletar, S.L.; Zolopa, A.; Alston Smith, B.; Zackin, R.; Bistrian, B. Evaluation of high-protein supplementation in weight-stable HIV-positive subjects with a history of weight loss: A randomized, double-blind, multicenter trial. Am. J. Clin. Nutr. 2008, 88, 1313–1321. [Google Scholar] [CrossRef]
- Fiatarone, M.A.; O’Neill, E.F.; Ryan, N.D.; Clements, K.M.; Solares, G.R.; Nelson, M.E.; Roberts, S.B.; Kehayias, J.J.; Lipsitz, L.A.; Evans, W.J. Exercise training and nutritional supplementation for physical frailty in very elderly people. N. Engl. J. Med. 1994, 330, 1769–1775. [Google Scholar] [CrossRef]
- Altorf-van der Kuil, W.; Engberink, M.F.; Brink, E.J.; van Baak, M.A.; Bakker, S.J.; Navis, G.; van’t Veer, P.; Geleijnse, J.M. Dietary protein and blood pressure: A systematic review. PLoS ONE 2010, 5, e12102. [Google Scholar] [CrossRef]
- Kerstetter, J.E.; Bihuniak, J.D.; Brindisi, J.; Sullivan, R.R.; Mangano, K.M.; Larocque, S.; Kotler, B.M.; Simpson, C.A.; Cusano, A.M.; Gaffney-Stomberg, E.; et al. The Effect of a Whey Protein Supplement on Bone Mass in Older Caucasian Adults. J. Clin. Endocrinol. Metab. 2015, 100, 2214–2222. [Google Scholar] [CrossRef]
- Roldan-Tapia, M.D.; Canovas, R.; Leon, I.; Garcia-Garcia, J. Cognitive Vulnerability in Aging May Be Modulated by Education and Reserve in Healthy People. Front. Aging Neurosci. 2017, 9, 340. [Google Scholar] [CrossRef]
- Heffernan, K.S.; Jae, S.Y.; Wilund, K.R.; Woods, J.A.; Fernhall, B. Racial differences in central blood pressure and vascular function in young men. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H2380–H2387. [Google Scholar] [CrossRef]
- Exner, D.V.; Dries, D.L.; Domanski, M.J.; Cohn, J.N. Lesser response to angiotensin-converting-enzyme inhibitor therapy in black as compared with white patients with left ventricular dysfunction. N. Engl. J. Med. 2001, 344, 1351–1357. [Google Scholar] [CrossRef]
