Low Levels of Vitamin D Promote Memory B Cells in Lupus
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Enrollment
2.2. Vitamin D3 Dietary Intervention and Animal Care
2.3. Organ Harvest/Preparation
2.4. Immunofluorescence Staining of Kidney Tissue
2.5. Serum Cytokine Flow Cytometry
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. Flow Cytometry
2.8. Real-Time Reverse-Transcriptase PCR
2.9. Statistical Analyses
3. Results
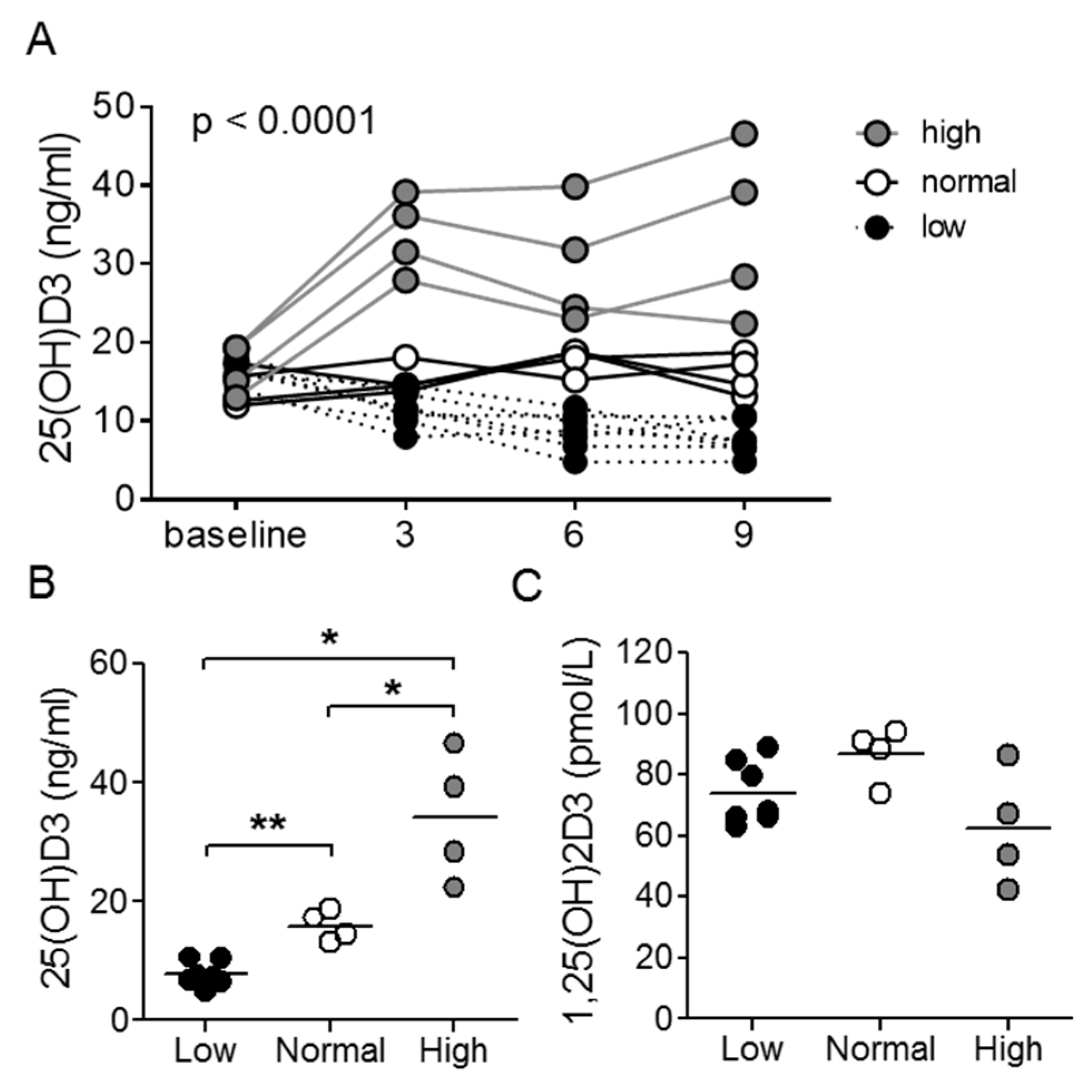
3.1. Serum 25(OH)D3 Levels Plateau after Controlled Dietary Vitamin D3 Intake
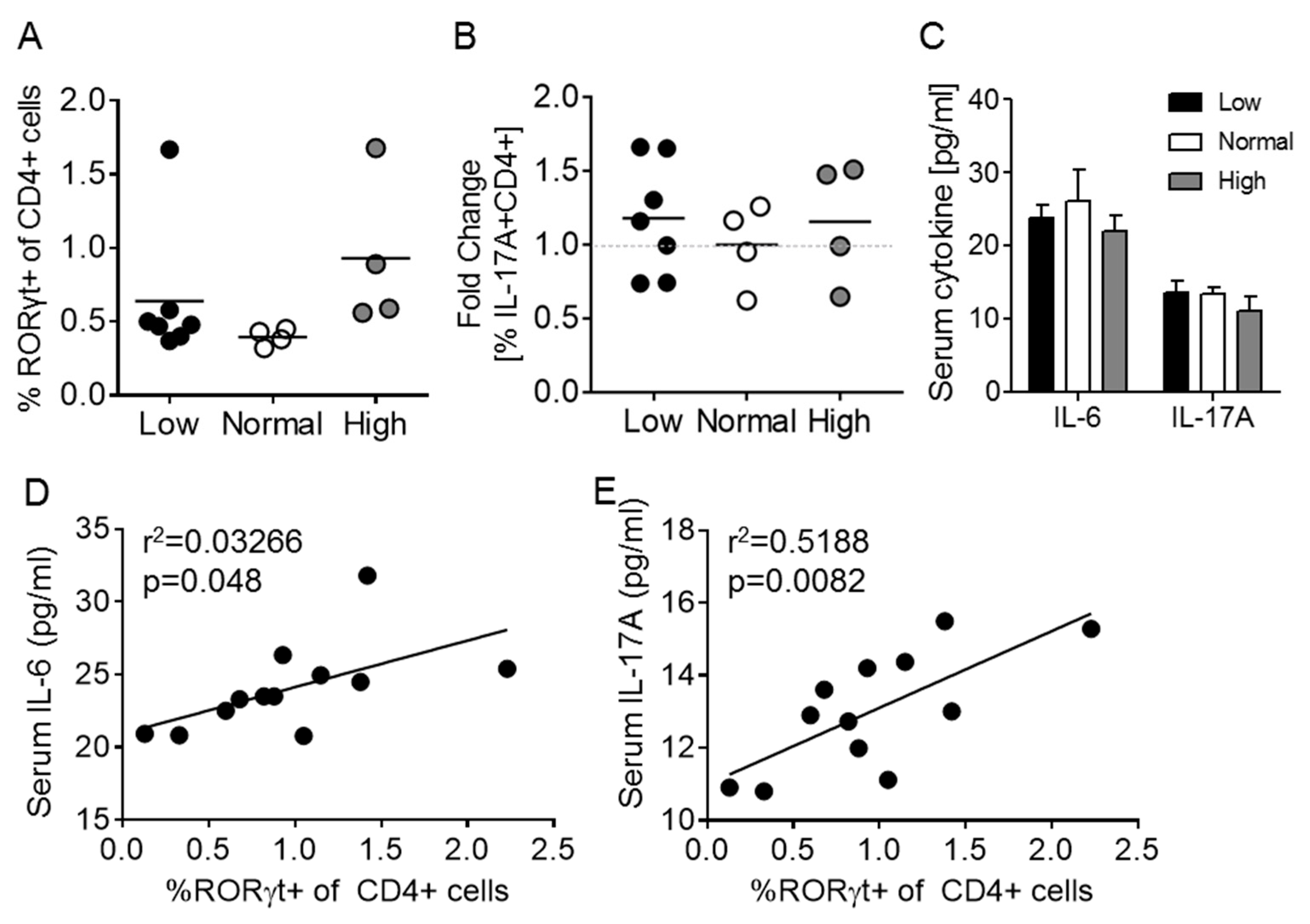
3.2. Vitamin D3 Manipulation Does Not Affect Th17 Cells in the Act1-/- Mouse
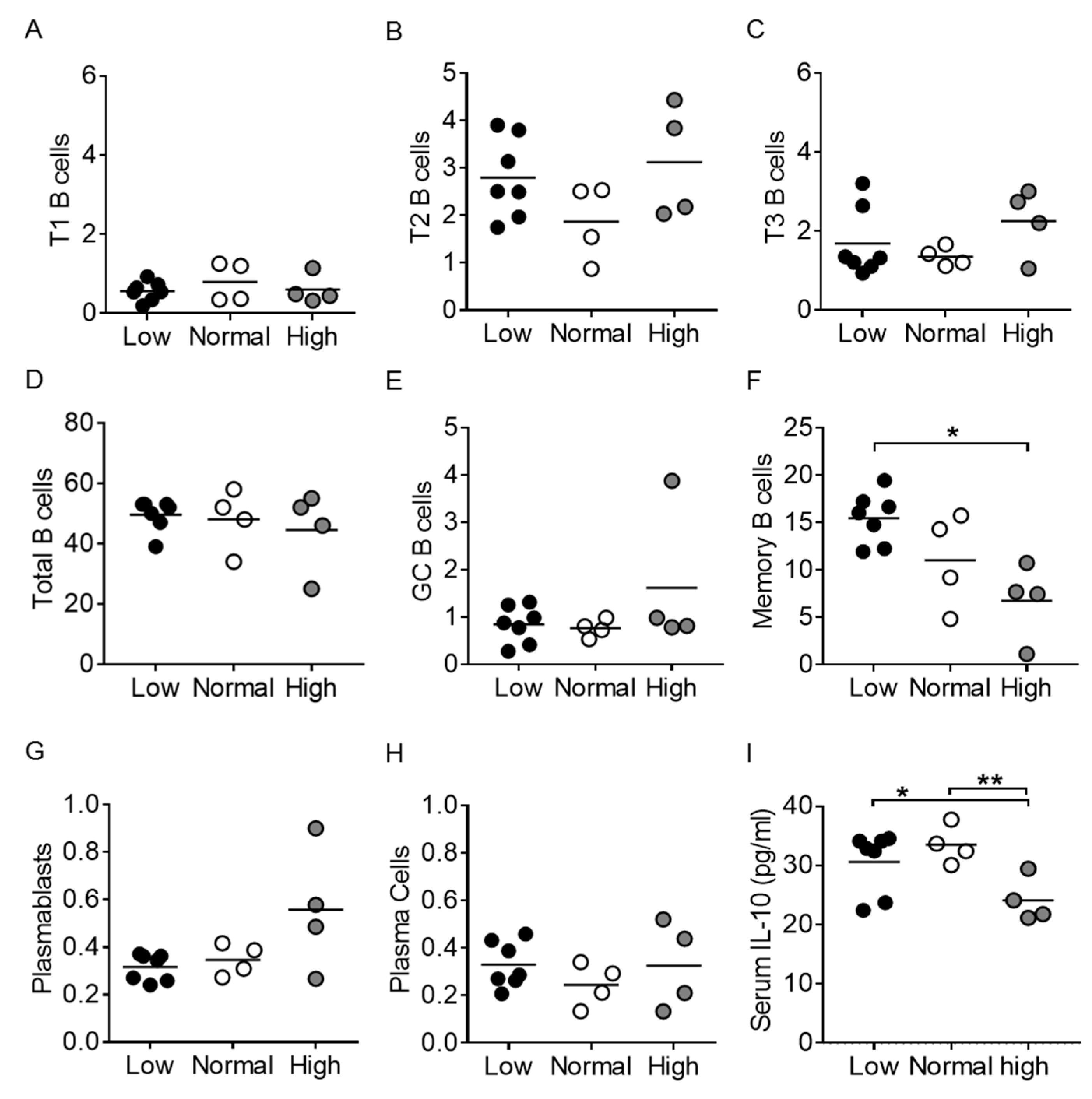
3.3. Vitamin D3 Restriction Specifically Augments Memory B Cells
3.4. Elevated Immunoglobulin Production and Anti-dsDNA in Vitamin D3-Restricted Mice
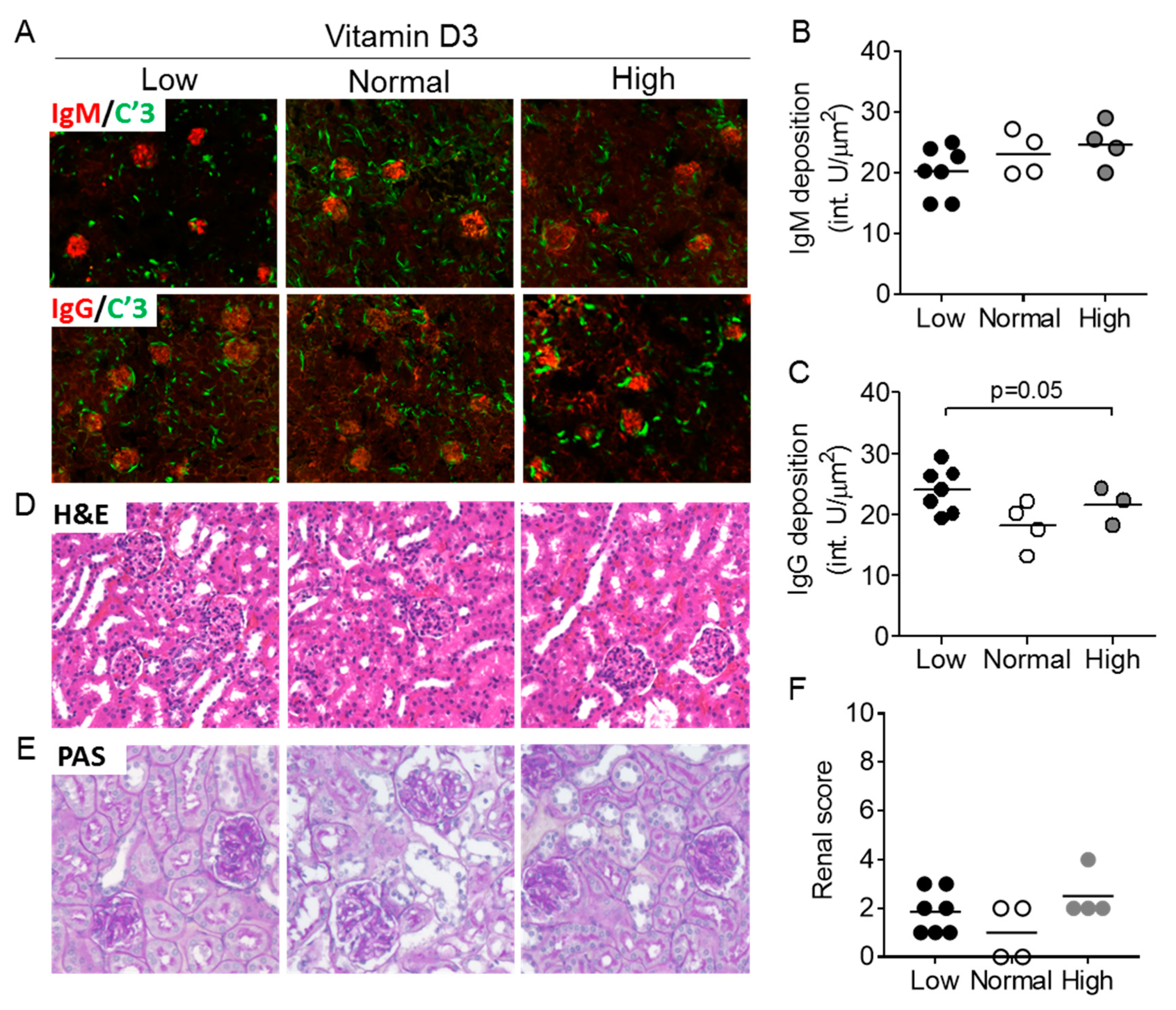
3.5. Vitamin D3 Exposure Does Not Affect Features of Nephritis after 9 Weeks
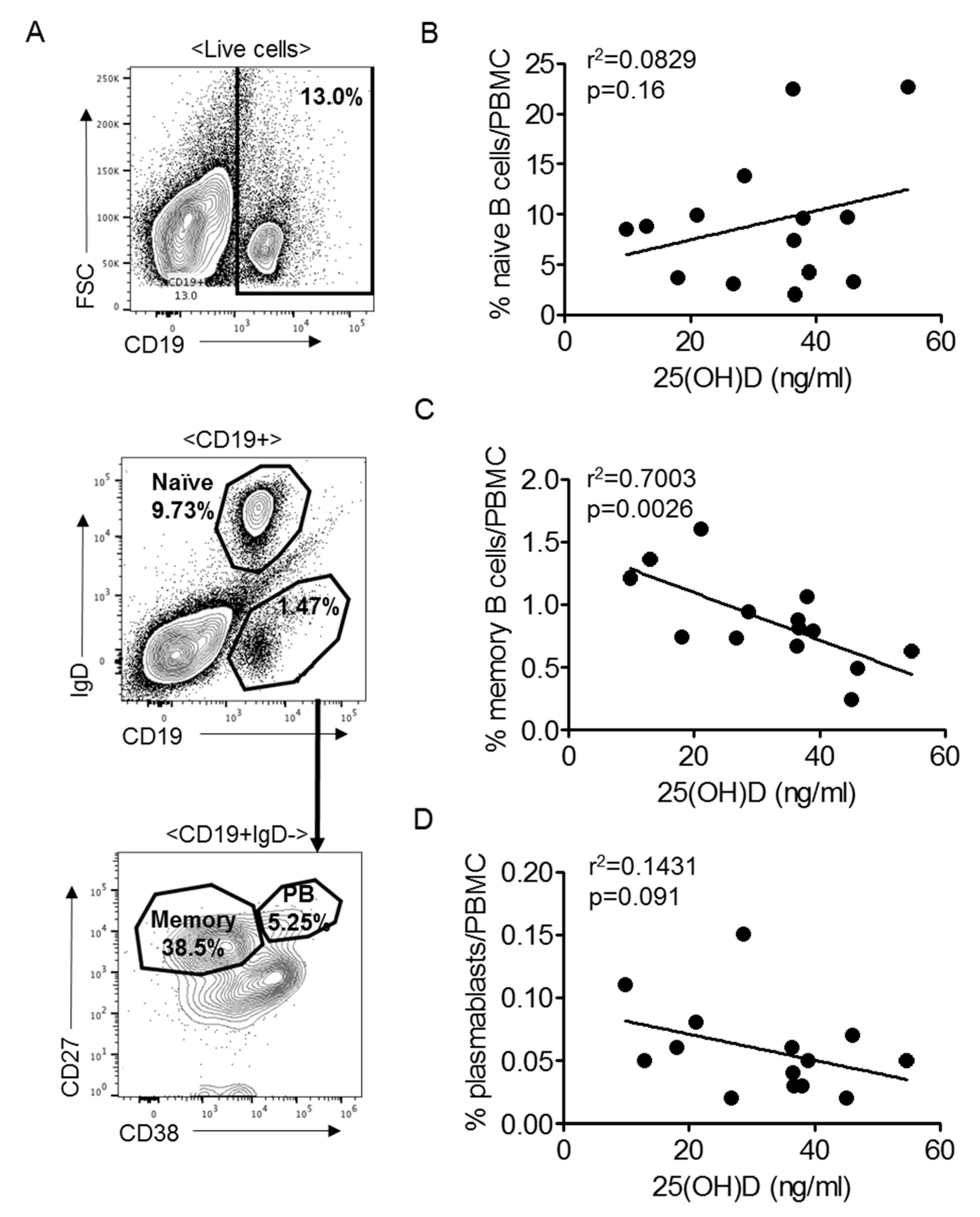
3.6. Memory B Cells Are Negatively Associated with Vitamin D3 in SLE
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tsokos, G.C. Systemic lupus erythematosus. N. Engl. J. Med. 2007, 369, 587–596. [Google Scholar]
- Harvey, P.R.; Gordon, C. B-Cell Targeted Therapies in Systemic Lupus Erythematosus. BioDrugs 2013, 27, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Liossis, S.-N.C.; Staveri, C. B Cell-Based Treatments in SLE: Past Experience and Current Directions. Curr. Rheumatol. Rep. 2017, 19, 78. [Google Scholar] [CrossRef]
- Sakthiswary, R.; Raymond, A.A. The Clinical Significance of Vitamin D in Systemic Lupus Erythematosus: A Systematic Review. PLoS ONE 2013, 8, e55275. [Google Scholar] [CrossRef]
- Carvalho, C.; Marinho, A.; Leal, B.; Bettencourt, A.; Boleixa, D.; Almeida, I.; Farinha, F.; Costa, P.P.; Vasconcelos, C.; Silva, B.M. Association between vitamin D receptor (VDR) gene polymorphisms and systemic lupus erythematosus in Portuguese patients. Lupus 2015, 24, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Abe, J.; Nakamura, K.; Takita, Y.; Nakano, T.; Irie, H.; Nishii, Y. Prevention of immunological disorders in MRL/l mice by a new synthetic analogue of vitamin D3: 22-oxa-1.ALPHA., 25-dihydroxyvitamin D3. J. Nutr. Sci. Vitaminol. 