Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Selection and Extraction
2.2. Infusion Trials
2.3. Analytic Procedures
2.4. Muscle Fractional Synthetic Rate
2.5. Whole-Body Protein Synthesis
2.6. Amino Acid Pharmacokinetics
2.7. Statistical Analysis
3. Results
3.1. Stepwise Regression
3.2. Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gwin, J.A.; Church, D.D.; Wolfe, R.R.; Ferrando, A.A.; Pasiakos, S.M. Muscle Protein Synthesis and Whole-Body Protein Turnover Responses to Ingesting Essential Amino Acids, Intact Protein, and Protein-Containing Mixed Meals with Considerations for Energy Deficit. Nutrients 2020, 12, 2457. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.P.; Wolfe, R.R. Is there a maximal anabolic response to protein intake with a meal? Clin. Nutr. 2012, 32, 309–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biolo, G.; Fleming, R.Y.; Maggi, S.P.; Wolfe, R.R. Transmembrane transport and intracellular kinetics of amino acids in human skeletal muscle. Am. J. Physiol. Metab. 1995, 268, E75–E84. [Google Scholar] [CrossRef] [PubMed]
- Bohé, J.; Low, A.; Wolfe, R.R.; Rennie, M.J. Human Muscle Protein Synthesis is Modulated by Extracellular, Not Intramuscular Amino Acid Availability: A Dose-Response Study. J. Physiol. 2003, 552, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Tipton, K.D.; Ferrando, A.A.; Phillips, S.M.; Doyle, D.; Wolfe, R.R. Postexercise net protein synthesis in human muscle from orally administered amino acids. Am. J. Physiol. Metab. 1999, 276, E628–E634. [Google Scholar] [CrossRef] [PubMed]
- Volpi, E.; Ferrando, A.A.; Yeckel, C.W.; Tipton, K.D.; Wolfe, R.R. Exogenous amino acids stimulate net muscle protein synthesis in the elderly. J. Clin. Investig. 1998, 101, 2000–2007. [Google Scholar] [CrossRef] [Green Version]
- Volpi, E.; Kobayashi, H.; Sheffield-Moore, M.; Mittendorfer, B.; Wolfe, R.R. Essential amino acids are primarily responsible for the amino acid stimulation of muscle protein anabolism in healthy elderly adults. Am. J. Clin. Nutr. 2003, 78, 250–258. [Google Scholar] [CrossRef]
- Church, D.D.; Gwin, J.A.; Wolfe, R.R.; Pasiakos, S.M.; Ferrando, A.A. Mitigation of Muscle Loss in Stressed Physiology: Military Relevance. Nutrients 2019, 11, 1703. [Google Scholar] [CrossRef] [Green Version]
- Smeets, J.S.J.; Horstman, A.M.H.; Vles, G.F.; Emans, P.J.; Goessens, J.P.B.; Gijsen, A.P.; Van Kranenburg, J.M.X.; Van Loon, L.J.C. Protein synthesis rates of muscle, tendon, ligament, cartilage, and bone tissue in vivo in humans. PLoS ONE 2019, 14, e0224745. [Google Scholar] [CrossRef]
- Smeets, J.S.J.; Horstman, A.M.H.; Schijns, O.E.M.G.; Dings, J.T.A.; Hoogland, G.; Gijsen, A.P.; Goessens, J.P.B.; Bouwman, F.G.; Wodzig, W.K.W.H.; Mariman, E.C.; et al. Brain tissue plasticity: Protein synthesis rates of the human brain. Brain 2018, 141, 1122–1129. [Google Scholar] [CrossRef]
