Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence
Abstract
1. Background
2. Methods
3. Results
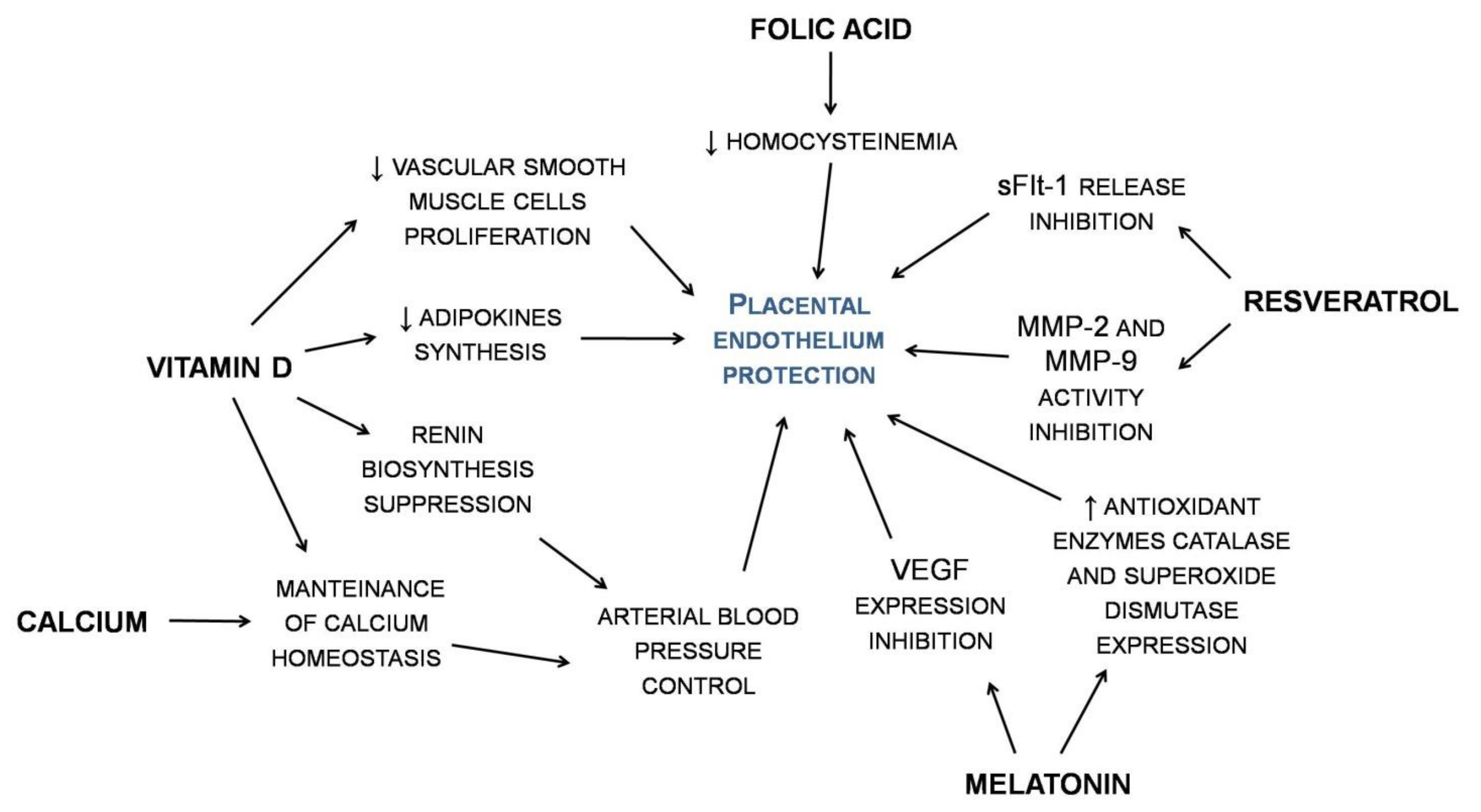
3.1. Calcium
3.2. Vitamin D
3.3. Folic Acid
3.4. Resveratrol
3.5. Sodium and Potassium
3.6. Zinc
3.7. Melatonin
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Villar, J.; Carroli, G.; Wojdyla, D.; Abalos, E.; Giordano, D.; Ba’aqeel, H.; Farnot, U.; Bergsjø, P.; Bakketeig, L.; Lumbiganon, P.; et al. Preeclampsia, gestational hypertension and intrauterine growth restriction, related or independent conditions? Am. J. Obstet. Gynecol. 2006, 94, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Hutcheon, J.A.; Lisonkova, S.; Joseph, K.S. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 391–403. [Google Scholar] [CrossRef] [PubMed]
- LeFevre, N.; Krumm, E. Antihypertensive Drug Therapy for Mild to Moderate Hypertension During Pregnancy. Am. Fam. Physician 2019, 100, 403–405. [Google Scholar] [PubMed]
- Regitz-Zagrosek, V.; Roos-Hesselink, J.W.; Bauersachs, J.; Blomström-Lundqvist, C.; Cífková, R.; De Bonis, M.; Iung, B.; Johnson, M.R.; Kintscher, U.; Kranke, P.; et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. Eur. Heart J. 2018, 39, 3165–3241. [Google Scholar] [CrossRef] [PubMed]
- ACOG. Practice Bulletin No. 202: Gestational Hypertension and Preeclampsia. Obstet. Gynecol. 2019, 133, e1–e25. [Google Scholar] [CrossRef]
- Poon, L.C.; Shennan, A.; Hyett, J.A.; Kapur, A.; Hadar, E.; Divakar, H.; McAuliffe, F.; da Silva Costa, F.; von Dadelszen, P.; McIntyre, H.D.; et al. The International Federation of Gynecology and Obstetrics (FIGO) initiative on pre-eclampsia: A pragmatic guide for first-trimester screening and prevention. Int. J. Gynaecol. Obstet. 2019, 145 (Suppl. S1), 1–33. [Google Scholar] [CrossRef]
- Ding, J.; Kang, Y.; Fan, Y.; Chen, Q. Efficacy of resveratrol to supplement oral nifedipine treatment in pregnancy-induced preeclampsia. Endocr. Connect. 2017, 6, 595–600. [Google Scholar] [CrossRef]
- Guideline: Calcium Supplementation in Pregnant Women; World Health Organization: Geneva, Switzerland, 2013.
- Hanzlik, R.P.; Fowler, S.C.; Fisher, D.H. Relative bioavailability of calcium from calcium formate, calcium citrate, and calcium carbonate. J. Pharmacol. Exp. Ther. 2005, 313, 1217–1222. [Google Scholar] [CrossRef]
- Khanam, F.; Hossain, B.; Mistry, S.K.; Mitra, D.K.; Raza, W.A.; Rifat, M.; Afsana, K.; Rahman, M. The association between daily 500 mg calcium supplementation and lower pregnancy-induced hypertension risk in Bangladesh. BMC Pregnancy Childbirth 2018, 18, 406. [Google Scholar] [CrossRef]
- Hofmeyr, G.J.; Lawrie, T.A.; Atallah, Á.N.; Torloni, M.R. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst. Rev. 2018, 10, CD001059. [Google Scholar] [CrossRef]