1990, 36, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Lemire, J.; Ince, A.; Takashima, M. 1,25-Dihydroxyvitamin D3 attenuates the expression of experimental murine lupus of MRL/1 mice. Autoimmunity 1992, 12, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, T.; Nakao, Y.; Matsui, T.; Nakagawa, T.; Matsuda, S.; Komoriya, K.; Kanai, Y.; Fujita, T. Effects of Corticosteroid and 1,24R-Dihydroxy-Vitamin D3 Administration on Lymphoproliferation and Autoimmune Disease in MRL/MP-lpr/lpr Mice. Int. Arch. Allergy Immunol. 1985, 77, 396–404. [Google Scholar] [CrossRef]
- Yamamoto, E.; Jørgensen, T.N. Immunological effects of vitamin D and their relations to autoimmunity. J. Autoimmun. 2019, 100, 7–16. [Google Scholar] [CrossRef]
- Costenbader, K.H.; Feskanich, D.; Holmes, M.; Karlson, E.W.; Benito-Garcia, E. Vitamin D intake and risks of systemic lupus erythematosus and rheumatoid arthritis in women. Ann. Rheum. Dis. 2008, 67, 530–535. [Google Scholar] [CrossRef]
- Ruiz-Irastorza, G.; Egurbide, M.V.; Olivares, N.; Martinez-Berriotxoa, A.; Aguirre, C. Vitamin D deficiency in systemic lupus erythematosus: Prevalence, predictors and clinical consequences. Rheumatology 2008, 47, 920–923. [Google Scholar] [CrossRef]
- Petri, M.; Bello, K.J.; Fang, H.; Magder, L.S. Vitamin D in systemic lupus erythematosus: Modest association with disease activity and the urine protein-to-creatinine ratio. Arthritis Rheum. 2013, 65, 1865–1871. [Google Scholar] [CrossRef]
- Abou-Raya, A.; Abou-Raya, S.; Helmii, M. The effect of vitamin D supplementation on inflammatory and hemostatic markers and disease activity in patients with Systemic Lupus Erythematosus: A randomized placebo-controlled trial. J. Rheumatol. 2013, 40, 265–272. [Google Scholar] [CrossRef]
- Palmer, M.T.; Lee, Y.K.; Maynard, C.L.; Oliver, J.R.; Bikle, D.D.; Jetten, A.M.; Weaver, C.T. Lineage-specific effects of 1,25-dihydroxyvitamin D3 on the development of effector CD4 T cells. J. Biol. Chem. 2011, 286, 997–1004. [Google Scholar] [CrossRef]
- Kang, S.W.; Kim, S.H.; Lee, N.; Lee, W.-W.; Hwang, K.-A.; Shin, M.S.; Lee, S.-H.; Kim, W.-U.; Kang, I. 1,25-Dihyroxyvitamin D3 promotes FOXP3 expression via binding to vitamin D response elements in its conserved noncoding sequence region. J. Immunol. 2012, 188, 5276–5282. [Google Scholar] [CrossRef]
