Her-2 Breast Cancer Outcomes Are Mitigated by Consuming n-3 Polyunsaturated, Saturated, and Monounsaturated Fatty Acids Compared to n-6 Polyunsaturated Fatty Acids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Mammary Tumour Measurements and Tissue Collection
2.3. Fatty Lipid Analysis
2.4. Statistical Analysis
3. Results
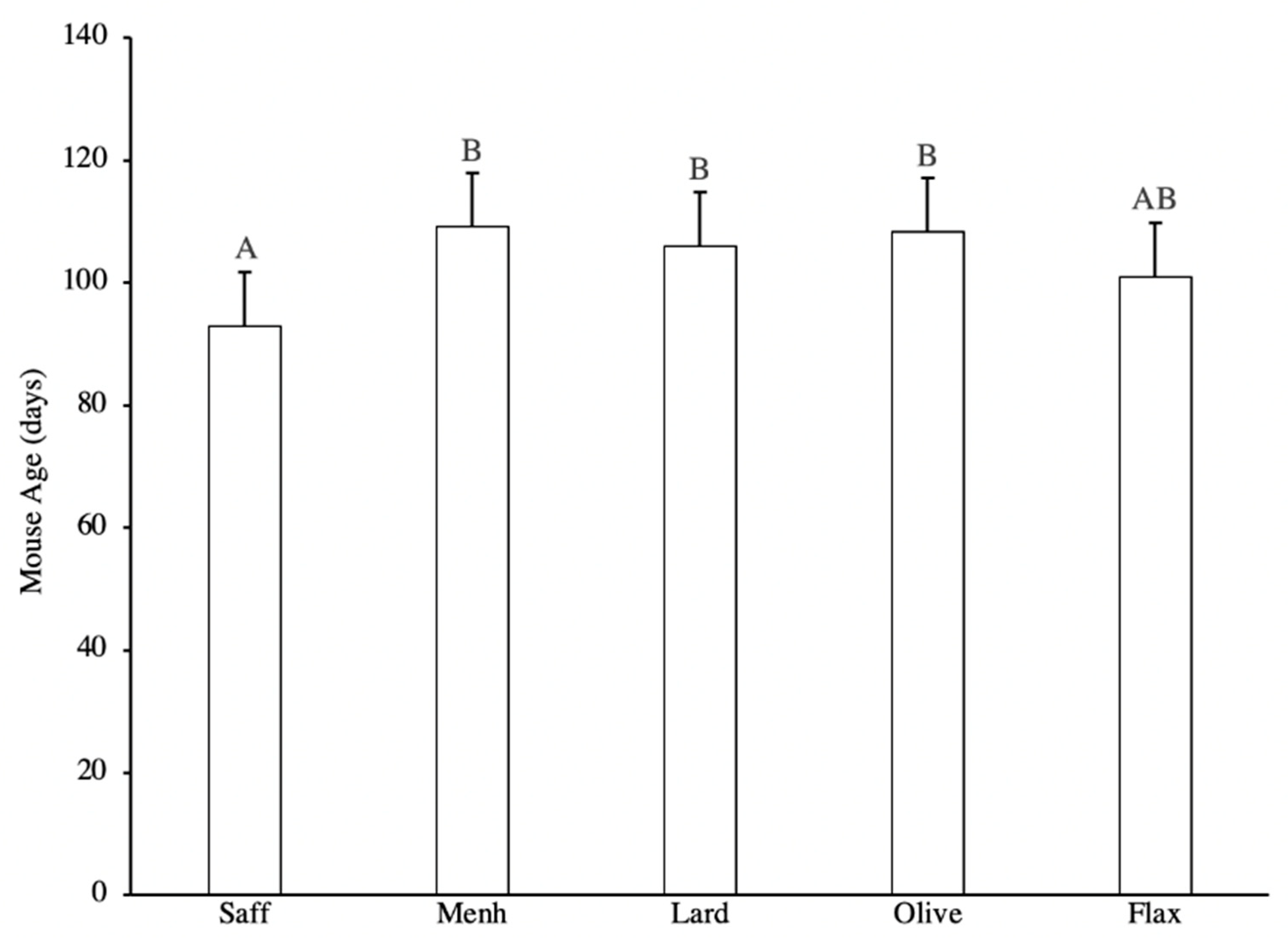
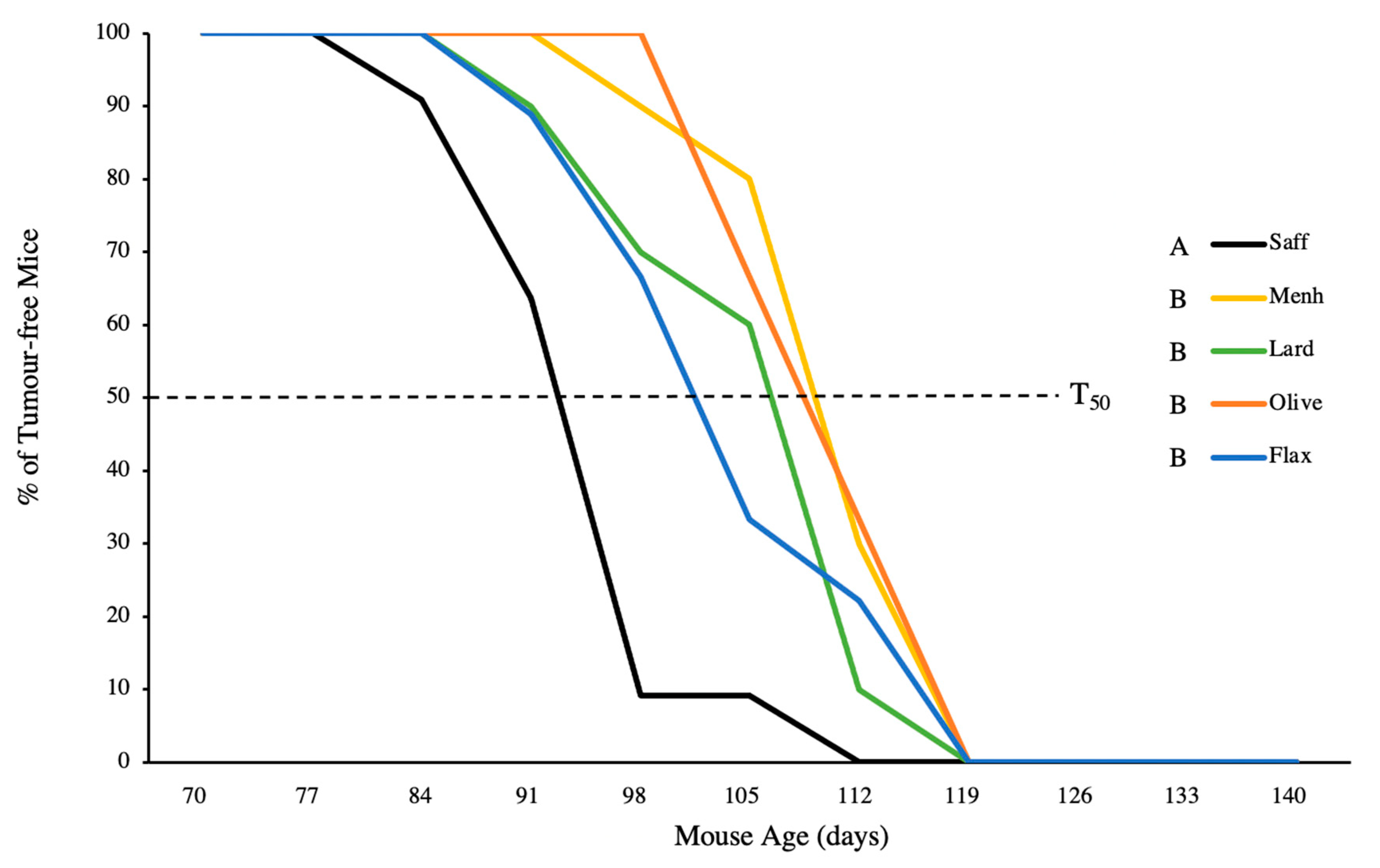
3.1. Tumour Latency and Tumour-Free Status
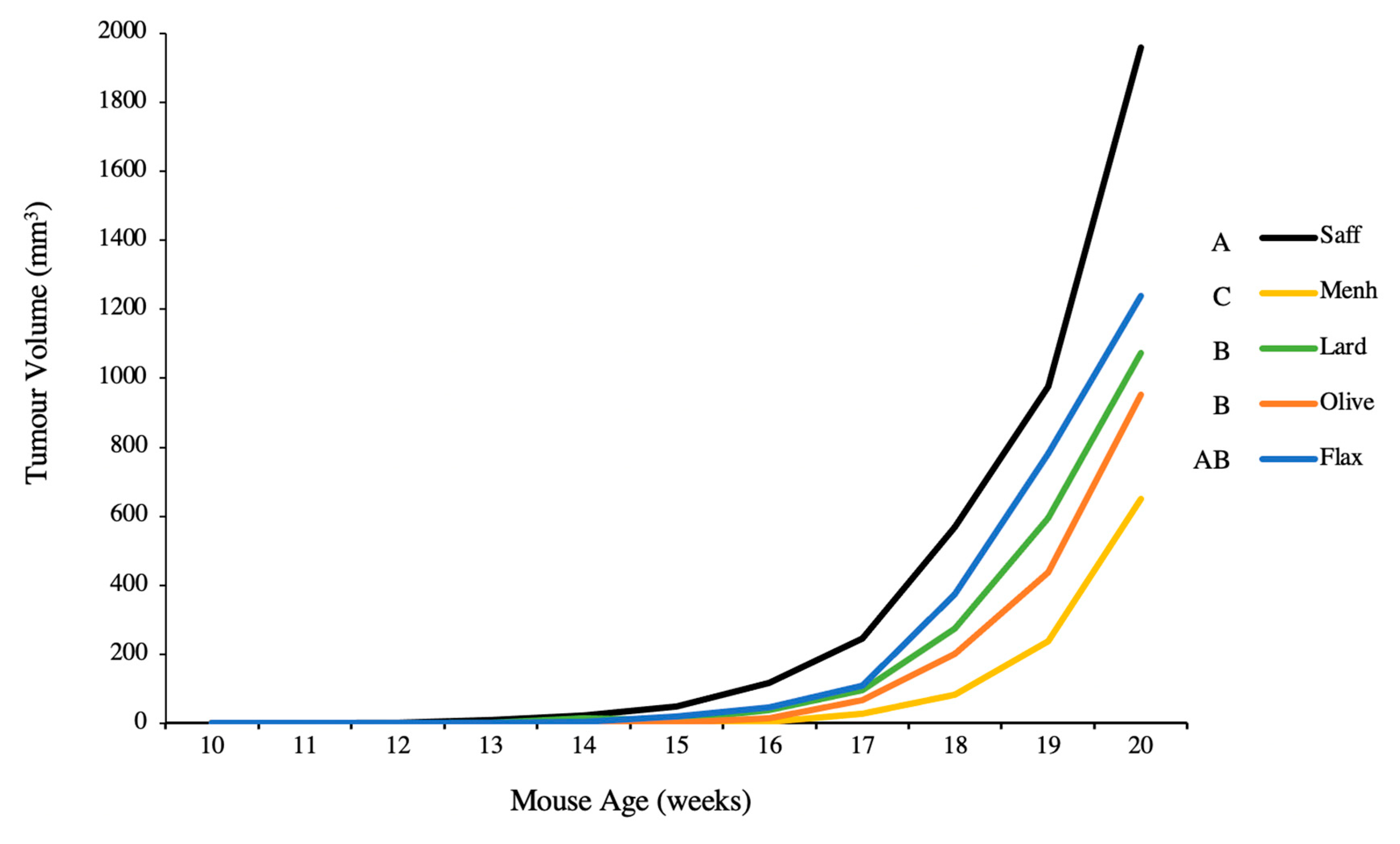
3.2. Tumour Volume
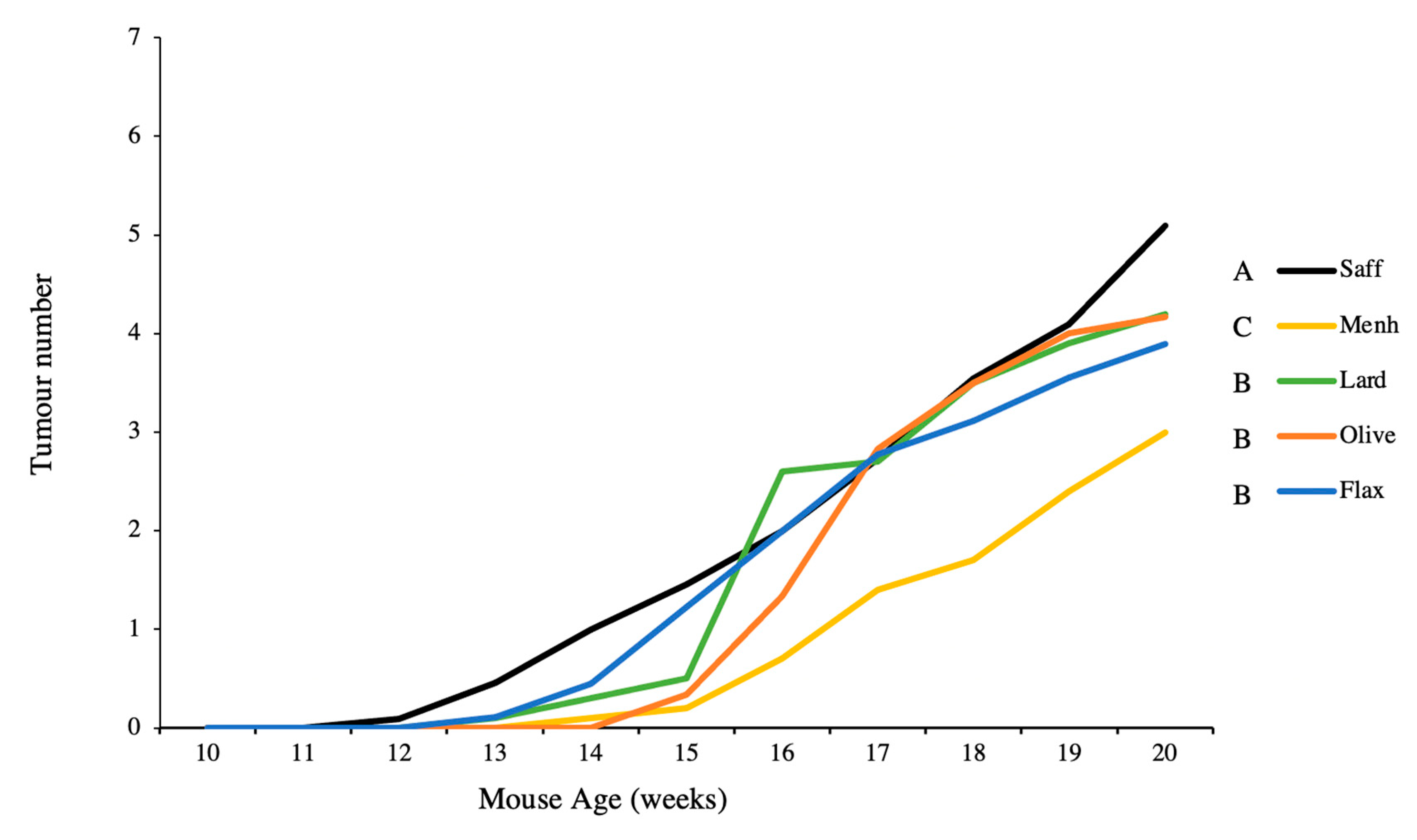
3.3. Tumour Multiplicity
3.4. Fatty Acid Composition of Tumour Phospholipids
4. Discussion
4.1. Role of Dietary Fat in Modulating Mammary Tumour Outcomes
4.2. Tumour Fatty Acid Composition and Tumourigenesis
4.3. Relevance of Model and Diets
