Possible Involvement of Vitamin C in Periodontal Disease-Diabetes Mellitus Association
Abstract
1. Introduction
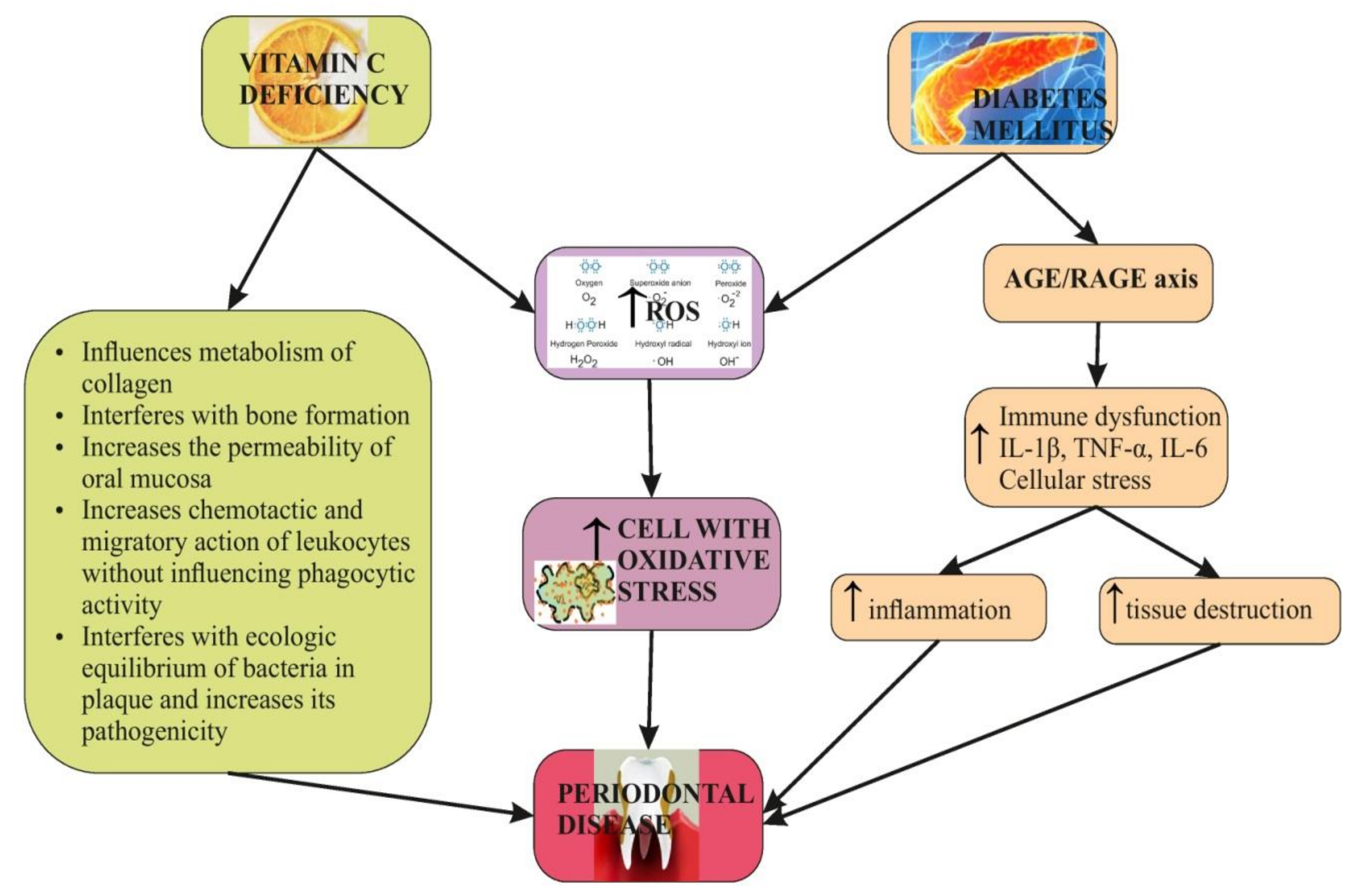
1.1. Diabetes Mellitus, Periodontal Disease and Their Interaction
1.2. Oxidative Stress and Reactive Oxygen Species—Background
1.3. Vitamin C, DM, and PD
2. Methods
2.1. Study Selection Criteria
2.2. Literature Search
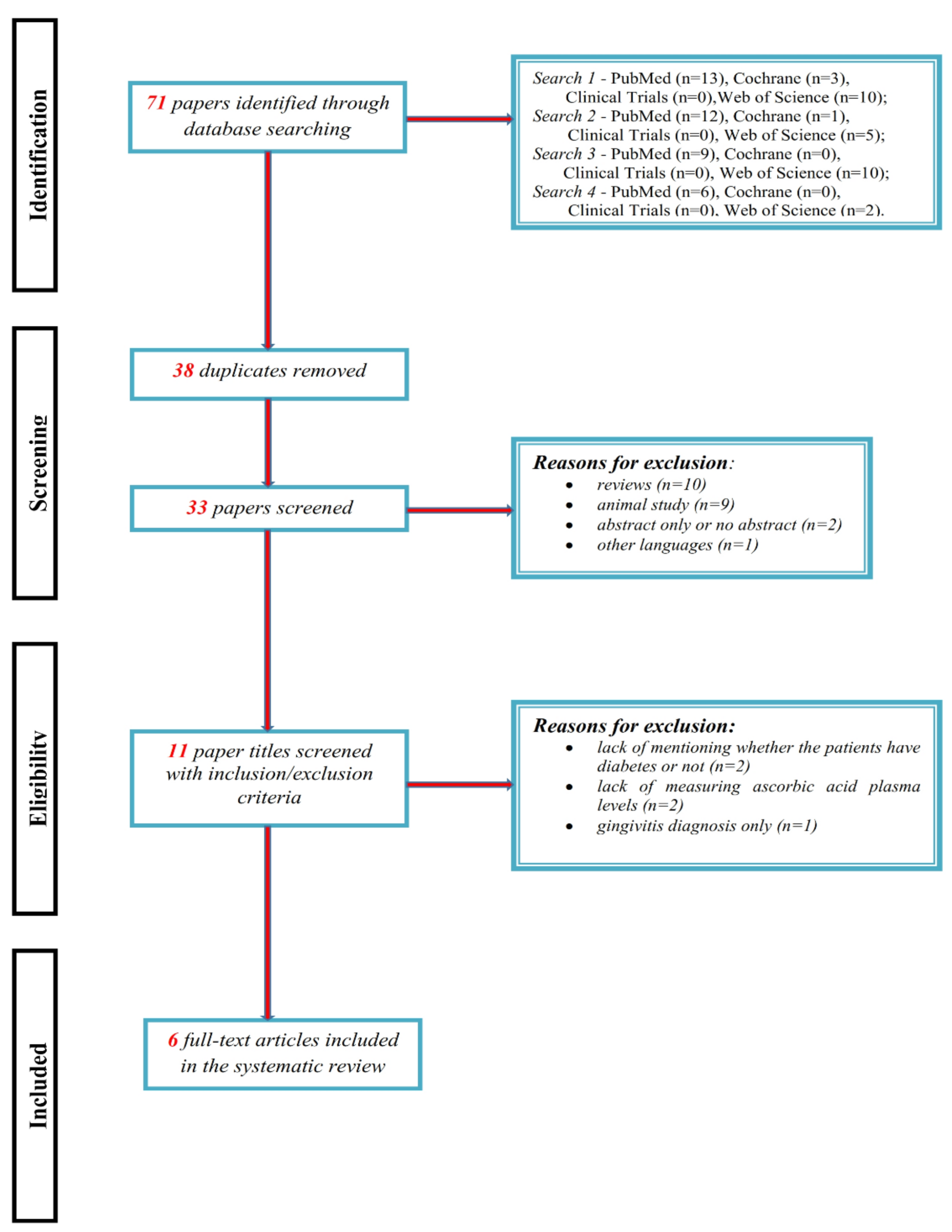
2.3. Selection of Studies
2.4. Data Extraction and Analysis
3. Results
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013, 36 (Suppl. S1), S67–S74. [Google Scholar]
- Liccardo, D.; Cannavo, A.; Spagnuolo, G.; Ferrara, N.; Cittadini, A.; Rengo, C.; Rengo, G. Periodontal Disease: A Risk Factor for Diabetes and Cardiovascular Disease. Int. J. Mol. Sci. 2019, 20, 1414. [Google Scholar] [CrossRef]
- Van der Velden, U.; Kuzmanova, D.; Chapple, I.L. Micronutritional approaches to periodontal therapy. J. Clin. Periodontol. 2011, 38 (Suppl. S11), 142–158. [Google Scholar] [CrossRef]
- Bui, F.Q.; Almeida-da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between periodontal pathogens and systemic disease. Biomed. J. 2019, 42, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Martu, M.A.; Solomon, S.M.; Toma, V.; Maftei, G.A.; Iovan, A.; Gamen, A.; Hurjui, L.; Rezus, E.; Foia, L.; Forna, N.C. The importance of cytokines in periodontal disease and rheumatoid arthritis. Review. Rom. J. Oral Rehabil. 2019, 11, 230–240. [Google Scholar]
- How, K.Y.; Song, K.P.; Chan, K.G. Porphyromonasgingivalis: An Overview of Periodontopathic Pathogen below the Gum Line. Front. Microbiol. 2016, 7, 53. [Google Scholar] [CrossRef]
- Mysak, J.; Podzimek, S.; Sommerova, P.; Lyuya-Mi, Y.; Bartova, J.; Janatova, T.; Prochazkova, J.M.; Duskova, J. Porphyromonas gingivalis:major periodontopathic pathogen overview. J. Immunol. Res. 2014, 2014, 476068. [Google Scholar] [CrossRef] [PubMed]
- Belibasakis, G.N.; Maula, T.; Bao, K.; Lindholm, M.; Bostanci, N.; Oscarsson, J.; Ihalin, R.; Johansson, A. Virulence and Pathogenicity Properties of Aggregatibacteractino mycetemcomitans. Pathogens 2019, 8, 222. [Google Scholar] [CrossRef] [PubMed]
- Zong, G.; Scott, A.E.; Griffiths, H.R.; Zockm, P.L.; Dietrich, T.; Newson, R.S. Serum alpha-Tocopherol Has a Nonlinear Inverse Association with Periodontitis among US Adults. J. Nutr. 2015, 145, 893–899. [Google Scholar] [CrossRef]
- Varela-Lopez, A.; Navarro-Hortal, M.D.; Giampieri, F.; Bullon, P.; Battino, M.; Quiles, J.L. Nutraceuticals in Periodontal Health: A Systematic Review on the Role of Vitamins in Periodontal Health Maintenance. Molecules 2018, 23, 1226. [Google Scholar] [CrossRef]
- Alagl, A.S.; Bhat, S.G. Ascorbic acid: New role of an age-old micronutrient in the management of periodontal disease in older adults. Geriatr. Gerontol. Int. 2015, 15, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes. Diabetes Care 2012, 35, S11. [Google Scholar] [CrossRef] [PubMed]
