Influence of HLA-DQ2.5 Dose on Clinical Picture of Unrelated Celiac Disease Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. Histology
2.3. Serology
2.4. Genetic Analysis
2.5. Statistics
3. Results
4. Discussion
4.1. Strengths and Weaknesses
4.2. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gray, A.M.; Papanicolas, I.N. Impact of symptoms on quality of life before and after diagnosis of coeliac disease: Results from a UK population survey. BMC Health Serv. Res. 2010, 10, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Norström, F.; Lindholm, L.; Sandström, O.; Nordyke, K.; Ivarsson, A. Delay to celiac disease diagnosis and its implications for health-related quality of life. BMC Gastroenterol. 2011, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Sabatino, A.; Corazza, G.R. Coeliac disease. Lancet 2009, 373, 1480–1493. [Google Scholar] [CrossRef]
- Caio, G.; Volta, U.; Sapone, A.; Leffler, D.A.; De Giorgio, R.; Catassi, C.; Fasano, A. Celiac disease: A comprehensive current review. BMC Med. 2019, 17, 1–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Midhagen, G.; Hallert, C. High rate of gastrointestinal symptoms in celiac patients living on a gluten-free diet: Controlled study. Am. J. Gastroenterol. 2003, 98, 2023–2026. [Google Scholar] [CrossRef] [PubMed]
- Wahab, P.J.; Meijer, J.W.R.; Mulder, C.J.J. Histologic follow-up of people with celiac disease on a gluten-free diet: Slow and incomplete recovery. Am. J. Clin. Pathol. 2002, 118, 459–463. [Google Scholar] [CrossRef]
- Hujoel, I.A.; Murray, J.A. Refractory Celiac Disease. Curr. Gastroenterol. Rep. 2020, 22, 18. [Google Scholar] [CrossRef]
- Singh, P.; Arora, S.; Lal, S.; Strand, T.A.; Makharia, G.K. Risk of celiac disease in the first- and second-degree relatives of patients with celiac disease: A systematic review and meta-analysis. Am. J. Gastroenterol. 2015, 110, 1539–1548. [Google Scholar] [CrossRef]
- Van De Wal, Y.; Kooy, Y.M.C.; Drijfhout, J.W.; Amons, R.; Koning, F. Peptide binding characteristics of the coeliac disease associated DQ(α1(*)0501, β1(*)0201) molecule. Immunogenetics 1996, 44, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Vartdal, F.; Johansen, B.H.; Friede, T.; Thorpe, C.J.; Stevanović, S.; Eriksen, J.E.; Sletten, K.; Thorsby, E.; Rammensee, H.G.; Sollid, L.M. The peptide binding motif of the disease associated HLA-DQ (α 1(*) 0501, β 1(*) 0201) molecule. Eur. J. Immunol. 1996, 26, 2764–2772. [Google Scholar] [CrossRef] [PubMed]
- Sollid, L.M.; Thorsby, E. HLA susceptibility genes in celiac disease: Genetic mapping and role in pathogenesis. Gastroenterology 1993, 105, 910–922. [Google Scholar] [CrossRef]
- Ploski, R.; Ek, J.; Thorsby, E.; Sollid, L.M. On the HLA-DQ(α1*0501, β1*0201)-associated susceptibility in celiac disease: A possible gene dosage effect of DQB1*0201. Tissue Antigens 1993, 41, 173–177. [Google Scholar] [CrossRef] [PubMed]
