Risk Factors for Anemia and Micronutrient Deficiencies among Women of Reproductive Age—The Impact of the Wheat Flour Fortification Program in Uzbekistan
Abstract
1. Introduction
2. Materials and Methods
2.1. Survey Design and Participants
2.2. Data Collection and Laboratory Analysis
2.3. Parameters and Clinical Thresholds
2.4. Data Management and Statistical Analysis
2.5. Ethics and Consent
3. Results
3.1. Household and Demographic Characteristics of Respondents
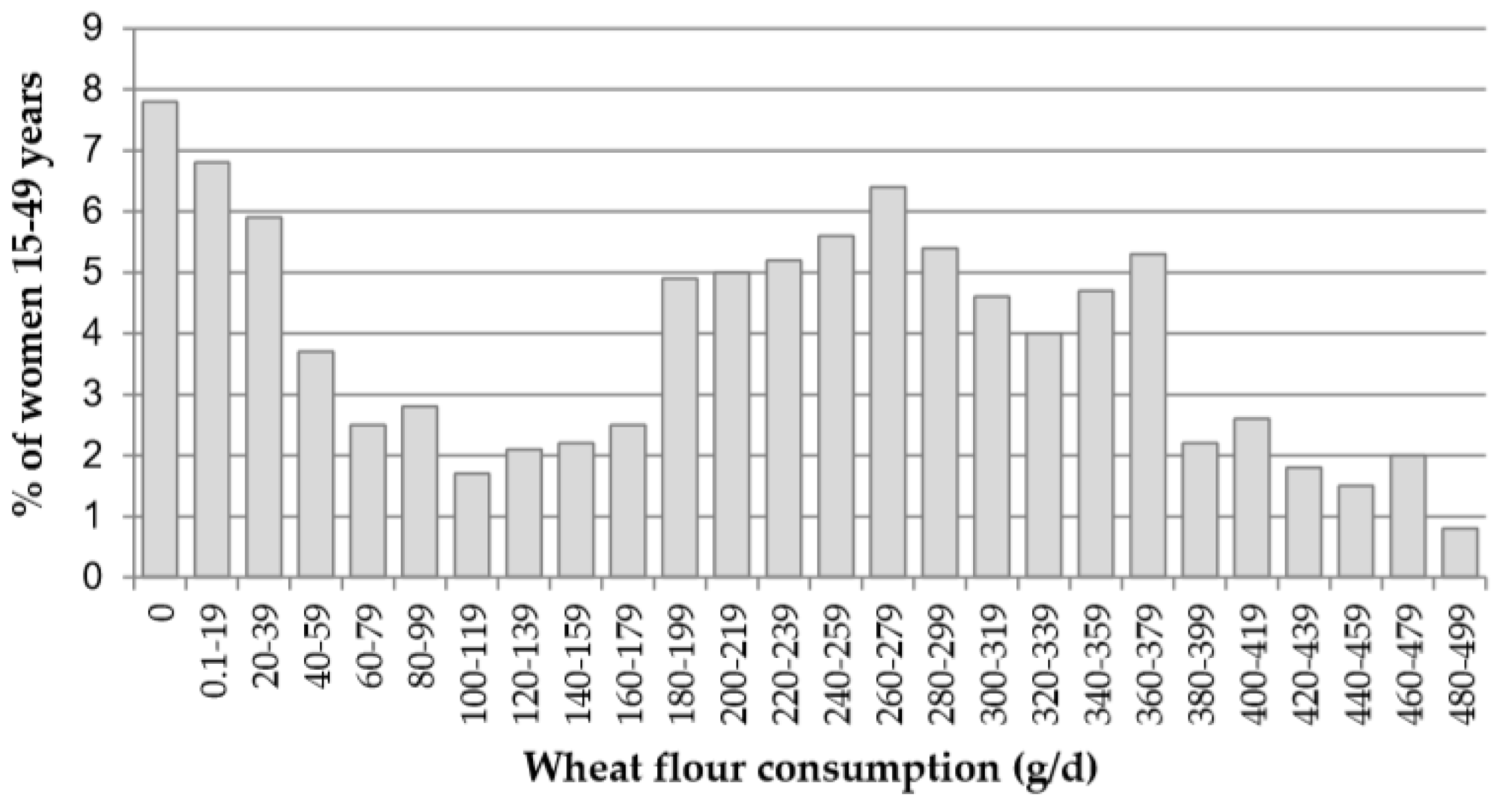
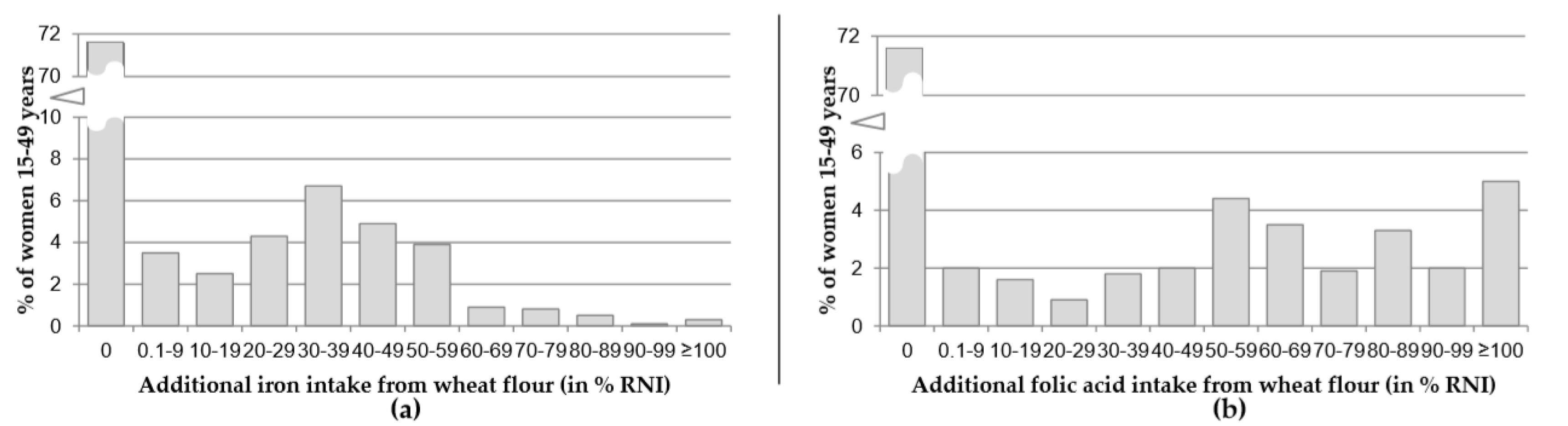
3.2. Flour Consumption and Intake of Iron and Folate from Fortified Flour
3.3. Risk Factors for Anemia, Iron and Folate Deficiencies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Global Burden of Disease Study Collaborators 2013; Vos, T.; Allen, C.; Arora, M.; Barber, R.M.; Bhutta, Z.A.; Brown, A.; Liang, X.; Kawashima, T.; Coggeshall, M.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 743–800. [Google Scholar] [CrossRef]
- Rogers, L.M.; Cordero, A.M.; Pfeiffer, C.M.; Hausman, D.B.; Tsang, B.L.; De-Regil, L.M.; Rosenthal, J.; Razzaghi, H.; Wong, E.C.; Bailey, L.B.; et al. Global folate status in women of reproductive age: A systematic review with emphasis on methodological issues. Ann. N. Y. Acad. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Peña-Rosas, J.P.; Fernández-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Keats, E.; Haider, B.; Tam, E.; Bhutta, Z. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Van der Spoel, E.; Rozing, M.P.; Houwing-Duistermaat, J.J.; Slagboom, P.E.; Beekman, M.; de Craen, A.J.; Westendorp, R.; van Heemst, D. Optimal Serum and Red Blood Cell Folate Conentration in Women of Reproductive Age for Prevention of Neural Tube Defects; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar] [CrossRef]