5. Strengths, Limitations, and Future Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| 10% Safflower a (D04092701) | 3% Menhaden b (D04092703) | 3% Flaxseed c (D04092711N) | 10% Olive d (D16012101) | 10% Lard e (D16012401) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Macronutrient | g% | kcal% | g% | kcal% | g% | kcal% | g% | kcal% | g% | kcal% |
| Protein | 21 | 20 | 21 | 20 | 21 | 20 | 21 | 20 | 21 | 20 |
| Carbohydrate | 60 | 58 | 60 | 58 | 60 | 58 | 60 | 58 | 60 | 58 |
| Fat | 10 | 22 | 10 | 22 | 10 | 22 | 10 | 22 | 10 | 22 |
| Total | 100 | 100 | 100 | 100 | 100 | |||||
| kcal/g | 4 | 4 | 4 | 4 | 4 | |||||
| Ingredient | g | kcal | g | kcal | g | kcal | g | kcal | g | kcal |
| Casein | 200 | 800 | 200 | 800 | 200 | 800 | 200 | 800 | 200 | 800 |
| L-Cystine | 3 | 12 | 3 | 12 | 3 | 12 | 3 | 12 | 3 | 12 |
| Corn starch | 337 | 1347 | 337 | 1347 | 337 | 1347 | 337 | 1347 | 337 | 134 |
| Maltodextrin 10 | 132 | 528 | 132 | 528 | 132 | 528 | 132 | 528 | 132 | 528 |
| Sucrose | 100 | 400 | 100 | 400 | 100 | 400 | 100 | 400 | 100 | 400 |