- Nakshabendi, I.M.; McKee, R.; Downie, S.; Russell, R.I.; Rennie, M.J. Rates of small intestinal mucosal protein synthesis in human jejunum and ileum. Am. J. Physiol. 1999, 277, E1028–E1031. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Paddon-Jones, D.; Layman, D.K. Optimizing Adult Protein Intake during Catabolic Health Conditions. Adv. Nutr. 2020, 11, S1058–S1069. [Google Scholar] [CrossRef] [PubMed]
- Rennie, M.J.; Tipton, K.D. Protein and amino acid metabolism during and after exercise and the effects of nutrition. Annu. Rev. Nutr. 2000, 20, 457–483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Chinkes, D.L.; Sakurai, Y.; Wolfe, R.R. An isotopic method for measurement of muscle protein fractional breakdown rate in vivo. Am. J. Physiol. 1996, 270, E759–E767. [Google Scholar] [CrossRef]
- Zhang, X.-J.; Chinkes, D.L.; Wolfe, R.R. Measurement of muscle protein fractional synthesis and breakdown rates from a pulse tracer injection. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E753–E764. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.-J.; Chinkes, D.L.; Herndon, D.N.; Wolfe, R.R. Measurement of protein fractional synthesis and breakdown rates in the skin of rabbits using a subflooding dose method. Metab. Clin. Exp. 2009, 58, 1239–1247. [Google Scholar] [CrossRef]
- Crossland, H.; Smith, K.; Atherton, P.J.; Wilkinson, D.J. A novel stable isotope tracer method to simultaneously quantify skeletal muscle protein synthesis and breakdown. Metabol. Open 2020, 5, 100022. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Park, S.; Kim, I.-Y.; Starck, C.; Marquis, B.J.; Ferrando, A.A.; Moughan, P.J. Quantifying the contribution of dietary protein to whole body protein kinetics: Examination of the intrinsically labeled proteins method. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E74–E84. [Google Scholar] [CrossRef]
- Trommelen, J.; Holwerda, A.M.; Nyakayiru, J.; Gorissen, S.H.M.; Rooyackers, O.; Burd, N.A.; Boirie, Y.; Van Loon, L.J.C. The intrinsically labeled protein approach is the preferred method to quantify the release of dietary protein-derived amino acids into the circulation. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E433–E434. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Park, S.; Kim, I.-Y.; Starck, C.; Marquis, B.J.; Ferrando, A.A.; Moughan, P.J. Reply to Letter to the Editor: “The intrinsically labeled protein approach is the preferred method to quantify the release of dietary protein-derived amino acids into the circulation. ” Am. J. Physiol. Endocrinol. Metab. 2019, 317, E435. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Park, S.; Kim, I.-Y.; Moughan, P.J.; Ferrando, A.A. Advances in stable isotope tracer methodology part 2: New thoughts about an “old” method-measurement of whole body protein synthesis and breakdown in the fed state. J. Investig. Med. 2020, 68, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R.; Chinkes, D.L. Isotope Tracers in Metabolic Research: Principles and Practice of Kinetic Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2004; ISBN 978-0-471-46209-5. [Google Scholar]