- van Belzen, M.J.; Koeleman, B.P.C.; Crusius, J.B.A.; Meijer, J.W.R.; Bardoel, A.F.J.; Pearson, P.L.; Sandkuijl, L.A.; Houwen, R.H.J.; Wijmenga, C. Defining the contribution of the HLA region to cis DQ2-positive coeliac disease patients. Genes Immun. 2004, 5, 215–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Margaritte-Jeannin, P.; Babron, M.C.; Bourgey, M.; Louka, A.S.; Clot, F.; Percopo, S.; Coto, I.; Hugot, J.P.; Ascher, H.; Sollid, L.M.; et al. HLA-DQ relative risks for coeliac disease in European populations: A study of the European Genetics Cluster on Coeliac Disease. Tissue Antigens 2004, 63, 562–567. [Google Scholar] [CrossRef]
- Vader, W.; Stepniak, D.; Kooy, Y.; Mearin, L.; Thompson, A.; van Rood, J.J.; Spaenij, L.; Koning, F. The HLA-DQ2 gene dose effect in celiac disease is directly related to the magnitude and breadth of gluten-specific T cell responses. Proc. Natl. Acad. Sci. USA 2003, 100, 12390–12395. [Google Scholar] [CrossRef] [Green Version]
- Bajor, J.; Szakács, Z.; Farkas, N.; Hegyi, P.; Illés, A.; Solymár, M.; Pétervári, E.; Balaskó, M.; Pár, G.; Sarlós, P.; et al. Classical celiac disease is more frequent with a double dose of HLA-DQB102: A systematic review with meta-analysis. PLoS ONE 2019, 14, e0212329. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef]
- Dickson, B.C.; Streutker, C.J.; Chetty, R. Coeliac disease: An update for pathologists. J. Clin. Pathol. 2006, 59, 1008–1016. [Google Scholar] [CrossRef] [Green Version]
- Dieterich, W.; Ehnis, T.; Bauer, M.; Donner, P.; Volta, U.; Riecken, E.O.; Schuppan, D. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat. Med. 1997, 3, 797–801. [Google Scholar] [CrossRef]
- Chorzelski, T.P.; Sulej, J.; Tchorzewska, H.; Jablonska, S.; Beutner, E.H.; Kumar, V. IgA Class Endomysium Antibodies in Dermatitis Herpetiformis and Coeliac Disease. Ann. N. Y. Acad. Sci. 1983, 420, 325–334. [Google Scholar] [CrossRef]
- Eade, O.E.; Lloyd, R.S.; Lang, C.; Wright, R. IgA and IgG reticulin antibodies in coeliac and non-coeliac patients. Gut 1977, 18, 991–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sulkanen, S.; Collin, P.; Laurila, K.; Mäki, M. IgA- and IgG-class antihuman umbilical cord antibody tests in adult coeliac disease. Scand. J. Gastroenterol. 1998, 33, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Monsuur, A.J.; de Bakker, P.I.W.; Zhernakova, A.; Pinto, D.; Verduijn, W.; Romanos, J.; Auricchio, R.; Lopez, A.; van Heel, D.A.; Crusius, J.B.A.; et al. Effective detection of human leukocyte antigen risk alleles in celiac disease using tag single nucleotide polymorphisms. PLoS ONE 2008, 3, e2270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koskinen, L.; Romanos, J.; Kaukinen, K.; Mustalahti, K.; Korponay-Szabo, I.; Barisani, D.; Bardella, M.T.; Ziberna, F.; Vatta, S.; Széles, G.; et al. Cost-effective HLA typing with tagging SNPs predicts celiac disease risk haplotypes in the Finnish, Hungarian, and Italian populations. Immunogenetics 2009, 61, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ojinaga, E.; Fernández-Prieto, M.; Molina, M.; Polanco, I.; Urcelay, E.; Núñez, C. Influence of HLA on clinical and analytical features of pediatric celiac disease. BMC Gastroenterol. 2019, 19, 91. [Google Scholar] [CrossRef] [PubMed]
- du Pré, M.F.; Blazevski, J.; Dewan, A.E.; Stamnaes, J.; Kanduri, C.; Sandve, G.K.; Johannesen, M.K.; Lindstad, C.B.; Hnida, K.; Fugger, L.; et al. B cell tolerance and antibody production to the celiac disease autoantigen transglutaminase 2. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef]