| Cellulose, BW200 | 50 | 0 | 50 | 0 | 50 | 0 | 50 | 0 | 50 | 0 |
| Safflower oil | 97 | 873 | 68 | 611 | 68 | 611 | 0 | 0 | 0 | 0 |
| Menhaden oil | 0 | 0 | 29 | 262 | 0 | 0 | 0 | 0 | 0 | 0 |
| Flax oil | 0 | 0 | 0 | 0 | 29 | 262 | 0 | 0 | 0 | 0 |
| Olive oil | 0 | 0 | 0 | 0 | 0 | 0 | 97 | 873 | 0 | 0 |
| Lard | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 97 | 873 |
| t-Butylhydroquinone | 0.02 | 0 | 0.02 | 0 | 0.02 | 0 | 0.02 | 0 | 0.02 | 0 |
| Mineral Mix S10022 | 35 | 0 | 35 | 0 | 35 | 0 | 35 | 0 | 35 | 0 |
| Vitamin mix V10037 | 10 | 40 | 10 | 40 | 10 | 40 | 10 | 40 | 10 | 40 |
| Choline bitartrate | 2.5 | 0 | 2.5 | 0 | 2.5 | 0 | 2.5 | 0 | 2.5 | 0 |
| Total | 966 | 4000 | 966 | 4000 | 966 | 4000 | 966 | 4000 | 966 | 4000 |
References
- American Cancer Society. Breast Cancer Facts & Figures 2019–2020. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2019-2020.pdf (accessed on 30 July 2020).
