Mumefural Improves Blood Flow in a Rat Model of FeCl3-Induced Arterial Thrombosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. FeCl3-Induced Vascular Injury and Drug Treatment
2.3. Immunostaining
2.4. Pharmacokinetics Analysis
2.5. Statistical Analysis
3. Results
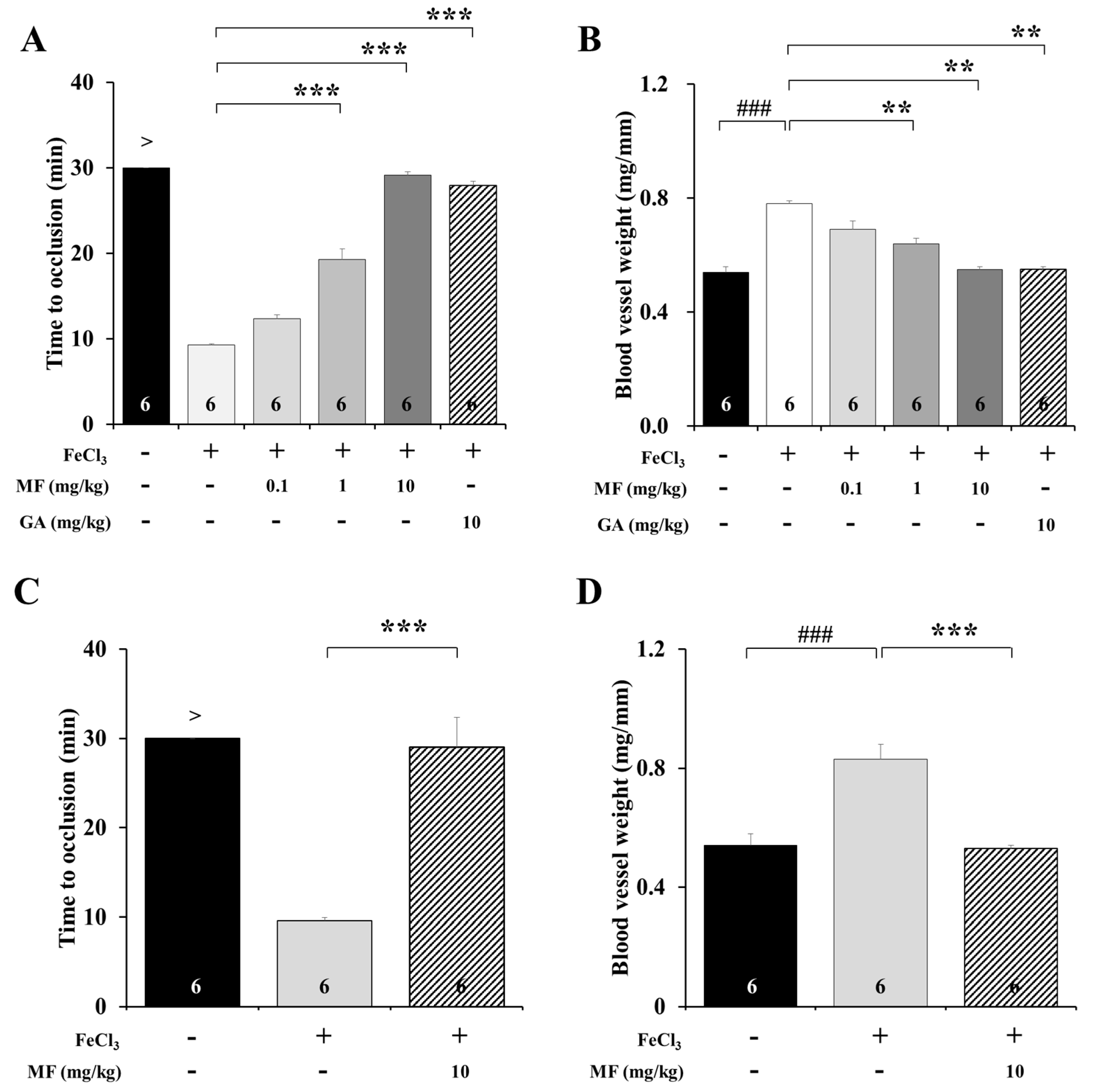
3.1. MF Improves Blood Flow in an FeCl3-Induced Araterial Thrombosis Model
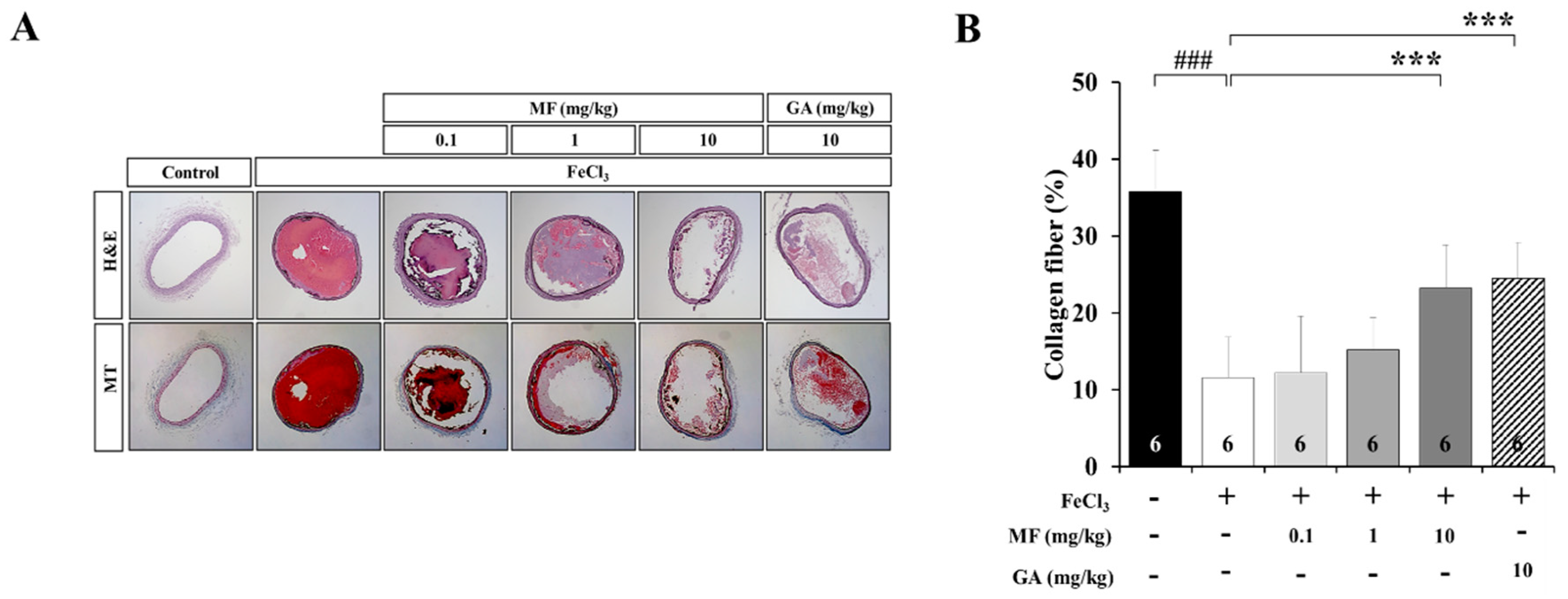
3.2. MF Inhibits Collagen Fiber Damage in an FeCl3-Induced Araterial Thrombosis Model
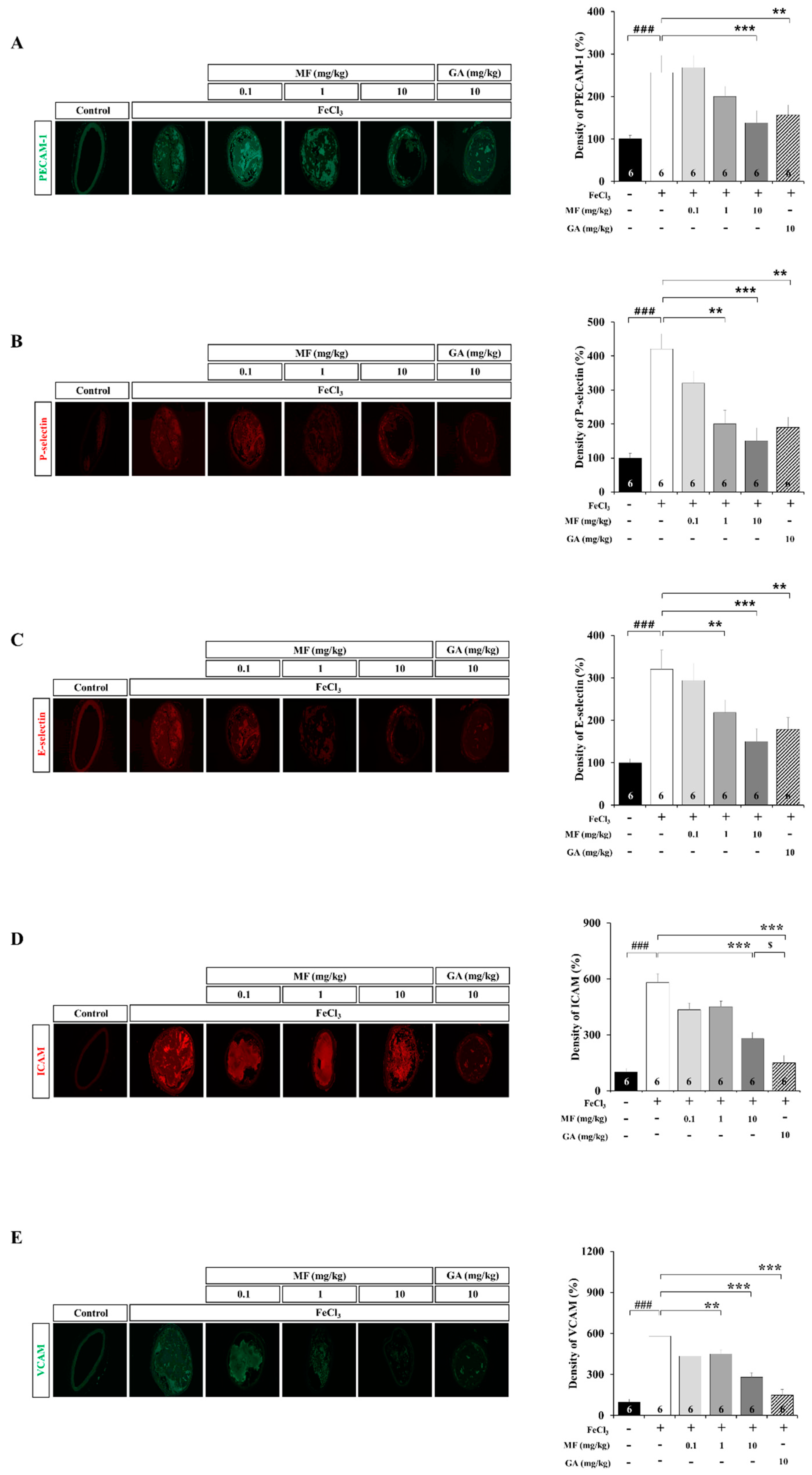
3.3. MF Inhibits Cell Adhesion Molecules (CAMs) in an FeCl3-Induced Arterial Thrombosis Model
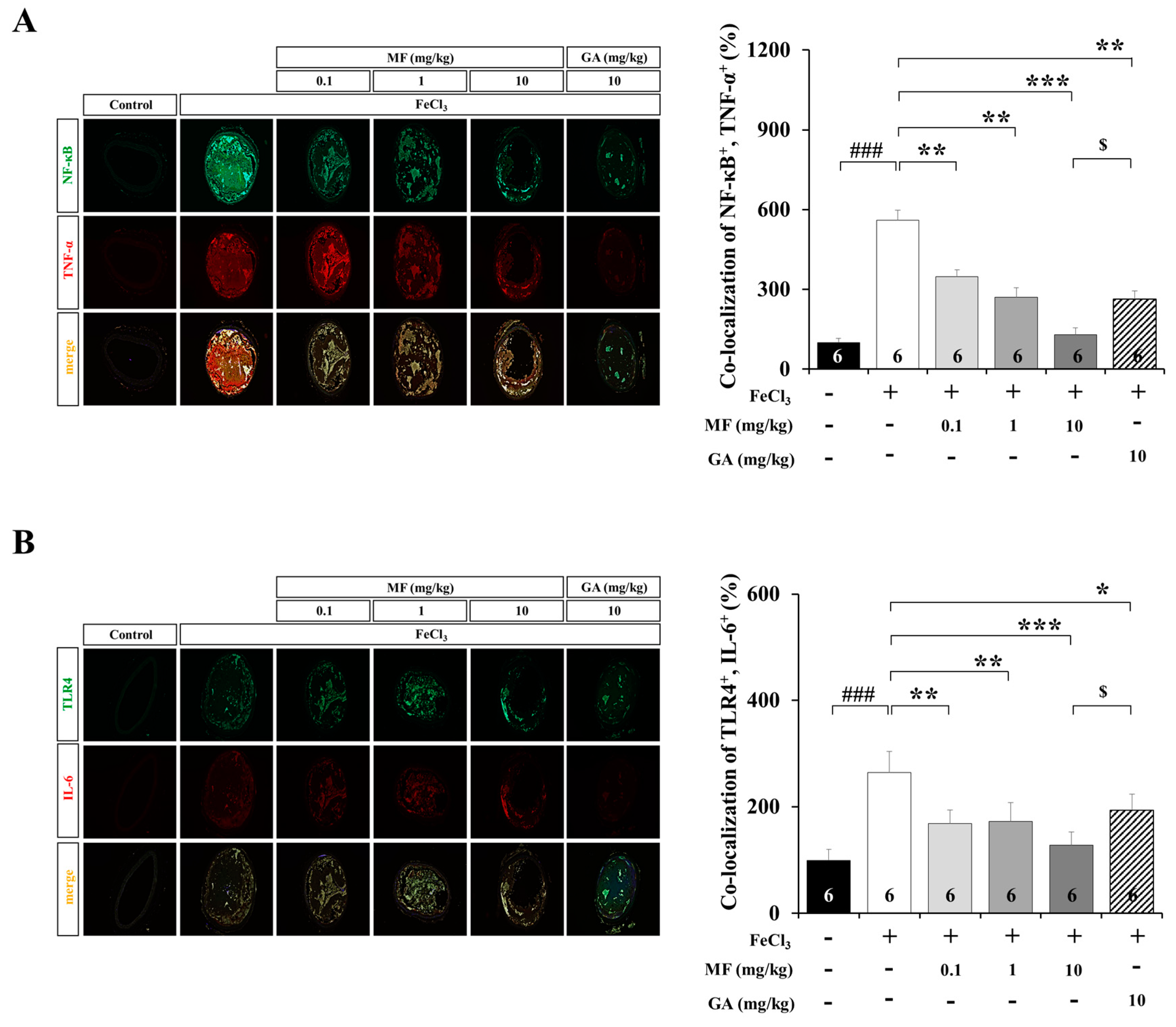
3.4. MF Inhibits Inflammatory Response in an FeCl3-Induced Arterial Thrombosis Model
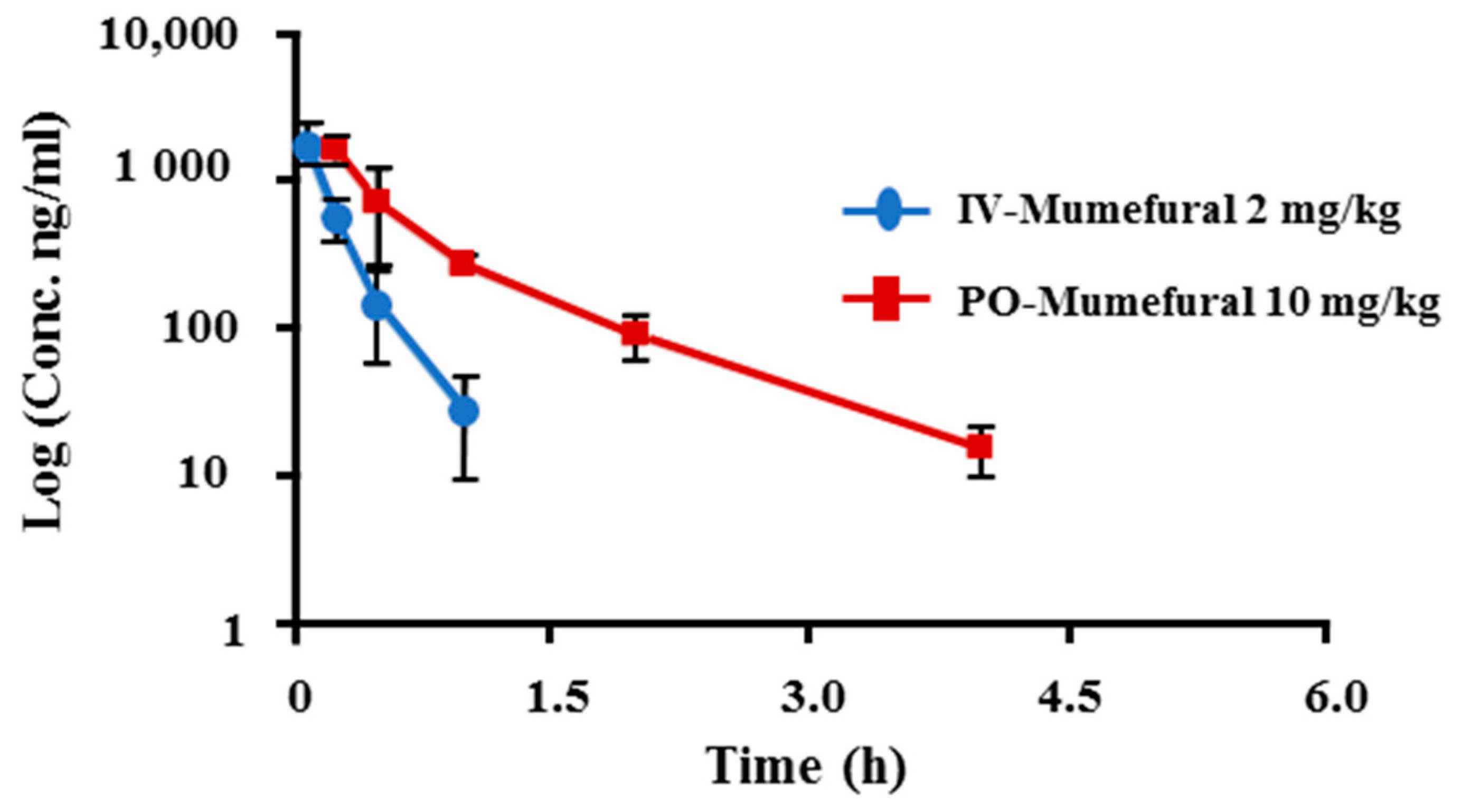
3.5. Pharmacokinetic Parameters of MF in an FeCl3-Induced Arterial Thrombosis Model
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Donahue, M.J.; Hendrikse, J. Improved detection of cerebrovascular disease processes: Introduction to the Journal of cerebral blood flow and metabolism special issue on cerebrovascular disease. J. Cereb. Blood Flow Metab. 2018, 38, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Lebas, H.; Yahiaoui, K.; Martos, R.; Boulaftali, Y. Platelets are at the nexus of vascular diseases. Front. Cardiovasc. Med. 2019, 6, 132. [Google Scholar] [CrossRef] [PubMed]
- Furie, B.; Furie, B.C. Mechanisms of disease: Mechanisms of thrombus formation. N. Engl. J. Med. 2008, 359, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Yeung, J.; Li, W.; Holinstat, M. Platelet signaling and disease: Targeted therapy for thrombosis and other related diseases. Pharmacol. Rev. 2018, 70, 526–548. [Google Scholar] [CrossRef] [PubMed]
