Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study
Abstract
1. Introduction
1.1. Nickel and Allergic Contact Mucositis
1.2. Endometriosis
1.3. Gastrointestinal Disorders and Endometriosis
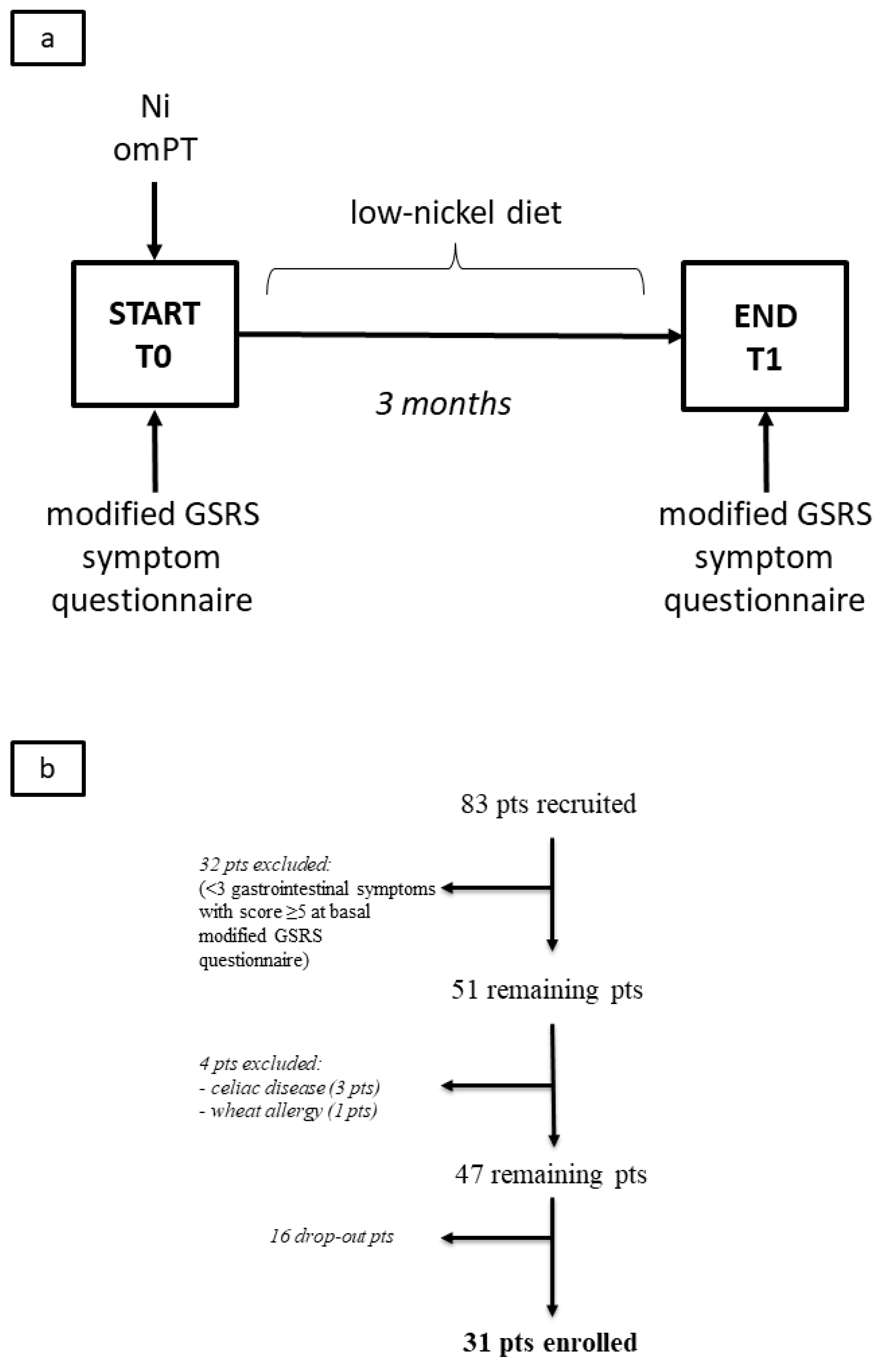
2. Materials and Methods
2.1. Patients
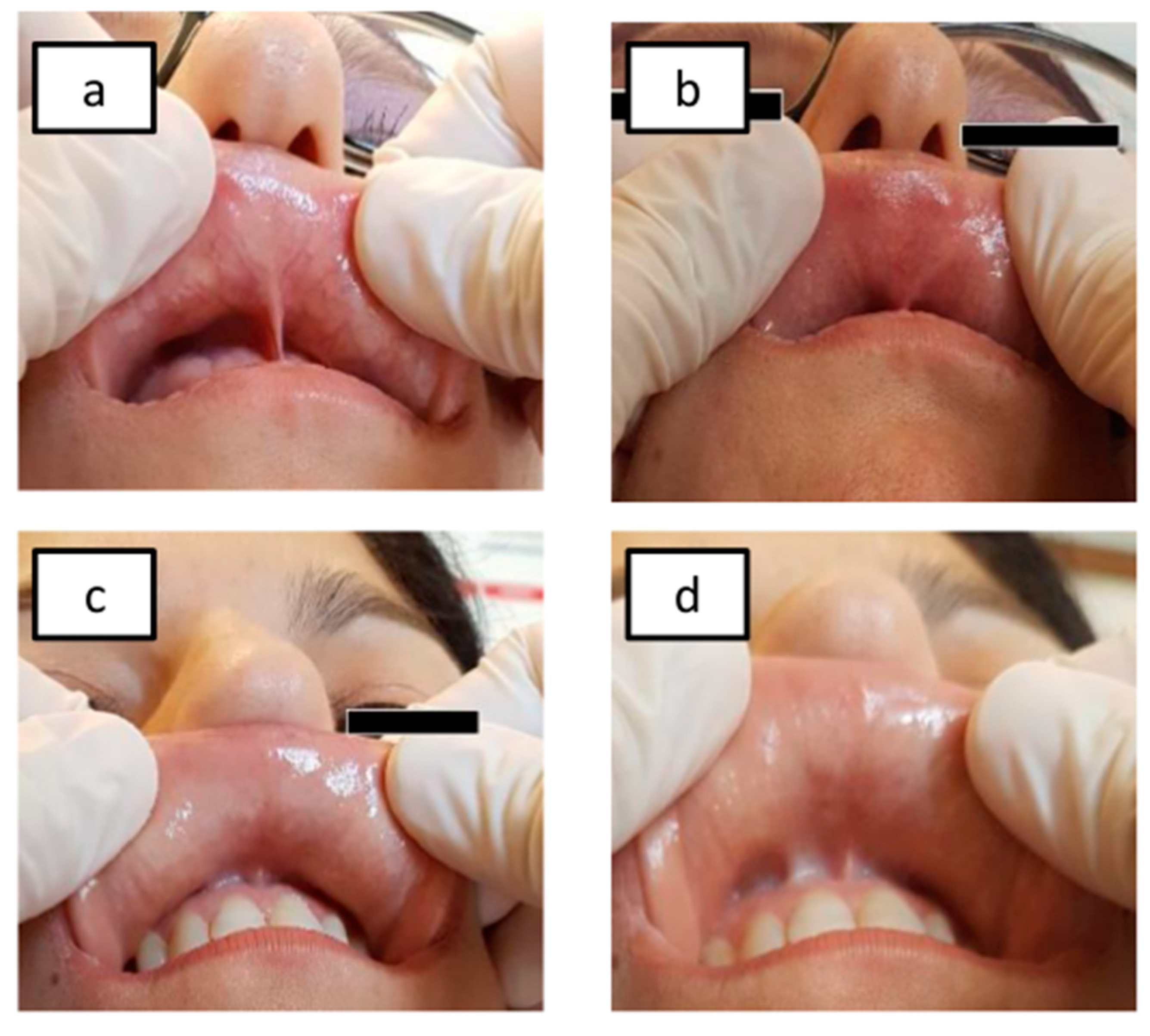
2.2. Nickel Oral Mucosa Patch Test
2.3. Symptom Questionnaire
2.4. Low-Nickel Diet
2.5. Statistical Analysis
3. Results
3.1. Patients
3.2. Nickel Oral Mucosa Patch Test
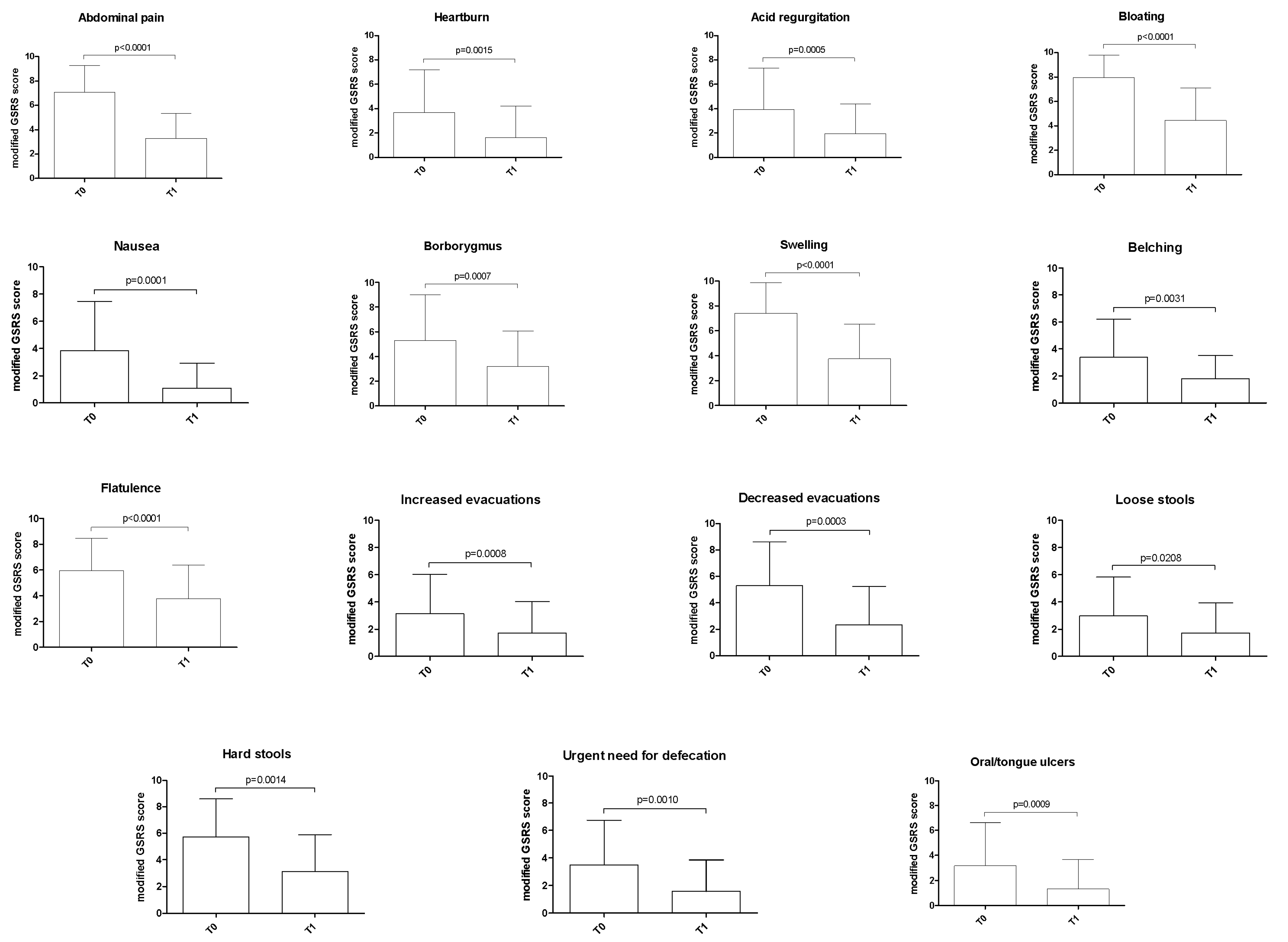
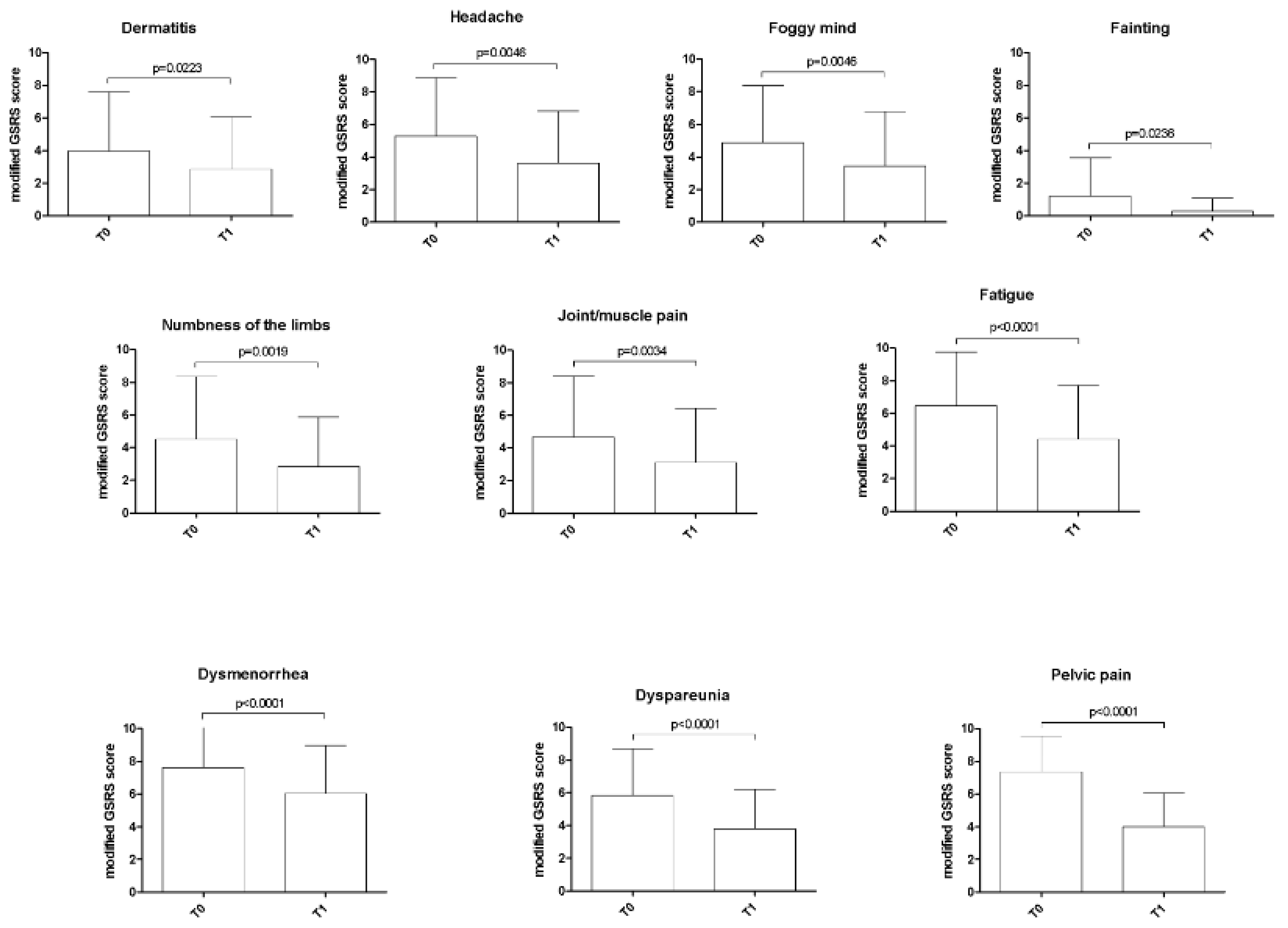
3.3. Symptom Questionnaire
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barceloux, D.G. Nickel. J. Toxicol. Clin. Toxicol. 1999, 37, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Solomons, N.W.; Viteri, F. Bioavailabilty of nickel in man: Effects of foods and chemically-defined dietary constituents on the absorption of inorganic nickel. J. Nutr. 1982, 112, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.D. Low nickel diet in dermatology. Indian J. Dermatol 2013, 58, 240. [Google Scholar] [CrossRef] [PubMed]
- Wittsiepe, J.; Schnell, K.; Hilbig, A.; Schrey, P.; Kersting, M.; Wilhelm, M. Dietary intake of nickel and zinc by young children—Results from food duplicate portion measurements in comparison to data calculated from dietary records and available data on levels in food groups. J. Trace Elem. Med. Biol. 2009, 23, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Borghini, R.; Donato, G.; Alvaro, D. New insights in IBS-like disorders: Pandora’s box has been opened; a review. Gastroenterol. Hepatol. Bed Bench. 2017, 10, 79–89. [Google Scholar] [PubMed]
- Brera, S.; Nicolini, A. Respiratory manifestations due to nickel. Acta Otorhinolaryngol Ital 2005, 25, 113–115. [Google Scholar]
- Pizzutelli, S. Systemic nickel hypersensitivity and diet: Myth or reality? Eur. Ann. Allergy Clin. Immunol. 2011, 43, 5–18. [Google Scholar]
- Marcusson, J.A.; Lindh, G.; Evengård, B. Chronic fatigue syndrome and nickel allergy. Contact Dermatitis 1999, 40, 269–272. [Google Scholar] [CrossRef]
- Rizzi, A.; Nucera, E.; Laterza, L.; Gaetani, E.; Valenza, V.; Corbo, G.M.; Inchingolo, R.; Buonomo, A.; Schiavino, D.; Gasbarrini, A. Irritable Bowel Syndrome and Nickel Allergy: What Is the Role of the Low Nickel Diet? J. Neurogastroenterol Motil. 2017, 23, 101–108. [Google Scholar] [CrossRef]
