Prevalence of Low Plasma Vitamin B1 in the Stroke Population Admitted to Acute Inpatient Rehabilitation
Abstract
1. Introduction
2. Methods
2.1. Patient Population
2.2. VitB1 Measurement
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
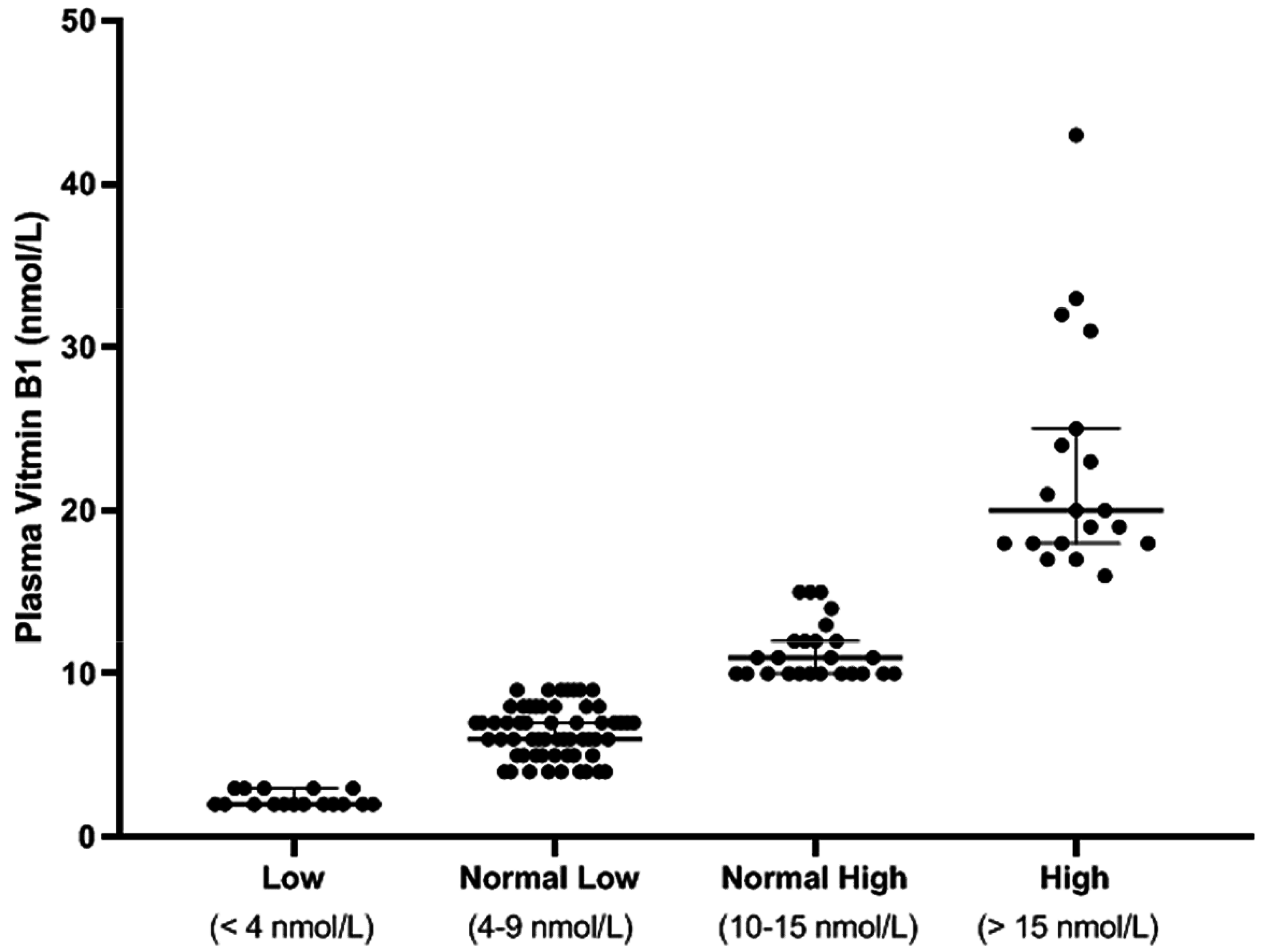
3.2. Plasma Thiamine Levels
3.2.1. Low VitB1
3.2.2. Normal Low VitB1
3.2.3. Normal High VitB1
3.2.4. High VitB1
4. Discussion
4.1. VitB1 Deficiency Mimicking Stroke
4.2. VitB1 and Stroke
4.3. Plasma VitB1
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kennedy, D.O. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 27, 68. [Google Scholar] [CrossRef]
- Raichle, M.E. Two views of brain function. Trends Cogn. Sci. 2010, 14, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Spector, R.; Johanson, C.E. Vitamin transport and homeostasis in mammalian brain: Focus on Vitamins B and E. J. Neurochem. 2007, 103, 425–438. [Google Scholar] [CrossRef] [PubMed]
- Spector, R. Nutrient transport systems in brain: 40 years of progress. J. Neurochem. 2009, 111, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Kerns, J.C.; Arundel, C.; Chawla, L.S. Thiamin deficiency in people with obesity. Adv. Nutr. 2015, 6, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, J.A.; Parrott, J. New considerations on the neuromodulatory role of thiamine. Pharmacology 2012, 89, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Bâ, A. Metabolic and structural role of thiamine in nervous tissues. Cell Mol. Neurobiol. 2008, 28, 923–931. [Google Scholar] [CrossRef]
- Oliveira, F.A.; Galan, D.T.; Ribeiro, A.M.; Santos Cruz, J. Thiamine deficiency during pregnancy leads to cerebellar neuronal death in rat offspring: Role of voltage-dependent K+ channels. Brain Res. 2007, 23, 79–86. [Google Scholar] [CrossRef]
- Rindi, G.; Patrini, C.; Comincioli, V.; Reggiani, C. Thiamine content and turnover rates of some rat nervous regions, using labeled thiamine as a tracer. Brain Res. 1980, 181, 369–380. [Google Scholar] [CrossRef]
- Rindi, G. Metabolism of thiamin and its phosphoric esters in different regions of the nervous system: A new approach. Acta Vitam. Enzym. 1982, 4, 59–68. [Google Scholar]