| WPI n = 53 | CHO n = 46 | p Value | df | |
|---|---|---|---|---|
| Female Sex | 26(49.1) | 19(41.3) | 0.54 | 98 |
| Asthma | 1(1.9) | 0(0.0) | 1.00 | 98 |
| Hypertension | 23(43.4) | 17(37.0) | 0.54 | 98 |
| Dyslipidemia | 23(43.4) | 28(60.9) | 0.11 | 98 |
| Fam Hx CVD | 32(60.4) | 28(62.2) | 1.00 | 98 |
| Handedness | 0.25 | 98 | ||
| Right | 42(79.2) | 40(87.0) | ||
| Left | 9(17.0) | 3(6.5) | ||
| Ambidextrous | 2(3.8) | 3(6.5) | ||
| Education | 0.08 | 98 | ||
| High School | 5(9.4) | 1(2.2) | ||
| Some college | 8(15.1) | 3(6.5) | ||
| 2-yr Degree | 3(5.7) | 1(2.2) | ||
| College | 12(22.6) | 21(45.7) | ||
| Graduate degree | 25(47.2) | 20(43.5) | ||
| Age (years) ^ | 69 ± 7 | 67 ± 6 | 0.25 | 98 |
| Height (m) ^ | 1.67 ± 0.13 | 1.68 ± 0.10 | 0.95 | 98 |
| CESD Score ^ | 7 ± 6 | 7 ± 8 | 0.89 | 95 |
| MOCA Score ^ | 27 ± 2 | 27 ± 2 | 0.70 | 98 |
| WPI | CHO | Effects, p-Value (Partial η2) | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 12 weeks | Baseline | 12 weeks | Group | Time | Group × Time | df | |
| Weight (kg) | 78.2 ± 16.3 | 78.1 ± 16.3 | 76.3 ± 14.5 | 77.3 ± 15.0 | 0.67(0.00) | 0.18(0.02) | 0.051(0.04) | 98 |
| BMI (kg/m2) | 27.9 ± 5.6 | 27.8 ± 5.6 | 27.0 ± 3.9 | 27.4 ± 4.1 * | 0.65(0.00) | 0.18(0.02) | 0.04(0.04) | 98 |
| Body fat (%) | 30.9 ± 12.3 | 29.8 ± 13.3 | 29.4 ± 9.9 | 29.8 ± 10.7 | 0.76(0.00) | 0.51(0.01) | 0.14(0.02) | 93 |
| Waist Circ (cm) | 96.6 ± 13.2 | 96.1 ± 13.8 | 95.8 ± 12.0 | 96.3 ± 12.5 | 0.91(0.00) | 0.97(0.00) | 0.32(0.01) | 94 |
| Total cholesterol (mg/dL) | 189 ± 34 | 184 ± 36 | 182 ± 33 | 183 ± 34 | 0.51(0.01) | 0.41(0.01) | 0.20(0.02) | 93 |
| HDL (mg/dL) | 60 ± 22 | 58 ± 21 | 57 ± 19 | 55 ± 19 | 0.53(0.00) | 0.11(0.03) | 0.82(0.00) | 92 |
| LDL (mg/dL) | 105 ± 30 | 103 ± 34 | 98 ± 27 | 101 ± 26 | 0.39(0.01) | 0.80(0.00) | 0.36(0.01) | 83 |
| Triglycerides (mg/dL) | 112 ± 46 | 107 ± 48 | 129 ± 78 | 133 ± 57 | 0.08(0.03) | 0.70(0.00) | 0.11(0.03) | 91 |