- Wald, N.J.; Morris, J.K.; Blakemore, C. Public health failure in the prevention of neural tube defects: Time to abandon the tolerable upper intake level of folate. Public Health Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Keats, E.C.; Neufeld, L.M.; Garrett, G.S.; Mbuya, M.N.N.; Bhutta, Z.A. Improved micronutrient status and health outcomes in low-and middle-income countries following large-scale fortification: Evidence from a systematic review and meta-Analysis. Am. J. Clin. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kancherla, V.; Wagh, K.; Johnson, Q.; Oakley, G.P. A 2017 global update on folic acid-preventable spina bifida and anencephaly. Birth Defects Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.; Ranum, P.; de Pee, S.; Biebinger, R.; Hulthen, L.; Johnson, Q.; Lynch, S. Revised recommendations for iron fortification of wheat flour and an evaluation of the expected impact of current national wheat flour fortification programs. Food Nutr. Bull. 2010, 31, S7–S21. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.; Bothwell, T.; Cook, J.D.; Dary, O.; Davidsson, L.; Fairweather-Tait, S.; Hallberg, L.; Lynch, S.; Rosado, J.; Whittaker, P.; et al. The usefulness of elemental iron for cereal flour fortification: A SUSTAIN Task Force report. Sharing United States Technology to Aid in the Improvement of Nutrition. Nutr. Rev. 2002, 60, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Babitt, J.L. Hepcidin regulation in the anemia of inflammation. Curr. Opin. Hematol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health Uzbekistan, UNICEF, GroundWork. Uzbekistan Nutrition Survey 2017. 2019. Available online: http://groundworkhealth.org/wp-content/uploads/2019/11/Uzbekistan_Nutrition_Survey_en_web.pdf (accessed on 10 January 2020).
