Differential Impact of Calcium and Vitamin D on Body Composition Changes in Post-Menopausal Women Following a Restricted Energy Diet and Exercise Program
Abstract
1. Introduction
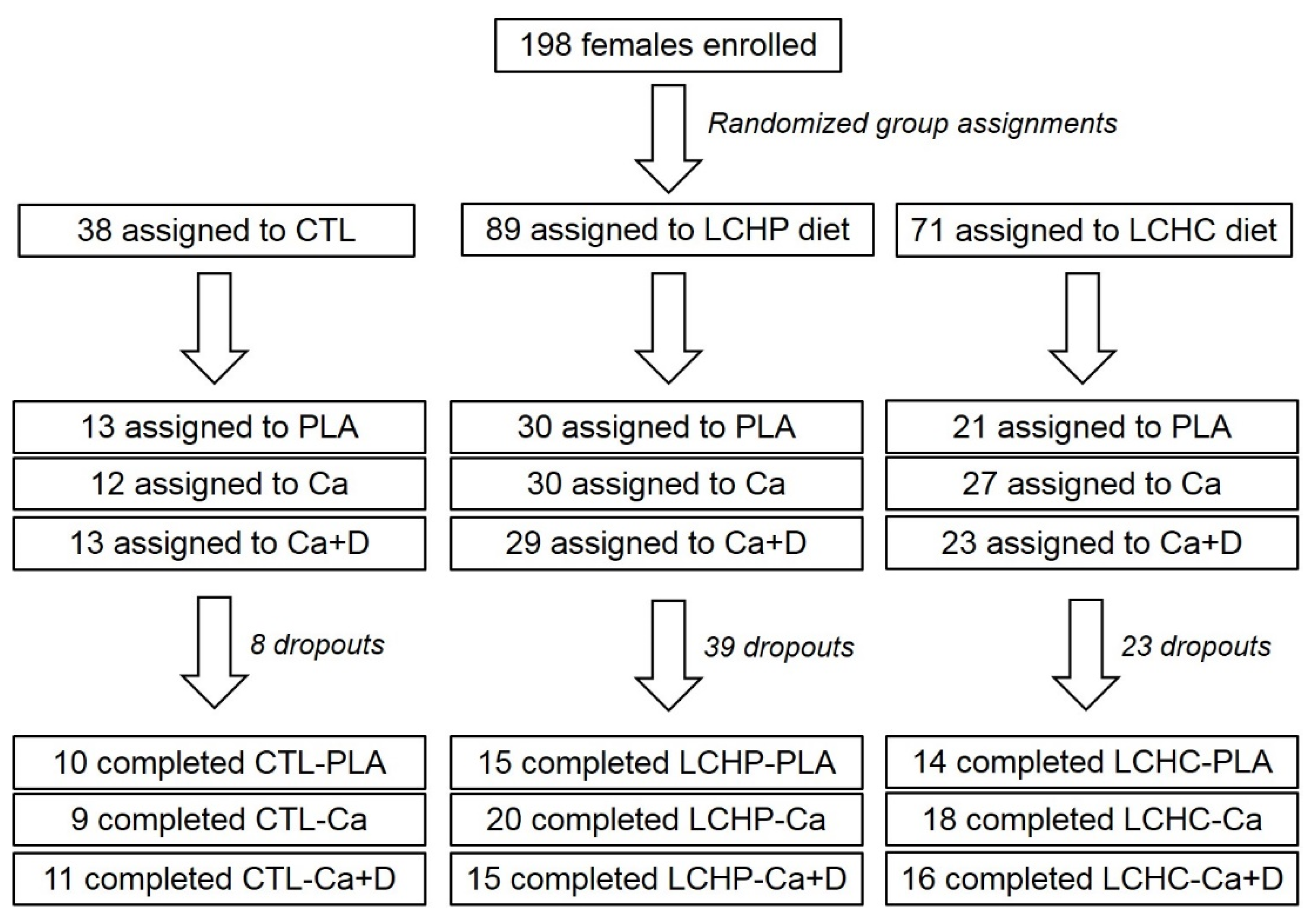
2. Methods
2.1. Experimental Design
2.2. Participants
2.3. Exercise Intervention
2.4. Diet Intervention
2.5. Supplementation Protocol
3. Procedures
3.1. Dietary Assessment
3.2. Resting Energy Expenditure and Metabolism
3.3. Body Weight and Composition
3.4. Resting Hemodynamics and Exercise Capacity
3.5. Muscular Strength and Endurance
3.6. Blood Collection and Analysis
3.7. Statistical Analysis
4. Results
4.1. Baseline Characteristics
4.2. Energy and Macronutrient Intake
4.3. Resting Energy Expenditure and Metabolism
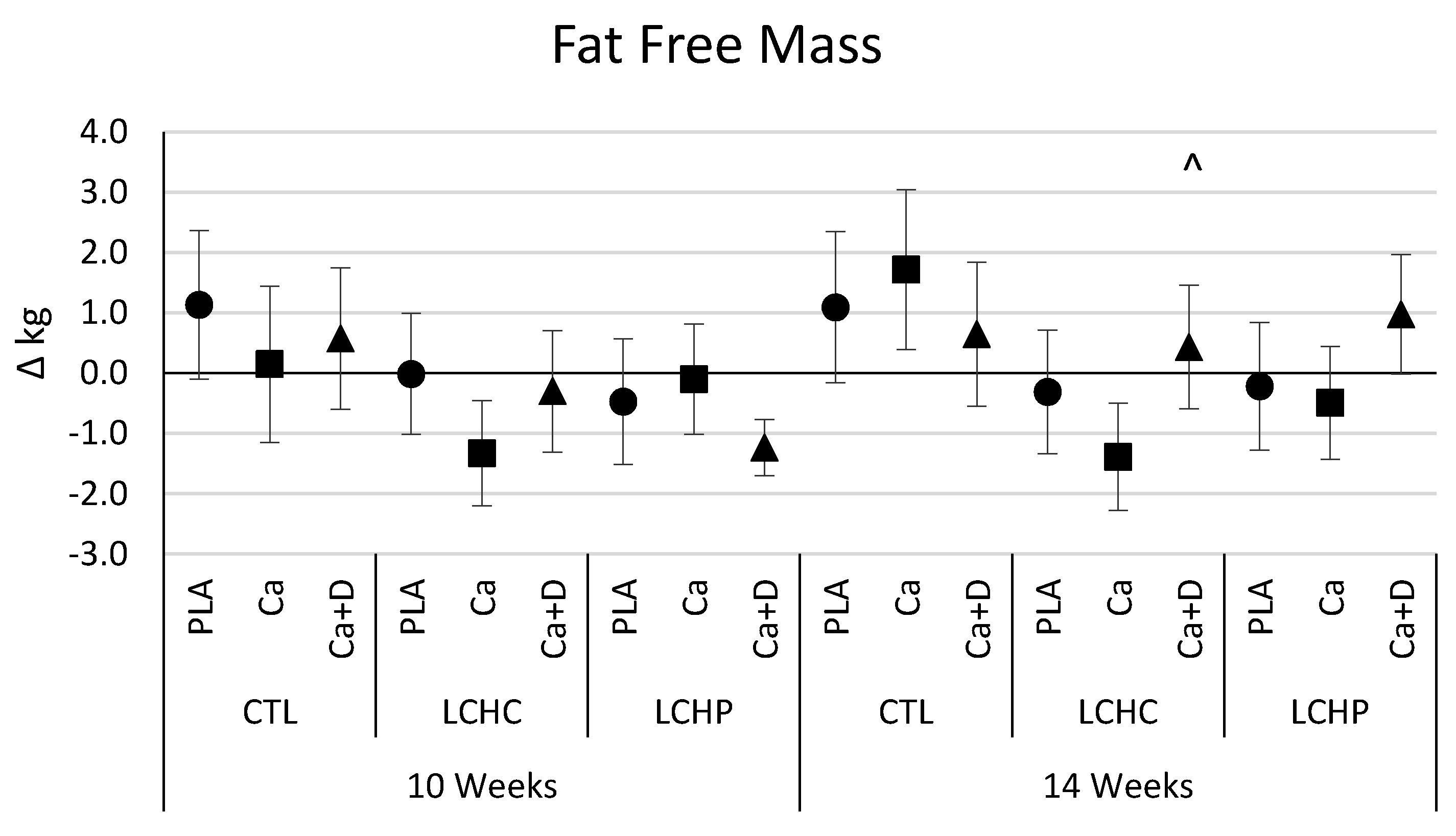
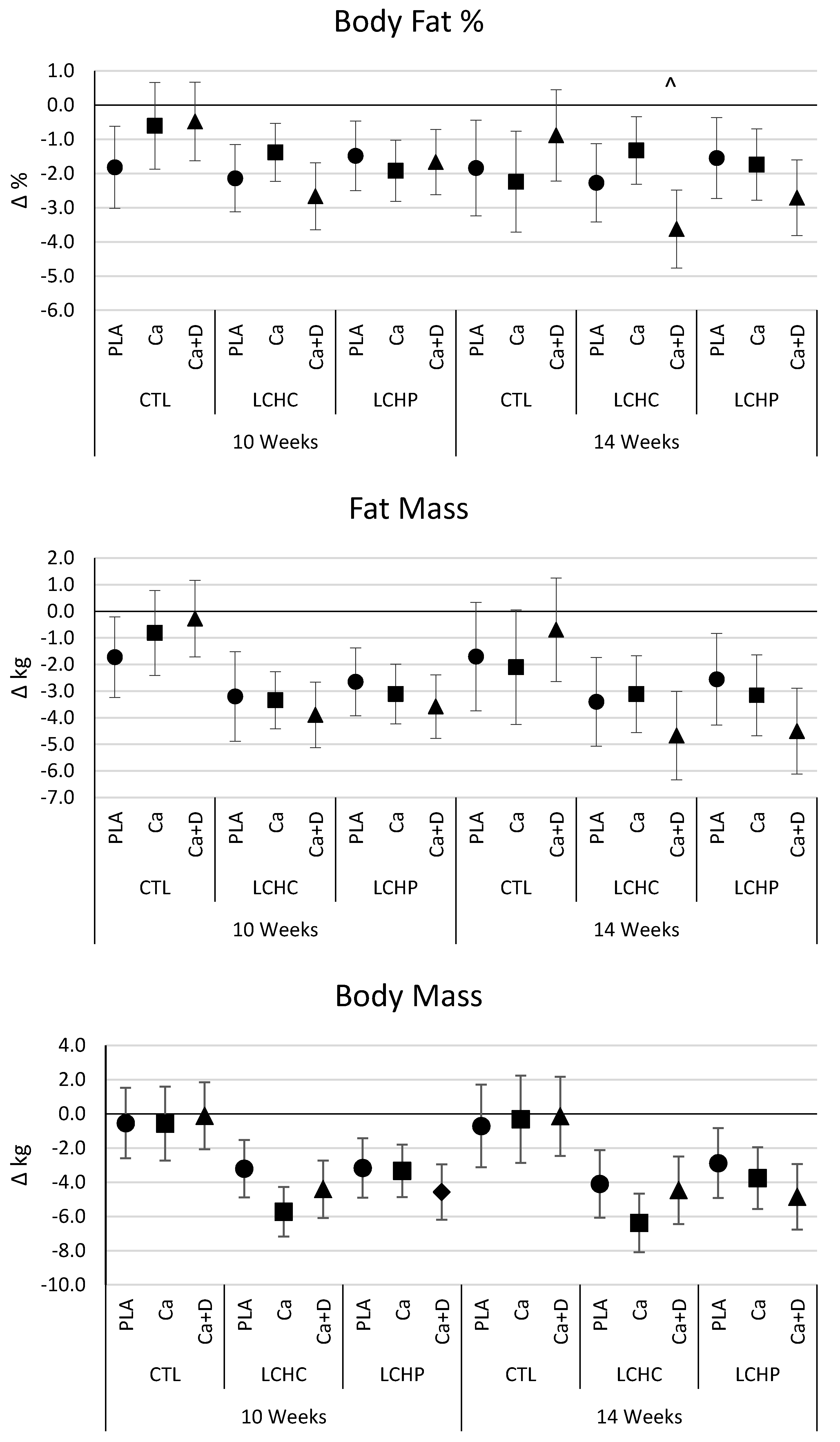
4.4. Body Weight and Composition
4.5. Resting Hemodynamics and Aerobic Capacity
4.6. Muscular Strength and Endurance
4.7. Blood Glucose and Lipids
5. Discussion and Conclusions
5.1. Primary Outcome—Weight Loss and Body Composition
5.2. Secondary Outcomes—Markers of Health & Fitness
5.3. Limitations
5.4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef]
- Ward, Z.J.; Bleich, S.N.; Cradock, A.L.; Barrett, J.L.; Giles, C.M.; Flax, C.; Long, M.W.; Gortmaker, S.L. Projected U.S. state-level prevalence of adult obesity and severe obesity. N. Engl. J. Med. 2019, 381, 2440–2450. [Google Scholar] [CrossRef]
- Dee, A.; Kearns, K.; O’Neill, C.; Sharp, L.; Staines, A.; O’Dwyer, V.; Fitzgerald, S.; Perry, I.J. The direct and indirect costs of both overweight and obesity: A systematic review. BMC Res. Notes 2014, 7, 242. [Google Scholar] [CrossRef]
- Wouterse, B.; Huisman, M.; Meijboom, B.R.; Deeg, D.J.; Polder, J.J. The effect of trends in health and longevity on health services use by older adults. BMC Health Serv. Res. 2015, 15, 574. [Google Scholar] [CrossRef]
- Withrow, D.; Alter, D.A. The economic burden of obesity worldwide: A systematic review of the direct costs of obesity. Obes. Rev. 2011, 12, 131–141. [Google Scholar] [CrossRef]
- Cawley, J.; Meyerhoefer, C. The medical care costs of obesity: An instrumental variables approach. J. Health Econ. 2012, 31, 219–230. [Google Scholar] [CrossRef]
- Fontaine, K.R.; Redden, D.T.; Wang, C.; Westfall, A.O.; Allison, D.B. Years of life lost due to obesity. JAMA 2003, 289, 187–193. [Google Scholar] [CrossRef]
