Inflammation (IL-1β) Modifies the Effect of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids on Core Symptoms of Autism Spectrum Disorder—An Exploratory Pilot Study †
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Measurements
2.3. Biochemical Analysis
2.4. Statistical Analysis
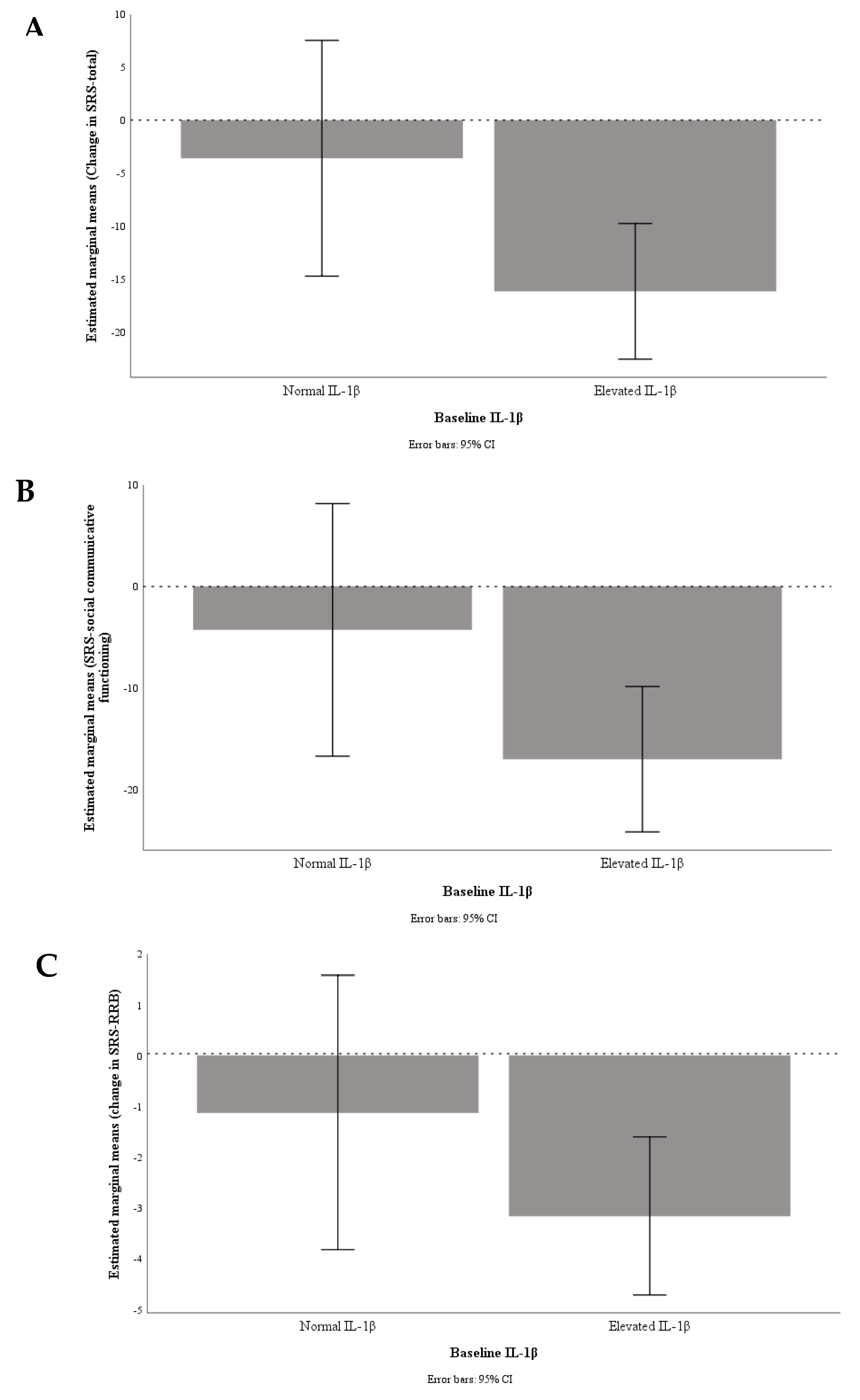
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5™, 5th ed.; American Psychiatric Association: Washinton, DC, USA, 2013. [Google Scholar]
- Mayes, S.D.; Calhoun, S.L.; Murray, M.J.; Ahuja, M.; Smith, L.A. Anxiety, depression, and irritability in children with autism relative to other neuropsychiatric disorders and typical development. Res. Autism Spectr. Disord. 2011, 5, 474–485. [Google Scholar] [CrossRef]
- Ohja, K.; Gozal, E.; Fahnestock, M.; Cai, L.; Cai, J.; Freedman, J.H.; Switala, A.; El-Baz, A.; Barnes, G.N. Neuroimmunologic and Neurotrophic Interactions in Autism Spectrum Disorders: Relationship to Neuroinflammation. Neuromol. Med. 2018, 20, 161–173. [Google Scholar]
- Alabdali, A.; Al-Ayadhi, L.; El-Ansary, A. Association of social and cognitive impairment and biomarkers in autism spectrum disorders. J. Neuroinflamm. 2014, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Matta, S.M.; Hill-Yardin, E.L.; Crack, P.J. The influence of neuroinflammation in Autism Spectrum Disorder. Brain Behav. Immun. 2019, 79, 75–90. [Google Scholar] [CrossRef]
- Estes, M.L.; McAllister, A.K. Immune mediators in the brain and peripheral tissues in autism spectrum disorder. Nat. Rev. Neurosci. 2015, 16, 469–486. [Google Scholar] [CrossRef]
- Chez, M.G.; Dowling, T.; Patel, P.B.; Khanna, P.; Kominsky, M. Elevation of tumor necrosis factor-alpha in cerebrospinal fluid of autistic children. Pediatr. Neurol. 2007, 36, 361–365. [Google Scholar] [CrossRef]
- Napolioni, V.; Ober-Reynolds, B.; Szelinger, S.; Corneveaux, J.J.; Pawlowski, T.; Ober-Reynolds, S.; Kirwan, J.; Persico, A.M.; Melmed, R.D.; Craig, D.W.; et al. Plasma cytokine profiling in sibling pairs discordant for autism spectrum disorder. J. Neuroinflamm. 2013, 10, 38. [Google Scholar] [CrossRef]
- Molloy, C.A.; Morrow, A.L.; Meinzen-Derr, J.; Schleifer, K.; Dienger, K.; Manning-Courtney, P.; Altaye, M.; Wills-Karp, M. Elevated cytokine levels in children with autism spectrum disorder. J. Neuroimmunol. 2006, 172, 198–205. [Google Scholar] [CrossRef]
- Ashwood, P.; Krakowiak, P.; Hertz-Picciotto, I.; Hansen, R.; Pessah, I.; Van de Water, J. Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain Behav. Immun. 2011, 25, 40–45. [Google Scholar] [CrossRef]
- Shen, L.; Feng, C.; Zhang, K.; Chen, Y.; Gao, Y.; Ke, J.; Chen, X.; Lin, J.; Li, C.; Iqbal, J.; et al. Proteomics Study of Peripheral Blood Mononuclear Cells (PBMCs) in Autistic Children. Front. Cell. Neurosci. 2019, 13, 105. [Google Scholar] [CrossRef]
- Giulian, D.; Young, D.G.; Woodward, J.; Brown, D.C.; Lachman, L.B. Interleukin-1 is an astroglial growth factor in the developing brain. J. Neurosci. 1988, 8, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Borsini, A.; Alboni, S.; Horowitz, M.A.; Tojo, L.M.; Cannazza, G.; Su, K.P.; Pariante, C.M.; Zunszain, P.A. Rescue of IL-1β-induced reduction of human neurogenesis by omega-3 fatty acids and antidepressants. Brain Behav. Immun. 2017, 65, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Li, X.; Leonard, B.E.; Horrobin, D.F. Effects of dietary n-3 or n-6 fatty acids on interleukin-1beta-induced anxiety, stress, and inflammatory responses in rats. J. Lipid Res. 2003, 44, 1984–1991. [Google Scholar] [CrossRef] [PubMed]