- Kennedy, S.; Bergqvist, A.; Chapron, C.; D’Hooghe, T.; Dunselman, G.; Greb, R.; Hummelshoj, L.; Prentice, A.; Saridogan, E.; ESHRE Special Interest Group for Endometriosis and Endometrium Guideline Development Group. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum. Reprod. 2005, 20, 2698–2704. [Google Scholar] [CrossRef]
- Giudice, L.C. Clinical practice. Endometriosis. N Engl J Med. 2010, 362, 2389–2398. [Google Scholar] [CrossRef] [PubMed]
- Meuleman, C.; Vandenabeele, B.; Fieuws, S.; Spiessens, C.; Timmerman, D.; D’Hooghe, T. High prevalence of endometriosis in infertile women with normal ovulation and normospermic partners. Fertil. Steril. 2009, 92, 68–74. [Google Scholar] [CrossRef]
- Parazzini, F.; Esposito, G.; Tozzi, L.; Noli, S.; Bianchi, S. Epidemiology of endometriosis and its comorbidities. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 209, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Heilier, J.F.; Donnez, J.; Nackers, F.; Rousseau, R.; Verougstraete, V.; Rosenkranz, K.; Donnez, O.; Grandjean, F.; Lison, D.; Tonglet, R. Environmental and host-associated risk factors in endometriosis and deep endometriotic nodules: A matched case-control study. Environ. Res. 2007, 103, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Yeaman, G.R.; Crispens, M.A.; Igarashi, T.M.; Osteen, K.G. Dioxin may promote inflammation-related development of endometriosis. Fertil. Steril. 2008, 89, 1287–1298. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.; Divekar, S.D.; Storchan, G.B.; Parodi, D.A.; Martin, M.B. Metals and breast cancer. J. Mammary Gland Biol. Neoplasia 2013, 18, 63–73. [Google Scholar] [CrossRef]
- Martin, M.B.; Reiter, R.; Pham, T.; Avellanet, Y.R.; Camara, J.; Lahm, M.; Pentecost, E.; Pratap, K.; Gilmore, B.A.; Divekar, S.; et al. Estrogen-like activity of metals in MCF-7 breast cancer cells. Endocrinology 2003, 144, 2425–2436. [Google Scholar] [CrossRef]
- Lappano, R.; Malaguarnera, R.; Belfiore, A.; Maggiolini, M. Recent advances on the stimulatory effects of metals in breast cancer. Mol. Cell Endocrinol. 2017, 457, 49–56. [Google Scholar] [CrossRef]
