Ethanolic Extract of Moringa oleifera Leaves Influences NF-κB Signaling Pathway to Restore Kidney Tissue from Cobalt-Mediated Oxidative Injury and Inflammation in Rats
Abstract
1. Introduction
2. Material and Methods
2.1. Ethical Statement
2.2. Tested Chemicals and Plant Extract
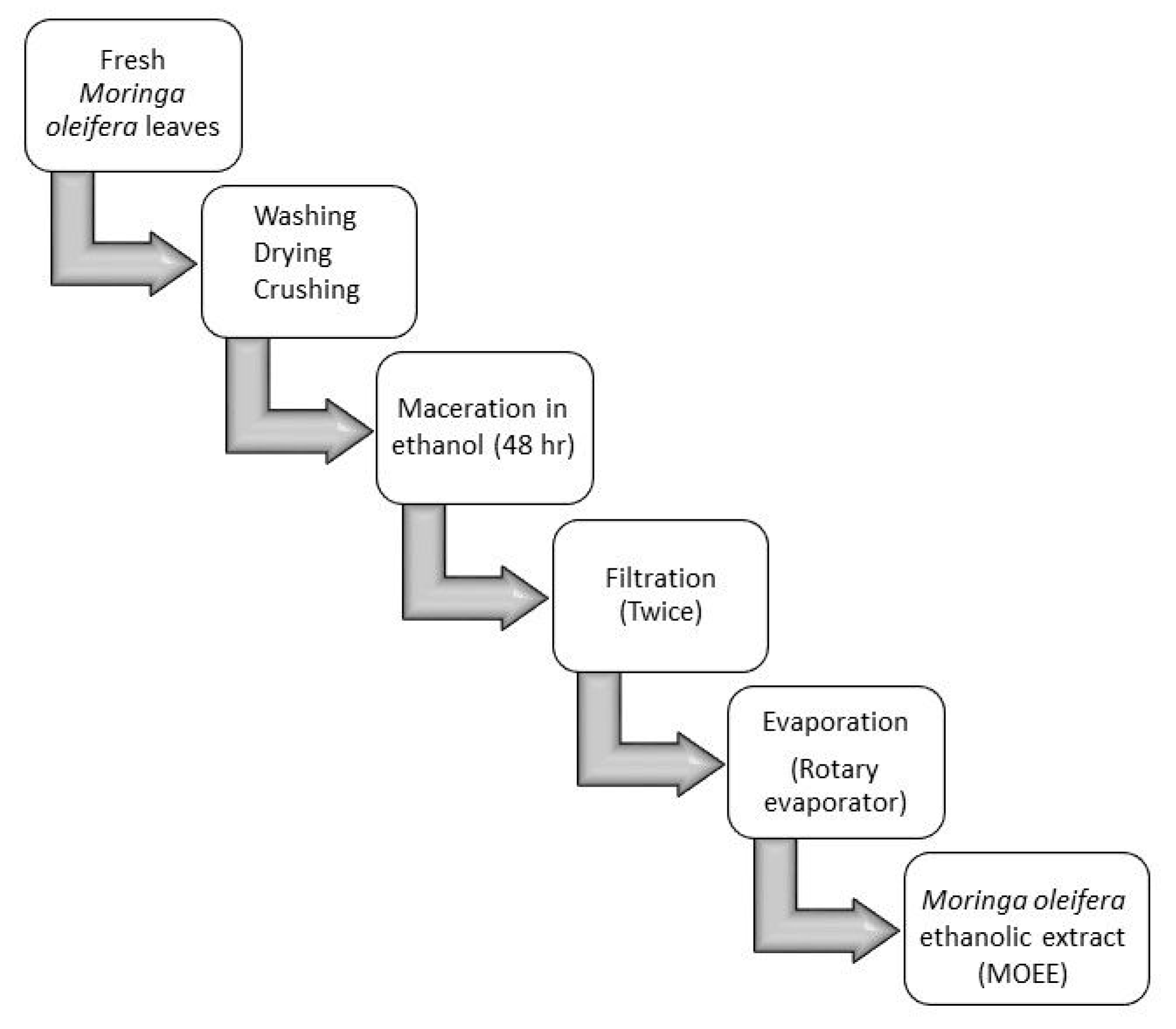
2.3. Preparation of the Ethanolic Extract of the Leaves of Moringa oleifera
2.4. Gas Chromatography/Mass Spectrometry Analysis (GC–MS) of MOEE Bioactive Chemical Constituents
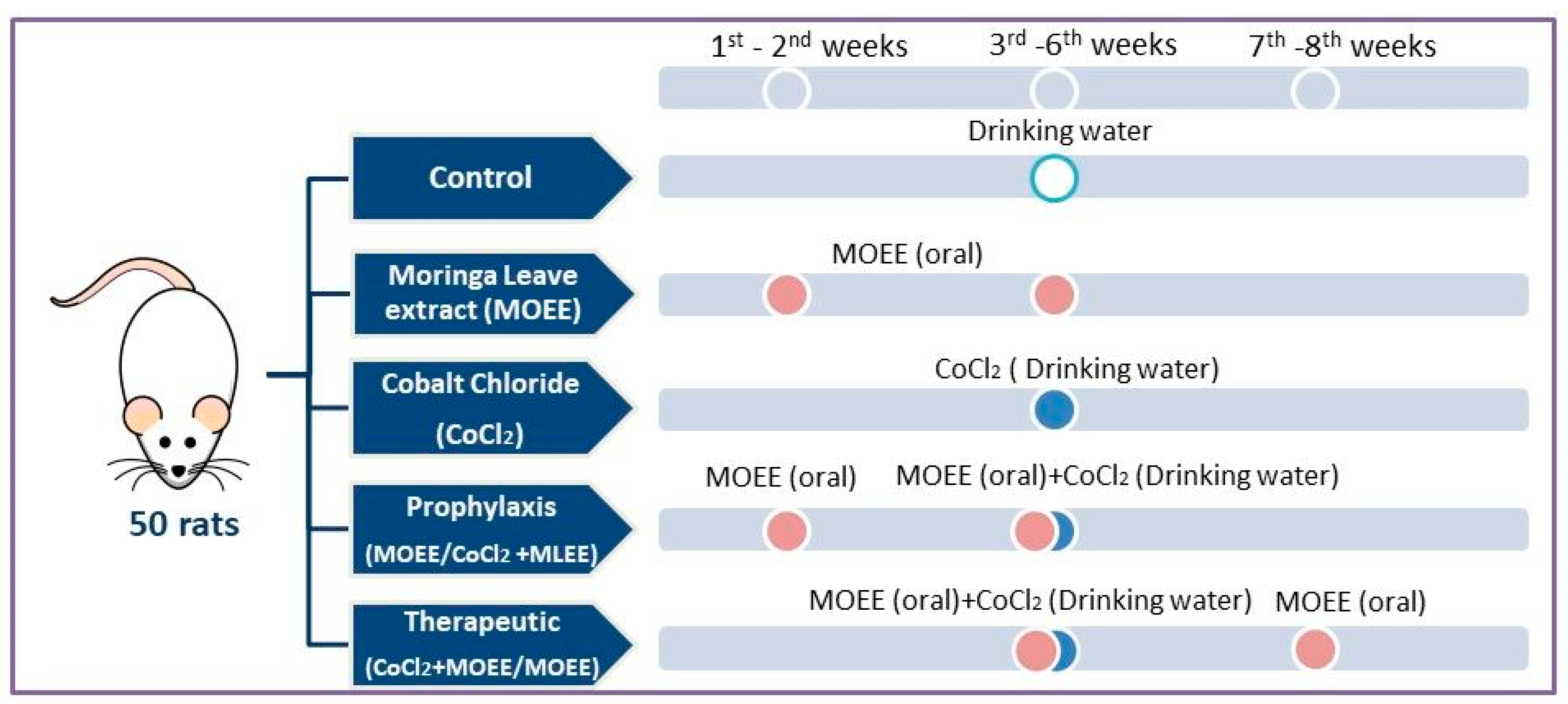
2.5. Experimental Animals and Procedures
2.6. Biochemical Estimation
2.6.1. Renal Injury Markers and Protein Profile
2.6.2. Oxidative Stress/Antioxidant Status Markers
2.6.3. Inflammatory Response Markers
2.7. Real-Time Quantitative PCR (RT-qPCR) Analysis of Genes Encoding Inflammation (Pro-Inflammatory Cytokines)
2.8. Data Analysis
3. Results
3.1. GC-MS Profiling of the MOEE
3.2. Mortalities, Signs, Body Weight Changes, And Relative Kidney Weight
3.3. Renal Injury Markers and Protein Profile
3.4. Oxidative Stress/Antioxidant Status Markers
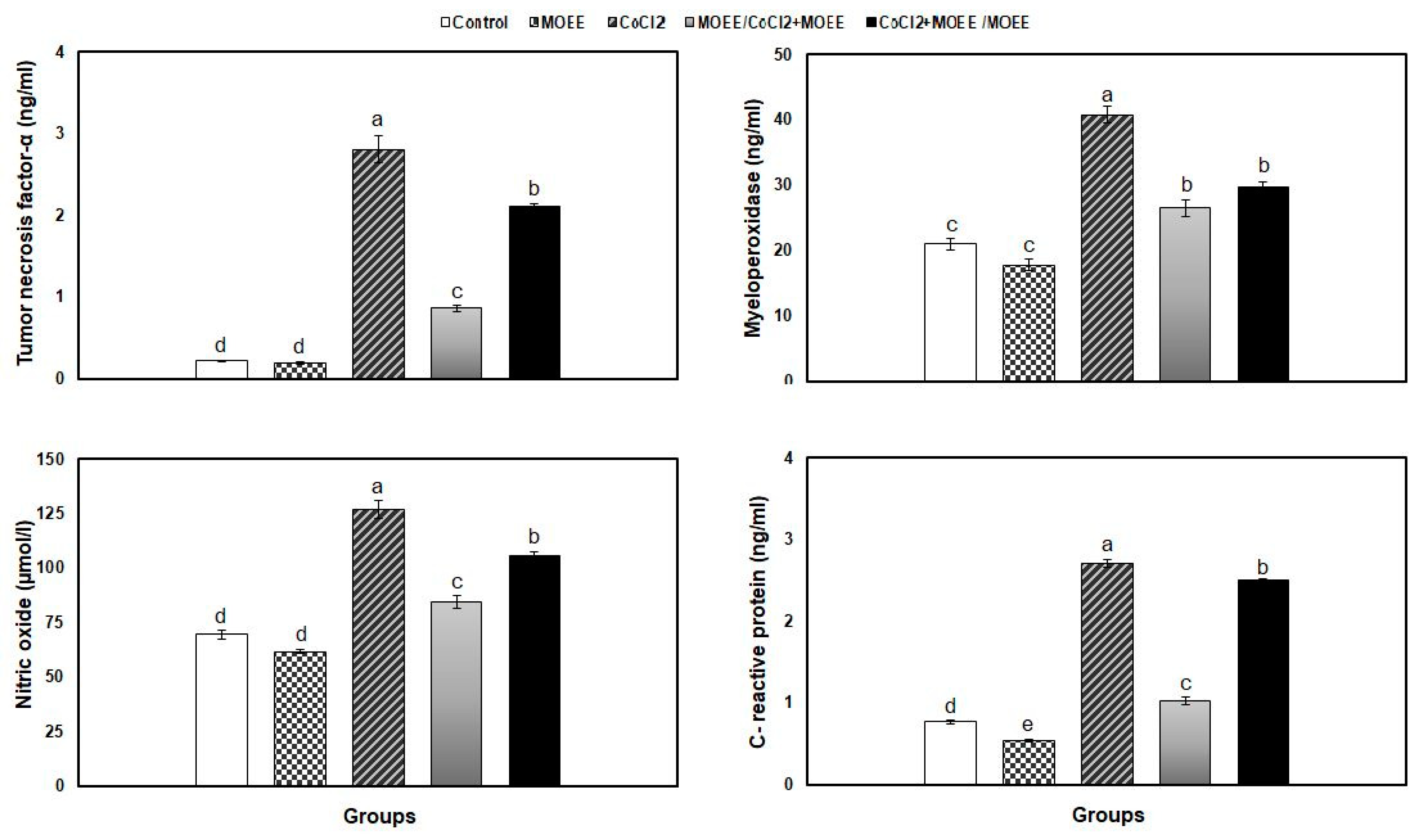
3.5. Inflammatory Response Markers in Kidney Tissue
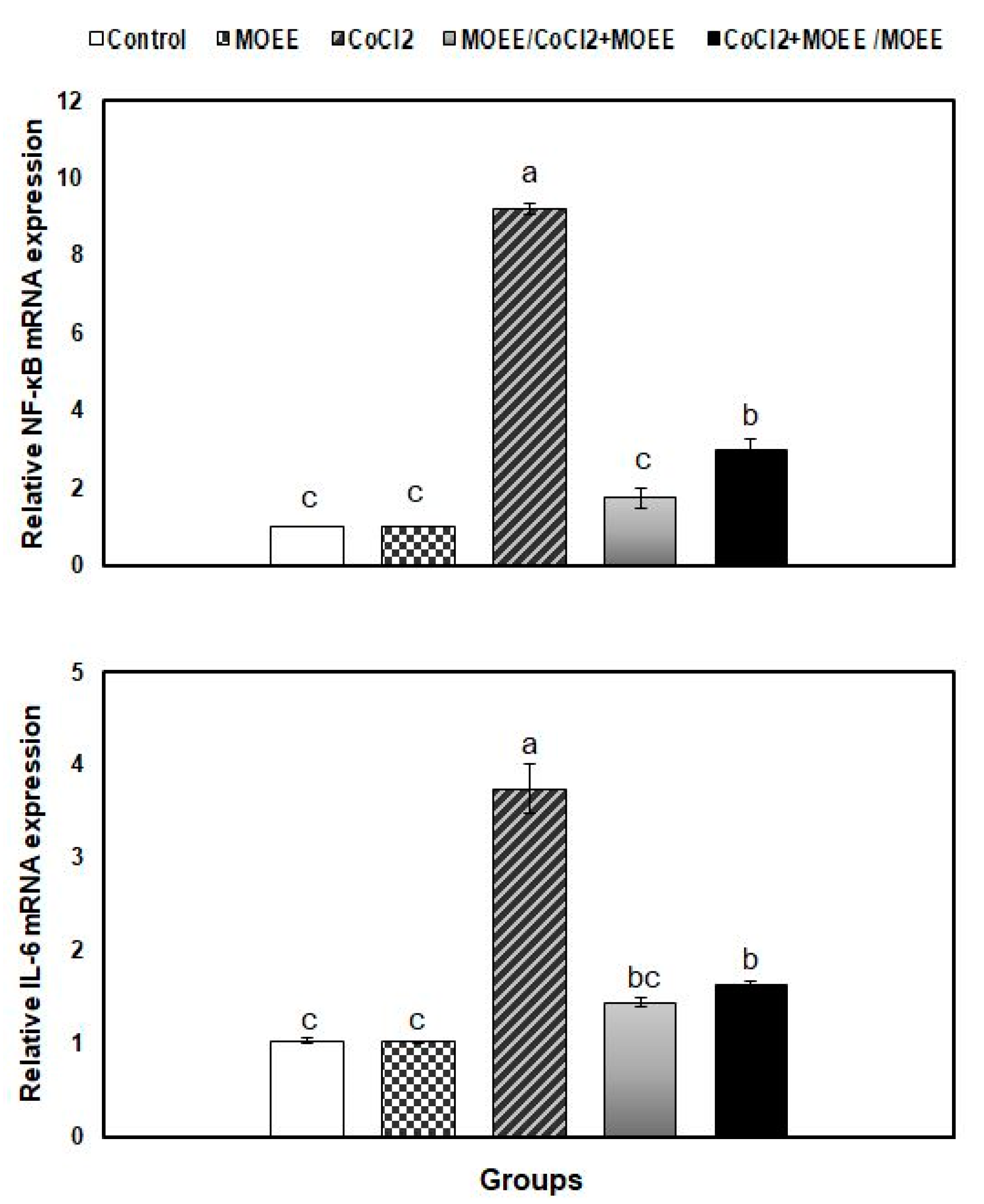
3.6. Relative mRNA Levels of Inflammatory Cytokines in the Kidney Tissue
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef]
- Cheyns, K.; Nkulu, C.B.L.; Ngombe, L.K.; Asosa, J.N.; Haufroid, V.; De Putter, T.; Nawrot, T.; Kimpanga, C.M.; Numbi, O.L.; KabylaIlunga, B.; et al. Pathways of human exposure to cobalt in Katanga, a mining area of the DR Congo. Sci. Total Environ. 2014, 490, 313–321. [Google Scholar] [CrossRef]
- Mobasheri, A.; Proudman, C.J. Cobalt chloride doping in racehorses: Concerns over a potentially lethal practice. Veterinary J. 2015, 205, 335–338. [Google Scholar] [CrossRef][Green Version]
- Koizumi, H.; Takeuchi, Y.; Imai, H.; Kawai, T.; Yoneyama, T. Application of titanium and titanium alloys to fixed dental prostheses. J. Prosthodont. Res. 2019, 63, 266–270. [Google Scholar] [CrossRef]
- Kitagawa, M.; Murakami, S.; Akashi, Y.; Oka, H.; Shintani, T.; Ogawa, I.; Inoue, T.; Kurihara, H. Current status of dental metalallergy in Japan. J. Prosthodont. Res. 2019, 63, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Lin, Y.; Jiang, L.; Lin, H.; Xu, C.; Lin, D.; Cheng, H. Removal of dental alloys and titanium attenuates trace metals and biological effects on liver and kidney. Chemosphere 2020, 243, 125205. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; Cobalt in Hard Metals and Cobalt Sulfate, Gallium Arsenide, Indium Phosphide and Vanadium Pentoxide; World Health Organization: Lyon, France, 2006; Volume 86. [Google Scholar]
- Leyssens, L.; Vinck, B.; Van Der Straeten, C.; Wuyts, F.; Maes, L. Cobalt toxicity in humans—A review of the potential sources and systemic health effects. Toxicology 2017, 387, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Uboldi, C.; Orsière, T.; Darolles, C.; Aloin, V.; Tassistro, V.; George, I.; Malard, V. Poorly soluble cobalt oxide particles trigger genotoxicity via multiple pathways. Part. Fibre Toxicol. 2015, 13, 5. [Google Scholar] [CrossRef]
- Apostoli, P.; Catalani, S.; Zaghini, A.; Mariotti, A.; Poliani, P.L.; Vielmi, V.; Semeraro, F.; Duse, S.; Porzionato, A.; Macchi, V.; et al. High doses of cobalt induce optic and auditory neuropathy. Exp. Toxicol. Pathol. 2013, 65, 719–727. [Google Scholar] [CrossRef]