- Adzemovic, M.Z.; Zeitelhofer, M.; Hochmeister, S.; Gustafsson, S.A.; Jagodic, M. Efficacy of vitamin D in treating multiple sclerosis-like neuroinflammation depends on developmental stage. Exp. Neurol. 2013, 249, 39–48. [Google Scholar] [CrossRef]
- Drozdenko, G.; Heine, G.; Worm, M. Oral vitamin D increases the frequencies of CD38 + human B cells and ameliorates IL-17-producing T cells. Exp. Dermatol. 2014, 23, 107–112. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory Effects of 1,25-Dihydroxyvitamin D3 on Human B Cell Differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef]
- Haas, J.; Schwarz, A.; Korporal-Kuhnke, M.; Faller, S.; Jarius, S.; Wildemann, B. Hypovitaminosis D upscales B-cell immunoreactivity in multiple sclerosis. J. Neuroimmunol. 2016, 294, 18–26. [Google Scholar] [CrossRef]
- Chen, W.C.; Vayuvegula, B.; Gupta, S. 1,25-Dihydroxyvitamin D3-mediated inhibition of human B cell differentiation. Clin. Exp. Immunol. 1987, 69, 639–646. [Google Scholar]
- Heine, G.; Anton, K.; Henz, B.M.; Worm, M. 1alpha,25-dihydroxyvitamin D3 inhibits anti-CD40 plus IL-4-mediated IgE production in vitro. Eur. J. Immunol. 2002, 32, 3395–3404. [Google Scholar] [PubMed]
- Hartmann, B.; Heine, G.; Babina, M.; Steinmeyer, A.; Zügel, U.; Radbruch, A.; Worm, M. Targeting the vitamin D receptor inhibits the B cell-dependent allergic immune response. Allergy 2011, 66, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Qin, J.; Cui, G.; Naramura, M.; Snow, E.; Ware, C.F.; Fairchild, R.L.; Omori, S.A.; Rickert, R.C.; Scott, M.; et al. Act1, a Negative Regulator in CD40- and BAFF-Mediated B Cell Survival. Immunity 2004, 21, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Giltiay, N.; Xiao, J.; Wang, Y.; Qin, J.; Tian, J.; Shuhua, H.; Scott, M.; Carter, R.; Li, X. Deficiency of Act1, a critical modulator of B cell function, leads to development of Sjogren’s syndrome 1. Eur. J. Immunol. 2008, 38, 2219–2228. [Google Scholar] [CrossRef]
- Giltiay, N.V.; Lu, Y.; Allman, D.; Jørgensen, T.N.; Li, X. The adaptor molecule Act1 regulates BAFF responsiveness and self-reactive B cell selection during transitional B cell maturation. J. Immunol. 2010, 185, 99–109. [Google Scholar] [CrossRef]