- Lonsdale, D. Thiamine metabolism in disease. CRC Crit. Rev. Clin. Lab. Sci. 1975, 5, 289–313. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D. A review of the biochemistry, metabolism and clinical benefits of thiamin (e) and its derivatives. Evid. Based Complement. Altern. Med. ECAM 2006, 3, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D. Thiamine and magnesium deficiencies: Keys to disease. Med. Hypotheses 2015, 84, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Donnino, M.W.; Vega, J.; Miller, J.; Walsh, M. Myths and misconceptions of Wernicke’s encephalopathy: What every emergency physician should know. Ann. Emerg. Med. 2007, 50, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.D.; Marshall, E.J. The natural history and pathophysiology of Wernicke’s Encephalopathy and Korsakoff’s Psychosis. Alcohol. Alcohol. Oxf. Oxfs. 2006, 41, 151–158. [Google Scholar] [CrossRef]
- Thomson, A.D.; Guerrini, I.; Marshall, E.J. The evolution and treatment of Korsakoff’s syndrome: Out of sight, out of mind? Neuropsychol. Rev. 2012, 22, 81–92. [Google Scholar] [CrossRef]
- Manzanares, W.; Hardy, G. Thiamine supplementation in the critically ill. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 610–617. [Google Scholar] [CrossRef]
- O’Keeffe, S.T.; Tormey, W.P.; Glasgow, R.; Lavan, J.N. Thiamine deficiency in hospitalized elderly patients. Gerontology 1994, 40, 18–24. [Google Scholar] [CrossRef]
- Butterworth, R.F.; Gaudreau, C.; Vincelette, J.; Bourgault, A.M.; Lamothe, F.; Nutini, A.M. Thiamine deficiency and Wernicke’s encephalopathy in AIDS. Metab. Brain Dis. 1991, 6, 207–212. [Google Scholar] [CrossRef]
- Harper, C. Wernicke’s encephalopathy: A more common disease than realised. A neuropathological study of 51 cases. J. Neurol. Neurosurg. Psychiatry 1979, 42, 226–231. [Google Scholar] [CrossRef]
- Williams, R.D.; Mason, H.L.; Power, M.H.; Wilder, R.M. Induced Thiamine (Vitamin B1) Deficiency in Man: Relation of Depletion of Thiamine to Development of Biochemical Defect and of Polyneuropathy. Arch. Intern. Med. 1943, 71, 38–53. [Google Scholar] [CrossRef]
- Walker, J.; Kepner, A. Wernicke’s encephalopathy presenting as acute psychosis after gastric bypass. J. Emerg. Med. 2012, 43, 811–814. [Google Scholar] [CrossRef] [PubMed]
- Frank, L.L. Thiamin in Clinical Practice. JPEN J. Parenter. Enteral. Nutr. 2015, 39, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Stroh, C.; Meyer, F.; Manger, T. Beriberi, a severe complication after metabolic surgery—Review of the literature. Obes. Facts. 2014, 7, 246–252. [Google Scholar] [CrossRef]
- Office of Dietary Supplements—Thiamin. Available online: https://ods.od.nih.gov/factsheets/Thiamin-HealthProfessional/ (accessed on 23 April 2019).