- Bluthé, R.; Parnet, P.; Dantzer, R.; Kelley, K.J.N.R.C. Interleukin-1 receptor antagonist blocks effects of IL-1α and IL-1β on social behaviour and body weight in mice. Symptom Res. CAO 1991, 9, 151–158. [Google Scholar]
- Masi, A.; Breen, E.J.; Alvares, G.A.; Glozier, N.; Hickie, I.B.; Hunt, A.; Hui, J.; Beilby, J.; Ravine, D.; Wray, J.; et al. Cytokine levels and associations with symptom severity in male and female children with autism spectrum disorder. Mol. Autism 2017, 8, 63. [Google Scholar] [CrossRef]
- Emanuele, E.; Orsi, P.; Boso, M.; Broglia, D.; Brondino, N.; Barale, F.; di Nemi, S.U.; Politi, P. Low-grade endotoxemia in patients with severe autism. Neurosci. Lett. 2010, 471, 162–165. [Google Scholar] [CrossRef]
- Enstrom, A.M.; Onore, C.E.; Van de Water, J.A.; Ashwood, P. Differential monocyte responses to TLR ligands in children with autism spectrum disorders. Brain Behav. Immun. 2010, 24, 64–71. [Google Scholar] [CrossRef]
- Jyonouchi, H.; Sun, S.; Le, H. Proinflammatory and regulatory cytokine production associated with innate and adaptive immune responses in children with autism spectrum disorders and developmental regression. J. Neuroimmunol. 2001, 120, 170–179. [Google Scholar] [CrossRef]
- Handley, M.T.; Lian, L.Y.; Haynes, L.P.; Burgoyne, R.D. Structural and functional deficits in a neuronal calcium sensor-1 mutant identified in a case of autistic spectrum disorder. PLoS ONE 2010, 5, e10534. [Google Scholar] [CrossRef]
- Tsai, S.J.; Hong, C.J.; Liu, M.E.; Hou, S.J.; Yen, F.C.; Hsieh, C.H.; Liou, Y.J. Interleukin-1 beta (C-511T) genetic polymorphism is associated with cognitive performance in elderly males without dementia. Neurobiol. Aging 2010, 31, 1950–1955. [Google Scholar] [CrossRef]
- Li, X.; Chauhan, A.; Sheikh, A.M.; Patil, S.; Chauhan, V.; Li, X.M.; Ji, L.; Brown, T.; Malik, M. Elevated immune response in the brain of autistic patients. J. Neuroimmunol. 2009, 207, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Ortiz, L.; Plotkin, S.R.; Kastin, A.J. Human interleukin (IL) 1 alpha, murine IL-1 alpha and murine IL-1 beta are transported from blood to brain in the mouse by a shared saturable mechanism. J. Pharmacol. Exp. Ther. 1991, 259, 988–996. [Google Scholar] [PubMed]
- Wang, Y.; Jin, S.; Sonobe, Y.; Cheng, Y.; Horiuchi, H.; Parajuli, B.; Kawanokuchi, J.; Mizuno, T.; Takeuchi, H.; Suzumura, A. Interleukin-1beta induces blood-brain barrier disruption by downregulating Sonic hedgehog in astrocytes. PLoS ONE 2014, 9, e110024. [Google Scholar]
- Clifford, S.; Dissanayake, C.; Bui, Q.M.; Huggins, R.; Taylor, A.K.; Loesch, D.Z. Autism spectrum phenotype in males and females with fragile X full mutation and premutation. J. Autism Dev. Disord. 2007, 37, 738–747. [Google Scholar] [CrossRef]
- Mazahery, H.; Camargo, C.A., Jr.; Conlon, C.; Beck, K.L.; Kruger, M.C.; von Hurst, P.R. Vitamin D and autism spectrum disorder: A literature review. Nutrients 2016, 8, 236. [Google Scholar] [CrossRef]