- Donato, G.B.; Fuchs, S.C.; Oppermann, K.; Bastos, C.; Spritzer, P.M. Association between menopause status and central adiposity measured at different cutoffs of waist circumference and waist-to-hip ratio. Menopause 2006, 13, 280–285. [Google Scholar] [CrossRef]
- Hong, S.C.; Yoo, S.W.; Cho, G.J.; Kim, T.; Hur, J.Y.; Park, Y.K.; Lee, K.W.; Kim, S.H. Correlation between estrogens and serum adipocytokines in premenopausal and postmenopausal women. Menopause 2007, 14, 835–840. [Google Scholar] [CrossRef]
- Morton, R.W.; McGlory, C.; Phillips, S.M. Nutritional interventions to augment resistance training-induced skeletal muscle hypertrophy. Front. Physiol. 2015, 6, 245. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Gallagher, D.; Poehlman, E.T.; Wolper, C.; Nonas, K.; Nelson, D.; Wang, Z.M. Menopausal changes in body composition and energy expenditure. Exp. Gerontol. 1994, 29, 377–389. [Google Scholar] [CrossRef]
- Johansson, K.; Neovius, M.; Hemmingsson, E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 99, 14–23. [Google Scholar] [CrossRef]
- Poirier, P.; Despres, J.P. Exercise in weight management of obesity. Cardiol. Clin. 2001, 19, 459–470. [Google Scholar] [CrossRef]
- Kerksick, C.; Thomas, A.; Campbell, B.; Taylor, L.; Wilborn, C.; Marcello, B.; Roberts, M.; Pfau, E.; Grimstvedt, M.; Opusunju, J.; et al. Effects of a popular exercise and weight loss program on weight loss, body composition, energy expenditure and health in obese women. Nutr. Metab. (Lond.) 2009, 6, 23. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Wismann-Bunn, J.; Fogt, D.; Thomas, A.R.; Taylor, L.; Campbell, B.I.; Wilborn, C.D.; Harvey, T.; Roberts, M.D.; La Bounty, P.; et al. Changes in weight loss, body composition and cardiovascular disease risk after altering macronutrient distributions during a regular exercise program in obese women. Nutr. J. 2010, 9, 59. [Google Scholar] [CrossRef]
- Meckling, K.A.; Sherfey, R. A randomized trial of a hypocaloric high-protein diet, with and without exercise, on weight loss, fitness, and markers of the Metabolic Syndrome in overweight and obese women. Appl. Physiol. Nutr. Metab. 2007, 32, 743–752. [Google Scholar] [CrossRef]