- Kauma, S.; Kaukinen, K.; Huhtala, H.; Kivelä, L.; Pekki, H.; Salmi, T.; Saavalainen, P.; Lindfors, K.; Kurppa, K.; Kauma, S.; et al. The Phenotype of Celiac Disease Has Low Concordance between Siblings, Despite a Similar Distribution of HLA Haplotypes. Nutrients 2019, 11, 479. [Google Scholar] [CrossRef] [Green Version]
- Wacklin, P.; Kaukinen, K.; Tuovinen, E.; Collin, P.; Lindfors, K.; Partanen, J.; Mäki, M.; Mättuö, J. The duodenal microbiota composition of adult celiac disease patients is associated with the clinical manifestation of the disease. Inflamm. Bowel Dis. 2013, 19, 934–941. [Google Scholar] [CrossRef] [Green Version]
- Al-Toma, A.; Goerres, M.S.; Meijer, J.W.R.; Peña, A.S.; Crusius, J.B.A.; Mulder, C.J.J. Human leukocyte antigen-DQ2 homozygosity and the development of refractory celiac disease and enteropathy-associated T-cell lymphoma. Clin. Gastroenterol. Hepatol. 2006, 4, 315–319. [Google Scholar] [CrossRef]
- Ilus, T.; Kaukinen, K.; Virta, L.J.; Pukkala, E.; Collin, P. Incidence of malignancies in diagnosed celiac patients: A population-based estimate. Am. J. Gastroenterol. 2014, 109, 1471–1477. [Google Scholar] [CrossRef]
- Paarlahti, P.; Kurppa, K.; Ukkola, A.; Collin, P.; Huhtala, H.; Mäki, M.; Kaukinen, K. Predictors of persistent symptoms and reduced quality of life in treated coeliac disease patients: A large cross-sectional study. BMC Gastroenterol. 2013, 13, 75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wacklin, P.; Laurikka, P.; Lindfors, K.; Collin, P.; Salmi, T.; Lähdeaho, M.L.; Saavalainen, P.; Mäki, M.; Mättö, J.; Kurppa, K.; et al. Altered Duodenal microbiota composition in celiac disease patients suffering from persistent symptoms on a long-term gluten-free diet. Am. J. Gastroenterol. 2014, 109, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M. High prevalence of small intestinal bacterial overgrowth in celiac patients with persistence of gastrointestinal symptoms after gluten withdrawal. Am. J. Gastroenterol. 2003, 98, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Rahim, M.W.; See, J.A.; Lahr, B.D.; Wu, T.T.; Murray, J.A. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am. J. Gastroenterol. 2010, 105, 1412–1420. [Google Scholar] [CrossRef] [Green Version]
- Sollid, L.M. The roles of MHC class II genes and post-translational modification in celiac disease. Immunogenetics 2017, 69, 605–616. [Google Scholar] [CrossRef]
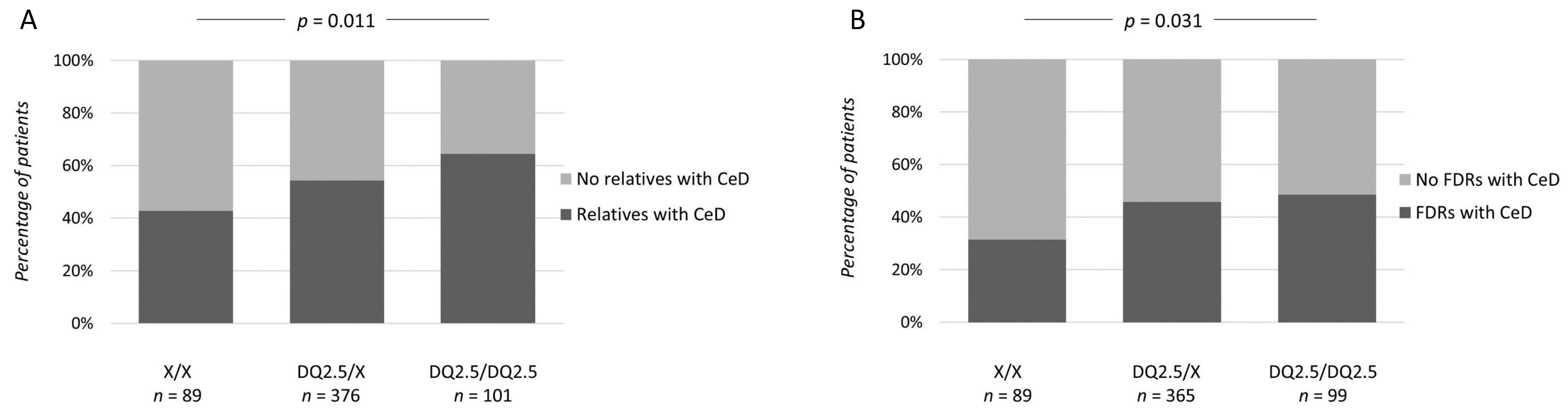
| X/X | DQ2.5/X | DQ2.5/DQ2.5 | p | |
|---|---|---|---|---|
| n = 100 | n = 401 | n = 104 | ||
| % (n) | % (n) | % (n) | ||
| Females | 78.0 (78) | 73.3 (294) | 76.0 (79) | 0.589 |
| Age group | 0.996 * | |||
| 0–6 years | 7.1 (7) | 6.8 (27) | 7.7 (8) | |
| 7–20 years | 13.1 (13) | 13.6 (54) | 14.4 (15) | |
| 21–65 years | 75.8 (75) | 76.1 (302) | 73.1 (76) | |
| >65 years | 4.0 (4) | 3.5 (14) | 4.8 (5) | |
| Symptoms in childhood | 48.9 (43) | 43.8 (161) | 51.5 (51) | 0.334 |
| Diagnostic delay | 0.908 * | |||
| 0 1 | 4.3 (4) | 4.9 (18) | 3.0 (3) | |
| <1 year | 20.7 (19) | 23.6 (86) | 25.7 (26) | |
| 1–5 years | 37.0 (34) | 31.5 (115) | 32.7 (33) | |