- Giltiay, N.V.; Lu, Y.; Cullen, J.L.; Trine, N.; Shlomchik, M.J.; Li, X. Spontaneous loss of tolerance of autoreactive B cells in Act1-deficient rheumatoid factor transgenic mice. J. Immunol. 2013, 191, 2155–2163. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- Jørgensen, T.N.; Thurman, J.; Izui, S.; Falta, M.T.; Metzger, E.T.; Flannery, A.S.; Kappler, J.; Marrack, P.; Kotzin, B.L. Genetic susceptibility to PolyI:C-induced IFNα/β-dependent accelerated disease in lupus-prone mice. Genes Immun. 2006, 7, 555–567. [Google Scholar] [CrossRef]
- Smith, J.E.; Goodman, D.S. The turnover and transport of vitamin D and of a polar metabolite with the properties of 25-hydroxycholecalciferol in human plasma. J. Clin. Investig. 1971, 50, 2159–2167. [Google Scholar] [CrossRef]
- Gray, R.W.; Caldas, A.E.; Wilz, D.R.; Lemann, J.; Smith, G.A.; DeLuca, H.F. Metabolism and Excretion of 3 H-1,2 5-(OH)2-Vitamin D3 in Healthy Adults. J. Clin. Endocrinol. Metab. 1978, 46, 756–765. [Google Scholar] [CrossRef]
- Seeman, E.; Kumar, R.; Hunder, G.G.; Scott, M.; Heath, H.; Riggs, B.L. Production, degradation, and circulating levels of 1,25-dihydroxyvitamin D in health and in chronic glucocorticoid excess. J. Clin. Investig. 1980, 66, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, L.; Bulek, K.; Martin, B.N.; Zepp, J.A.; Kang, Z.; Liu, C.; Herjan, T.; Misra, S.; Carman, J.A.; et al. The psoriasis-associated D10N variant of the adaptor Act1 with impaired regulation by the molecular chaperone hsp90. Nat. Immunol. 2013, 14, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wang, C.; Boisson, B.; Misra, S.; Rayman, P.; Finke, J.H.; Puel, A.; Casanova, J.-L.; Li, X. The differential regulation of human ACT1 isoforms by Hsp90 in IL-17 signaling. J. Immunol. 2014, 193, 1590–1599. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-J.; Wang, C.; Jiang, M.; Gu, C.; Xiao, J.; Chen, X.; Martin, B.N.; Tang, F.; Yamamoto, E.; Xian, Y.; et al. Act1 is a negative regulator in T and B cells via direct inhibition of STAT3. Nat. Commun. 2018, 9, 2745. [Google Scholar] [CrossRef]
- Waddell, A.; Zhao, J.; Cantorna, M.T. NKT cells can help mediate the protective effects of 1,25-dihydroxyvitamin D3 in experimental autoimmune encephalomyelitis in mice. Int. Immunol. 2015, 27, 237–244. [Google Scholar] [CrossRef]
- Joshi, S.; Pantalena, L.-C.; Liu, X.K.; Gaffen, S.L.; Liu, H.; Rohowsky-Kochan, C.; Ichiyama, K.; Yoshimura, A.; Steinman, L.; Christakos, S.; et al. 1,25-Dihydroxyvitamin D3 Ameliorates Th17 Autoimmunity via Transcriptional Modulation of Interleukin-17A. Mol. Cell. Biol. 2011, 31, 3653–3669. [Google Scholar] [CrossRef]