| Glucose (mg/dL) | 94 ± 14 | 95 ± 15 | 92 ± 11 | 94 ± 11 | 0.43(0.01) | 0.09(0.03) | 0.53(0.00) | 93 |
| 6 m Walk (s) | 4.72 ± 0.64 | 4.78 ± 0.72 | 4.59 ± 0.64 | 4.86 ± 0.71 | 0.80(0.00) | <0.01(0.08) | 0.07(0.04) | 88 |
| IPAQ ^ | 3065 ± 3023 | 2602 ± 2552 | 4597 ± 4009 | 4367 ± 4314 | 0.15(0.22) | 0.32(0.18) | 0.99(0.02) | 78 |
| MVPA (min/d) ^ | 23.2 ± 19.3 | 19.2 ± 15.8 | 26.0 ± 27.6 | 30.9 ± 34.0 | 0.99(0.08) | 0.99(0.04) | 0.13(0.25) | 65 |
| WPI | CHO | Effects, p-Value (Partial η2) | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 12 weeks | Baseline | 12 weeks | Group | Time | Group × Time | df | |
| Brachial | ||||||||
| Systolic pressure (mmHg) | 125 ± 13 | 123 ± 12 | 127 ± 11 | 128 ± 12 | 0.10(0.03) | 0.42(0.01) | 0.13(0.02) | 96 |
| Diastolic pressure (mmHg) | 79 ± 8 | 76 ± 7 | 79 ± 5 | 78 ± 6 | 0.30(0.01) | <0.01(0.09) | 0.20(0.02) | 96 |
| Pulse pressure (mmHg) | 46 ± 9 | 46 ± 9 | 48 ± 8 | 49 ± 9 | 0.13(0.02) | 0.28(0.01) | 0.28(0.01) | 96 |
| Mean pressure (mmHg) | 94 ± 9 | 92 ± 8 | 95 ± 7 | 95 ± 7 | 0.12(0.03) | 0.03(0.05) | 0.13(0.02) | 96 |
| Aorta | ||||||||
| Augmentation index 75 | 24 ± 10 | 23 ± 11 | 25 ± 9 | 25 ± 8 | 0.25(0.01) | 0.50(0.01) | 0.52(0.00) | 96 |
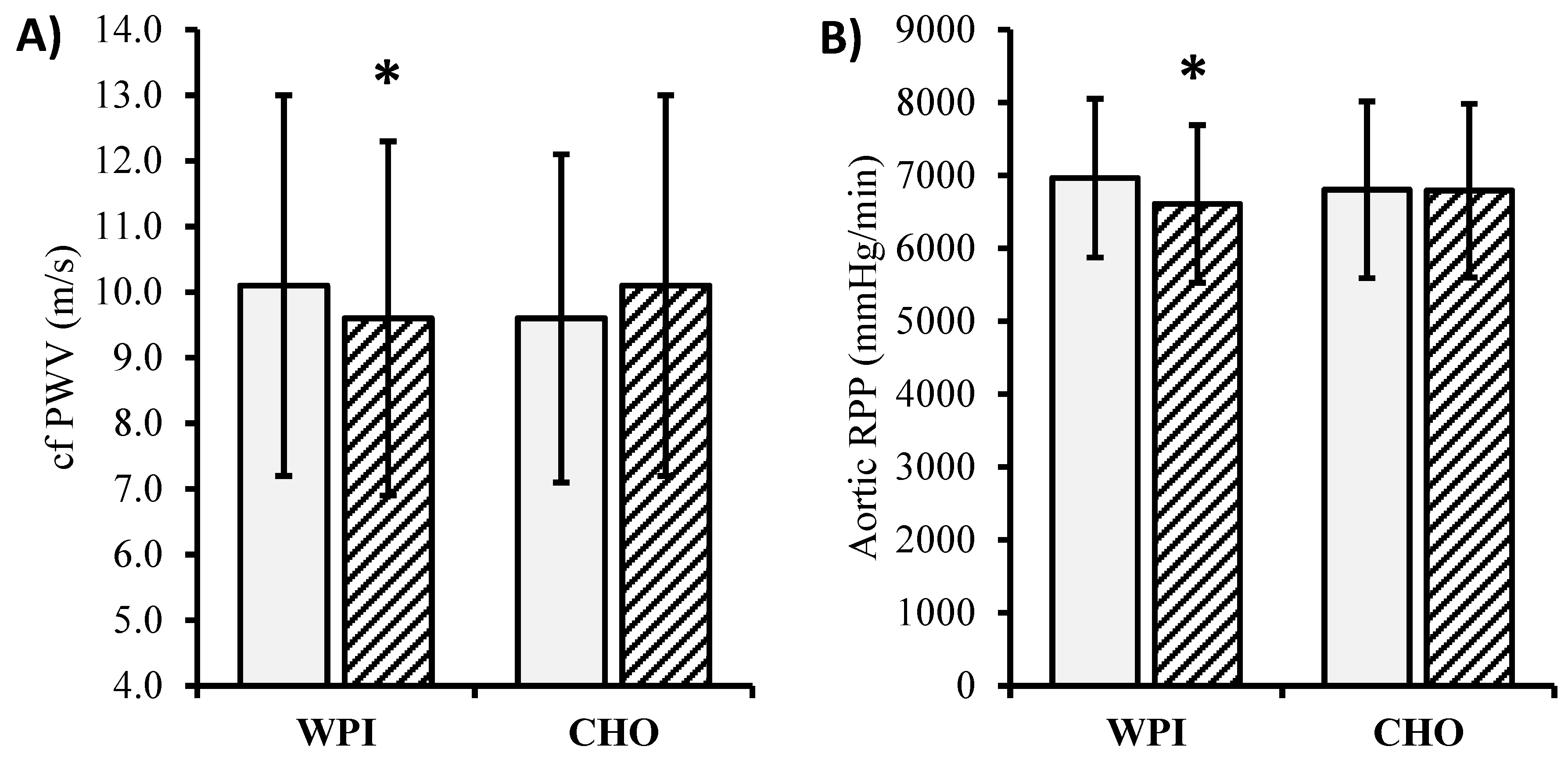
| Systolic pressure (mmHg) | 115 ± 13 | 112 ± 12 | 117 ± 11 | 119 ± 12 | 0.04(0.04) | 0.48(0.00) | 0.09(0.03) | 97 |
| Pulse pressure (mmHg) | 35 ± 10 | 35 ± 7 | 37 ± 8 | 39 ± 10 | 0.06(0.04) | 0.24(0.01) | 0.26(0.01) | 97 |
| Heart rate (b/min) | 60 ± 8 | 56 ± 8 * | 57 ± 9 | 56 ± 9 | 0.39(0.01) | <0.001(0.13) | <0.01(0.07) | 96 |
| Cf-PWV/MAP (m/s/mmHg × 102) | 10.7 ± 3.1 | 10.5 ± 3.0 | 10.0 ± 2.3 | 10.6 ± 2.8 * | 0.73(0.00) | 0.46(0.01) | 0.03(0.05) | 95 |
| WPI | CHO | Effects, p-Value (Partial η2) | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 12 weeks | Baseline | 12 weeks | Group | Time | Group × Time | df | |
| Common Carotid Artery | ||||||||