- Park, S.; Church, D.D.; Azhar, G.; Schutzler, S.E.; Ferrando, A.A.; Wolfe, R.R. Anabolic response to essential amino acid plus whey protein composition is greater than whey protein alone in young healthy adults. J. Int. Soc. Sports Nutr. 2020, 17, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Church, D.D.; Ferrando, A.A.; Wolfe, R.R. Improved Muscle Protein Synthesis is Achieved with 3.6 g of Free Form Essential Amino Acid Ingestion in Elderly. J. Int. Soc. Sports Nutr. 2019, 17, 13. [Google Scholar]
- Park, S.; Church, D.D.; Starck, C.; Schutzler, S.E.; Azhar, G.; Kim, I.-Y.; Ferrando, A.A.; Moughan, P.J.; Wolfe, R.R. The impact of Hayward green kiwifruit on dietary protein digestion and protein metabolism. Eur. J. Nutr. 2020. [Google Scholar] [CrossRef]
- Park, S.; Church, D.D.; Schutzler, S.E.; Azhar, G.; Kim, I.Y.; Ferrando, A.A.; Wolfe, R.R. Metabolic evaluation of the Dietary Guideline’s ounce equivalents of protein food sources in young adults: A randomized controlled trial. J. Nutr. 2020, in press. [Google Scholar]
- Abumrad, N.N.; Rabin, D.; Diamond, M.P.; Lacy, W.W. Use of a heated superficial hand vein as an alternative site for the measurement of amino acid concentrations and for the study of glucose and alanine kinetics in man. Metabolism 1981, 30, 936–940. [Google Scholar] [CrossRef]
- De Betue, C.T.I.; Joosten, K.F.M.; Deutz, N.E.P.; Vreugdenhil, A.C.E.; Van Waardenburg, D.A. Arginine appearance and nitric oxide synthesis in critically ill infants can be increased with a protein-energy-enriched enteral formula. Am. J. Clin. Nutr. 2013, 98, 907–916. [Google Scholar] [CrossRef] [Green Version]
- Baumann, P.Q.; Stirewalt, W.S.; O’Rourke, B.D.; Howard, D.; Nair, K.S. Precursor pools of protein synthesis: A stable isotope study in a swine model. Am. J. Physiol. 1994, 267, E203–E209. [Google Scholar] [CrossRef]
- Gwin, J.A.; Church, D.D.; Hatch-McChesney, A.; Howard, E.E.; Carrigan, C.T.; Murphy, N.E.; Wilson, M.A.; Margolis, L.M.; Carbone, J.W.; Wolfe, R.R.; et al. Effects of high versus standard essential amino acid intakes on whole-body protein turnover and mixed muscle protein synthesis during energy deficit: A randomized, crossover study. Clin. Nutr. 2020. [Google Scholar] [CrossRef]
- Witard, O.C.; Jackman, S.R.; Breen, L.; Smith, K.; Selby, A.; Tipton, K.D. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 2014, 99, 86–95. [Google Scholar] [CrossRef] [Green Version]
- Reeds, P.J.; Hachey, D.L.; Patterson, B.W.; Motil, K.J.; Klein, P.D. VLDL apolipoprotein B-100, a potential indicator of the isotopic labeling of the hepatic protein synthetic precursor pool in humans: Studies with multiple stable isotopically labeled amino acids. J. Nutr. 1992, 122, 457–466. [Google Scholar] [CrossRef]