- Ikeda, U.; Wakita, D.; Ohkuri, T.; Chamoto, K.; Kitamura, H.; Iwakura, Y.; Nishimura, T. 1α,25-Dihydroxyvitamin D3 and all-trans retinoic acid synergistically inhibit the differentiation and expansion of Th17 cells. Immunol. Lett. 2010, 134, 7–16. [Google Scholar] [CrossRef]
- Johnson, A.C.; Davison, L.M.; Giltiay, N.V.; Vareechon, C.; Li, X.; Jørgensen, T.N. Lack of T cells in Act1-deficient mice results in elevated IgM-specific autoantibodies but reduced lupus-like disease. Eur. J. Immunol. 2012, 42, 1695–1705. [Google Scholar] [CrossRef][Green Version]
- Bhattacharya, D.; Cheah, M.T.; Franco, C.B.; Hosen, N.; Pin, C.L.; Sha, W.C.; Weissman, I.L. Transcriptional Profiling of Antigen-Dependent Murine B Cell Differentiation and Memory Formation. J. Immunol. 2007, 179, 6808–6819. [Google Scholar] [CrossRef]
- Jones, G.W.; McLoughlin, R.M.; Hammond, V.J.; Parker, C.R.; Williams, J.D.; Malhotra, R.; Scheller, J.; Williams, A.S.; Rose-John, S.; Topley, N.; et al. Loss of CD4+ T cell IL-6R expression during inflammation underlines a role for IL-6 trans signaling in the local maintenance of Th17 cells. J. Immunol. 2010, 184, 2130–2139. [Google Scholar] [CrossRef]
- McFarland-Mancini, M.M.; Funk, H.M.; Paluch, A.M.; Zhou, M.; Giridhar, P.V.; Mercer, C.A.; Kozma, S.C.; Drew, A.F. Differences in Wound Healing in Mice with Deficiency of IL-6 versus IL-6 Receptor. J. Immunol. 2010, 184, 7219–7228. [Google Scholar] [CrossRef]
- Crotty, S. Follicular Helper CD4 T Cells (TFH). Annu. Rev. Immunol. 2011, 29, 621–663. [Google Scholar] [CrossRef]
- Kaji, T.; Hijikata, A.; Ishige, A.; Kitami, T.; Watanabe, T.; Ohara, O.; Yanaka, N.; Okada, M.; Shimoda, M.; Taniguchi, M.; et al. CD4 memory T cells develop and acquire functional competence by sequential cognate interactions and stepwise gene regulation. Int. Immunol. 2016, 28, 267–282. [Google Scholar] [CrossRef]
- Saxena, A.; Khosraviani, S.; Noel, S.; Mohan, D.; Donner, T.; Hamad, A.R.A. Interleukin-10 paradox: A potent immunoregulatory cytokine that has been difficult to harness for immunotherapy. Cytokine 2015, 74, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Guthmiller, J.J.; Graham, A.C.; Zander, R.A.; Pope, R.L.; Butler, N.S. Cutting Edge: IL-10 is essential for the generation of germinal center B cell responses and anti-plasmodium humoral immunity. J. Immunol. 2017, 198, 617–622. [Google Scholar] [CrossRef]