- Caton, J.; Armitage, G.; Berglundh, T.; Berglundh, T.; Chapple, I.L.C.; Jepsen, S.; Kornman, K.S.; Mealey, B.L.; Papapanou, P.N.; Sanz, M.; et al. A new classification scheme for periodontal and peri-implant diseases and conditions – Introduction and keychanges from the 1999 classification. J. Clin. Periodontol. 2018, 45 (Suppl. S20), S1–S8. [Google Scholar]
- Sanz, M.; Ceriello, A.; Buysschaert, M.; Chapple, I.; Demmer, R.T.; Graziani, F.; Herrera, D.; Jepsen, S.; Lione, L.; Madianos, P.; et al. Scientific evidence on the links between periodontal diseases and diabetes: Consensus report and guidelines of the joint workshop on periodontal diseases and diabetes by the International Diabetes Federation and the EuropeanFederation of Periodontology. J. Clin. Periodontol. 2018, 45, 138–149. [Google Scholar] [CrossRef]
- Gurav, A.; Jadhav, V. Periodontitis and risk of diabetes mellitus. J. Diabetes 2011, 3, 21–28. [Google Scholar] [CrossRef]
- Demmer, R.T.; Jacobs, D.R., Jr.; Desvarieux, M. Periodontal disease and incident type 2 diabetes: Results from the First National Health and Nutrition Examination Survey and its epidemiologic follow-up study. Diabetes Care 2008, 31, 1373–1379. [Google Scholar] [CrossRef]
- Huang, J.; Xiao, Y.; Xu, A.; Zhou, Z. Neutrophils in type 1 diabetes. J. Diabetes Investig. 2016, 7, 652–663. [Google Scholar] [CrossRef]
- Olteanu, M.; Surlin, P.; Oprea, B.; Rauten, A.M.; Popescu, R.M.; Niţu, M.; Camen, G.C.; Caraivan, O. Gingival inflammatory infiltrate analysis in patients with chronic periodontitis and diabetes mellitus. Rom. J. Morphol. Embryol. 2011, 52, 1311–1317. [Google Scholar]
- Șurlin, P.; Camen, A.; Stratul, S.I.; Roman, A.; Gheorghe, D.N.; Herăscu, E.; Osiac, E.; Rogoveanu, I. Optical coherence tomography assessment of gingival epithelium inflammatory status in periodontal—Systemic affected patients. Ann. Anat. 2018, 219, 51–56. [Google Scholar] [CrossRef]
- Wendt, T.; Tanji, N.; Guo, J.; Hudson, B.I.; Bierhaus, A.; Ramasamy, R.; Arnold, B.; Nawroth, P.P.; Yan, S.F.; D’Agati, V.; et al. Glucose, glycation, and RAGE: Implications for amplification of cellular dysfunction in diabetic nephropathy. J. Am. Soc. Nephrol. 2003, 14, 1383–1395. [Google Scholar] [CrossRef] [PubMed]
- Rafatjou, R.; Razavi, Z.; Tayebi, S.; Khalili, M.; Farhadian, M. Dental Health Status and Hygiene in Children and Adolescents with Type 1 Diabetes Mellitus. J. Res. Health Sci. 2016, 16, 122–126. [Google Scholar] [PubMed]
- Mirza, B.A.; Syed, A.; Izhar, F.; Ali Khan, A. Bidirectional relationship between diabetes and periodontal disease: Review of evidence. J. Pak. Med. Assoc. 2010, 60, 766–768. [Google Scholar] [PubMed]
- Mizuno, H.; Ekuni, D.; Maruyama, T.; Kataoka, K.; Yoneda, T.; Fukuhara, D.; Sugiura, Y.; Tomofuji, T.; Wada, J.; Morita, M. The effects of non-surgical periodontal treatment on glycemic control, oxidative stress balance and quality of life in patients with type 2 diabetes: A randomized clinical trial. PLoS ONE 2017, 12, e0188171. [Google Scholar] [CrossRef] [PubMed]
- Dağ, A.; Firat, E.T.; Arikan, S.; Kadiroğlu, A.K.; Kaplan, A. The effect of periodontal therapy on serum TNF-alpha and HbA1c levels in type 2 diabetic patients. Aust. Dent. J. 2009, 54, 17–22. [Google Scholar] [CrossRef]