- Estevez, B.; Du, X. New concepts and mechanisms of platelet activation signaling. Physiology 2017, 32, 162–177. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.Z.; Kang, L.Y.; Gao, X.M.; Shang, H.C.; Zhang, J.H.; Zhang, B.L. Strategies for antiplatelet targets and agents. Thromb. Res. 2008, 123, 35–49. [Google Scholar] [CrossRef]
- Abou-Saleh, H.; Yacoub, D.; Theoret, J.F.; Gillis, M.A.; Neagoe, P.E.; Labarthe, B.; Theroux, P.; Sirois, M.G.; Tabrizian, M.; Thorin, E.; et al. Endothelial progenitor cells bind and inhibit platelet function and thrombus formation. Circulation 2009, 120, 2230–2239. [Google Scholar] [CrossRef]
- Hirsch, G.E.; Viecili, P.R.N.; de Almeida, A.S.; Nascimento, S.; Porto, F.G.; Otero, J.; Schmidt, A.; da Silva, B.; Parisi, M.M.; Klafke, J.Z. Natural products with antiplatelet action. Curr. Pharm. Des. 2017, 23, 1228–1246. [Google Scholar] [CrossRef]
- Olas, B. Dietary supplements with antiplatelet activity: A solution for everyone? Adv. Nutr. 2018, 9, 51–57. [Google Scholar] [CrossRef]
- Tsuji, R.; Koizumi, H.; Fujiwara, D. Effects of a plum (Prunus mume Siebold and Zucc.) ethanol extract on the immune system in vivo and in vitro. Biosci. Biotechnol. Biochem. 2011, 75, 2011–2013. [Google Scholar] [CrossRef]
- Bailly, C. Anticancer properties of Prunus mume extracts (Chinese plum, Japanese apricot). J. Ethnopharmacol. 2020, 246, 112215. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Ohnishi, Y.; Kotani, T.; Gato, N. Effects of new dietary fiber from Japanese Apricot (Prunus mume Sieb. et Zucc.) on gut function and intestinal microflora in adult mice. Int. J. Mol. Sci. 2011, 12, 2088–2099. [Google Scholar] [CrossRef] [PubMed]
- Yingsakmongkon, S.; Miyamoto, D.; Sriwilaijaroen, N.; Fujita, K.; Matsumoto, K.; Jampangern, W.; Hiramatsu, H.; Guo, C.T.; Sawada, T.; Takahashi, T.; et al. In vitro inhibition of human influenza A virus infection by fruit-juice concentrate of Japanese plum (Prunus mume SIEB. et