- Vaisberg, M.W.; Kaneno, R.; Franco, M.F.; Mendes, N.F. Influence of cholecalciferol (vitamin D3) on the course of experimental systemic lupus erythematosus in F1 (NZBxW) mice. J. Clin. Lab. Anal. 2000, 14, 91–96. [Google Scholar] [CrossRef][Green Version]
- Briere, F. Human interleukin 10 induces naive surface immunoglobulin D+ (sIgD+) B cells to secrete IgG1 and IgG3. J. Exp. Med. 1994, 179, 757–762. [Google Scholar] [CrossRef]
- Ishida, H. Continuous administration of anti-interleukin 10 antibodies delays onset of autoimmunity in NZB/W F1 mice. J. Exp. Med. 1994, 179, 305–310. [Google Scholar] [CrossRef]
- Ravirajan, C.T.; Wang, Y.; Matis, L.A.; Papadaki, L.; Griffiths, M.H.; Latchman, D.S.; Isenberg, D.A. Effect of neutralizing antibodies to IL-10 and C5 on the renal damage caused by a pathogenic human anti-dsDNA antibody. Rheumatology 2004, 43, 442–447. [Google Scholar] [CrossRef]
- Park, Y.B.; Lee, S.K.; Kim, D.S.; Lee, J.; Lee, C.H.; Song, C.H. Elevated interleukin-10 levels correlated with disease activity in systemic lupus erythematosus. Clin. Exp. Rheumatol. 1998, 16, 283–288. [Google Scholar]
- Hedrich, C.M.; Rauen, T.; Apostolidis, S.A.; Grammatikos, A.P.; Rodriguez, N.R.; Ioannidis, C.; Kyttaris, V.C.; Crispín, J.C.; Tsokos, G.C. Stat3 promotes IL-10 expression in lupus T cells through trans-activation and chromatin remodeling. Proc. Natl. Acad. Sci. USA 2014, 111, 13457–13462. [Google Scholar] [CrossRef]
- Enghard, P.; Langnickel, D.; Riemekasten, G. T cell cytokine imbalance towards production of IFN-γ and IL-10 in NZB/W F1 lupus-prone mice is associated with autoantibody levels and nephritis. Scand. J. Rheumatol. 2006, 35, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Franco, A.S.; Freitas, T.Q.; Bernardo, W.M.; Pereira, R.M.R. Vitamin D supplementation and disease activity in patients with immune-mediated rheumatic diseases: A systematic review and meta-analysis. Medicine 2017, 96, e7024. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.K.; Lammas, D.A.; Raza, K.; Sansom, D.M. 1,25-Dihydroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef]