- Li, Q.; Hao, S.; Fang, J.; Xie, J.; Kong, X.H.; Yang, J.X. Effect of non-surgical periodontal treatment on glycemic control of patients with diabetes: A meta-analysis of randomized controlled trials. Trials 2015, 16, 291. [Google Scholar] [CrossRef]
- Isola, G.; Matarese, G.; Ramaglia, L.; Pedullà, E.; Rapisarda, E.; Iorio-Siciliano, V. Association between periodontitis and glycosylated haemoglobin before diabetes onset: A cross-sectional study. Clin. Oral Investig. 2019. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef]
- Ray, R.; Shah, A.M. NADPH oxidase and endothelial cell function. Clin. Sci. 2005, 109, 217–226. [Google Scholar] [CrossRef]
- Griendling, K.K.; FitzGerald, G.A. Oxidative stress and cardiovascular injury: Part I: Basic mechanisms and in vivo monitoring of ROS. Circulation 2003, 108, 1912–1916. [Google Scholar] [CrossRef]
- Ebrahimian, T.; Li, M.W.; Lemarie, C.A.; Simeone, S.M.; Pagano, P.J.; Gaestel, M.; Paradis, P.; Wassmann, S.; Schiffrin, E.L. Mitogen-activated protein kinase-activated protein kinase 2 in angiotensin II-induced inflammation and hypertension: Regulation of oxidative stress. Hypertension 2011, 57, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Yamamoto, M. Molecular mechanisms activating the Nrf2-Keap1 pathway of antioxidant gene regulation. Antioxid. Redox Signal. 2005, 7, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Burke-Gaffney, A.; Evans, T.W. Lest we forget the endothelial glycocalyx in sepsis. Crit. Care 2012, 16, 121. [Google Scholar] [CrossRef] [PubMed]
- Gumuz, M.P.; Kanmaz, B.; Buduneli, M. The role of oxidative stress in the interaction of periodontal disease with systemic diseases or conditions. Oxid. Antioxid. Med. Sci. 2016, 5, 33–38. [Google Scholar] [CrossRef]
- Kinane, D.F.; Preshaw, P.M.; Loos, B.G. Working Group 2 of Seventh European Workshop on P. Host-response: Understanding the cellular and molecular mechanisms of host-microbial interactions—consensus of the Seventh European Workshop on Periodontology. J. Clin. Periodontol. 2011, 38 (Suppl. S11), 44–48. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.; Bereswill, S.; Heimesaat, M.M. Immunomodulatory and Antimicrobial Effects of Vitamin C. Eur. J. Microbiol. Immunol. 2019, 9, 73–79. [Google Scholar] [CrossRef]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral in takes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (A review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Tveden-Nyborg, P. The Pharmacokinetics of Vitamin C. Nutrients 2019, 11, 2412. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Chalker, E. Vitamin C Can Shorten the Length of Stayin the ICU: A Meta-Analysis. Nutrients 2019, 11, 708. [Google Scholar] [CrossRef] [PubMed]
- Liugan, M.; Carr, A.C. Vitamin C and Neutrophil Function: Findings from Randomized Controlled Trials. Nutrients 2019, 11, 2102. [Google Scholar] [CrossRef] [PubMed]
- Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef]
- Wang, Y.; Chun, O.K.; Song, W.O. Plasma and dietary antioxidant status as cardiovascular disease risk factors: A review of human studies. Nutrients 2013, 5, 2969–3004. [Google Scholar] [CrossRef]