- Claessens, M.; van Baak, M.A.; Monsheimer, S.; Saris, W.H. The effect of a low-fat, high-protein or high-carbohydrate ad libitum diet on weight loss maintenance and metabolic risk factors. Int. J. Obes. (Lond.) 2009, 33, 296–304. [Google Scholar] [CrossRef]
- Arsenault, B.J.; Cote, M.; Cartier, A.; Lemieux, I.; Despres, J.P.; Ross, R.; Earnest, C.P.; Blair, S.N.; Church, T.S. Effect of exercise training on cardiometabolic risk markers among sedentary, but metabolically healthy overweight or obese post-menopausal women with elevated blood pressure. Atherosclerosis 2009, 207, 530–533. [Google Scholar] [CrossRef]
- Gordon, M.M.; Bopp, M.J.; Easter, L.; Miller, G.D.; Lyles, M.F.; Houston, D.K.; Nicklas, B.J.; Kritchevsky, S.B. Effects of dietary protein on the composition of weight loss in post-menopausal women. J. Nutr. Health Aging 2008, 12, 505–509. [Google Scholar] [CrossRef]
- Gonzalez, J.T.; Rumbold, P.L.; Stevenson, E.J. Effect of calcium intake on fat oxidation in adults: A meta-analysis of randomized, controlled trials. Obes. Rev. 2012, 13, 848–857. [Google Scholar] [CrossRef]
- Major, G.C.; Alarie, F.P.; Dore, J.; Tremblay, A. Calcium plus vitamin D supplementation and fat mass loss in female very low-calcium consumers: Potential link with a calcium-specific appetite control. Br. J. Nutr. 2009, 101, 659–663. [Google Scholar] [CrossRef]
- Narvaez, C.J.; Matthews, D.; Broun, E.; Chan, M.; Welsh, J. Lean phenotype and resistance to diet-induced obesity in vitamin D receptor knockout mice correlates with induction of uncoupling protein-1 in white adipose tissue. Endocrinology 2009, 150, 651–661. [Google Scholar] [CrossRef]
- Yin, Y.; Yu, Z.; Xia, M.; Luo, X.; Lu, X.; Ling, W. Vitamin D attenuates high fat diet-induced hepatic steatosis in rats by modulating lipid metabolism. Eur. J. Clin. Investig. 2012, 42, 1189–1196. [Google Scholar] [CrossRef]
- Zemel, M.B. Role of dietary calcium and dairy products in modulating adiposity. Lipids 2003, 38, 139–146. [Google Scholar] [CrossRef]
- Zemel, M.B.; Shi, H.; Greer, B.; Dirienzo, D.; Zemel, P.C. Regulation of adiposity by dietary calcium. FASEB J. 2000, 14, 1132–1138. [Google Scholar] [CrossRef]
- Zemel, M.B. Mechanisms of dairy modulation of adiposity. J. Nutr. 2003, 133, 252S–256S. [Google Scholar] [CrossRef]