- Lu, J.; Frank, E.L. Rapid HPLC measurement of thiamine and its phosphate esters in whole blood. Clin. Chem. 2008, 54, 901–906. [Google Scholar] [CrossRef]
- Pourhassan, M.; Angersbach, B.; Lueg, G.; Klimek, C.N.; Wirth, R. Blood Thiamine Level and Cognitive Function in Older Hospitalized Patients. J. Geriatr. Psychiatry Neurol. 2018, 32, 90–96. [Google Scholar] [CrossRef]
- Latt, N.; Dore, G. Thiamine in the treatment of Wernicke encephalopathy in patients with alcohol use disorders. Intern. Med. J. 2014, 44, 911–915. [Google Scholar] [CrossRef]
- Fama, R.; Le Berre, A.-P.; Hardcastle, C.; Sassoon, S.A.; Pfefferbaum, A.; Sullivan, E.V.; Zahr, N.M. Neurological, nutritional and alcohol consumption factors underlie cognitive and motor deficits in chronic alcoholism. Addict. Biol. 2019, 24, 290–302. [Google Scholar] [CrossRef]
- Lu, J.; Pan, X.; Fei, G.; Wang, C.; Zhao, L.; Sang, S.; Liu, H.; Liu, M.; Wang, H.; Wang, Z.; et al. Correlation of thiamine metabolite levels with cognitive function in the non-demented elderly. Neurosci. Bull. 2015, 31, 676–684. [Google Scholar] [CrossRef]
- Dabar, G.; Harmouche, C.; Habr, B.; Riachi, M.; Jaber, B. Shoshin Beriberi in Critically-Ill patients: Case series. Nutr. J. 2015, 13, 51. [Google Scholar] [CrossRef]
- Attaluri, P.; Castillo, A.; Edriss, H.; Nugent, K. Thiamine Deficiency: An Important Consideration in Critically Ill Patients. Am. J. Med. Sci. 2018, 356, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Oudman, E.; Wijnia, J.W.; van Dam, M.; Biter, L.U.; Postma, A. Preventing Wernicke Encephalopathy after Bariatric Surgery. Obes. Surg. 2018, 28, 2060–2068. [Google Scholar] [CrossRef] [PubMed]
- Lopes da Silva, S.; Vellas, B.; Elemans, S.; Luchsinger, J.; Kamphuis, P.; Yaffe, K.; Sijben, J.; Groenendijk, M.; Stijnen, T. Plasma nutrient status of patients with Alzheimer’s disease: Systematic review and meta-analysis. Alzheimers Dement. J. Alzheimers Assoc. 2014, 10, 485–502. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.; Nguyễn, L.T. The role of thiamine in HIV infection. Int. J. Infect. Dis. IJID Off. Publ. Int. Soc. Infect. Dis. 2013, 17, e221–e227. [Google Scholar]
- Lu’o’ng, K.V.Q.; Nguyễn, L.T.H. The role of thiamine in cancer: Possible genetic and cellular signaling mechanisms. Cancer Genom. Proteom. 2013, 10, 169–185. [Google Scholar]
- Page, G.L.J.; Laight, D.; Cummings, M.H. Thiamine deficiency in diabetes mellitus and the impact of thiamine replacement on glucose metabolism and vascular disease. Int. J. Clin. Pract. 2011, 65, 684–690. [Google Scholar] [CrossRef]