- Shaik-Dasthagirisaheb, Y.B.; Varvara, G.; Murmura, G.; Saggini, A.; Caraffa, A.; Antinolfi, P.; Tete, S.; Tripodi, D.; Conti, F.; Cianchetti, E.; et al. Role of vitamins D, E and C in immunity and inflammation. J. Biol. Regul. Homeost. Agents 2013, 27, 291–295. [Google Scholar]
- Sailaja Devi, M.M.; Das, U.N. Effect of prostaglandins against alloxan-induced diabetes mellitus. Prostaglandins Leukot. Essent. Fat. Acids 2006, 74, 39–60. [Google Scholar] [CrossRef]
- Das, U.N. Vitamin C for Type 2 Diabetes Mellitus and Hypertension. Arch. Med Res. 2019, 50, 11–14. [Google Scholar] [CrossRef]
- Mason, S.A.; Rasmussen, B.; van Loon, L.J.C.; Salmon, J.; Wadley, G.D. Ascorbic acid supplementation improves postprandial glycaemic control and blood pressure in individuals with type 2 diabetes: Findings of a randomized cross-over trial. Diabetes Obes. Metab. 2019, 21, 674–682. [Google Scholar] [CrossRef]
- Asmat, U.; Abad, K.; Ismail, K. Diabetes mellitus and oxidative stress-A concise review. Saudi Pharm. J. 2016, 24, 547–553. [Google Scholar] [CrossRef]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B., 3rd. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O. Management of diabetes mellitus: Could simultaneous targeting of hyperglycemia and oxidative stress be a better panacea? Int. J. Mol. Sci. 2012, 13, 2965–2972. [Google Scholar] [CrossRef] [PubMed]
- Nishida, M.; Grossi, S.G.; Dunford, R.G.; Ho, A.W.; Trevisan, M.; Genco, R.J. Dietary vitamin C and the risk for periodontal disease. J. Periodontol. 2000, 71, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Kuzmanova, D.; Jansen, I.D.; Schoenmaker, T.; Nazmi, K.; Teeuw, W.J.; Bizzarro, S.; Loos, B.G.; van der Velden, U. Vitamin C in plasma and leucocytes in relation to periodontitis. J. Clin. Periodontol. 2012, 39, 905–912. [Google Scholar] [CrossRef]
- Petti, S.; Cairella, G.; Tarsitani, G. Nutritional variables related to gingival health in adolescent girls. Community Dent. Oral Epidemiol. 2000, 28, 407–413. [Google Scholar] [CrossRef]
- Tada, A.; Miura, H. The Relationship between Vitamin C and Periodontal Diseases: A Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 2472. [Google Scholar] [CrossRef]
- Monea, A.; Mezei, T.; Popsor, S.; Monea, M. Oxidative Stress: A Link between Diabetes Mellitus and Periodontal Disease. Int. J. Endocrinol. 2014, 2014, 917631. [Google Scholar] [CrossRef]
- Li, X.; Sun, X.; Zhang, X.; Mao, Y.; Ji, Y.; Shi, L.; Cai, W.; Wang, P.; Wu, G.; Gan, X.; et al. Enhanced Oxidative Damage and Nrf2 Downregulation Contribute to the Aggravation of Periodontitis by Diabetes Mellitus. Oxidative Med. Cell. Longev. 2018, 2018, 9421019. [Google Scholar] [CrossRef]
- Lee, J.H.; Shin, M.S.; Kim, E.J.; Ahn, Y.B.; Kim, H.D. The association of dietary vitamin C intake with periodontitis among Korean adults: Results from KNHANES. PLoS ONE 2017, 12, e0177074. [Google Scholar] [CrossRef]
- Dietrich, T.; Kaye, E.K.; Nunn, M.E.; Van Dyke, T.; Garcia, R.I. Gingivitis susceptibility and its relation to periodontitis in men. J. Dent. Res. 2006, 85, 1134–1137. [Google Scholar] [CrossRef]