- Anand, P.; Kunnumakara, A.B.; Sundaram, C.; Harikumar, K.B.; Thakaran, S.T.; Lai, O.K.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that required major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef]
- Parkin, D.M.; Boyd, L.; Walker, L.C. 16. The fraction of cancer attributable to lifestyle and environmental factors in the UK in 2010. Br. J. Cancer 2011, 105, S77–S81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerber, M. Omega-3 fatty acids and cancers: A systematic update review of epidemiological studies. Br. J. Nutr. 2012, 107, S228–S239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, B.M.; Ma, D.W. Are all n-3 polyunsaturated fatty acids created equal? Lipids Health Dis. 2009, 8, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Ma, D.W.L. The role of n-3 polyunsaturated fatty acids in the prevention and treatment of breast cancer. Nutrients 2014, 6, 5184–5223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziegler, R.G.; Hoover, R.N.; Pike, M.C.; Hildesheim, A.; Nomura, A.M.; West, D.W.; Wu-Williams, A.H.; Kolonel, L.N.; Horn-Ross, P.L.; Rosenthal, J.F.; et al. Migration patterns and breast cancer risk in Asian-American women. J. Natl. Cancer Inst. 1993, 85, 1819–1827. [Google Scholar] [CrossRef]
- Saadatian-Elahi, M.; Norat, T.; Goudable, J.; Riboli, E. Biomarkers of dietary fatty acid intake and the risk of breast cancer: A meta-analysis. Int. J. Cancer 2004, 111, 584–591. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Hilakivi-Clarke, L.; Clarke, R.; Onojafe, I.; Raygada, M.; Cho, E.; Lippman, M. A maternal diet high in n-6 polyunsaturated fats alters mammary gland development, puberty onset, and breast cancer risk among female offspring. Nutrition 1997, 15, 392–401. [Google Scholar] [CrossRef]
- Liu, J. “Plant- and Marine-derived N-3 Polyunsaturated Fatty Acids Prevent Mammary Tumor Development.” The Atrium, University of Guelph. 2015. Available online: https://atrium.lib.uoguelph.ca/xmlui/handle/10214/9126 (accessed on 9 June 2020).
- Winikka, L.; Quach, D.; Harlow, B.; Brenner, A.; Munoz, N.; Tiziani, S.; de Graffenried, L. Abstract P1-03-12: The ratio of omega-3 to omega-6 PUFAs impact cancer cell phenotype in the tumor microenvironment. Cancer Res. 2018, 78. [Google Scholar] [CrossRef]
- Dydjow-Bendek, D.; Zagoźdźon, P. Total Dietary Fats, Fatty Acids, and Omega-3/Omega-6 Ratio as Risk Factors of Breast Cancer in the Polish Population—A Case-Control Study. In Vivo 2019, 34, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Mourouti, N.; Panagiotakos, D.B. The beneficial effect of a Mediterranean diet supplemented with extra virgin olive oil in the primary prevention of breast cancer among women at high cardiovascular risk in the PREDIMED Trial. Evid. Based Nurs. 2016, 19, 71. [Google Scholar] [CrossRef] [PubMed]
- Menendez, J.A.; Papadimitropoulou, A.; Vellon, L.; Lupu, R. A genomic explanation connecting “Mediterranean diet,” olive oil and cancer: Oleic acid, the main monounsaturated fatty acid of olive oil, induces formation of inhibitory “PEA3 transcription factor PEA3 DNA binding site” complexes at the Her-2/neu (erbB-2) oncogene promoter in breast, ovarian and stomach cancer cells. Eur. J. Cancer 2006, 42, 2425–2432. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Landau, J.M.; Huang, M.T.; Newmark, H.L. Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu. Rev. Nutr. 2001, 21, 281–406. [Google Scholar] [CrossRef] [Green Version]
- Van den Brandt, P.A.; Schulpen, M. Mediterranean diet adherance and risk of postmenopausal breast cancer: Results of a cohort study and meta-analysis. Int. J. Cancer 2017, 140, 2220–2231. [Google Scholar] [CrossRef]
- Sieri, S.; Chiodini, P.; Agnoli, C.; Pala, V.; Berrino, F.; Trichopoulou, A. Dietary fat intake and development of specific breast cancer subtypes. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef] [Green Version]
- Turner, L.B. A meta-analysis of fat intake, reproduction, and breast cancer risk: An evolutionary perspective. Am. J. Hum. Biol. 2011, 23, 601–608. [Google Scholar] [CrossRef]
- Bassett, J.K.; Hodge, A.M.; English, D.R.; MacInnis, R.J.; Giles, G.G. Plasma phospholipid fatty acids, dietary fatty acids, and breast cancer risk. Cancer Causes Control 2016, 27, 759–773. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Fats and Fatty Acids in Human Nutrition: Report of an Expert Consultation. Available online: http://www.fao.org/3/a-i1953e.pdf (accessed on 9 June 2020).
- Xia, H.; Ma, S.; Wang, S.; Sun, G. Meta-Analysis of Saturated Fatty Acid Intake and Breast Cancer Risk. Medicine 2015, 94, e2391. [Google Scholar] [CrossRef]
- Liu, J.; Abdelmagid, S.A.; Pinelli, C.J.; Monk, J.M.; Liddle, D.M.; Hillyer, L.M.; Hucik, B.; Silva, A.; Subedi, S.; Wood, G.A.; et al. Marine fish oil is more potent than plant-based n-3 polyunsaturated fatty acids in the prevention of mammary tumors. J. Nutr. Biochem. 2018, 55, 41–52. [Google Scholar] [CrossRef]
- Salem, N.; Litman, B.; Kim, H.Y.; Gawrisch, K. Mechanisms of action of docosahexanoic acid in the nervous system. Lipids 2001, 36, 945–959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacLennan, M.B.; Clarke, S.E.; Perez, K.; Wood, G.A.; Muller, W.J.; Kang, J.X.; Ma, D.W. Mammary tumor development is directly inhibited by lifelong n-3 polyunsaturated fatty acids. J. Nutr. Biochem. 2013, 24, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Hamid, R.; Singh, J.; Reddy, B.S.; Cohen, L.A. Inhibition by dietary menhaden oil of cyclooxygenase-1 and -2 in N-nitrosomethylurea-induced rat mammary tumors. Int. J. Oncol. 1999, 14, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Hardman, W.E.; Avula, C.P.; Fernandes, G.; Cameron, I.L. Three percent dietary fish oil concentrate increased efficacy of doxorubicin against MDA-MB 231 breast cancer xenografts. Clin. Cancer Res. 2001, 7, 2041–2049. [Google Scholar] [PubMed]
- Hardman, W.E. Omega-3 fatty acids to augment cancer therapy. J. Nutr. 2002, 132, 3508S–3512S. [Google Scholar] [CrossRef] [Green Version]
- Rose, D.P.; Connolly, J.M.; Rayburn, J.; Coleman, M. Influence of diets containing eicosapentaenoic or docosahexaenoic acid on growth and metastasis of breast cancer cells in nude mice. J. Natl. Cancer Inst. 1995, 87, 587–592. [Google Scholar] [CrossRef]
- Rose, D.P.; Connolly, J.M. Regulation of tumor angiogenesis by dietary fatty acids and eicosanoids. Nutr. Cancer 2000, 37, 119–127. [Google Scholar] [CrossRef]