- Mohamed, A.A.R.; Metwally, M.M.; Khalil, S.R.; Salem, G.A.; Ali, H.A. Moringa oleifera extract attenuates the CoCl2 induced hypoxia of rat’s brain: Expression pattern of HIF-1α, NF-kB, MAO and EPO. Biomed. Pharmacother. 2019, 109, 1688–1697. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, Y.; Hong, H.; Wang, W.; Liu, F. Protective effects of Zn2+ against cobalt nanoparticles and cobalt chloride-induced cytotoxicity of RAW 264.7 cells via ROS pathway. Biochem. Biophys. Res. Commun. 2017, 486, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Daliu, P.; Santini, A.; Novellino, E. A decade of nutraceutical patents: Where are we now in 2018? Expert Opin. Ther. Pat. 2018, 28, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef] [PubMed]
- Khalil, S.R.; Salem, H.F.; Metwally, M.M.; Emad, R.M.; Elbohi, K.M.; Ali, S.A. Protective effect of Spirulina platensis against physiological, ultrastructural and cell proliferation damage induced by furan in kidney and liver of rat. Ecotoxicol. Environ. Saf. 2020, 192, 110256. [Google Scholar] [CrossRef] [PubMed]
- Khalil, S.R.; Mohammed, A.T.; El-fattah, A.H.A.; Zaglool, A.W. Intermediate filament protein expression pattern and inflammatory response changes in kidneys of rats receiving doxorubicin chemotherapy and quercetin. Toxicol. Lett. 2018, 288, 89–98. [Google Scholar] [CrossRef]
- Daghestani, M.H.; Selim, M.E.; Abd-Elhakim, Y.M.; Said, E.N.; El-Hameed, N.E.A.; Khalil, S.R.; El-Tawil, O.S. The role of apitoxin in alleviating propionic acid-induced neurobehavioral impairments in rat pups: The expression pattern of Reelin gene. Biomed. Pharmacother. 2017, 93, 48–56. [Google Scholar] [CrossRef]
- Zakaria, N.; Khalil, S.R.; Awad, A.; Khairy, G.M. Quercetin Reverses Altered Energy Metabolism in the Heart of Rats Receiving Adriamycin Chemotherapy. Cardiovasc. Toxicol. 2018, 18, 109–119. [Google Scholar] [CrossRef]
- Toughan, H.; Khalil, S.R.; El-Ghoneimy, A.A.; Awad, A.; Seddek, A.S. Effect of dietary supplementation with Spirulina platensis on Atrazine-induced oxidative stress-mediated hepatic damage and inflammation in the common carp (Cyprinus carpio L.). Ecotoxicol. Environ. Saf. 2018, 149, 135–142. [Google Scholar] [CrossRef]
- Pandey, A.; Pandey, R.V.; Tripathi, P.; Gupta, P.P.; Haider, J.; Bhatt, S.; Singh, A.V. Moringa oleifera Lam. (Sahijan)—A plant with a plethora of diverse therapeutic benefits: An updated retrospection. Med. Aromat. Plants 2012, 1, 101. [Google Scholar] [CrossRef]
- Stohs, S.J.; Hartman, M.J. Review of the safety and efficacy of Moringa oleifera. Phytother. Res. 2015, 29, 796–804. [Google Scholar] [CrossRef]
- Sharifudin, S.A.; Fakurazi, S.; Hidayat, M.T.; Hairuszah, I.; Aris Mohd Moklas, M.; Arulselvan, P. Therapeutic potential of Moringa oleifera extracts against acetaminophen-induced hepatotoxicity in rats. Pharm. Biol. 2012, 51, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Taha, N.R.; Amin, H.A.; Sultan, A.A. The protective effect of Moringa oleifera leaves against cyclophosphamide-induced urinary bladder toxicity in rats. Tissue Cell 2015, 47, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Abdou, K.H.; Moselhy, W.A.; Mohamed, H.M.; El-Nahass, E.S.; Khalifa, A.G. Moringa oleifera leaves extract protects titanium dioxide nanoparticles-induced nephrotoxicity via Nrf2/HO-1 signaling and amelioration of oxidative stress. Biol. Trace Elem. Res. 2019, 187, 181–191. [Google Scholar] [CrossRef]
- Khalil, S.R.; Abdel-Motal, S.M.; Abd-Elsalam, M.; El-Hameed, N.E.A.; Awad, A. Restoring strategy of ethanolic extract of Moringa oleifera leaves against Tilmicosin-induced cardiac injury in rats: Targeting cell apoptosis-mediated pathways. Gene 2020, 730, 144272. [Google Scholar] [CrossRef]
- Khalil, S.R.; Mohammed, W.A.; Zaglool, A.W.; Elhady, W.M.; Farag, M.R. Inflammatory and oxidative injury is induced in cardiac and pulmonary tissue following fipronil exposure in Japanese quail: mRNA expression of the genes encoding interleukin 6, nuclear factor kappa B, and tumor necrosis factor-alpha. Environ. Poll. 2019, 251, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Lugrin, J.; Rosenblatt-Velin, N.; Parapanov, R.; Liaudet, L. The role of oxidative stress during inflammatory processes. Biol. Chem. 2014, 395, 203–230. [Google Scholar] [CrossRef]