- Silva, N.; Senanayake, H.; Waduge, V. Elevated levels of whole blood nickel in a group of Sri Lankan women with endometriosis: A case control study. BMC Res. Notes 2013, 6, 13. [Google Scholar] [CrossRef]
- Yuk, J.S.; Kim, Y.J.; Yi, K.W.; Tak, K.; Hur, J.Y.; Shin, J.H. High rate of nickel allergy in women with endometriosis: A 3-year population-based study. J. Obstet. Gynaecol. Res. 2015, 41, 1255–1259. [Google Scholar] [CrossRef]
- Simoens, S.; Dunselman, G.; Dirksen, C.; Hummelshoj, L.; Bokor, A.; Brandes, I.; Brodszky, V.; Canis, M.; Colombo, G.L.; DeLeire, T.; et al. The burden of endometriosis: Costs and quality of life of women with endometriosis and treated in referral centres. Hum. Reprod. 2012, 27, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Porpora, M.G.; Koninckx, P.R.; Piazze, J.; Natili, M.; Colagrande, S.; Cosmi, E.V. Correlation between endometriosis and pelvic pain. J. Am. Assoc. Gynecol. Laparosc. 1999, 6, 429–434. [Google Scholar] [CrossRef]
- Manganaro, L.; Porpora, M.G.; Vinci, V.; Bernardo, S.; Lodise, P.; Sollazzo, P.; Sergi, M.E.; Saldari, M.; Pace, G.; Vittori, G.; et al. Diffusion tensor imaging and tractography to evaluate sacral nerve root abnormalities in endometriosis-related pain: A pilot study. Eur. Radiol. 2014, 24, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Stratton, P.; Berkley, K.J. Chronic pelvic pain and endometriosis: Translational evidence of the relationship and implications. Hum. Reprod. Update 2011, 17, 327–346. [Google Scholar] [CrossRef] [PubMed]
- Howard, F.M. Endometriosis and mechanisms of pelvic pain. J. Minim. Invasive Gynecol. 2009, 16, 540–550. [Google Scholar] [CrossRef]
- Stein, S.L. Chronic pelvic pain. Gastroenterol. Clin. North Am. 2013, 42, 785–800. [Google Scholar] [CrossRef]
- Ferrero, S.; Ragni, N.; Remorgida, V. Deep dyspareunia: Causes, treatments, and results. Curr. Opin. Obstet. Gynecol. 2008, 20, 394–399. [Google Scholar] [CrossRef]
- Dunselman, G.A.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’Hooghe, T.; De Bie, B.; Heikinheimo, O.; Horne, A.W.; Kiesel, L.; Nap, A.; et al. ESHRE guideline: Management of women with endometriosis. Hum. Reprod. 2014, 29, 400–412. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Bonsignore, L.; Olive, D.; Samuels, S.; Vercellini, P. Validation study of nonsurgical diagnosis of endometriosis. Fertil. Steril. 2001, 76, 929–935. [Google Scholar] [CrossRef]