- Chang, J.-H.; Cha, H.-R.; Lee, D.-S.; Seo, K.Y.; Kweon, M.-N. 1,25-Dihydroxyvitamin D3 inhibits the differentiation and migration of T(H)17 cells to protect against experimental autoimmune encephalomyelitis. PLoS ONE 2010, 5, e12925. [Google Scholar] [CrossRef]
- Nakayama, Y.; Stabach, P.; Maher, S.E.; Mahajan, M.C.; Masiar, P.; Liao, C.; Zhang, X.; Ye, Z.-J.; Tuck, D.; Bothwell, A.L.; et al. A limited number of genes are involved in the differentiation of germinal center B cells. J. Cell. Biochem. 2006, 99, 1308–1325. [Google Scholar] [CrossRef] [PubMed]
- Defrance, T.; Taillardet, M.; Genestier, L. T cell-independent B cell memory. Curr. Opin. Immunol. 2011, 23, 330–336. [Google Scholar] [CrossRef]
- Obukhanych, T.V.; Nussenzweig, M.C. T-independent type II immune responses generate memory B cells. J. Exp. Med. 2006, 203, 305–310. [Google Scholar] [CrossRef]
- Snapper, C.M.; McIntyre, T.M.; Mandler, R.; Pecanha, L.M.; Finkelman, F.D.; Lees, A.; Mond, J.J. Induction of IgG3 secretion by interferon gamma: A model for T cell- independent class switching in response to T cell-independent type 2 antigens. J. Exp. Med. 1992, 175, 1367–1371. [Google Scholar] [CrossRef]
- DeLuca, H.F.; Cantorna, M.T. Vitamin D: Its role and uses in immunology. FASEB J. 2001, 15, 2579–2585. [Google Scholar] [CrossRef]
- Tebben, P.J.; Singh, R.J.; Kumar, R. Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment. Endocr. Rev. 2016, 37, 521–547. [Google Scholar] [CrossRef] [PubMed]

| Vitamin D3 | |||
|---|---|---|---|
| Gene | Low | Normal | High |
| Rgs13 | 1.2 ± 0.1 | 1.1 ± 0.1 | 1.4 ± 0.2 |
| Bcl6 | 0.8 ± 0.1 | 1.0 ± 0.1 | 0.3 ± 0.02 ac |
| Aicda | 2.5 ± 0.5 | 2.4 ± 0.9 | 1.1 ± 0.3 a |
| Itga4 | 0.8 ± 0.1 | 1.0 ± 0.1 | 0.6 ± 0.2 b |
| Il6ra | 1.3 ± 0.3 | 1.0 ± 0.1 | 1.4 ± 0.2 |
| Irf4 | 1.2 ± 0.1 | 1.0 ± 0.1 | 0.8 ± 0.2 |
| Pt | Sex | Age | Race | 25(OH)D3 (ng/mL) | SLEDAI -2K | anti-dsDNA (IU/mL) | C3/C4 | Cr | IL- 17A | Th17 cells | %Naive B cells | %Memory B cells | %Plasma-blasts | Medication |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 33 | Other | 45.1 | 6 | - | 114/46 | - | 14.0 | 0.4 | 9.73% | 0.24% | 0.02% | Hydroxychloroquine, Mycophenolate |
| 2 | F | 36 | White | 38.0 | 6 | n.d. | - | - | 6.0 | 0.68 | 9.58% | 1.06% | 0.03% | - |
| 3 | F | 68 | Black | 46.1 | 2 | 102 | - | 0.81 | 10.0 | 0.91 | 3.27% | 0.49% | 0.07% | Hydroxychloroquine, Oral steroids |