- Hund, L.; Northrop-Clewes, C.A.; Nazario, R.; Suleymanova, D.; Mirzoyan, L.; Irisova, M.; Pagano, M.; Valadez, J.J. A novel approach to evaluating the iron and folate status of women of reproductive age in Uzbekistan after 3 years of flour fortification with micronutrients. PLoS ONE 2013. [Google Scholar] [CrossRef] [PubMed]
- Vitamin-Mineral Mixture for the Enrichment of Wheat Flour for Baking—O’z DST 1098:2005; State Standard of the Republic of Uzbekistan: Tashkent, Uzbekistan, 2005.
- Vitamin-Mineral Mixture for the Enrichment of Wheat Flour Baking—O’z DSt 3221:2017; State Standard of the Republic of Uzbekistan: Tashkent, Uzbekistan, 2017.
- World Health Organization. WHO STEPS Surveillance Manual: The WHO STEPwise Approach to Non-Communicable Disease Risk Factor Surveillance; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Vitamin and Mineral Nutrition Information System (WHO/NMH/NHD/MNM/11.1); World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Namaste, S.M.; Rohner, F.; Huang, J.; Bhushan, N.L.; Flores-Ayala, R.; Kupka, R.; Northrop-Clewes, C.A.; Mei, Z.; Rawat, R.; Raiten, D.J.; et al. Approaches to understand and adjust ferritin concentrations in the presence of inflammation in preschool-aged children and women of reproductive age: A multi-country analysis. Am. J. Clin. Nutr. 2017, 106, 359S–371S. [Google Scholar] [PubMed]
- World Health Organization/Centers for Disease Control and Prevention. Assessing the Iron Status of a Population, 2nd ed.; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- World Health Organization. Serum Retinol Concentrations for Determining the Prevalence of Vitamin A Deficiency in populations. In Vitamin and Mineral Nutrition Information System; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Tanumihardjo, S.A. Biomarkers of vitamin A status: What do they mean? World Health Organization: Report. Priorities in Assessment of Vitamin A and Iron Status in Populations. Biomarkers 2012, 2, 2. [Google Scholar]
- Larson, L.M.; Namaste, S.M.; Williams, A.M.; Engle-Stone, R.; Addo, O.Y.; Suchdev, P.S.; Wirth, J.P.; Temple, V.; Serdula, M.; Northrop-Clewes, C.A. Adjusting retinol-binding protein concentrations for inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) project. Am. J. Clin. Nutr. 2017, 106, 390S–401S. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.; DeBenoist, B.; Dary, O.; Hurrell, R. Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Thurnham, D.I.; McCabe, G.P.; Northrop-Clewes, C.A.; Nestel, P. Effects of subclinical infection on plasma retinol concentrations and assessment of prevalence of vitamin a deficiency: Meta-analysis. Lancet 2003. [Google Scholar] [CrossRef]
- Shetty, P.S.; James, W.P.T. Body Mass Index: A Measure of Chronic Energy Deficiency in Adults; Food and Agriculture Organization: Rome, Italy, 1994. [Google Scholar]
- Filmer, D.; Pritchett, L.H. Estimating Wealth Effects Without Expenditure Data—Or Tears: An Application To Educational Enrollments In States Of India. Demography 2001, 38, 115–132. [Google Scholar] [PubMed]
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Policy Plan. 2006. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Human Energy Requirements: Report of a Joint FAO/WHO/UNU Expert Consultation; Food and Agriculture Organization: Rome, Italy, 2001. [Google Scholar]
- Weisell, R.; Dop, M. The Adult Male Equivalent concept and its application to Household Consumption and Expenditures Surveys (HCES). Food Nutr. Bull. 2012. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization; World Health Organization. Vitamin and Mineral Requirements in Human Nutrition, 2nd ed.; World Health Organization: Geneva, Switzerland, 2004; pp. 1–20. [Google Scholar]
- World Health Organization/UNICEF Joint Monitoring Programme for Water Supply Sanitation and Hygiene (JMP). In JMP Methodology—2017 Update & SDG Baselines; World Health Organization: Geneva, Switzerland, 2017.