- Sun, X.; Li, H.; He, X.; Li, M.; Yan, P.; Xun, Y.; Lu, C.; Yang, K.; Zhang, X. The association between calcium supplement and preeclampsia and gestational hypertension: A systematic review and meta-analysis of randomized trials. Hypertens. Pregnancy 2019, 38, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Bortolotto, M.R.; Francisco, R.P.V.; Zugaib, M. Resistant Hypertension in Pregnancy: How to Manage? Curr. Hypertens. Rep. 2018, 20, 63. [Google Scholar] [CrossRef] [PubMed]
- Cormick, G.; Ciapponi, A.; Cafferata, M.L.; Belizán, J.M. Calcium supplementation for prevention of primary hypertension. Cochrane Database Syst. Rev. 2015, CD010037. [Google Scholar] [CrossRef] [PubMed]
- Jarjou, L.M.; Laskey, M.A.; Sawo, Y.; Goldberg, G.R.; Cole, T.J.; Prentice, A. Effect of calcium supplementation in pregnancy on maternal bone outcomes in women with a low calcium intake. Am. J. Clin. Nutr. 2010, 92, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, B.J.; Taylor, B.J.; de Lange, M.; Harper, M.J.; Jones, S.; Mekhail, A.; Houghton, L.A. A Longitudinal Study of 25-Hydroxy Vitamin D and Parathyroid Hormone Status throughout Pregnancy and Exclusive Lactation in New Zealand Mothers and Their Infants at 45° S. Nutrients 2018, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Karras, S.N.; Wagner, C.L.; Angeloudi, E.; Kotsa, K. Maternal vitamin D status during pregnancy in Europe: The two sides of the story. Eur. J. Nutr. 2017, 56, 2207–2208. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.D.; Wang, Y.; Guo, J.J.; Zhou, L.; Wang, N. Vitamin D Enhances Efficacy of Oral Nifedipine in Treating Preeclampsia with Severe Features: A Double Blinded, Placebo-Controlled and Randomized Clinical Trial. Front. Pharm. 2017, 8, 865. [Google Scholar] [CrossRef]
- Fogacci, S.; Fogacci, F.; Banach, M.; Michos, E.D.; Hernandez, A.V.; Lip, G.Y.H.; Blaha, M.J.; Toth, P.P.; Borghi, C.; Cicero, A.F.G.; et al. Vitamin D supplementation and incident preeclampsia: A systematic review and meta-analysis of randomized clinical trials. Clin. Nutr. 2019, S0261, in press. [Google Scholar] [CrossRef]
- Evans, K.N.; Bulmer, J.N.; Kilby, M.D.; Hewison, M. Vitamin D and placental-decidual function. J. Soc. Gynecol. Investig. 2004, 11, 263–271. [Google Scholar] [CrossRef]
- Dinca, M.; Serban, M.C.; Sahebkar, A.; Mikhailidis, D.P.; Toth, P.P.; Martin, S.S.; Blaha, M.J.; Blüher, M.; Gurban, C.; Penson, P.; et al. Does vitamin D supplementation alter plasma adipokines concentrations? A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2016, 107, 360–371. [Google Scholar] [CrossRef]
- Walker, M.C.; Finkelstein, S.A.; Rennicks White, R.; Shachkina, S.; Smith, G.N.; Wen, S.W.; Rodger, M. The Ottawa and Kingston (OaK) Birth Cohort: Development and achievements. J. Obs. Gynaecol. Can. 2011, 33, 1124–1133. [Google Scholar] [CrossRef]