ZUCC). Biol. Pharm. Bull. 2008, 3, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Takemura, S.; Yoshimasu, K.; Fukumoto, J.; Mure, K.; Nishio, N.; Kishida, K.; Yano, F.; Mitani, T.; Takeshita, T.; Miyashita, K. Safety and adherence of Umezu polyphenols in the Japanese plum (Prunus mume) in a 12-week double-blind randomized placebo-Controlled pilot trial to evaluate antihypertensive effects. Environ. Health. Prev. Med. 2014, 19, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M.; Yamazaki, M.; Matsuda, H.; Gato, N.; Kotani, T. Effect of fruit-juice concentrate of Japanese apricot (Prunus mume SEIB. et ZUCC) on improving blood fluidity. Nat. Med. 2005, 59, 22–27. [Google Scholar]
- Lee, K.M.; Bang, J.; Kim, B.Y.; Lee, I.S.; Han, J.S.; Hwang, B.Y.; Jeon, W.K. Fructus mume alleviates chronic cerebral hypoperfusion-induced white matter and hippocampal damage via inhibition of inflammation and downregulation of TLR4 and p38 MAPK signaling. BMC Complement. Altern. Med. 2015, 22, 125. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Bang, J.H.; Lee, J.; Han, J.S.; Kang, H.W.; Jeon, W.K. Fructus mume ethanol extract prevents inflammation and normalizes the septohippocampal cholinergic system in a rat model of chronic cerebral hypoperfusion. J. Med. Food 2016, 19, 196–204. [Google Scholar] [CrossRef]
- Nishimura, M.; Kume, H.; Kadowaki, A.; Gato, N.; Nishihira, J. Effects and safety of daily ingestion of plum extract on blood pressure: Randomized, double-blinded, placebo-Controlled parallel group comparison study. Funct. Foods Health Dis. 2017, 7, 873–888. [Google Scholar] [CrossRef]
- Lee, S.; Chang, W.; Lu, K.; Lo, D.; Wu, M. Antioxidant capacity and Hepatoprotective effect on ethanol-injured liver cell of lemon juice concentrates and its comparison with commercial Japanese apricot juice concentrates. Res. J. Pharmaceut. Sci. 2013, 2, 7–14. [Google Scholar]