- Nath, A.; Tran, T.; Shope, T.R.; Koch, T.R. Prevalence of clinical thiamine deficiency in individuals with medically complicated obesity. Nutr. Res. 2017, 37, 29–36. [Google Scholar] [CrossRef]
- Swenson, A.J.; St Louis, E.K. Computed tomography findings in thiamine deficiency-induced coma. Neurocrit. Care 2006, 5, 45–48. [Google Scholar] [CrossRef]
- Helbok, R.; Beer, R.; Engelhardt, K.; Broessner, G.; Lackner, P.; Brenneis, C.; Pfausler, B.; Schmutzhard, E. Intracerebral haemorrhage in a malnourished patient, related to Wernicke’s encephalopathy. Eur. J. Neurol. 2008, 15, e99–e100. [Google Scholar] [CrossRef]
- Al-Bayati, A.R.; Nichols, J.; Jovin, T.G.; Jadhav, A.P. Thiamine Deficiency Presenting as Intraventricular Hemorrhage. Stroke 2016, 47, e95–e97. [Google Scholar] [CrossRef]
- Karapanayiotides, T.; Anastasiou, A.; Barmpas, N.; Grigoriadis, N.; Karacostas, D. A “posterior circulation stroke” that benefits from vitamins. Am. J. Med. 2014, 127, e1–e2. [Google Scholar] [CrossRef]
- Blum, A.; Ovadia, M.; Rosen, G.; Simsolo, C. Immediate recovery of an “ischemic stroke” following treatment with intravenous thiamine (vitamin B1). Isr. Med. Assoc. J. IMAJ 2014, 16, 518–519. [Google Scholar] [PubMed]
- Choi, J.Y.; Scarborough, T.K. Stroke and seizure following a recent laparoscopic Roux-en-Y gastric bypass. Obes. Surg. 2004, 14, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Geng, X.; Khan, H.; Pendy, J.T., Jr.; Peng, C.; Li, X.; Rafols, J.A.; Ding, Y. Exacerbation of Brain Injury by Post-Stroke Exercise Is Contingent Upon Exercise Initiation Timing. Front Cell Neurosci. 5 October 2017. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5633611/ (accessed on 22 April 2019).
- Martin, P.R.; Singleton, C.K.; Hiller-Sturmhöfel, S. The role of thiamine deficiency in alcoholic brain disease. Alcohol. Res. Health 2003, 27, 134–142. [Google Scholar] [PubMed]
- Corcoran, P.J.; Jebsen, R.H.; Brengelmann, G.L.; Simons, B.C. Effects of plastic and metal leg braces on speed and energy cost of hemiparetic ambulation. Arch. Phys. Med. Rehabil. 1970, 51, 69–77. [Google Scholar]
- Gersten, J.W.; Orr, W. External work of walking in hemiparetic patients. Scand. J. Rehabil. Med. 1971, 3, 85–88. [Google Scholar]
- Laur, C.; McCullough, J.; Davidson, B.; Keller, H. Becoming Food Aware in Hospital: A Narrative Review to Advance the Culture of Nutrition Care in Hospitals. Healthcare 2015, 3, 393–407. [Google Scholar] [CrossRef]
- Ross, A.C.; Caballero, B.H.; Cousins, R.J.; Tucker, K.L.; Ziegler, T.R. Modern Nutrition in Health and Disease, 11th ed.; Wolters Kluwer Health Adis (ESP), 2012; Available online: https://jhu.pure.elsevier.com/en/publications/modern-nutrition-in-health-and-disease-eleventh-edition (accessed on 17 April 2019).