| Pulsatility index | 1.37 ± 0.24 | 1.40 ± 0.30 | 1.40 ± 0.28 | 1.41 ± 0.29 | 0.77(0.00) | 0.34(0.01) | 0.84(0.00) | 96 |
| β-stiffness | 8.5 ± 4.3 | 8.5 ± 3.8 | 8.0 ± 2.5 | 9.3 ± 5.0 | 0.39(0.01) | 0.45(0.01) | 0.16(0.02) | 94 |
| W1 (mmHg/m/s3) | 7.1 ± 4.7 | 7.2 ± 4.1 | 7.2 ± 4.2 | 7.6 ± 6.6 | 0.84(0.00) | 0.91(0.00) | 0.75(0.00) | 93 |
| NA (mmHg/m/s2) | 22.9 ± 11.6 | 24.5 ± 14.4 | 25.2 ± 16.6 | 26.0 ± 23.8 | 0.80(0.00) | 0.80(0.00) | 0.34(0.01) | 93 |
| Mean Diameter (mm) | 5.92 ± 0.62 | 5.88 ± 0.63 | 5.86 ± 0.65 | 5.86 ± 0.67 | 0.48(0.01) | 0.55(0.00) | 0.48(0.01) | 96 |
| ∆Diameter (mm) | +0.11 ± 0.15 | +0.17 ± 0.15 | +0.10 ± 0.20 | +0.13 ± 0.13 | 0.08(0.03) | 0.31(0.01) | 0.57(0.00) | 95 |
| IMT (mm) | 0.66 ± 0.10 | 0.68 ± 0.12 | 0.67 ± 0.14 | 0.67 ± 0.14 | 0.80(0.00) | 0.24(0.01) | 0.09(0.03) | 96 |
| Mean velocity (cm/s) | 54 ± 12 | 52 ± 14 | 53 ± 15 | 51 ± 14 | 0.85(0.00) | <0.01(0.09) | 0.65(0.00) | 92 |
| Middle Cerebral Artery | ||||||||
| Mean velocity (cm/s) | 54 ± 12 | 52 ± 14 | 53 ± 15 | 51 ± 14 | 0.85(0.00) | <0.01(0.09) | 0.65(0.00) | 93 |
| ∆Mean velocity (cm/s) | +5 ± 7 | +4 ± 5 | +5 ± 6 | +5 ± 5 | 0.38(0.01) | 0.36(0.01) | 0.57(0.00) | 92 |
| Pulsatility index | 0.86 ± 0.15 | 0.88 ± 0.18 | 0.87 ± 0.13 | 0.88 ± 0.18 | 0.79(0.00) | 0.67(0.00) | 0.62(0.00) | 93 |
| ∆Pulsatility index | +0.00 ± 0.06 | −0.02 ± 0.05 | −0.00 ± 0.05 | −0.01 ± 0.05 | 0.70(0.00) | 0.02(0.06) | 0.42(0.01) | 92 |
| Conductance (cm/s/mmHg × 102) | 58.0 ± 15.2 | 57.2 ± 17.0 | 55.6 ± 16.8 | 54.5 ± 17.1 | 0.54(0.00) | 0.13(0.02) | 0.77(0.00) | 93 |
| ∆Conductance (cm/s/mmHg × 102) | +0.2 ± 6.0 | −0.9 ± 6.6 | +0.2 ± 6.5 | +0.4 ± 5.6 | 0.51(0.01) | 0.60(0.00) | 0.41(0.01) | 90 |
| WPI | CHO | Effects, p-Value (z/sqrt(n)) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Domain/Construct | Task | Baseline | 12 weeks | Baseline | 12 weeks | Group | Time | Group × Time | df |