- Chuda, Y.; Ono, H.; Ohnishi-Kameyama, M.; Matsumoto, K.; Nagata, T.; Kikuchi, Y. Mumefural, citric acid derivative improving blood fluidity from fruit-juice concentrate of Japanese apricot (Prunus mume Sieb. et Zucc). J. Agric. Food Chem. 1999, 3, 828–831. [Google Scholar] [CrossRef]
- Bang, J.; Kim, M.S.; Jeon, W.K. Mumefural ameliorates cognitive impairment in chronic cerebral hypoperfusion via regulating the septohippocampal cholinergic system and neuroinflammation. Nutrients 2019, 11, 2755. [Google Scholar] [CrossRef] [PubMed]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach, 3rd ed.; John Wiley and Sons: London, UK, 2012; pp. 230–232. [Google Scholar]
- Jeon, W.K.; Lee, I.S.; Choi, S. Optimization of ferric chloride induced carotid artery thrombosis model in a rat: Effect of Ginkgo biloba extracts. Korean J. Clin. Lab. Sci. 2011, 4, 179–187. [Google Scholar]
- Committee on the Compilation of Textbooks. Parmacognosy, 3rd ed.; Dongmyungsa: Seoul, Korea, 2001. [Google Scholar]
- Utsunomiya, H.; Takekoshi, S.; Gato, N.; Utatsu, H.; Motley, E.D.; Eguchi, K.; Fitzgerald, T.G.; Mifune, M.; Frank, G.D.; Eguchi, S. Fruit-juice concentrate of Asian plum inhibits growth signals of vascular smooth muscle cells induced by angiotensin II. Life Sci. 2002, 72, 659–667. [Google Scholar] [CrossRef]
- Sriwilaijaroen, N.; Kadowaki, A.; Onishi, Y.; Gato, N.; Ujike, M.; Odagiri, T.; Tashiro, M.; Suzuki, Y. Mumefural and related HMF derivatives from Japanese apricot fruit juice concentrate show multiple inhibitory effects on pandemic influenza A (H1N1) virus. Food Chem. 2011, 127, 1–9. [Google Scholar] [CrossRef]