- Scheubert, K.; Hufsky, F.; Böcker, S. Computational mass spectrometry for small molecules. J. Cheminformatics 2013, 5, 12. [Google Scholar] [CrossRef]
- Awoyemi, O.V.; Okotie, U.J.; Oyagbemi, A.A.; Omobowale, T.O.; Asenuga, E.R.; Ola-Davies, O.E.; Ogunpolu, B.S. Cobalt chloride exposure dose-dependently induced hepatotoxicity through enhancement of cyclooxygenase-2 (COX-2)/B-cell associated protein X (BAX) signaling and genotoxicity in Wistar rats. Environ. Toxicol. 2017, 32, 1899–1907. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Sinha, A.K. Colorimetric assay of catalase. Anal. Biochem. 1972, 47, 389–394. [Google Scholar] [CrossRef]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972, 247, 3170–3175. [Google Scholar] [PubMed]
- Beutler, E. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar] [PubMed]
- Khalil, S.R.; Awad, A.; Mohammed, H.H.; Nassan, M.A. Imidacloprid insecticide exposure induces stress and disrupts glucose homeostasis in male rats. Environ. Toxicol. Pharmacol. 2017, 55, 165–174. [Google Scholar] [CrossRef]
- Awad, A.; Khalil, S.R.; Farag, M.R.; Nassan, M.A. Differential susceptibility of kidneys and livers to proliferative processes and transcriptional level of the genes encoding desmin, vimentin, connexin 43, and nestin in rats exposed to furan. Ecotoxicol. Environ. Saf. 2018, 162, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-DDCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, G.G.; Khalil, S.; Hussein, M.M.; Labib, M. The cytotoxic and ultrastrctural perturbations of aluminum exposed Nile catfish with special reference to the mitigating effect of vitamin C. Life Sci. J. 2012, 9, 5198–5210. [Google Scholar]
- Khalil, S.R.; Hussein, M.M. Neurotransmitters and neuronal apoptotic cell death of chronically aluminum intoxicated Nile catfish (Clarias gariepinus) in response to ascorbic acid supplementation. Neurotoxicology 2015, 51, 184–191. [Google Scholar] [CrossRef]
- Khalil, S.R.; Khalifa, H.A.; Abdel-Motal, S.M.; Mohammed, H.H.; Elewa, Y.H.; Mahmoud, H.A. Spirulina platensis attenuates the associated neurobehavioral and inflammatory response impairments in rats exposed to lead acetate. Ecotoxicol. Environ. Saf. 2018, 157, 255–265. [Google Scholar] [CrossRef]
- Khalil, S.R.; Elhady, W.M.; Elewa, Y.H.; El-Hameed, N.E.A.; Ali, S.A. Possible role of Arthrospira platensis in reversing oxidative stress-mediated liver damage in rats exposed to lead. Biomed. Pharmacother. 2018, 97, 1259–1268. [Google Scholar] [CrossRef]
- Khalil, S.R.; Awad, A.; Mohammed, H.H. Behavioral response and gene expression changes in fipronil-administered male Japanese quail (Coturnix japonica). Environ. Poll. 2017, 223, 51–61. [Google Scholar] [CrossRef]
- Hashemipour, H.; Kermanshahi, H.; Golian, A.; Veldkamp, T. Effect of thymol and carvacrol feed supplementation on performance, antioxidant enzyme activities, fatty acid composition, digestive enzyme activities, and immune response in broiler chickens. Poult. Sci. 2013, 92, 2059–2069. [Google Scholar] [CrossRef] [PubMed]
- Vinodini, N.A.; Chatterjee, P.K.; Chatterjee, P.; Chakraborti, S.; Nayanatara, A.K.; Bhat, R.M.; Rashmi, K.S.; Suman, V.B.; Shetty, S.B.; Pai, S.R. Protective role of aqueous leaf extract of Moringa oleifera on blood parameters in cadmium exposed adult Wistar albino rats. Int. J. Curr. Res. Acad. Rev. 2015, 3, 192–199. [Google Scholar]