- Talwar, D.; Davidson, H.; Cooney, J.; St JO’Reilly, D. Vitamin B(1) status assessed by direct measurement of thiamin pyrophosphate in erythrocytes or whole blood by HPLC: Comparison with erythrocyte transketolase activation assay. Clin. Chem. 2000, 46, 704–710. [Google Scholar] [CrossRef]
- Tallaksen, C.M.; Sande, A.; Bøhmer, T.; Bell, H.; Karlsen, J. Kinetics of thiamin and thiamin phosphate esters in human blood, plasma and urine after 50 mg intravenously or orally. Eur. J. Clin. Pharm. 1993, 44, 73–78. [Google Scholar] [CrossRef]
- Tallaksen, C.M.; Bøhmer, T.; Bell, H.; Karlsen, J. Concomitant determination of thiamin and its phosphate esters in human blood and serum by high-performance liquid chromatography. J. Chromatogr. 1991, 564, 127–136. [Google Scholar] [CrossRef]
- Herve, C.; Beyne, P.; Lettéron, P.; Delacoux, E. Comparison of erythrocyte transketolase activity with thiamine and thiamine phosphate ester levels in chronic alcoholic patients. Clin. Chim. Acta Int. J. Clin Chem. 1995, 234, 91–100. [Google Scholar] [CrossRef]
- Lynch, P.L.; Young, I.S. Determination of thiamine by high-performance liquid chromatography. J. Chromatogr. A 2000, 881, 267–284. [Google Scholar] [CrossRef]
- Jenčo, J.; Krčmová, L.K.; Solichová, D.; Solich, P. Recent trends in determination of thiamine and its derivatives in clinical practice. J. Chromatogr. A 2017, 1510, 1–12. [Google Scholar] [CrossRef]
- Donnino, M.W.; Carney, E.; Cocchi, M.N.; Barbash, I.; Chase, M.; Joyce, N.; Chou, P.P.; Ngo, L. Thiamine deficiency in critically ill patients with sepsis. J. Crit. Care 2010, 25, 576–581. [Google Scholar] [CrossRef]
- Donnino, M.W.; Cocchi, M.N.; Smithline, H.; Carney, E.; Chou, P.P.; Salciccioli, J. Coronary artery bypass graft surgery depletes plasma thiamine levels. Nutrition 2010, 26, 133–136. [Google Scholar] [CrossRef]
- Saltzman, E.; Karl, J.P. Nutrient deficiencies after gastric bypass surgery. Annu. Rev. Nutr. 2013, 33, 183–203. [Google Scholar] [CrossRef]
- Koch, T.R.; Finelli, F.C. Postoperative metabolic and nutritional complications of bariatric surgery. Gastroenterol. Clin. N. Am. 2010, 39, 109–124. [Google Scholar] [CrossRef]
- Frank, T.; Czeche, K.; Bitsch, R.; Stein, G. Assessment of thiamin status in chronic renal failure patients, transplant recipients and hemodialysis patients receiving a multivitamin supplementation. Int. J. Vitam. Nutr. Res. 2000, 70, 159–166. [Google Scholar] [CrossRef]
- Thornalley, P.J.; Babaei-Jadidi, R.; Al Ali, H.; Rabbani, N.; Antonysunil, A.; Larkin, J.; Ahmed, A.; Rayman, G.; Bodmer, C.W. High prevalence of low plasma thiamine concentration in diabetes linked to a marker of vascular disease. Diabetologia 2007, 50, 2164–2170. [Google Scholar] [CrossRef]
- Dancy, M.; Evans, G.; Gaitonde, M.K.; Maxwell, J.D. Blood thiamine and thiamine phosphate ester concentrations in alcoholic and non-alcoholic liver diseases. Br. Med. J. Clin. Res. Ed. 1984, 14, 79–82. [Google Scholar] [CrossRef]
- McCann, A.; Midttun, Ø.; Whitfield, K.C.; Kroeun, H.; Borath, M.; Sophonneary, P.; Ueland, P.M.; Green, T.J. Comparable Performance Characteristics of Plasma Thiamine and Erythrocyte Thiamine Diphosphate in Response to Thiamine Fortification in Rural Cambodian Women. Nutrients 2017, 29, 676. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.E.; Icke, G.C.; Thom, J.; Riley, W.J. Intestinal absorption of thiamin in man compared with folate and pyridoxal and its subsequent urinary excretion. J. Nutr. Sci. Vitam. (Tokyo) 1984, 30, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Coats, D.; Frank, E.L.; Reid, J.M.; Ou, K.; Chea, M.; Khin, M.; Preou, C.; Enders, F.T.; Fischer, P.R.; Topazian, M.; et al. Thiamine pharmacokinetics in Cambodian mothers and their breastfed infants. Am. J. Clin. Nutr. 2013, 98, 839–844. [Google Scholar] [CrossRef]