- Chen, H.Y.; Chang, S.S.; Chan, Y.C.; Chen, C.Y. Discovery of novel insomnia leads from screening traditional Chinese medicine database. J. Biomol. Struct. Dyn. 2014, 32, 776–791. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, L.; Zhao, Y.; Han, H.; Hu, Y.; Liang, D.; Yu, B.; Kou, J. The myosin II inhibitor, blebbistatin, ameliorates FeCl3-induced arterial thrombosis via the GSK3β-NF-κB pathway. Int. J. Biol. Sci. 2017, 5, 630–639. [Google Scholar] [CrossRef][Green Version]
- Tseng, M.T.; Dozier, A.; Haribabu, B.; Graham, U.M. Transendothelial migration of ferric ion in FeCl3 injured murine common carotid artery. Thromb. Res. 2006, 2, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Eckly, A.; Hechler, B.; Freund, M.; Zerr, M.; Cazenave, J.P.; Lanza, F.; Mangin, P.H.; Gachet, C. Mechanisms underlying FeCl3-induced arterial thrombosis. J. Thromb. Haemost. 2011, 4, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Neil, D.A. CD31 highlights platelet-rich microthrombi. Histophathology 2009, 3, 387–388. [Google Scholar] [CrossRef] [PubMed]
- Caligiuri, G.; Groyer, E.; Khallou-Laschet, J.; Al Haj Zen, A.; Sainz, J.; Urbain, D.; Gaston, A.T.; Lemitre, M.; Nicoletti, A.; Lafont, A. Reduced immunoregulatory CD31+ T cells in the blood of atherosclerotic mice with plaque thrombosis. Arterioscler. Thromb. Vasc. Biol. 2005, 8, 1659–1664. [Google Scholar] [CrossRef]
- Graczyk, M.; Przybyszewski, M.; Kuzminski, A.; Tlappa, J.; Mucka, J.; Napiorkowska, K.; Szynkiewicz, E.; Zbikowska-Gotz, M.; Bartuzi, Z. Role of E-selectin and platelet endothelial cell adhesion molecule 1 in gastritis in food allergy patients. Postepy Dermatol. Alergol. 2013, 5, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Gupta, G.S. (Ed.) Selectins and associated adhesion proteins in inflammatory disorders. In Animal Lectins: Form, Function and Clinical Applications; Springer: Vienna, Austria, 2012; p. 991. [Google Scholar]