- Senzaki, H.; Iwamoto, S.; Ogura, E.; Kiyozuka, Y.; Arita, S.; Kurebayashi, J.; Takada, H.; Hioki, K.; Tsubura, A. Dietary effects of fatty acids on growth and metastasis of KPL-1 human breast cancer cells in vivo and in vitro. Anticancer Res. 1998, 18, 1621–1627. [Google Scholar]
- Li, S.; Zhou, T.; Li, C.; Dai, Z.; Che, D.; Yao, Y.; Li, L.; Ma, J.; Yang, X.; Gao, G. High Metastaticgastric and breast cancer cells consume oleic acid in an AMPK dependent manner. PLoS ONE 2014, 9, e97330. [Google Scholar] [CrossRef]
- Banim, P.R.; Luben, R.; Mctaggart, A.; Khaw, K.-T.; Wareham, N.; Hart, A. Do Oleic Acid and N-3 Fatty Acids Prevent Pancreatic Cancer? Data from a UK Prospective Cohort Study Using 7-Day Food Diaries. Gastroenterology 2011, 140. [Google Scholar] [CrossRef]
- Akl, M.R.; Ayoub, N.M.; Mohyeldin, M.M.; Busnena, B.A.; Foudah, A.I.; Liu, Y.Y.; Sayed, K.A. Olive phenolics as c-Met inhibitors: (-)-Oleocanthal attenuates cell proliferation, invasiveness, and tumor growth in breast cancer models. PLoS ONE 2014, 9, e97622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Storniolo, C.E.; Martínez-Hovelman, N.; Martínez-Huélamo, M.; Lamuela-Raventos, R.M.; Moreno, J.J. Extra Virgin Olive Oil Minor Compounds Modulate Mitogenic Action of Oleic Acid on Colon Cancer Cell Line. J. Agric. Food Chem. 2019, 67, 11420–11427. [Google Scholar] [CrossRef] [PubMed]
- Liotti, A.; Cosimato, V.; Mirra, P.; Calì, G.; Conza, D.; Secondo, A.; Luongo, G.; Terracciano, D.; Formisano, P.; Beguinot, F.; et al. Oleic acid promotes prostate cancer malignant phenotype via the G protein-coupled receptor FFA1/GPR40. J. Cell Physiol. 2018, 233, 7367–7378. [Google Scholar] [CrossRef] [PubMed]
- Xiang, F.; Wu, K.; Liu, Y.; Shi, L.; Wang, D.; Li, G.; Tao, K.; Wang, G. Omental adipocytes enhance the invasiveness of gastric cancer cells by oleic acid-induced activation of the PI3K-Akt signaling pathway. Int. J. Biochem. Cell Biol. 2017, 84, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Su, C.; Luo, X.; Zeng, H.; Zhao, L.; Wei, L.; Zhang, X.; Varghese, Z.; Moorhead, J.F.; Chen, Y.; et al. Dietary oleic acid-induced CD36 promotes cervical cancer cell growth and metastasis via up-regulation Src/ERK pathway. Cancer Lett. 2018, 438, 76–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coccia, A.; Mosca, L.; Puca, R.; Mangino, G.; Rossi, A.; Lendaro, E. Extra-virgin olive oil phenols block cell cycle progression and modulate chemotherapeutic toxicity in bladder cancer cells. Oncol. Rep. 2016, 36, 3095–3104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, Y.; Yuan, C.; Zhang, J.; Zhang, F.; Fu, Q.; Zhu, X.; Shu, G.; Wang, L.; Gao, P.; Xi, Q.; et al. Stearic acid suppresses mammary gland development by inhibiting PI3K/Akt signaling pathway through GPR120 in pubertal mice. Biochem. Biophys. Res. Comm. 2017, 491, 192–197. [Google Scholar] [CrossRef]
- Meng, Y.; Zhang, J.; Zhang, F.; Ai, W.; Zhu, X.; Shu, G.; Wang, L.; Gao, P.; Xi, Q.; Zhang, Y.; et al. Lauric acid stimulates mammary gland development of pubertal mice through activation of GPR84 and PI3L/Akt Signaling Pathway. J. Agric. Food Chem. 2017, 65, 95–103. [Google Scholar] [CrossRef]
- Binker-Cosen, M.J.; Richards, D.; Oliver, B.; Gaisano, H.Y.; Binker, M.G.; Cosen-Binker, L.I. Palmitic acid increases invasiveness of pancreatic cancer cells AsPC-1 through TLR4/ROS/NF-κB/MMP-9 signaling pathway. Biochem. Biophys. Res. Commun. 2017, 484, 152–158. [Google Scholar] [CrossRef]
- Wicha, M.S.; Lance, A.L.; Kidwell, W.R. Effects of Free Fatty Acids on the Growth of Normal and Neoplastic Rat Mammary Epithelial Cells. Cancer Res. 1979, 39, 426–435. [Google Scholar]
- Dong, J.Y.; Zhang, L.; He, K.; Qin, L.Q. Dairy consumption and risk of breast cancer: A meta-analysis of prospective cohort studies. Breast Cancer Res. Treat. 2011, 127, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Wei, W.; Zhan, L. Red and processed meat intake and risk of breast cancer: A meta-analysis of prospective studies. Breast Cancer Res. Treat. 2015, 151, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patra, S.K. Dissecting lipid raft facilitated cell signaling pathways in cancer. Biochem. Biophys. Acta 2008, 1785, 182–206. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Yun, U.J.; Koo, K.H.; Sung, J.Y.; Shim, J.; Ye, S.K.; Hong, K.M.; Kim, Y.N. Down-regulation of lipid raft-associated onco-proteins via cholesterol-dependent lipid raft internalization in docosahexaenoic acid-induced apoptosis. Biochim. Biophys. Acta 2014, 1841, 190–203. [Google Scholar] [CrossRef]
- Burdge, G.C.; Calder, P.C. Conversion of α-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597. [Google Scholar] [CrossRef]
- Benoit, V.; Relic, B.; Leval, X.; Chariot, A.; Merville, M.P.; Bours, V. Regulation of HER-2 oncogene expression by cyclooxygenase-2 and prostaglandin E2. Oncogene 2004, 23, 1631–1635. [Google Scholar] [CrossRef] [Green Version]
- Larsson, S.C.; Kumlin, M.; Ingelman-Sundberg, M.; Wolk, A. Dietary long-chain n-3 fatty acids for the prevention of cancer: A review of potential mechanisms. Am. J. Clin. Nutr. 2004, 79, 935–945. [Google Scholar] [CrossRef]
- Wang, D.D.; Hu, F.B. Dietary Fat and Risk of Cardiovascular Disease: Recent Controversies and Advances. Annu. Rev. Nutr. 2017, 37, 423–446. [Google Scholar] [CrossRef]
- Burns, J.L.; Nakamura, M.T.; Ma, D.W. Differentiating the biological effects of linoleic acid from arachidonic acid in health and disease. Prostaglandins Leukot. Essent. Fat. Acids 2018, 135, 1–4. [Google Scholar] [CrossRef]
- Li, Q.; Wang, M.; Tan, L.; Wang, C.; Ma, J.; Li, N.; Li, Y.; Xu, G.; Li, J. Docosahexaenoic acid changes lipid composition and interleukin-2 receptor signaling in membrane rafts. J. Lipid Res. 2005, 46, 1904–1913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravacci, G.R.; Brentani, M.M.; Tortelli, T.; Torrinhas, R.S.; Saldanha, T.; Torres, E.A.; Waitzberg, D.L. Lipid raft disruption by docosahexaenoic acid induces apoptosis in transformed human mammary luminal epithelial cells harboring her-2 overexpression. J. Nutr. Biochem. 2013, 24, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Muller, W.J.; Sinn, E.; Pattengale, P.K.; Wallace, R.; Leder, P. Single-step induction of mammary adenocarcinoma in transgenic mice bearing the activated c-neu oncogene. Cell 1988, 54, 105–115. [Google Scholar] [CrossRef]
- Statistics Canada. Table 13-10-0769-01 Percentage of Total Energy Intake from Fat, by Dietary Reference Intake Age-Sex Group, Household Population Aged 1 and over, Canadian Community Health Survey (CCHS)—Nutrition, Canada and Provinces. Available online: https://doi.org/10.25318/1310076901-eng (accessed on 20 June 2020).