- Ouédraogo, M.; Lamien-Sanou, A.; Ramdé, N.; Ouédraogo, A.S.; Ouédraogo, M.; Zongo, S.P.; Goumbri, O.; Duez, P.; Guissou, P.I. Protective effect of Moringa oleifera leaves against gentamicin-induced nephrotoxicity in rabbits. Exp. Toxicol. Pathol. 2013, 65, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Karthivashan, G.; Kura, A.U.; Arulselvan, P.; Isa, N.M.; Fakurazi, S. The modulatory effect of Moringa oleifera leaf extract on endogenous antioxidant systems and inflammatory markers in an acetaminophen-induced nephrotoxic mice model. PeerJ 2016, 4, e2127. [Google Scholar] [CrossRef]
- Simonsen, L.O.; Harbak, H.; Bennekou, P. Cobalt metabolism and toxicology—A brief update. Sci. Total Environ. 2012, 432, 210–215. [Google Scholar] [CrossRef]
- Velaga, M.K.; Daughtry, L.K.; Jones, A.C.; Yallapragada, P.R.; Rajanna, S.; Rajanna, B. Attenuation of lead-induced oxidative stress in rat brain, liver, kidney and blood of male Wistar rats by Moringa oleifera seed powder. J. Environ. Pathol. Toxicol. Oncol. 2014, 33, 323–337. [Google Scholar] [CrossRef]
- Gupta, R.; Kannan, G.M.; Sharma, M.; Flora, S.J. Therapeutic effects of Moringa oleifera on arsenic-induced toxicity in rats. Environ. Toxicol. Pharmacol. 2005, 20, 456–464. [Google Scholar] [CrossRef]
- Flora, S.J.S. Nutritional components modify metal absorption, toxicresponse and chelation therapy. J. Nutr. Environ. Med. 2002, 12, 53–67. [Google Scholar] [CrossRef]
- Wang, X.Y.; Yokoi, I.; Liu, J.K.; Mori, A. Cobalt (II) and nickel (II) ions as promoters of free radicals in vivo: Detected directly using electron spin resonance spectrometry in circulating blood in rats. Arch. Biochem. Biophys. 1993, 306, 402–406. [Google Scholar] [CrossRef]
- Zeeshan, M.; Murugadas, A.; Ghaskadbi, S.; Ramaswamy, B.R.; Akbarsha, M.A. Ecotoxicological assessment of cobalt using Hydra model: ROS, oxidative stress, DNA damage, cell cycle arrest, and apoptosis as mechanisms of toxicity. Environ. Poll. 2017, 224, 54–69. [Google Scholar] [CrossRef]
- Yamamoto, K.; Inoue, S.; Yamazaki, A.; Yoshinaga, T.; Kawanishi, S. Site-specific DNA damage induced by cobalt(II) ion and hydrogen peroxide: Role of singlet oxygen. Chem. Res. Toxicol. 1989, 2, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Oyagbemi, A.A.; Omobowale, T.O.; Awoyomi, O.V.; Ajibade, T.O.; Falayi, O.O.; Ogunpolu, B.S.; Okotie, U.J.; Asenuga, E.R.; Adejumobi, O.A.; Hassan, F.O.; et al. Cobalt chloride toxicity elicited hypertension and cardiac complication via induction of oxidative stress and upregulation of COX-2/Bax signaling pathway. Hum. Exp. Toxicol. 2019, 38, 519–532. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Morris, H.; Cronin, M.T. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef]
- Lison, D.; DeBoeck, M.; Verougstraete, V.; Kirsch-Volder, M. Update on the genotoxicity and carcinogenicity of cobalt compounds. Occup. Environ. Med. 2001, 58, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Beyersmann, D.; Hartwig, A. Carcinogenic metal compounds: Recent insights into molecular and cellular mechanisms. Arch. Toxicol. 2008, 82, 493–512. [Google Scholar] [CrossRef] [PubMed]
- Alarifi, S.; Ali, D.; Al Omar Suliman, Y.; Ahamed, M.; Siddiqui, M.A.; Al-Khedhairy, A.A. Oxidative stress contributes to cobalt oxide nanoparticles-induced cytotoxicity and DNA damage in human hepatocarcinoma cells. Int. J. Nanomed. 2013, 8, 189. [Google Scholar]
- Tumer, T.B.; Rojas-Silva, P.; Poulev, A.; Raskin, I.; Waterman, C. Direct and indirect antioxidant activity of polyphenol-and isothiocyanate-enriched fractions from Moringa oleifera. J. Agr. Food Chem. 2015, 63, 1505–1513. [Google Scholar] [CrossRef]
- Sreelatha, S.; Padma, P.R. Modulatory effects of Moringa oleifera extracts against hydrogen peroxide-induced cytotoxicity and oxidative damage. Hum. Exp. Toxicol. 2011, 30, 1359–1368. [Google Scholar] [CrossRef]