- Mazahery, H.; Stonehouse, W.; Delshad, M.; Kruger, M.C.; Conlon, C.A.; Beck, K.L.; von Hurst, P.R. Relationship between Long Chain n-3 polyunsaturated fatty acids and autism spectrum disorder: Systematic review and meta-analysis of case-control and randomised controlled trials. Nutrients 2017, 9, 155. [Google Scholar] [CrossRef]
- Morgese, M.G.; Schiavone, S.; Maffione, A.B.; Tucci, P.; Trabace, L. Depressive-like phenotype evoked by lifelong nutritional omega-3 deficiency in female rats: Crosstalk among kynurenine, Toll-like receptors and amyloid beta oligomers. Brain Behav. Immun. 2020. [Google Scholar] [CrossRef]
- Calton, E.K.; Keane, K.N.; Newsholme, P.; Soares, M.J. The Impact of vitamin D levels on inflammatory status: A systematic review of immune cell studies. PLoS ONE 2015, 10, e0141770. [Google Scholar] [CrossRef]
- Reid, D.; Toole, B.J.; Knox, S.; Talwar, D.; Harten, J.; O’Reilly, D.S.J.; Blackwell, S.; Kinsella, J.; McMillan, D.C.; Wallace, A.M. The relation between acute changes in the systemic inflammatory response and plasma 25-hydroxyvitamin D concentrations after elective knee arthroplasty. Am. J. Clin. Nutr. 2011, 93, 1006–1011. [Google Scholar] [CrossRef]
- Kang, J.X.; Weylandt, K.H. Modulation of inflammatory cytokines by omega-3 fatty acids. Subcell. Biochem. 2008, 49, 133–143. [Google Scholar]
- Griffin, M.D.; Lutz, W.; Phan, V.A.; Bachman, L.A.; McKean, D.J.; Kumar, R. Dendritic cell modulation by 1α,25 dihydroxyvitamin D3 and its analogs: A vitamin D receptor-dependent pathway that promotes a persistent state of immaturity in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2001, 98, 6800–6805. [Google Scholar] [CrossRef] [PubMed]
- Takano, T. Role of microglia in autism: Recent advances. Dev. Neurosci. 2015, 37, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Vasileva, S.; Langguth, M.; Alexander, S.; Cui, X.; Whitehouse, A.; McGrath, J.J.; Eyles, D. Developmental vitamin D deficiency produces behavioral phenotypes of relevance to autism in an animal model. Nutrients 2019, 11, 1187. [Google Scholar] [CrossRef] [PubMed]
- Pietropaolo, S.; Goubran, M.G.; Joffre, C.; Aubert, A.; Lemaire-Mayo, V.; Crusio, W.E.; Laye, S. Dietary supplementation of omega-3 fatty acids rescues fragile X phenotypes in Fmr1-Ko mice. Psychoneuroendocrinology 2014, 49, 119–129. [Google Scholar] [CrossRef]
- Weiser, M.J.; Mucha, B.; Denheyer, H.; Atkinson, D.; Schanz, N.; Vassiliou, E.; Benno, R.H. Dietary docosahexaenoic acid alleviates autistic-like behaviors resulting from maternal immune activation in mice. Prostaglandins Leukot Essent Fat. Acids 2016, 106, 27–37. [Google Scholar] [CrossRef]
- Vuillermot, S.; Luan, W.; Meyer, U.; Eyles, D. Vitamin D treatment during pregnancy prevents autism-related phenotypes in a mouse model of maternal immune activation. Mol. Autism 2017, 8, 9. [Google Scholar] [CrossRef]
- Vinkhuyzen, A.A.E.; Eyles, D.W.; Burne, T.H.J.; Blanken, L.M.E.; Kruithof, C.J.; Verhulst, F.; Jaddoe, V.W.; Tiemeier, H.; McGrath, J.J. Gestational vitamin D deficiency and autism-related traits: The generation r study. Mol. Psychiatry 2018, 23, 240–246. [Google Scholar] [CrossRef]
- Wu, D.M.; Wen, X.; Han, X.R.; Wang, S.; Wang, Y.J.; Shen, M.; Fan, S.H.; Zhuang, J.; Li, M.Q.; Hu, B.; et al. Relationship between neonatal vitamin D at birth and risk of autism spectrum disorders: The NBSIB study. J. Bone Miner. Res. 2018, 33, 458–466. [Google Scholar] [CrossRef]