- Shi, H.; Dirienzo, D.; Zemel, M.B. Effects of dietary calcium on adipocyte lipid metabolism and body weight regulation in energy-restricted aP2-agouti transgenic mice. FASEB J. 2001, 15, 291–293. [Google Scholar] [CrossRef]
- Wang, H.; Bua, P.; Capodice, J. A comparative study of calcium absorption following a single serving administration of calcium carbonate powder versus calcium citrate tablets in healthy premenopausal women. Food Nutr. Res. 2014, 58. [Google Scholar] [CrossRef]
- Shahar, D.R.; Schwarzfuchs, D.; Fraser, D.; Vardi, H.; Thiery, J.; Fiedler, G.M.; Bluher, M.; Stumvoll, M.; Stampfer, M.J.; Shai, I.; et al. Dairy calcium intake, serum vitamin D, and successful weight loss. Am. J. Clin. Nutr. 2010, 92, 1017–1022. [Google Scholar] [CrossRef]
- Wagner, G.; Kindrick, S.; Hertzler, S.; DiSilvestro, R.A. Effects of various forms of calcium on body weight and bone turnover markers in women participating in a weight loss program. J. Am. Coll. Nutr. 2007, 26, 456–461. [Google Scholar] [CrossRef]
- Shapses, S.A.; Heshka, S.; Heymsfield, S.B. Effect of calcium supplementation on weight and fat loss in women. J. Clin. Endocrinol. Metab. 2004, 89, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.J.; Chan She Ping-Delfos, W.; Ghanbari, M.H. Calcium and vitamin D for obesity: A review of randomized controlled trials. Eur. J. Clin. Nutr. 2011, 65, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K. Protein quantity and quality at levels above the RDA improves adult weight loss. J. Am. Coll. Nutr. 2004, 23, 631S–636S. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K.; Boileau, R.A.; Erickson, D.J.; Painter, J.E.; Shiue, H.; Sather, C.; Christou, D.D. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J. Nutr. 2003, 133, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K.; Evans, E.; Baum, J.I.; Seyler, J.; Erickson, D.J.; Boileau, R.A. Dietary protein and exercise have additive effects on body composition during weight loss in adult women. J. Nutr. 2005, 135, 1903–1910. [Google Scholar] [CrossRef]
- World Medical, A. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Farris, G.; Wismann, J.; Farris, R.; Gandy, N.; Long, L.; Pfau, E.; Campbell, B.; La Bounty, P.; Rasmussen, C.; Wilson, R.; et al. Exercise intensity and energy expenditure analysis of women participating in the Curves® exercise program. FASEB J. 2006, LB, 93–94. [Google Scholar]