- Di Simone, N.; Riccardi, P.; Maggiano, N.; Piacentani, A.; D’Asta, M.; Capelli, A.; Caruso, A. Effect of folic acid on homocysteine-induced trophoblast apoptosis. Mol. Hum. Reprod. 2004, 10, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Merchant, A.T.; Msamanga, G.; Villamor, E.; Saathoff, E.; O’brien, M.; Hertzmark, E.; Hunter, D.J.; Fawzi, W.W. Multivitamin supplementation of HIV-positive women during pregnancy reduces hypertension. J. Nutr. 2005, 135, 1776–1781. [Google Scholar] [CrossRef]
- Wen, S.W.; Zhou, J.; Yang, Q.; Fraser, W.; Olatunbosun, O.; Walker, M. Maternal exposure to folic acid antagonists and placenta-mediated adverse pregnancy outcomes. CMAJ 2008, 179, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liu, C.; Wang, Q.; Zhang, Z. Supplementation of folic acid in pregnancy and the risk of preeclampsia and gestational hypertension: A meta-analysis. Arch. Gynecol. Obstet. 2018, 298, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.W.; White, R.R.; Rybak, N.; Gaudet, L.M.; Robson, S.; Hague, W.; Simms-Stewart, D.; Carroli, G.; Smith, G.; Fraser, W.D.; et al. Effect of high dose folic acid supplementation in pregnancy on pre-eclampsia (FACT): Double blind, phase III, randomised controlled, international, multicentre trial. BMJ 2018, 362, k3478. [Google Scholar] [CrossRef] [PubMed]
- Fogacci, F.; Tocci, G.; Presta, V.; Fratter, A.; Borghi, C.; Cicero, A.F.G. Effect of resveratrol on blood pressure: A systematic review and meta-analysis of randomized, controlled, clinical trials. Crit. Rev. Food Sci. Nutr. 2019, 59, 1605–1618. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Fogacci, F.; Colletti, A. Food and plant bioactives for reducing cardiometabolic disease risk: An evidence based approach. Food Funct. 2017, 8, 2076–2088. [Google Scholar] [CrossRef]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Sridharan, K.; Sequeira, R.P. Drugs for treating severe hypertension in pregnancy: A network meta-analysis and trial sequential analysis of randomized clinical trials. Br. J. Clin. Pharmacol. 2018, 84, 1906–1916. [Google Scholar] [CrossRef]
- Cudmore, M.J.; Ramma, W.; Cai, M.; Fujisawa, T.; Ahmad, S.; Al-Ani, B.; Ahmed, A. Resveratrol inhibits the release of soluble fms-like tyrosine kinase (sFlt-1) from human placenta. Am. J. Obstet. Gynecol. 2012, 206, e10–e15. [Google Scholar] [CrossRef]
- Pandey, A.K.; Bhattacharya, P.; Shukla, S.C.; Paul, S.; Patnaik, R. Resveratrol inhibits matrix metalloproteinases to attenuate neuronal damage in cerebral ischemia: A molecular docking study exploring possible neuroprotection. Neural. Regen. Res. 2015, 10, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Palei, A.C.; Granger, J.P.; Tanus-Santos, J.E. Matrix metalloproteinases as drug targets in preeclampsia. Curr. Drug. Targets 2013, 14, 325–334. [Google Scholar] [PubMed]