- Steenweg-de Graaff, J.; Tiemeier, H.; Ghassabian, A.; Rijlaarsdam, J.; Jaddoe, V.W.V.; Verhulst, F.C.; Roza, S.J. Maternal fatty acid status during pregnancy and child autistic traits: The Generation R study. Am. J. Epidemiol. 2016, 183, 792–799. [Google Scholar] [CrossRef]
- Lee, B.K.; Eyles, D.W.; Magnusson, C.; Newschaffer, C.J.; McGrath, J.J.; Kvaskoff, D.; Ko, P.; Dalman, C.; Karlsson, H.; Gardner, R.M. Developmental vitamin D and autism spectrum disorders: Findings from the Stockholm Youth Cohort. Mol. Psychiatry 2019. [Google Scholar] [CrossRef]
- Morgese, M.G.; Trabace, L. Maternal malnutrition in the etiopathogenesis of psychiatric diseases: Role of polyunsaturated fatty acids. Brain Sci. 2016, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, A., Jr.; Meyer, B.J.; Tsang, B.; Jones, B.; et al. A randomised-controlled trial of vitamin d and omega-3 long chain polyunsaturated fatty acids in the treatment of core symptoms of autism spectrum disorder in children. J. Autism Dev. Disord. 2019, 49, 1778–1794. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.; Beaulieu, A.A. Psychotropic medications in children with autism spectrum disorders: A systematic review and synthesis for evidence-based practice. J. Autism Dev. Disord. 2012, 42, 1592–1605. [Google Scholar] [CrossRef] [PubMed]
- Masi, A.; Lampit, A.; Glozier, N.; Hickie, I.B.; Guastella, A.J. Predictors of placebo response in pharmacological and dietary supplement treatment trials in pediatric autism spectrum disorder: A meta-analysis. Transl. Psychiatry 2015, 5, e640. [Google Scholar] [CrossRef]
- Labrousse, V.F.; Leyrolle, Q.; Amadieu, C.; Aubert, A.; Sere, A.; Coutureau, E.; Gregoire, S.; Bretillon, L.; Pallet, V.; Gressens, P.; et al. Dietary omega-3 deficiency exacerbates inflammation and reveals spatial memory deficits in mice exposed to lipopolysaccharide during gestation. Brain Behav. Immun. 2018, 73, 427–440. [Google Scholar] [CrossRef]
- Grudet, C.; Malm, J.; Westrin, Å.; Brundin, L. Suicidal patients are deficient in vitamin D, associated with a pro-inflammatory status in the blood. Psychoneuroendocrinology 2014, 50, 210–219. [Google Scholar] [CrossRef]
- Accortt, E.E.; Schetter, C.D.; Peters, R.M.; Cassidy-Bushrow, A.E. Lower prenatal vitamin D status and postpartum depressive symptomatology in African American women: Preliminary evidence for moderation by inflammatory cytokines. Arch. Womens Ment. Health 2016, 19, 373–383. [Google Scholar] [CrossRef]
- Mazahery, H.; Conlon, C.; Beck, K.L.; Kruger, M.C.; Stonehouse, W.; Camargo, A., Jr.; Meyer, B.J.; Tsang, B.; Mugridge, O.; von Hurst, P.R. Vitamin D and omega-3 fatty acid supplements in children with autism spectrum disorder: A study protocol for a factorial randomised, double-blind, placebo-controlled trial. Trials 2016, 17, 295. [Google Scholar] [CrossRef]
- Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, A., Jr.; Meyer, B.J.; Jones, B.; von Hurst, P.R. A randomised controlled trial of vitamin D and omega-3 long chain polyunsaturated fatty acids in the treatment of irritability and hyperactivity among children with autism spectrum disorder. J. Steroid Biochem. Mol. Biol. 2018. [Google Scholar] [CrossRef]
- Ministry of Health. Food and Nutrition Guidelines for Healthy Children and Young People (Aged 2–18 Years) A background Paper; Health, Minsitry of Health: Wellington, New Zealand, 2012.