- Harris, W.S.; Mozaffarian, D.; Rimm, E.; Kris-Etherton, P.; Rudel, L.L.; Appel, L.J.; Engler, M.M.; Engler, M.B.; Sacks, F. Omega-6 fatty acids and risk for cardiovascular disease: A science advisory from the American Heart Association Nutrition Subcommittee of the Council on Nutrition, Physical Activity, and Metabolism; Council on Cardiovascular Nursing; and Council on Epidemiology and Prevention. Circulation 2009, 119, 902–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, W.; McMurray, D.N.; Chapkin, R.S. n-3 polyunsaturated fatty acids—Physiological relevance of dose. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 155–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- U.S. Departments of Health and Human Services (HHS) and Agriculture (USDA), Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee. Available online: http://www.health.gov/dietaryguidelines/2015-scientific-report/ (accessed on 9 June 2020).
- Agence Française de Sécurité Sanitaire des Aliments (AFSSA). Avis de l’Agence française de sécurité sanitaire des aliments relatif à l’actualisation des apports nutritionnels conseillés pour les acides gras (Opinion of the French Food Safety Agency on the Update of French Population Reference Intakes (RDIs) for Fatty Acids). Request No. 2006-SA-0359. 2010. Available online: https://www.anses.fr/fr/content/avis-de-l%E2%80%99agence-fran%C3%A7aise-de-s%C3%A9curit%C3%A9-sanitaire-des-aliments-relatif-%C3%A0-l%E2%80%99actualisation-de-2 (accessed on 15 December 2020).
- Barlow, L.J.; Zhang, J.-T. Abstract 4415: Fatty acid synthase-mediated palmitate production impacts epidermal growth factor receptor signaling to regulate specificity protein 1 in breast cancer cells. Mol. Cell Biol. Genet. 2017, 77. [Google Scholar] [CrossRef]
- Holder, A.M.; Gonzalez-Angulo, A.M.; Chen, H.; Akcakanat, A.; Anh-Do, K.; Symmans, F.; Pusztai, L.; Hortobagyi, G.; Mills, G.B.; Meric-Bernstam, F. Abstract 682: Increased stearoyl-CoA desaturase 1 expression is associated with shorter survival in breast cancer patients. Clin. Res. 2012, 72. [Google Scholar] [CrossRef]
| Fatty Acid | 10% Safflower Oil | 3% Menhaden Oil | 3% Flaxseed Oil | 10% Olive Oil | 10% Lard |
|---|---|---|---|---|---|
| 12:0 | 0.04 | 0.08 | 0.03 | 0.04 | 1.49 |
| 14:0 | 0.24 | 2.67 | 0.22 | 0.16 | 1.28 |
| 15:0 | 0 | 0.26 | 0.03 | 0 | 0.11 |
| 16:0 | 6.91 | 10.11 | 6.52 | 14.51 | 21.27 |
| 16:1c9 | 0.15 | 3.99 | 1.13 | 1.98 | 1.67 |
| 17:1c10 | 0 | 0.19 | 0.02 | 0.13 | 0.24 |
| 18:0 | 2.59 | 2.85 | 2.89 | 2.11 | 11.51 |
| 18:1c9 | 15.25 | 13.78 | 16.59 | 64.50 | 35.76 |
| 18:1c11 | 0.80 | 1.47 | 0.78 | 3.51 | 2.33 |
| 18:2n6 | 71.93 | 51.71 | 53.81 | 11.15 | 20.85 |
| 18:3n6 | 0.11 | 0.16 | 0 | 0 | 0.11 |
| 19:0 | 0 | 0 | 0 | 0 | 0.10 |
| 19:1c7 | 0 | 0.18 | 0 | 0 | 0 |
| 18:3n3 | 0.31 | 0.86 | 17.30 | 0.66 | 1.56 |
| 18:4n3 | 0.13 | 0.90 | 0.14 | 0 | 0.17 |
| 20:0 | 0.36 | 0.36 | 0.51 | 0.39 | 0.22 |
| 20:1c5&8 | 0.10 | 0 | 0 | 0 | 0 |
| 20:1c11 | 0.22 | 0.49 | 0.34 | 0.37 | 0.79 |
| 20:2n6 | 0.05 | 0.12 | 0.09 | 0 | 0.84 |
| 20:3n6 | 0 | 0.10 | 0 | 0 | 0.17 |
| 20:4n6 | 0 | 0.49 | 0 | 0 | 0.37 |
| 20:3n3 | 0 | 0.20 | 0 | 0 | 0.25 |
| 20:5n3 | 0.05 | 4.20 | 0 | 0 | 0 |
| 22:0 | 0.30 | 0.27 | 0.28 | 0.13 | 0 |
| 22:1n9 | 0.02 | 0.09 | 0.02 | 0.03 | 0.02 |
| 22:2n6 | 0 | 0.27 | 0 | 0 | 0 |
| 22:4n6 | 0.11 | 0.14 | 0 | 0 | 0.12 |
| 22:5n3 | 0 | 0.75 | 0 | 0 | 0.12 |
| 24:0 | 0.14 | 0.02 | 0.16 | 0.12 | 0.01 |
| 22:6n3 | 0 | 3.08 | 0 | 0 | 0 |