- Isola, G.; Polizzi, A.; Muraglie, S.; Leonardi, R.; Lo Giudice, A. Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease. Nutrients 2019, 11, 2956. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Gokhale, N.H.; Acharya, A.B.; Patil, V.S.; Trivedi, D.J.; Thakur, S.L. A Short-Term Evaluation of the Relationship Between Plasma Ascorbic Acid Levels and Periodontal Disease in Systemically Healthy and Type 2 Diabetes Mellitus Subjects. J. Diet. Suppl. 2013, 10, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Kunsongkeit, P.; Okuma, N.; Rassameemasmaung, S.; Chaivanit, P. Effect of vitamin C as an adjunct in nonsurgical periodontal therapy in uncontrolled type 2 diabetes mellitus patients. Eur. J. Dent. 2019, 13, 444–449. [Google Scholar] [CrossRef][Green Version]
- Thomas, B.; Kumari, S.; Ramitha, K.; Ashwini Kumari, M.B. Ashwini Kumari, Comparative evaluation of micronutrient status in the serum of diabetes mellitus patients and healthy individuals with periodontitis. J. Indian Soc. Periodontol. 2010, 14, 46–49. [Google Scholar] [CrossRef]
- Gümüş, P.; Buduneli, N.; Cetinkalp, S.; Hawkins, S.I.; Renaud, D.; Kinane, D.F.; Scott, D.A. Salivary antioxidants in patients with type 1 or 2 diabetes and inflammatory periodontal disease: A case-control study. J. Periodontol. 2009, 80, 1440–1446. [Google Scholar] [CrossRef]
- Patil, V.S.; Patil, V.P.; Gokhale, N.; Acharya, A.; Kangokar, P. Chronic periodontitis in type 2 diabetes mellitus: Oxidative stress as a common factor in periodontal tissue injury. J. Clin. Diagn. Res. 2016, 10, 12–16. [Google Scholar] [CrossRef]
- Amaliya, A.; Laine, M.L.; Delanghe, J.R.; Loos, B.G.; Van Wijk, A.J.; Van der Velden, U. Javaproject on periodontal diseases. Periodontal bone loss in relation to environmental to environmental and systemic conditions. J. Clin. Periodontol. 2015, 42, 325–332. [Google Scholar] [CrossRef]
- Bogdan, M.; Silosi, I.; Surlin, P.; Tica, A.A.; Tica, O.S.; Balseanu, T.A.; Rauten, A.M.; Camen, A. Salivary and serum biomarkers for the study of side effects of aripiprazole coprescribed with mirtazapine in rats. Int. J. Clin. Exp. Med. 2015, 8, 8051–8059. [Google Scholar]
- Gariballa, S. Poor vitamin C status is associated with increased depression symptoms following acute illness in older people. Int. J. Vitam. Nutr. Res. 2014, 84, 12–17. [Google Scholar] [CrossRef]
- Kocot, J.; Luchowska-Kocot, D.; Kielczykowska, M.; Musik, I.; Kurzepa, J. Does Vitamin C Influence Neurodegenerative Diseases and Psychiatric Disorders? Nutrients 2017, 9, 659. [Google Scholar] [CrossRef] [PubMed]
- Salinero-Fort, M.A.; Gómez-Campelo, P.; San Andrés-Rebollo, F.J.; Cárdenas-Valladolid, J.; Abánades-Herranz, J.C.; Carrillo de Santa Pau, E.; Chico-Moraleja, R.M.; Beamud-Victoria, D.; de Miguel-Yanes, J.M.; Jimenez-Garcia, R.; et al. Prevalence of depression in patients with type 2 diabetes mellitus in Spain (the DIADEMA Study): Results from the MADIABETES cohort. BMJ Open 2018, 8, e020768. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.H.; Park, S.G. The relationship between depression and periodontal diseases. Community Dent. Health 2018, 35, 23–29. [Google Scholar] [PubMed]