- Kim, D.Y.; Won, K.J.; Hwang, D.I.; Park, S.M.; Kim, B.; Lee, H.M. Chemical Composition, Antioxidant and Anti-melanogenic Activities of Essential Oils from Chrysanthemum boreale Makino at Different Harvesting Stages. Chem. Biodivers. 2018, 15, e1700506. [Google Scholar] [CrossRef]
- Sueishi, Y.; Nii, R. Comparative Profiling of Clove Extract and Its Component Antioxidant Activities Against Five Reactive Oxygen Species Using Multiple Free Radical Scavenging. Food Sci. Technol. Res. 2019, 25, 885–890. [Google Scholar] [CrossRef]
- Chniguir, A.; Pintard, C.; Liu, D.; Dang, P.M.C.; El-Benna, J.; Bachoual, R. Eugenol prevents fMLF-induced superoxide anion production in human neutrophils by inhibiting ERK1/2 signaling pathway and p47phox phosphorylation. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Sakat, M.S.; Kilic, K.; Akdemir, F.N.E.; Yildirim, S.; Eser, G.; Kiziltunc, A. The effectiveness of eugenol against cisplatin-induced ototoxicity. Braz. J. Otorhinolaryngol. 2019, 85, 766–773. [Google Scholar] [CrossRef]
- Mesole, S.B.; Alfred, O.O.; Yusuf, U.A.; Lukubi, L.; Ndhlovu, D. Apoptotic Inducement of Neuronal Cells by Aluminium Chloride and the Neuroprotective Effect of Eugenol in Wistar Rats. Oxid. Med. Cell. Longev. 2020, 2020, 8425643. [Google Scholar] [CrossRef] [PubMed]
- Salah, A.; Bouaziz, C.; Amara, I.; Abid-Essefi, S.; Bacha, H. Eugenol protects against citrinin-induced cytotoxicity and oxidative damages in cultured human colorectal HCT116 cells. Environ. Sci. Poll. Res. 2019, 26, 31374–31383. [Google Scholar] [CrossRef] [PubMed]
- Deniz, G.Y. The Protective Effects of Thymol Against Ketoprofen Induced Damages on Pancreatic Acinar and Islet of Langerhans Cells in Rats. J. Essent. Oil Bear. Plants 2019, 22, 604–613. [Google Scholar] [CrossRef]
- Meeran, M.F.N.; Laham, F.; Al-Taee, H.; Azimullah, S.; Ojha, S. Protective effects of α-bisabolol on altered hemodynamics, lipid peroxidation, and nonenzymatic antioxidants in isoproterenol-induced myocardial infarction: In vivo and in vitro evidences. J. Biochem. Mol. Toxicol. 2018, 32, e22200. [Google Scholar] [CrossRef]
- Meeran, M.F.N.; Laham, F.; Azimullah, S.; Tariq, S.; Ojha, S. α-Bisabolol abrogates isoproterenol-induced myocardial infarction by inhibiting mitochondrial dysfunction and intrinsic pathway of apoptosis in rats. Mol. Cell. Biochem. 2019, 453, 89–102. [Google Scholar] [CrossRef]
- Sampaio, T.L.; de Menezes, R.R.P.P.B.; da Costa, M.F.B.; Meneses, G.C.; Arrieta, M.C.V.; Chaves Filho, A.J.M.; de Morais, G.B.; Liborio, A.B.; Alves, R.S.; Evangelista, J.S.A.M.; et al. Nephroprotective effects of (−)-α-bisabolol against ischemic-reperfusion acute kidney injury. Phytomedicine 2016, 23, 1843–1852. [Google Scholar] [CrossRef]
- Sampaio, T.L.; Menezes, R.R.P.P.B.; Lima, D.B.; Silva, R.A.C.; de Azevedo, I.E.P.; Magalhães, E.P.; Marinho, M.M.; Santos, R.P.; Martins, A.M.C. Involvement of NADPH-oxidase enzyme in the nephroprotective effect of (−)-α-bisabolol on HK2 cells exposed to ischemia–Reoxygenation. Eur. J. Pharmacol. 2019, 855, 1–9. [Google Scholar] [CrossRef]
- Tanaka, Y.; Uchi, H.; Furue, M. Antioxidant cinnamaldehyde attenuates UVB-induced photoaging. J. Dermatol. Sci. 2019, 96, 151–158. [Google Scholar] [CrossRef]
- Wang, D.; Hou, J.; Yang, Y.; Zhou, P.; Liu, S.; Wan, J.; Wang, P. Cinnamaldehyde Ameliorates High-Glucose–Induced Oxidative Stress and Cardiomyocyte Injury Through Transient Receptor Potential Ankyrin 1. J. Cardiovasc. Pharmacol. 2019, 74, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Sanz, A.B.; Santamaria, B.; Ruiz-Ortega, M.; Egido, J.; Ortiz, A. Mechanisms of renal apoptosis in health and disease. J. Am. Soc. Nephrol. 2008, 19, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Roth, J.; Oppenheim, J.J.; Tracey, K.J.; Vogl, T.; Feldmann, M.; Horwood, N.; Nanchahal, J. Alarmins: Awaiting a clinical response. J. Clin. Investig. 2012, 122, 2711–2719. [Google Scholar] [CrossRef] [PubMed]