- Pennings, B.; Boirie, Y.; Senden, J.M.G.; Gijsen, A.P.; Kuipers, H.; Van Loon, L.J.C. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 2011, 93, 997–1005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, G.I.; Patterson, B.W.; Mittendorfer, B. Human muscle protein turnover—Why is it so variable? J. Appl. Physiol. 2011, 110, 480–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volpi, E.; Mittendorfer, B.; Wolf, S.E.; Wolfe, R.R. Oral amino acids stimulate muscle protein anabolism in the elderly despite higher first-pass splanchnic extraction. Am. J. Physiol. 1999, 277, E513–E520. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, W.K.; Phillips, B.E.; Williams, J.P.; Rankin, D.; Lund, J.N.; Wilkinson, D.J.; Smith, K.; Atherton, P.J. The impact of delivery profile of essential amino acids upon skeletal muscle protein synthesis in older men: Clinical efficacy of pulse vs. bolus supply. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E450–E457. [Google Scholar] [CrossRef] [Green Version]
- Pennings, B.; Groen, B.; de Lange, A.; Gijsen, A.P.; Zorenc, A.H.; Senden, J.M.G.; Van Loon, L.J.C. Amino acid absorption and subsequent muscle protein accretion following graded intakes of whey protein in elderly men. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E992–E999. [Google Scholar] [CrossRef] [Green Version]
- Kramer, I.F.; Verdijk, L.B.; Hamer, H.M.; Verlaan, S.; Luiking, Y.C.; Kouw, I.W.K.; Senden, J.M.; Van Kranenburg, J.; Gijsen, A.P.; Bierau, J.; et al. Both basal and post-prandial muscle protein synthesis rates, following the ingestion of a leucine-enriched whey protein supplement, are not impaired in sarcopenic older males. Clin. Nutr. 2017, 36, 1440–1449. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Crombag, J.J.; Langer, H.; Bierau, J.; Respondek, F.; Van Loon, L.J. Ingestion of Wheat Protein Increases In Vivo Muscle Protein Synthesis Rates in Healthy Older Men in a Randomized Trial. J. Nutr. 2016, 146, 1651–1659. [Google Scholar] [CrossRef]
- Symons, T.B.; Schutzler, S.E.; Cocke, T.L.; Chinkes, D.L.; Wolfe, R.R.; Paddon-Jones, D. Aging does not impair the anabolic response to a protein-rich meal. Am. J. Clin. Nutr. 2007, 86, 451–456. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, C.J.; Hermans, W.J.H.; Holwerda, A.M.; Smeets, J.S.J.; Senden, J.M.; Van Kranenburg, J.; Gijsen, A.P.; Wodzig, W.K.H.W.; Schierbeek, H.; Verdijk, L.B.; et al. Branched-chain amino acid and branched-chain ketoacid ingestion increases muscle protein synthesis rates in vivo in older adults: A double-blind, randomized trial. Am. J. Clin. Nutr. 2019, 110, 862–872. [Google Scholar] [CrossRef] [Green Version]
- Bendtsen, L.Q.; Thorning, T.K.; Reitelseder, S.; Ritz, C.; Hansen, E.T.; Van Hall, G.; Astrup, A.; Sjödin, A.; Holm, L. Human Muscle Protein Synthesis Rates after Intake of Hydrolyzed Porcine-Derived and Cows’ Milk Whey Proteins-A Randomized Controlled Trial. Nutrients 2019, 11, 989. [Google Scholar] [CrossRef] [Green Version]