- Constantino, J.; Gruber, C. The Social Responsiveness Scale, 2nd ed.; Western Psychological Services: Los Angeles, CA, USA, 2012. [Google Scholar]
- Cholemkery, H.; Kitzerow, J.; Rohrmann, S.; Freitag, C. Validity of the social responsiveness scale to differentiate between autism spectrum disorders and disruptive behaviour disorders. Eur. Child Adolesc. Psychiatry 2014, 23, 81–93. [Google Scholar] [CrossRef]
- Bolte, S.; Poustka, F.; Constantino, J.N. Assessing autistic traits: Cross-cultural validation of the social responsiveness scale (SRS). Autism Res. 2008, 1, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, A.; Ferrucci, L.; Sparvieri, E.; Cherubini, A.; Volpato, S.; Corsi, A.; Bonafe, M.; Franceschi, C.; Abate, G.; Paganelli, R. Serum IL-1beta levels in health and disease: A population-based study. ‘The InCHIANTI study’. Cytokine 2003, 22, 198–205. [Google Scholar] [CrossRef]
- Siniscalco, D.; Schultz, S.; Brigida, A.L.; Antonucci, N. Inflammation and neuro-immune dysregulations in autism spectrum disorders. Pharmaceuticals 2018, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- Tsilioni, I.; Taliou, A.; Francis, K.; Theoharides, T.C. Children with autism spectrum disorders, who improved with a luteolin-containing dietary formulation, show reduced serum levels of TNF and IL-6. Transl. Psychiatry 2015, 5, e647. [Google Scholar] [CrossRef]
- Mazereeuw, G.; Herrmann, N.; Andreazza, A.C.; Scola, G.; Ma, D.W.L.; Oh, P.I.; Lanctôt, K.L. Baseline oxidative stress is associated with memory changes in omega-3 fatty acid treated coronary artery disease patients. Cardiovasc. Psychiatry Neurol. 2017, 2017, 7. [Google Scholar] [CrossRef]
- Suzuki, K.; Matsuzaki, H.; Iwata, K.; Kameno, Y.; Shimmura, C.; Kawai, S.; Yoshihara, Y.; Wakuda, T.; Takebayashi, K.; Takagai, S.; et al. Plasma cytokine profiles in subjects with high-functioning autism spectrum disorders. PLoS ONE 2011, 6, e20470. [Google Scholar] [CrossRef]
- Arteaga-Henriquez, G.; Simon, M.S.; Burger, B.; Weidinger, E.; Wijkhuijs, A.; Arolt, V.; Birkenhager, T.K.; Musil, R.; Muller, N.; Drexhage, H.A. Low-grade inflammation as a predictor of antidepressant and anti-inflammatory therapy response in MDD patients: A systematic review of the literature in combination with an analysis of experimental data collected in the EU-MOODINFLAME consortium. Front. Psychiatry 2019, 10, 458. [Google Scholar] [CrossRef]
- Mondelli, V.; Ciufolini, S.; Belvederi Murri, M.; Bonaccorso, S.; Di Fort, M.I.; Giordano, A.; Marques, T.R.; Zunszain, P.A.; Morgan, C.; Murray, R.M.; et al. Cortisol and inflammatory biomarkers predict poor treatment response in first episode psychosis. Schizophr. Bull. 2015, 41, 1162–1170. [Google Scholar] [CrossRef]
- Lasselin, J.; Kemani, M.K.; Kanstrup, M.; Olsson, G.L.; Axelsson, J.; Andreasson, A.; Lekander, M.; Wicksell, R.K. Low-grade inflammation may moderate the effect of behavioral treatment for chronic pain in adults. J. Behav. Med. 2016, 39, 916–924. [Google Scholar] [CrossRef]
- Raison, C.L.; Rutherford, R.E.; Woolwine, B.J.; Shuo, C.; Schettler, P.; Drake, D.F.; Haroon, E.; Miller, A.H. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: The role of baseline inflammatory biomarkers. JAMA Psychiatry 2013, 70, 31–41. [Google Scholar] [CrossRef]
- Eisenberger, N.I.; Inagaki, T.K.; Mashal, N.M.; Irwin, M.R. Inflammation and social experience: An inflammatory challenge induces feelings of social disconnection in addition to depressed mood. Brain Behav. Immun. 2010, 24, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Gladysz, D.; Krzywdzinska, A.; Hozyasz, K.K. Immune abnormalities in autism spectrum disorder-could they hold promise for causative treatment? Mol. Neurobiol. 2018, 55, 6387–6435. [Google Scholar] [CrossRef] [PubMed]