| 5–10 years | 7.6 (7) | 10.4 (38) | 6.9 (7) | |
| >10 years | 30.4 (28) | 29.6 (108) | 31.7 (32) | |
| Classical symptoms 2 | 30.0 (30) | 16.7 (67) | 21.2 (22) | 0.007 |
| Abdominal symptoms 3 | 86.9 (86) | 81.3 (322) | 81.7 (85) | 0.426 |
| Diarrhea | 44.7 (42) | 37.1 (144) | 41.7 (43) | 0.337 |
| Anemia | 30.3 (30) | 28.5 (113) | 37.5 (39) | 0.209 |
| Extraintestinal manifestations 4 | 44.4 (44) | 49.5 (196) | 47.1 (49) | 0.646 |
| Dermatitis herpetiformis | 11.0 (10) | 17.1 (63) | 19.2 (19) | 0.270 |
| Severity of symptoms 5 | 0.484 * | |||
| No symptoms | 5.0 (4) | 5.9 (17) | 4.0 (3) | |
| Mild | 23.8 (19) | 25.9 (74) | 33.3 (25) | |
| Moderate | 15.0 (12) | 12.2 (35) | 5.3 (4) | |
| Severe | 56.3 (45) | 55.9 (160) | 57.3 (43) | |
| Celiac disease antibody positivity 6,7 | 77.8 (49) | 86.8 (217) | 94.9 (56) | 0.021 |
| Severity of mucosal damage 8 | 0.546 * | |||
| Normal morphology | 2.4 (2) | 3.0 (10) | 2.4 (2) | |
| Partial villous atrophy | 27.7 (23) | 33.8 (111) | 25.3 (21) | |
| Subtotal/total villous atrophy | 69.9 (58) | 63.1 (207) | 72.3 (60) |
| X/X | DQ2.5/X | DQ2.5/DQ2.5 | ||
|---|---|---|---|---|
| n = 100 | n = 401 | n = 104 | ||
| % (n) | % (n) | % (n) | p | |
| Adherence to GFD 1 | 0.025 * | |||
| Strict GFD 1 | 93.8 (91) | 97.4 (376) | 91.1 (92) | |
| Dietary lapses | 5.2 (5) | 2.3 (9) | 7.9 (8) | |
| No GFD 1 | 1.0 (1) | 0.3 (1) | 1.0 (1) | |
| Self-reported current symptoms 2,3 | 44.8 (26) | 22.9 (59) | 32.3 (20) | 0.002 |
| Celiac disease antibody positivity 4,5 | 28.6 (4) | 23.8 (10) | 9.1 (1) | 0.535 |
| Severity of mucosal damage 6 | 0.108 * | |||
| Normal morphology | 72.9 (35) | 55.6 (104) | 46.9 (23) | |
| Partial villous atrophy | 25.0 (12) | 38.0 (71) | 44.9 (22) | |
| Subtotal/total villous atrophy | 2.1 (1) | 6.4 (12) | 8.2 (4) | |
| Other illnesses | ||||
| Any autoimmune disease 7 | 29.8 (28) | 23.6 (91) | 18.0 (18) | 0.155 |
| Type 1 diabetes | 3.2 (3) | 3.4 (13) | 1.0 (1) | 0.504 |
| Thyroidal disease | 17.9 (17) | 14.0 (54) | 9.7 (10) | 0.248 |
| Malignancy | 3.2 (3) | 3.9 (15) | 5.8 (6) | 0.581 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Airaksinen, L.; Laurikka, P.; Huhtala, H.; Kurppa, K.; Salmi, T.; Saavalainen, P.; Kaukinen, K.; Lindfors, K. Influence of HLA-DQ2.5 Dose on Clinical Picture of Unrelated Celiac Disease Patients. Nutrients 2020, 12, 3775. https://doi.org/10.3390/nu12123775
Airaksinen L, Laurikka P, Huhtala H, Kurppa K, Salmi T, Saavalainen P, Kaukinen K, Lindfors K. Influence of HLA-DQ2.5 Dose on Clinical Picture of Unrelated Celiac Disease Patients. Nutrients. 2020; 12(12):3775. https://doi.org/10.3390/nu12123775
Chicago/Turabian StyleAiraksinen, Laura, Pilvi Laurikka, Heini Huhtala, Kalle Kurppa, Teea Salmi, Päivi Saavalainen, Katri Kaukinen, and Katri Lindfors. 2020. "Influence of HLA-DQ2.5 Dose on Clinical Picture of Unrelated Celiac Disease Patients" Nutrients 12, no. 12: 3775. https://doi.org/10.3390/nu12123775
APA StyleAiraksinen, L., Laurikka, P., Huhtala, H., Kurppa, K., Salmi, T., Saavalainen, P., Kaukinen, K., & Lindfors, K. (2020). Influence of HLA-DQ2.5 Dose on Clinical Picture of Unrelated Celiac Disease Patients. Nutrients, 12(12), 3775. https://doi.org/10.3390/nu12123775