- Glynn, E.L.; Fry, C.S.; Drummond, M.J.; Timmerman, K.L.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Excess leucine intake enhances muscle anabolic signaling but not net protein anabolism in young men and women. J. Nutr. 2010, 140, 1970–1976. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Hamer, H.M.; de Lange, A.; Kiskini, A.; Groen, B.B.L.; Senden, J.M.G.; Gijsen, A.P.; Verdijk, L.B.; Van Loon, L.J.C. Leucine co-ingestion improves post-prandial muscle protein accretion in elderly men. Clin. Nutr. 2013, 32, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Chanet, A.; Verlaan, S.; Salles, J.; Giraudet, C.; Patrac, V.; Pidou, V.; Pouyet, C.; Hafnaoui, N.; Blot, A.; Cano, N.; et al. Supplementing Breakfast with a Vitamin D and Leucine-Enriched Whey Protein Medical Nutrition Drink Enhances Postprandial Muscle Protein Synthesis and Muscle Mass in Healthy Older Men. J. Nutr. 2017, 147, 2262–2271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheffield-Moore, M.; Wolfe, R.R.; Gore, D.C.; Wolf, S.E.; Ferrer, D.M.; Ferrando, A.A. Combined effects of hyperaminoacidemia and oxandrolone on skeletal muscle protein synthesis. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E273–E279. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2005, 19, 422–424. [Google Scholar] [CrossRef]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef]
- Yang, Y.; Breen, L.; Burd, N.A.; Hector, A.J.; Churchward-Venne, T.A.; Josse, A.R.; Tarnopolsky, M.A.; Phillips, S.M. Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br. J. Nutr. 2012, 108, 1780–1788. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Churchward-Venne, T.A.; Burd, N.A.; Breen, L.; Tarnopolsky, M.A.; Phillips, S.M. Myofibrillar protein synthesis following ingestion of soy protein isolate at rest and after resistance exercise in elderly men. Nutr. Metab. 2012, 9, 57. [Google Scholar] [CrossRef] [Green Version]
- Robinson, M.J.; Burd, N.A.; Breen, L.; Rerecich, T.; Yang, Y.; Hector, A.J.; Baker, S.K.; Phillips, S.M. Dose-dependent responses of myofibrillar protein synthesis with beef ingestion are enhanced with resistance exercise in middle-aged men. Appl. Physiol. Nutr. Metab. 2013, 38, 120–125. [Google Scholar] [CrossRef] [Green Version]
- Holwerda, A.M.; Paulussen, K.J.M.; Overkamp, M.; Goessens, J.P.B.; Kramer, I.F.; Wodzig, W.K.W.H.; Verdijk, L.B.; Van Loon, L.J.C. Dose-Dependent Increases in Whole-Body Net Protein Balance and Dietary Protein-Derived Amino Acid Incorporation into Myofibrillar Protein During Recovery from Resistance Exercise in Older Men. J. Nutr. 2019, 149, 221–230. [Google Scholar] [CrossRef]
- Dunlop, M.V.; Kilroe, S.P.; Bowtell, J.L.; Finnigan, T.J.A.; Salmon, D.L.; Wall, B.T. Mycoprotein represents a bioavailable and insulinotropic non-animal-derived dietary protein source: A dose-response study. Br. J. Nutr. 2017, 118, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, C.S.; Kobayashi, H.; Sheffield-Moore, M.; Aarsland, A.; Wolfe, R.R. Aging is associated with diminished accretion of muscle proteins after the ingestion of a small bolus of essential amino acids. Am. J. Clin. Nutr. 2005, 82, 1065–1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, S.L.; Tipton, K.D.; Chinkes, D.L.; Wolf, S.E.; Wolfe, R.R. Independent and combined effects of amino acids and glucose after resistance exercise. Med. Sci. Sports Exerc. 