- Yılmaz, Z.V.; Akkaş, E.; Türkmen, G.G.; Kara, Ö.; Yücel, A.; Uygur, D. Dietary sodium and potassium intake were associated with hypertension, kidney damage and adverse perinatal outcome in pregnant women with preeclampsia. Hypertens. Pregnancy 2017, 36, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Birukov, A.; Andersen, L.B.; Herse, F.; Rakova, N.; Kitlen, G.; Kyhl, H.B.; Golic, M.; Haase, N.; Kräker, K.; Müller, D.N.; et al. Aldosterone, Salt, and Potassium Intakes as Predictors of Pregnancy Outcome, Including Preeclampsia. Hypertension 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Arvizu, M.; Bjerregaard, A.A.; Madsen, M.T.; Granström, C.; Halldorsson, T.I.; Olsen, S.F.; Gaskins, A.J.; Rich-Edwards, J.W.; Rosner, B.A.; Chavarro, J.E. Sodium Intake during Pregnancy, but Not Other Diet Recommendations Aimed at Preventing Cardiovascular Disease, Is Positively Related to Risk of Hypertensive Disorders of Pregnancy. J. Nutr. 2019, nxz197, in press. [Google Scholar] [CrossRef]
- Asayama, K.; Imai, Y. The impact of salt intake during and after pregnancy. Hypertens. Res. 2018, 41, 1–5. [Google Scholar] [CrossRef]
- Gallery, E.D.; Brown, M.A. Control of sodium excretion in human pregnancy. Am. J. Kidney Dis. 1987, 9, 290–295. [Google Scholar] [CrossRef]
- He, L.; Lang, L.; Li, Y.; Liu, Q.; Yao, Y. Comparison of serum zinc, calcium, and magnesium concentrations in women with pregnancy-induced hypertension and healthy pregnant women: A meta-analysis. Hypertens. Pregnancy 2016, 35, 202–209. [Google Scholar] [CrossRef]
- Food and Nutrition Board: Institute of Medicine. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; The National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Caulfield, L.E.; Black, R.E. Zinc deficiency. In Comparative Quantification of Health Risks: Global and Regional Burden of Disease Attributable to Selected Major Risk Factors Geneva; World Health Organization: Geneva, Switzerland, 2004; pp. 257–279. [Google Scholar]
- National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand; NHMRC: Canberra, Australia, 2005. [Google Scholar]
- Caulfield, L.E.; Zavaleta, N.; Shankar, A.H.; Merialdi, M. Potential contribution of maternal zinc supplementation during pregnancy to maternal and child survival. Am. J. Clin. Nutr. 1998, 68, 499S–508S. [Google Scholar] [CrossRef]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef]
- Ota, E.; Mori, R.; Middleton, P.; Tobe-Gai, R.; Mahomed, K.; Miyazaki, C.; Bhutta, Z.A. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015, CD000230. [Google Scholar] [CrossRef] [PubMed]