- Vercellini, P.; Carmignani, L.; Rubino, T.; Barbara, G.; Abbiati, A.; Fedele, L. Surgery for deep endometriosis: A pathogenesis-oriented approach. Gynecol. Obstet. Invest. 2009, 68, 88–103. [Google Scholar] [CrossRef]
- Porpora, M.G.; Pallante, D.; Ferro, A.; Crisafi, B.; Bellati, F.; Benedetti Panici, P. Pain and ovarian endometrioma recurrence after laparoscopic treatment of endometriosis: A long-term prospective study. Fertil. Steril. 2010, 93, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Schroder, A.K.; Diedrich, K.; Ludwig, M. Medical management of endometriosis: A systematic review. IDrugs. 2004, 7, 451–463. [Google Scholar] [PubMed]
- Vercellini, P.; Crosignani, P.; Somigliana, E.; Viganò, P.; Frattaruolo, M.P.; Fedele, L. ‘Waiting for Godot’: A commonsense approach to the medical treatment of endometriosis. Hum. Reprod. 2011, 26, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Hernando, L.; Muñoz-Gonzalez, J.L.; Marqueta-Marques, L.; Alvarez-Conejo, C.; Tejerizo-García, Á.; Lopez-Gonzalez, G.; Villegas-Muñoz, E.; Martin-Jimenez, A.; Jiménez-López, J.S. Endometriosis: Alternative methods of medical treatment. Int. J. Womens Health 2015, 7, 595–603. [Google Scholar] [PubMed]
- Maroun, P.; Cooper, M.J.; Reid, G.D.; Keirse, M.J. Relevance of gastrointestinal symptoms in endometriosis. Aust. N Z J Obstet. Gynaecol. 2009, 49, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Remorgida, V.; Ferrero, S.; Fulcheri, E.; Ragni, N.; Martin, D.C. Bowel endometriosis: Presentation, diagnosis, and treatment. Obstet. Gynecol. Surv. 2007, 62, 461–470. [Google Scholar] [CrossRef]
- Fauconnier, A.; Chapron, C.; Dubuisson, J.B.; Vieira, M.; Dousset, B.; Bréart, G. Relation between pain symptoms and the anatomic location of deep infiltrating endometriosis. Fertil. Steril. 2002, 78, 719–726. [Google Scholar] [CrossRef]
- Seaman, H.E.; Ballard, K.D.; Wright, J.T.; de Vries, C.S. Endometriosis and its coexistence with irritable bowel syndrome and pelvic inflammatory disease: Findings from a national case-control study—Part 2. BJOG. 2008, 115, 1392–1396. [Google Scholar] [CrossRef]