- Franco, A.T.; Corken, A.; Ware, J. Platelets at the interface of thrombosis, inflammation, and cancer. Blood 2015, 5, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Myers, D.D., Jr.; Rectenwald, J.E.; Bedard, P.W.; Kaila, N.; Shaw, G.D.; Schaub, R.G.; Farris, D.M.; Hawley, A.E.; Wrobleski, S.K.; Henke, P.K.; et al. Decreased venous thrombosis with an oral inhibitor of P selectin. J. Vasc. Surg. 2005, 2, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Myers, D., Jr.; Farris, D.; Hawley, A.; Wrobleski, S.; Chapman, A.; Stoolman, L.; Knibbs, R.; Strieter, R.; Wakefield, T. Selectins influence thrombosis in a mouse model of experimental deep venous thrombosis. J. Surg. Res. 2002, 2, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Del Bo’, C.; Marino, M.; Riso, P.; Moller, P.; Porrini, M. Anthocyanins and metabolites resolve TNF-α-mediated production of E-selectin and adhesion of monocytes to endothelial cells. Chem. Biol. Interact. 2019, 300, 49–55. [Google Scholar] [CrossRef] [PubMed]
- McEver, R.P. Selectins: Initiators of leucocyte adhesion and signalling at the vascular wall. Cardiovasc. Res. 2015, 3, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Videm, V.; Albrigtsen, M. Soluble ICAM-1 and VCAM-1 as markers of endothelial activation. Scand. J. Immunol. 2008, 5, 523–531. [Google Scholar] [CrossRef]
- Castro-Ferreira, R.; Cardoso, R.; Leite-Moreira, A.; Mansilha, A. The role of endothelial dysfunction and inflammation in chronic venous disease. Ann. Vasc. Surg. 2018, 46, 380–393. [Google Scholar] [CrossRef]
- Mussbacher, M.; Salzmann, M.; Brostjan, C.; Hoesel, B.; Schoergenhofer, C.; Datler, H.; Hohensinner, P.; Basilio, J.; Petzelbauer, P.; Assinger, A.; et al. Cell type-specific roles of NF-κB linking inflammation and thrombosis. Front. Immunol. 2019, 10, 85. [Google Scholar] [CrossRef]
- Zang, G.; Han, J.; Welch, E.J.; Ye, R.D.; Voyno-Yasenetskaya, T.A.; Malik, A.B.; Du, X.; Li, Z. Lipopolysaccharide stimulates platelet secretion and potentiates platelet aggregation via TLR4/MyD88 and the cGMP-dependent protein kinase pathway. J. Immunol. 2009, 182, 7997–8004. [Google Scholar] [CrossRef]