- Kreider, R.; Parker, A.; Moreillon, J.; Rasmussen, C.; Greenwood, M. Energy expenditure analysis of women participating in a computerized hydraulic circuit training program. J. Strength Cond. Res. 2008, 22, A69–A70. [Google Scholar]
- La Bounty, P.; Wilborn, C.; Marcello, B.; Campbell, B.; Faries, M.; Shim, J.; Rasmussen, C.; Kreider, R. Analysis of exercise intensities of women using the Curves® hydraulic training equipment. FASEB J. 2006, LB, 93. [Google Scholar]
- Kreider, R.B.; Rasmussen, C.; Kerksick, C.M.; Wilborn, C.; Taylor, L.T.; Campbell, B.; Magrans-Courtney, T.; Fogt, D.; Ferreira, M.; Li, R.; et al. A carbohydrate-restricted diet during resistance training promotes more favorable changes in body composition and markers of health in obese women with and without insulin resistance. Physician Sportsmed. 2011, 39, 27–40. [Google Scholar] [CrossRef]
- Galbreath, M.; Campbell, B.; LaBounty, P.; Bunn, J.; Dove, J.; Harvey, T.; Hudson, G.; Gutierrez, J.L.; Levers, K.; Galvan, E.; et al. Effects of adherence to a higher protein diet on weight loss, markers of health, and functional capacity in older women participating in a resistance-based exercise program. Nutrients 2018, 10, 1070. [Google Scholar] [CrossRef] [PubMed]
- Baetge, C.; Earnest, C.P.; Lockard, B.; Coletta, A.M.; Galvan, E.; Rasmussen, C.; Levers, K.; Simbo, S.Y.; Jung, Y.P.; Koozehchian, M.; et al. Efficacy of a randomized trial examining commercial weight loss programs and exercise on metabolic syndrome in overweight and obese women. Appl. Physiol. Nutr. Metab. 2017, 42, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Serra, M.; Beavers, K.M.; Moreillon, J.; Kresta, J.Y.; Byrd, M.; Oliver, J.M.; Gutierrez, J.; Hudson, G.; Deike, E.; et al. A structured diet and exercise program promotes favorable changes in weight loss, body composition, and weight maintenance. J. Am. Diet. Assoc. 2011, 111, 828–843. [Google Scholar] [CrossRef] [PubMed]
- Magrans-Courtney, T.; Wilborn, C.; Rasmussen, C.; Ferreira, M.; Greenwood, L.; Campbell, B.; Kerksick, C.M.; Nassar, E.; Li, R.; Iosia, M.; et al. Effects of diet type and supplementation of glucosamine, chondroitin, and MSM on body composition, functional status, and markers of health in women with knee osteoarthritis initiating a resistance-based exercise and weight loss program. J. Int. Soc. Sports Nutr. 2011, 8, 8. [Google Scholar] [CrossRef]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000. [Google Scholar]