- Combs, G.F. The Vitamins Fundamental Aspects in Nutrition and Health, 4th ed.; Elsevier/Academic Press: Amsterdam, The Netherlands, 2012; p. 599. [Google Scholar]
- Gropper, S.S.; Smith, J.L. Advanced Nutrition and Human Metabolism, 6th ed.; Cengage Learning: Belmont, CA, USA, 2012; p. 608. [Google Scholar]
- Galvin, R.; Bråthen, G.; Ivashynka, A.; Hillbom, M.; Tanasescu, R.; Leone, M.A. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur. J. Neurol. 2010, 17, 1408–1418. [Google Scholar] [CrossRef] [PubMed]
- Singleton, C.K.; Martin, P.R. Molecular mechanisms of thiamine utilization. Curr. Mol. Med. 2001, 1, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ariaey-Nejad, M.R.; Balaghi, M.; Baker, E.M.; Sauberlich, H.E. Thiamin metabolism in man. Am. J. Clin. Nutr. 1970, 23, 764–778. [Google Scholar] [CrossRef] [PubMed]
| Low | Normal Low | Normal High | High | p | |
|---|---|---|---|---|---|
| N | 17 | 58 | 25 | 19 | |
| Age (years) | 58.18 (17.72) | 61.29 (15.62) | 66.2 (13.84) | 63.37 (15.53) | 0.433 |
| BMI | 30.23 (11.66) | 28.52 (7.25) | 26.25 (5.08) | 27.9 (8.33) | 0.512 |
| Gender | |||||
| Male | 9 | 33 | 12 | 10 | |
| Female | 8 | 25 | 13 | 9 | 0.901 |
| Ethnicity | |||||
| White | 11 | 40 | 19 | 15 | |
| Non-White | 6 | 18 | 6 | 4 | 0.722 |
| Stroke Type | |||||
| Hemorrhagic | 2 | 12 | 8 | 7 | |
| Ischemic | 13 | 40 | 15 | 11 | |
| Both | 2 | 6 | 2 | 1 | 0.620 |
| Comorbidities | |||||
| Hypertension | 11 | 48 | 20 | 16 | |
| Hyperlipidemia | 8 | 28 | 13 | 7 | |
| Cardiac Dysfunction | 4 | 19 | 4 | 6 | |
| Renal Dysfunction | 2 | 14 | 4 | 9 | |
| Diabetes Mellitus | 9 | 18 | 8 | 7 | 0.626 |
| Low | Normal Low | Normal High | High | |
|---|---|---|---|---|
| Number of values | 17 | 58 | 25 | 19 |
| Minimum | 2 | 4 | 10 | 16 |
| 25% Percentile | 2 | 5 | 10 | 18 |
| Median | 2 | 6 | 11 | 20 |
| 75% Percentile | 3 | 8 | 12 | 25 |
| Maximum | 3 | 9 | 15 | 43 |
| 95% CI of median | ||||
| Actual confidence level | 95.10% | 95.21% | 95.67% | 98.08% |
| Lower confidence limit | 2 | 6 | 10 | 18 |
| Upper confidence limit | 3 | 7 | 12 | 25 |
| Mean | 2.294 | 6.414 | 11.36 | 22.74 |
| Std. Deviation | 0.4697 | 1.579 | 1.753 | 7.164 |
| Std. Error of Mean | 0.1139 | 0.2073 | 0.3506 | 1.643 |
| Coefficient of variation | 20.47% | 24.61% | 15.43% | 31.51% |
| Skewness | 0.9936 | 0.02553 | 1.161 | 1.607 |
| Kurtosis | −1.166 | −0.9582 | 0.1225 | 2.277 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ehsanian, R.; Anderson, S.; Schneider, B.; Kennedy, D.; Mansourian, V. Prevalence of Low Plasma Vitamin B1 in the Stroke Population Admitted to Acute Inpatient Rehabilitation. Nutrients 2020, 12, 1034. https://doi.org/10.3390/nu12041034
Ehsanian R, Anderson S, Schneider B, Kennedy D, Mansourian V. Prevalence of Low Plasma Vitamin B1 in the Stroke Population Admitted to Acute Inpatient Rehabilitation. Nutrients. 2020; 12(4):1034. https://doi.org/10.3390/nu12041034
Chicago/Turabian StyleEhsanian, Reza, Sean Anderson, Byron Schneider, David Kennedy, and Vartgez Mansourian. 2020. "Prevalence of Low Plasma Vitamin B1 in the Stroke Population Admitted to Acute Inpatient Rehabilitation" Nutrients 12, no. 4: 1034. https://doi.org/10.3390/nu12041034
APA StyleEhsanian, R., Anderson, S., Schneider, B., Kennedy, D., & Mansourian, V. (2020). Prevalence of Low Plasma Vitamin B1 in the Stroke Population Admitted to Acute Inpatient Rehabilitation. Nutrients, 12(4), 1034. https://doi.org/10.3390/nu12041034