- Moieni, M.; Irwin, M.R.; Jevtic, I.; Breen, E.C.; Eisenberger, N.I. Inflammation impairs social cognitive processing: A randomized controlled trial of endotoxin. Brain Behav. Immun. 2015, 48, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Bollen, J.; Trick, L.; Llewellyn, D.; Dickens, C. The effects of acute inflammation on cognitive functioning and emotional processing in humans: A systematic review of experimental studies. J. Psychosom. Res. 2017, 94, 47–55. [Google Scholar] [CrossRef]
- Marchezan, J.; Winkler Dos Santos, E.G.A.; Deckmann, I.; Riesgo, R.D.S. Immunological dysfunction in autism spectrum disorder: A potential target for therapy. Neuroimmunomodulation 2018, 25, 300–319. [Google Scholar] [CrossRef]
- Asadabadi, M.; Mohammadi, M.R.; Ghanizadeh, A.; Modabbernia, A.; Ashrafi, M.; Hassanzadeh, E.; Forghani, S.; Akhondzadeh, S. Celecoxib as adjunctive treatment to risperidone in children with autistic disorder: A randomized, double-blind, placebo-controlled trial. Psychopharmacology 2013, 225, 51–59. [Google Scholar] [CrossRef]
- Victoria, R.; Caroline Flora, S.; Youssef, D.; Jules Alexandre, D. Prediction by pharmacogenetics of safety and efficacy of non-steroidal anti- inflammatory drugs: A review. Curr. Drug Metab. 2014, 15, 326–343. [Google Scholar]
- Fattorusso, A.; Di Genova, L.; Dell’Isola, G.B.; Mencaroni, E.; Esposito, S. Autism spectrum disorders and the gut microbiota. Nutrients 2019, 11, 521. [Google Scholar] [CrossRef]
- Cantorna, M.T.; Snyder, L.; Arora, J. Vitamin A and vitamin D regulate the microbial complexity, barrier function, and the mucosal immune responses to ensure intestinal homeostasis. Crit. Rev. Biochem. Mol. Biol. 2019, 54, 184–192. [Google Scholar] [CrossRef]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of omega-3 fatty acids on the gut microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef]
- Ge, X.; Wang, L.; Li, M.; Xu, N.; Yu, F.; Yang, F.; Li, R.; Zhang, F.; Zhao, B.; Du, J. Vitamin D/VDR signaling inhibits LPS-induced IFNγ and IL-1β in Oral epithelia by regulating hypoxia-inducible factor-1α signaling pathway. Cell Commun. Signal. 2019, 17, 18. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Fragala, M.S.; McElhaney, J.E.; Kuchel, G.A. Conceptual and methodological issues relevant to cytokine and inflammatory marker measurements in clinical research. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 541–547. [Google Scholar] [CrossRef] [PubMed]
| Nutritional Deficiency | Management |
|---|---|
| Vitamin D | Participants with serum 25(OH)D concentrations <25 nmol/L were offered supplementation of 400IU per day 1 |
| Iron | Children with iron deficiency were offered iron supplements and postponed entry into the trial after 3 months. Children were retested. A child was iron deficient when 2 of the following pools were abnormal: red cell pool (haemoglobin <111 gr/L, red blood cell distribution width >14%), transport iron (iron saturation <16%) and/or storage iron (serum ferritin ≤15 µg/L) 1. Criteria for treatment were according to the New Zealand Ministry of Health guidelines 2 |
| Vitamin B12 | Children with serum levels <110 pmol/L were offered the option of prescribed supplements or dietary advice to improve status |
| Variables | Total (n = 67) | IL-1β | ||
|---|---|---|---|---|
| Undetectable/Normal (n = 15) | Elevated (n = 52) | p-Value * | ||
| Age (years), mean ± SD | 5.3 ± 1.4 | 5.5 ± 1.4 | 5.3 ± 1.4 | 0.61 |
| Sex, n (%) | 0.67 | |||
| Male | 56 (84) | 12 (21) | 44 (79) | |
| Female | 11 (16) | 3 (27) | 8 (73) | |
| Ethnicity, n (%) | 0.64 | |||