- Cuka, S.; Dvornik, S.; Drazenovic, K.; Mihic, J. Evaluation of the Dade Behring Dimension RxL clinical chemistry analyzer. Clin. Lab. 2001, 47, 35–40. [Google Scholar] [PubMed]
- Lockard, B.; Earnest, C.P.; Oliver, J.; Goodenough, C.; Rasmussen, C.; Greenwood, M.; Kreider, R.B. Retrospective Analysis of Protein- and Carbohydrate-Focused Diets Combined with Exercise on Metabolic Syndrome Prevalence in Overweight and Obese Women. Metab. Syndr. Relat. Disord. 2016, 14, 228–237. [Google Scholar] [CrossRef]
- Page, P. Beyond statistical significance: Clinical interpretation of rehabilitation research literature. Int. J. Sports Phys. Ther. 2014, 9, 726–736. [Google Scholar]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Gotting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef]
- Vimaleswaran, K.S.; Berry, D.J.; Lu, C.; Tikkanen, E.; Pilz, S.; Hiraki, L.T.; Cooper, J.D.; Dastani, Z.; Li, R.; Houston, D.K.; et al. Causal relationship between obesity and vitamin D status: Bi-directional Mendelian randomization analysis of multiple cohorts. PLoS Med. 2013, 10, e1001383. [Google Scholar] [CrossRef]
- Major, G.C.; Alarie, F.; Dore, J.; Phouttama, S.; Tremblay, A. Supplementation with calcium + vitamin D enhances the beneficial effect of weight loss on plasma lipid and lipoprotein concentrations. Am. J. Clin. Nutr. 2007, 85, 54–59. [Google Scholar] [CrossRef]
- Holecki, M.; Zahorska-Markiewicz, B.; Wiecek, A.; Mizia-Stec, K.; Nieszporek, T.; Zak-Golab, A. Influence of calcium and vitamin D supplementation on weight and fat loss in obese women. Obes. Facts 2008, 1, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Deeg, D.J.; Lips, P.; Longitudinal Aging Study Amsterdam. Low vitamin D and high parathyroid hormone levels as determinants of loss of muscle strength and muscle mass (sarcopenia): The Longitudinal Aging Study Amsterdam. J. Clin. Endocrinol. Metab. 2003, 88, 5766–5772. [Google Scholar] [CrossRef] [PubMed]
- Gerdhem, P.; Ringsberg, K.A.; Obrant, K.J.; Akesson, K. Association between 25-hydroxy vitamin D levels, physical activity, muscle strength and fractures in the prospective population-based OPRA Study of Elderly Women. Osteoporos. Int. 2005, 16, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Inderjeeth, C.A.; Glennon, D.; Petta, A.; Soderstrom, J.; Boyatzis, I.; Tapper, J. Vitamin D and muscle strength in patients with previous fractures. N. Z. Med. J. 2007, 120, U2730. [Google Scholar]