- Gloire, G.; Legrand-Poels, S.; Piette, J. NF-κB activation by reactive oxygen species: Fifteen years later. Biochem. Pharmacol. 2006, 72, 1493–1505. [Google Scholar] [CrossRef]
- Madrigal-Estebas, L.; Doherty, D.G.; O’Donoghue, D.P.; Feighery, C.; O’Farrelly, C. Differential expression and upregulation of interleukin-1a, interleukin-1b and interleukin-6 by freshly isolated human small intestinal epithelial cells. Mediat. Inflamm. 2002, 11, 313–319. [Google Scholar] [CrossRef]
- Pedersen, B.K. Muscles and their myokines. J. Exp. Biol. 2011, 214, 337–346. [Google Scholar] [CrossRef]
- Radi, R. Peroxynitrite, a stealthy biological oxidant. J. Biol. Chem. 2013, 288, 26464–26472. [Google Scholar] [CrossRef]
- Cook, H.T.; Ebrahim, H.; Jansen, A.S.; Foster, G.R.; Largen, P.; Cattell, V. Expression of the gene for inducible nitric oxide synthase in experimental glomerulonephritis in the rat. Clin. Exp. Immunol. 1994, 97, 315–320. [Google Scholar] [CrossRef]
- Arnhold, J.; Osipov, A.N.; Spalteholz, H.; Panasenko, O.M.; Schiller, J. Effects of hypochlorous acid on unsaturated phosphatidylcholines. Free Radic. Biol. Med. 2001, 31, 1111–1119. [Google Scholar] [CrossRef]
- Akinrinde, A.S.; Oyagbemi, A.A.; Omobowale, T.O.; Asenuga, E.R.; Ajibade, T.O. Alterations in blood pressure, antioxidant status and caspase 8 expression in cobalt chloride-induced cardio-renal dysfunction are reversed by Ocimum gratissimum and gallic acid in Wistar rats. J. Trace Elem. Med. Biol. 2016, 36, 27–37. [Google Scholar] [CrossRef]
- Cheng, W.N.; Jeong, C.H.; Seo, H.G.; Han, S.G. Moringa Extract Attenuates Inflammatory Responses and Increases Gene Expression of Casein in Bovine Mammary Epithelial Cells. Animals 2019, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Minaiyan, M.; Asghari, G.; Taheri, D.; Saeidi, M.; Nasr-Esfahani, S. Anti-inflammatory effect of Moringa oleifera Lam. seeds on acetic acid-induced acute colitis in rats. Avicenna J. Phytomed. 2014, 4, 127–136. [Google Scholar] [PubMed]
- Fontinele, L.L.; Heimfarth, L.; Pereira, E.W.M.; Rezende, M.M.; Lima, N.T.; de Carvalho, Y.M.B.G.; Pires, E.A.M.; Guimarães, A.G.; de Carvalho, M.T.B.; Barreto, R.S.S.; et al. Anti-hyperalgesic effect of (-)-α-bisabolol and (-)-α-bisabolol/β-Cyclodextrin complex in a chronic inflammatory pain model is associated with reduced reactive gliosis and cytokine modulation. Neurochem. Int. 2019, 131, 104530. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, H.A.O.; Silva-Filho, S.E.; Wiirzler, L.A.M.; Cardia, G.F.E.; Uchida, N.S.; Silva-Comar, F.M.S.; Bersani-Amado, C.A.; Cuman, R.K.N. Effect of (-)-α-Bisabolol on the Inflammatory Response in Systemic Infection Experimental Model in C57BL/6 Mice. Inflammation 2020, 43(1), 193–203. [Google Scholar] [CrossRef]
- Fernandes, M.Y.D.; do Carmo, M.R.S.; Fonteles, A.A.; de Sousa Neves, J.C.; da Silva, A.T.A.; Pereira, J.F.; Ferreira, E.O.; Lima, N.M.R.; Neves, K.R.T.; de Andrade, G.M. (-)-α-bisabolol prevents neuronal damage and memory deficits through reduction of proinflammatory markers induced by permanent focal cerebral ischemia in mice. Eur. J. Pharmacol. 2019, 842, 270–280. [Google Scholar] [CrossRef]
- Meeran, M.N.; Azimullah, S.; Laham, F.; Tariq, S.; Goyal, S.N.; Adeghate, E.; Ojha, S. α-Bisabolol protects against β-adrenergic agonist-induced myocardial infarction in rats by attenuating inflammation, lysosomal dysfunction, NLRP3 inflammasome activation and modulating autophagic flux. Food Funct. 2020, 11, 965–976. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Hung, S.L.; Pai, S.F.; Lee, Y.H.; Yang, S.F. Eugenol suppressed the expression of lipopolysaccharide-induced proinflammatory mediators in human macrophages. J. Endod. 2007, 33, 698–702. [Google Scholar] [CrossRef]