- Andonegui, G.; Kerfoot, S.M.; McNagny, K.; Ebbert, K.V.; Patel, K.D.; Kubes, P. Platelets express functional Toll-like receptor-4. Blood 2005, 106, 2417–2423. [Google Scholar] [CrossRef] [PubMed]
| Primary Antibody | Company | Dilution |
|---|---|---|
| Anti-P-selectin | Thermo Scientific | 1:250 |
| Anti-E-selectin | Abcam | 1:250 |
| Anti-platelet/endothelial cell adhesion molecule-1 (PECAM-1) | Abcam | 1:100 |
| Anti-intercellular Adhesion Molecule 1 (ICAM) | Abcam | 1:500 |
| Anti-vascular cell adhesion molecule (VCAM) | Abcam | 1:500 |
| Anti-nuclear factor (NF)-κB | Santa Cruz | 1:1000 |
| Anti-tumor necrosis factor (TNF)-α | Santa Cruz | 1:500 |
| Anti-interleukin (IL)-6 | Santa Cruz | 1:500 |
| Anti-Toll like receptor 4 (TLR4) | Santa Cruz | 1:1000 |
| IV MF (2 mg/kg) | ||||||
| Blood concentration (ng/mL) | ||||||
| Time (h) | Rat 1 | Rat 2 | Rat 3 | Rat 4 | Mean | SD |
| 0 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 0.083 | 1635.95 | 1329.02 | 1928.95 | 2684.24 | 1894.54 | 580.66 |
| 0.25 | 826.55 | 434.42 | 429.42 | 658.24 | 587.16 | 191.98 |
| 0.5 | 197.32 | 84.42 | 67.56 | 272.14 | 155.36 | 96.85 |
| 1 | 23.08 | 13.16 | BLQ | 49.44 | 28.56 | 18.75 |
| 2 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 4 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 6 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 8 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 24 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| PO MF (10 mg/kg) | ||||||
| Blood concentration (ng/mL) | ||||||
| Time (h) | Rat 1 | Rat 2 | Rat 3 | Rat 4 | Mean | SD |
| 0 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 0.25 | 2107.84 | 1268.53 | 1648.32 | 1759.75 | 1696.11 | 345.77 |
| 0.5 | 432.06 | 1410.53 | 424.37 | 686.58 | 738.38 | 464.36 |