- Roman, H.; Ness, J.; Suciu, N.; Bridoux, V.; Gourcerol, G.; Leroi, A.M.; Tuech, J.J.; Ducrotté, P.; Savoye-Collet, C.; Savoye, G. Are digestive symptoms in women presenting with pelvic endometriosis specific to lesion localizations? A preliminary prospective study. Hum. Reprod. 2012, 27, 3440–3449. [Google Scholar] [CrossRef]
- Mathias, J.R.; Franklin, R.; Quast, D.C.; Fraga, N.; Loftin, C.A.; Yates, L.; Harrison, V. Relation of endometriosis and neuromuscular disease of the gastrointestinal tract: New insights. Fertil. Steril. 1998, 70, 81–88. [Google Scholar] [CrossRef]
- Berkley, K.J.; Rapkin, A.J.; Papka, R.E. The pains of endometriosis. Science 2005, 308, 1587–1589. [Google Scholar] [CrossRef] [PubMed]
- Tokushige, N.; Markham, R.; Russell, P.; Fraser, I.S. High density of small nerve fibres in the functional layer of the endometrium in women with endometriosis. Hum. Reprod. 2006, 21, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Sand, E.; Bergvall, M.; Ekblad, E.; D’Amato, M.; Ohlsson, B. Expression and distribution of GnRH, LH, and FSH and their receptors in gastrointestinal tract of man and rat. Regul. Pept. 2013, 187, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, B. Gonadotropin-Releasing Hormone and Its Physiological and Pathophysiological Roles in Relation to the Structure and Function of the Gastrointestinal Tract. Eur. Surg. Res. 2016, 57, 22–33. [Google Scholar] [CrossRef]
- Lea, R.; Whorwell, P.J. Irritable bowel syndrome or endometriosis, or both? Eur. J. Gastroenterol. Hepatol. 2003, 15, 1131–1133. [Google Scholar] [CrossRef]
- Yuk, J.S.; Shin, J.S.; Shin, J.Y.; Oh, E.; Kim, H.; Park, W.I. Nickel Allergy Is a Risk Factor for Endometriosis: An 11-Year Population-Based Nested Case-Control Study. PLoS ONE. 2015, 10, e0139388. [Google Scholar] [CrossRef]
- Borghini, R.; Puzzono, M.; Rosato, E.; Di Tola, M.; Marino, M.; Greco, F.; Picarelli, A. Nickel-Related Intestinal Mucositis in IBS-Like Patients: Laser Doppler Perfusion Imaging and Oral Mucosa Patch Test in Use. Biol. Trace Elem. Res. 2016, 173, 55–61. [Google Scholar] [CrossRef]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Picarelli, A.; Di Tola, M.; Vallecoccia, A.; Libanori, V.; Magrelli, M.; Carlesimo, M.; Rossi, A. Oral mucosa patch test: A new tool to recognize and study the adverse effects of dietary nickel exposure. Biol. Trace Elem. Res. 2011, 139, 151–159. [Google Scholar] [CrossRef]