- Food and Agriculture Organization; U.S. Agency for International Development-Food and Nutrition Technical Assistance. Minimum Dietary Diversity for Women—A Guide to Measurement; Food and Agriculture Organization: Rome, Italy, 2018. [Google Scholar]
- Chen, M.Y.; Rose, C.E.; Qi, Y.P.; Williams, J.L.; Yeung, L.F.; Berry, R.J.; Hao, L.; Cannon, M.J.; Crider, K.S. Defining the plasma folate concentration associated with the red blood cell folate concentration threshold for optimal neural tube defects prevention: A population-based, randomized trial of folic acid supplementation. Am. J. Clin. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Serum and Red Blood Cell Folate Concentrations for Assessing Folate Status in Populations; World Health Organization: Geneva, Switzerland, 2015; Available online: https://apps.who.int/iris/bitstream/handle/10665/75584/WHO_NMH_NHD_EPG_12.1_eng.pdf;sequence=1 (accessed on 10 January 2020).
- Kassebaum, N.J.; Jasrasaria, R.; Naghavi, M.; Wulf, S.K.; Johns, N.; Lozano, R.; Regan, M.; Chou, D.P.; Pullan, R.L.; Flaxman, S.R.; et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood 2014. [Google Scholar] [CrossRef]
| Variable | Number with Characteristic (N) | % or Mean a | (95% CI) b |
|---|---|---|---|
| Household characteristics | |||
| Household size, mean | 1806 | 6.0 | (5.8, 6.2) |
| Urban residence, % | 505 | 28.2% | (23.9, 33.0) |
| Male headed household, % | 1452 | 79.3% | (76.4, 81.9) |
| Safe drinking water c, % | 1802 | 99.9% | (98.9, 100.0) |
| Improved sanitary sanitation c, % | 1600 | 89.2% | (86.9, 91.0) |
| Fortified wheat flour d, % | 517 | 29.2% | (26.1, 32.5) |
| Non-pregnant women (NPW) characteristics | |||
| Age (years), mean | 1806 | 31.2 | (30.8, 31.6) |
| Higher education e, % | 1041 | 58.4% | (55.5, 61.2) |
| Currently married, % | 1329 | 73.1% | (70.8, 75.2) |
| Currently breastfeeding, % | 341 | 19.1% | (17.1, 21.3) |
| Given birth in past 2 years, % | 872 | 49.5% | (47.0, 52.1) |
| Number of prior pregnancies, mean | 1806 | 2.3 | (2.2, 2.4) |
| Meeting minimum dietary diversity f, % | 716 | 40.6% | (37.7, 43.5) |
| Consumption of predominant type of flour (g/d), mean | 1806 | 210 | (199, 221) |
| Characteristic | Category | Unadjusted (Bivariate Analysis) | Adjusted (Poisson Regression) | ||
|---|---|---|---|---|---|
| Relative Risk | 95% CI | Relative Risk | 95% CI | ||
| Anemia a (N = 1473) | |||||
| Iron intake from fortified flour (as %RNI) | ≥40% | 1.30 | (0.96, 1.70) | 1.20 | (0.91, 1.50) |
| 0.1–39.9% | 1.10 | (0.87, 1.43) | 1.20 | (0.93, 1.40) | |
| 0% | referent | - | referent | - | |
| Iron deficiency b (ID) | Yes | 4.24 | (3.15, 5.70) | 4.70 | (3.50, 6.50) |
| No | referent | - | referent | - | |
| Vitamin A insufficiency c (VAI) | Yes | 1.89 | (1.52, 2.34) | 1.50 | (1.30, 1.90) |