- Magalhães, C.B.; Riva, D.R.; DePaula, L.J.; Brando-Lima, A.; Koatz, V.L.; Leal-Cardoso, J.H.; Zin, W.A.; Faffe, D.S. In vivo anti-inflammatory action of eugenol on lipopolysaccharide-induced lung injury. J. Appl. Physiol. 2010, 108, 845–851. [Google Scholar] [CrossRef]
- Chang, H.J.; Kim, J.M.; Lee, J.C.; Kim, W.K.; Chun, H.S. Protective effect of βcaryophyllene, a natural bicyclic sesquiterpene, against cerebral ischemic injury. J. Med. Food 2013, 16, 471–480. [Google Scholar] [CrossRef]
- Wan, L.; Meng, D.; Wang, H.; Wan, S.; Jiang, S.; Huang, S.; Wei, L.; Yu, P. Preventive and therapeutic effects of thymol in a lipopolysaccharide-induced acute lung injury mice model. Inflammation 2018, 41, 183–192. [Google Scholar] [CrossRef]
- Wu, H.; Jiang, K.; Yin, N.; Ma, X.; Zhao, G.; Qiu, C.; Deng, G. Thymol mitigates lipopolysaccharide-induced endometritis by regulating the TLR4- and ROS-mediated NF-kappaB signaling pathways. Oncotarget 2017, 8, 20042–20055. [Google Scholar] [PubMed]
- Wang, Q.; Cheng, F.; Xu, Y.; Zhang, J.; Qi, J.; Liu, X.; Wang, R. Thymol alleviates lipopolysaccharide-stimulated inflammatory response via downregulation of RhoA-mediated NF-κB signalling pathway in human peritoneal mesothelial cells. Eur. J. Pharmacol. 2018, 833, 210–220. [Google Scholar] [CrossRef] [PubMed]
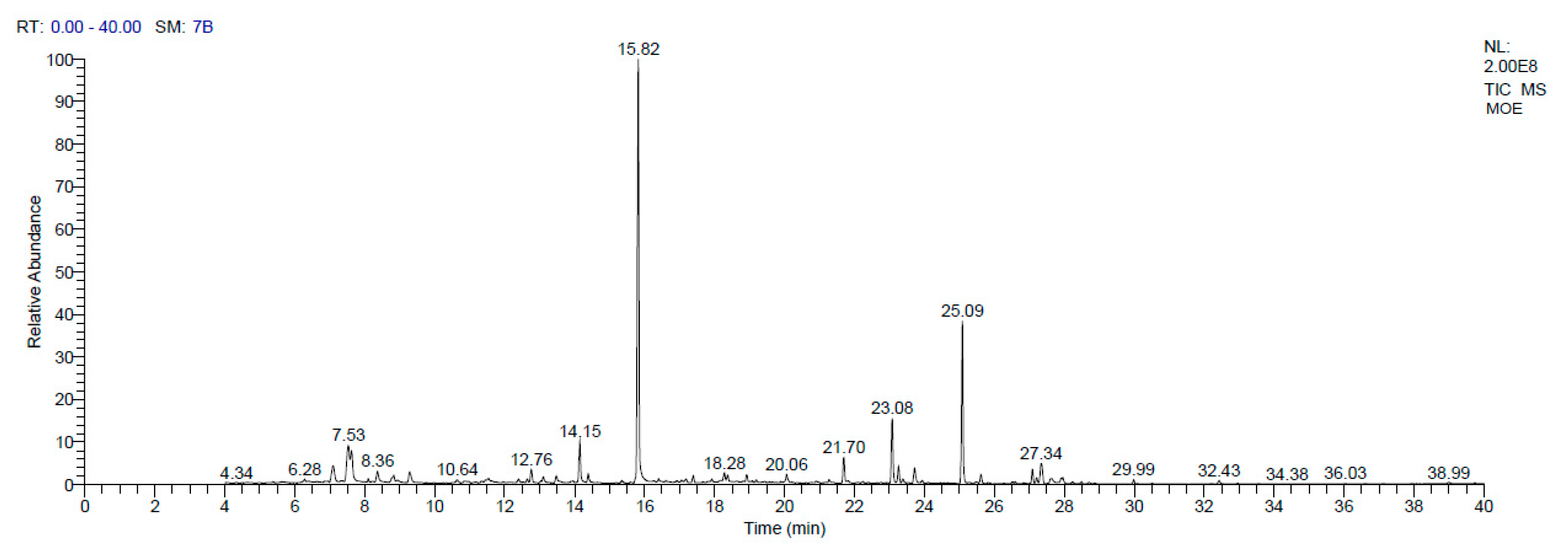
| Bioactive Chemical Constituents | Chemical Formula | Mass Weight (MW) | Matching Factor (MF) | Retention Time (RT) (min) | Area % |
|---|---|---|---|---|---|
| Eugenol: 4-allyl-2-methoxyphenol | C10H12O2 | 164 | 951 | 15.82 | 39.38 |
| α-Bisabolol | C15H26O | 222 | 877 | 25.09 | 15.00 |
| Bisabolol oxide B: 2-Furanmethanol,tetrahydro-à,à,5-trimethyl-5-(4-methyl-3-cyclohexen-1-yl)-, [2S-[2à,5á(R*)]]- | C15H26O2 | 238 | 927 | 23.08 | 6.19 |
| E-dipropylene glycol | C6H14O3 | 134 | 943 | 7.53 | 4.84 |
| Thymol: 2-isopropyl-5-methylphenol | C10H14O | 150 | 918 | 14.15 | 4.74 |
| Menthol: 1’-(butyn-3-one-1-yl)-, (1S,2S,5R)- 4-(1-Hydroxy-2-isopropyl-5-methylcyclohexyl)-3-butyn-2 -one | C15H24 | 204 | 701 | 21.70 | 3.41 |
| 2-(2-Hydroxypropoxy)-1-propanol | C6H14O3 | 134 | 950 | 7.62 | 3.30 |
| 7-Acetyl-6-ethyl-1,1,4,4-tetramethyltetralin | C18H26O | 258 | 930 | 27.34 | 2.78 |
| 2-Propanol,1,1-oxybis- | C18H32O2 | 134 | 892 | 7.10 | 2.67 |
| (-)-α-Bisabolol oxide A: 2H-Pyran-3-ol, tetrahydro-2,2,6-trimethyl-6-(4-methyl-3-cyclohexen-1-yl)-, [3S[3alpha,6alpha (R*)]] | C15H26O2 | 236 | 845 | 23.72 | 1.80 |
| 7a-Isopropenyl-4,5-dimethyloctahydroinden-4-yl)methanol | C15H26O | 222 | 816 | 23.26 | 1.53 |
| 2-phenylethanol | C8H10O | 122 | 921 | 9.29 | 1.39 |
| D- Carvone: 2-Cyclohexen-1-one, 2-methyl-5-(1-methylethenyl)- | C10H14O | 150 | 938 | 12.76 | 1.36 |
| Malonic acid: bis(1-methyl-2-hydroxyethyl)ether | C3H4O4 | 104 | 999 | 8.36 | 1.28 |
| Neophytadiene: 1,3-butadiene, 2-(4,8,12-trimethyltridecyl)- | C20H38 | 278 | 917 | 27.09 | 1.23 |
| (E)-Cinnamaldehyde: trans-3-phenyl-2-propenal | C9H8O | 132 | 871 | 13.47 | 1.04 |
| 1H-3a,7-Methanoazulene,2,3,6,7,8,8a-hexahydro-1,4,9,9-tetramethyl-(1à,3aà,7à,8aá)- | C15H24 | 204 | 868 | 18.28 | 0.97 |
| Phthalic acid: 1,2-Benzenedicarboxylic acid, diethyl ester | C12H14O4 | 222 | 942 | 20.06 | 0.96 |
| 1-Chloro-7-heptadecyne | C17H31Cl | 270 | 743 | 27.96 | 0.90 |
| 1-chlorotetradecane | C14H29Cl | 232 | 733 | 8.83 | 0.87 |
| Acetyl cedrene, Ethanone | C17H26O | 246 | 940 | 25.62 | 0.82 |
| Nerol: (Z)-3,7-dimethyl-2,6-octadien-1-ol | C10H18O | 154 | 907 | 13.11 | 0.80 |