| New Zealand European | 34 (52) | 10 (29) | 24 (71) | |
| Māori | 9 (14) | 2 (22) | 7 (78) | |
| Pacific | 2 (3) | 0 (0) | 2 (100) | |
| Asian | 10 (15) | 1 (10) | 9 (90) | |
| Others | 11 (17) | 2 (18) | 9 (82) | |
| Annual household income (NZ$), n (%) | 0.24 | |||
| <60,000 | 15 (25) | 1 (7) | 14 (93) | |
| 60,000–120,000 | 32 (53) | 8 (25) | 24 (75) | |
| >120,000 | 13 (22) | 4 (31) | 9 (69) | |
| Season of blood collection, n (%) | 0.25 | |||
| Summer and Autumn | 36 (54) | 10 (28) | 26 (72) | |
| Winter and Spring | 31 (46) | 5 (16) | 26 (84) | |
| BMI-for-age categories, n (%) | 0.93 | |||
| Normal (<85th percentile) | 44 (66) | 10 (23) | 34 (77) | |
| Overweight/obese (≥85th percentiles) | 23 (34) | 5 (22) | 18 (78) | |
| Severity of ASD (based on confirmed medical diagnosis), n (%) | 0.29 | |||
| Mild | 29 (43) | 9 (31) | 20 (69) | |
| Moderate | 29 (43) | 4 (14) | 25 (86) | |
| Severe | 9 (14) | 2 (22) | 7 (78) | |
| Scores on ASD behavioural symptoms (assessed using SRS), mean ± SD | ||||
| Total | 101 ± 26 | 98 ± 26 | 102 ± 27 | 0.66 |
| Social and communicative functioning | 82 ± 22 | 79 ± 22 | 83 ± 22 | 0.55 |
| Awareness | 13 ± 4.0 | 14 ± 4.0 | 13 ± 4.0 | 0.64 |
| Cognition | 20 ± 5.7 | 19 ± 5.9 | 21 ± 5.6 | 0.26 |
| Communication | 34 ± 10 | 33 ± 10 | 34 ± 10 | 0.89 |
| Motivation | 14 ± 5.6 | 13 ± 4.2 | 15 ± 5.9 | 0.14 |
| RRB | 20 ± 6.0 | 20 ± 5.1 | 20 ± 6.3 | 0.93 |
| Biochemical markers | ||||
| Serum 25(OH)D (nmol/L), mean ± SD | 62 ± 24 | 66 ± 21 | 61 ± 25 | 0.41 |
| Omega-3 index **, median (25th, 75th percentiles) | 4.7 (4.4, 5.2) | 4.7 (4.3, 6.2) | 4.7 (4.4, 5.3) | 0.55 |
| Outcome Variables | Study Groups | |||
|---|---|---|---|---|
| VID (n = 15) | OM (n = 21) | VIDOM (n = 15) | Placebo (n = 16) | |
| Total | ||||
| Baseline | 100 ± 24 | 100 ± 26 | 97 ± 29 | 108 ± 27 |
| Endpoint | 92 ± 32 | 82 ± 31 | 84 ± 33 | 102 ± 24 |
| Change | −8.6 ± 25 | −18±18 | −13 ± 21 | −5.8 ± 12 |
| P-value 1 | 0.56 | 0.06 | 0.11 | |
| Effect size | <0.01 | 0.07 | 0.04 | |
| Social communicative functioning | ||||
| Baseline | 82 ± 20 | 81 ± 21 | 78 ± 23 | 88 ± 24 |
| Endpoint | 74 ± 26 | 67 ± 25 | 62 ± 28 | 82 ± 19 |
| Change | −7.8 ± 20 | −14 ± 16 | −16 ± 24 | −5.6 ± 11 |
| P-value 1 | 0.62 | 0.11 | 0.05 | |
| Effect size | <0.01 | 0.05 | 0.07 | |
| Social awareness | ||||
| Baseline | 13 ± 2.9 | 13 ± 3.9 | 13 ± 4.4 | 13 ± 4.8 |
| Endpoint | 13 ± 3.5 | 12 ± 4.3 | 11 ± 5.1 | 13 ± 4.0 |
| Change | −0.5 ± 2.7 | −1.6 ± 2.4 | −1.7 ± 3.5 | 0.4 ± 2.8 |
| P-value 1 | 0.26 | 0.01 | 0.01 | |
| Effect size | 0.02 | 0.11 | 0.11 | |
| Social cognition | ||||
| Baseline | 20 ± 5.8 | 20 ± 5.4 | 19 ± 6.6 | 22 ± 5.1 |
| Endpoint | 18 ± 7.9 | 19 ± 14 | 17 ± 7.0 | 20 ± 5.5 |
| Change | −1.4 ± 4.8 | −0.9 ± 12 | −2.3 ± 4.1 | −2.3 ± 2.8 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | <0.01 | <0.01 | <0.01 | |
| Communication | ||||
| Baseline | 34 ± 9.8 | 32 ± 9.5 | 33 ± 12 | 37 ± 11 |
| Endpoint | 30 ± 12 | 26 ± 11 | 28 ± 12 | 34 ± 9.2 |
| Change | −3.9 ± 10 | −5.8 ± 8.1 | −5.3 ± 9.8 | −2.4 ± 7.1 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | 0.01 | 0.04 | 0.05 | |
| Social motivation | ||||
| Baseline | 15 ± 5.4 | 15 ± 5.5 | 13 ± 5.3 | 16 ± 6.2 |
| Endpoint | 13 ± 5.9 | 11 ± 5.6 | 11 ± 5.5 | 14 ± 4.0 |
| Change | −1.9 ± 4.7 | −3.8 ± 4.3 | −2.0 ± 4.5 | −1.1 ± 4.3 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | <0.01 | 0.04 | 0.01 | |
| Repetitive and restricted interest and behaviour | ||||
| Baseline | 20 ± 6.0 | 19 ± 6.6 | 18 ± 5.6 | 22 ± 5.7 |
| Endpoint | 18 ± 6.6 | 16 ± 7.8 | 17 ± 7.0 | 20 ± 5.5 |
| Change | −2.7 ± 5.1 | −3.5 ± 4.3 | −1.4 ± 4.9 | −1.8 ± 5.4 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | <0.01 | 0.02 | <0.01 | |