| No | referent | - | referent | - | |
| ID (N = 1473) | |||||
| Iron intake from fortified flour (as %RNI) | ≥40% | 0.97 | (0.81, 1.20) | 1.00 | (0.86, 1.20) |
| 0.1–39.9% | 0.90 | (0.76, 1.06) | 0.91 | (0.78, 1.05) | |
| 0% | referent | - | referent | - | |
| Educational level d | Secondary or less | 0.86 | (0.77, 0.95) | 0.85 | (0.77, 0.94) |
| Special secondary or more | referent | - | referent | - | |
| Currently breastfeeding | Yes | 1.15 | (1.02, 1.30) | 1.10 | (0.99, 1.20) |
| No | referent | - | referent | - | |
| Underweight (BMI < 18.5) | Yes | 0.81 | (0.62, 1.05) | 0.74 | (0.58, 0.96) |
| No | referent | - | referent | - | |
| VAI | Yes | 1.45 | (1.30, 1.61) | 1.40 | (1.30, 1.60) |
| No | referent | - | referent | - | |
| Any inflammation e | Yes | 0.88 | (0.75, 1.03) | 0.86 | (0.74, 1.01) |
| No | referent | - | referent | - | |
| FD f (N = 1550) | |||||
| Folic acid from fortified flour (as %RNI) | ≥70% | 0.62 | (0.49, 0.79) | 0.67 | (0.53, 0.85) |
| 0.1–69.9% | 0.70 | (0.58, 0.86) | 0.74 | (0.62, 0.88) | |
| 0% | referent | - | referent | - | |
| Currently breastfeeding | Yes | 1.27 | (1.13, 1.43) | 1.20 | (1.10, 1.40) |
| No | referent | - | referent | - | |
| Any inflammation | Yes | 1.23 | (1.07, 1.42) | 1.20 | (1.03, 1.30) |
| No | referent | - | referent | - | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petry, N.; Nizamov, F.; Woodruff, B.A.; Ishmakova, R.; Komilov, J.; Wegmüller, R.; Wirth, J.P.; Arifdjanova, D.; Guo, S.; Rohner, F. Risk Factors for Anemia and Micronutrient Deficiencies among Women of Reproductive Age—The Impact of the Wheat Flour Fortification Program in Uzbekistan. Nutrients 2020, 12, 714. https://doi.org/10.3390/nu12030714
Petry N, Nizamov F, Woodruff BA, Ishmakova R, Komilov J, Wegmüller R, Wirth JP, Arifdjanova D, Guo S, Rohner F. Risk Factors for Anemia and Micronutrient Deficiencies among Women of Reproductive Age—The Impact of the Wheat Flour Fortification Program in Uzbekistan. Nutrients. 2020; 12(3):714. https://doi.org/10.3390/nu12030714
Chicago/Turabian StylePetry, Nicolai, Fakhriddin Nizamov, Bradley A. Woodruff, Regina Ishmakova, Jasur Komilov, Rita Wegmüller, James P. Wirth, Diyora Arifdjanova, Sufang Guo, and Fabian Rohner. 2020. "Risk Factors for Anemia and Micronutrient Deficiencies among Women of Reproductive Age—The Impact of the Wheat Flour Fortification Program in Uzbekistan" Nutrients 12, no. 3: 714. https://doi.org/10.3390/nu12030714
APA StylePetry, N., Nizamov, F., Woodruff, B. A., Ishmakova, R., Komilov, J., Wegmüller, R., Wirth, J. P., Arifdjanova, D., Guo, S., & Rohner, F. (2020). Risk Factors for Anemia and Micronutrient Deficiencies among Women of Reproductive Age—The Impact of the Wheat Flour Fortification Program in Uzbekistan. Nutrients, 12(3), 714. https://doi.org/10.3390/nu12030714