- Steele, J.C.; Bruce, A.J.; Davis, M.D.; Torgerson, R.R.; Drage, L.A.; Rogers, R.S., 3rd. Clinically relevant patch test results in patients with burning mouth syndrome. Dermatitis 2012, 23, 61–70. [Google Scholar] [CrossRef]
- Kim, T.W.; Kim, W.I.; Mun, J.H.; Song, M.; Kim, H.S.; Kim, B.S.; Kim, M.B.; Ko, H.C. Patch Testing with Dental Screening Series in Oral Disease. Ann. Dermatol. 2015, 27, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Porpora, M.G.; Medda, E.; Abballe, A.; Bolli, S.; De Angelis, I.; di Domenico, A.; Ferro, A.; Ingelido, A.M.; Maggi, A.; Panici, P.B.; et al. Endometriosis and organochlorinated environmental pollutants: A case-control study on Italian women of reproductive age. Environ. Health Perspect. 2009, 117, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Aquino, N.B.; Sevigny, M.B.; Sabangan, J.; Louie, M.C. The role of cadmium and nickel in estrogen receptor signaling and breast cancer: Metalloestrogens or not? J. Environ. Sci. Health C Environ. Carcinog Ecotoxicol. Rev. 2012, 30, 189–224. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D. Irritable bowel syndrome, chronic pelvic inflammatory disease and endometriosis. Eur. J. Gastroenterol. Hepatol. 2004, 16, 1251–1252. [Google Scholar] [CrossRef] [PubMed]
- Issa, B.; Onon, T.S.; Agrawal, A.; Shekhar, C.; Morris, J.; Hamdy, S.; Whorwell, P.J. Visceral hypersensitivity in endometriosis: A new target for treatment? Gut 2012, 61, 367–372. [Google Scholar] [CrossRef]
- Moore, J.S.; Gibson, P.R.; Perry, R.E.; Burgell, R.E. Endometriosis in patients with irritable bowel syndrome: Specific symptomatic and demographic profile, and response to the low FODMAP diet. Aust. N Z J Obstet. Gynaecol. 2017, 57, 201–205. [Google Scholar] [CrossRef]
| Foodstuffs | Ni-containing foods |
|---|---|
| Fishes | Herring, mackerel, salmon, shellfish, tuna |
| Vegetables | Fresh and dried legumes (chickpeas, lentils, peanuts, peas, red kidney beans, soya beans, and soy products), garlic, green leafy vegetables (spinach), onion, raw carrots, tomatoes |