2003, 35, 449–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordon, B.S.; Kazi, A.A.; Coleman, C.S.; Dennis, M.D.; Chau, V.; Jefferson, L.S.; Kimball, S.R. RhoA modulates signaling through the mechanistic target of rapamycin complex 1 (mTORC1) in mammalian cells. Cell. Signal. 2014, 26, 461–467. [Google Scholar] [CrossRef] [Green Version]
- Verhoeven, S.; Vanschoonbeek, K.; Verdijk, L.B.; Koopman, R.; Wodzig, W.K.W.H.; Dendale, P.; Van Loon, L.J.C. Long-term leucine supplementation does not increase muscle mass or strength in healthy elderly men. Am. J. Clin. Nutr. 2009, 89, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Church, D.D.; Schwarz, N.A.; Spillane, M.B.; McKinley-Barnard, S.K.; Andre, T.L.; Ramirez, A.J.; Willoughby, D.S. l-Leucine Increases Skeletal Muscle IGF-1 but Does Not Differentially Increase Akt/mTORC1 Signaling and Serum IGF-1 Compared to Ursolic Acid in Response to Resistance Exercise in Resistance-Trained Men. J. Am. Coll. Nutr. 2016, 35, 627–638. [Google Scholar] [CrossRef]
- DE Andrade, I.T.; Gualano, B.; Hevia-LarraÍn, V.; Neves-Junior, J.; Cajueiro, M.; Jardim, F.; Gomes, R.L.; Artioli, G.G.; Phillips, S.M.; Campos-Ferraz, P.; et al. Leucine Supplementation Has No Further Effect on Training-induced Muscle Adaptations. Med. Sci. Sports Exerc. 2020, 52, 1809–1814. [Google Scholar] [CrossRef]
- Backx, E.M.P.; Horstman, A.M.H.; Marzuca-Nassr, G.N.; Van Kranenburg, J.; Smeets, J.S.; Fuchs, C.J.; Janssen, A.A.W.; de Groot, L.C.P.G.M.; Snijders, T.; Verdijk, L.B.; et al. Leucine Supplementation Does Not Attenuate Skeletal Muscle Loss during Leg Immobilization in Healthy, Young Men. Nutrients 2018, 10, 635. [Google Scholar] [CrossRef] [Green Version]
- Ferrando, A.A.; Williams, B.D.; Stuart, C.A.; Lane, H.W.; Wolfe, R.R. Oral Branched-Chain Amino Acids Decrease Whole-Body Proteolysis. J. Parenter. Enter. Nutr. 1995, 19, 47–54. [Google Scholar] [CrossRef]
- Gonzalez, A.M.; Church, D.D.; Townsend, J.R.; Bagheri, R. Emerging Nutritional Supplements for Strength and Hypertrophy. Strength Cond. J. 2020, 42, 57–70. [Google Scholar] [CrossRef]
- Nair, K.S.; Schwartz, R.G.; Welle, S. Leucine as a regulator of whole body and skeletal muscle protein metabolism in humans. Am. J. Physiol. 1992, 263, E928–E934. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.-Y.; Schutzler, S.; Schrader, A.; Spencer, H.J.; Azhar, G.; Ferrando, A.A.; Wolfe, R.R. The anabolic response to a meal containing different amounts of protein is not limited by the maximal stimulation of protein synthesis in healthy young adults. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E73–E80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Churchward-Venne, T.A.; Pinckaers, P.J.M.; Smeets, J.S.J.; Betz, M.W.; Senden, J.M.; Goessens, J.P.B.; Gijsen, A.P.; Rollo, I.; Verdijk, L.B.; Van Loon, L.J. Dose-response effects of dietary protein on muscle protein synthesis during recovery from endurance exercise in young men: A double-blind randomized trial. Am. J. Clin. Nutr. 2020, 112, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Rémond, D.; Machebeuf, M.; Yven, C.; Buffière, C.; Mioche, L.; Mosoni, L.; Patureau Mirand, P. Postprandial whole-body protein metabolism after a meat meal is influenced by chewing efficiency in elderly subjects. Am. J. Clin. Nutr. 2007, 85, 1286–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, S.; Chinkes, D.; MacLean, D.A.; Gore, D.; Wolfe, R.R. In vivo muscle amino acid transport involves two distinct processes. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E136–E141. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Song, J.; Sun, J.; Zhang, X. Total aminoacyl-transfer RNA pool is greater in liver than muscle in rabbits. J. Nutr. 2007, 137, 2333–2338. [Google Scholar] [CrossRef] [Green Version]
- Wolfe, R.R. The 2017 Sir David P Cuthbertson lecture. Amino acids and muscle protein metabolism in critical care. Clin. Nutr. 2018, 37, 1093–1100. [Google Scholar] [CrossRef]
| Study | Group | N (M/F) | Age (y) | BM (kg) | BMI (kg/m2) | LBM (kg) | BF (%) |
|---|---|---|---|---|---|---|---|
| Park 2020 European Journal of Nutrition [25] | 3.5 oz Ground Beef and 2 green kiwis | 11 (5/5) | 72.5 ± 1.9 | 82.5 ± 2.2 | 28.7 ± 0.8 | 48.5 ± 2.5 | 36.4 ± 2.1 |
| 3.5 oz Ground Beef and 2 gold kiwis | |||||||
| Park 2020 Journal of International Society of Sports Nutrition [23] | 2.4 g Whey + 3.2 g Free Form EAA | 8 (3/5) | 21.4 ± 0.5 | 73.8 ± 4.8 | 24.6 ± 0.8 | 51.6 ± 4.9 | 21.1 ± 2.2 |
| 4.8 g Whey + 6.4 g Free Form EAA | |||||||
| 12.6 g Whey | 8 (4/4) | 26.9 ± 2.0 | 76.2 ± 3.1 | 25.7 ± 1.6 | 49.5 ± 2.6 | 24.8 ± 4.1 | |
| Park 2020 Journal of Nutrition [26] | 2 oz Ground Beef | 8 (4/4) | 21.8 ± 2.2 | 76.3 ± 4.6 | 24.9 ± 1.0 | 49.2 ± 4.1 | 30.1 ± 3.1 |
| 2 oz Beef Sirloin | 8 (4/4) | 23.9 ± 1.6 | 68.0 ± 4.0 | 23.5 ± 1.0 | 43.5 ± 3.3 | 31.0 ± 2.4 | |
| 2 Cooked Eggs | 8 (4/4) | 23.9 ± 1.9 | 74.2 ± 5.4 | 24.4 ± 1.3 | 49.1 ± 3.4 | 27.5 ± 2.5 | |
| 2 oz Pork Loin | 8 (4/4) | 22.1 ± 1.0 | 74.3 ± 3.5 | 24.5 ± 0.9 | 51.8 ± 4.6 | 29.9 ± 3.9 | |
| 1/2C Kidney Beans | 8 (4/4) | 23.8 ± 1.9 | 69.8 ± 5.7 | 24.1 ± 1.7 | 44.8 ± 3.1 | 30.1 ± 2.5 | |
| 2T Peanut Butter | 8 (4/4) | 20.3 ± 1.5 | 70.4 ± 3.8 | 24.1 ± 1.1 | 48.1 ± 3.8 | 27.1 ± 2.3 | |
| 4 oz Tofu | 8 (4/4) | 25.9 ± 2.2 | 75.9 ± 2.2 | 25.9 ± 1.0 | 49.7 ± 4.2 | 33.0 ± 3.0 | |
| 1 oz Mixed Nuts | 8 (4/4) | 24.3 ± 2.1 | 74.3 ± 5.3 | 24.9 ± 1.2 | 49.3 ± 3.9 | 32.2 ± 2.2 | |
| Church 2020 Journal of International Society of Sports Nutrition [24] | 3.6 g Free Form EAA | 11 (5/6) | 68.8 ± 1.8 | 81.4 ± 5.69 | 31.8 ± 5.7 | 49.7 ± 3.6 | 35.7 ± 2.2 |
| 10.8 g Free Form EAA | 12 (8/4) | 67.4 ± 1.5 | 83.4 ± 5.5 | 27.5 ± 1.3 | 53.1 ± 3.9 | 35.4 ± 3.2 | |