| 24:1 | 0.17 | 0.23 | 0.15 | 0.19 | 0 |
| Total n-6 | 72.20 | 52.99 | 53.90 | 11.15 | 22.46 |
| Total n-3 | 0.49 | 9.99 | 17.44 | 0.66 | 2.1 |
| Total SFA | 10.58 | 16.62 | 10.64 | 17.46 | 35.99 |
| Total MUFA | 16.71 | 43.19 | 19.03 | 70.71 | 40.81 |
| Total PUFA | 72.69 | 62.98 | 71.34 | 11.81 | 24.56 |
| Fatty Acids | 10% Safflower | 3% Menhaden | 3% Flaxseed | 10% Olive | 10% Lard |
|---|---|---|---|---|---|
| 12:0 | 0.04 ± 0.06 | 0.15 ± 0.09 | 0.09 ± 0.01 | 0.30 ± 0.29 | 0.07 ± 0.03 |
| 14:0 | 1.06 ± 0.31 | 1.25 ± 0.34 | 0.81 ± 0.06 | 0.83 ± 0.33 | 1.25 ± 0.16 |
| 15:0 | 0.24 ± 0.04 | 0.24 ± 0.17 | 0.22 ± 0.03 | 0.24 ± 0.05 | 0.25 ± 0.02 |
| 16:0 | 29.21 ± 1.16 | 29.39 ± 1.01 | 27.75 ± 2.08 | 25.81 ± 2.77 | 27.74 ± 3.07 |
| 18:0 | 8.32 ± 1.04 | 8.61 ± 2.58 | 8.24 ± 0.82 | 6.95 ± 1.68 | 7.25 ± 1.21 |
| 19:0 | 0.13 ± 0.08 | 0.14 ± 0.07 | 0.13 ± 0.02 | 0.15 ± 0.08 | 0.10 ± 0.03 |
| 20:0 | 0.11 ± 0.03 | 0.12 ± 0.05 | 0.19 ± 0.18 | 0.08 ± 0.02 | 0.08 ± 0.01 |
| 24:0 | 0.33 ± 0.13 B | 0.56 ± 0.23 A | 0.24 ± 0.05 B | 0.24 ± 0.06 B | 0.14 ± 0.07 B |
| Total SFA | 39.44 | 40.47 | 37.68 | 34.60 | 36.88 |
| 16:1c-9 | 2.80 ± 0.79 | 3.34 ± 1.01 | 2.84 ± 0.33 | 3.62 ± 1.06 | 3.84 ± 0.44 |
| 18:1c-9 | 13.03 ± 1.60 C | 13.76 ± 1.72 C | 13.80 ± 1.61 C | 23.63 ± 1.23A | 19.88 ± 1.10 B |
| 18:1c-11 | 5.16 ± 0.61 B | 6.92 ± 1.94 AB | 5.37 ± 0.92 B | 7.86 ± 1.03 A | 8.14 ± 1.28 A |
| 20:1c-11 | 0.86 ± 0.31 | 0.83 ± 0.41 | 0.74 ± 0.33 | 1.28 ± 0.27 | 1.11 ± 0.20 |
| 22:1n-9 | 0.48 ± 0.14 | 0.37 ± 0.17 | 0.18 ± 0.09 | 0.37 ± 0.18 | 0.34 ± 0.24 |
| 24:1n-9 | 0.52 ± 0.25 | 0.59 ± 0.09 | 0.26 ± 0.05 | 0.57 ± 0.38 | 0.41 ± 0.08 |
| Total MUFA | 22.85 | 25.80 | 23.19 | 37.33 | 33.71 |
| 18:2n-6 | 13.11 ± 1.70 B | 12.34 ± 1.83 B | 17.33 ± 2.16 A | 4.79 ± 0.37 C | 7.20 ± 0.58 C |
| 18:3n-6 | 0.22 ± 0.09 A | 0.11 ± 0.05 B | 0.17 ± 0.01 AB | 0.13 ± 0.03 AB | 0.10 ± 0.02 B |
| 18:3n-3 | 0.21 ± 0.22 B | 0.21 ± 0.08 B | 0.49 ± 0.19 A | 0.19 ± 0.08 B | 0.18 ± 0.05 B |
| 20:2n-6 | 2.24 ± 0.75 A | 1.77 ± 0.65 AB | 1.98 ± 0.61 A | 0.45 ± 0.11 C | 1.02 ± 0.17 BC |
| 20:3n-6 | 3.40 ± 1.43 BC | 2.92 ± 0.51 BC | 4.18 ± 0.67 B | 2.21 ± 0.36 C | 2.47 ± 0.27 C |
| 20:4n-6 | 13.89 ± 3.43 AB | 8.85 ± 2.13 C | 10.68 ± 1.93 BC | 16.81 ± 2.50 A | 15.03 ± 1.66 AB |
| 20:5n-3 | 0.54 ± 0.73 BC | 0.89 ± 0.31 B | 0.50 ± 0.16 BC | 0.16 ± 0.05 C | 0.19 ± 0.04 BC |
| 22:2n-6 | 0.31 ± 0.10 A | 0.22 ± 0.06 AB | 0.20 ± 0.07 AB | 0.17 ± 0.09 AB | 0.11 ± 0.07 B |
| 22:4n-6 | 1.20 ± 0.16 A | 0.51 ± 0.16 B | 0.39 ± 0.06 B | 0.74 ± 0.36 B | 0.61 ± 0.06 B |
| 22:5n-6 | 1.10 ± 0.34 A | 0.47 ± 0.19 B | 0.18 ± 0.04 B | 0.46 ± 0.23 B | 0.38 ± 0.09 B |
| 22:5n-3 | 0.41 ± 0.31 B | 1.49 ± 0.61 A | 0.83 ± 0.03 B | 0.36 ± 0.15 B | 0.36 ± 0.10 B |
| 22:6n-3 | 1.06 ± 0.50 B | 3.95 ± 2.89 A | 2.19 ± 0.29 AB | 1.62 ± 0.22 B | 1.77 ± 0.11 B |
| Total PUFA | 37.70 | 33.73 | 39.13 | 28.07 | 29.41 |
| Fatty Acids | 10% Safflower | 3% Menhaden | 3% Flaxseed | 10% Olive | 10% Lard |
|---|---|---|---|---|---|
| 12:0 | 0.04 ± 0.10 B | 0.05 ± 0.08 B | 0.13 ± 0.07 AB | 0.26 ± 0.07 A | 0.23 ± 0.12 A |
| 14:0 | 0.40 ± 0.12 | 0.42 ± 0.19 | 0.39 ± 0.07 | 0.33 ± 0.14 | 0.53 ± 0.16 |
| 15:0 | 0.18 ± 0.14 | 0.09 ± 0.11 | 0.15 ± 0.09 | 0.20 ± 0.13 | 0.24 ± 0.11 |
| 16:0 | 6.79 ± 0.74 A | 7.48 ± 0.65 A | 7.22 ± 0.66 A | 5.00 ± 0.50 B | 6.70 ± 0.25 A |
| 18:0 | 14.25 ± 1.28 | 13.93 ± 2.46 | 13.70 ± 1.12 | 12.03 ± 1.95 | 11.84 ± 1.32 |
| 19:0 | 0.14 ± 0.13 | 0.04 ± 0.07 | 0.12 ± 0.09 | 0.04 ± 0.10 | 0.08 ± 0.10 |