| Fruits | Fresh and dried fruits (almonds, hazelnuts, walnuts) |
| Cereals | Buckwheat, maize, millet, oat, rye, whole grain, whole wheat |
| Beverages | Beer, coffee, initial water flow from the tap (especially in the morning), red wine, tea |
| Others | Baking powder, canned foods, cocoa and chocolate, foods cooked in stainless steel utensils (especially if acidic foods as tomatoes), gelatin, linseeds, marzipan, Ni-containing vitamin supplements, strong licorice, sunflower seeds |
| Pt n. | Age (yrs) | Endometriosis Duration (yrs) | Endometriotic Lesion’s Site | Size Score | Endometriosis Stage | Hormonal Treatment Duration | Surgery | Comorbidities | Concomitant Therapies | Ni omPT Result |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 38 | 3 | LO EC | 2 | n.a. | - | - | - | - | Positive |
| 2 | 25 | 2 | rectum | n.a. | n.a. | EP (≥ 5 yrs) | - | PCOS | - | Positive |
| 3 | 45 | 20 | LO EC; POD; peritoneum | 0 | IV | - | LPS | folate-deficiency anemia | folic acid | Positive |
| 4 | 31 | 5 | LO EC; peritoneum | 1 | III | EP (≥ 5 yrs) | LPS | - | - | Positive |
| 5 | 38 | 4 | cesarean section scar | 0 | iatrogenic | EP (1-3 yrs) | LPT | generalized anxiety disorder | escitalopram | Negative |
| 6 | 46 | 17 | bilateral ECs; peritoneum | 1 | IV | EP (1-3 yrs) | LPS | - | - | Positive |
| 7 | 31 | 4 | bilateral ECs | 1 | IV | P (1-3 yrs) | LPS | - | - | Positive |
| 8 | 49 | 21 | bilateral ECs | 2 | IV | - | LPT; LPS | - | - | Positive |
| 9 | 30 | 3 | LO EC | 2 | n.a. | P (1-3 yrs) | - | - | - | Negative |
| 10 | 32 | 3 | LO EC | 1 | n.a. | EP (1-3 yrs) | - | - | - | Positive |
| 11 | 39 | 20 | RO EC | 1 | III | - | LPS | bilateral breast fibroadenomas | - | Positive |
| 12 | 31 | 7 | LO EC | 0 | n.a. | - | - | - | - | Positive |
| 13 | 37 | 21 | uterine cervix | 0 | n.a. | - | - | fibromyalgia | - | Positive |
| 14 | 25 | 4 | LO EC | 2 | III | P (1-3 yrs) | LPS | Hashimoto thyroiditis | - | Positive |
| 15 | 27 | 3 | RO; peritoneum | 2 | III | EP (1-3 yrs) | LPS | - | - | Positive |
| 16 | 31 | 9 | LO EC; peritoneum | 2 | III | EP (≥ 5 yrs) | LPS | - | - | Positive |
| 17 | 38 | 3 | peritoneum | 0 | III | EP (≥ 5 yrs) | LPS | - | - | Positive |
| 18 | 25 | 4 | LO EC | 0 | n.a. | - | - | - | - | Positive |