- Stockton, K.A.; Mengersen, K.; Paratz, J.D.; Kandiah, D.; Bennell, K.L. Effect of vitamin D supplementation on muscle strength: A systematic review and meta-analysis. Osteoporos. Int. 2011, 22, 859–871. [Google Scholar] [CrossRef]
- Volek, J.S.; Vanheest, J.L.; Forsythe, C.E. Diet and exercise for weight loss: A review of current issues. Sports Med. 2005, 35, 1–9. [Google Scholar] [CrossRef]
- Stiegler, P.; Cunliffe, A. The role of diet and exercise for the maintenance of fat-free mass and resting metabolic rate during weight loss. Sports Med. 2006, 36, 239–262. [Google Scholar] [CrossRef]
- Samaha, F.F.; Iqbal, N.; Seshadri, P.; Chicano, K.L.; Daily, D.A.; McGrory, J.; Williams, T.; Williams, M.; Gracely, E.J.; Stern, L. A low-carbohydrate as compared with a low-fat diet in severe obesity. N. Engl. J. Med. 2003, 348, 2074–2081. [Google Scholar] [CrossRef]
- Yancy, W.S., Jr.; Olsen, M.K.; Guyton, J.R.; Bakst, R.P.; Westman, E.C. A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: A randomized, controlled trial. Ann. Intern. Med. 2004, 140, 769–777. [Google Scholar] [CrossRef]
- Nordmann, A.J.; Nordmann, A.; Briel, M.; Keller, U.; Yancy, W.S., Jr.; Brehm, B.J.; Bucher, H.C. Effects of low-carbohydrate vs. low-fat diets on weight loss and cardiovascular risk factors: A meta-analysis of randomized controlled trials. Ann. Intern. Med. 2006, 166, 285–293. [Google Scholar] [CrossRef]
- Wood, R.J.; Fernandez, M.L.; Sharman, M.J.; Silvestre, R.; Greene, C.M.; Zern, T.L.; Shrestha, S.; Judelson, D.A.; Gomez, A.L.; Kraemer, W.J.; et al. Effects of a carbohydrate-restricted diet with and without supplemental soluble fiber on plasma low-density lipoprotein cholesterol and other clinical markers of cardiovascular risk. Metab. Clin. Exp. 2007, 56, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Volek, J.S.; Sharman, M.J.; Love, D.M.; Avery, N.G.; Gomez, A.L.; Scheett, T.P.; Kraemer, W.J. Body composition and hormonal responses to a carbohydrate-restricted diet. Metab. Clin. Exp. 2002, 51, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Dansinger, M.L.; Gleason, J.A.; Griffith, J.L.; Selker, H.P.; Schaefer, E.J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: A randomized trial. JAMA 2005, 293, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Tay, J.; Brinkworth, G.D.; Noakes, M.; Keogh, J.; Clifton, P.M. Metabolic effects of weight loss on a very-low-carbohydrate diet compared with an isocaloric high-carbohydrate diet in abdominally obese subjects. J. Am. Coll. Cardiol. 2008, 51, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Naude, C.E.; Schoonees, A.; Senekal, M.; Young, T.; Garner, P.; Volmink, J. Low carbohydrate versus isoenergetic balanced diets for reducing weight and cardiovascular risk: A systematic review and meta-analysis. PLoS ONE 2014, 9, e100652. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kerksick, C.M.; Roberts, M.D.; Campbell, B.I.; Galbreath, M.M.; Taylor, L.W.; Wilborn, C.D.; Lee, A.; Dove, J.; Bunn, J.W.; Rasmussen, C.J.; et al. Differential Impact of Calcium and Vitamin D on Body Composition Changes in Post-Menopausal Women Following a Restricted Energy Diet and Exercise Program. Nutrients 2020, 12, 713. https://doi.org/10.3390/nu12030713
Kerksick CM, Roberts MD, Campbell BI, Galbreath MM, Taylor LW, Wilborn CD, Lee A, Dove J, Bunn JW, Rasmussen CJ, et al. Differential Impact of Calcium and Vitamin D on Body Composition Changes in Post-Menopausal Women Following a Restricted Energy Diet and Exercise Program. Nutrients. 2020; 12(3):713. https://doi.org/10.3390/nu12030713
Chicago/Turabian StyleKerksick, Chad M., Michael D. Roberts, Bill I. Campbell, Melyn M. Galbreath, Lemuel W. Taylor, Colin D. Wilborn, Ashli Lee, Jacqueline Dove, Jennifer W. Bunn, Christopher J. Rasmussen, and et al. 2020. "Differential Impact of Calcium and Vitamin D on Body Composition Changes in Post-Menopausal Women Following a Restricted Energy Diet and Exercise Program" Nutrients 12, no. 3: 713. https://doi.org/10.3390/nu12030713
APA StyleKerksick, C. M., Roberts, M. D., Campbell, B. I., Galbreath, M. M., Taylor, L. W., Wilborn, C. D., Lee, A., Dove, J., Bunn, J. W., Rasmussen, C. J., & Kreider, R. B. (2020). Differential Impact of Calcium and Vitamin D on Body Composition Changes in Post-Menopausal Women Following a Restricted Energy Diet and Exercise Program. Nutrients, 12(3), 713. https://doi.org/10.3390/nu12030713