| 20:0 | 0.15 ± 0.03 | 0.11 ± 0.04 | 0.10 ± 0.02 | 0.14 ± 0.05 | 0.09 ± 0.09 |
| 24:0 | 0.79 ± 0.29 A | 0.63 ± 0.28 AB | 0.36 ± 0.19 B | 0.54 ± 0.13 AB | 0.33 ± 0.22 B |
| Total SFA | 22.75 | 22.75 | 22.18 | 18.54 | 20.04 |
| 16:1c9 | 1.45 ± 0.34 B | 1.90 ± 0.37 A | 1.90 ± 0.22 A | 1.27 ± 0.28 B | 1.92 ± 0.17 A |
| 18:1c9 | 17.55 ± 2.61 C | 18.20 ± 4.59 C | 19.96 ± 1.32 BC | 25.04 ± 3.17 A | 24.08 ± 0.73 AB |
| 18:1c11 | 4.77 ± 0.96 B | 5.65 ± 1.48 AB | 5.38 ± 0.89 AB | 5.83 ± 1.23 AB | 6.83 ± 1.22 A |
| 20:1c11 | 1.07 ± 0.34 AB | 0.88 ± 0.45 AB | 0.74 ± 0.31 B | 1.34 ± 0.30 A | 1.23 ± 0.28 AB |
| 22:1n-9 | 1.36 ± 1.37 A | 0.28 ± 0.09 AB | 0.06 ± 0.10 B | 0.34 ± 0.11 AB | 0.21 ± 0.02 B |
| 24:1n-9 | 0.23 ± 0.08 AB | 0.10 ± 0.12 B | 0.17 ± 0.06 AB | 0.32 ± 0.10 A | 0.15 ± 0.07 B |
| Total MUFA | 26.44 | 27.02 | 28.21 | 34.14 | 34.41 |
| 18:2n-6 | 7.52 ± 0.80 B | 8.75 ± 1.23 B | 11.10 ± 1.98 A | 2.41 ± 0.47 D | 4.38 ± 0.37 C |
| 18:3n-6 | 0.19 ± 0.16 A | 0.05 ± 0.06 AB | 0.12 ± 0.11 AB | 0.01 ± 0.03 B | 0.04 ± 0.04 AB |
| 18:3n-3 | 0.22 ± 0.34 AB | 0.02 ± 0.05 B | 0.46 ± 0.26 A | 0.13 ± 0.14 AB | 0.23 ± 0.12 AB |
| 20:2n-6 | 1.72 ± 0.55 A | 1.14 ± 0.30 ABC | 1.25 ± 0.29 AB | 0.60 ± 0.18 C | 0.72 ± 0.12 BC |
| 20:3n-6 | 3.71 ± 1.54 AB | 2.96 ± 0.84 AB | 4.31 ± 0.44 A | 2.31 ± 0.51 B | 2.65 ± 0.30 B |
| 20:4n-6 | 21.19 ± 4.59 BC | 17.76 ± 3.98 C | 19.01 ± 1.57 C | 27.75 ± 1.91 A | 25.49 ± 0.80 AB |
| 20:5n-3 | 1.11 ± 1.67 AB | 1.69 ± 0.42 A | 0.86 ± 0.15 AB | 0.29 ± 0.11 B | 0.30 ± 0.05 B |
| 22:2n-6 | 0.13 ± 0.10 AB | 0.05 ± 0.10 B | 0.00 ± 0.00 B | 0.27 ± 0.12 A | 0.03 ± 0.04 B |
| 22:4n-6 | 4.13 ± 0.18 A | 1.10 ± 0.52 C | 0.88 ± 0.22 C | 2.50 ± 0.99 B | 1.67 ± 0.34 BC |
| 22:5n-6 | 4.65 ± 1.80 C | 0.65 ± 0.23 B | 0.47 ± 0.13 B | 1.25 ± 0.48 B | 0.82 ± 0.18 B |
| 22:5n-3 | 1.45 ± 1.18 BC | 3.68 ± 1.06 A | 2.63 ± 0.45 AB | 1.08 ± 0.24 C | 1.21 ± 0.13 C |
| 22:6n-3 | 4.78 ± 1.70 C | 12.38 ± 4.10 A | 8.53 ± 1.38 B | 8.72 ± 0.65 B | 8.03 ± 0.59 BC |
| Total PUFA | 50.81 | 50.23 | 49.61 | 47.32 | 45.55 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hillyer, L.M.; Hucik, B.; Baracuhy, E.M.; Lin, Z.; Muller, W.J.; Robinson, L.E.; Ma, D.W.L. Her-2 Breast Cancer Outcomes Are Mitigated by Consuming n-3 Polyunsaturated, Saturated, and Monounsaturated Fatty Acids Compared to n-6 Polyunsaturated Fatty Acids. Nutrients 2020, 12, 3901. https://doi.org/10.3390/nu12123901
Hillyer LM, Hucik B, Baracuhy EM, Lin Z, Muller WJ, Robinson LE, Ma DWL. Her-2 Breast Cancer Outcomes Are Mitigated by Consuming n-3 Polyunsaturated, Saturated, and Monounsaturated Fatty Acids Compared to n-6 Polyunsaturated Fatty Acids. Nutrients. 2020; 12(12):3901. https://doi.org/10.3390/nu12123901
Chicago/Turabian StyleHillyer, Lyn M., Barbora Hucik, Enzo M. Baracuhy, Zhen Lin, William J. Muller, Lindsay E. Robinson, and David W. L. Ma. 2020. "Her-2 Breast Cancer Outcomes Are Mitigated by Consuming n-3 Polyunsaturated, Saturated, and Monounsaturated Fatty Acids Compared to n-6 Polyunsaturated Fatty Acids" Nutrients 12, no. 12: 3901. https://doi.org/10.3390/nu12123901
APA StyleHillyer, L. M., Hucik, B., Baracuhy, E. M., Lin, Z., Muller, W. J., Robinson, L. E., & Ma, D. W. L. (2020). Her-2 Breast Cancer Outcomes Are Mitigated by Consuming n-3 Polyunsaturated, Saturated, and Monounsaturated Fatty Acids Compared to n-6 Polyunsaturated Fatty Acids. Nutrients, 12(12), 3901. https://doi.org/10.3390/nu12123901