- Ward, M.S.; Lamb, J.; May, J.M.; Harrison, F.E. Behavioral and monoamine changes following severe vitamin C deficiency. J. Neurochem. 2013, 124, 363–375. [Google Scholar] [CrossRef]
- Moretti, M.; Colla, A.; de Oliveira Balen, G.; dos Santos, D.B.; Budni, J.; de Freitas, A.E.; Farina, M.; Severo Rodrigues, A.L. Ascorbic acid treatment, similarly to fluoxetine, reverses depressive-like behavior and brain oxidative damage induced by chronic unpredictable stress. J. Psychiatr. Res. 2012, 46, 331–340. [Google Scholar] [CrossRef]
- Badescu, S.V.; Tataru, C.; Kobylinska, L.; Georgescu, E.L.; Zahiu, D.M.; Zagrean, A.M.; Zagrean, L. The association between Diabetes mellitus and Depression. J. Med. Life 2016, 9, 120–125. [Google Scholar]
- Fatima, Z.; Bey, A.; Azmi, S.A.; Gupta, N.D.; Khan, A. Mental depression as a risk factor for periodontal disease: A case-control study. Gen. Dent. 2016, 5, 86–89. [Google Scholar] [CrossRef]
- Penmetsa, G.S.; Seethalakshmi, P. Effect of stress, depression, and anxiety over periodontal health indicators among health professional students. J. Indian Assoc. Public Health Dent. 2019, 17, 36–40. [Google Scholar]
- Nascimento, G.G.; Gastal, M.T.; Leite, F.R.M.; Quevedo, L.A.; Peres, K.G.; Peres, M.A.; Horta, B.L.; Barros, F.C.; Demarco, F.F. Is there an association between depression and periodontitis? A birth cohort study. J. Clin. Periodontol. 2019, 46, 31–39. [Google Scholar] [CrossRef]
- Turcu-Stiolica, A.; Taerel, A.E.; Turcu-Stiolica, R. Identifying and measuring compliance and adherence in antidepressants taking. Procedia Econ. Financ. 2014, 15, 836–839. [Google Scholar] [CrossRef]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B.; Lowe, B. The Patient Health Questionnaire Somatic, Anxiety, and Depressive Symptom Scales: A systematic review. Gen. Hosp. Psychiatry 2010, 32, 345–359. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Kathariya, R.; Bansal, S.; Singh, A.; Shahakar, D. Dietary antioxidants and their indispensable role in periodontal health. J. Food Drug Anal. 2016, 24, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Muniz, F.W.; Nogueira, S.B.; Mendes, F.L.; Rösing, C.K.; Moreira, M.M.; de Andrade, G.M.; Carvalho Rde, S. The impact of antioxidant agents complimentary to periodontal therapy on oxidative stress and periodontal outcomes: A systematic review. Arch. Oral Biol. 2015, 60, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
| Reference | Study Type | Country | Participant Characteristics (Study Sample) | Periodontal Status | Measurement of Periodontal Status | Intervention | Diabetes Status | Measurement of Diabetes | ||
|---|---|---|---|---|---|---|---|---|---|---|
| No. | Age | Gender | ||||||||