| Methyl pimar-8-en-18-oate | C21H34O2 | 318 | 623 | 27.62 | 0.76 |
| Caryophyllene: Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene-, [1R-(1R*,4E,9S*)]- | C15H24 | 204 | 888 | 17.39 | 0.68 |
| Alloaromadendrene: (1aS,4aR,7S,7aS,7bR)-1,1,7-Trimethyl-4-methylendecahydro-1H-cyclopropa[e]azulen | C15H24 | 204 | 907 | 18.37 | 0.65 |
| α-Isomethyl ionone: 4-(2,6,6-Trimethyl 2-cyclohexen-1-yl)-3-methyl-3-buten-2-one | C14H22O | 206 | 890 | 18.92 | 0.64 |
| 2-Pentadecanone, 6,10,14-trimethyl- | C18H36O | 268 | 876 | 27.21 | 0.43 |
| Parameters | Experimental Groups | ||||
|---|---|---|---|---|---|
| Control | MOEE | CoCl2 | MOEE/CoCl2+MOEE | CoCl2+MOEE /MOEE | |
| Body weight change (gm) | 12.00 b ± 1.09 | 18.16 a ± 1.44 | −11.00 d ± 0.73 | −5.00 c ± 2.67 | −8.00 cd ± 0.57 |
| Relative kidney weight | 0.25 ± 0.01 | 0.26 ± 0.01 | 0.27 ± 0.02 | 0.26 ± 0.02 | 0.28 ± 0.02 |
| Kidney tissue injury markers | |||||
| Urea (mg/dL) | 21.18 d ± 0.80 | 20.54 d ± 0.61 | 43.48 a ± 1.69 | 26.65 c ± 1.15 | 37.51 b ± 1.56 |
| Creatinine (mg/dL) | 0.83 d ± 0.02 | 0.84 d ± 0.04 | 1.97 a ± 0.06 | 1.09 c ± 0.05 | 1.49 b ± 0.06 |
| Protein profile | |||||
| Total protein (gm/dL) | 6.47 a ± 0.25 | 6.43 a ± 0.29 | 3.97 c ± 0.29 | 5.39 b ± 0.21 | 4.20 c ± 0.31 |
| Albumin (gm/dL) | 3.97 a ± 0.23 | 4.09 a ± 0.23 | 2.34 c ± 0.16 | 3.24 b ± 0.08 | 2.56 bc ± 0.22 |
| Globulin (gm/dL) | 2.49 a ± 0.09 | 2.35 a ± 0.13 | 1.63 b ± 0.15 | 2.15 ab ± 0.18 | 1.64 b ± 0.12 |
| A/G ratio | 1.60 ± 0.09 | 1.75 ± 0.11 | 1.46 ± 0.07 | 1.56 ± 0.14 | 1.56 ± 0.12 |
| Parameters | Experimental Groups | ||||
|---|---|---|---|---|---|
| Control | MOEE | CoCl2 | MOEE/CoCl2+MOEE | CoCl2 + MOEE /MOEE | |
| Antioxidants biomarkers | |||||
| SOD activity (U/gm tissue) | 3.77 a ± 0.31 | 3.82 a ± 0.23 | 1.63 c ± 0.19 | 2.48 b ± 0.12 | 1.81b c ± 0.20 |
| CAT activity (U/gm tissue) | 1.14 a ± 0.03 | 1.21 a ± 0.05 | 0.54 d ± 0.04 | 0.97 b ± 0.01 | 0.73 c ± 0.05 |
| GSH level (mmol/gm tissue) | 0.56 a ± 0.03 | 0.55 a ± 0.03 | 0.21 d ± 0.02 | 0.42 b ± 0.02 | 0.32 c ± 0.01 |
| Oxidative injury biomarkers | |||||
| H2O2 (ng/mL) | 1.12 d ± 0.02 | 1.06 d ± 0.03 | 2.77 a ± 0.03 | 1.66 c ± 0.04 | 2.22 b ± 0.05 |
| MDA (nmoL/gm tissue) | 9.97 c ± 0.12 | 9.49 c ± 0.38 | 25.06 a ± 2.23 | 15.18 b ± 1.12 | 21.04 a ± 0.70 |
| 8OHdG (ng/mL) | 0.09 d ± 0.01 | 0.07 d ± 0.01 | 0.37 a ± 0.02 | 0.16 c ± 0.01 | 0.31 b ± 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Daim, M.M.; Khalil, S.R.; Awad, A.; Abu Zeid, E.H.; El-Aziz, R.A.; El-Serehy, H.A. Ethanolic Extract of Moringa oleifera Leaves Influences NF-κB Signaling Pathway to Restore Kidney Tissue from Cobalt-Mediated Oxidative Injury and Inflammation in Rats. Nutrients 2020, 12, 1031. https://doi.org/10.3390/nu12041031
Abdel-Daim MM, Khalil SR, Awad A, Abu Zeid EH, El-Aziz RA, El-Serehy HA. Ethanolic Extract of Moringa oleifera Leaves Influences NF-κB Signaling Pathway to Restore Kidney Tissue from Cobalt-Mediated Oxidative Injury and Inflammation in Rats. Nutrients. 2020; 12(4):1031. https://doi.org/10.3390/nu12041031
Chicago/Turabian StyleAbdel-Daim, Mohamed M., Samah R. Khalil, Ashraf Awad, Ehsan H. Abu Zeid, Reda Abd El-Aziz, and Hamed A. El-Serehy. 2020. "Ethanolic Extract of Moringa oleifera Leaves Influences NF-κB Signaling Pathway to Restore Kidney Tissue from Cobalt-Mediated Oxidative Injury and Inflammation in Rats" Nutrients 12, no. 4: 1031. https://doi.org/10.3390/nu12041031
APA StyleAbdel-Daim, M. M., Khalil, S. R., Awad, A., Abu Zeid, E. H., El-Aziz, R. A., & El-Serehy, H. A. (2020). Ethanolic Extract of Moringa oleifera Leaves Influences NF-κB Signaling Pathway to Restore Kidney Tissue from Cobalt-Mediated Oxidative Injury and Inflammation in Rats. Nutrients, 12(4), 1031. https://doi.org/10.3390/nu12041031