| Executive function | Maze | −0.34 ± 2.80 | 0.34 ± 2.14 | −0.29 ± 2.08 | 0.74 ± 2.62 | 0.99(0.07) | <0.01(0.33) | 0.99(0.08) | 93 |
| Impulsivity | Go-no-go | −0.23 ± 1.65 | 0.28 ± 1.16 | −0.12 ± 1.54 | 0.18 ± 1.12 | 0.99(0.04) | 0.051(0.25) | 0.77(0.12) | 92 |
| Emotion Identification | Recognition | −0.14 ± 1.15 | 0.43 ± 1.37 * | −0.15 ± 1.98 | −0.11 ± 2.06 | 0.87(0.11) | <0.01(0.31) | 0.04(0.25) | 96 |
| Memory | Recall | 0.30 ± 5.76 | 0.66 ± 5.25 | −0.21 ± 5.29 | −0.43 ± 6.40 | 0.53(0.14) | 0.99(0.05) | 0.36(0.16) | 94 |
| Information Processing | Interference | 0.85 ± 4.73 | 2.30 ± 3.62 | −0.06 ± 5.75 | 0.62 ± 5.19 | 0.98(0.46) | 0.045(0.27) | 0.92(0.11) | 81 |
| Attention | CPT | 0.02 ± 2.05 | 0.03 ± 1.82 | −0.10 ± 2.34 | 0.28 ± 1.56 | 0.99(0.04) | 0.79(0.11) | 0.77(0.12) | 95 |
| Working memory | Digit span | 0.23 ± 1.65 | 0.27 ± 1.93 | −0.38 ± 2.23 | 0.00 ± 1.71 | 0.52(0.09) | 0.99(0.09) | 0.99(0.05) | 87 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lefferts, W.K.; Augustine, J.A.; Spartano, N.L.; Hughes, W.E.; Babcock, M.C.; Heenan, B.K.; Heffernan, K.S. Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial. Nutrients 2020, 12, 1054. https://doi.org/10.3390/nu12041054
Lefferts WK, Augustine JA, Spartano NL, Hughes WE, Babcock MC, Heenan BK, Heffernan KS. Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial. Nutrients. 2020; 12(4):1054. https://doi.org/10.3390/nu12041054
Chicago/Turabian StyleLefferts, Wesley K., Jacqueline A. Augustine, Nicole L. Spartano, William E. Hughes, Matthew C. Babcock, Brigid K. Heenan, and Kevin S. Heffernan. 2020. "Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial" Nutrients 12, no. 4: 1054. https://doi.org/10.3390/nu12041054
APA StyleLefferts, W. K., Augustine, J. A., Spartano, N. L., Hughes, W. E., Babcock, M. C., Heenan, B. K., & Heffernan, K. S. (2020). Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial. Nutrients, 12(4), 1054. https://doi.org/10.3390/nu12041054