| Outcome Variables | Study Groups | |||
|---|---|---|---|---|
| VID (n = 9) | OM (n = 16) | VIDOM (n = 12) | Placebo (n = 15) | |
| Total | ||||
| Baseline | 98 ± 24 | 104 ± 27 | 97 ± 29 | 106 ± 28 |
| Endpoint | 83 ± 32 | 82 ± 33 | 86 ± 33 | 100 ± 23 |
| Change | −15 ± 23 | −21 ± 19 | −11 ± 23 | −6.3 ± 13 |
| P-value 1 | 0.07 | 0.01 | 0.17 | |
| Effect size 2 | 0.08 | 0.14 | 0.04 | |
| Social communicative functioning | ||||
| Baseline | 81 ± 21 | 84 ± 21 | 78 ± 23 | 86 ± 24 |
| Endpoint | 67 ± 27 | 66 ± 26 | 63 ± 28 | 80 ± 19 |
| Change | −14 ± 17 | −17 ± 16 | −15 ± 26 | −6.1 ± 11 |
| P-value 1 | 0.09 | 0.03 | 0.05 | |
| Effect size 2 | 0.07 | 0.11 | 0.09 | |
| Social awareness | ||||
| Baseline | 13 ± 2.3 | 14 ± 4.1 | 13 ± 4.4 | 13 ± 5.0 |
| Endpoint | 12 ± 3.6 | 12 ± 4.4 | 12 ± 5.2 | 13 ± 4.0 |
| Change | −1.4 ± 2.7 | −1.8 ± 2.5 | −1.0 ± 2.6 | 0.5 ± 2.9 |
| P-value 1 | 0.01 | 0.003 | 0.01 | |
| Effect size 2 | 0.14 | 0.18 | 0.14 | |
| Social cognition | ||||
| Baseline | 19 ± 4.9 | 21 ± 5.9 | 21 ± 6.7 | 22 ± 5.0 |
| Endpoint | 16 ± 7.9 | 19 ± 16 | 18 ± 7.3 | 19 ± 6.0 |
| Change | −3.1 ± 4.3 | −1.6 ± 13 | −2.6 ± 4.4 | −2.2 ± 2.9 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | <0.01 | <0.01 | <0.01 | |
| Communication | ||||
| Baseline | 33 ± 11 | 33 ± 9.6 | 33 ± 11 | 36 ± 11 |
| Endpoint | 27 ± 12 | 26 ± 11 | 29 ± 11 | 33 ± 10 |
| Change | −6.4 ± 8.8 | −6.5 ± 7.8 | −4.3 ± 10 | −3.0 ± 7.0 |
| P-value 1 | 0.07 | 0.10 | >0.1 | |
| Effect size 2 | 0.07 | 0.06 | 0.05 | |
| Social motivation | ||||
| Baseline | 16 ± 6.8 | 16 ± 4.9 | 13 ± 5.6 | 15 ± 6.0 |
| Endpoint | 13 ± 6.9 | 12 ± 5.9 | 11 ± 5.5 | 14 ± 4.0 |
| Change | −2.8 ± 3.9 | −4.3 ± 4.2 | −1.5 ± 4.7 | −1.3 ± 4.2 |
| P-value 1 | >0.1 | 0.05 | >0.1 | |
| Effect size 2 | 0.03 | 0.09 | <0.01 | |
| Repetitive and restricted interest and behaviour | ||||
| Baseline | 19 ± 6.5 | 20 ± 6.9 | 18 ± 6.1 | 22 ± 6.0 |
| Endpoint | 16 ± 5.5 | 16 ± 8.2 | 17 ± 7.7 | 20 ± 6.0 |
| Change | −3.8 ± 4.6 | −3.8 ± 4.8 | −1.4 ± 5.5 | −1.8 ± 5.6 |
| P-value 1 | >0.1 | >0.1 | >0.1 | |
| Effect size 2 | 0.04 | 0.03 | <0.01 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, C.A., Jr.; Meyer, B.J.; Tsang, B.; von Hurst, P.R. Inflammation (IL-1β) Modifies the Effect of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids on Core Symptoms of Autism Spectrum Disorder—An Exploratory Pilot Study . Nutrients 2020, 12, 661. https://doi.org/10.3390/nu12030661
Mazahery H, Conlon CA, Beck KL, Mugridge O, Kruger MC, Stonehouse W, Camargo CA Jr., Meyer BJ, Tsang B, von Hurst PR. Inflammation (IL-1β) Modifies the Effect of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids on Core Symptoms of Autism Spectrum Disorder—An Exploratory Pilot Study . Nutrients. 2020; 12(3):661. https://doi.org/10.3390/nu12030661
Chicago/Turabian StyleMazahery, Hajar, Cathryn A. Conlon, Kathryn L. Beck, Owen Mugridge, Marlena C. Kruger, Welma Stonehouse, Carlos A. Camargo, Jr., Barbara J. Meyer, Bobby Tsang, and Pamela R. von Hurst. 2020. "Inflammation (IL-1β) Modifies the Effect of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids on Core Symptoms of Autism Spectrum Disorder—An Exploratory Pilot Study " Nutrients 12, no. 3: 661. https://doi.org/10.3390/nu12030661
APA StyleMazahery, H., Conlon, C. A., Beck, K. L., Mugridge, O., Kruger, M. C., Stonehouse, W., Camargo, C. A., Jr., Meyer, B. J., Tsang, B., & von Hurst, P. R. (2020). Inflammation (IL-1β) Modifies the Effect of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids on Core Symptoms of Autism Spectrum Disorder—An Exploratory Pilot Study . Nutrients, 12(3), 661. https://doi.org/10.3390/nu12030661