| 4 | F | 57 | White | 36.4 | 4 | n.d. | 160/37 | 0.62 | 9.5 | 0.81 | 22.44% | 0.67% | 0.06% | Methotrexate, Oral steroids |
| 5 | M | 70 | White | 54.7 | 2 | n.d. | - | 2.07 | 7.0 | 0.89 | 22.65% | 0.63% | 0.05% | Hydroxychloroquine |
| 6 | F | 54 | White | 38.9 | 2 | n.d. | 146/26 | 0.77 | 8.0 | 0.66 | 4.24% | 0.79% | 0.05% | Hydroxychloroquine |
| 7 | F | 36 | Other | 28.7 | 6 | 46 | 105/14 | 0.67 | 64.6 | 1.42 | 13.83% | 0.94% | 0.15% | Hydroxychloroquine, Oral steroids |
| 8 | F | 39 | Black | 13.1 | 6 | n.d. | 105/18 | 0.75 | 32.7 | 0.8 | 8.75% | 1.36% | 0.05% | Hydroxychloroquine, Mycophenolate, Oral steroids |
| 9 | F | 45 | White | 18.1 | 4 | n.d. | 186/35 | 0.67 | 1.8 | 2.19 | 3.72% | 0.74% | 0.06% | Hydroxychloroquine, Oral steroids |
| 10 | F | 45 | White | 36.8 | 4 | n.d. | 145/44 | 0.89 | 23.8 | 0.76 | 2.04% | 0.81% | 0.03% | Hydroxychloroquine, Oral steroids |
| 11 | F | 28 | Black | 9.9 | 2 | n.d. | 117/23 | 0.52 | 2.2 | 1 | 8.52% | 1.21% | 0.11% | - |
| 12 | F | 39 | White | 26.8 | 0 | n.d. | 145/30 | 2.63 | 16.5 | 1.47 | 3.10% | 0.73% | 0.02% | Hydroxychloroquine, Oral steroids |
| 13 | F | 39 | Other | 21.1 | 3 | - | 65/12 | 0.7 | 17.8 | 0.56 | 9.87% | 1.60% | 0.08% | Hydroxychloroquine, Mycophenolate |
| 14 | F | 61 | White | 36.6 | 8 | 12 | 121/28 | 0.83 | 306.2 | 0.12 | 7.41% | 0.88% | 0.04% | Oral steroids |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamamoto, E.A.; Nguyen, J.K.; Liu, J.; Keller, E.; Campbell, N.; Zhang, C.-J.; Smith, H.R.; Li, X.; Jørgensen, T.N. Low Levels of Vitamin D Promote Memory B Cells in Lupus. Nutrients 2020, 12, 291. https://doi.org/10.3390/nu12020291
Yamamoto EA, Nguyen JK, Liu J, Keller E, Campbell N, Zhang C-J, Smith HR, Li X, Jørgensen TN. Low Levels of Vitamin D Promote Memory B Cells in Lupus. Nutrients. 2020; 12(2):291. https://doi.org/10.3390/nu12020291
Chicago/Turabian StyleYamamoto, Erin A., Jane K. Nguyen, Jessica Liu, Emma Keller, Nicole Campbell, Cun-Jin Zhang, Howard R. Smith, Xiaoxia Li, and Trine N Jørgensen. 2020. "Low Levels of Vitamin D Promote Memory B Cells in Lupus" Nutrients 12, no. 2: 291. https://doi.org/10.3390/nu12020291
APA StyleYamamoto, E. A., Nguyen, J. K., Liu, J., Keller, E., Campbell, N., Zhang, C.-J., Smith, H. R., Li, X., & Jørgensen, T. N. (2020). Low Levels of Vitamin D Promote Memory B Cells in Lupus. Nutrients, 12(2), 291. https://doi.org/10.3390/nu12020291