| Sample Means | - | 39.9 ± 2.0 | 76.1 ± 1.2 | 26.1 ± 0.6 | 49.5 ± 0.9 | 32.2 ± 0.8 | |
| Best Predictor | R2 | p | Formula |
|---|---|---|---|
| ΔFSR | |||
| EAACMax | 0.341 | ≤0.001 | (0.00001933 × EAACMax) − 0.017 |
| Postprandial FSR | |||
| EAACMax | 0.524 | ≤0.001 | (0.00003307 × EAACMax) + 0.016 |
| ΔWBPS | |||
| ΔEAA | 0.345 | ≤0.001 | (0.001 × ΔEAA) + 0.242 |
| kcals | Protein (g) | EAA (g) | CHO (g) | Fat (g) | EAA:Protein | ||
|---|---|---|---|---|---|---|---|
| Postprandial FSR (%/h) | R | −0.334 * | −0.189 * | 0.230 * | −0.210 * | −0.402 * | 0.644 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 | |
| ΔFSR (%/h) | R | −0.346 * | −0.205 * | 0.227 * | −0.255 * | −0.383 * | 0.585 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 | |
| ΔWBPS (g/h) | R | −0.227 * | 0.033 | 0.386 * | −0.269 * | −0.279 * | 0.409 * |
| N | 112 | 112 | 112 | 112 | 112 | 112 | |
| EAA AUCi (umol/L/min) | R | 0.096 | 0.400 * | 0.822 * | 0.063 | −0.121 | 0.379 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 | |
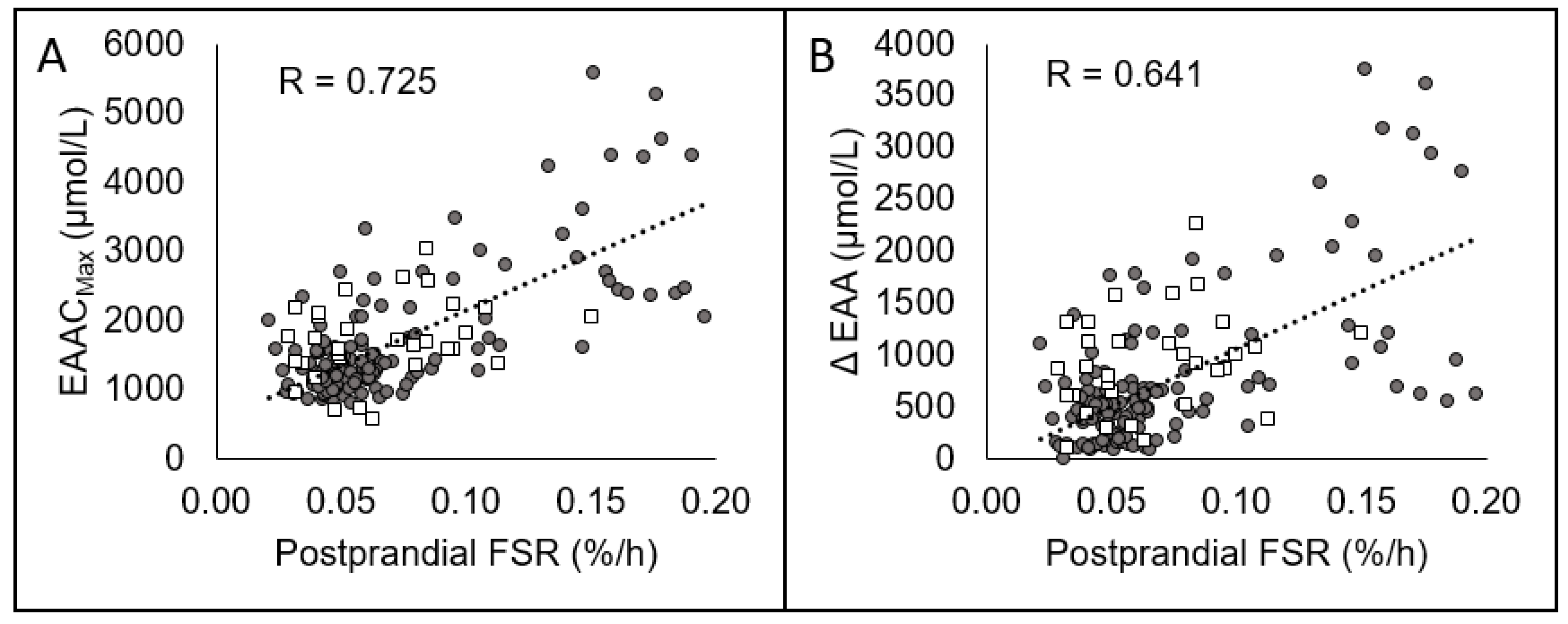
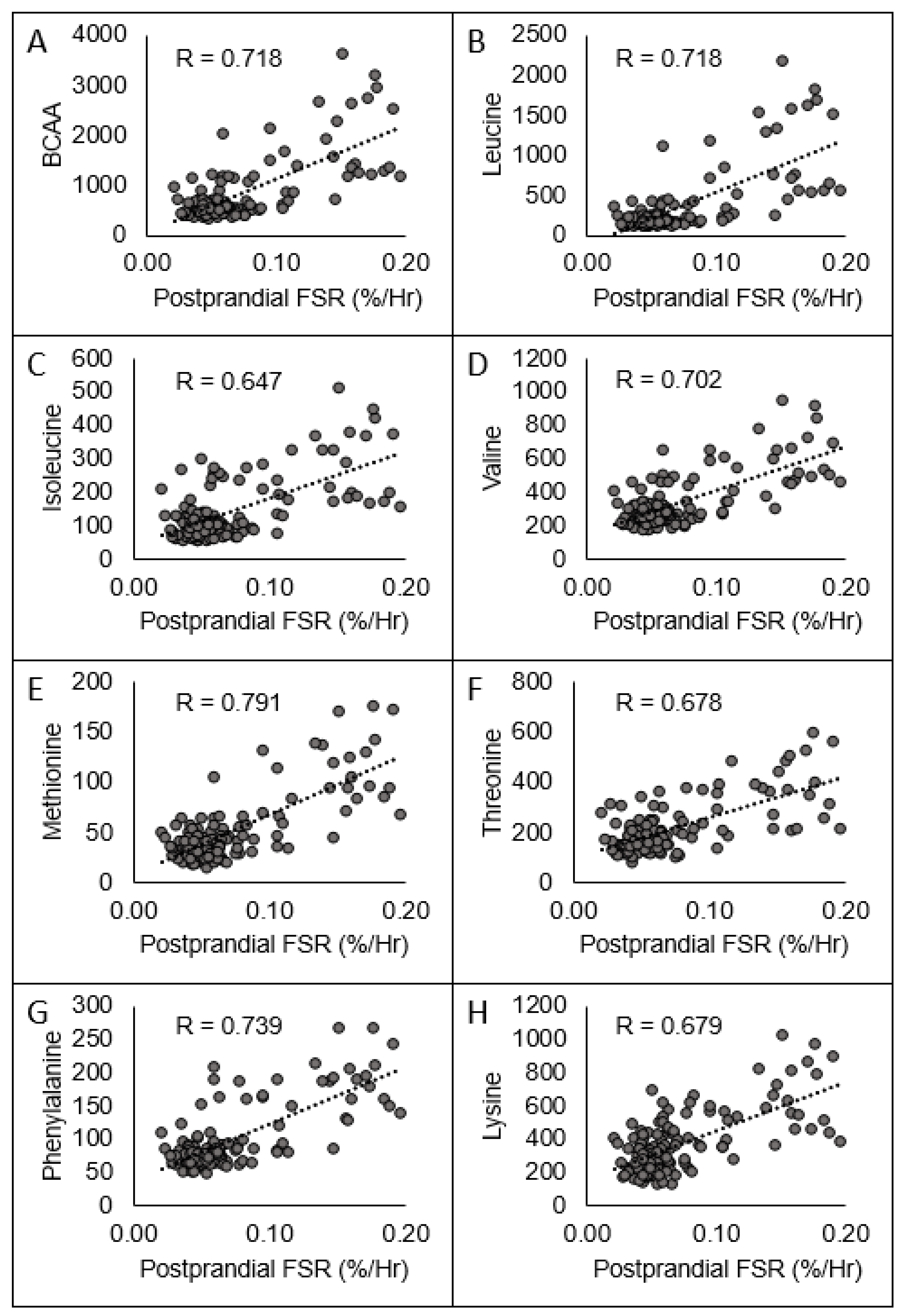
| EAACmax (umol/L) | R | −0.429 * | −0.169 | 0.510 * | −0.342 * | −0.524 * | 0.781 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 | |
| Δ[EAA] (umol/L) | R | −0.383 * | −0.094 | 0.604 * | −0.323 * | −0.495 * | 0.733 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 | |
| Rate to Peak EAACmax (umol/L/min) | R | −0.546 * | −0.303 * | 0.418 * | −0.430 * | −0.611 * | 0.841 * |
| N | 134 | 134 | 134 | 134 | 134 | 134 |
| Postprandial FSR (%/h) | ΔFSR (%/h) | ΔWBPS (g/h) | ||
|---|---|---|---|---|
| EAA AUCi (umol/L/min) | R | 0.475 * | 0.420 * | 0.438 * |
| N | 130 | 130 | 112 | |
| EAACmax (umol/L) | R | 0.724 * | 0.584 * | 0.559 * |
| N | 130 | 130 | 112 | |
| Δ[EAA] (umol/L) | R | 0.641 * | 0.562 * | 0.587 * |
| N | 130 | 130 | 112 | |
| Rate to Peak EAACmax (umol/L/min) | R | 0.626 * | 0.577 * | 0.444 * |
| N | 130 | 130 | 112 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Church, D.D.; Hirsch, K.R.; Park, S.; Kim, I.-Y.; Gwin, J.A.; Pasiakos, S.M.; Wolfe, R.R.; Ferrando, A.A. Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding. Nutrients 2020, 12, 3717. https://doi.org/10.3390/nu12123717
Church DD, Hirsch KR, Park S, Kim I-Y, Gwin JA, Pasiakos SM, Wolfe RR, Ferrando AA. Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding. Nutrients. 2020; 12(12):3717. https://doi.org/10.3390/nu12123717
Chicago/Turabian StyleChurch, David D., Katie R. Hirsch, Sanghee Park, Il-Young Kim, Jess A. Gwin, Stefan M. Pasiakos, Robert R. Wolfe, and Arny A. Ferrando. 2020. "Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding" Nutrients 12, no. 12: 3717. https://doi.org/10.3390/nu12123717
APA StyleChurch, D. D., Hirsch, K. R., Park, S., Kim, I.-Y., Gwin, J. A., Pasiakos, S. M., Wolfe, R. R., & Ferrando, A. A. (2020). Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding. Nutrients, 12(12), 3717. https://doi.org/10.3390/nu12123717