| 19 | 19 | 3 | peritoneum | 0 | n.a. | EP (1-3 yrs) | - | pollen allergy | - | Positive |
| 20 | 43 | 4 | bilateral ovarian ECs | 1 | n.a. | EP (< 1 yrs) | - | - | - | Positive |
| 21 | 27 | n.a. | LO EC | 0 | n.a. | - | - | - | - | Positive |
| 22 | 40 | 20 | bilateral ovarian ECs | 0 | n.a. | P (1-3 yrs) | - | PRL-secreting pituitary adenomas | cabergoline | Positive |
| 23 | 35 | 10 | RO EC | 1 | II | P (1-3 yrs) | LPS | hypothyroidism | levothyroxine | Negative |
| 24 | 43 | 18 | bilateral ovarian ECs | 1 | IV | P (1-3 yrs) | LPS | Hashimoto thyroiditis | - | Positive |
| 25 | 27 | 1 | RO EC | 0 | n.a. | - | - | psoriatic arthritis, fibromyalgia | - | Positive |
| 26 | 20 | 1 | 3 LO ECs | 1 | III | EP (< 1 yr) | LPS | - | - | Positive |
| 27 | 28 | 8 | LO EC | 0 | III | P (≥ 5 yrs) | LPS | mild depressive disorder | amitriptyline | Positive |
| 28 | 36 | 21 | LO EC | 0 | n.a. | - | - | - | Positive | |
| 29 | 40 | 13 | RO EC | 0 | IV | P (≥ 5 yrs) | LPS | Hashimoto’s thyroiditis | levothyroxine | Positive |
| 30 | 38 | 2 | bilateral ovarian ECs | 2 | III | EP (1-3 yrs) | LPS | fibromyalgia | Positive | |
| 31 | 24 | 1 | 3 RO ECs | 0 | n.a. | - | - | - | Positive |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borghini, R.; Porpora, M.G.; Casale, R.; Marino, M.; Palmieri, E.; Greco, N.; Donato, G.; Picarelli, A. Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study. Nutrients 2020, 12, 341. https://doi.org/10.3390/nu12020341
Borghini R, Porpora MG, Casale R, Marino M, Palmieri E, Greco N, Donato G, Picarelli A. Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study. Nutrients. 2020; 12(2):341. https://doi.org/10.3390/nu12020341
Chicago/Turabian StyleBorghini, Raffaele, Maria Grazia Porpora, Rossella Casale, Mariacatia Marino, Emilia Palmieri, Nicoletta Greco, Giuseppe Donato, and Antonio Picarelli. 2020. "Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study" Nutrients 12, no. 2: 341. https://doi.org/10.3390/nu12020341
APA StyleBorghini, R., Porpora, M. G., Casale, R., Marino, M., Palmieri, E., Greco, N., Donato, G., & Picarelli, A. (2020). Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study. Nutrients, 12(2), 341. https://doi.org/10.3390/nu12020341