| Gumus, 2009 [66] | CC | Turkey | 65 | 17–73 years old | M and F | 16 patients with type 1 DM (5 M, 11F, age: 17–73), 25 patients with type 2 (11 M, 14F, age: 42–69) and 24 systematically healthy (control group, 10 M, 14 F, age: 22–60), all with PD | plaque-induced inflammatory PD, non-aggressive. PII, PB, gingival recession, CAL, BOP, recorded for 6 sites per tooth. | full-mouth periodontal clinical measurements | type 1 or type 2 | FBS, HbA1C, and diabetes complications |
| Thomas, 2010 [65] | CC | India | 60 | adults | M and F | 3 groups: group 1—20 patients with type 2 DM and PD, group 2—20 healthy patients with PD and group 3—20 healthy patients without PD | CAL measured with a Williams periodontal probe and BOP | examinations | type 2 | RBS, FBS |
| Gokhale, 2013 [63] | RS | India | 120 | 30–60 years old | M and F | 4 groups of 30 patients each group 1: no PD, group 2: chronic gingivitis, group 3: chronic periodontitis, group 4: chronic periodontitis and recently diagnosed type 2 diabetes; randomized subjects of groups 2–4, were grouped to receive SRP with dietary supplementation of ascorbic acid for 2 weeks or only SRP; chronic periodontitis—PPD and assessment of gingival bleeding | PII, SBI for gingivitis, PPDs for chronic periodontitis, gingival bleeding | randomized subjects within groups 2–4, divided to receive either SRP or not | type 2 | CPG, FBS, PPG |
| Amaliya, 2015 [68] | CH | Indonesia | 98 | 39–50 years old | 45 M and 53 F | remote populations deprived of oral health care—natural development of periodontitis; subjects of this population showed a mean of 30% ABL in their dentition, ranging from 19% to 54% | dental radiographs, ABL, periapical radiologic transparency | examinations | a small number of subjects (70% in prediabetic state and 6% having undiagnosed diabetes) with HbA1c values ≥6.5% | HbA1C |
| Patil, 2016 [67] | CS | India | 100 | adults | M and F | 4 groups: 25 normal healthy controls, 25 gingivitis patients, 25 chronic periodontitis patients, 25 chronic periodontitis and type 2 diabetes | BOP, SBI, PPD | examinations | type 2 | FBS, PPG |
| Kunsongkeit, 2019 [64] | Double-blind, placebo-controlled, clinical trial | Thailand | 31 | 43–72 years old | 9 M and 22 F | moderate chronic periodontitis, 2 groups: n = 15 who received periodontal therapy and vitamin C for 2 months and n = 16 who received periodontal therapy and placebo | PII, SBI, gingival index, PPD | full SRP and examinations | type 2 uncontrolled (FBS > 150 mg/dL, HbA1c > 7%) | FBS, HbA1C |
| Reference | Duration | Experimental Design | Measurement of Vitamin C | Main Results | ||
|---|---|---|---|---|---|---|
| Dosage | Administration | Frequency | ||||
| Gumus, 2009 [66] | 2 and 1/2 years | none | none | none | measurement of antioxidants’ salivary concentrations in whole saliva samples | Subjects with type 2 DM had fewer teeth and more sites with probing depths (>4 mm) than patients with type 1 DM. Despite this, total antioxidant capacity and vitamin C concentrations did not seem to play a major role in the pathogenesis of periodontitis correlated with DM. |
| Thomas, 2010 [65] | Not mentioned | none | none | none | venous blood samples collected | Diabetic patients with periodontitis revealed a significant decrease in vitamin C levels. |