| 1 | 272.87 | 303.51 | 232.32 | 306.84 | 278.89 | 34.60 |
| 2 | 55.41 | 121.98 | 85.24 | 116.78 | 94.85 | 30.90 |
| 4 | 21.39 | 9.52 | BLQ | 18.01 | 16.31 | 6.12 |
| 6 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 8 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 10 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| 24 | BLQ | BLQ | BLQ | BLQ | NA | NA |
| t1/2 (h) | Tmax (h) | Cmax (ng/mL) | AUC(0–t) (h*ng/mL) | F (%) | |
|---|---|---|---|---|---|
| IV Mumefural 2 mg/kg | |||||
| Rat1 | 0.15 | 0.083 | 1635.95 | 551.91 | - |
| Rat 2 | 0.14 | 0.083 | 1329.02 | 387.8 | - |
| Rat 3 | 0.09 | 0.083 | 1928.95 | 508 | - |
| Rat 4 | 0.2 | 0.083 | 2684.24 | 811.19 | - |
| Mean | 0.14 | 0.08 | 1894.54 | 564.73 | - |
| SD | 0.05 | 0 | 580.66 | 178.35 | - |
| PO Mumefural 10 mg/kg | |||||
| Rat 1 | 0.8 | 0.25 | 2107.84 | 998.14 | 35.35 |
| Rat 2 | 0.59 | 0.5 | 1410.53 | 1266.2 | 44.84 |
| Rat 3 | 0.65 | 0.25 | 1648.32 | 788.08 | 27.91 |
| Rat 4 | 0.73 | 0.25 | 1759.75 | 1120.72 | 39.69 |
| Mean | 0.69 | 0.31 | 1731.61 | 1043.28 | 36.95 |
| SD | 0.69 | 0.13 | 290.64 | 202.37 | 7.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bang, J.; Jeon, W.K. Mumefural Improves Blood Flow in a Rat Model of FeCl3-Induced Arterial Thrombosis. Nutrients 2020, 12, 3795. https://doi.org/10.3390/nu12123795
Bang J, Jeon WK. Mumefural Improves Blood Flow in a Rat Model of FeCl3-Induced Arterial Thrombosis. Nutrients. 2020; 12(12):3795. https://doi.org/10.3390/nu12123795
Chicago/Turabian StyleBang, Jihye, and Won Kyung Jeon. 2020. "Mumefural Improves Blood Flow in a Rat Model of FeCl3-Induced Arterial Thrombosis" Nutrients 12, no. 12: 3795. https://doi.org/10.3390/nu12123795
APA StyleBang, J., & Jeon, W. K. (2020). Mumefural Improves Blood Flow in a Rat Model of FeCl3-Induced Arterial Thrombosis. Nutrients, 12(12), 3795. https://doi.org/10.3390/nu12123795