- Lanoix, D.; Beghdadi, H.; Lafond, J.; Vaillancourt, C. Human placental trophoblasts synthesize melatonin and express its receptors. J. Pineal. Res. 2008, 45, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, F.J.; Vera, J.; Venegas, C.; Pino, F.; Lagunas, C. Circadian System and Melatonin Hormone: Risk Factors for Complications during Pregnancy. Obstet. Gynecol. Int. 2015, 825802. [Google Scholar] [CrossRef] [PubMed]
- Lanoix, D.; Guérin, P.; Vaillancourt, C. Placental melatonin production and melatonin receptor expression are altered in preeclampsia: New insights into the role of this hormone in pregnancy. J. Pineal. Res. 2012, 53, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, L.; D’Angelo, G.; Manti, S.; Reiter, R.J.; Gitto, E. Potential Utility of Melatonin in Preeclampsia, Intrauterine Fetal Growth Retardation, and Perinatal Asphyxia. Reprod. Sci. 2016, 23, 970–977. [Google Scholar] [CrossRef]
- Hobson, S.R.; Gurusinghe, S.; Lim, R.; Alers, N.O.; Miller, S.L.; Kingdom, J.C.; Wallace, E.M. Melatonin improves endothelial function in vitro and prolongs pregnancy in women with early-onset preeclampsia. J. Pineal. Res. 2018, 65, e12508. [Google Scholar] [CrossRef]
- Borghi, C.; Cicero, A.F.; Degli Esposti, D.; Immordino, V.; Bacchelli, S.; Rizzo, N.; Santi, F.; Ambrosioni, E. Hemodynamic and neurohumoral profile in patients with different types of hypertension in pregnancy. Intern. Emerg. Med. 2011, 6, 227–234. [Google Scholar] [CrossRef]
- Cicero, A.F.; Degli Esposti, D.; Immordino, V.; Morbini, M.; Baronio, C.; Rosticci, M.; Borghi, C. Independent Determinants of Maternal and Fetal Outcomes in a Sample of Pregnant Outpatients With Normal Blood Pressure, Chronic Hypertension, Gestational Hypertension, and Preeclampsia. J. Clin. Hypertens. 2015, 17, 777–782. [Google Scholar] [CrossRef]
- De Boer, A.; Bast, A.; Godschalk, R. Dietary supplement intake during pregnancy; better safe than sorry? Regul. Toxicol. Pharm. 2018, 95, 442–447. [Google Scholar] [CrossRef]
- Patti, A.M.; Giglio, R.V.; Pafili, K.; Rizzo, M.; Papanas, N. Pharmacotherapy for gestational diabetes. Expert Opin. Pharm. 2018, 19, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fogacci, S.; Fogacci, F.; Cicero, A.F.G. Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence. Nutrients 2020, 12, 378. https://doi.org/10.3390/nu12020378
Fogacci S, Fogacci F, Cicero AFG. Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence. Nutrients. 2020; 12(2):378. https://doi.org/10.3390/nu12020378
Chicago/Turabian StyleFogacci, Silvia, Federica Fogacci, and Arrigo F.G. Cicero. 2020. "Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence" Nutrients 12, no. 2: 378. https://doi.org/10.3390/nu12020378
APA StyleFogacci, S., Fogacci, F., & Cicero, A. F. G. (2020). Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence. Nutrients, 12(2), 378. https://doi.org/10.3390/nu12020378