| Gokhale, 2013 [63] | 4 months | 450 mg | subgroups randomly divided using a coin-toss method: subgroup A (15) 450 mg chewable tablet and subgroup B (15) placebo chewable tablet | daily intake for 2 weeks or only SRP | plasma measurement | Plasma measured AAL were below the normal range in systemically healthy subjects with gingivitis and diabetics with periodontitis. Dietary AA supplementation associated with SRP improved the SBI in patients with gingivitis and PD–DM. |
| Amaliya, 2015 [68] | 1 year | food products categorized as high (>60 mg), fair (31–60 mg), low (2–30 mg) or no vitamin C (<2 mg vitamin C/100 g) | Number of guava fruit servings | food frequency taken in the last month | plasma measurement, based on the values provided by the National Nutrient Database for standard reference | 45% of the participants showed vitamin C depletion/deficiency, 70% were in a prediabetic state, 6% had untreated diabetes. Still, it has been shown that guava fruit consumption might have played a protective role against periodontitis in a malnourished population, regarding the extent and severity of ABL (at least 10% of the participants had a low BMI and were considered as malnourished). |
| Patil, 2016 [67] | 1 year | none | none | none | plasma measurement | A significant decrease in vitamin C was observed in the diabetic periodontitis group as compared with healthy control groups. Type 2 diabetic subjects revealed excessive ROS concentration, therefore more periodontal tissue destruction. |
| Kunsongkeit, 2019 [64] | 2 months | 500 mg | tablets | daily for 2 months | plasma measurement | Periodontitis patients with uncontrolled type 2 DM did not have evident benefits by supplementation of 500 mg/day vitamin C. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogdan, M.; Meca, A.D.; Boldeanu, M.V.; Gheorghe, D.N.; Turcu-Stiolica, A.; Subtirelu, M.-S.; Boldeanu, L.; Blaj, M.; Botnariu, G.E.; Vlad, C.E.; et al. Possible Involvement of Vitamin C in Periodontal Disease-Diabetes Mellitus Association. Nutrients 2020, 12, 553. https://doi.org/10.3390/nu12020553
Bogdan M, Meca AD, Boldeanu MV, Gheorghe DN, Turcu-Stiolica A, Subtirelu M-S, Boldeanu L, Blaj M, Botnariu GE, Vlad CE, et al. Possible Involvement of Vitamin C in Periodontal Disease-Diabetes Mellitus Association. Nutrients. 2020; 12(2):553. https://doi.org/10.3390/nu12020553
Chicago/Turabian StyleBogdan, Maria, Andreea Daniela Meca, Mihail Virgil Boldeanu, Dorin Nicolae Gheorghe, Adina Turcu-Stiolica, Mihaela-Simona Subtirelu, Lidia Boldeanu, Mihaela Blaj, Gina Eosefina Botnariu, Cristiana Elena Vlad, and et al. 2020. "Possible Involvement of Vitamin C in Periodontal Disease-Diabetes Mellitus Association" Nutrients 12, no. 2: 553. https://doi.org/10.3390/nu12020553
APA StyleBogdan, M., Meca, A. D., Boldeanu, M. V., Gheorghe, D. N., Turcu-Stiolica, A., Subtirelu, M.-S., Boldeanu, L., Blaj, M., Botnariu, G. E., Vlad, C. E., Foia, L. G., & Surlin, P. (2020). Possible Involvement of Vitamin C in Periodontal Disease-Diabetes Mellitus Association. Nutrients, 12(2), 553. https://doi.org/10.3390/nu12020553










